Foliar Nutrition Influences Yield, Nut Quality and Kernel Composition in Hazelnut cv Mortarella
Abstract
1. Introduction
2. Results
2.1. Yield and Biometrical Characteristics of Nuts and Kernels
2.2. Leaf Mineral Composition
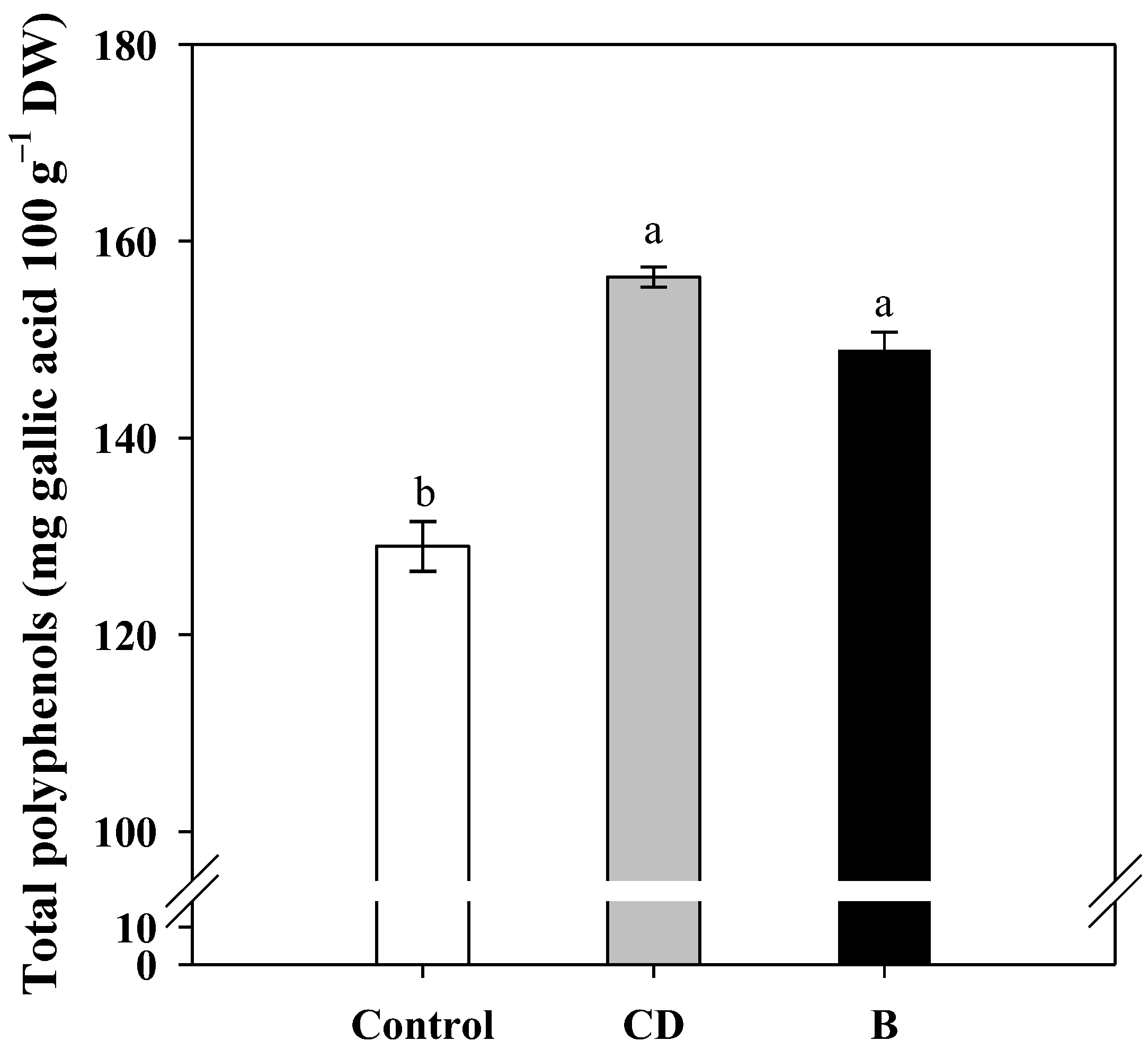
2.3. Kernel Composition and Total Polyphenols Content
2.4. Lipid Alteration
2.5. Oil Fatty Acid Composition
3. Discussion
3.1. Effects of Foliar Fertilization on Leaf Mineral Composition and Yield Components
3.2. Effects of Foliar Fertilization on Kernel Composition and Quality
4. Materials and Methods
4.1. Plant Material and Experimental Site
4.2. Foliar Nutrition Treatments and Foliar Mineral Analysis
4.3. Foliar Analyses
4.4. Yield, Nut and Kernel Biometrical Traits
4.5. Kernel Constituents and Fatty Acid Composition
4.6. Analyses of Lipid Alteration and Oxidation
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- International Nut and Dried Fruit Foundation. Nuts and Dried Fruits Statistical Yearbook 2020–2021; International Nut and Dried Fruit Foundation: Reus, Spain, 2021; Volume 26, p. 29. [Google Scholar]
- Sands, R.; Mulligan, D.R. Water and Nutrient Dynamics and Tree Growth. For. Ecol. Manag. 1990, 30, 91–111. [Google Scholar] [CrossRef]
- Rombolà, A.D.; Tagliavini, M. Iron Nutrition of Fruit Tree Crops. In Iron Nutrition Plants Rhizospheric Microorg; Barton, L.L., Abadia, J., Eds.; Springer: Dordrecht, The Nederlands, 2006; pp. 61–83. [Google Scholar] [CrossRef]
- McMichael, B.L.; Quisenberry, J.E. The Impact of the Soil Environment on the Growth of Root Systems. Environ. Exp. Bot. 1993, 33, 53–61. [Google Scholar] [CrossRef]
- Mengel, K. Alternative or Complementary Role of Foliar Supply in Mineral Nutrition. Acta Hortic. 2002, 594, 33–47. [Google Scholar] [CrossRef]
- Silvestri, C.; Bacchetta, L.; Bellincontro, A.; Cristofori, V. Advances in Cultivar Choice, Hazelnut Orchard Management, and Nut Storage to Enhance Product Quality and Safety: An Overview. J. Sci. Food Agric. 2021, 101, 27–43. [Google Scholar] [CrossRef]
- Germain, E.; Sarraquigne, J.P. Le Noisetier; Centre Technique Interprofessionnel des Fruits et Legumes, Instituit National de La Recherche Agronomique, Association National des Producteurs de Noisette: Paris, France, 2004; pp. 170–189. [Google Scholar]
- Olsen, J.L. Nutrient management guide: Hazelnuts. Oregon State University Extension Service 001, 1–4. Available online: https://agsci.oregonstate.edu/sites/agscid7/files/horticulture/attachments/em8786-e.pdf (accessed on 20 January 2023).
