Magnesium-Deficiency Effects on Pigments, Photosynthesis and Photosynthetic Electron Transport of Leaves, and Nutrients of Leaf Blades and Veins in Citrus sinensis Seedlings
Abstract
1. Introduction
2. Results
2.1. Mg-Deficiency Symptoms in C. sinensis Seedlings
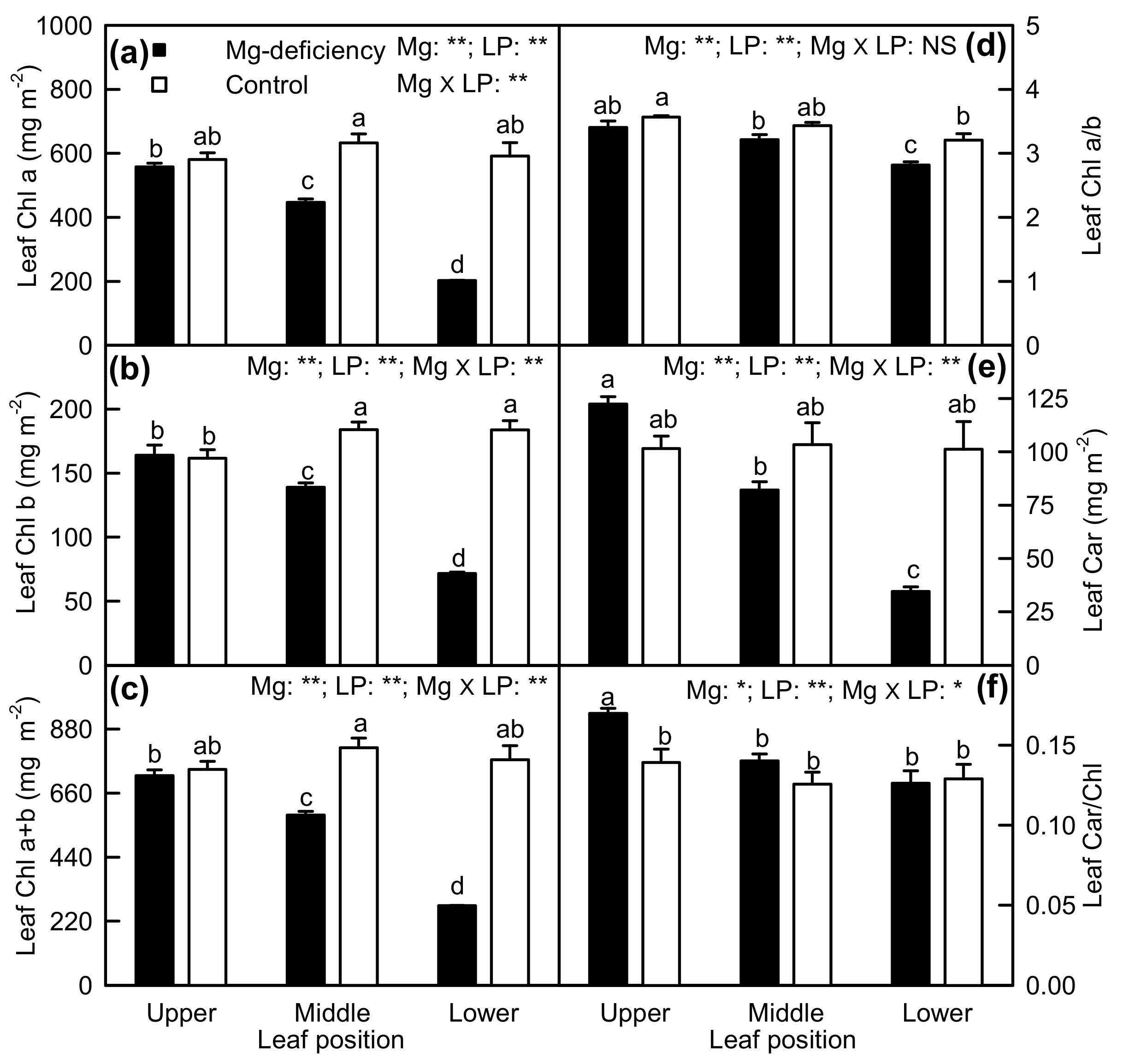
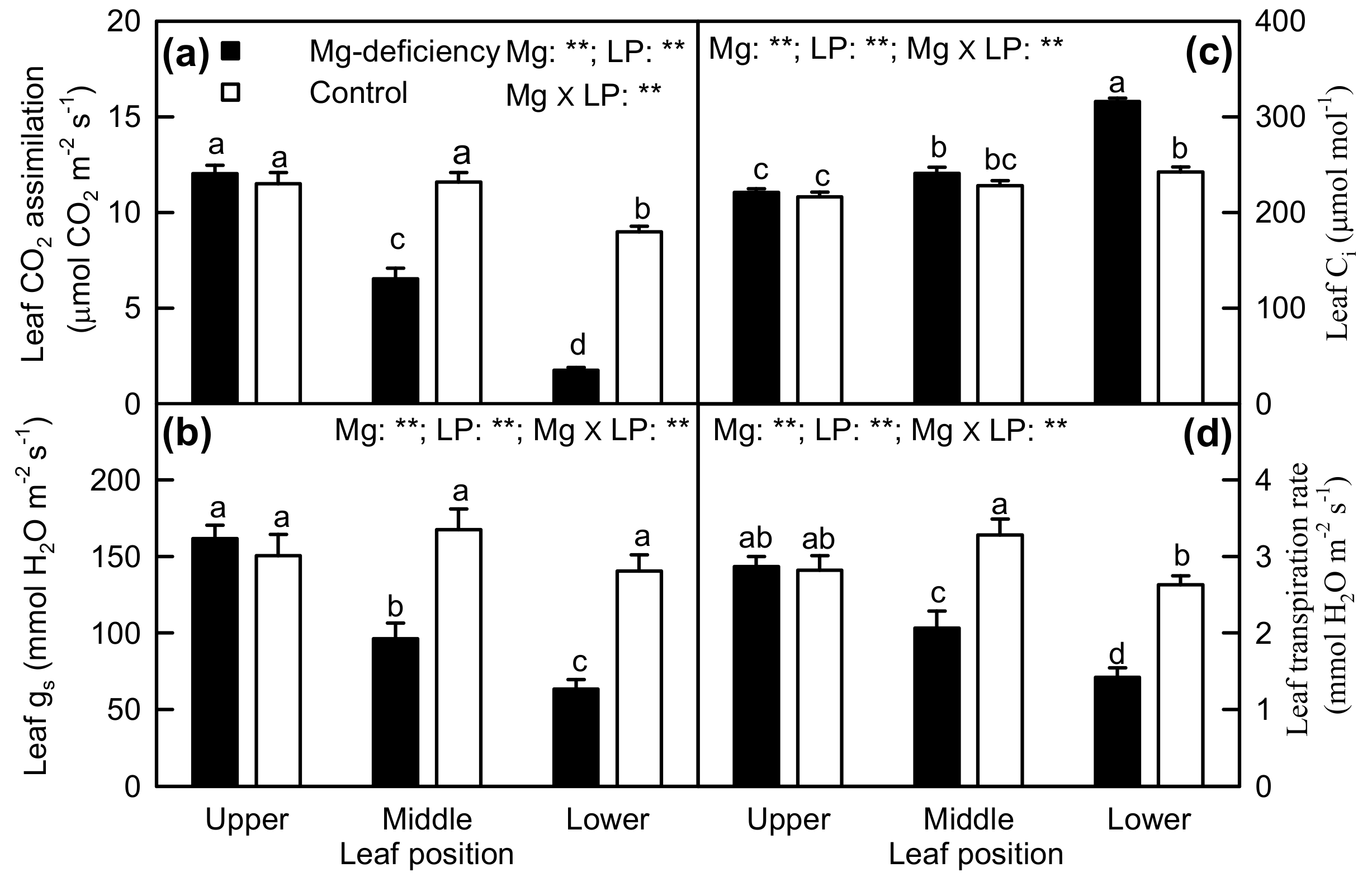
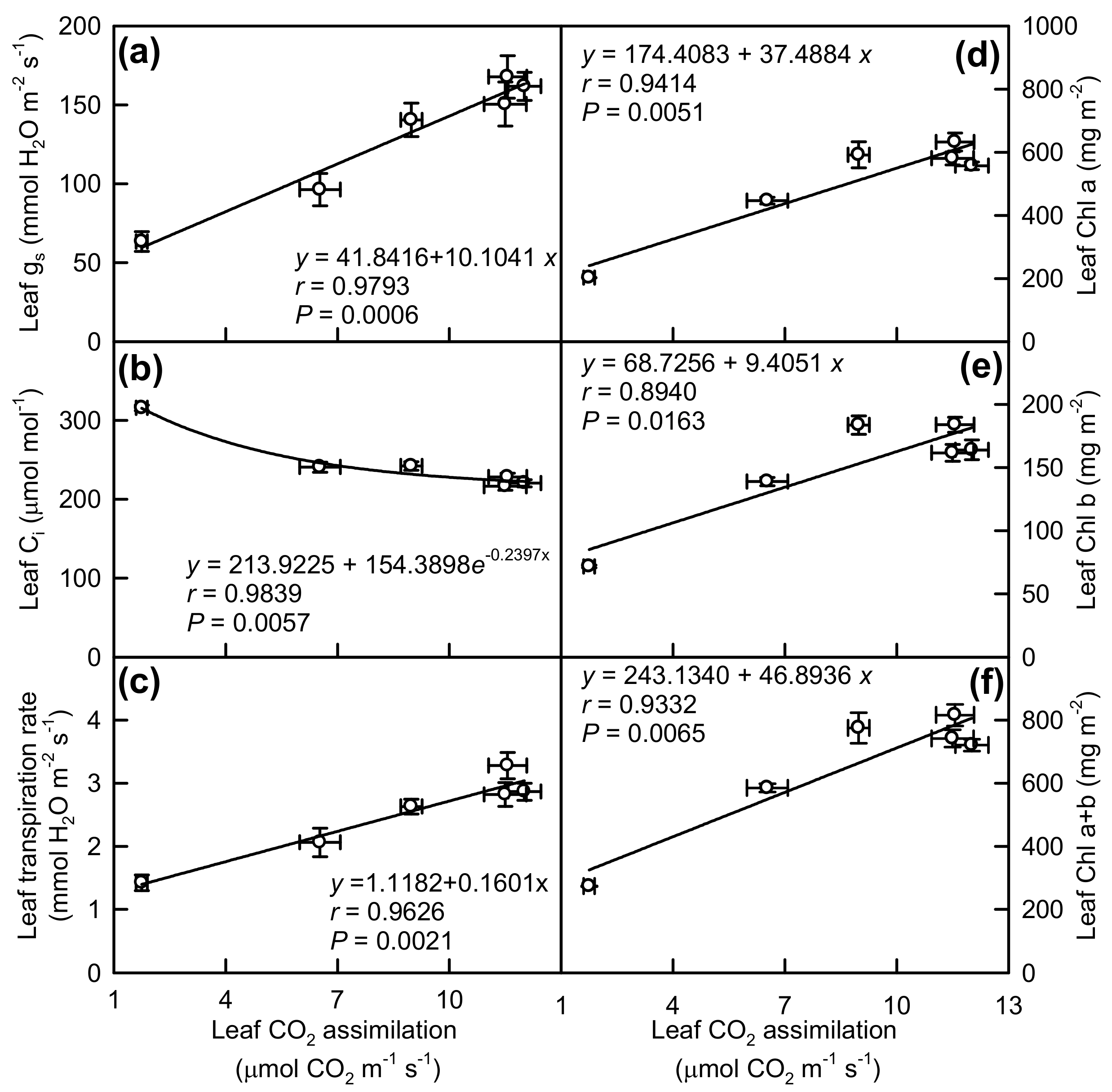
2.2. Leaf Pigments and Gas Exchange
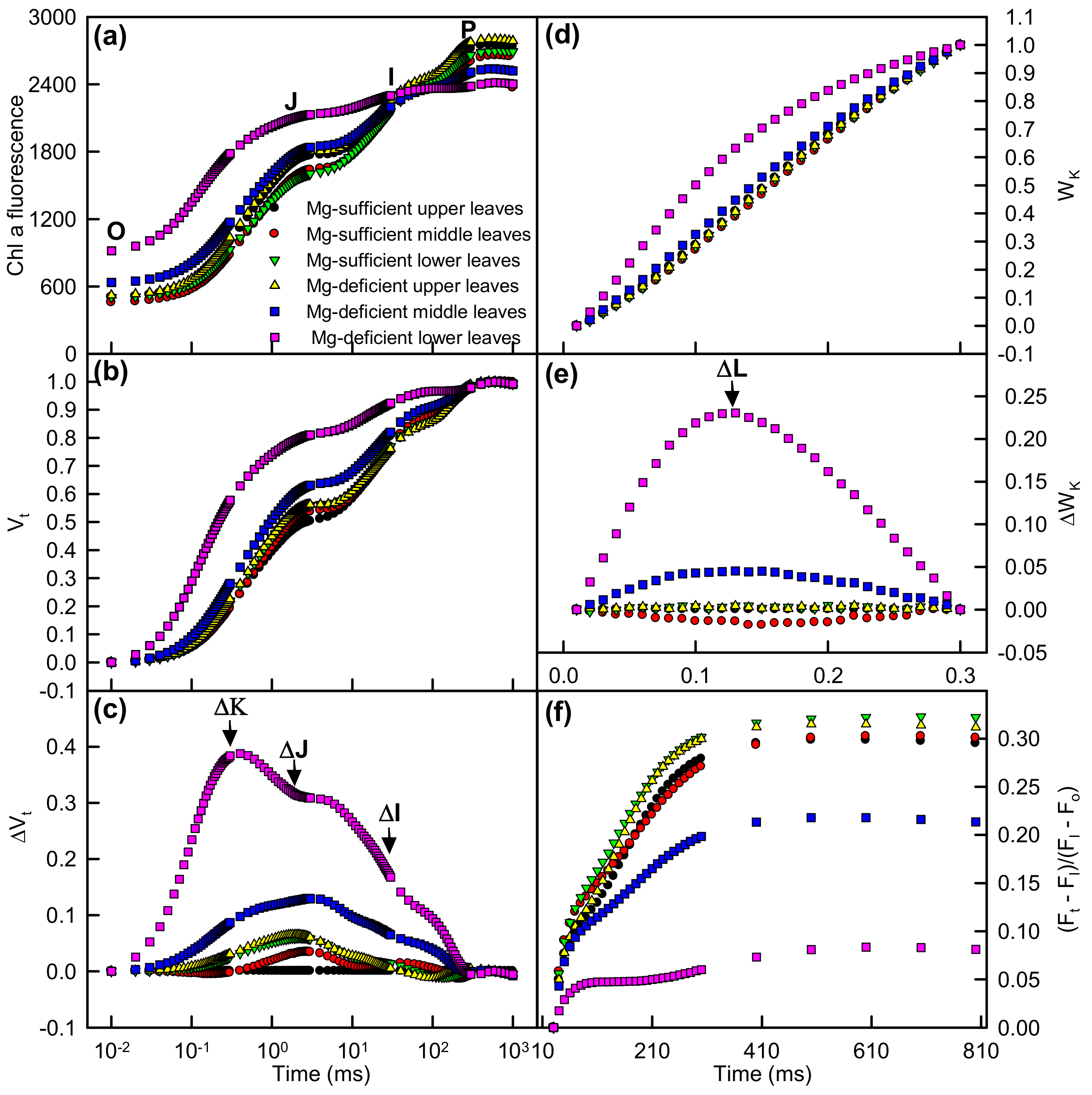
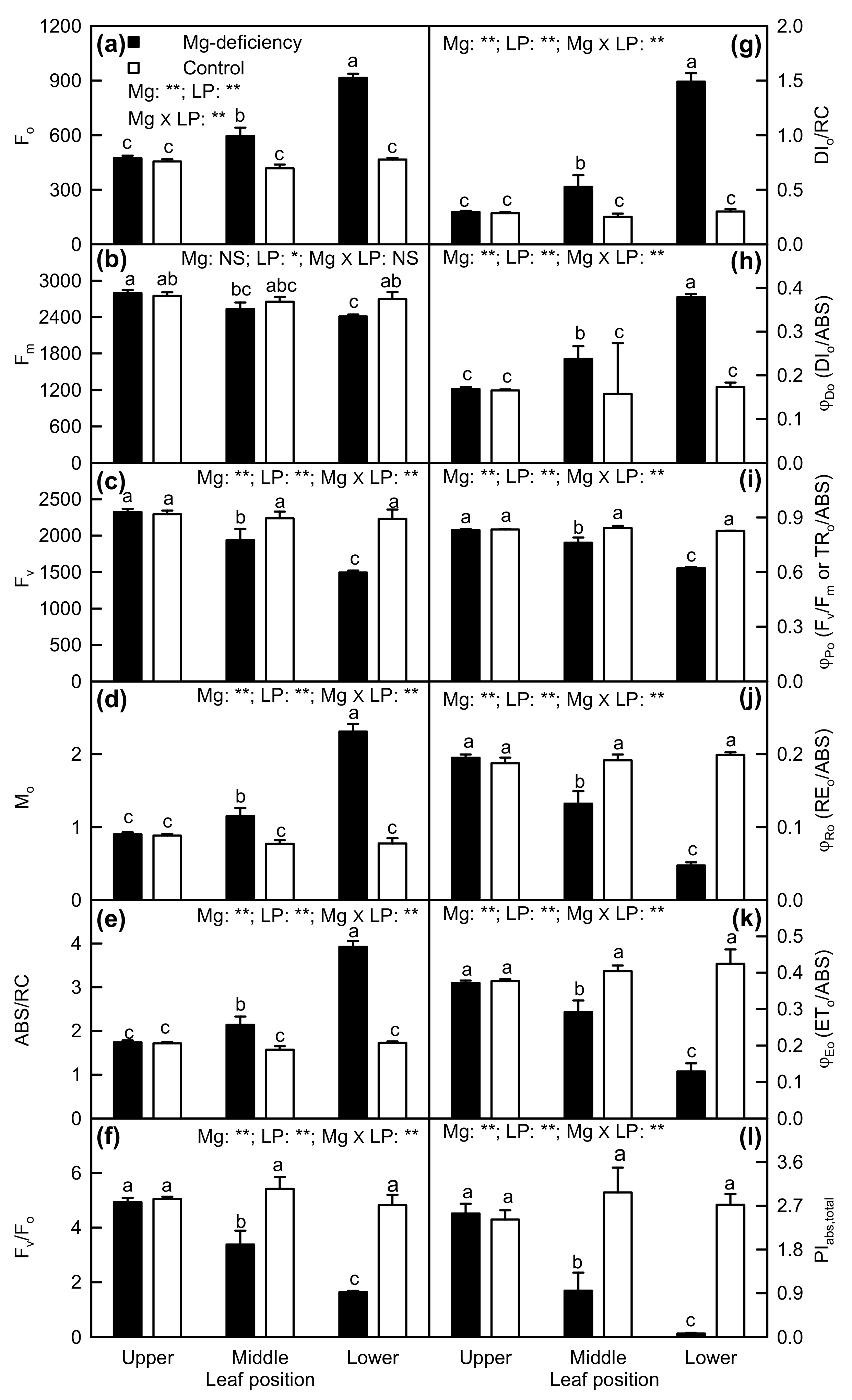
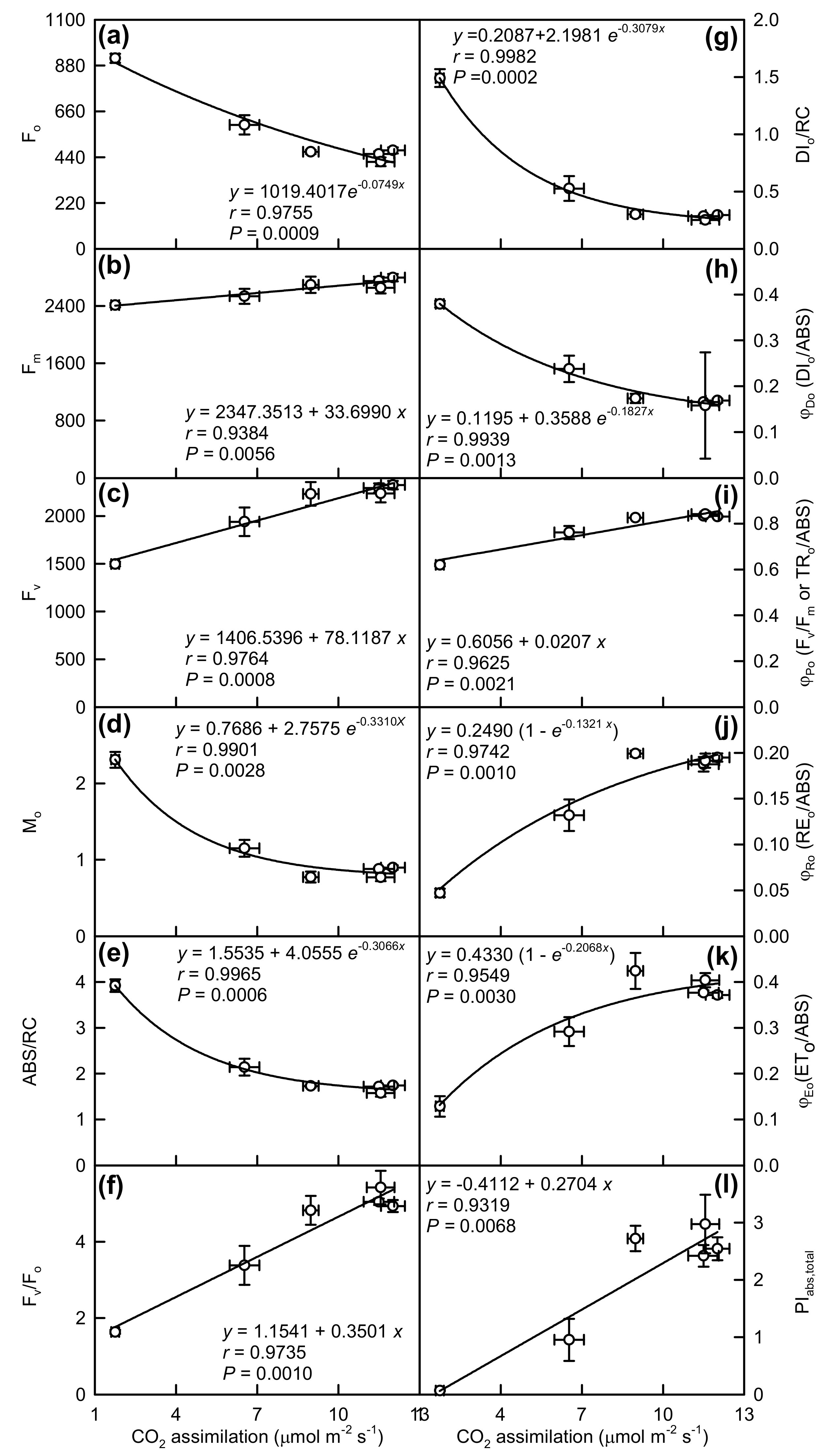
2.3. Leaf OJIP Transients and Related Parameters
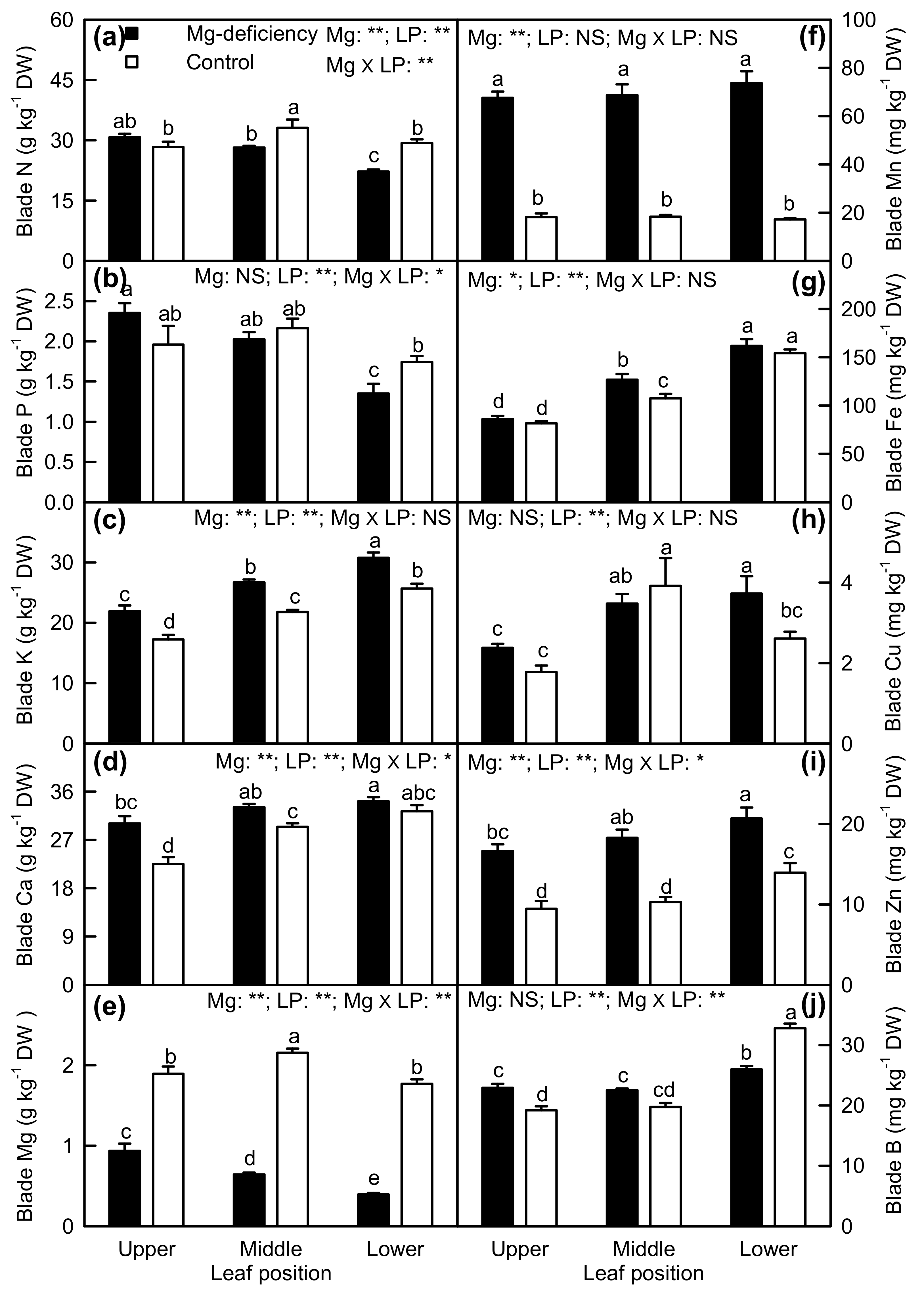
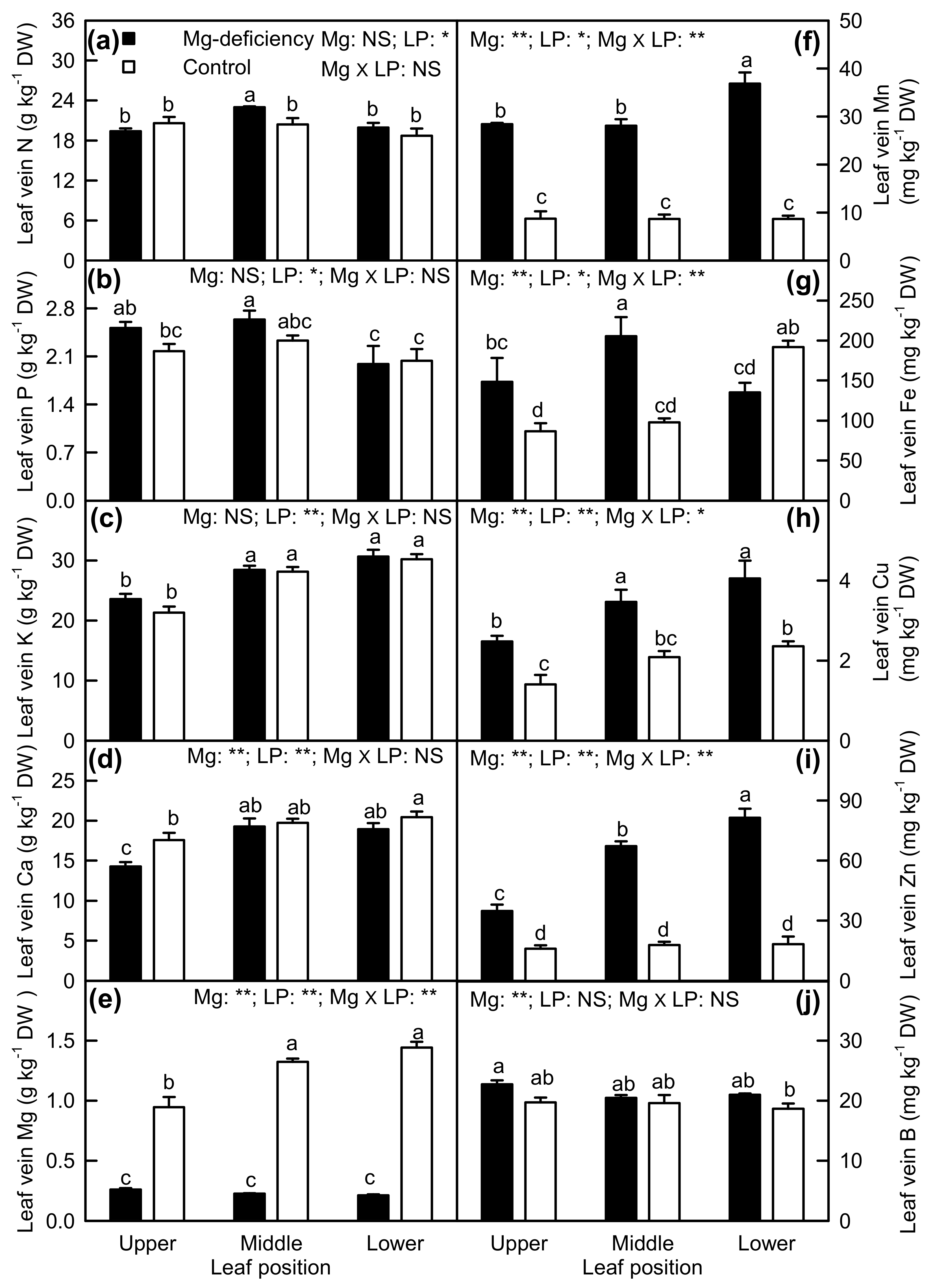
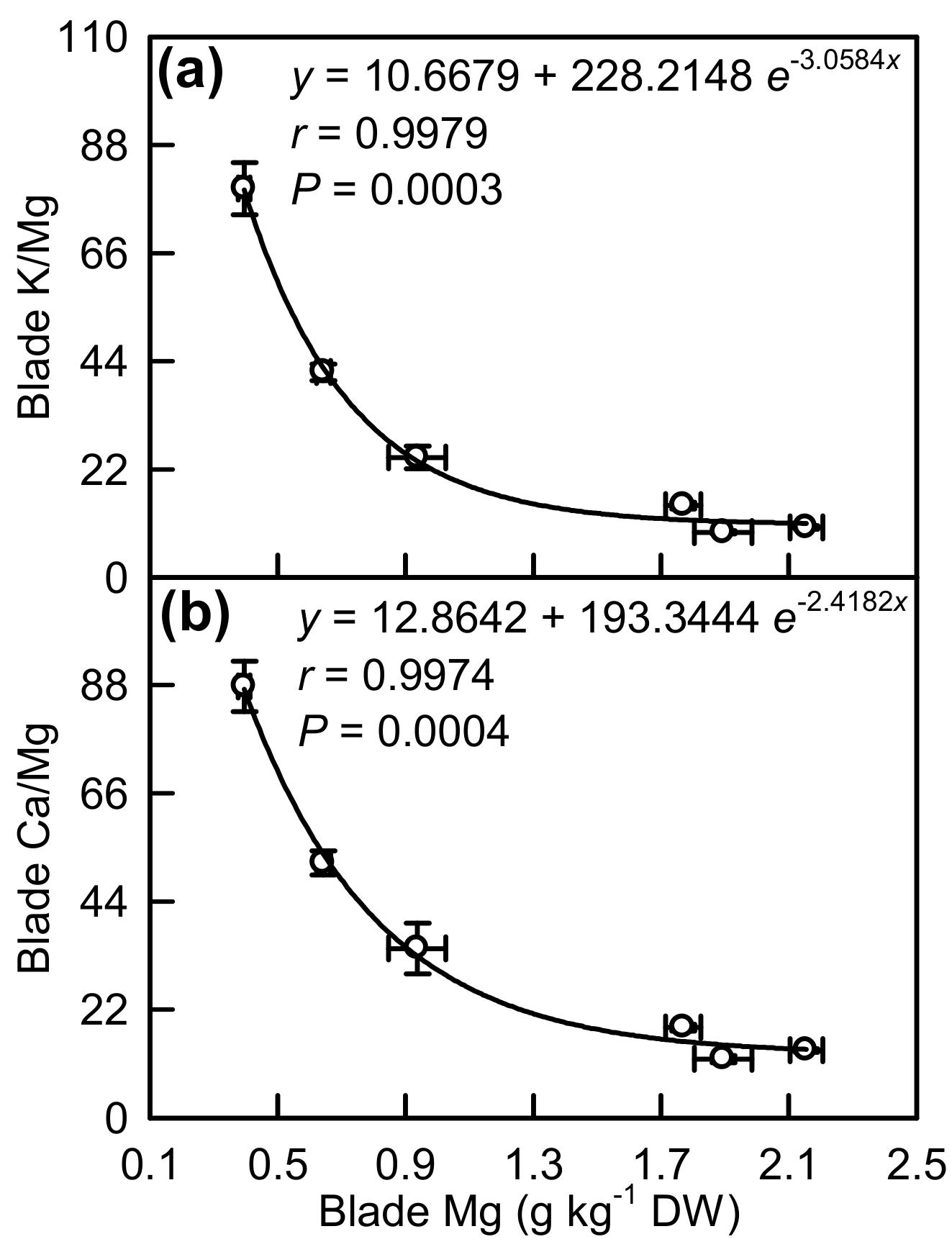
2.4. Element Concentrations in Leaf Blades and Veins
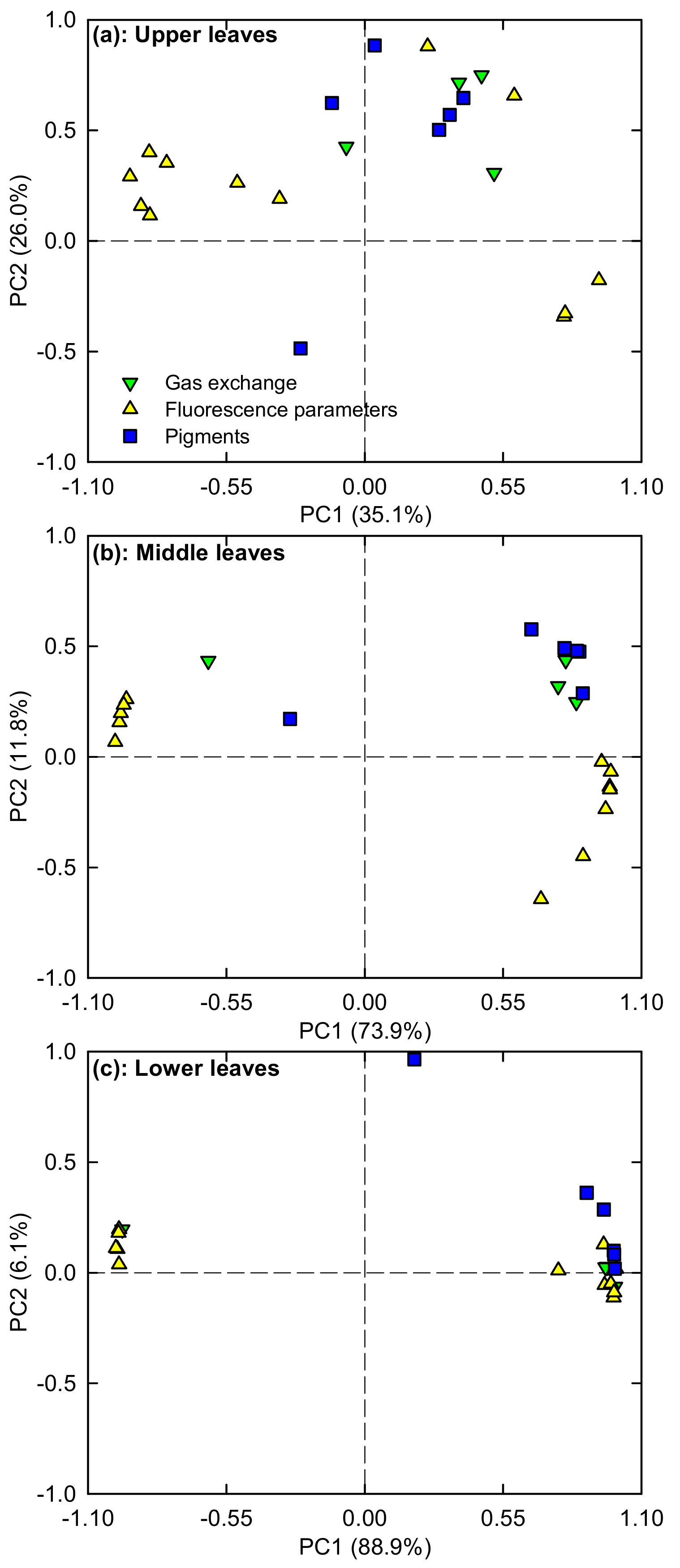
2.5. PCA Loading Plots
3. Discussion
3.1. Responses of Leaf Photosynthesis and Related Parameters to Mg-Deficiency Increased with Increasing Leaf Age, and Mg-Deficiency Increased Their Differences Among Leaves with Various Ages
3.2. Antagonistic Interactions Existed between Mg and Some Elements
3.3. Mg-Deficiency-Induced Alterations of Some Nutrient Concentrations Differed between Leaf Blades and Veins, and Mg-Deficiency-Induced Enlargement and Corkiness of Veins Were Not Caused by Mg-Deficiency-Induced B-Starvation
3.4. Impairment of Photosynthetic Electron Transport Chain Might Contribute to Mg-Deficiency-Induced Decline in Leaf CO2 Assimilation
4. Materials and Methods
4.1. Plant Culture and Mg Treatments
4.2. Leaf Pigments and Gas Exchange
4.3. Leaf OJIP Transients and Related Parameters
4.4. Assays of Elements
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Billard, V.; Maillard, A.; Coquet, L.; Jouenne, T.; Cruz, F.; Garcia-Mina, J.M.; Yvin, J.-C.; Ourry, A.; Etienne, P. Mg deficiency affects leaf Mg remobilization and the proteome in Brassica napus. Plant Physiol. Biochem. 2016, 107, 337–343. [Google Scholar] [CrossRef]
- Jezek, M.; Geilfus, C.M.; Bayer, A.; Mühling, K.H. Photosynthesis capacity, nutrient status, and growth of maize (Zea mays L.) upon MgSO4 leaf-application. Front. Plant Sci. 2015, 5, 781. [Google Scholar] [CrossRef]
