Current Status of Soybean Anthracnose Associated with Colletotrichum truncatum in Brazil and Argentina
Abstract
:1. Introduction
2. Results
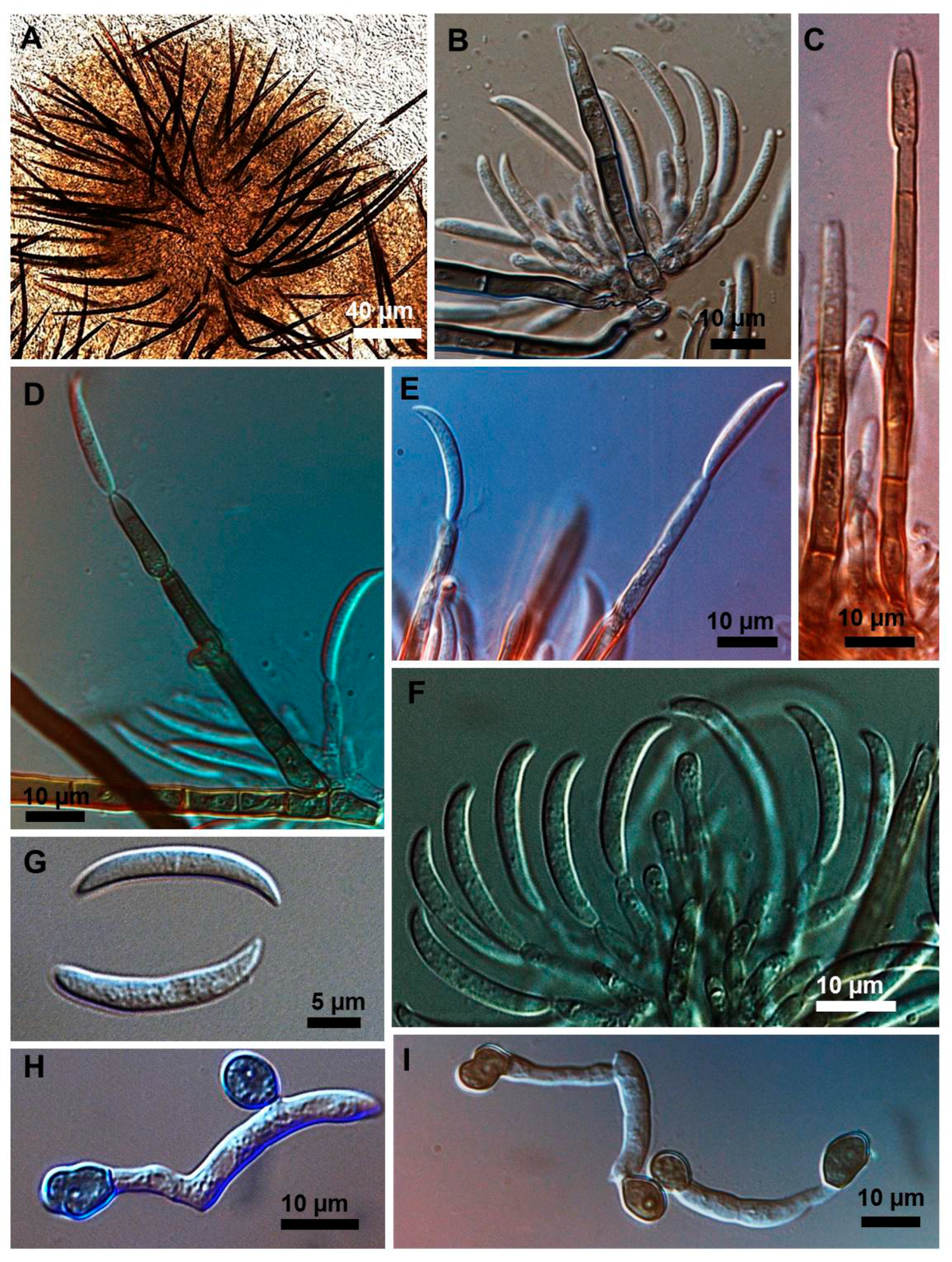
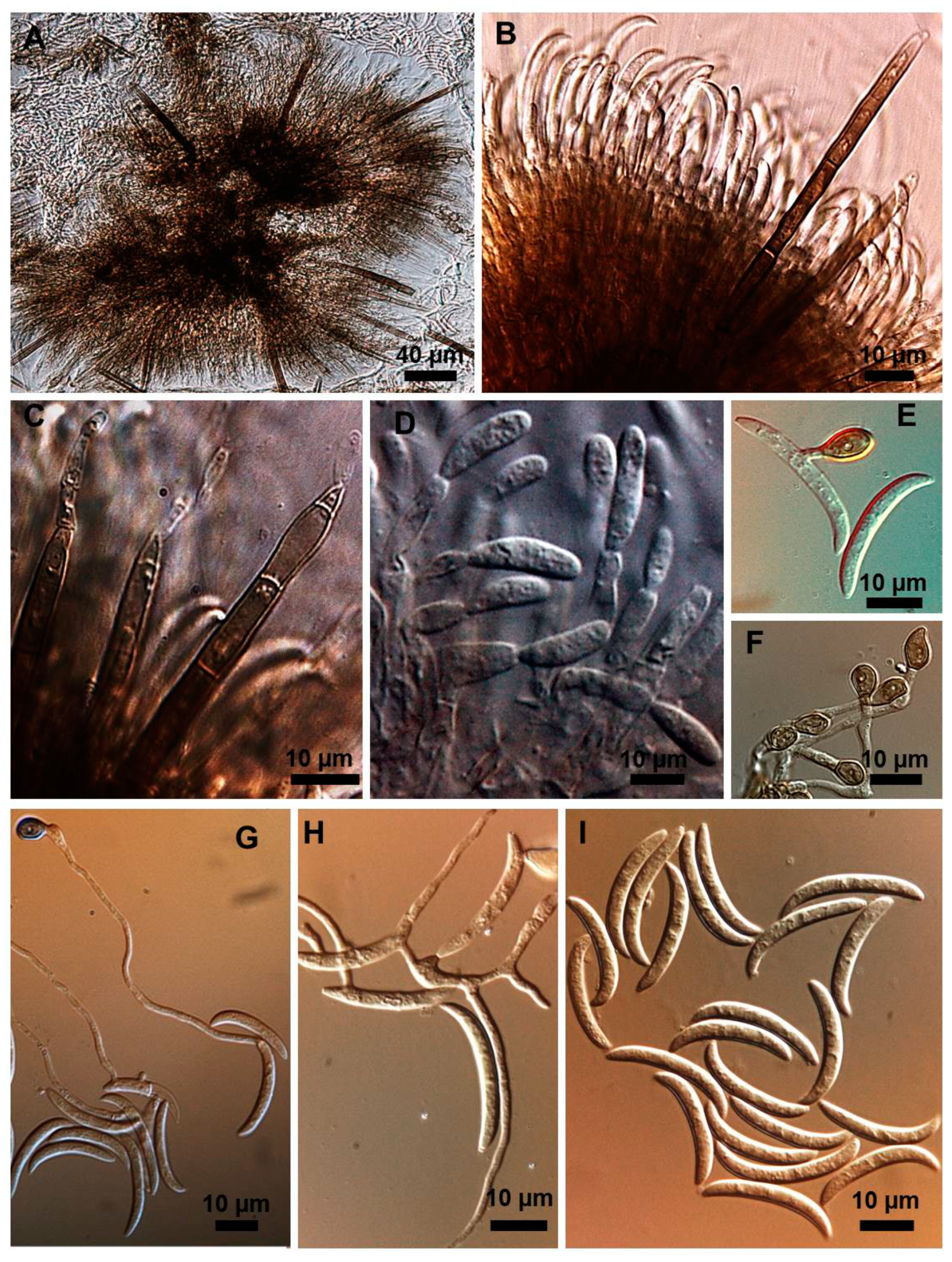
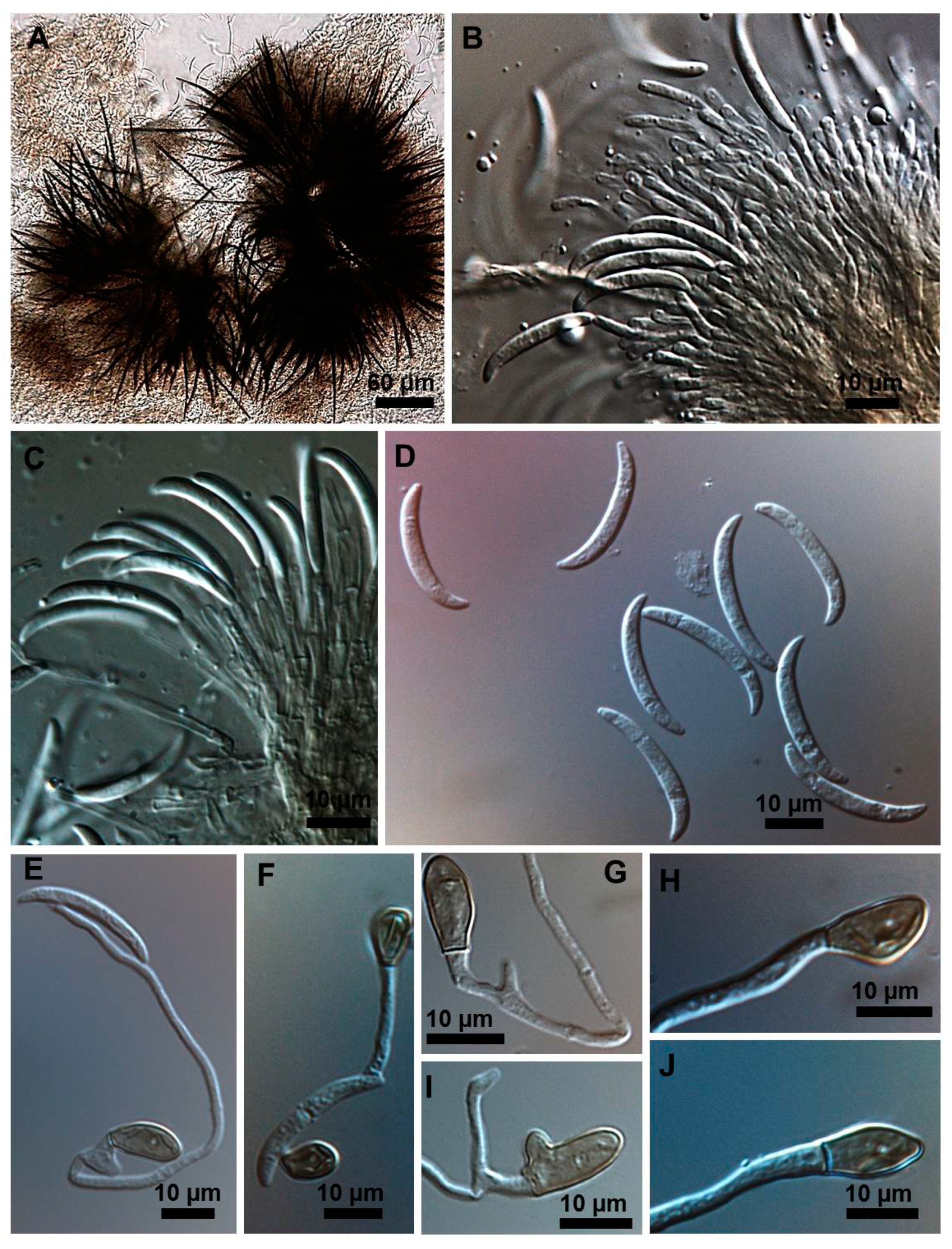
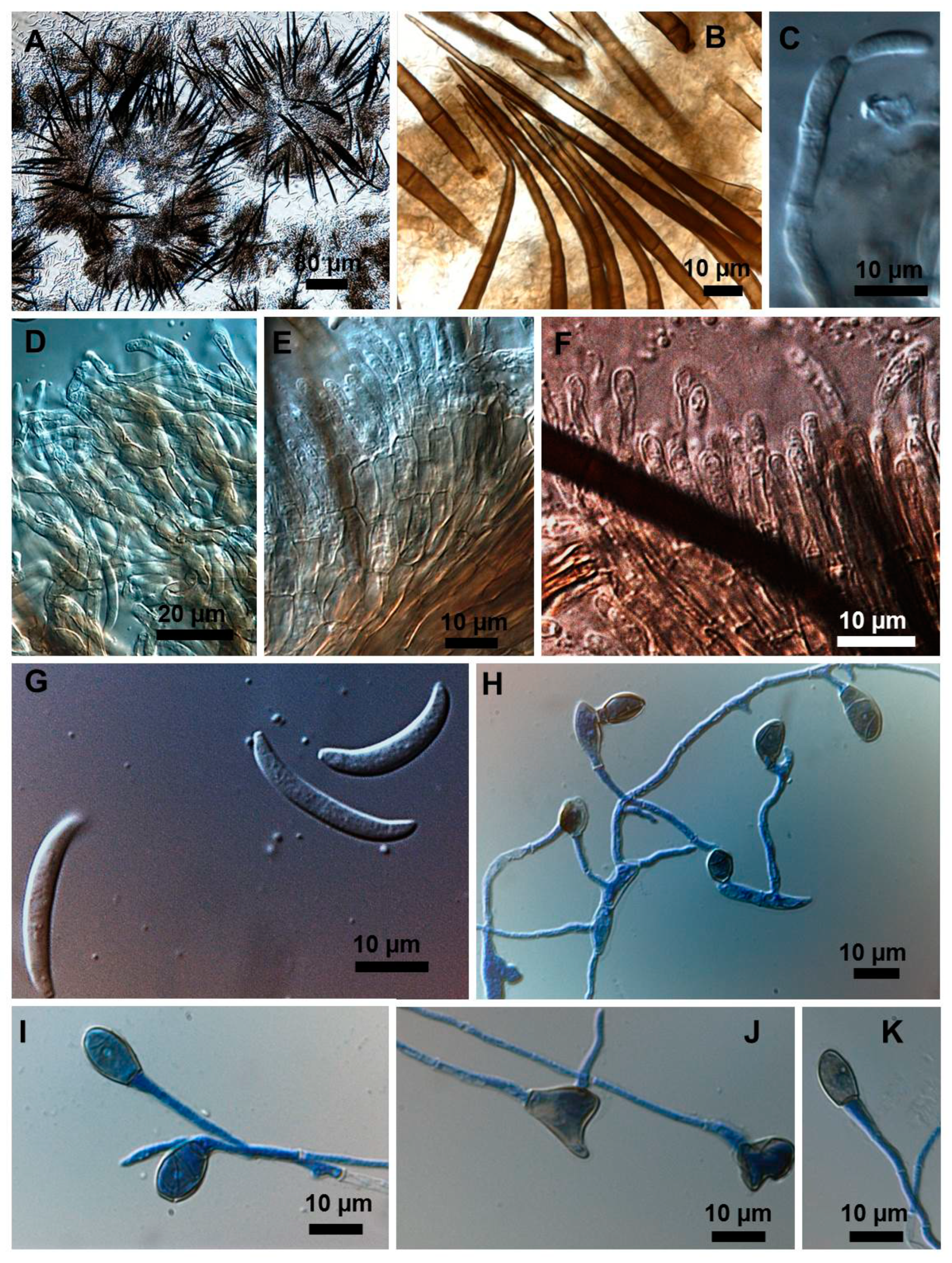
