Heterophylly: Phenotypic Plasticity of Leaf Shape in Aquatic and Amphibious Plants
Abstract
1. What is Heterophylly?
2. History of Research on Heterophylly
3. Environmental Factors that Induce Heterophylly
3.1. CO2
3.2. Light Intensity
3.3. Light Quality
3.4. Light Photoperiod
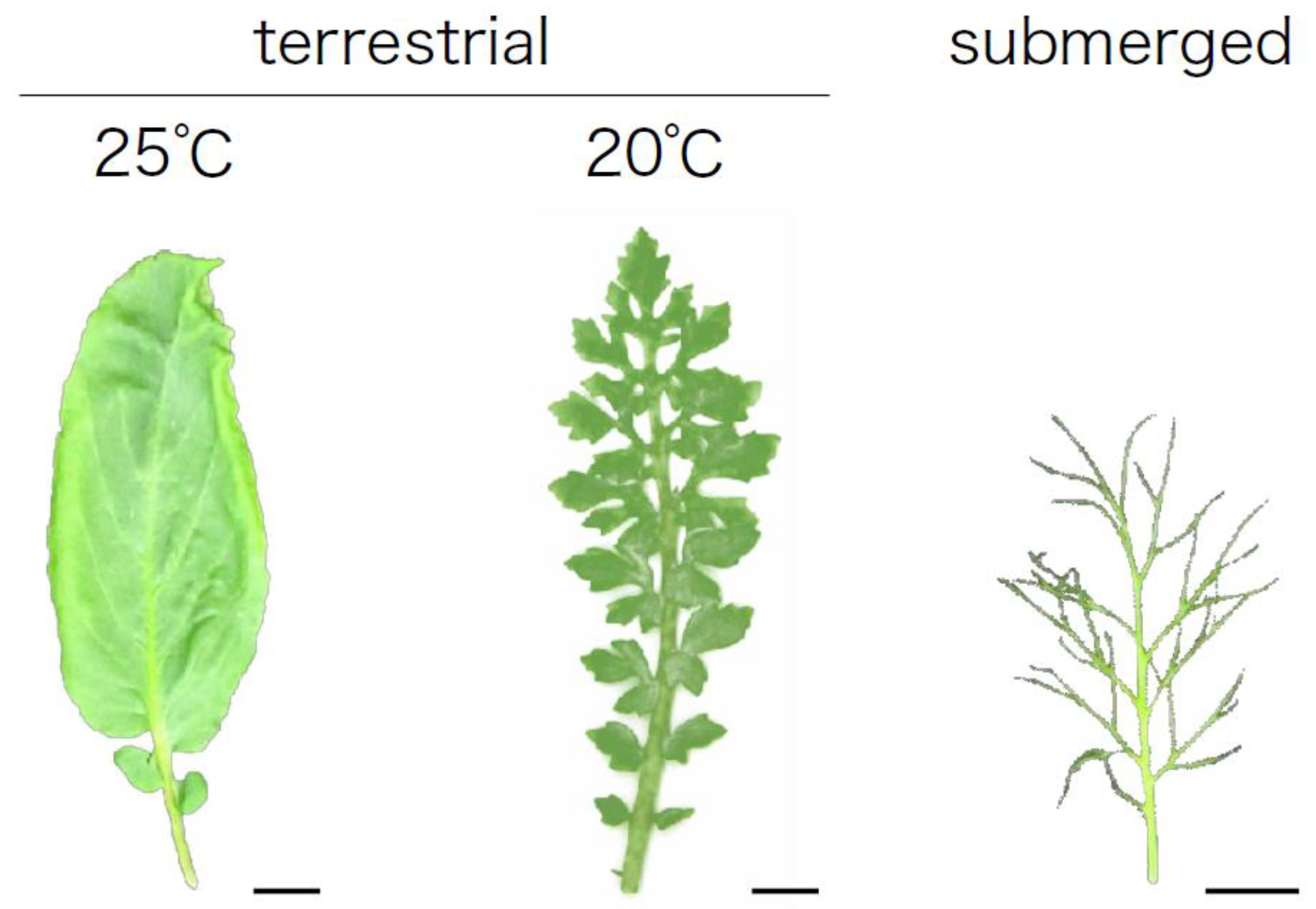
3.5. Temperature
3.6. Osmotic Stress
3.7. Water Depth
3.8. Water Flow
4. Molecular Basis for Heterophylly
4.1. North American Lake Cress
4.2. Threadleaf Crowfoot
4.3. Potamogeton octandrus
4.4. Water-Wisteria, Hygrophila Difformis (Acanthaceae)
5. Future Perspectives for Studies on Heterophylly
5.1. Sensing Mechanisms for Submergence
5.2. Epigenetic Regulation of Heterophylly
5.3. Adaptive Significance of Heterophylly
5.4. Evolution of Heterophylly
Author Contributions
Funding
Conflicts of Interest
References
- Zotz, G.; Wilhelm, K.; Becker, A. Heteroblasty—A review. Bot. Rev. 2011, 77, 109–151. [Google Scholar] [CrossRef]
- Wells, C.L.; Pigliucci, M. Adaptive phenotypic plasticity: The case of heterophylly in aquatic plants. Perspect. Plant Ecol. Evol. Syst. 2000, 3, 1–18. [Google Scholar] [CrossRef]
- Wanke, D. The ABA-mediated switch between submersed and emersed life-styles in aquatic macrophytes. J. Plant Res. 2011, 124, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.W. Abscisic Acid Induces Formation of Floating Leaves in the Heterophyllous Aquatic Angiosperm Potamogeton nodosus. Science 1978, 201, 1135–1138. [Google Scholar] [CrossRef]
- Goliber, T.E.; Feldman, L.J. Developmental analysis of leaf plasticity in the heterophyllous aquatic plant Hippuris vulgaris. Am. J. Bot. 1990, 77, 399–412. [Google Scholar] [CrossRef]
- Kuwabara, A.; Nagata, T. Cellular basis of developmental plasticity observed in heterophyllous leaf formation of Ludwigia arcuata (Onagraceae). Planta 2006, 224, 761–770. [Google Scholar] [CrossRef]
- Dengler, N.G. Anisophylly and dorsiventral shoot symmetry. Int. J. Plant Sci. 1999, 160, S67–S80. [Google Scholar] [CrossRef]
- Davis, G.J. Proserpinaca: Photoperiodic and chemical differentiation of leaf development and flowering. Plant Physiol. 1967, 42, 667–668. [Google Scholar] [CrossRef]
- Kane, M.E.; Albert, L.S. Integrative regulation of leaf morphogenesis by gibberellic and abscisic acids in the aquatic angiosperm Proserpinaca palustris L. Aquat. Bot. 1987, 28, 89–96. [Google Scholar] [CrossRef]
- Kane, M.E.; Albert, L.S. Abscisic acid induction of aerial leaf development in Myriophyllum and Proserpinaca species cultured in vitro. J. Aquat. Plant Manag. 1989, 27, 102–111. [Google Scholar]
- Deschamp, P.A.; Cooke, T.J. Causal mechanisms of leaf dimorphism in the aquatic angiosperm Callitriche heterophylla. Am. J. Bot. 1984, 71, 319–329. [Google Scholar] [CrossRef]