- Li, C.-P.; Qi, Y.-P.; Zhang, J.; Yang, L.-T.; Wang, D.-H.; Ye, X.; Lai, N.-W.; Tan, L.-L.; Li, D.; Chen, L.-S. Magnesium-deficiency-induced alterations of gas exchange, major metabolites and key enzymes differ among roots, lower and upper leaves of Citrus sinensis seedlings. Tree Physiol. 2017, 37, 1564–1581. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.-Y.; Qi, Y.-P.; Lee, J.; Yang, L.-T.; Guo, P.; Jiang, H.-X.; Chen, L.-S. Proteomic analysis of Citrus sinensis roots and leaves in response to long-term magnesium-deficiency. BMC Genom. 2015, 16, 253. [Google Scholar] [CrossRef]
- Ma, C.-L.; Qi, Y.-P.; Liang, W.-W.; Yang, L.-T.; Lu, Y.-B.; Guo, P.; Ye, X.; Chen, L.-S. MicroRNA regulatory mechanisms on Citrus sinensis leaves to magnesium-deficiency. Front. Plant Sci. 2016, 7, 201. [Google Scholar] [CrossRef]
- Cakmak, I.; Yazici, A.M. Magnesium: A forgotten element in crop production. Better Crops 2010, 94, 23–25. [Google Scholar]
- Verbruggen, N.; Hermans, C. Physiological and molecular responses to magnesium nutritional imbalance in plants. Plant Soil 2013, 368, 87–99. [Google Scholar] [CrossRef]
- Cai, Y.-T.; Zhang, H.; Qi, Y.-P.; Ye, X.; Huang, Z.-R.; Guo, J.-X.; Chen, L.-S.; Yang, L.-T. Responses of reactive oxygen species and methylglyoxal metabolisms to magnesium-deficiency differ greatly among the roots, upper and lower leaves of Citrus sinensis. BMC Plant Biol. 2019, 19, 76. [Google Scholar] [CrossRef]
- Guo, W.; Hussain, H.; Liang, Z.; Yang, D. Magnesium deficiency in plants: An urgent problem. Crop J. 2016, 4, 83–91. [Google Scholar] [CrossRef]
- Ling, L.-L.; Peng, L.-Z.; Cao, L.; Jiang, C.-L.; Chun, C.-P.; Zhang, G.-Y.; Wang, Z.-X. Effect of magnesium deficiency on photosynthesis characteristic of Beibei 447 Jinchen orange. J. Fruit Sci. 2009, 26, 275–280. [Google Scholar]
- Li, Y.; Han, M.-Q.; Lin, F.; Ten, Y.; Lin, J.; Zhu, D.-H.; Guo, P.; Weng, Y.-B.; Chen, L.-S. Soil chemical properties, ‘Guanximiyou’ pummelo leaf mineral nutrient status and fruit quality in the southern region of Fujian province, China. J. Soil Sci. Plant Nutr. 2015, 15, 615–628. [Google Scholar] [CrossRef]
- Yang, G.-H.; Yang, L.-T.; Jiang, H.-X.; Wang, P.; Chen, L.-S. Physiological impacts of magnesium-deficiency in Citrus seedlings: Photosynthesis, antioxidant system and carbohydrates. Trees Struct. Funct. 2012, 26, 1237–1250. [Google Scholar] [CrossRef]
- Li, Y.; Liu, X.-H.; Zhuang, W.-M. The effect of magnesium deficiency on photosynthesis of longan (Dimocarpus longan Lour.) seedlings. Acta Hortic. Sin. 2001, 28, 101–106. [Google Scholar]
- Hermans, C.; Verbruggen, N. Physiological characterization of magnesium deficiency in Arabidopsis thaliana. J. Exp. Bot. 2005, 56, 2153–2161. [Google Scholar] [CrossRef]