- Cristofori, V.; Bertazza, G.; Bignami, C. Changes in Kernel Chemical Composition during Nut Development of Three Italian Hazelnut Cultivars. Fruits 2015, 70, 311–322. [Google Scholar] [CrossRef]
- Wells, M.L.; Conner, P.J.; Funderburk, J.F.; Price, J.G. Effects of Foliar-Applied Boron on Fruit Retention, Fruit Quality, and Tissue Boron Concentration of Pecan. HortScience Horts 2008, 43, 696–699. [Google Scholar] [CrossRef]
- Stephenson, R.A.; Gallagher, E.C. Effects of Foliar Boron Sprays on Yield and Quality of Macadamia Nuts. Sci. Hortic. 1987, 32, 97–103. [Google Scholar] [CrossRef]
- .Nyomora, A.M.S.; Brown, P.H.; Krueger, B. Rate and Time of Boron Application Increase Almond Productivity and Tissue Boron Concentration. HortScience 1999, 34, 242–245. [Google Scholar] [CrossRef]
- Borges, O.M.P.; Carvalho, J.L.R.S.; Silva, A.P.; Santos, A. Effects of Foliar Boron Sprays on Yield and Nut Quality of “Segorbe” and “Fertile de Coutard” Hazelnuts. Acta Hortic. 2001, 556, 299–302. [Google Scholar] [CrossRef]
- Alkoshab, O.; Righetti, T.L.; Dixon, A.R. Evaluation of DRIS for Judging the Nutritional Status of Hazelnuts. J. Am. Soc. Hortic. Sci. 2022, 113, 643–647. [Google Scholar] [CrossRef]
- Painter, J.H.; Hammar, H.E. Effects of Differential Applications of Nitrogen, Potassium, Magnesium, Boron, and Phosphorus on Their Concentration in Leaves of Filbert Trees. Proc. Amer. Soc. Hort. Sci. 1962, 80, 315–326. [Google Scholar]
- Baron, L.C.; Riggert, C.; Stebbins, R.L.; Bell, S.M. Growing Hazelnuts in Oregon. Or. State Univ. Ext. Serv. 1985, 1–20. Available online: https://ir.library.oregonstate.edu/concern/administrative_report_or_publications/n296wz40s (accessed on 20 January 2023).
- Shrestha, G.K.; Thompson, M.M.; Righetti, T.L. Foliar-Applied Boron Increases Fruit Set in ‘Barcelona’ Hazelnut. J. Am. Soc. Hortic. Sci. 2022, 112, 412–416. [Google Scholar] [CrossRef]
- Ferrán, X.; Tous, J.; Romero, A.; Lloveras, J.; Pericón, J.R. Boron Does Not Increase Hazelnut Fruit Set and Production. HortScience 1997, 32, 1053–1055. [Google Scholar] [CrossRef]
- Paula Silva, A.; Rosa, E.; Haneklaus, S.H. Influence of Foliar Boron Application on Fruit Set and Yield of Hazelnut. J. Plant Nutr. 2003, 26, 561–569. [Google Scholar] [CrossRef]
- Meriño-Gergichevich, C.; Luengo-Escobar, A.; Alarcón, D.; Reyes-Díaz, M.; Ondrasek, G.; Morina, F.; Ogass, K. Combined Spraying of Boron and Zinc During Fruit Set and Premature Stage Improves Yield and Fruit Quality of European Hazelnut Cv. Tonda Di Giffoni. Front. Plant Sci. 2021, 12, 661542. [Google Scholar] [CrossRef]
- Olsen, J.L.; Cacka, J.F. Foliar Fertilizers on Hazelnuts in Oregon, USA. Acta Hortic. 2009, 845, 349–352. [Google Scholar] [CrossRef]
- Cacka, J.F.; Smith, F. Foliar Nutrition Applied at Early Hazelnut Development Shows Positive Yield and Quality Factors in the Willamette Valley of Oregon. Acta Hortic. 2009, 845, 343–348. [Google Scholar] [CrossRef]
- Nicolosi, E.; Leotta, G.; Raiti, G. Effect of Foliar Fertilization on Hazelnuts Growing in Mount Etna Area. Acta Hortic. 2009, 845, 373–378. [Google Scholar] [CrossRef]
- Solar, A.; Stampar, F. Characterisation of Selected Hazelnut Cultivars: Phenology, Growing and Yielding Capacity, Market Quality and Nutraceutical Value. J. Sci. Food Agric. 2011, 91, 1205–1212. [Google Scholar] [CrossRef] [PubMed]
- Tous, J.; Romero, A.; PLana, J.; Sentis, X.; Ferrán, J. Effect of Nitrogen, Boron and Iron Fertilization on Yiel and Nut Quality of “Negret” Hazelnut Trees. Acta Hortic. 2005, 686, 277–280. [Google Scholar] [CrossRef]
- Erdogan, V.; Aygun, A. Effect of Foliar Boron Application on Fruit Set in “Tombul” Hazelnut. Acta Hortic. 2009, 845, 331–336. [Google Scholar] [CrossRef]