- Lin, B.; Yang, W. Blue light and abscisic acid independently induce heterophyllous switch in Marsilea quadrifolia. Plant Physiol. 1999, 119, 429–434. [Google Scholar] [CrossRef]
- Hsu, T.; Liu, H.; Wang, J.; Chen, R.; Wang, Y.; Lin, B. Early genes responsive to abscisic acid during heterophyllous induction in Marsilea quadrifolia. Plant Mol. Biol. 2001, 47, 703–715. [Google Scholar] [CrossRef]
- Kuwabara, A.; Tsukaya, H.; Nagata, T. Identification of factors that cause heterophylly in Ludwigia arcuata Walt. (Onagraceae). Plant Biol. 2001, 3, 98–105. [Google Scholar] [CrossRef]
- Nakayama, H.; Nakayama, N.; Nakamasu, A.; Sinha, N.; Kimura, S. Toward elucidating the mechanisms that regulate heterophylly. Plant Morphol. 2012, 24, 57–63. [Google Scholar] [CrossRef][Green Version]
- Klimenko, E.N. Structural and functional aspects of the Nuphar lutea (L.) Smith heterophylly: Ultrastructure and photosynthesis. Cytol. Genet. 2012, 46, 272–279. [Google Scholar] [CrossRef]
- Kane, M.E.; Albert, L.S. Abscisic acid induces aerial leaf morphology and vasculature in submerged Hippuris vulgaris L. Aquat. Bot. 1987, 28, 81–88. [Google Scholar] [CrossRef]
- Tsukaya, H. Comparative leaf development in angiosperms. Curr. Opin. Plant Biol. 2014, 17, 103–109. [Google Scholar] [CrossRef]
- Arber, A. On Heterophylly in Water Plants. Am. Nat. 1919, 53, 272–278. [Google Scholar] [CrossRef]
- McComb, A.J. The control of elongation in Callitriche shoots by environment and gibberellic acid. Ann. Bot. 1965, 29, 445–458. [Google Scholar] [CrossRef]
- Deschamp, P.A.; Cooke, T.J. Leaf dimorphism in aquatic angiosperms: Significance of turgor pressure and cell expansion. Science 1983, 219, 505–507. [Google Scholar] [CrossRef]
- Jones, H. Variation in Leaf Form in Callitriche intermedia. Nature 1952, 170, 848–849. [Google Scholar] [CrossRef]
- Jones, H. Heterophylly in some Species of Callitriche, with especial reference to Callitriche intermedia. Ann. Bot. 1955, 19, 226–245. [Google Scholar] [CrossRef]
- Bodkin, B.C.; Spence, D. Photoreversible control of heterophylly in Hippuris vulgaris L. New Phytol. 1980, 84, 533–542. [Google Scholar] [CrossRef]
- McCully, M.E.; Dale, H.M. Heterophylly in Hippuris, a problem in identification. Botany 1961, 39, 1099–1116. [Google Scholar] [CrossRef]
- Li, G.; Hu, S.; Yang, J.; Schultz, E.A.; Clarke, K.; Hou, H. Water-Wisteria as an ideal plant to study heterophylly in higher aquatic plants. Plant Cell Rep. 2017, 36, 1225–1236. [Google Scholar] [CrossRef]
- Horiguchi, G.; Nemoto, K.; Yokoyama, T.; Hirotsu, N. Photosynthetic acclimation of terrestrial and submerged leaves in the amphibious plant Hygrophila difformis. AoB Plants 2019, 11, plz009. [Google Scholar] [CrossRef]