- da Silva, D.M.; Brandão, I.R.; Alves, J.D.; de Santos, M.O.; de Souza, K.R.D.; de Silveira, H.R.O. Physiological and biochemical impacts of magnesium-deficiency in two cultivars of coffee. Plant Soil 2014, 382, 133–150. [Google Scholar] [CrossRef]
- Ceylan, Y.; Kutman, U.B.; Mengutay, M.; Cakmak, I. Magnesium applications to growth medium and foliage affect the starch distribution, increase the grain size and improve the seed germination in wheat. Plant Soil 2016, 406, 145–1561. [Google Scholar] [CrossRef]
- Fischer, E.S.; Lohaus, G.; Heineke, D.; Heldt, H.W. Magnesium deficiency results in accumulation of carbohydrates and amino acids in source and sink leaves of spinach. Physiol. Plant 1998, 102, 16–20. [Google Scholar] [CrossRef]
- Kobayashi, N.I.; Saito, T.; Iwata, N.; Ohmae, Y.; Iwata, R.; Tanoi, K.; Nakanishi, T.M. Leaf senescence in rice due to magnesium deficiency mediated defect in transpiration rate before sugar accumulation and chlorosis. Physiol. Plant 2013, 148, 490–501. [Google Scholar] [CrossRef]
- Han, S.; Tang, N.; Jiang, H.-X.; Yang, L.-T.; Li, Y.; Chen, L.-S. CO2 assimilation, photosystem II photochemistry, carbohydrate metabolism and antioxidant system of Citrus leaves in response to boron stress. Plant Sci. 2009, 176, 143–153. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Oukarroum, A.; Alexandrov, V.; Kouzmanova, M.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Allakhverdiev, S.I.; Goltsev, V. Identification of nutrient deficiency in maize and tomato plants by in vivo chlorophyll a fluorescence measurements. Plant Physiol. Biochem. 2014, 81, 16–25. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Bąba, W.; Gediga, K.; Goltsev, V.; Samborska, I.A.; Cetner, M.D.; Dimitrovam, S.; Piszcz, U.; Bielecki, K.; Karmowska, K.; et al. Chlorophyll fluorescence as a tool for nutrient status identification in rapeseed plants. Photosynth. Res. 2018, 136, 329–343. [Google Scholar] [CrossRef]
- Srinivasarao, C.; Shanker, A.K.; Kundu, S.; Reddy, S. Chlorophyll fluorescence induction kinetics and yield responses in rainfed crops with variable potassium nutrition in K deficient semi-arid alfisols. J. Photochem. Photobiol. B 2016, 160, 86–95. [Google Scholar] [CrossRef]
- Carstensen, A.; Herdean, A.; Schmidt, S.B.; Sharma, A.; Spetea, C.; Pribil, M.; Husted, S. The mmpacts of phosphorus deficiency on the photosynthetic electron transport chain. Plant Physiol. 2018, 177, 271–284. [Google Scholar]
