The Design of a Plant-Based Biopesticide Formulation with Extracts and Derivatives Containing Naphthoquinones
Abstract
1. Introduction
2. Results and Discussion
2.1. Antifungal Activity of Plant-Derived Secondary Metabolites
2.2. Antifungal Activity of Plant Extracts with Naphthoquinones
2.3. Characterization of the Drosera intermedia Ethyl Acetate Extract
2.4. Design of an Antifungal Formulation Using Drosera intermedia
3. Material and Methods
3.1. Chemicals and Reagents
3.2. Plant Material
3.3. Fungi Material
3.4. Plant Material Extraction
3.5. Chromatographic Analysis
3.5.1. HPLC-PAD and HPLC-MS Analysis Instrumentation
3.5.2. Identification and Quantification of Individual Polyphenols
3.6. Antifungal Activity
3.7. Antifungal Formulation
3.8. Characterization of the Physicochemical Properties of the Formulation
3.9. Statistical Analysis
4. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mincuzzi, A.; Sanzani, S.M.; Palou, L.; Ragni, M.; Ippolito, A. Postharvest Rot of Pomegranate Fruit in Southern Italy: Characterization of the Main Pathogens. J. Fungi 2022, 8, 475. [Google Scholar] [CrossRef] [PubMed]
- Manzoor, S.; Fayaz, U.; Dar, A.H.; Dash, K.K.; Shams, R.; Bashir, I.; Pandey, V.K.; Abdi, G. Sustainable development goals through reducing food loss and food waste: A comprehensive review. Future Foods 2024, 9, 100362. [Google Scholar] [CrossRef]
- UN. Transforming Our World: The 2030 Agenda for Sustainable Development. Resolution Adopted by the General Assembly on 25 September 2015; United Nations: New York, NY, USA, 2015; pp. 1–13. [Google Scholar]
- Sánchez-Torres, P. Molecular Mechanisms Underlying Fungicide Resistance in Citrus Postharvest Green Mold. J. Fungi 2021, 7, 783. [Google Scholar] [CrossRef]
- Davies, C.R.; Wohlgemuth, F.; Young, T.; Violet, J.; Dickinson, M.; Sanders, J.W.; Vallieres, C.; Avery, S.V. Evolving challenges and strategies for fungal control in the food supply chain. Fungal Biol. Rev. 2021, 36, 15–26. [Google Scholar] [CrossRef]
- Chen, Z.F.; Ying, G.G. Occurrence, fate and ecological risk of five typical azole fungicides as therapeutic and personal care products in the environment: A review. Environ. Int. 2015, 84, 142–153. [Google Scholar] [CrossRef]
- Vitiello, A.; Ferrara, F.; Boccellino, M.; Ponzo, A.; Cimmino, C.; Comberiati, E.; Zovi, A.; Clemente, S.; Sabbatucci, M. Antifungal Drug Resistance: An Emergent Health Threat. Biomedicines 2023, 11, 1063. [Google Scholar] [CrossRef]
- Pawłowska, A.; Stepczyńska, M. Natural Biocidal Compounds of Plant Origin as Biodegradable Materials Modifiers. J. Polym. Environ. 2022, 30, 1683–1708. [Google Scholar] [CrossRef] [PubMed]
- Faisal, M.; Fauzi Abubakar, Y.; Heriansyah, M.B.; Desvita, H. Application of edible coating based on durian seed starch and peel liquid smoke as natural preservatives for tomatoes (Solanum lycopersicum L.). S. Afr. J. Chem. Eng. 2025, 54, 79–88. [Google Scholar] [CrossRef]
- Wang, Z.; Wu, M.; Liao, Q.; Wang, Y.; Sui, Y.; Gong, C. Current status and future trends of eco-friendly management of postharvest fungal decays in tomato fruit. npj Sci. Food 2025, 9, 104. [Google Scholar] [CrossRef]
- Alvarez, M.V.; Pérez-Gago, M.B.; Taberner, V.; Settier-Ramírez, L.; Martínez-Blay, V.; Palou, L. Postharvest Application of Novel Bio-Based Antifungal Composite Edible Coatings to Reduce Sour Rot and Quality Losses of ‘Valencia’ Oranges. Coatings 2023, 13, 1412. [Google Scholar] [CrossRef]
- Matrose, N.A.; Obikeze, K.; Belay, Z.A.; Caleb, O.J. Plant extracts and other natural compounds as alternatives for post-harvest management of fruit fungal pathogens: A review. Food Biosci. 2021, 41, 100840. [Google Scholar] [CrossRef]
- Hussein, H.Y.; Hasan, A.H.; Hussein, A.J.; Ayoob, M.M.; Samad, M.K.; Hussen, N.H.; Hawaiz, F.E.; Shakya, S.; Muzaffar, S.; Jamalis, J. Novel pyrazoline-thiazole hybrids containing azo group as antibacterial agents: Design, synthesis, in vitro bioactivity, in silico molecular docking, ADME profile and DFT studies. Res. Chem. Intermed. 2024, 50, 4551–4578. [Google Scholar] [CrossRef]
- Moradinezhad, F.; Ranjbar, A. Efficacy of active and passive modified atmosphere packaging on quality preservation and storage life of pomegranate fruit and arils: A review. Adv. Hort. Sci. 2024, 38, 83–96. [Google Scholar] [CrossRef]
- Silva, L.M.A.; Andrade, R.C.G. Phenolic compounds and their antimicrobial activities. J. Appl. Pharm. Sci. 2017, 7, 1–7. [Google Scholar]
- Zhang, C.W.; Zhong, X.J.; Zhao, Y.Z.; Riaz Rajoka, M.S.; Hashmi, M.; Zhai, P.; Xun Song, X. Antifungal natural products and their derivatives: A review of their activity and mechanism of actions. Pharmacol. Res. Mod. Chin. Med. 2023, 7, 100262. [Google Scholar] [CrossRef]