- Pansecchi, A.; Roversi, A.; Malvicini, G.L. Some Years of Trials on the Effectiveness of Hazelnut Foliar Fertilization. Acta Hortic. 2014, 1052, 195–199. [Google Scholar] [CrossRef]
- Cristofori, V.; Bortolato, A.; Valentini, R.; Stelliferi, R.; Valentini, B. Total Foliar Nutrition Applied on European Hazelnut. Acta Hortic. 2018, 1226, 273–279. [Google Scholar] [CrossRef]
- Ellena, M.; Sandoval, P.; Montenegro, A.; Gonzalez, A.; Azocar, G. Effect of Foliar Nutrient Applications on Fruit Set in “Chilean Barcelona” Hazelnut, in Southern Chile. Acta Hortic. 2014, 1052, 231–234. [Google Scholar] [CrossRef]
- Cabo, S.; Aires, A.; Carvalho, R.; Vilela, A.; Pascual-Seva, N.; Silva, A.P.; Gonçalves, B. Kaolin, Ascophyllum Nodosum and Salicylic Acid Mitigate Effects of Summer Stress Improving Hazelnut Quality. J. Sci. Food Agric. 2021, 101, 459–475. [Google Scholar] [CrossRef] [PubMed]
- Solar, A.; Jakopic, J.; Stampar, F.; Veberic, R. Soil Applied Nitrogen and Foliar Fertilizers Together with Biostimulators Increase Productivity but Decrease Antioxidative Potential in Hazelnut. Acta Hortic. 2018, 1226, 281–288. [Google Scholar] [CrossRef]
- Milosevic, T.; Milosevic, N.; Glisic, I.; Paunovic, G. Leaf Nutritional Status and Macronutrient Dynamics in European Hazelnut (Corylus avellana L.) under Western Serbian Conditions. Pak. J. Bot. 2009, 41, 3169–3178. [Google Scholar]
- Hänsch, R.; Mendel, R.R. Physiological Functions of Mineral Micronutrients (Cu, Zn, Mn, Fe, Ni, Mo, B, Cl). Curr. Opin. Plant Biol. 2009, 12, 259–266. [Google Scholar] [CrossRef]
- Barker, A.V.; Pilbeam, D.J. (Eds.) Handbook of Plant Nutrition, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2015; ISBN 9781420014877. [Google Scholar] [CrossRef]
- Serdar, U.; Horuz, A.; Demir, T. The Effects of B-Zn Fertilization on Yield, Cluster Drop and Nut Traits in Hazelnut. J. Biol. Sci. 2005, 5, 786–789. [Google Scholar] [CrossRef]
- Solar, A.; Stampar, F. Influence of Boron and Zinc Application on Flowering and Nut Set in “Tonda Di Giffoni” Hazelnut. Acta Hortic. 2001, 556, 307–312. [Google Scholar] [CrossRef]
- Wang, N.; Yang, C.; Pan, Z.; Liu, Y.; Peng, S. Boron Deficiency in Woody Plants: Various Responses and Tolerance Mechanisms. Front. Plant Sci. 2015, 6, 916. [Google Scholar] [CrossRef]
- Saadati, S.; Moallemi, N.; Mortazavi, S.M.H.; Seyyednejad, S.M. Effects of Zinc and Boron Foliar Application on Soluble Carbohydrate and Oil Contents of Three Olive Cultivars during Fruit Ripening. Sci. Hortic. 2013, 164, 30–34. [Google Scholar] [CrossRef]
- Desouky, I.M.; Haggag, L.F.; Abd El-Migeed, M.M.M.; Kishk, Y.F.M.; El-Hady, E.S. Effect of Boron and Calcium Nutrients Sprays on Fruit Set, Oil Content and Oil Quality of Some Olive Oil Cultivars. World J. Agric. Sci. 2009, 5, 180–185. [Google Scholar]
- Sawan, Z.M.; Hafez, S.A.; Basyony, A.E. Effect of Nitrogen and Zinc Fertilization and Plant Growth Retardants on Cottonseed, Protein, Oil Yields, and Oil Properties. J. Am. Oil Chem. Soc. 2001, 78, 1087–1092. [Google Scholar] [CrossRef]
- Yang, M.; Shi, L.; Xu, F.S.; Lu, J.W.; Wang, Y.H. Effects of B, Mo, Zn, and Their Interactions on Seed Yield of Rapeseed (Brassica napus L.). Pedosphere 2009, 19, 53–59. [Google Scholar] [CrossRef]
- Mekhail Tawfik, M.; Gobarah, M.E.; Mohamed, M.H.; Tawfik, M. Effect of Phosphorus Fertilizer and Foliar Spraying with Zinc on Growth, Yield and Quality of Groundnut under Reclaimed Sandy Soils. J. Appl. Sci. Res. 2006, 2, 491–496. [Google Scholar]
- Ashraf, N.; Ashraf, M.; Tomar, C.S. Effect of Foliar Application of Nutrients and Biostimulant on Growth, Phenology and Yield Attributes of Pecan Nut Cv. “Western Schley”. J. Food Agric. Environ. 2013, 11, 1222–1226. [Google Scholar]