- Carstensen, A.; Szameitat, A.E.; Frydenvang, J.; Husted, S. Chlorophyll a fluorescence analysis can detect phosphorus deficiency under field conditions and is an effective tool to prevent grain yield reductions in spring barley (Hordeum vulgare L.). Plant Soil 2019, 434, 79–91. [Google Scholar] [CrossRef]
- Aleksandrov, V.; Krasteva, V.; Paunov, M.; Chepisheva, M.; Kousmanova, M.; Kalaji, H.M.; Goltsev, V. Deficiency of some nutrient elements in bean and maize plants analyzed by luminescent method. Bulg. J. Agric. Sci. 2014, 20, 24–30. [Google Scholar]
- Lin, Z.-H.; Chen, L.-S.; Chen, R.-B.; Zhang, F.-Z.; Jiang, H.-X.; Tang, N. CO2 assimilation, ribulose-1, 5-bisphosphate carboxylase/oxygenase, carbohydrates and photosynthetic electron transport probed by the JIP-test, of tea leaves in response to phosphorus supply. BMC Plant Biol 2009, 9, 43. [Google Scholar] [CrossRef]
- Schmidt, S.B.; Pedas, P.; Laursen, K.H.; Schjoerring, J.K.; Husted, S. Latent manganese deficiency in barley can be diagnosed and remediated on the basis of chlorophyll a fluorescence measurements. Plant Soil 2013, 372, 417–429. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Schansker, G.; Ladle, R.J.; Goltsev, V.; Bosa, K.; Allakhverdiev, S.I.; Brestic, M.; Bussotti, F.; Calatayud, A.; Dąbrowski, P.; et al. Frequently asked questions about in vivo chlorophyll fluorescence: Practical issues. Photosynth. Res. 2014, 122, 121–158. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Schansker, G.; Brestic, M.; Bussotti, F.; Calatayud, A.; Ferroni, L.; Goltsev, V.; Guidi, L.; Jajoo, A.; Li, P.; et al. Frequently asked questions about chlorophyll fluorescence, the sequel. Photosynth. Res. 2017, 132, 13–66. [Google Scholar] [CrossRef]
- Strasser, R.J.; Tsimilli-Micheal, M.; Srivastava, A. Analysis of the chlorophyll a fluorescence transient. In Chlorophyll a Fluorescence: A Signature of Photosynthesis; Papageorgiou, G.C., Ed.; Springer: Berlin, Germany, 2004; Volume 19, pp. 321–362. [Google Scholar]
- Jiang, H.-X.; Chen, L.-S.; Zheng, J.-G.; Han, S.; Tang, N.; Smith, B.R. Aluminum-induced effects on photosystem II photochemistry in Citrus leaves assessed by the chlorophyll a fluorescence transient. Tree Physiol. 2008, 28, 1863–1871. [Google Scholar] [CrossRef]