- Li, Z.J.; Liu, M.; Dawuti, G.; Dou, Q.; Ma, Y.; Liu, H.G.; Aibai, S. Antifungal Activity of Gallic Acid In Vitro and In Vivo. Phytother Res. 2017, 31, 1039–1045. [Google Scholar] [CrossRef] [PubMed]
- Barral, B.; Chillet, M.; Minier, J.; Léchaudel, M.; Schorr-Galindo, S. Evaluating the response to Fusarium ananatum inoculation and antifungal activity of phenolic acids in pineapple. Fungal Biol. 2017, 121, 1045–1053. [Google Scholar] [CrossRef]
- Morales, J.; Mendoza, L.; Cotoras, M. Alteration of oxidative phosphorylation as a possible mechanism of the antifungal action of p-coumaric acid against Botrytis cinerea. J. Appl. Microbiol. 2017, 123, 969–976. [Google Scholar] [CrossRef]
- Yuan, S.; Li, W.; Li, Q.; Wang, L.; Cao, J.; Jiang, W. Defense Responses, Induced by p-Coumaric Acid and Methyl p-Coumarate, of Jujube (Ziziphus jujuba Mill.) Fruit against Black Spot Rot Caused by Alternaria alternata. J. Agric. Food Chem. 2019, 67, 2801–2810. [Google Scholar] [CrossRef]
- Alfarrayeh, I.; Pollák, E.; Czéh, Á.; Vida, A.; Das, S.; Papp, G. Antifungal and Anti-Biofilm Effects of Caffeic Acid Phenethyl Ester on Different Candida Species. Antibiotics 2021, 10, 1359. [Google Scholar] [CrossRef]
- Zduńska, K.; Dana, A.; Kolodziejczak, A.; Rotsztejn, H. Antioxidant Properties of Ferulic Acid and Its Possible Application. Skin Pharmacol Physiol. 2018, 31, 332–336. [Google Scholar] [CrossRef]
- Lončar, M.; Gašo-Sokač, D.; Molnar, M. Coumarin Derivatives as Antifungal Agents—A Review. Czech J. Food Sci. 2023, 41, 79–91. [Google Scholar] [CrossRef]
- Ji, Q.; Ge, Z.; Ge, Z.; Chen, K.; Wu, H.; Liu, X.; Huang, Y.; Yuan, L.; Yang, X.; Liao, F. Synthesis and biological evaluation of novel phosphoramidate derivatives of coumarin as chitin synthase inhibitors and antifungal agents. Eur. J. Med. Chem. 2016, 108, 166–176. [Google Scholar] [CrossRef]
- De Araújo, R.S.A.; Guerra, F.Q.S.; De Oliviera Lima, E.; De Simone, C.A.; Tavares, J.F.; Scotti, L.; Scotti, M.T.; De Aquino, T.M.; De Moura, R.O.; Mendonça, F.J.B.; et al. Synthesis, Structure-Activity Relationships (SAR) and in Silico Studies of Coumarin Derivatives with Antifungal Activity. Int. J. Mol. Sci. 2013, 14, 1293–1309. [Google Scholar] [CrossRef]
- Al Aboody, M.S.; Mickymaray, S. Anti-Fungal Efficacy and Mechanisms of Flavonoids. Antibiotics 2020, 9, 45. [Google Scholar] [CrossRef]
- Henry, E.J.; Bird, S.J.; Gowland, P.; Collins, M.; Cassella, J.P. Ferrocenyl chalcone derivatives as possible antimicrobial agents. J. Antibiot. 2020, 73, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.; Billing, D.; Connolly, P.; Smith, W.; Cooney, J.; Burdon, J. Maturity, storage and ripening effects on anti-fungal compounds in the skin of ‘Hass’ avocado fruit. Postharvest Biol. Technol. 2018, 146, 43–50. [Google Scholar] [CrossRef]
- Duarte, S.; Magro, A.; Tomás, J.; Hilário, C.; Ferreira, R.B.; Carvalho, M.O. Antifungal Activity of Benzoquinones Produced by Tribolium castaneum in Maize-Associated Fungi. Insects 2022, 13, 868. [Google Scholar] [CrossRef]
- Navarro-Tovar, G.; Vega-Rodríguez, S.; Leyva, E.; Loredo-Carrillo, S.; de Loera, D.; López-López, L.I. The Relevance and Insights on 1,4-Naphthoquinones as Antimicrobial and Antitumoral Molecules: A Systematic Review. Pharmaceuticals 2023, 16, 496. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Chang, C.W.T.; Zhang, Q. 1,4-Naphthoquinone Analogs and Their Application as Antibacterial Agents. ChemistrySelect 2022, 7, e202203330. [Google Scholar] [CrossRef]
- Futuro, D.O.; Ferreira, P.G.; Nicoletti, C.D.; Borba-Santos, L.P.; Silva, F.C.D.; Rozental, S.; Ferreira, V.F. The Antifungal Activity of Naphthoquinones: An Integrative Review. An. Acad. Bras. Cienc. 2018, 90, 1187–1214. [Google Scholar] [CrossRef]
- Barilli, E.; Agudo, F.J.; Masi, M.; Nocera, P.; Evidente, A.; Rubiales, D. Anthraquinones and their analogues as potential biocontrol agents of rust and powdery mildew diseases of field crops. Pest. Manag. Sci. 2022, 78, 3489–3497. [Google Scholar] [CrossRef]
- Raghuveer, D.; Pai, V.V.; Murali, T.S.; Nayak, R. Exploring Anthraquinones as Antibacterial and Antifungal Agents. ChemistrySelect 2023, 8, e202204537. [Google Scholar] [CrossRef]
- Angulo-Elizari, E.; Henriquez-Figuereo, A.; Morán-Serradilla, C.; Plano, D.; Sanmartín, C. Unlocking the potential of 1,4-naphthoquinones: A comprehensive review of their anticancer properties. Eur. J. Med. Chem. 2024, 268, 116249. [Google Scholar] [CrossRef]
- Rahman, M.M.; Islam, M.R.; Akash, S.; Shohag, S.; Ahmed, L.; Supti, F.A.; Rauf, A.; Aljohani, A.S.M.; Al Abdulmonem, W.; Khalil, A.A.; et al. Naphthoquinones and derivatives as potential anticancer agents: An updated review. Chem. Biol. Interact. 2022, 368, 110198. [Google Scholar] [CrossRef] [PubMed]