- Ramteke, V.; Preethi, P.; Veena, G.L.; Nirala, Y.S. Impact of Foliar Application of Primary Nutrients on Growth and Yield Contributing Traits in Cashew (Anacardium occidentale L.). J. Environ. Biol. 2022, 43, 477–483. [Google Scholar] [CrossRef]
- Shchitaeva, V.A. Effect of Zinc on the Metabolic Activity of Fine-Fibered Cotton Root System. Izv. Akad. Nauk. Turkm. SSR. Seriia Biol. Nauk. 1984, 8–13. [Google Scholar]
- El-Kader, A.; Mona, G. Effect of Sulfur Application and Foliar Spraying with Zinc and Boron on Yield, Yield Components, and Seed Quality of Peanut (Arachis hypogaea L.). Res. J. Agric. Biol. Sci. 2013, 9, 127–135. [Google Scholar]
- Bi, G.; Scagel, C.F.; Cheng, L.; Fuchigami, L.H. Soil and Foliar Nitrogen Supply Affects the Composition of Nitrogen and Carbohydrates in Young Almond Trees. J. Hortic. Sci. Biotechnol. 2004, 79, 175–181. [Google Scholar] [CrossRef]
- Asare, E.; Scarisbrick, D.H. Rate of Nitrogen and Sulphur Fertilizers on Yield, Yield Components and Seed Quality of Oilseed Rape (Brassica napus L.). Field Crops Res. 1995, 44, 41–46. [Google Scholar] [CrossRef]
- Pannico, A.; Schouten, R.E.; Basile, B.; Romano, R.; Woltering, E.J.; Cirillo, C. Non-Destructive Detection of Flawed Hazelnut Kernels and Lipid Oxidation Assessment Using NIR Spectroscopy. J. Food Eng. 2015, 160, 42–48. [Google Scholar] [CrossRef]
- Dag, A.; Ben-David, E.; Kerem, Z.; Ben-Gal, A.; Erel, R.; Basheer, L.; Yermiyahu, U. Olive Oil Composition as a Function of Nitrogen, Phosphorus and Potassium Plant Nutrition. J. Sci. Food Agric. 2009, 89, 1871–1878. [Google Scholar] [CrossRef]
- Ghasemnezhad, A.; Honermeier, B. Yield, Oil Constituents, and Protein Content of Evening Primrose (Oenothera biennis L.) Seeds Depending on Harvest Time, Harvest Method and Nitrogen Application. Ind. Crops Prod. 2008, 28, 17–23. [Google Scholar] [CrossRef]
- Bendini, A.; Cerretani, L.; Carrasco-Pancorbo, A.; Gómez-Caravaca, A.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Lercker, G. Phenolic Molecules in Virgin Olive Oils: A Survey of Their Sensory Properties, Health Effects, Antioxidant Activity and Analytical Methods. An Overview of the Last Decade. Molecules 2007, 12, 1679–1719. [Google Scholar] [CrossRef] [PubMed]
- Servili, M.; Selvaggini, R.; Esposto, S.; Taticchi, A.; Montedoro, G.; Morozzi, G. Health and Sensory Properties of Virgin Olive Oil Hydrophilic Phenols: Agronomic and Technological Aspects of Production That Affect Their Occurrence in the Oil. J. Chromatogr. A 2004, 1054, 113–127. [Google Scholar] [CrossRef] [PubMed]
- Pelvan, E.; Alasalvar, C.; Uzman, S. Effects of roasting on the antioxidant status and phenolic profiles of commercial Turkish hazelnut varieties (Corylus avellana L.). J. Agric. Food Chem. 2012, 60, 1218–1223. [Google Scholar] [CrossRef]
- Cakmak, I.; Römheld, V. Boron Deficiency-Induced Impairments of Cellular Functions in Plants. Plant Soil 1997, 193, 71–83. [Google Scholar] [CrossRef]
- Pfeffer, H.; Dannel, F.; Römheld, V. Are There Connections between Phenol Metabolism, Ascorbate Metabolism and Membrane Integrity in Leaves of Boron-Deficient Sunflower Plants? Physiol. Plant. 1998, 104, 479–485. [Google Scholar] [CrossRef]
- Soil Texture Calculator [Internet]. Natural Resource Conservation Service, United States Department of Agriculture. Available online: https://www.nrcs.usda.gov/resources/education-and-teaching-materials/soil-texture-calculator (accessed on 16 May 2023).
- Pannico, A.; El-Nakhel, C.; Kyriacou, M.C.; Giordano, M.; Stazi, S.R.; De Pascale, S.; Rouphael, Y. Combining Micronutrient Deficiency and Enhancing Food Functional Quality Through Selenium Fortification of Select Lettuce Genotypes Grown in a Closed Soilless System. Front. Plant Sci. 2019, 10, 1495. [Google Scholar] [CrossRef]