- Živčák, M.; Olšovská, K.; Slamka, P.; Galambošová, J.; Rataj, V.; Shao, H.B.; Brestič, M. Application of chlorophyll fluorescence performance indices to assess the wheat photosynthetic functions influenced by nitrogen deficiency. Plant Soil Environ. 2015, 60, 210–215. [Google Scholar] [CrossRef]
- Huang, J.-H.; Xu, J.; Ye, X.; Luo, T.-Y.; Ren, L.-H.; Fan, G.-C.; Qi, Y.-P.; Li, Q.; Ferrarezi, R.S.; Chen, L.-S. Magnesium deficiency affects secondary lignification of the vascular system in Citrus sinensis seedlings. Trees Struct. Funct. 2019, 33, 171–182. [Google Scholar] [CrossRef]
- Cao, W.X.; Tibbitts, T.W. Growth, carbon dioxide exchange and mineral accumulation in potatoes grown at different magnesium concentrations. J. Plant Nutr. 1992, 15, 1359–1371. [Google Scholar] [CrossRef]
- Moss, G.I.; Higgins, M.L. Magnesium influences on the fruit quality of sweet orange (Citrus sinensis L. Osbeck). Plant Soil 1974, 41, 103–112. [Google Scholar] [CrossRef]
- Kleiber, T.; Golcz, A.; Krzesinski, W. Effect of magnesium nutrition on onion (Allium cepa L.). Part, I. Yielding and nutrient status. Ecol. Chem. Eng. 2012, 19, 97–105. [Google Scholar] [CrossRef]
- Alim, K.; Frey, E. Quantification of leaf vein patterning. Biophys. J. 2009, 96, 631a. [Google Scholar] [CrossRef][Green Version]
- Zhao, H.; Wu, L.; Chai, T.; Zhang, Y.; Tan, J.; Ma, S. The effects of copper, manganese and zinc on plant growth and elemental accumulation in the manganese-hyperaccumulator Phytolacca americana. J. Plant Physiol. 2012, 169, 1243–1252. [Google Scholar] [CrossRef]
- Vogel-Mikuš, K.; Simčič, J.; Pelicon, P.; Budnar, M.; Kump, P.; Nečemer, M.; Mesjasz-Przybyłowicz, J.; Przybyłowicz, W.J.; Regvar, M. Comparison of essential and non-essential element distribution in leaves of the Cd/Zn hyperaccumulator Thlaspi praecox as revealed by micro-PIXE. Plant Cell Environ. 2008, 31, 1484–1496. [Google Scholar] [CrossRef]
- Han, S.; Chen, L.-S.; Jiang, H.-X.; Smith, B.R.; Yang, L.-T.; Xie, C.-Y. Boron deficiency decreases growth and photosynthesis, and increases starch and hexoses in leaves of Citrus seedlings. J. Plant Physiol. 2008, 165, 1331–1341. [Google Scholar] [CrossRef]
- Chatterjee, C.; Nautiyal, N.; Agarwala, S.C. Influence of changes in manganese and magnesium supply on some aspects of wheat physiology. Soil Sci. Plant Nutr. 1994, 40, 191–197. [Google Scholar] [CrossRef]
- Löhnis, M.P. Effect of magnesium and calcium supply on the uptake of manganese by various crop plants. Plant Soil 1960, 12, 339–376. [Google Scholar] [CrossRef]
