Differential Responses of Rice Genotypes to Nitrogen Supply: Impacts on Nitrogen Metabolism and Chlorophyll Fluorescence Kinetics
Abstract
1. Introduction
2. Results
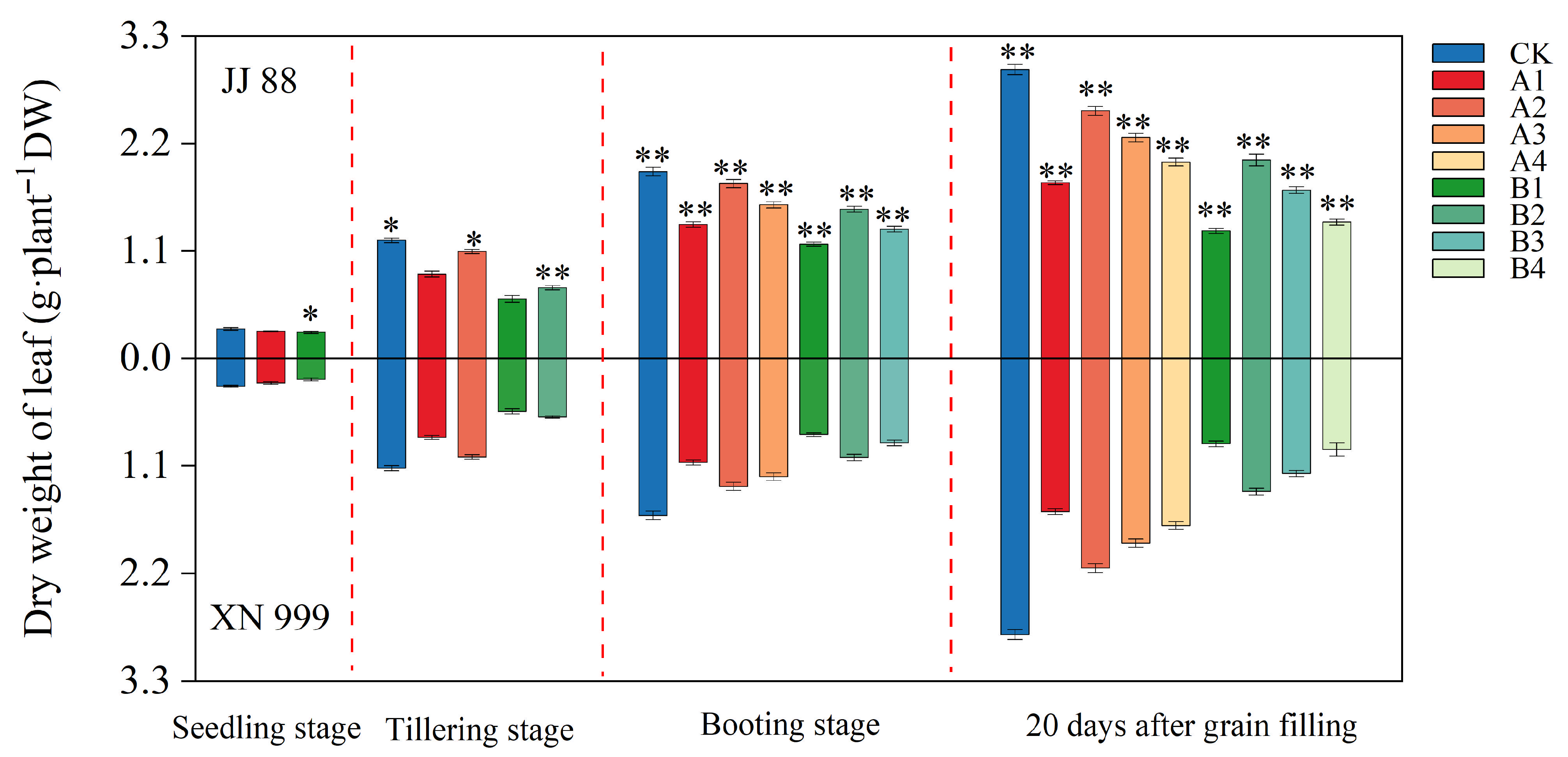
2.1. Dry Matter Accumulation
2.2. Nitrogenous Compounds
2.3. Soluble Sugar Content, Amino Acid and Proline Content
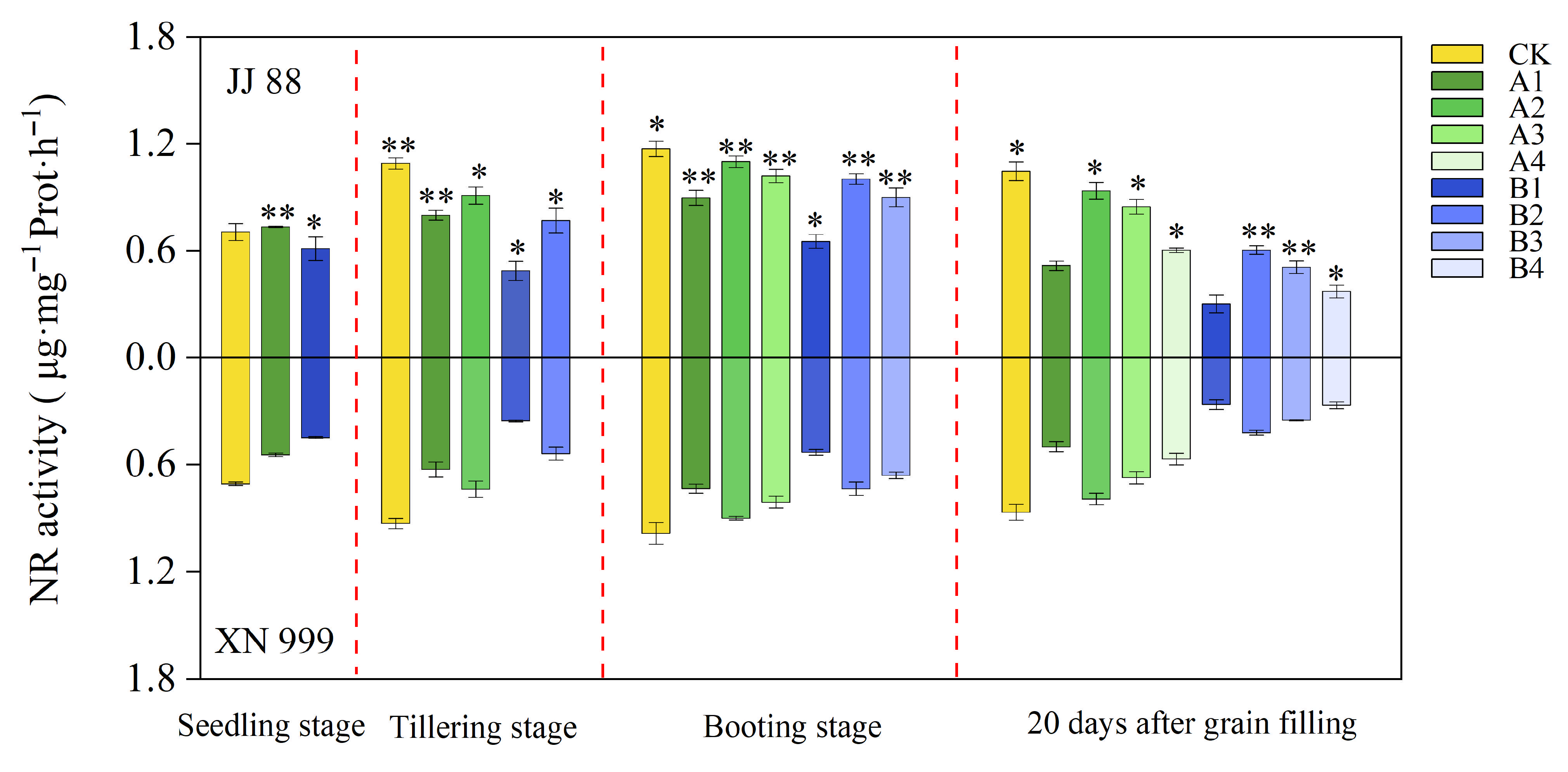
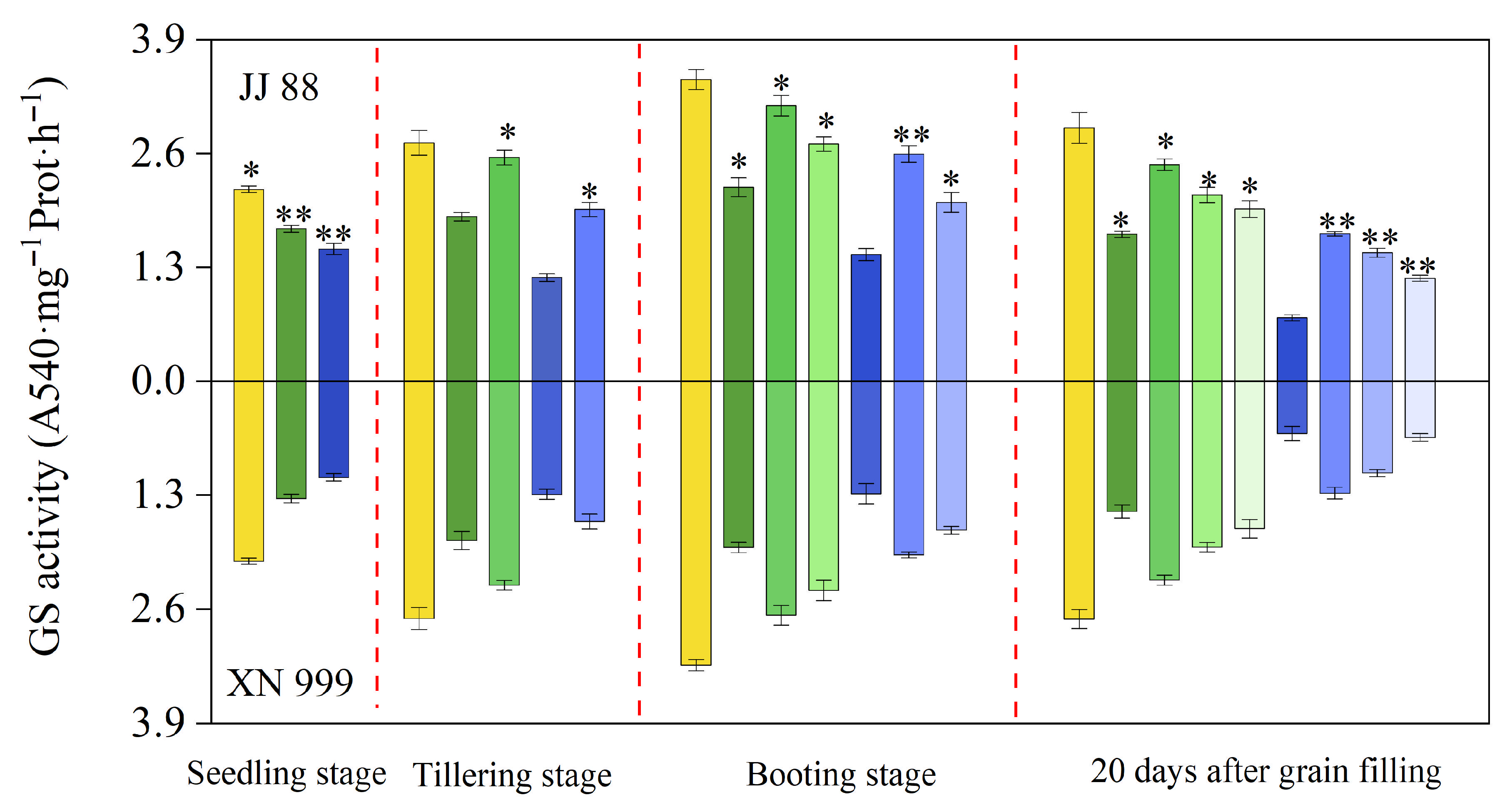
2.4. NR and Gs Activity
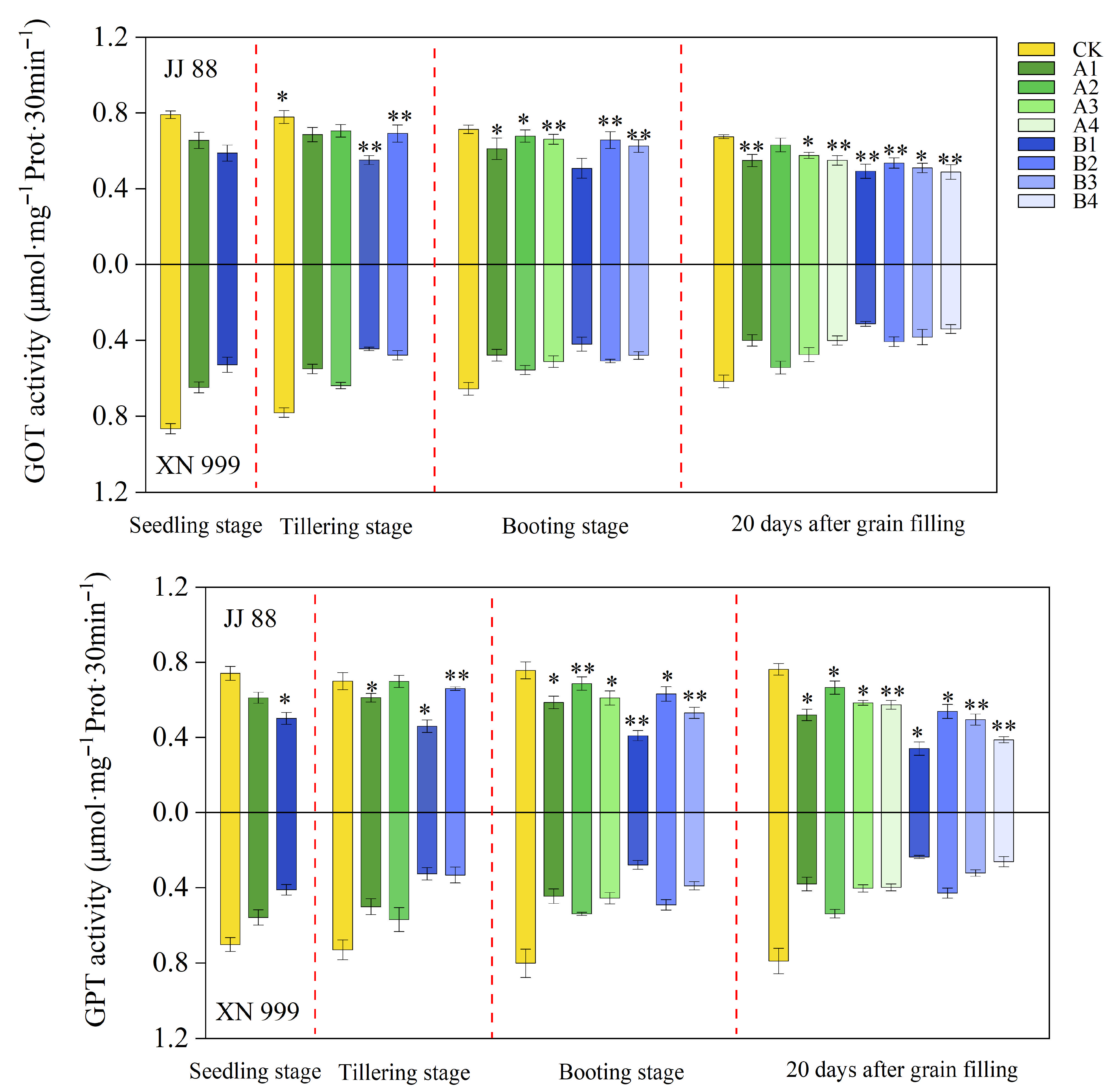
2.5. GOT and GPT Activity
2.6. Chlorophyll Fluorescence Related Parameters
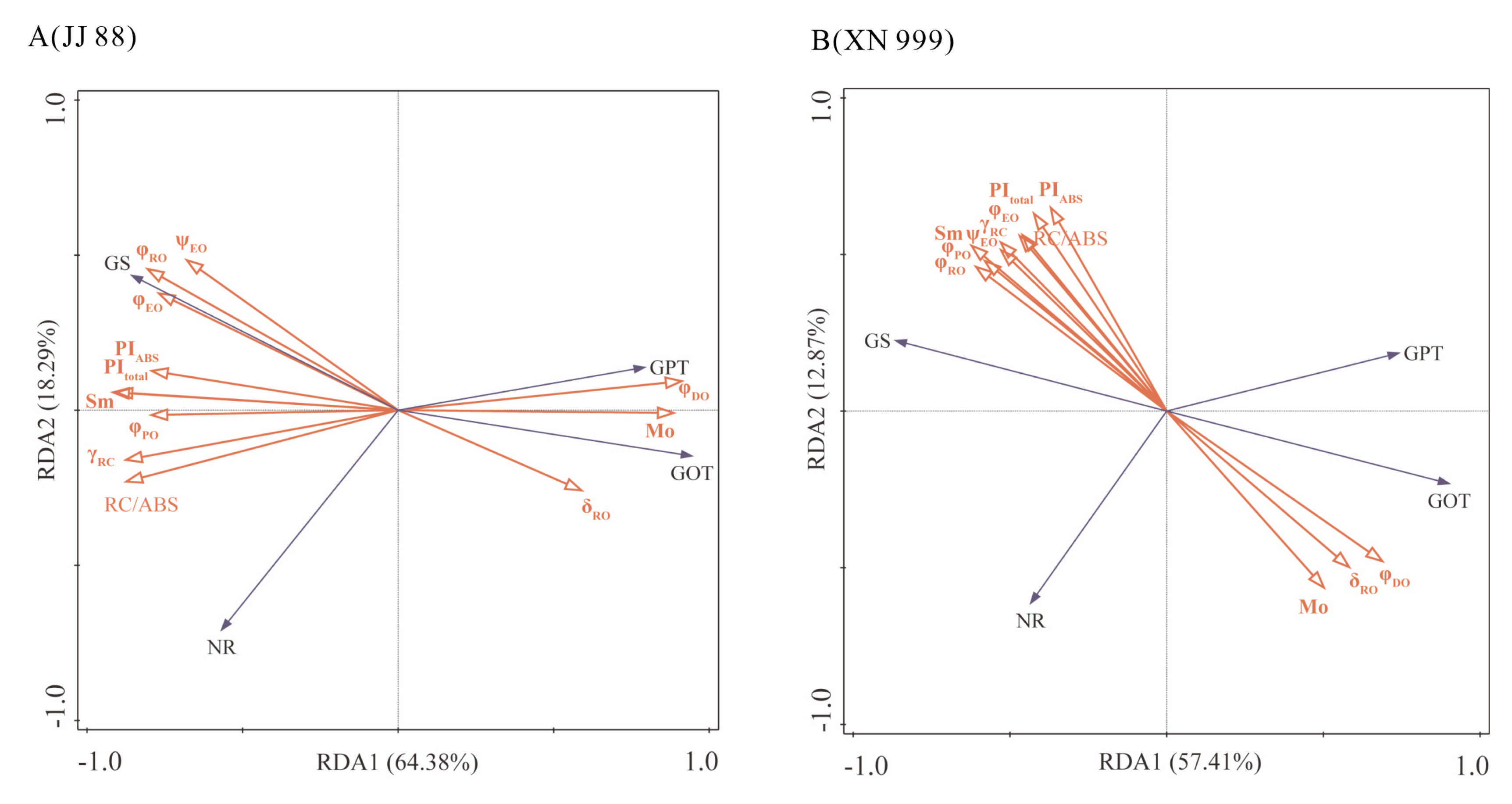
2.7. Redundancy Analysis
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Experimental Design
4.3. Measurement Indicators and Methods
4.3.1. Chlorophyll a Fluorescence Induction Kinetics (OJIP) Analysis
4.3.2. Biochemistry
4.3.3. Enzyme Activity Assay
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ogawa, T.; Oikawa, S.; Hirose, T. Nitrogen-utilization efficiency in rice: An analysis at leaf, shoot, and whole-plant level. Plant Soil 2016, 404, 321–344. [Google Scholar] [CrossRef]
- Lee, S. Recent advances on nitrogen use efficiency in rice. Agronomy 2021, 11, 753. [Google Scholar] [CrossRef]
- FAO. Rice Market Monitor; FAO: Rome, Italy, 2018. [Google Scholar]
- Wang, B.; Zhou, G.; Guo, S.; Li, X.; Yuan, J.; Hu, A. Improving nitrogen use efficiency in rice for sustainable agriculture: Strategies and future perspectives. Life 2022, 12, 1653. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Wang, W.; Chen, J.; Liu, Y.; Chu, C. Genetic improvement toward nitrogen-use efficiency in rice: Lessons and perspectives. Mol. Plant 2023, 16, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Okamoto, M.; Beatty, P.H.; Rothstein, S.J.; Good, A.G. The genetics of nitrogen use efficiency in crop plants. Ann. Rev. Genet. 2015, 49, 269–289. [Google Scholar] [CrossRef]