- Herrera, C.M.; Bazaga, P. Epigenetic correlates of plant phenotypic plasticity: DNA methylation differs between prickly and nonprickly leaves in heterophyllous Ilex aquifolium (Aquifoliaceae) trees. Bot. J. Linn. Soc. 2013, 171, 441–452. [Google Scholar] [CrossRef]
- Kuwabara, A.; Ikegami, K.; Koshiba, T.; Nagata, T. Effects of ethylene and abscisic acid upon heterophylly in Ludwigia arcuata (Onagraceae). Planta 2003, 217, 880–887. [Google Scholar] [CrossRef]
- Sato, M.; Tsutsumi, M.; Ohtsubo, A.; Nishii, K.; Kuwabara, A.; Nagata, T. Temperature-dependent changes of cell shape during heterophyllous leaf formation in Ludwigia arcuata (Onagraceae). Planta 2008, 228, 27–36. [Google Scholar] [CrossRef]
- Bristow, J.M.; Looi, A. Effects of Carbon Dioxide on the Growth and Morphogenesis of Marsilea. Am. J. Bot. 1968, 55, 884–889. [Google Scholar] [CrossRef]
- Gaudet, J. Marsilea vestita: Conversion of the Water Form to the Land Form by Darkness and by Far-Red Light. Science 1963, 140, 975–976. [Google Scholar] [CrossRef]
- Bristow, J.M. The effects of carbon dioxide on the growth and development of amphibious plants. Botany 1969, 47, 1803–1807. [Google Scholar] [CrossRef]
- Titus, J.E.; Sullivan, P.G. Heterophylly in the yellow waterlily, Nuphar variegata (Nymphaeaceae): Effects of CO2, natural sediment type, and water depth. Am. J. Bot. 2001, 88, 1469–1478. [Google Scholar] [CrossRef]
- Chitwood, D.H.; Sinha, N.R. Evolutionary and environmental forces sculpting leaf development. Curr. Biol. 2016, 26, R297–R306. [Google Scholar] [CrossRef]
- He, D.; Guo, P.; Gugger, P.F.; Guo, Y.; Liu, X.; Chen, J. Investigating the molecular basis for heterophylly in the aquatic plant Potamogeton octandrus (Potamogetonaceae) with comparative transcriptomics. PeerJ 2018, 6, e4448. [Google Scholar] [CrossRef]
- McCallum, W.B. On the nature of the stimulus causing the change of form and structure in Proserpinaca palustris. Bot. Gaz. 1902, 34, 93–108. [Google Scholar] [CrossRef]
- Wallenstein, A.; Albert, L. Plant Morphology: Its control in proserpinaca by photoperiod, temperature, and gibberellic acid. Science 1963, 140, 998–1000. [Google Scholar] [CrossRef]
- Kane, M.E.; Albert, L.S. Environmental and growth regulator effects on heterophylly and growth of Proserpinaca intermedia (Haloragaceae). Aquat. Bot. 1982, 13, 73–85. [Google Scholar] [CrossRef]
- Cook, C.D. On the determination of leaf form in Ranunculus aquatilis. New Phytol. 1969, 68, 469–480. [Google Scholar] [CrossRef]
- Young, J.P.; Dengler, N.G.; Donnelly, P.M.; Dickinson, T.A. Heterophylly in Ranunculus flabellaris: The effect of abscisic acid on leaf ultrastructure. Ann. Bot. 1990, 65, 603–615. [Google Scholar] [CrossRef]