- Cunniff, P. Official Methods of AOAC Analysis; Association of Official Analytical Chemists: Washington, DC, USA, 1995. [Google Scholar]
- Oliveira, I.; Sousa, A.; Morais, J.S.; Ferreira, I.C.F.R.; Bento, A.; Estevinho, L.; Pereira, J.A. Chemical Composition, and Antioxidant and Antimicrobial Activities of Three Hazelnut (Corylus avellana L.) Cultivars. Food Chem. Toxicol. 2008, 46, 1801–1807. [Google Scholar] [CrossRef]
- Jakopic, J.; Petkovsek, M.M.; Likozar, A.; Solar, A.; Stampar, F.; Veberic, R. HPLC-MS Identification of Phenols in Hazelnut (Corylus avellana L.) Kernels. Food Chem. 2011, 124, 1100–1106. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A., Jr.; Rossi, J.A., Jr. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar] [CrossRef]
- International Union of Pure and Applied Chemistry. Standard Methods for the Analysis of Oils, Fats and Derivatives; International Union of Pure and Applied Chemistry: Zurich, Switzerland, 1987; pp. 99–102. [Google Scholar]
- Pannico, A.; Cirillo, C.; Giaccone, M.; Scognamiglio, P.; Romano, R.; Caporaso, N.; Sacchi, R.; Basile, B. Fruit Position within the Canopy Affects Kernel Lipid Composition of Hazelnuts. J. Sci. Food Agric. 2017, 97, 4790–4799. [Google Scholar] [CrossRef]
- San Martín, M.B.; Fernández-García, T.; Romero, A.; López, A. Effect of Modified Atmosphere Storage on Hazelnut Quality. J. Food Process. Preserv. 2001, 25, 309–321. [Google Scholar] [CrossRef]
- Lòpez, A.; Piqué, M.T.; Boatella, J.; Parcerosa, J.; Romero, A.; Ferrà, A.; Garci, J. Influence of Drying Conditions on the Hazelnut Quality. I. Lipid Oxidation. Dry. Technol. 1997, 15, 965–977. [Google Scholar] [CrossRef]
- García, J.M.; Gutiérrez, F.; Castellano, J.M.; Perdiguero, S.; Morilla, A.; Albi, M.A. Influence of Storage Temperature on Fruit Ripening and Olive Oil Quality. J. Agric. Food Chem. 1996, 44, 264–267. [Google Scholar] [CrossRef]
| Source of Variance | Yield | Nut Number | Blank | Nut Weight | Kernel Dry Weight |
|---|---|---|---|---|---|
| (kg Plant−1) | (no. Plant−1) | (%) | (g) | (g) | |
| Year (Y) | |||||
| 2011 | 3.29 ± 0.16 b | 1357 ± 48.37 b | 4.42 ± 0.34 b | 2.42 ± 0.06 b | 1.07 ± 0.01 b |
| 2012 | 3.30 ± 0.10 b | 1245 ± 41.66 b | 3.87 ± 0.53 c | 2.66 ± 0.05 a | 1.21 ± 0.02 a |
| 2013 | 4.36 ± 0.26 a | 1613 ± 99.45 a | 5.17 ± 0.34 a | 2.71 ± 0.02 a | 1.24 ± 0.01 a |
| *** | *** | *** | *** | *** | |
| Fertilization (F) | |||||
| Control | 3.56 ± 0.21 b | 1400 ± 72.83 b | 5.86 ± 0.19 a | 2.55 ± 0.07 | 1.14 ± 0.03 b |
| CD | 4.16 ± 0.29 a | 1557 ± 103.1 a | 4.04 ± 0.25 b | 2.67 ± 0.04 | 1.20 ± 0.03 a |
| B | 3.23 ± 0.13 b | 1257 ± 36.32 c | 3.57 ± 0.39 c | 2.57 ± 0.07 | 1.17 ± 0.03 ab |
| *** | *** | *** | ns | * | |
| Y × F | |||||
| 2011 × Control | 3.24 ± 0.05 cd | 1418 ± 30.77 bcd | 5.54 ± 0.18 ab | 2.28 ± 0.05 c | 1.04 ± 0.01 |
| 2011 × CD | 3.86 ± 0.03 bc | 1472 ± 43.30 bc | 3.23 ± 0.08 e | 2.62 ± 0.06 ab | 1.10 ± 0.01 |
| 2011 × B | 2.77 ± 0.12 d | 1179 ± 31.91 cd | 4.49 ± 0.10 cd | 2.35 ± 0.04 bc | 1.07 ± 0.02 |
| 2012 × Control | 3.08 ± 0.06 cd | 1156 ± 46.18 d | 5.60 ± 0.40 ab | 2.67 ± 0.06 a | 1.19 ± 0.01 |
| 2012 × CD | 3.45 ± 0.25 cd | 1273 ± 60.58 cd | 3.96 ± 0.20 de | 2.70 ± 0.09 a | 1.23 ± 0.04 |
| 2012 × B | 3.37 ± 0.17 cd | 1305 ± 94.16 cd | 2.05 ± 0.09 f | 2.60 ± 0.11 abc | 1.21 ± 0.03 |
| 2013 × Control | 4.37 ± 0.16 ab | 1627 ± 69.58 ab | 6.43 ± 0.07 a | 2.69 ± 0.02 a | 1.20 ± 0.02 |
| 2013 × CD | 5.17 ± 0.37 a | 1925 ± 101.1 a | 4.93 ± 0.07 bc | 2.68 ± 0.05 a | 1.26 ± 0.03 |
| 2013 × B | 3.56 ± 0.08 bcd | 1286 ± 36.80 cd | 4.17 ± 0.16 cd | 2.77 ± 0.05 a | 1.24 ± 0.01 |
| * | *** | *** | * | ns |
| Source of Variance | NO3 | NH4 | P | K | Ca | Mg | Zn | Fe | B | Mn |