- Tang, N.; Li, Y.; Chen, L.-S. Magnesium deficiency-induced impairment of photosynthesis in leaves of fruiting Citrus reticulata trees accompanied by up-regulation of antioxidant metabolism to avoid photooxidative damage. J. Plant Nutr. Soil Sci. 2012, 175, 784–793. [Google Scholar] [CrossRef]
- Jin, X.-L.; Ma, C.-L.; Yang, L.-T.; Chen, L.-S. Alterations of physiology and gene expression due to long-term magnesium-deficiency differ between leaves and roots of Citrus reticulata. J. Plant Physiol. 2016, 198, 103–115. [Google Scholar] [CrossRef]
- Mehne-Jakobs, B. Magnesium deficiency treatment causes reductions in photosynthesis of well-nourished Norway spruce. Trees Struct. Funct. 1996, 10, 293–300. [Google Scholar] [CrossRef]
- Peaslee, D.-E.; Moss, D.N. Photosynthesis in K- and Mg-deficient maize (Zea mays L.) leaves. Soil Sci. Soc. Am. J. 1966, 30, 220–223. [Google Scholar] [CrossRef]
- Sheen, J. Feedback control of gene expression. Photosynth. Res. 1994, 39, 427–438. [Google Scholar] [CrossRef]
- Cave, G.; Tolley, L.C.; Strain, B.R. Effect of carbon dioxide enrichment on chlorophyll content, starch content and starch grain structure in Trifolium subteraneum leaves. Physiol. Plant 1981, 51, 171–174. [Google Scholar] [CrossRef]
- Cetner, M.D.; Kalaji, H.M.; Goltsev, V.; Aleksandrov, V.; Kowalczyk, K.; Borucki, W.; Jajoo, A. Effects of nitrogen-deficiency on efficiency of light-harvesting apparatus in radish. Plant Physiol. Biochem. 2017, 119, 81–92. [Google Scholar] [CrossRef]
- Srivastava, A.; Guisse, B.; Greppin, H.; Strasser, R.J. Regulation of antenna structure and electron transport in photosystem II of Pisum sativum under elevated temperature probed by the fast polyphasic chlorophyll a fluorescence transient: OKJIP. Biochim. Biophys. Acta 1997, 1320, 95–106. [Google Scholar] [CrossRef]
- Hakala, M.; Tuominen, I.; Kerӓnen, M.; Tyystjӓrvi, T.; Tyystjӓrvi, E. Eviddence for the role of the oxygen-evolving manganese complex in photoinhibition of photosystem II. Biochim. Biophys. Acta 2005, 1706, 68–80. [Google Scholar] [CrossRef]
- Schansker, G.; Tüth, S.Z.; Strasser, R.J. Methylviologen and dibromothymoquinone treatments of pea leaves reveal the role of photosystem I in the Chl a fluorescence rise OJIP. Biochim. Biophys. Acta 2005, 1706, 250–261. [Google Scholar] [CrossRef]
- Liao, X.-Y.; Yang, L.-T.; Lu, Y.-B.; Ye, X.; Chen, L.-S. Roles of rootstocks and scions in aluminum-tolerance of Citrus. Acta Physiol. Plant 2015, 37, 1743. [Google Scholar] [CrossRef]