- Rani, R.; Sethi, K.; Gupta, S.; Varma, R.S.; Kumar, R. Mechanism of Action and Implication of Naphthoquinone as Potent Anti-trypanosomal Drugs. Curr. Top. Med. Chem. 2022, 22, 2087–2105. [Google Scholar] [CrossRef] [PubMed]
- Oda, Y.; Nakashima, S.; Kondo, E.; Nakamura, S.; Yano, M.; Kubota, C.; Masumoto, Y.; Hirao, M.; Ogawa, Y.; Matsuda, H. Comparison of lawsone contents among Lawsonia inermis plant parts and neurite outgrowth accelerators from branches. J. Nat. Med. 2018, 72, 890–896. [Google Scholar] [CrossRef]
- Batiha, G.E.; Teibo, J.O.; Shaheen, H.M.; Babalola, B.A.; Teibo, T.K.A.; Al-Kuraishy, H.M.; Al-Garbeeb, A.I.; Alexiou, A.; Papadakis, M. Therapeutic potential of Lawsonia inermis Linn: A comprehensive overview. Naunyn Schmiedebergs Arch. Pharmacol. 2024, 397, 3525–3540. [Google Scholar] [CrossRef]
- Muheyuddeen, G.; Divy, S.R.; Gautam, S.K.; Gupta, S.K. Lawsonia inermis L. Phytopharmacological Characteristics and Recent Advancement. Res. J. Pharmacogn. Phytochem. 2023, 15, 11–13. [Google Scholar] [CrossRef]
- Li, B.; Cui, C.; Zhang, C.; Lui, J.; Hao, F.; Han, L.; Bai, C. Traditional Applications, Ethnopharmacology, and Phytochemistry of Walnut Green Husk (Juglans regia L.): A Review. Nat. Prod. Commun. 2024, 19, 1934578X241262156. [Google Scholar] [CrossRef]
- Grevenstuk, T.; Gonçalves, S.; Almeida, S.; Coelho, N.; Quintas, C.; Gaspar, M.N.; Romano, A. Evaluation of the antioxidant and antimicrobial properties of in vitro cultured Drosera intermedia extracts. Nat. Prod. Commun. 2009, 4, 1063–1068. [Google Scholar] [CrossRef] [PubMed]
- Parachur, V.A.; Ravichandran, S. Patent on the Ethyl Acetate Extracts or Active Ingredients Isolated from Ethyl Acetate Extract of Lawsonia inermis Against Fungal Infections. 2017. Available online: https://patentimages.storage.googleapis.com/a8/1c/9f/ade52b2d22f1c4/US20170042955A1.pdf (accessed on 21 July 2025).
- Bendre, N.; Gonjari, G. Antiangiogenic Potential of Endophytic Fungi Alternaria alternata Isolated From Lawsonia inermis Linn. Biosci. Biotechnol. Res. Commun. 2019, 12, 820–828. [Google Scholar] [CrossRef]
- Xi, M.; Hou, Y.; Cai, Y.; Shen, H.; Ao, J.; Li, M.; Wang, J.; Luo, A. Antioxidant and antimicrobial characteristics of ethyl acetate polar fractions from walnut green husk. J. Food Sci. 2023, 88, 1060–1074. [Google Scholar] [CrossRef]
- Abedi, P.; Yaralizadeh, M.; Fatahinia, M.; Namjoyan, F.; Nezamivand-Chegini, S.; Yaralizadeh, M. Comparison of the effects of Juglans nigra green husk and clotrimazole on Candida albicans in rats. Jundishapur J. Microbiol. 2018, 11, 7–9. [Google Scholar] [CrossRef]
- Grevenstuk, T.; Gonçalves, S.; Domingos, T.; Quintas, C.; van der Hooft, J.J.; Vervoort, J.; Romano, A. Inhibitory activity of plumbagin produced by Drosera intermedia on food spoilage fungi. J. Sci. Food Agric. 2012, 92, 1638–1642. [Google Scholar] [CrossRef]
- Ahmad, M.; Tahir, M.; Hong, Z.; Zia, M.A.; Rafeeq, H.; Ahmad, M.S.; Rehman, S.U.; Sun, J. Plant and marine-derived natural products: Sustainable pathways for future drug discovery and therapeutic development. Front. Pharmacol. 2025, 6, 1497668. [Google Scholar] [CrossRef]
- Anand, U.; Jacobo-Herrera, N.; Altemimi, A.; Lakhssassi, N. A Comprehensive Review on Medicinal Plants as Antimicrobial Therapeutics: Potential Avenues of Biocompatible Drug Discovery. Metabolites 2019, 9, 258. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Yu, L.; Bi, C.; Huang, L.; Su, B.; Nie, J.; Dou, Z.; Yang, S.; Li, Y. A new paradigm for drug discovery in the treatment of complex diseases: Drug discovery and optimization. Chin. Med. 2025, 20, 40. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information (2025a). PubChem Compound Summary for CID 10205, Plumbagin. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Plumbagin (accessed on 22 July 2025).
- National Center for Biotechnology Information (2025b). PubChem Compound Summary for CID 26905, 7-Methyljuglone. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/7-Methyljuglone (accessed on 22 July 2025).
- National Center for Biotechnology Information (2025c). PubChem Compound Summary for CID 5281672, Myricetin. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Myricetin (accessed on 22 July 2025).
- National Center for Biotechnology Information (2025d). PubChem Compound Summary for CID 5280343, Quercetin. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Quercetin (accessed on 22 July 2025).
- National Center for Biotechnology Information (2025e). PubChem Compound Summary for CID 5280863, Kaempferol. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Kaempferol (accessed on 22 July 2025).