- Ladha, J.K.; Jat, M.L.; Stirling, C.M.; Chakraborty, D.; Pradhan, P.; Krupnik, T.J.; Sapkota, T.B.; Pathak, H.; Rana, D.S.; Tesfaye, K.; et al. Achieving the sustainable development goals in agriculture: The crucial role of nitrogen in cereal-based systems. Adv. Agron. 2020, 163, 39–116. [Google Scholar] [CrossRef]
- Neeraja, C.; Subramanyam, D.; Surekha, K.; Rao, P.R.; Rao, L.S.; Babu, M.P.; Voleti, S.R. Advances in genetic basis of nitrogen use efficiency of rice. Indian J. Plant Physiol. 2016, 21, 504–513. [Google Scholar] [CrossRef]
- Evans, J.R.; Clarke, V.C. The nitrogen cost of photosynthesis. J. Exp. Bot. 2019, 70, 7–15. [Google Scholar] [CrossRef]
- Zhao, L.S.; Li, K.; Wang, Q.M.; Song, X.Y.; Su, H.N.; Xie, B.B.; Zhang, X.Y.; Huang, F.; Chen, X.L.; Zhou, B.C.; et al. Nitrogen starvation impacts the photosynthetic performance of Porphyridium cruentum as revealed by chlorophyll a fluorescence. Sci. Rep. 2017, 7, 8542. [Google Scholar] [CrossRef]
- Cen, H.; Weng, H.; Yao, J.; He, M.; Lv, J.; Hua, S.; Li, H.Y.; He, Y. Chlorophyll fluorescence imaging uncovers photosynthetic fingerprint of citrus Huanglongbing. Front. Plant Sci. 2017, 8, 1509. [Google Scholar] [CrossRef]
- Zhong, C.; Cao, X.; Bai, Z.; Zhang, J.; Zhu, L.; Huang, J.; Jin, Q. Nitrogen metabolism correlates with the acclimation of photosynthesis to short-term water stress in rice (Oryza sativa L.). Plant Physiol. Biochem. 2018, 125, 52–62. [Google Scholar] [CrossRef]
- Guo, P.; Ren, J.; Shi, X.; Xu, A.; Zhang, P.; Guo, F.; Feng, Y.; Zhao, X.; Yu, H.; Jiang, C. Optimized nitrogen application ameliorates the photosynthetic performance and yield potential in peanuts as revealed by OJIP chlorophyll fluorescence kinetics. BMC Plant Biol. 2024, 24, 774. [Google Scholar] [CrossRef]
