Comparative Study on the Phenolic Fingerprint and Antioxidant Activity of Strawberry Tree (Arbutus unedo L.) Leaves and Fruits
Abstract
:1. Introduction
2. Results and Discussion
2.1. Qualitative Phenolic Profile of A. unedo Leaves and Fruits
2.1.1. Phenolic Acids and Their Derivatives
2.1.2. Flavonoids and Their Derivatives
2.1.3. Other Phenolic Compounds
2.2. Quantitative Phenolic Profile of A. unedo Leaves and Fruit
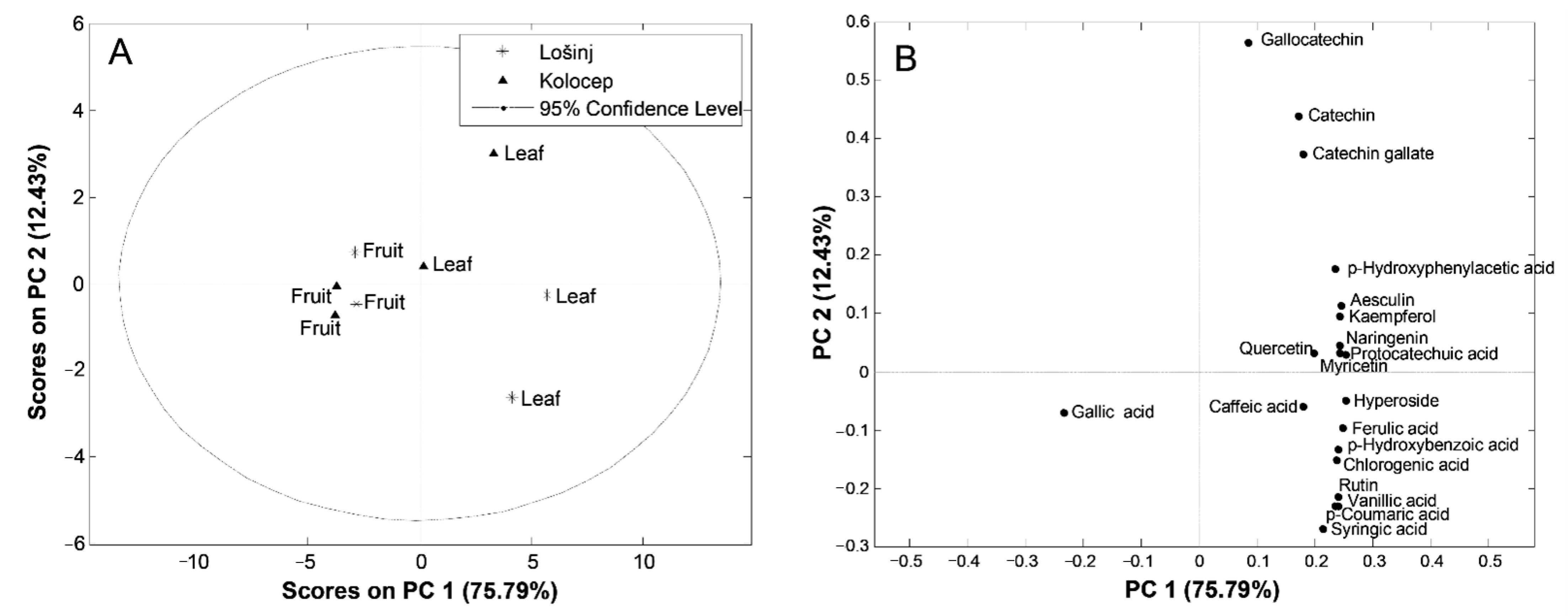
Principal Component Analysis
2.3. Determination of TPC and RSA
2.4. Strengths and Limitations of the Study
3. Materials and Methods
3.1. Chemicals and Materials
3.2. Samples
3.3. Preparation of the Extracts
3.4. UHPLC-LTQ OrbiTrap MS Analysis of Phenolic Compounds
3.5. Total Phenolic Content (TPC)
3.6. DPPH Radical Scavenging Activity (RSA)
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Díaz-Guerra, L.; Llorens, L.; Julkunen-Tiitto, R.; Nogués, I.; Font, J.; González, J.A.; Verdaguer, D. Leaf biochemical adjustments in two Mediterranean resprouter species facing enhanced UV levels and reduced water availability before and after aerial biomass removal. Plant Physiol. Biochem. 2019, 137, 130–143. [Google Scholar] [CrossRef]
- Jurica, K.; Brčić Karačonji, I.; Mikolić, A.; Milojković-Opsenica, D.; Benković, V.; Kopjar, N. In vitro safety assessment of the strawberry tree (Arbutus unedo L.) water leaf extract and arbutin in human peripheral blood lymphocytes. Cytotechnology 2018, 70, 1261–1278. [Google Scholar] [CrossRef] [PubMed]
- Tenuta, M.C.; Tundis, R.; Xiao, J.; Loizzo, M.R.; Dugay, A.; Deguin, B. Arbutus species (Ericaceae) as source of valuable bioactive products. Crit. Rev. Food Sci. Nutr. 2019, 59, 864–881. [Google Scholar] [CrossRef] [PubMed]
- Morgado, S.; Morgado, M.; Plácido, A.I.; Roque, F.; Duarte, A.P. Arbutus unedo L.: From traditional medicine to potential uses in modern pharmacotherapy. J. Ethnopharmacol. 2018, 225, 90–102. [Google Scholar] [CrossRef]
- Dib, M.E.A.; Allali, H.; Bendiabdellah, A.; Meliani, N.; Tabti, B. Antimicrobial activity and phytochemical screening of Arbutus unedo L. J. Saudi Chem. Soc. 2013, 17, 381–385. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, I.; Baptista, P.; Bento, A.; Pereira, J.A. Arbutus unedo L. and its benefits on human health. J. Food Nutr. Res. 2011, 50, 73–85. [Google Scholar]