- Johnson, M.P. Temperature Dependent Leaf Morphogenesis in Ranunculus flabellaris. Nature 1967, 214, 1354–1355. [Google Scholar] [CrossRef]
- Kim, J.; Joo, Y.; Kyung, J.; Jeon, M.; Park, J.Y.; Lee, H.G.; Chung, D.S.; Lee, E.; Lee, I. A molecular basis behind heterophylly in an amphibious plant, Ranunculus trichophyllus. PLoS Genet. 2018, 14, e1007208. [Google Scholar] [CrossRef]
- Nakayama, H.; Fukushima, K.; Fukuda, T.; Yokoyama, J.; Kimura, S. Molecular Phylogeny Determined Using Chloroplast DNA Inferred a New Phylogenetic Relationship of Rorippa aquatica (Eaton) EJ Palmer & Steyermark (Brassicaceae)—Lake Cress. Am. J. Plant Sci. 2014, 5, 48–54. [Google Scholar]
- Nakayama, H.; Nakayama, N.; Seiki, S.; Kojima, M.; Sakakibara, H.; Sinha, N.; Kimura, S. Regulation of the KNOX-GA gene module induces heterophyllic alteration in North American Lake Cress. Plant Cell 2014, 26, 4733–4748. [Google Scholar] [CrossRef]
- Momokawa, N.; Kadono, Y.; Kudoh, H. Effects of light quality on leaf morphogenesis of a heterophyllous amphibious plant, Rotala hippuris. Ann. Bot. 2011, 108, 1299–1306. [Google Scholar] [CrossRef]
- Raven, J.A.; Beardall, J. The ins and outs of CO2. J. Exp. Bot. 2015, 67, 1–13. [Google Scholar]
- Jackson, S.D. Plant responses to photoperiod. New Phytol. 2009, 181, 517–531. [Google Scholar] [CrossRef]
- Upadhyaya, H.; Sahoo, L.; Panda, S. Molecular Stress Physiology of Plants; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Al-Karaki, G.N. Growth, water use efficiency, and sodium and potassium acquisition by tomato cultivars grown under salt stress. J. Plant Nutr. 2000, 23, 1–8. [Google Scholar] [CrossRef]
- Najla, S.; Vercambre, G.; Pagès, L. Tomato plant architecture as affected by salinity: Descriptive analysis and integration in a 3-D simulation model. Botany 2009, 87, 893–904. [Google Scholar] [CrossRef]
- Hu, X.; Li, N.; Wu, L. Quantitative iTRAQ-based proteomic analysis of phosphoproteins and ABA-regulated phosphoproteins in maize leaves under osmotic stress. Sci. Rep. 2015, 5, 15626. [Google Scholar] [CrossRef] [PubMed]
- Skirycz, A.; Claeys, H.; De Bodt, S. Pause-and-Stop: The Effects of Osmotic Stress on Cell Proliferation during Early Leaf Development in Arabidopsis and a Role for Ethylene Signaling in Cell Cycle Arrest. Plant Cell 2011, 23, 1876–1888. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Yang, R.; Huo, Y. Expression of cotton PLATZ1 in transgenic Arabidopsis reduces sensitivity to osmotic and salt stress for germination and seedling establishment associated with modification of the abscisic acid, gibberellin, and ethylene signalling pathways. BMC Plant Biol. 2018, 18, 218. [Google Scholar] [CrossRef] [PubMed]
- Spence, D.H.N. The Zonation of Plants in Freshwater Lakes. Adv. Ecol. Res. 1982, 12, 37–125. [Google Scholar]