- Baranyai, B.; Bäcker, C.; Reich, C. The Production of 7-Methyljuglone, Plumbagin and Quercetin in Wild and Cultivated Drosera rotundifolia and Drosera intermedia. Mires Peat 2016, 18, 19. [Google Scholar] [CrossRef]
- Braunberger, C.; Zehl, M.; Conrad, J.; Fischer, S.; Adhami, H.R.; Beifuss, U.; Krenn, L. LC-NMR, NMR, and LC-MS identification and LC-DAD quantification of flavonoids and ellagic acid derivatives in Drosera peltata. J. Chromatogr. B Analyt Technol. Biomed. Life Sci. 2013, 932, 111–116. [Google Scholar] [CrossRef]
- Schlauer, J.; Fleischmann, A.; Hartmeyer, S.R.H.; Hartmeyer, I.; Rischer, H. Distribution of Acetogenic Naphthoquinones in Droseraceae and Their Chemotaxonomic Utility. Biology 2024, 13, 97. [Google Scholar] [CrossRef]
- Wójciak, M.; Feldo, M.; Stolarczyk, P.; Płachno, B.J. Carnivorous Plants from Nepenthaceae and Droseraceae as a Source of Secondary Metabolites. Molecules 2023, 28, 2155. [Google Scholar] [CrossRef]
- Grevenstuk, T.; van der Hooft, J.J.J.; Vervoort, J.; Gonçalves, S.; Romano, A. Identification of antimicrobial agents from Drosera intermedia using HPLC-MS/HPLC-SPE-NMR. Planta Med. 2009, 75, PA-34. [Google Scholar] [CrossRef]
- Liyanapathiranage, A.; Dassanayake, R.S.; Gamage, A.; Karri, R.R.; Manamperi, A.; Evon, P.; Jayakodi, Y.; Madhujith, T.; Merah, O. Recent Developments in Edible Films and Coatings for Fruits and Vegetables. Coatings 2023, 13, 1177. [Google Scholar] [CrossRef]
- Piluharto, B.; Indarti, D.; Rohman, L.; Lelono, A.; Larasati, K.A.; Wedi Pratama, A.W. Stability Studies of Neem Seed Oil-in-Water Emulsion Formulation. Case Stud. Chem. Environ. Eng. 2025, 11, 101201. [Google Scholar] [CrossRef]
- Perez-Vazquez, A.; Barciela, P.; Carpena, M.; Prieto, M.A. Edible Coatings as a Natural Packaging System to Improve Fruit and Vegetable Shelf Life and Quality. Foods 2023, 12, 3570. [Google Scholar] [CrossRef]
- Krstonošić, V.; Dokić, L.; Nikolić, I.; Milanović, M. Influence of xanthan gum on oil-in-water emulsion characteristics stabilized by OSA starch. Food Hydrocoll. 2015, 45, 9–17. [Google Scholar] [CrossRef]
- Baptista, S.; Baptista, F.; Freitas, F. Development of Emulsions Containing L-Ascorbic Acid and α-Tocopherol Based on the Polysaccharide FucoPol: Stability Evaluation and Rheological and Texture Assessment. Cosmetics 2023, 10, 56. [Google Scholar] [CrossRef]