- Song, X.; Zhou, G.; Ma, B.-L.; Wu, W.; Ahmad, I.; Zhu, G.; Yan, W.; Jiao, X. Nitrogen application improved photosynthetic productivity, chlorophyll fluorescence, yield and yield components of two oat genotypes under saline conditions. Agronomy 2019, 9, 115. [Google Scholar] [CrossRef]
- Zhong, C.; Cao, X.; Hu, J.; Zhu, L.; Zhang, J.; Huang, J.; Jin, Q. Nitrogen metabolism in adaptation of photosynthesis to water stress in rice grown under different nitrogen levels. Front. Plant Sci. 2017, 8, 1079. [Google Scholar] [CrossRef]
- Zhou, C.; Huang, Y.; Jia, B.; Wang, Y.; Wang, Y.; Xu, Q.; Li, R.; Wang, S.; Dou, F. Effects of cultivar, nitrogen rate, and planting density on rice-grain quality. Agronomy 2018, 8, 246. [Google Scholar] [CrossRef]
- Kaltrina, R.; Kristi, B.; Dea, Z.; Lulezim, S.; René, H.; Jakob, S.; Reinhard, B. Alpine ecology, plant biodiversity and photosynthetic performance of marker plants in a nitrogen gradient induced by Alnus bushes. BMC Ecol. 2020, 20, 23. [Google Scholar] [CrossRef] [PubMed]
- Shao, A.; Sun, Z.; Fan, S.; Xu, X.; Wang, W.; Amombo, E.; Yin, Y.; Li, X.; Wang, G.; Wang, H. Moderately low nitrogen application mitigate the negative effects of salt stress on annual ryegrass seedlings. PeerJ 2020, 8, e10427. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wan, L.; Igathinathane, C.; Zhang, Z.; Guo, Y.; Sun, D.; Cen, H. Spatiotemporal heterogeneity of chlorophyll content and fluorescence response within rice (Oryza sativa L.) canopies under different nitrogen treatments. Front. Plant Sci. 2021, 12, 645977. [Google Scholar] [CrossRef]
- Zhao, M.; Li, J.; Zhang, B.; Dong, Z.; Wang, M. The compensatory mechanism in exploring crop production potential. Acta Agron. Sin. 2006, 32, 1566–1573. [Google Scholar]
- Tsukaguchi, T.; Taniguchi, Y.; Ito, R. The effects of nitrogen uptake before and after heading on grain protein content and the occurrence of basal-and back-white grains in rice (Oryza sativa L.). Plant Prod. Sci. 2016, 19, 508–517. [Google Scholar] [CrossRef]