- Vervuren, P.; Blom, C.; Kroon, H. Extreme flooding events on the Rhine and the survival and distribution of riparian plant species. J. Ecol. 2003, 91, 135–146. [Google Scholar] [CrossRef]
- Richards, P.; Sculthorpe, C. The Biology of Aquatic Vascular Plants Edward Arnold. J. Ecol. 1968, 56, 915. [Google Scholar] [CrossRef]
- Paterson, D.M.; Black, K.S. Water flow, sediment dynamics and benthic biology. Adv. Ecol. Res. 1999, 29, 155–193. [Google Scholar]
- Robionek, A.; Banas, K.; Chmara, R.; Szmeja, J. The avoidance strategy of environmental constraints by an aquatic plant Potamogeton alpinus in running waters. Ecol. Evol. 2015, 5, 3327–3337. [Google Scholar] [CrossRef]
- Bociag, K.; Galka, A.; Lazarewicz, T.; Szmeja, J. Mechanical strength of stems in aquatic macrophytes. Acta Soc. Bot. Pol. 2011, 78, 181–187. [Google Scholar] [CrossRef]
- Minorsky, P.V. The hot and the classic. Plant Physiol. 2003, 133, 1671–1672. [Google Scholar] [CrossRef]
- Nakayama, H.; Sinha, N.R.; Kimura, S. How do plants and phytohormones accomplish heterophylly, leaf phenotypic plasticity, in response to environmental cues. Front. Plant Sci. 2017, 8, 1717. [Google Scholar] [CrossRef] [PubMed]
- Schlichting, C.D.; Wund, M.A. Phenotypic plasticity and epigenetic marking: An assessment of evidence for genetic accommodation. Evolution 2014, 68, 656–672. [Google Scholar] [CrossRef] [PubMed]
- Legris, M.; Klose, C.; Burgie, E.S.; Rojas, C.C.R.; Neme, M.; Hiltbrunner, A.; Wigge, P.A.; Schafer, E.; Vierstra, R.D.; Casal, J.J. Phytochrome B integrates light and temperature signals in Arabidopsis. Science 2016, 354, 897–900. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Domijan, M.; Klose, C.; Biswas, S.; Ezer, D.; Gao, M.; Khattak, A.K.; Box, M.; Charoensawan, V.; Cortijo, S.; et al. Phytochromes function as thermosensors in Arabidopsis. Science 2016, 354, 886–889. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, T.; Kimura, S. Plant temperature sensors. Sensors 2018, 18, 4365. [Google Scholar] [CrossRef]
- Li, X.; He, D.; Guo, Y. Morphological structure and physiological research of heterophylly in Potamogeton octandrus. Plant Syst. Evol. 2019, 305, 223–232. [Google Scholar] [CrossRef]
- Hattori, Y.; Nagai, K.; Furukawa, S.; Song, X.J.; Kawano, R.; Sakakibara, H.; Wu, J.; Matsumoto, T.; Yoshimura, A.; Kitano, H.; et al. The ethylene response factors SNORKEL1 and SNORKEL2 allow rice to adapt to deep water. Nature 2009, 460, 1026–1030. [Google Scholar] [CrossRef]
- Dittrich, M.; Mueller, H.M.; Bauer, H.; Peirats-Llobet, M.; Rodriguez, P.L.; Geilfus, C.-M.; Carpentier, S.C.; Rasheid, K.S.; Kollist, H.; Merilo, E.; et al. The role of Arabidopsis ABA receptors from the PYR/PYL/RCAR family in stomatal acclimation and closure signal integration. Nat. Plants 2019. [Google Scholar] [CrossRef]