- Yazdanfar, N.; Manafi, L.; Ebrahiminejad, B.; Mazaheri, Y.; Sadighara, P.; Basaran, B.; Mohamadi, S. Evaluation of Sodium Benzoate and Potassium Sorbate Preservative Concentrations in Different Sauce Samples in Urmia, Iran. J. Food Prot. 2023, 86, 100118. [Google Scholar] [CrossRef]
- Benamar, K.; Lahlali, R.; Ezzouggari, R.; El Ouassete, M.; Dehbi, I.; Khadiri, M.; Radi, M.; Ait Haddou, L.; Ibnsouda Koraichi, S.; Benamar, S.; et al. Antifungal Efficacy of Ethanolic Extracts from Four Medicinal Plants Against Major Postharvest Fungal Pathogens of Apple Fruit. Agronomy 2025, 15, 1577. [Google Scholar] [CrossRef]
- Khadiri, M.; Boubaker, H.; Askarne, L.; Farhaoui, A.; Taoussi, M.; Haddou, L.A.; Cherrate, M.; Ouchari, W.; Zerrouk, M.H.; Sobeh, M.; et al. Exploration of the antifungal potential of aqueous extracts from two agave species against major postharvest diseases in apples. Postharvest Biol. Technol. 2024, 214, 112992. [Google Scholar] [CrossRef]
- Nawaz, M.; Pan, J.; Liu, H.; Umer, M.J.; Liu, J.; Yang, W.; Lv, Z.; Zhang, Q.; Jiao, Z. Integrated evaluation of antifungal activity of pomegranate peel polyphenols against a diverse range of postharvest fruit pathogens. Bioresour. Bioprocess. 2025, 12, 34. [Google Scholar] [CrossRef]
- Zhang, J.; Ma, S.; Du, S.; Chen, S.; Sun, H. Antifungal activity of thymol and carvacrol against postharvest pathogens Botrytis cinerea. J. Food Sci. Technol. 2019, 56, 2611–2620. [Google Scholar] [CrossRef]
- Mehani, M.; Segni, L.; Terzi, V.; Morcia, C.; Ghizzoni, R.; Goudjil, M.; Bencheikh, S. Antibacterial, antifungal activity and chemical composition study of essential oil of Mentha piperita from the south Algerian. Der Pharma Chem. 2015, 7, 382–387. [Google Scholar]
- Prakash, B.; Kujur, A.; Yadav, A.; Kumar, A.; Singh, P.P.; Dubey, N. Nanoencapsulation: An efficient technology to boost the antimicrobial potential of plant essential oils in food system. Food Control 2018, 89, 1–11. [Google Scholar] [CrossRef]
- Motallebi, P.; Negahban, M. Neem (Azadirachta indica) Seed Extract Formulation for Managing Anthracnose and Gray Mold Diseases in Strawberry. S. Afr. J. Bot. 2024, 169, 66–71. [Google Scholar] [CrossRef]
- McDaniel, A.; Tonyali, B.; Yucel, U.; Trinetta, V. Formulation and development of lipid nanoparticle antifungal packaging films to control postharvest disease. J. Agric. Food Res. 2019, 1, 100013. [Google Scholar] [CrossRef]