- Chen, X.; Huang, L.; Zhong, L.; Huang, W.; He, H. Nitrogen deficiency and compensatory effects in super hybrid late rice during different growth and development stages. Acta Agric. Univ. Jiangxiensis 2015, 37, 11–19. [Google Scholar] [CrossRef]
- Xiong, Q.; Tang, G.; Zhong, L.; He, H.; Chen, X. Response to nitrogen deficiency and compensation on physiological characteristics, yield formation, and nitrogen utilization of rice. Front. Plant Sci. 2018, 9, 1075. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, Y.; Li, K.; Yan, M.; Zhang, J.; Yu, M.; Tang, S.; Wang, L.; Qu, H.; Luo, L. Nitrogen mediates flowering time and nitrogen use efficiency via floral regulators in rice. Curr. Biol. 2021, 31, 671–683. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Weng, J.; Zhang, Q.; Yu, L.; Yao, Q.; Chang, L.; Niu, Q.L. Physiological and biochemical responses of Cucumis melo L. chloroplasts to low-phosphate stress. Front. Plant Sci. 2018, 9, 1525. [Google Scholar] [CrossRef]
- Forde, B.G.; Clarkson, D.T. Nitrate and ammonium nutrition of plants: Physiological and molecular perspectives. Adv. Bot. Res. 1999, 30, 1–90. [Google Scholar] [CrossRef]
- Foyer, C.H.; Valadier, M.H.; Migge, A.; Becker, T.W. Drought-induced effects on nitrate reductase activity and mRNA and on the coordination of nitrogen and carbon metabolism in maize leaves. Plant Physiol. 1998, 117, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Tan, J. Recent advances in the application of chlorophyll a fluorescence from photosystem II. Photochem. Photobiol. 2015, 91, 1–14. [Google Scholar] [CrossRef]
- Thu, H.N.T.; Shim, I.S.; Kobayashi, K.; Kenji, U. Accumulation of some nitrogen compounds in response to salt stress and their relationships with salt tolerance in rice (Oryza sativa L.) seedlings. J. Plant Growth Regul. 2003, 41, 159–164. [Google Scholar] [CrossRef]