- Pallauf, K.; Rivas-Gonzalo, J.C.; del Castillo, M.D.; Cano, M.P.; de Pascual-Teresa, S. Characterization of the antioxidant composition of strawberry tree (Arbutus unedo L.) fruits. J. Food Compos. Anal. 2008, 21, 273–281. [Google Scholar] [CrossRef] [Green Version]
- Fiorentino, A.; Castaldi, S.; D’Abrosca, B.; Natale, A.; Carfora, A.; Messere, A.; Monaco, P. Polyphenols from the hydroalcoholic extract of Arbutus unedo living in a monospecific Mediterranean woodland. Biochem. Syst. Ecol. 2007, 35, 809–811. [Google Scholar] [CrossRef]
- Maleš, Ž.; Šarić, D.; Bojić, M. Quantitative determination of flavonoids and chlorogenic acid in the leaves of Arbutus unedo L. using thin layer chromatography. J. Anal. Methods Chem. 2013, 2013, 8–9. [Google Scholar] [CrossRef] [Green Version]
- Mendes, L.; de Freitas, V.; Baptista, P.; Carvalho, M. Comparative antihemolytic and radical scavenging activities of strawberry tree (Arbutus unedo L.) leaf and fruit. Food Chem. Toxicol. 2011, 49, 2285–2291. [Google Scholar] [CrossRef]
- Tavares, L.; Fortalezas, S.; Carrilho, C.; McDougall, G.J.; Stewart, D.; Ferreira, R.B.; Santos, C.N. Antioxidant and antiproliferative properties of strawberry tree tissues. J. Berry Res. 2010, 1, 3–12. [Google Scholar] [CrossRef] [Green Version]
- Coimbra, A.T.; Luís, Â.F.S.; Batista, M.T.; Ferreira, S.M.P.; Duarte, A.P.C. Phytochemical characterization, bioactivities evaluation and synergistic effect of Arbutus unedo and Crataegus monogyna extracts with amphotericin B. Curr. Microbiol. 2020, 77, 2143–2154. [Google Scholar] [CrossRef] [PubMed]
- Maldini, M.; D’Urso, G.; Pagliuca, G.; Petretto, G.L.; Foddai, M.; Gallo, F.R.; Multari, G.; Caruso, D.; Montoro, P.; Pintore, G. HPTLC-PCA complementary to HRMS-PCA in the case study of Arbutus unedo antioxidant phenolic profiling. Foods 2019, 8, 294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayaz, F.A.; Kucukislamoglu, M.; Reunanen, M. Sugar, non-volatile and phenolic acids composition of strawberry tree (Arbutus unedo L. var. ellipsoidea) fruits. J. Food Compos. Anal. 2000, 13, 171–177. [Google Scholar] [CrossRef]