2.1. Morphological and Culture Observations
2.2. Morphometrics of Conidia and Appressoria
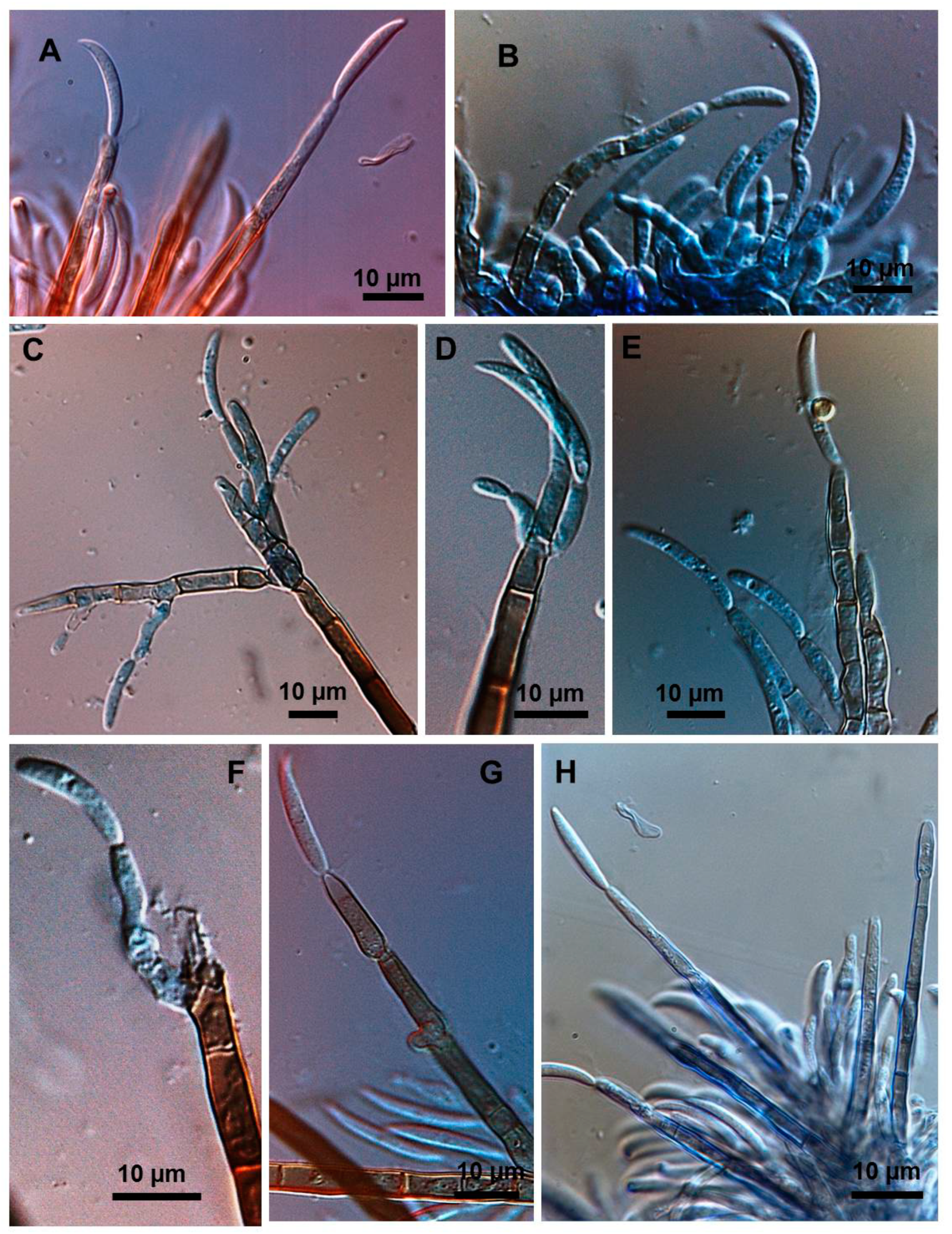
2.3. Types of Conidiogenesis Recorded in South American C. truncatum Isolates
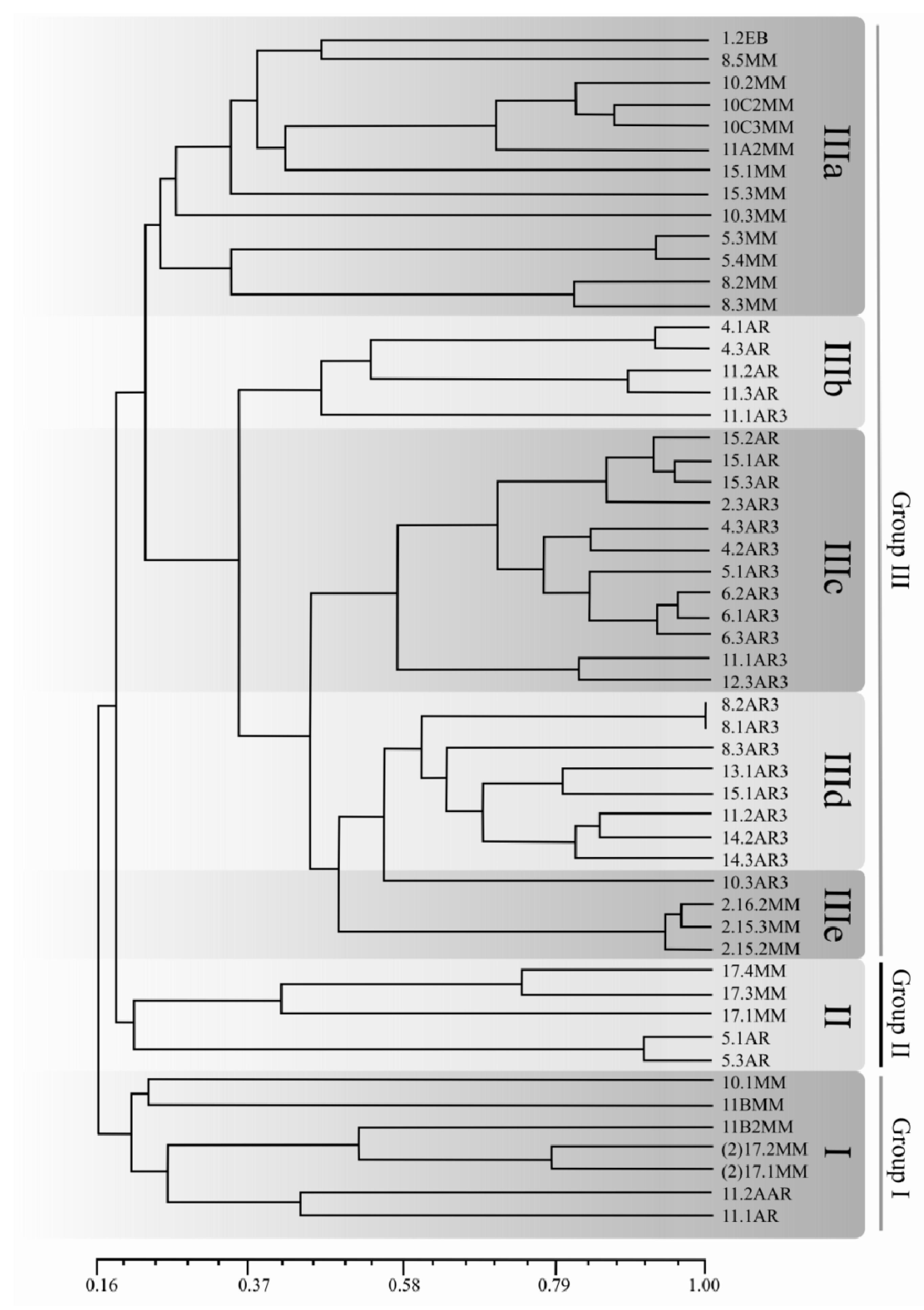
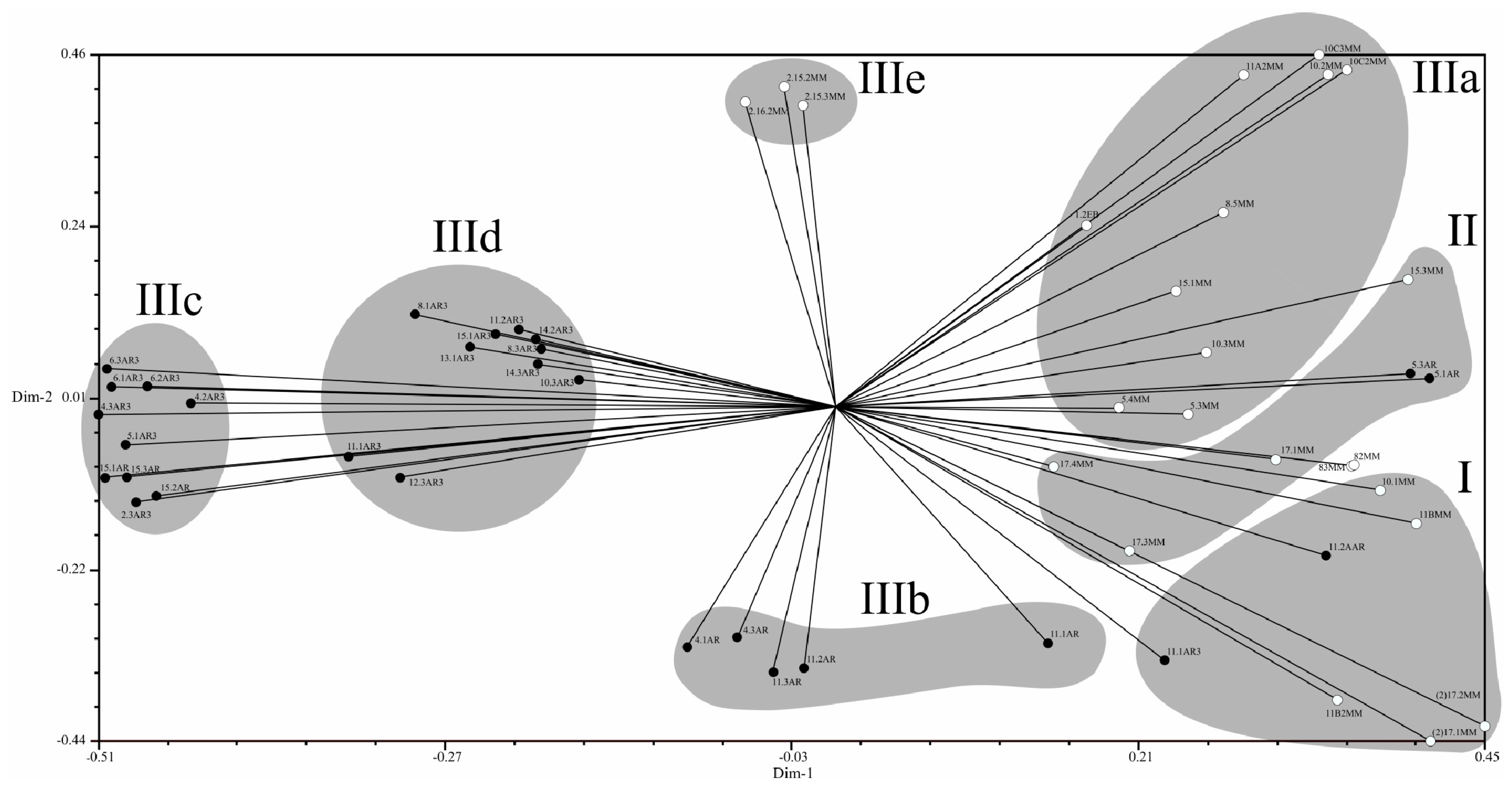