- Konishi, N.; Ishiyama, K.; Matsuoka, K.; Maru, I.; Hayakawa, T.; Yamaya, T.; Kojima, S. NADH-dependent glutamate synthase plays a crucial role in assimilating ammonium in the Arabidopsis root. Physiol. Plant. 2014, 152, 138–151. [Google Scholar] [CrossRef]
- Marlon, D.; Théo, P.; Francesc, M.; Leyre, U.; Jose, A.; Cédric, C.; Izargi, V.; Pilar, C.; Ernesto, I.; Yves, G.; et al. Natural variation in the adjustment of primary metabolism determines ammonium tolerance in the model grass Brachypodium distachyon. J. Exp. Bot. 2024, 22, 7237–7253. [Google Scholar] [CrossRef]
- Joshi, V.; Joung, J.G.; Fei, Z.; Jander, G. Interdependence of threonine, methionine and isoleucine metabolism in plants: Accumulation and transcriptional regulation under abiotic stress. Amino Acids 2010, 39, 933–947. [Google Scholar] [CrossRef]
- Ashraf, M.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Singh, M.; Singh, V.P.; Prasad, S.M. Responses of photosynthesis, nitrogen and proline metabolism to salinity stress in Solanum lycopersicum under different levels of nitrogen supplementation. Plant Physiol. Biochem. 2016, 109, 72–83. [Google Scholar] [CrossRef]
- Gao, J.; Liu, L.; Ma, N.; Yang, J.; Dong, Z.; Zhang, J.; Zhang, J.; Cai, M. Effect of ammonia stress on carbon metabolism in tolerant aquatic plant—Myriophyllum aquaticum. Environ. Pollut. 2020, 263, 114412. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, C.; Chaves, M. Photosynthesis and drought: Can we make metabolic connections from available data? J. Exp. Bot. 2011, 62, 869–882. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.; Jian, S.; Li, Y.; Yue, M.; Xiong, J.; Tao, L. Alterations of panicle antioxidant metabolism and carbohydrate content and pistil water potential involved in spikelet sterility in rice under water-deficit stress. Rice Sci. 2010, 17, 303–310. [Google Scholar] [CrossRef]