- Aro, E.-M.; McCaffery, S.; Anderson, J.M. Photoinhibition and D1 protein degradation in peas acclimated to different growth irradiances. Plant Physiol. 1993, 103, 835–843. [Google Scholar] [CrossRef]
- Šetlík, I.; Allakhverdiev, S.I.; Nedbal, L.; Šetlíková, E.; Klimov, V.V. Three types of photosystem II photoinactivation. Photosynth. Res. 1990, 23, 39–48. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence–a practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Force, L.; Critchley, C.; van Rensen, J.J.S. New fluorescence parameters for monitoring photosynthesis in plants. 1. The effect of illumination on the fluorescence parameters of the JIP-test. Photosynth. Res. 2003, 78, 17–33. [Google Scholar] [CrossRef]
- Pereira, W.E.; de Siqueira, D.L.; Martinez, C.A.; Puiatti, M. Gas exchange and chlorophyll fluorescence in four Citrus rootstocks under aluminum stress. J. Plant Physiol. 2000, 157, 513–520. [Google Scholar] [CrossRef]
- Lavon, R.; Goldschmidt, E.R.; Salomon, R.; Frank, A. Effect of potassium, magnesium, and calcium deficiencies on carbohydrate pools and metabolism in Citrus leaves. J. Am. Soc. Hortic. Sci. 1995, 120, 54–58. [Google Scholar] [CrossRef]
- Shen, Y.; Xiao, J.; Yang, H.; Zhang, S. Effects of magnesium stress on growth, distribution of several mineral elements and leaf ultrastructure of ‘Harumi’ tangor. Acta Hortiv. Sin. 2011, 38, 849–858. [Google Scholar]
- Li, Q.; Chen, L.-S.; Jiang, H.-X.; Tang, N.; Yang, L.-T.; Lin, Z.-H.; Li, Y.; Yang, G.-H. Effects of manganese-excess on CO2 assimilation, ribulose-1, 5-bisphosphate carboxylase/oxygenase, carbohydrates and photosynthetic electron transport of leaves, and antioxidant systems of leaves and roots in Citrus grandis seedlings. BMC Plant Biol. 2010, 10, 42. [Google Scholar] [CrossRef]
- Long, A.; Zhang, J.; Yang, L.-T.; Ye, X.; Lai, N.-W.; Tan, L.-L.; Lin, D.; Chen, L.-S. Effects of low pH on photosynthesis, related physiological parameters and nutrient profile of Citrus. Front. Plant Sci. 2017, 8, 185. [Google Scholar] [CrossRef]
- Yang, T.-Y.; Cai, L.-Y.; Qi, Y.-P.; Yang, L.-T.; Lai, N.-W.; Chen, L.-S. Increasing nutrient solution pH alleviated aluminum-induced inhibition of growth and impairment of photosynthetic electron transport chain in Citrus sinensis seedlings. BioMed. Res. Int. 2019, 2019, 9058715. [Google Scholar] [CrossRef]