- Vakili-Ghartavol, M.; Arouiee, H. Harnessing plant-derived antifungal agents and nanoencapsulation for postharvest pathogen management: A comprehensive review. J. Agric. Food Res. 2025, 24, 102280. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, X.; Sun, M.; Wang, L.; Zou, Y.; Fu, L.; Han, C.; Li, A.; Li, L.; Zhu, C. Impact of vanillin on postharvest disease control of apple. Front Microbiol. 2022, 13, 979737. [Google Scholar] [CrossRef]
- Casanova, E.; García-Mina, J.M.; Calvo, M.I. Antioxidant and Antifungal Activity of Verbena officinalis L. Leaves. Plant Foods Hum. Nutr. 2008, 63, 93–97. [Google Scholar] [CrossRef]
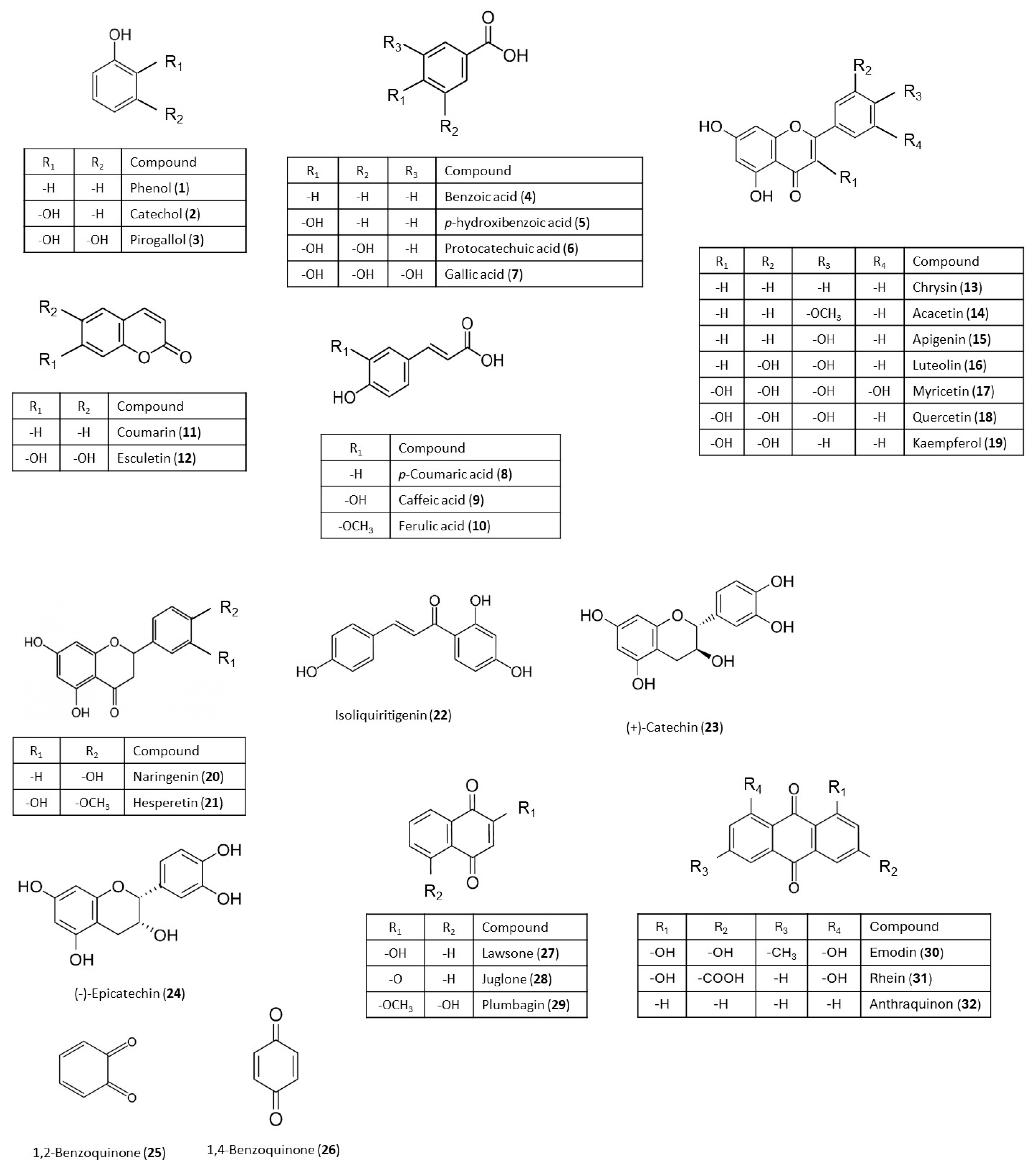
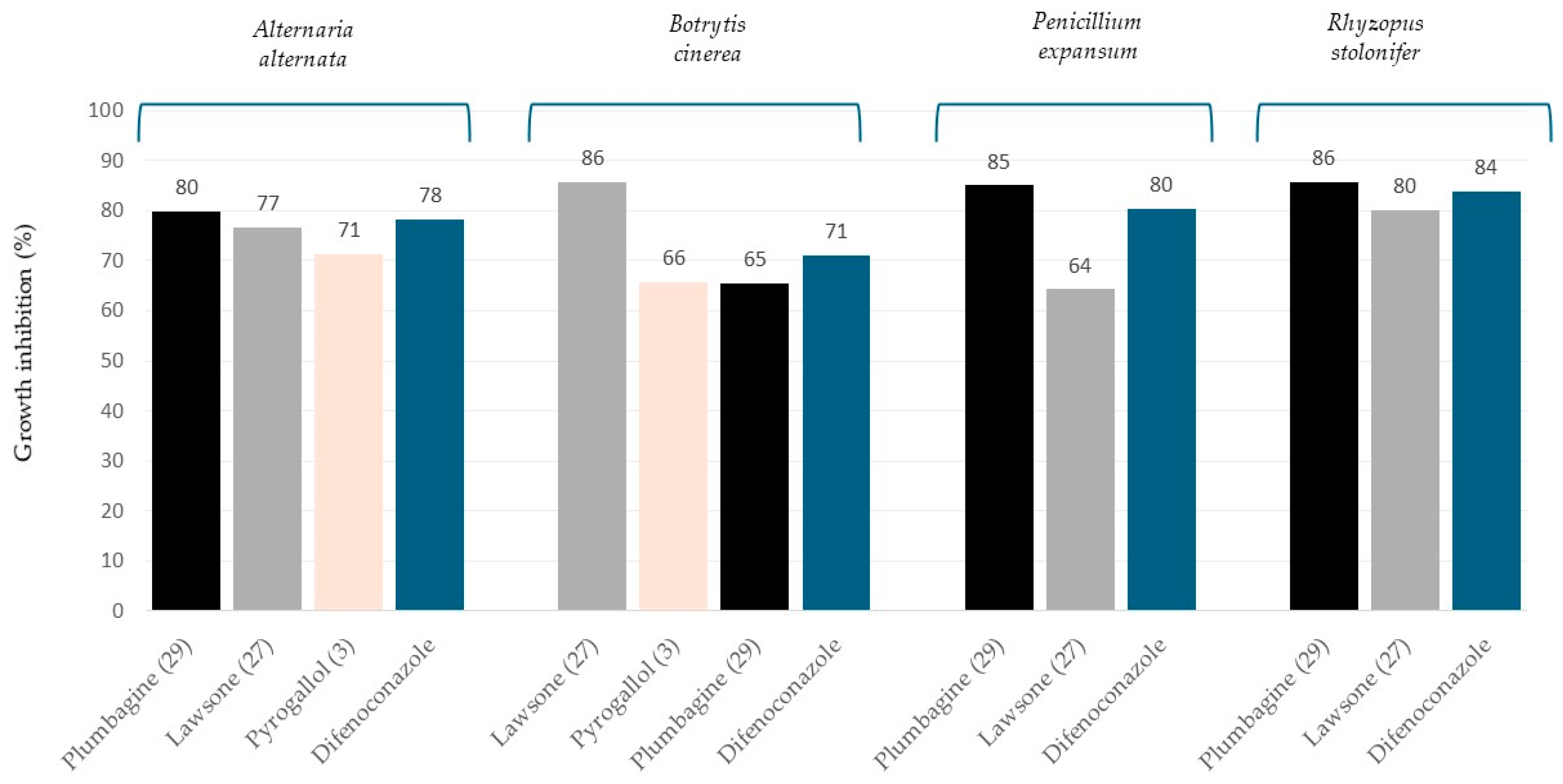
| Growth Inhibition (%) | |||||
|---|---|---|---|---|---|
| Alternaria alternata | Botrytis cinerea | Penicillium expansum | Rhizopus stolonifer | ||
| Phenols | Phenol (1) | - | 18 ± 1 | 4.5 ± 0.1 | - |
| Catechol (2) | - | 29 ± 1 | 19 ± 2 | 9.0 ± 0.7 | |
| Pyrogallol (3) | 71 ± 4 | 66 ± 2 | 3.0 ± 0.1 | - | |
| Hydroxyphenolic acids | Benzoic acid (4) | 0.6 ± 0.1 | - | - | - |
| p-hydroxybenzoic acid (5) | - | 14 ± 3 | - | - | |
| Protocatechuic acid (6) | - | 28 ± 1 | 41 ± 2 | 10 ± 1 | |
| Gallic acid (7) | 12.1 ± 0.4 | 57 ± 3 | 0.4 ± 0.1 | 62 ± 5 | |
| Hydroxycinnamic acids | p-coumaric acid (8) | 7.3 ± 0.7 | 2.0 ± 0.1 | - | - |
| Caffeic acid (9) | 25 ± 3 | 5.2 ± 0.1 | 3.0 ± 0.3 | - | |
| Ferulic acid (10) | 21 ± 2 | 5.5 ± 0.2 | - | - | |
| Coumarins | Coumarin (11) | - | - | - | - |
| Esculetin (12) | - | 7 ± 1 | 10 ± 2 | - | |
| Flavones | Chrysin (13) | - | - | - | - |
| Acacetin (14) | - | 0.9 ± 0.1 | - | - | |
| Apigenin (15) | - | 28 ± 2 | - | 19 ± 2 | |
| Luteolin (16) | - | 28 ± 3 | - | 19 ± 3 | |
| Flavonols | Myricetin (17) | - | - | - | 33 ± 2 |
| Quercetin (18) | - | - | - | 16 ± 1 | |
| Kaempferol (19) | - | - | - | - | |
| Flavanones | Naringenin (20) | - | 26 ± 2 | - | 14 ± 2 |
| Hesperetin (21) | - | 10 ± 1 | - | 29 ± 3 | |
| Chalcones | Isoliquiritigenin (22) | 39 ± 4 | 2.0 ± 0.1 | 25 ± 1 | 14 ± 2 |
| Catechins | Catechin (23) | - | - | - | 4.0 ± 0.4 |
| Epicatechin (24) | - | 7.9 ± 0.9 | - | 1.6 ± 0.1 | |
| Benzoquinones | 1,2-benzoquinone (25) | 13 ± 1 | - | 16 ± 1 | 19 ± 2 |
| 1,4-benzoquinone (26) | 25 ± 4 | 15.2 ± 0.6 | 28 ± 4 | 30 ± 2 | |
| Naphthoquinones | Lawsone (27) | 77 ± 5 | 86 ± 4 | 64 ± 1 | 80 ± 4 |
| Juglone (28) | 31 ± 2 | 0.15 ± 0.01 | 16.4 ± 0.8 | 50 ± 4 | |
| Plumbagin (29) | 80 ± 1 | 65 ± 1 | 85 ± 3 | 86 ± 5 | |
| Anthraquinones | Emodin (30) | 21 ± 2 | 49 ± 5 | - | - |
| Rhein (31) | 36 ± 5 | 47 ± 4 | 10 ± 1 | 2.8 ± 0.5 | |