2.4. Population Structure of C. Truncatum in South America
2.5. Reaction of Soybean Commercial Cultivars and Breeding Lines to Anthracnose
3. Discussion
4. Materials and Methods
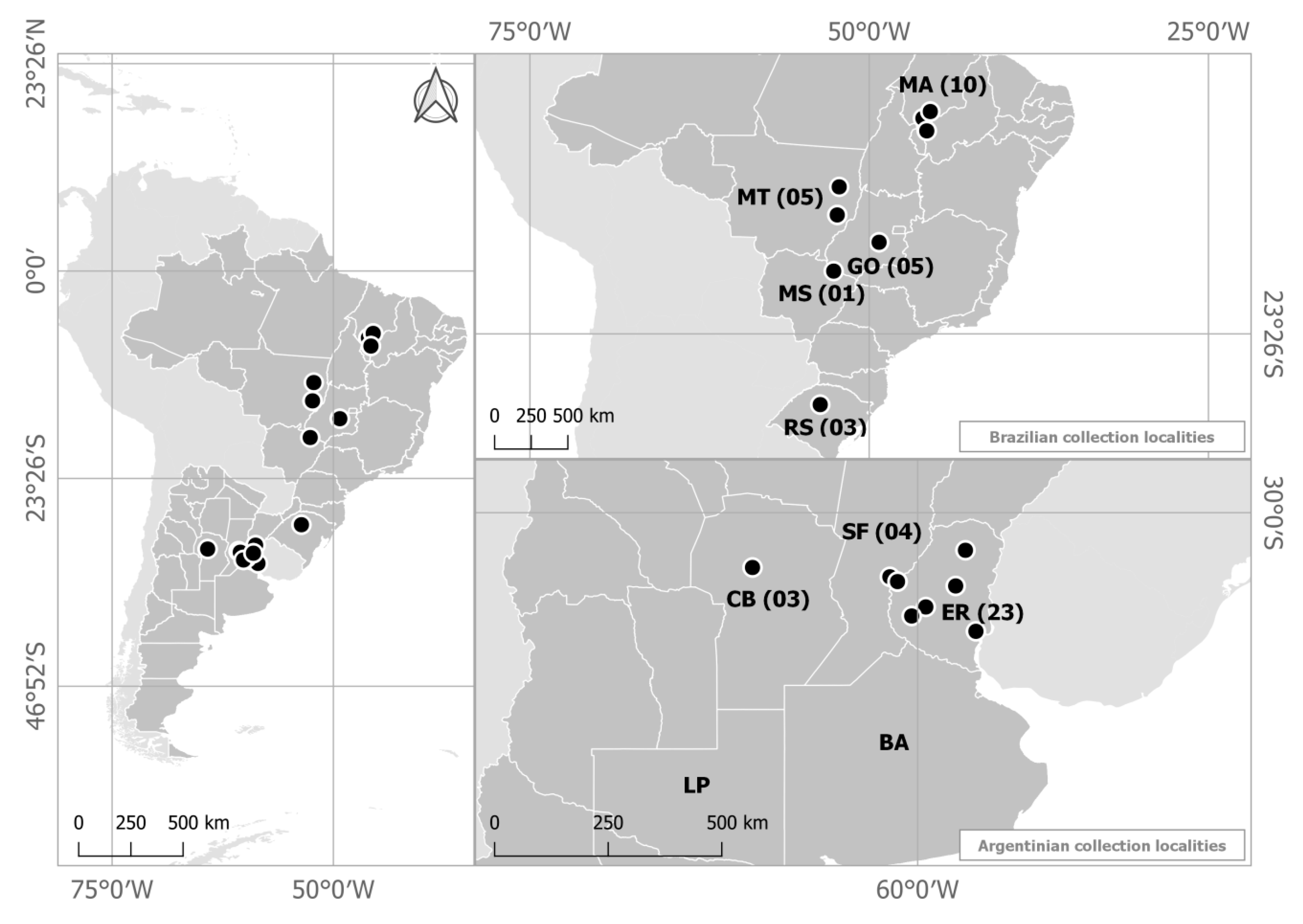
4.1. Origin of Isolates
4.2. Morphological and Cultural Studies
4.3. Production of Mycelium, Extraction, and Quantification of DNA
4.4. Identification of Isolates by Sequencing of the CHS-1 Gene
4.5. Diversity of C. truncatum Populations Based on RAPD Profiles
4.6. Data Analysis
4.7. Reaction of Commercial Cultivars and Breeding Lines of Soybean to Anthracnose
5. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- United States Department of Agriculture. World Agricultural Supply and Demand Estimates 2019; USDA: Washington, DC, USA, 2019. Available online: https://www.usda.gov/oce/commodity/wasde/wasde0919.pdf (accessed on 9 September 2019).