- Guo, P.; Li, Q.; Qi, Y.-P.; Yang, L.-T.; Ye, X.; Chen, H.-H.; Chen, L.-S. Sulfur-mediated-alleviation of aluminum-toxicity in Citrus grandis seedlings. Int. J. Mol. Sci. 2017, 18, 2570. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar]
- Guo, P.; Qi, Y.-P.; Cai, Y.-T.; Yang, T.-Y.; Yang, L.-T.; Huang, Z.-R.; Chen, L.-S. Aluminum effects on photosynthesis, reactive oxygen species and methylglyoxal detoxification in two Citrus species differing in aluminum tolerance. Tree Physiol. 2018, 38, 1548–1565. [Google Scholar] [CrossRef]
- Chen, L.-S.; Cheng, L. Photosystem 2 is more tolerant to high temperature in apple (Malus domestica Borkh.) leaves than in fruit peel. Photosynthetica 2009, 47, 112–120. [Google Scholar] [CrossRef]
- Banks, J.M. Continuous excitation chlorophyll fluorescence parameters: A review for practitioners. Tree Physiol. 2017, 37, 1128–1136. [Google Scholar] [CrossRef]
- Sang, W.; Huang, Z.-R.; Yang, L.-T.; Guo, P.; Ye, X.; Chen, L.-S. Effects of high toxic boron concentration on protein profiles in roots of two Citrus species differing in boron-tolerance revealed by a 2-DE based MS approach. Front. Plant Sci. 2017, 8, 180. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, X.; Chen, X.-F.; Deng, C.-L.; Yang, L.-T.; Lai, N.-W.; Guo, J.-X.; Chen, L.-S. Magnesium-Deficiency Effects on Pigments, Photosynthesis and Photosynthetic Electron Transport of Leaves, and Nutrients of Leaf Blades and Veins in Citrus sinensis Seedlings. Plants 2019, 8, 389. https://doi.org/10.3390/plants8100389
Ye X, Chen X-F, Deng C-L, Yang L-T, Lai N-W, Guo J-X, Chen L-S. Magnesium-Deficiency Effects on Pigments, Photosynthesis and Photosynthetic Electron Transport of Leaves, and Nutrients of Leaf Blades and Veins in Citrus sinensis Seedlings. Plants. 2019; 8(10):389. https://doi.org/10.3390/plants8100389
Chicago/Turabian StyleYe, Xin, Xu-Feng Chen, Chong-Ling Deng, Lin-Tong Yang, Ning-Wei Lai, Jiu-Xin Guo, and Li-Song Chen. 2019. "Magnesium-Deficiency Effects on Pigments, Photosynthesis and Photosynthetic Electron Transport of Leaves, and Nutrients of Leaf Blades and Veins in Citrus sinensis Seedlings" Plants 8, no. 10: 389. https://doi.org/10.3390/plants8100389
APA StyleYe, X., Chen, X.-F., Deng, C.-L., Yang, L.-T., Lai, N.-W., Guo, J.-X., & Chen, L.-S. (2019). Magnesium-Deficiency Effects on Pigments, Photosynthesis and Photosynthetic Electron Transport of Leaves, and Nutrients of Leaf Blades and Veins in Citrus sinensis Seedlings. Plants, 8(10), 389. https://doi.org/10.3390/plants8100389