- Li, M.; Xu, J.; Wang, X.; Fu, H.; Zhao, M.; Wang, H.; Shi, L. Photosynthetic characteristics and metabolic analyses of two soybean genotypes revealed adaptive strategies to low-nitrogen stress. J. Plant Physiol. 2018, 229, 132–141. [Google Scholar] [CrossRef]
- Gitelson, A.; Gritz, Y.; Merzlyak, M. Relationships between leaf chlorophyll content and spectral reflectance and algorithms for nondestructive chlorophyll assessment in higher plant leaves. J. Plant Physiol. 2003, 160, 271–282. [Google Scholar] [CrossRef]
- Richards, R. Selectable traits to increase crop photosynthesis and yield of grain crops. J. Exp. Bot. 2000, 51, 447–458. [Google Scholar] [CrossRef]
- He, L.; Yu, L.; Li, B.; Du, N.; Guo, S. The effect of exogenous calcium on cucumber fruit quality, photosynthesis, chlorophyll fluorescence, and fast chlorophyll fluorescence during the fruiting period under hypoxic stress. BMC Plant Biol. 2018, 18, 180. [Google Scholar] [CrossRef]
- Ye, X.; Chen, X.F.; Deng, C.L.; Yang, L.T.; Lai, N.W.; Guo, J.X. Magnesium-deficiency effects on pigments, photosynthesis and photosynthetic electron transport of leaves, and nutrients of leaf blades and veins in Citrus sinensis seedlings. Plants 2019, 8, 389. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Bąba, W.; Gediga, K.; Goltsev, V.; Samborska, I.A.; Cetner, M.D.; Dimitrova, S.; Piszcz, U.; Bielecki, K.; Karmowska, K. Chlorophyll fluorescence as a tool for nutrient status identification in rapeseed plants. Photosynth. Res. 2018, 136, 329–343. [Google Scholar] [CrossRef]