| Anthraquinone (32) | 11.0 ± 0.2 | 11 ± 2 | - | - | |
| Difenoconazole | 78 ± 2 | 71 ± 2 | 80 ± 2 | 84 ± 2 | |
| Yield of Extraction (%) | Growth Inhibition (%) | |||||
|---|---|---|---|---|---|---|
| Plant | Extract | Alternaria alternata | Botrytis cinerea | Penicillium expansum | Rhizopus stolonifer | |
| L. inermis | Dichloromethane | 4.26 | - | - | - | - |
| Ethyl acetate | 8.51 | 71 ± 5 | 38 ± 4 | 95 ± 2 | 37 ± 2 | |
| Methanol | 20.00 | 29 ± 2 | 26 ± 1 | 31 ± 3 | 2.9 ± 0.1 | |
| J. regia | Dichloromethane | 4.66 | - | - | 10 ± 1 | 19 ± 1 |
| Ethyl acetate | 8.22 | 38 ± 2 | - | 47 ± 3 | 84 ± 3 | |
| Methanol | 13.44 | 19 ± 2 | - | 10 ± 1 | 18 ± 1 | |
| D. intermedia | Dichloromethane | 5.44 | 2.1 ± 0.7 | 1.6 ± 0.6 | 38 ± 4 | 20 ± 2 |
| Ethyl acetate | 10.09 | 87 ± 3 | 96 ± 3 | 64 ± 3 | 85 ± 4 | |
| Methanol | 12.60 | 21 ± 1 | 5 ± 1 | 22 ± 2 | 14.7 ± 0.9 | |
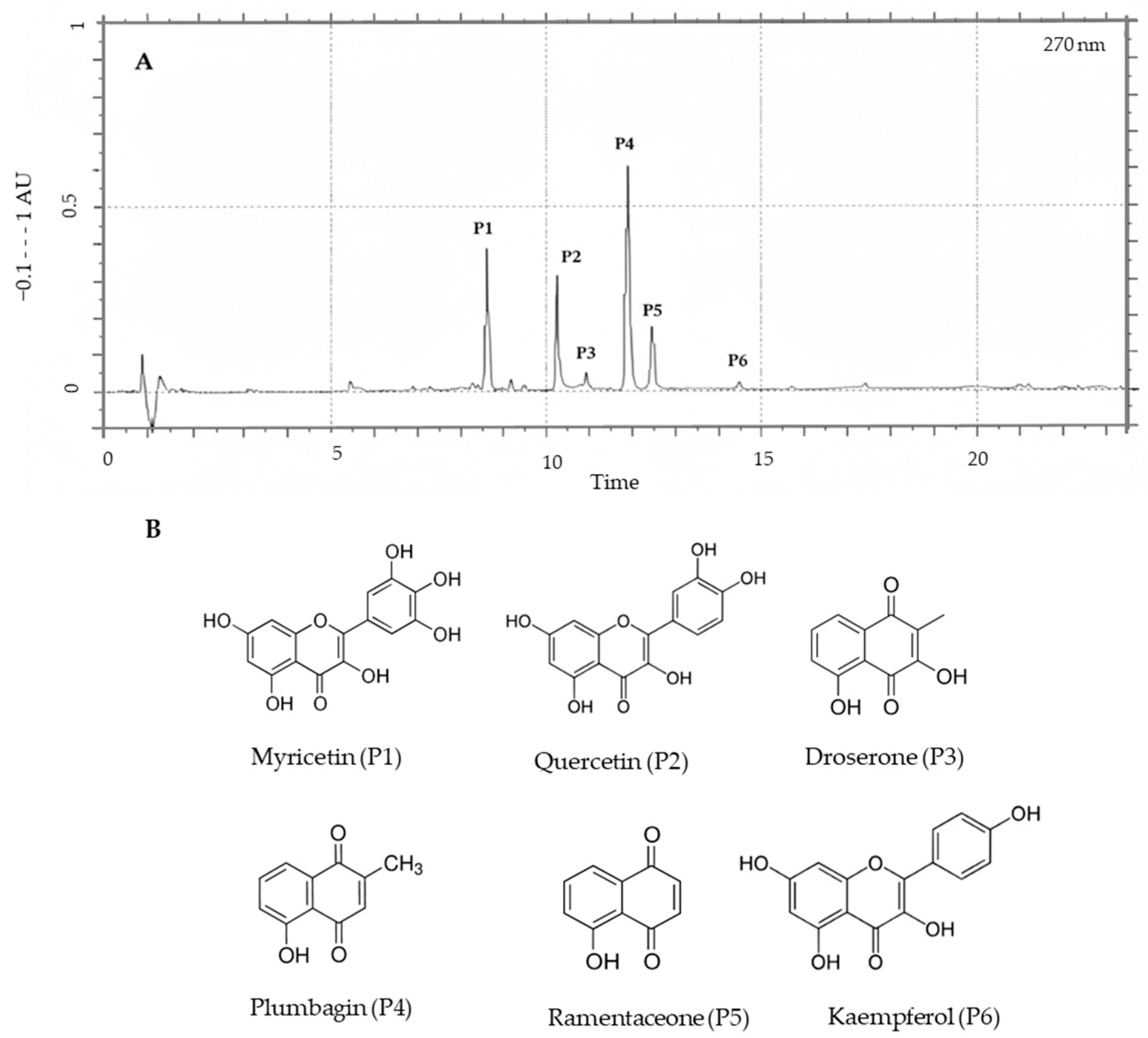
| Peak | Compound | tR (min.) | Chemical Formula | Abs. Max UV (nm) | Molecular Weight | Mass Spectrum, m/z (RA%) |
|---|---|---|---|---|---|---|
| P1 | Myricetin (17) | 8.87 | C15H10O8 | 254, 301sh, 374 | 318.2 | 319.4(100), 303.4(5), 287.0(30), 139.1(15), 121.2(10) |
| P2 | Quercetin (18) | 10.26 | C15H10O7 | 254, 301sh, 368 | 302.2 | 303.4(100), 246.9(30), 267.0(30), 193.1(10), 175.0(20) |
| P3 | Droserone * | 11.12 | C11H8O4 | 269, 422 | 204.2 | 205.0(100), 190.0(10), 187.0(30), 177.0(50), 172.0(10), 169.0(7), 162.0(22), 159.0(25) |
| P4 | Plumbagin (29) | 12.01 | C11H8O3 | 266, 414 | 188.2 | 189.1(100), 174.3(58), 161.5(7), 144.1 (32) |
| P5 | Ramentaceone (33) | 12.42 | C11H8O3 | 266, 415 | 188.2 | 189.0(100), 174.3(71), 161.3(12), 144.1 (25) |
| P6 | Kaempferol (19) | 14.50 | C15H10O6 | 265, 301sh, 365 | 286.2 | 287.0(15), 251.4(100), 211.4(50), 165.2(30) |
| Compound | mg/10 g Extract | g/100 g Dry Plant (%) | |
|---|---|---|---|
| Flavonoids | Myricetin (17) | 692 ± 27 | 0.69 ± 0.03 |
| Quercetin (18) | 618 ± 22 | 0.62 ± 0.02 | |
| Kaempferol (19) | 95 ± 2 | 0.09 ± 0.02 | |
| Σ Flavonoids | 1404 ± 50 | 1.40 ± 0.05 | |
| Naphthoquinones | Droserone * | 362 ± 5 | 0.36 ± 0.05 |
| Plumbagin (29) | 1456 ± 112 | 1.5 ± 0.1 | |
| Ramentaceone (33) | 929 ± 20 | 0.94 ± 0.02 | |
| Σ Naphthoquinones | 2748 ± 137 | 2.8 ± 0.1 |
| Component | Quantity (% w/w) | Function |
|---|---|---|
| Dry extract of D. intermedia (10:1, ethyl acetate) | 5.0% | Active Ingredient |
| Polysorbate 80 (Tween® 80) | 15.0% | Emulsifier/Surfactant |