|---|---|---|---|---|---|---|---|---|---|---|
| (g 100 g−1 DW) | (g 100 g−1 DW) | (g 100 g−1 DW) | (g 100 g−1 DW) | (g 100 g−1 DW) | (g 100 g−1 DW) | (mg kg−1 DW) | (mg kg−1 DW) | (mg kg−1 DW) | (mg kg−1 DW) | |
| Year (Y) | ||||||||||
| 2011 | 0.023 ± 0.002 | 0.024 ± 0.001 c | 0.233 ± 0.02 a | 0.815 ± 0.01 ab | 0.752 ± 0.02 a | 0.188 ± 0.01 a | 23.21 ± 1.99 b | 135.5 ± 7.02 a | 37.87 ± 5.25 c | 569.4 ± 27.1 a |
| 2012 | 0.015 ± 0.002 | 0.038 ± 0.002 a | 0.225 ± 0.01 a | 0.888 ± 0.03 a | 0.607 ± 0.03 b | 0.161 ± 0.01 b | 34.17 ± 4.83 a | 119.9 ± 5.15 b | 49.04 ± 9.80 b | 453.5 ± 18.0 b |
| 2013 | 0.017 ± 0.002 | 0.029 ± 0.001 b | 0.147 ± 0.01 b | 0.776 ± 0.03 b | 0.534 ± 0.02 b | 0.131 ± 0.01 c | 24.89 ± 2.95 b | 55.59 ± 7.10 c | 79.79 ± 23.11 a | 359.8 ± 5.39 c |
| ns | *** | *** | * | *** | *** | *** | *** | *** | *** | |
| Fertilization (F) | ||||||||||
| Control | 0.018 ± 0.003 | 0.032 ± 0.003 | 0.181 ± 0.02 b | 0.828 ± 0.02 | 0.596 ± 0.04 | 0.148 ± 0.01 b | 18.17 ± 0.74 b | 86.93 ± 13.1 c | 22.79 ± 1.24 c | 432.9 ± 15.5 c |
| CD | 0.018 ± 0.003 | 0.029 ± 0.002 | 0.218 ± 0.02 a | 0.789 ± 0.03 | 0.648 ± 0.04 | 0.172 ± 0.01 a | 31.68 ± 2.38 a | 118.1 ± 9.64 a | 39.19 ± 2.11 b | 489.7 ± 39.4 a |
| B | 0.020 ± 0.001 | 0.029 ± 0.002 | 0.206 ± 0.02 ab | 0.862 ± 0.03 | 0.649 ± 0.04 | 0.160 ± 0.01 ab | 32.43 ± 4.73 a | 105.9 ± 15.8 b | 104.7 ± 17.32 a | 460.2 ± 43.0 b |
| ns | ns | * | ns | ns | * | *** | *** | *** | *** | |
| Y × F | ||||||||||
| 2011 × control | 0.021 ± 0.003 | 0.025 ± 0.002 | 0.180 ± 0.02 bcd | 0.786 ± 0.02 | 0.745 ± 0.01 | 0.180 ± 0.01 | 16.92 ± 0.86 e | 111.9 ± 3.85 c | 19.46 ± 0.58 g | 463.9 ± 17.6 c |
| 2011 × CD | 0.026 ± 0.006 | 0.025 ± 0.001 | 0.281 ± 0.02 a | 0.799 ± 0.01 | 0.765 ± 0.04 | 0.203 ± 0.01 | 22.48 ± 1.26 d | 136.1 ± 4.00 b | 38.50 ± 0.31 de | 613.6 ± 0.40 a |
| 2011 × B | 0.022 ± 0.002 | 0.021 ± 0.001 | 0.239 ± 0.03 ab | 0.860 ± 0.02 | 0.745 ± 0.05 | 0.181 ± 0.01 | 30.24 ± 0.70 c | 158.5 ± 4.09 a | 55.64 ± 1.66 c | 630.7 ± 8.86 a |
| 2012 × control | 0.012 ± 0.005 | 0.043 ± 0.001 | 0.226 ± 0.02 abc | 0.864 ± 0.03 | 0.539 ± 0.04 | 0.143 ± 0.01 | 16.70 ± 0.34 e | 114.1 ± 1.14 bc | 27.40 ± 0.83 efg | 459.4 ± 4.70 c |
| 2012 × CD | 0.014 ± 0.006 | 0.034 ± 0.003 | 0.220 ± 0.02 abcd | 0.865 ± 0.06 | 0.624 ± 0.04 | 0.176 ± 0.01 | 36.14 ± 1.16 b | 136.1 ± 10.7 b | 32.29 ± 0.49 ef | 512.1 ± 4.48 b |
| 2012 × B | 0.020 ± 0.001 | 0.036 ± 0.002 | 0.229 ± 0.01 abc | 0.934 ± 0.06 | 0.660 ± 0.05 | 0.165 ± 0.01 | 49.67 ± 1.87 a | 109.5 ± 0.38 c | 87.43 ± 6.39 b | 389.1 ± 7.56 d |
| 2013 × control | 0.020 ± 0.004 | 0.029 ± 0.001 | 0.138 ± 0.01 d | 0.834 ± 0.03 | 0.505 ± 0.04 | 0.120 ± 0.01 | 20.88 ± 0.42 de | 34.80 ± 1.80 e | 21.52 ± 0.66 fg | 375.3 ± 6.48 de |
| 2013 × CD | 0.013 ± 0.001 | 0.027 ± 0.002 | 0.154 ± 0.02 bcd | 0.704 ± 0.06 | 0.555 ± 0.03 | 0.137 ± 0.01 | 36.40 ± 1.21 b | 82.16 ± 3.76 d | 46.79 ± 0.02 cd | 343.4 ± 2.23 e |
| 2013 × B | 0.019 ± 0.003 | 0.030 ± 0.001 | 0.150 ± 0.00 cd | 0.791 ± 0.05 | 0.541 ± 0.02 | 0.135 ± 0.01 | 17.39 ± 0.57 de | 49.82 ± 1.30 e | 171.1 ± 0.74 a | 360.9 ± 6.85 de |
| ns | ns | * | ns | ns | ns | *** | *** | *** | *** |
| Source of Variance | Fat Content | Protein Content | Carbohydrate Content | Ash Content |
|---|---|---|---|---|
| (g 100 g−1 DW) | (g 100 g−1 DW) | (g 100 g−1 DW) | (g 100 g−1 DW) | |
| Year (Y) | ||||
| 2012 | 63.47 ± 0.77 | 16.45 ± 0.81 | 17.81 ± 1.34 | 2.28 ± 0.03 |
| 2013 | 62.20 ± 0.63 | 16.47 ± 0.77 | 19.24 ± 1.19 | 2.08 ± 0.06 |
| t-test | ns | ns | ns | ** |
| Fertilization (F) | ||||
| Control | 60.18 ± 0.26 b | 14.77 ± 0.43 b | 22.94 ± 0.60 a | 2.10 ± 0.08 |
| CD | 63.80 ± 0.39 a | 19.47 ± 0.28 a | 14.59 ± 0.55 c | 2.15 ± 0.06 |
| B | 64.53 ± 0.48 a | 15.14 ± 0.14 b | 18.04 ± 0.56 b | 2.29 ± 0.04 |