- CONAB. Companhia Nacional de Abastecimento—Boletim Grãos Junho 2019. CONAB: Brasília, Brazil. Available online: https://www.conab.gov.br/info-agro/safras/graos/boletim-da-safra-de-graos (accessed on 9 September 2019).
- MINISTERIO DE AGRICULTURA, ARGENTINA. Estimaciones agrícolas al 05 de Septiembre 2019. Ministerio de Agricultura, Ganaderia y Pesca. Buenos Aires, Argentina. Available online: https://www.agroindustria.gob.ar/sitio/areas/estimaciones/ (accessed on 9 September 2019).
- Hartman, G.L.; Sinclair, J.B.; Rupe, J.C. Compendium of Soybean Diseases, 4th ed.; APS Press: St. Paul, MN, USA, 1999. [Google Scholar]
- Sharma, S.K.; Gupta, G.K.; Ramteke, R. Colletotrichum truncatum [(Schw.) Andrus & W.D. Moore], the causal agent of anthracnose of soybean [Glycine max (L.) Merrill]—A Review. Soybean Res. 2011, 9, 31–52. [Google Scholar]
- Dias, M.D.; Pinheiro, V.F.; Café-Filho, A.C. Impact of anthracnose on the yield of soybean subjected to chemical control in the north region of Brazil. Summa Phytopathol. 2016, 42, 18–23. [Google Scholar] [CrossRef]
- Armstrong-Cho, C.L.; Banniza, S. Glomerella truncata sp. nov., the teleomorph of Colletotrichum truncatum. Mycol. Res. 2006, 110, 951–956. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.D.; Cai, L.; Cannon, P.F.; Crouch, J.A.; Crous, P.W.; Damm, U.; Goodwin, P.H.; Chen, H.; Johnston, P.R.; Jones, E.B.G.; et al. Colletotrichum – Names in Current Use. Fungal Divers. 2009, 39, 147–183. [Google Scholar]
- Yang, H.-C.; Haudenshield, J.S.; Hartman, G.L. First report of Colletotrichum chlorophyti causing soybean anthracnose. Plant Dis. 2012, 96, 1699. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-C.; Haudenshield, J.S.; Hartman, G.L. Colletotrichum incanum sp. nov., a curved-conidial species causing soybean anthracnose in USA. Mycologia 2014, 106, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, M.C.G.; Ciampi-Guillardi, M.; Moraes, S.R.G.; Bonaldo, S.M.; Rogério, F.; Linhares, R.R.; Massola, N.S. First report of Colletotrichum cliviae causing anthracnose on soybean in Brazil. Plant Dis. 2017, 101, 1677. [Google Scholar] [CrossRef]
- Dias, M.D.; Fonseca, M.E.N.; Dias-Neto, J.J.; Santos, M.D.M.; Pandolfo, G.M.; Boiteux, L.S.; Café-Filho, A.C. Biology, pathogenicity, and haplotype analyses of Colletotrichum cliviae: A novel soybean anthracnose agent in warm tropical areas. Trop. Plant Pathol. 2018, 4, 439–451. [Google Scholar] [CrossRef]
- Damm, U.; Sato, T.; Alizadeh, A.; Groenewald, J.Z.; Crous, P.W. The Colletotrichum dracaenophilum, C. magnum and C. orchidearum species complexes. Stud. Mycol. 2019, 92, 1–46. [Google Scholar] [CrossRef] [PubMed]
- Araújo, A.G.; Café-Filho, A.C.; Cupertino, F.P. Antracnose da soja na região geoeconômica do Distrito Federal. Fitopatol. Bras. 1988, 13, 130. [Google Scholar]
- Rogério, F.; Gladieux, P.; Massola, N.S.; Ciampi-Guillardi, M. Multiple introductions without admixture of Colletotrichum truncatum associated with soybean anthracnose in Brazil. Phytopathology 2019, 109, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Rogério, F.; Ciampi-Guillardi, M.; Barbieri, M.C.G.; Bragança, C.A.D.; Seixas, C.D.S.; Almeida, A.M.R.; Massola, N.S. Phylogeny and variability of Colletotrichum truncatum associated with soybean anthracnose in Brazil. J. Appl. Microbiol. 2016, 122, 402–415. [Google Scholar] [CrossRef] [PubMed]
- Sutton, B.C. The Coelomycetes: Fungi Imperfecti with Pycnidia, Acervuli, and Stromata; Commonwealth Mycological Institute - Kew: Surrey, UK, 1980. [Google Scholar]
- Damm, U.; Woudenberg, J.H.C.; Cannon, P.F.; Crous, P.W. Colletotrichum species with curved conidia from herbaceous hosts. Fungal Divers. 2009, 39, 45–87. [Google Scholar]
- Khan, M. Pathogenicity of sclerotia- and nonsclerotia-forming isolates of Colletotrichum truncatum on soybean plants and roots. Phytopathology 1992, 82, 314. [Google Scholar] [CrossRef]
- Morrall, R.A.A. Evolution of lentil diseases over 25 years in western Canada. Can. J. Plant Pathol. 1997, 19, 197–207. [Google Scholar] [CrossRef]
- Lenné, J.M.; Thomas, D.; Andrade, R.P. Anthacnoses of Stylosanthes capitata: Implications for future disease evaluations of indigenous tropical pasture legumes. Phytopathology 1984, 74, 1070–1073. [Google Scholar] [CrossRef]