- Holmes, M.G.; Klein, W.H. The Light and Temperature Environments. In Plant life in aquatic and amphibious habitats; Blackwell Scientific Publisher: Oxford, UK, 1987; pp. 3–22. [Google Scholar]
- Liu, X.; Luo, M.; Yang, S. Role of epigenetic modifications in plant responses to environmental stresses. Setting the Stage for the Next Generation: Epigenetic Reprogramming During Sexual Plant Reproduction. In Nuclear Functions in Plant Transcription, Signaling and Development; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar]
- Baker-Brosh, K.F.; Peet, R.K. The ecological significance of lobed and toothed leaves in temperate forest trees. Ecology 1997, 78, 1250–1255. [Google Scholar] [CrossRef]
- Chitwood, D.H.; Kumar, R.; Headland, L.R. A quantitative genetic basis for leaf morphology in a set of precisely defined tomato introgression lines. Plant Cell 2013, 25, 2465–2481. [Google Scholar] [CrossRef]
- Andres, R.J.; Coneva, V.; Frank, M.H. Modifications to a LATE MERISTEM IDENTITY1 gene are responsible for the major leaf shapes of Upland cotton (Gossypium hirsutum L.). Proc. Natl. Acad. Sci. USA 2016, 114, E57. [Google Scholar] [CrossRef] [PubMed]
- Vuolo, F.; Mentink, R.A.; Hajheidari, M. Coupled enhancer and coding sequence evolution of a homeobox gene shaped leaf diversity. Genes Dev. 2016, 30, 2370–2375. [Google Scholar] [CrossRef] [PubMed]
- Helliker, B.R.; Richter, S.L. Subtropical to boreal convergence of tree-leaf temperatures. Nature 2008, 454, 511–514. [Google Scholar] [CrossRef] [PubMed]
- Vogel, S. Leaves in the lowest and highest winds: Temperature, force and shape. New Phytol. 2009, 183, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Palacio-lopez, K.; Beckage, B.; Scheiner, S.M.; Molofsky, J. The ubiquity of phenotypic plasticity in plants: A synthesis. Ecol. Evol. 2015, 5, 3389–3400. [Google Scholar] [CrossRef] [PubMed]
- Iida, S.; Ikeda, M.; Amano, M.; Sakayama, H.; Kadono, Y.; Kosuge, K. Loss of heterophylly in aquatic plants: Not ABA-mediated stress but exogenous ABA treatment induces stomatal leaves in Potamogeton perfoliatus. J. Plant Res. 2016, 129, 853–862. [Google Scholar] [CrossRef][Green Version]
| Species | Family | Common Name | Treatments | References |
|---|---|---|---|---|
| Alisma graminifolium | Alismataceae | - | light | [19] |
| Callitriche heterophylla | Callitrichaceae | two-headed water-starwort | ABA, GA, temperature, osmotic stress | [11,20,21] |
| Callitriche intermedia | Callitrichaceae | narrowleaf water-starwort | osmotic stress, temperature | [22,23] |
| Callitriche stagnalis | Callitrichaceae | pond water starwort | GA | [20,21] |