- Strasser, R.J.; Tsimilli, M.; Qiang, S.; Goltsev, V. Simultaneous in vivo recording of prompt and delayed fluorescence and 820-nm reflection changes during drying and after rehydration of the resurrection plant Haberlea rhodopensis. Biochim. Biophys. Acta Bioenerg. 2010, 1797, 1313–1326. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.; Ling, F.; Jia, D.; Cui, J.; Zhang, Z.; Xu, C.; Yu, L.; Guan, C.; Wang, Y.; Zhang, M. Effects of low nitrogen on seedling growth, photosynthetic characteristics and antioxidant system of rice varieties with different nitrogen efficiencies. Sci. Rep. 2023, 13, 19780. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Chen, G.; Zhou, Y.; Shen, Q. Ammonium nutrition increases photosynthesis rate under water stress at early development stage of rice (Oryza sativa L.). Plant Soil 2007, 296, 115–124. [Google Scholar] [CrossRef]
- Tsimilli-michael, M. Revisiting JIP-test: An educative review on concepts, assumptions, approximations, definitions and terminology. Photosynthetica 2020, 58, 275–292. [Google Scholar] [CrossRef]
- Cataldo, D.A.; Haroon, M.; Schrader, L.E.; Youngs, V.L. Rapid colorimetric determination of nitrate in plant tissue by nitration of salicylic acid. Commun. Soil Sci. Plant Anal. 1975, 6, 71–80. [Google Scholar] [CrossRef]
- Santoni, S.; Bonifacio, E.; Zanini, E. Indophenol blue colorimetric method for measuring cation exchange capacity in sandy soils. Commun. Soil Sci. Plant Anal. 2001, 32, 2519–2530. [Google Scholar] [CrossRef]
- Yokoyama, S.; Hiramatsu, J.I. A modified ninhydrin reagent using ascorbic acid instead of potassium cyanide. J. Biosci. Bioeng. 2003, 95, 204–205. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Khoo, K.H.; Chen, S.T.; Lin, C.C.; Wong, C.H.; Lin, C.H. Studies on the immuno-modulating and antitumor activities of Ganoderma lucidum (Reishi) polysaccharides: Functional and proteomic analyses of a fucose-containing glycoprotein fraction responsible for the activities. Bioorg. Med. Chem. 2002, 10, 1057–1062. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Hageman, R.H.; Reed, A.J. Nitrate reductase from higher plants. Meth. Enzymol. 1980, 69, 270–280. [Google Scholar] [CrossRef]