- Menezes, M.; Hanlin, R.T. Appressoria of Brazilian isolates of Colletotrichum gloeosporioides (Penz.) Sacc. causal agent of anthracnoses diseases. Rev. Microbiol. 1996, 27, 247–251. [Google Scholar]
- Castellani, A. Viability of some pathogenic fungi in distilled water. Am. J. Trop. Med. Hyg. 1939, 24, 270–276. [Google Scholar]
- Cai, L.; Hyde, K.D.; Taylor, P.W.J.; Weir, B.S.; Waller, J.; Abang, M.M.; Zhang, J.Z.; Yang, Y.L.; Phoulivong, S.; Liu, Z.Y.; et al. A polyphasic approach for studying Colletotrichum. Fungal Divers. 2009, 39, 183–204. [Google Scholar]
- Boiteux, L.S.; Fonseca, M.E.N.; Simon, P.W. Effects of Plant Tissue and DNA purification method on Randomly Amplified Polymorphic DNA-based genetic fingerprinting analysis in carrot. J. Am. Soc. Hortic. Sci. 1999, 124. [Google Scholar] [CrossRef]
- Ferreira, M.E.; Grattapaglia, D. Introducao ao Uso de Marcadores Moleculares em Analise Genetica, 3rd ed.; Embrapa-Cenargen, Doc. No. 20; Embrapa: Brasília, Brazil, 1998. [Google Scholar]
- Rohlf, F.J.; Applied Biostatistics, Inc.; Exeter Software (Firm). NTSYS-Pc: Numerical Taxonomy and Multivariate Analysis System; Applied Biostatistics, Inc.: Setauket, NY, USA, 1997. [Google Scholar]
- Vaillancourt, L.; Wang, J.; Hanau, R. Genetic regulation of sexual compatibility in Glomerella graminicola. In Colletotrichum: Host specificity, Pathology, and Host-pathogen Interaction; Prusky, D., Freeman, S., Dickman, M.B., Eds.; APS Press: St. Paul, MN, USA, 2000; pp. 29–44. [Google Scholar]

| No. | Isolate Code UB-UnB 1 | Geographic Origin 2 | Colony Color | Growth Rate 3 |
|---|---|---|---|---|
| 1 | 1.2EB | Chapadão do Sul/MT | Light Grey | Slow |
| 2 | 8.2MM | Tasso Fragoso/MA | Roseate beige | Slow |
| 3 | 8.3MM | Tasso Fragoso/MA | Roseate beige | Slow |
| 4 | 8.5MM | Tasso Fragoso/MA | Light grey | Fast |
| 5 | 11.A2MM | Cruz Alta/RS | Grey | Slow |
| 6 | 15.1MM | Nova Xavantina/ MT | Light Grey | Fast |
| 7 | 15.3MM | Nova Xavantina/ MT | Light Grey | Fast |
| 8 | 10C2MM | S. Rosa das Mangabeiras/MA | Light Grey | Slow |
| 9 | 10C3MM | S. Rosa das Mangabeiras/MA | Light Grey | Slow |
| 10 | 10.2MM | S. Rosa das Mangabeiras/MA | Roseate | Intermediate |
| 11 | 10.3MM | S. Rosa das Mangabeiras/MA | Roseate | Intermediate |
| 12 | 5.3MM | Balsas/MA | Light grey | Fast |
| 13 | 5.4MM | Balsas/MA | Light grey | Fast |
| 14 | 4.1AR | Paraná/Entre Ríos/AR | Grey | Intermediate |
| 15 | 4.3AR | Paraná/Entre Ríos/AR | Grey | Intermediate |
| 16 | 11.2AR | Paraná/Entre Ríos/AR | Light grey | Intermediate |
| 17 | 11.3AR | Paraná/Entre Ríos/AR | Light grey | Intermediate |
| 18 | 11.1AR | Paraná/Entre Ríos/AR | Light grey | Intermediate |
| 19 | 15.1AR | La Capital/Santa Fe/AR | Roseate | Slow |
| 20 | 15.2AR | La Capital/Santa Fe/AR | Roseate | Intermediate |
| 21 | 15.3AR | La Capital/Santa Fe/AR | Roseate | Slow |
| 22 | 2.3AR3 | Nogoyá/Entre Ríos/AR | Olivaceous | Intermediate |
| 23 | 4.3AR3 | Gualeguay/Entre Ríos/AR | Yellowish | Slow |
| 24 | 4.2AR3 | Entre Ríos/AR | Yellowish | Intermediate |
| 25 | 5.1AR3 | Entre Ríos/AR | Gray | Intermediate |
| 26 | 6.2AR3 | Victoria/Entre Ríos/AR | Yellowish | Intermediate |
| 27 | 6.1AR3 | Victoria/Entre Ríos/AR | Yellowish | Intermediate |
| 28 | 6.3AR3 | Victoria/Entre Ríos/AR | Yellowish | Intermediate |
| 29 | 11.1AR3 | Villaguay/Entre Ríos/AR | Roseate beige | Intermediate |
| 30 | 12.3AR3 | Gualeguay/Entre Ríos/AR | Yellowish | Intermediate |
| 31 | 8.2AR3 | Federal/Entre Ríos/AR | Yellowish | Intermediate |
| 32 | 8.1AR3 | Federal/Entre Ríos/AR | Yellowish | Intermediate |
| 33 | 8.3AR3 | Federal/Entre Ríos/AR | Yellowish | Intermediate |
| 34 | 13.1AR3 | Castellanos/Santa Fe/AR | Roseate grey | Intermediate |
| 35 | 15.AR3 | La Capital/Santa Fe/AR | Grey | Slow |
| 36 | 11.2AR3 | Villaguay/Entre Ríos/AR | Yellowish | Intermediate |
| 37 | 14.2AR3 | Castellanos/Córdoba/AR | Olivaceous | Intermediate |
| 38 | 14.3AR3 | Castellanos/Córdoba/AR | Olivaceous | Intermediate |
| 39 | 10.3AR3 | Villaguay/Entre Rios/AR | Roseate grey | Fast |