| Hippuris vulgaris | Hippuridaceae | common mare’s tail | ABA, temperature, light intensity, R/FR ratio, osmotic stress | [5,9,24,25] |
| Hygrophila difformis | Acanthaceae | Water-Wisteria | ABA, ethylene, GA, humidity, temperature | [26,27] |
| Ilex aquifolium | Aquifoliaceae | Common holly | mammalian browsing | [28] |
| Ludwigia arcuata | Onagraceae | piedmont primrose-willow | ABA, ethylene, temperature | [6,14,29,30] |
| Marsilea quadrifolia | Marsileaceae | European water clover | ABA, blue light, CO2 | [12,13] |
| Marsilea vestita | Marsileaceae | hairy water clover | CO2, light intnsity, light quality | [31,32] |
| Myriophyllum brasiliense | Haloragaceae | red stemmed parrot feather | CO2 | [33] |
| Nuphar variegate | Nymphaeaceae | yellow water lily | CO2, sediment type and water depth | [34] |
| Nuphar lutea | Nymphaeaceae | yellow pond-lily | water depth | [16] |
| Potamogeton nodosus | Potamogetonaceae | longleaf pondweed | ABA | [4] |
| Potamogeton alpinus | Potamogetonaceae | - | water flow | [35] |
| Potamogeton octandrus | Potamogetonaceae | - | submerged or floating condition, development | [36] |
| Proserpinaca palustris | Haloragidaceae | marsh mermaid-weed | ABA, GA, light intensity, humidity, osmotic stress, photoperiod | [17,37,38] |
| Proserpinaca intermedia | Haloragidaceae | intermediate mermaid-weed | photoperiod | [39] |
| Ranunculus aquatilis | Ranunculaceae | water crowfoot | photoperiod | [40] |
| Ranunculus flabellaris | Ranunculaceae | yellow water buttercup | ABA, temperature, CO2 | [33,41,42] |
| Ranunculus trichophyllus | Ranunculaceae | threadleaf crowfoot | ABA, ethylene, temperature, hypoxia | [43] |
| Rorippa aquatica | Brassicaceae | North American lake cress | GA, ethylene, temperature, light intensity | [15,44,45] |
| Rotala hippuris | Lythraceae | - | R/FR ratio, blue light intensity | [46] |
| Sagittaria sagittifolia | Alismataceae | arrowhead | light intensity | [19] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, G.; Hu, S.; Hou, H.; Kimura, S. Heterophylly: Phenotypic Plasticity of Leaf Shape in Aquatic and Amphibious Plants. Plants 2019, 8, 420. https://doi.org/10.3390/plants8100420
Li G, Hu S, Hou H, Kimura S. Heterophylly: Phenotypic Plasticity of Leaf Shape in Aquatic and Amphibious Plants. Plants. 2019; 8(10):420. https://doi.org/10.3390/plants8100420
Chicago/Turabian StyleLi, Gaojie, Shiqi Hu, Hongwei Hou, and Seisuke Kimura. 2019. "Heterophylly: Phenotypic Plasticity of Leaf Shape in Aquatic and Amphibious Plants" Plants 8, no. 10: 420. https://doi.org/10.3390/plants8100420
APA StyleLi, G., Hu, S., Hou, H., & Kimura, S. (2019). Heterophylly: Phenotypic Plasticity of Leaf Shape in Aquatic and Amphibious Plants. Plants, 8(10), 420. https://doi.org/10.3390/plants8100420