- Zhang, C.; Peng, S.; Peng, X.; Chavez, A.Q.; Bennett, J. Response of glutamine synthetase isoforms to nitrogen sources in rice (Oryza sativa L.) roots. Plant Sci. 1997, 125, 163–170. [Google Scholar] [CrossRef]
- Wu, L.; Jiang, S.; Tao, Q. The application of colormetric method on the determination of transaminase activity. Chin. J. Soil Sci. 1998, 3, 41–43. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, Z.; Sun, W.; Luo, C.; Zhang, Q.; Osman, F.M.; Guan, C.; Wang, Y.; Zhang, M.; Zhang, X.; Ding, J.; et al. Differential Responses of Rice Genotypes to Nitrogen Supply: Impacts on Nitrogen Metabolism and Chlorophyll Fluorescence Kinetics. Plants 2025, 14, 2467. https://doi.org/10.3390/plants14162467
Qi Z, Sun W, Luo C, Zhang Q, Osman FM, Guan C, Wang Y, Zhang M, Zhang X, Ding J, et al. Differential Responses of Rice Genotypes to Nitrogen Supply: Impacts on Nitrogen Metabolism and Chlorophyll Fluorescence Kinetics. Plants. 2025; 14(16):2467. https://doi.org/10.3390/plants14162467
Chicago/Turabian StyleQi, Zexin, Wenzheng Sun, Chun Luo, Qiang Zhang, Feisal Mohamed Osman, Chenglong Guan, Ye Wang, Mengru Zhang, Xiaotong Zhang, Jiale Ding, and et al. 2025. "Differential Responses of Rice Genotypes to Nitrogen Supply: Impacts on Nitrogen Metabolism and Chlorophyll Fluorescence Kinetics" Plants 14, no. 16: 2467. https://doi.org/10.3390/plants14162467
APA StyleQi, Z., Sun, W., Luo, C., Zhang, Q., Osman, F. M., Guan, C., Wang, Y., Zhang, M., Zhang, X., Ding, J., Zhang, Y., Ling, F., Liu, X., Zhang, Z., & Xu, C. (2025). Differential Responses of Rice Genotypes to Nitrogen Supply: Impacts on Nitrogen Metabolism and Chlorophyll Fluorescence Kinetics. Plants, 14(16), 2467. https://doi.org/10.3390/plants14162467