| *** | *** | *** | ns | |
| Y × F | ||||
| 2012 × control | 60.54 ± 0.33 | 14.71 ± 0.94 | 22.51 ± 1.24 | 2.24 ± 0.03 |
| 2012 × CD | 64.41 ± 0.30 | 19.45 ± 0.42 | 13.87 ± 0.66 | 2.27 ± 0.04 |
| 2012 × B | 65.44 ± 0.54 | 15.19 ± 0.19 | 17.05 ± 0.76 | 2.32 ± 0.06 |
| 2013 × control | 59.81 ± 0.29 | 14.84 ± 0.11 | 23.38 ± 0.25 | 1.97 ± 0.10 |
| 2013 × CD | 63.18 ± 0.53 | 19.49 ± 0.47 | 15.30 ± 0.76 | 2.03 ± 0.06 |
| 2013 × B | 63.62 ± 0.20 | 15.10 ± 0.24 | 19.03 ± 0.06 | 2.25 ± 0.05 |
| ns | ns | ns | ns |
| Source of Variance | Free Acidity | Peroxide Value | K232 | K270 |
|---|---|---|---|---|
| (% Oleic Acid) | ||||
| Year (Y) | ||||
| 2012 | 1.05 ± 0.01 | 2.31 ± 0.56 | 1.61 ± 0.04 | 0.36 ± 0.05 |
| 2013 | 0.42 ± 0.06 | 7.04 ± 0.21 | 1.83 ± 0.10 | 0.87 ± 0.08 |
| t-test | *** | *** | ns | *** |
| Fertilization (F) | ||||
| Control | 0.87 ± 0.09 a | 5.99 ± 0.73 a | 1.93 ± 0.09 a | 0.83 ± 0.12 a |
| CD | 0.66 ± 0.17 b | 3.55 ± 1.36 c | 1.48 ± 0.02 c | 0.40 ± 0.08 c |
| B | 0.69 ± 0.16 b | 4.48 ± 1.11 b | 1.74 ± 0.08 b | 0.61 ± 0.15 b |
| *** | *** | *** | *** | |
| Y × F | ||||
| 2012 × control | 1.07 ± 0.01 a | 4.37 ± 0.03 c | 1.76 ± 0.05 bc | 0.56 ± 0.01 c |
| 2012 × CD | 1.04 ± 0.01 a | 0.53 ± 0.03 e | 1.50 ± 0.01 d | 0.22 ± 0.02 d |
| 2012 × B | 1.05 ± 0.01 a | 2.03 ± 0.02 d | 1.57 ± 0.01 cd | 0.29 ± 0.01 d |
| 2013 × control | 0.66 ± 0.01 b | 7.62 ± 0.07 a | 2.11 ± 0.06 a | 1.10 ± 0.01 a |
| 2013 × CD | 0.28 ± 0.01 d | 6.57 ± 0.31 b | 1.47 ± 0.03 d | 0.58 ± 0.03 c |
| 2013 × B | 0.33 ± 0.02 c | 6.93 ± 0.38 ab | 1.91 ± 0.05 b | 0.94 ± 0.03 b |
| *** | *** | *** | *** |
| Fatty Acids (%) | Year (Y) | Fertilization (F) | Y × F | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2012 | 2013 | t-Test | Control | CD | B | Sig. | 2012 × Control | 2012 × CD | 2012 × B | 2013 × Control | 2013 × CD | 2013 × B | Sig. | |
| C16:0 | 5.98 ± 0.02 | 5.85 ± 0.09 | ns | 6.10 ± 0.04 a | 5.80 ± 0.09 b | 5.83 ± 0.04 b | *** | 6.03 ± 0.01 b | 6.00 ± 0.05 b | 5.92 ± 0.04 b | 6.18 ± 0.03 a | 5.61 ± 0.00 d | 5.75 ± 0.01 c | *** |
| C16:1 | 0.18 ± 0.004 | 0.18 ± 0.008 | ns | 0.19 ± 0.008 a | 0.17 ± 0.004 b | 0.17 ± 0.007 b | ** | 0.18 ± 0.011 abc | 0.18 ± 0.007 abc | 0.18 ± 0.006 ab | 0.20 ± 0.002 a | 0.17 ± 0.002 bc | 0.15 ± 0.002 c | *** |
| C17:0 | 0.043 ± 0.001 | 0.037 ± 0.001 | ** | 0.042 ± 0.002 | 0.038 ± 0.002 | 0.039 ± 0.002 | ns | 0.043 ± 0.004 | 0.043 ± 0.001 | 0.042 ± 0.001 | 0.041 ± 0.000 | 0.033 ± 0.001 | 0.035 ± 0.002 | ns |
| C17:1 | 0.066 ± 0.001 | 0.057 ± 0.001 | *** | 0.063 ± 0.002 | 0.060 ± 0.003 | 0.060 ± 0.002 | ns | 0.067 ± 0.002 | 0.065 ± 0.003 | 0.065 ± 0.001 | 0.060 ± 0.000 | 0.055 ± 0.002 | 0.055 ± 0.001 | ns |
| C18:0 | 3.60 ± 0.05 | 2.65 ± 0.04 | *** | 3.21 ± 0.24 a | 3.08 ± 0.25 b | 3.10 ± 0.15 ab | * | 3.74 ± 0.07 a | 3.64 ± 0.01 a | 3.43 ± 0.05 b | 2.67 ± 0.05 cd | 2.52 ± 0.01 d | 2.77 ± 0.02 c | *** |
| C18:1 n9 c | 82.56 ± 0.06 | 83.48 ± 0.08 | *** | 82.80 ± 0.17 c | 83.03 ± 0.25 b | 83.23 ± 0.20 a | *** | 82.44 ± 0.07 d | 82.46 ± 0.04 d | 82.79 ± 0.05 c | 83.17 ± 0.05 b | 83.60 ± 0.02 a | 83.67 ± 0.00 a | ** |
| C18:1 n9 t | 0.019 ± 0.002 | 0.015 ± 0.001 | ns | 0.017 ± 0.003 | 0.017 ± 0.001 | 0.017 ± 0.001 | ns | 0.021 ± 0.005 | 0.017 ± 0.003 | 0.018 ± 0.000 | 0.013 ± 0.000 | 0.017 ± 0.000 | 0.016 ± 0.002 | ns |
| C18:2 n6 c | 7.08 ± 0.02 | 7.31 ± 0.07 | ** | 7.12 ± 0.05 b | 7.35 ± 0.10 a | 7.11 ± 0.01 b | *** | 7.02 ± 0.01 d | 7.13 ± 0.02 c | 7.10 ± 0.01 cd | 7.22 ± 0.03 b | 7.58 ± 0.00 a | 7.13 ± 0.02 c | *** |
| C18:3 n3 | 0.11 ± 0.001 | 0.12 ± 0.003 | * | 0.12 ± 0.004 b | 0.12 ± 0.003 a | 0.11 ± 0.002 c | *** | 0.11 ± 0.001 c | 0.12 ± 0.002 b | 0.12 ± 0.002 b | 0.13 ± 0.001 a | 0.13 ± 0.001 a | 0.11 ± 0.001 c | *** |