| Propylene glycol | 12.0% | Co-solvent/Humectant |
| Glycerol | 5.0% | Humectant/Stabilizer |
| Xanthan gum | 0.2% | Suspending agent/Thickener |
| Citric acid | 0.2% | pH adjuster |
| Ascorbic acid | 0.1% | Antioxidant/Stabilizer |
| Sodium benzoate | 0.1% | Preservative |
| Purified water | Q.s. 100% | Diluent |
| pH | Droplet Size (µm) | PDI | Z Potential (mV) | Viscosity (cP) | |
|---|---|---|---|---|---|
| Day 0 | 5.0 ± 0.2 | 1.03 ± 0.01 | 0.17 ± 0.05 | −41 ± 1 | 3126 ± 38 |
| Day 3 | 5.0 ± 0.2 | 1.02 ± 0.03 | 0.17 ± 0.04 | −41 ± 2 | 3134 ± 29 |
| Day 5 | 5.0 ± 0.1 | 1.02 ± 0.04 | 0.19 ± 0.03 | −400 ± 0.7 | 3135 ± 34 |
| Day 7 | 5.0 ± 0.1 | 1.02 ± 0.03 | 0.19 ± 0.02 | −40.0 ± 0.7 | 3140 ± 30 |
| Dose (ppm) | Growth Inhibition (%) | |||||
|---|---|---|---|---|---|---|
| Alternaria alternata | Botrytis cinerea | Penicillium expansum | Rhizopus. stolonifer | |||
| Formulation | 250 | 0.09 ± 0.01 a | 0.15 ± 0.01 a | 1.1 ± 0.2 a | 4 ± 1 a | |
| 500 | 41 ± 2 b | 25 ± 1 b | 46 ± 1 b | 50 ± 1 b | ||
| 1000 | 80 ± 2 c | 75 ± 2 c | 65.1 ± 0.5 c | 85 ± 2 c | ||
| 2000 | 81 ± 2 c | 92 ± 2 d | 86 ± 3 c | 88 ± 2 c | ||
| Positive control | Neozil 50EC® | 1000 | 82.0 ± 0.7 c | 79 ± 2 c | 73 ± 1 c | 85 ± 2 c |
| Difenoconazole | 1000 | 78 ± 2 c | 71 ± 2 c | 80 ± 2 c | 84 ± 2 c | |
| Alternaria alternata | Botrytis cinerea | Penicillium expansum | Rhizopus. stolonifer | |
|---|---|---|---|---|
| Formulation | 595 ± 13 (560−766) | 645 ± 14 (612−748) | 584 ± 15 (455−624) | 500 ± 7 (458−618) |
| Difenoconazole | 503 ± 10 (482−573) | 435 ± 9 (418−452) | 471 ± 9 (453−519) | 582 ± 11 (561−603) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calvo, M.I. The Design of a Plant-Based Biopesticide Formulation with Extracts and Derivatives Containing Naphthoquinones. Plants 2025, 14, 3427. https://doi.org/10.3390/plants14223427
Calvo MI. The Design of a Plant-Based Biopesticide Formulation with Extracts and Derivatives Containing Naphthoquinones. Plants. 2025; 14(22):3427. https://doi.org/10.3390/plants14223427
Chicago/Turabian StyleCalvo, María Isabel. 2025. "The Design of a Plant-Based Biopesticide Formulation with Extracts and Derivatives Containing Naphthoquinones" Plants 14, no. 22: 3427. https://doi.org/10.3390/plants14223427
APA StyleCalvo, M. I. (2025). The Design of a Plant-Based Biopesticide Formulation with Extracts and Derivatives Containing Naphthoquinones. Plants, 14(22), 3427. https://doi.org/10.3390/plants14223427