| 40 | 2.16.2MM | Querência/MT | Light grey | Slow |
| 41 | 2.15.3MM | Nova Xavantina/MT | Light grey | Slow |
| 42 | 2.15.2MM | Nova Xavantina/MT | Light grey | Slow |
| 43 | 17.1MM | Goiânia/GO | Light grey | Slow |
| 44 | 17.3MM | Goiânia/GO | Light grey | Intermediate |
| 45 | 17.4MM | Goiânia/GO | Light grey | Slow |
| 46 | 5.1AR | Paraná/Entre Ríos/AR | Light grey | Slow |
| 47 | 5.2AR | Paraná/Entre Ríos/AR | Light grey | Slow |
| 48 | 11BMM | Cruz Alta/RS | Orange | Slow |
| 49 | 11B2MM | Cruz Alta/RS | Orange | Slow |
| 50 | 10.1MM | S. Rosa das Mangabeiras/MA | Roseate | Intermediate |
| 51 | 17.2MM | Goiânia/GO | Roseate orange | Intermediate |
| 52 | (2)17.1MM | Goiânia/GO | Roseate orange | Intermediate |
| 53 | 11.2AAR | Paraná/Entre Ríos/AR | Grey | Intermediate |
| 54 | 11.1AAR | Paraná/Entre Ríos/AR | Grey | Intermediate |
| Conidia (µm) | Appressoria (µm) | |||||||
|---|---|---|---|---|---|---|---|---|
| Length | Width | Length | Width | |||||
| Isolate | Average | Range | Average | Range | Average | Range | Average | Range |
| 11A2MM | 23.5 | 20.0–27.0 | 4.0 | 3.0–5.0 | 13.0 | 9.0–19.0 | 8.5 | 6.5–11.0 |
| 11.7MM | 26.0 | 24.0–28.0 | 3.5 | 3.0–4.0 | 13.5 | 9.0–19.0 | 7.0 | 5.0–9.0 |
| 5.1MM | 24.5 | 22.0–27.0 | 3.5 | 3.0–4.0 | 10.0 | 6.5–15.0 | 7.5 | 6.0–9.0 |
| 6.1MM | 20.0 | 15.0–25.0 | 3.5 | 2.0–5.0 | 13.5 | 11.5–18.0 | 7.0 | 5.5–8.5 |
| 10C3MM | 24.5 | 21.0–28.0 | 3.0 | 2.0–4.0 | 9.0 | 7.0–12.0 | 6.0 | 4.5–7.0 |
| Primer | Sequence | #Amplicons | Amplicon Size Range (bp) |
|---|---|---|---|
| OPA-3 | AGTCAGCCAC | 09 | 2500–200 |
| OPB-15 | GGAGGGTGTT | 08 | 1900–200 |
| OPD-3 | GTCGCCGTCA | 12 | 2000–300 |
| OPD-8 | GTGTGCCCCA | 06 | 2000–500 |
| OPE-17 | CTACTGCCGT | 06 | 1400–200 |
| OPE-8 | TCACCACGGT | 09 | 2100–200 |
| OPG-11 | TGCCCGTCGT | 09 | 2500–200 |
| OPG-17 | ACGACCGACA | 12 | 2500–300 |
| OPG-4 | AGCGTGTCTG | 10 | 1400–150 |
| OPK-17 | CCCAGCTGTG | 09 | 2500–300 |
| OPP-2 | TCGGCACGCA | 06 | 1750–200 |
| OPP-9 | GTGGTCCGCA | 08 | 2300–700 |
| Accession | Incidence on Cotyledons 1 | Incidence on Stems 1 | ||
|---|---|---|---|---|
| Assay 1 | Assay 2 | Assay 1 | Assay 2 | |
| BRS Pintado | 5.3 a | 9.3 a | 6.2 a | 4.4 n.s. |
| W877 | 6.2 a | 8.4 a | 9.8 a | 2.6 |
| W811 | 6.2 a | 8.9 a | 14.7 b | 1.7 |
| W791 | 8.0 a | 16.0 a | 13.3 b | 2.6 |
| W842 | 15.1 b | 11.6 a | 5.3 a | 2.2 |
| W828 | 15.6 b | 12.0 a | 17.0 b | 7.5 |
| TMG 115 | 5.3 a | 21.8 b | 12.9 b | 5.3 |
| P98Y11 | 13.3 b | 24.1 b | 13.3 b | 5.8 |
| W801 | 16.5 b | 25.4 b | 23.2 c | 8.4 |
| TMG127 | 15.2 b | 38.8 c | 14.2 b | 5.8 |
| W875 | 25.4 c | 14.7 a | 9.8 a | 5.3 |
| W810 | 19.1 c | 19.1 b | 14.7 b | 4.0 |
| W731 | 28.5 d | 39.6 c | 8.4 a | 0.8 |
| W787 | 16.9 b | 49.1 d | 8.0 a | 8.4 |
| W712 | 21.4 c | 58.0 e | 8.9 a | 2.6 |
| W891 | 32.8 d | 45.0 c | 25.8 c | 4.4 |
| Assay Average | 15.2 | 24.5 | 12.8 | 5.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dias, M.D.; Dias-Neto, J.J.; Santos, M.D.M.; Formento, A.N.; Bizerra, L.V.A.S.; Fonseca, M.E.N.; Boiteux, L.S.; Café-Filho, A.C. Current Status of Soybean Anthracnose Associated with Colletotrichum truncatum in Brazil and Argentina. Plants 2019, 8, 459. https://doi.org/10.3390/plants8110459
Dias MD, Dias-Neto JJ, Santos MDM, Formento AN, Bizerra LVAS, Fonseca MEN, Boiteux LS, Café-Filho AC. Current Status of Soybean Anthracnose Associated with Colletotrichum truncatum in Brazil and Argentina. Plants. 2019; 8(11):459. https://doi.org/10.3390/plants8110459
Chicago/Turabian StyleDias, Moab D., Justino J. Dias-Neto, Maria D.M. Santos, Angela Norma Formento, Lincoln V.A.S. Bizerra, Maria Esther N. Fonseca, Leonardo S. Boiteux, and Adalberto C. Café-Filho. 2019. "Current Status of Soybean Anthracnose Associated with Colletotrichum truncatum in Brazil and Argentina" Plants 8, no. 11: 459. https://doi.org/10.3390/plants8110459
APA StyleDias, M. D., Dias-Neto, J. J., Santos, M. D. M., Formento, A. N., Bizerra, L. V. A. S., Fonseca, M. E. N., Boiteux, L. S., & Café-Filho, A. C. (2019). Current Status of Soybean Anthracnose Associated with Colletotrichum truncatum in Brazil and Argentina. Plants, 8(11), 459. https://doi.org/10.3390/plants8110459