| C18:3 n6 | 0.15 ± 0.002 | 0.11 ± 0.002 | *** | 0.13 ± 0.007 a | 0.13 ± 0.012 ab | 0.12 ± 0.008 b | * | 0.15 ± 0.003 a | 0.15 ± 0.004 a | 0.14 ± 0.001 a | 0.12 ± 0.000 b | 0.10 ± 0.003 c | 0.11 ± 0.003 bc | ** |
| C20:1 | 0.12 ± 0.003 | 0.14 ± 0.004 | *** | 0.14 ± 0.003 | 0.13 ± 0.005 | 0.13 ± 0.010 | ns | 0.13 ± 0.005 bcd | 0.12 ± 0.004 cd | 0.11 ± 0.001 d | 0.14 ± 0.000 ab | 0.14 ± 0.003 bc | 0.16 ± 0.007 a | ** |
| C20:3 n6 | 0.030 ± 0.001 | 0.026 ± 0.001 | *** | 0.030 ± 0.001 b | 0.027 ± 0.002 b | 0.028 ± 0.001 a | *** | 0.032 ± 0.001 a | 0.031 ± 0.001 ab | 0.029 ± 0.000 abc | 0.028 ± 0.000 bc | 0.023 ± 0.001 d | 0.026 ± 0.000 c | *** |
| C23:0 | 0.049 ± 0.002 | 0.027 ± 0.000 | *** | 0.037 ± 0.004 | 0.040 ± 0.006 | 0.038 ± 0.005 | ns | 0.045 ± 0.002 a | 0.052 ± 0.003 a | 0.050 ± 0.001 a | 0.028 ± 0.001 b | 0.027 ± 0.000 b | 0.026 ± 0.001 b | * |
| SFA | 9.68 ± 0.07 | 8.56 ± 0.11 | *** | 9.39 ± 0.21 a | 8.96 ± 0.34 b | 9.01 ± 0.19 b | *** | 9.86 ± 0.07 a | 9.73 ± 0.05 a | 9.44 ± 0.05 b | 8.92 ± 0.07 c | 8.19 ± 0.01 e | 8.58 ± 0.03 d | *** |
| UFA | 90.32 ± 0.07 | 91.44 ± 0.11 | *** | 90.61 ± 0.21 b | 91.04 ± 0.34 a | 90.99 ± 0.19 a | *** | 90.14 ± 0.07 e | 90.27 ± 0.05 e | 90.56 ± 0.05 d | 91.08 ± 0.07 c | 91.81 ± 0.01 a | 91.42 ± 0.03 b | *** |
| MUFA | 82.95 ± 0.06 | 83.87 ± 0.07 | *** | 83.21 ± 0.17 c | 83.41 ± 0.25 b | 83.61 ± 0.20 a | *** | 82.83 ± 0.08 d | 82.84 ± 0.04 d | 83.17 ± 0.05 c | 83.59 ± 0.05 b | 83.97 ± 0.02 a | 84.05 ± 0.01 a | ** |
| PUFA | 7.37 ± 0.02 | 7.57 ± 0.07 | * | 7.40 ± 0.04 b | 7.63 ± 0.09 a | 7.38 ± 0.01 b | *** | 7.31 ± 0.01 d | 7.43 ± 0.02 bc | 7.39 ± 0.01 cd | 7.49 ± 0.03 b | 7.83 ± 0.01 a | 7.37 ± 0.02 cd | *** |
| Omega 3 | 0.11 ± 0.001 | 0.12 ± 0.003 | * | 0.12 ± 0.004 b | 0.12 ± 0.003 a | 0.11 ± 0.002 c | *** | 0.11 ± 0.001 c | 0.12 ± 0.002 b | 0.12 ± 0.002 b | 0.13 ± 0.001 a | 0.13 ± 0.001 a | 0.11 ± 0.001 c | *** |
| Omega 6 | 7.26 ± 0.02 | 7.44 ± 0.07 | * | 7.28 ± 0.04 b | 7.51 ± 0.09 a | 7.26 ± 0.01 b | *** | 7.20 ± 0.01 d | 7.31 ± 0.02 bc | 7.27 ± 0.01 cd | 7.36 ± 0.03 b | 7.70 ± 0.01 a | 7.26 ± 0.02 cd | *** |
| Description | Units | Value |
|---|---|---|
| Sand, coarse | % | 24 |
| Sand, fine | % | 11 |
| Silt | % | 34 |
| Clay | % | 31 |
| pH | - | 5.23 |
| Electric conductivity | mS/cm | 0.104 |
| Organic carbon | % | 1.36 |
| Organic matter | % | 2.34 |
| Total nitrogen | % | 0.15 |
| NO3- | ppm | 20.00 |
| NH3 | ppm | 21.00 |
| P2O5 | ppm | 61.83 |
| K2O | ppm | 490.20 |
| Boron | ppm | 1.1 |
| Iron | ppm | 32 |
| Manganese | ppm | 0.7 |
| Copper | ppm | 2.4 |
| Zinc | ppm | 0.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pannico, A.; Modarelli, G.C.; Stazi, S.R.; Giaccone, M.; Romano, R.; Rouphael, Y.; Cirillo, C. Foliar Nutrition Influences Yield, Nut Quality and Kernel Composition in Hazelnut cv Mortarella. Plants 2023, 12, 2219. https://doi.org/10.3390/plants12112219
Pannico A, Modarelli GC, Stazi SR, Giaccone M, Romano R, Rouphael Y, Cirillo C. Foliar Nutrition Influences Yield, Nut Quality and Kernel Composition in Hazelnut cv Mortarella. Plants. 2023; 12(11):2219. https://doi.org/10.3390/plants12112219
Chicago/Turabian StylePannico, Antonio, Giuseppe Carlo Modarelli, Silvia Rita Stazi, Matteo Giaccone, Raffaele Romano, Youssef Rouphael, and Chiara Cirillo. 2023. "Foliar Nutrition Influences Yield, Nut Quality and Kernel Composition in Hazelnut cv Mortarella" Plants 12, no. 11: 2219. https://doi.org/10.3390/plants12112219
APA StylePannico, A., Modarelli, G. C., Stazi, S. R., Giaccone, M., Romano, R., Rouphael, Y., & Cirillo, C. (2023). Foliar Nutrition Influences Yield, Nut Quality and Kernel Composition in Hazelnut cv Mortarella. Plants, 12(11), 2219. https://doi.org/10.3390/plants12112219