- Fortalezas, S.; Tavares, L.; Pimpão, R.; Tyagi, M.; Pontes, V.; Alves, P.M.; McDougall, G.; Stewart, D.; Ferreira, R.B.; Santos, C.N. Antioxidant properties and neuroprotective capacity of strawberry tree fruit (Arbutus unedo). Nutrients 2010, 2, 214–229. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, R.; Barros, L.; Dueñas, M.; Carvalho, A.M.; Queiroz, M.J.R.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Characterisation of phenolic compounds in wild fruits from Northeastern Portugal. Food Chem. 2013, 141, 3721–3730. [Google Scholar] [CrossRef] [Green Version]
- Pawlowska, A.M.; de Leo, M.; Braca, A. Phenolics of Arbutus unedo L. (Ericaceae) fruits: Identification of anthocyanins and gallic acid derivatives. J. Agric. Food Chem. 2006, 54, 10234–10238. [Google Scholar] [CrossRef]
- Mosele, J.I.; Macià, A.; Romero, M.P.; Motilva, M.J. Stability and metabolism of Arbutus unedo bioactive compounds (phenolics and antioxidants) under in vitro digestion and colonic fermentation. Food Chem. 2016, 201, 120–130. [Google Scholar] [CrossRef] [PubMed]
- El Cadi, H.; El Cadi, A.; Kounnoun, A.; Oulad El Majdoub, Y.; Palma Lovillo, M.; Brigui, J.; Dugo, P.; Mondello, L.; Cacciola, F. Wild strawberry (Arbutus unedo): Phytochemical screening and antioxidant properties of fruits collected in northern Morocco. Arab. J. Chem. 2020, 13, 6299–6311. [Google Scholar] [CrossRef]
- Zitouni, H.; Hssaini, L.; Zerhoune, M.; Ourradi, H.; Viuda-Martos, M.; Hernández, F.; Ercisli, S.; Hanine, H. Phytochemical components and bioactivity assessment among twelve strawberry (Arbutus unedo L.) genotypes growing in Morocco using chemometrics. Foods 2020, 9, 1345. [Google Scholar] [CrossRef]
- Salem, I.B.; Ouesleti, S.; Mabrouk, Y.; Landolsi, A.; Saidi, M.; Boulilla, A. Exploring the nutraceutical potential and biological activities of Arbutus unedo L. (Ericaceae) fruits. Ind. Crops Prod. 2018, 122, 726–731. [Google Scholar] [CrossRef]
- Macchioni, V.; Santarelli, V.; Carbone, K. Phytochemical profile, antiradical capacity and α-glucosidase inhibitory potential of wild Arbutus unedo L. fruits from Central Italy: A chemometric approach. Plants 2020, 9, 1785. [Google Scholar] [CrossRef]
- Tenuta, M.C.; Deguin, B.; Loizzo, M.R.; Dugay, A.; Acquaviva, R.; Malfa, G.A.; Bonesi, M.; Bouzidi, C.; Tundis, R. Contribution of flavonoids and iridoids to the hypoglycaemic, antioxidant, and nitric oxide (NO) inhibitory activities of Arbutus unedo L. Antioxidants 2020, 9, 184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malheiro, R.; Sá, O.; Pereira, E.; Aguiar, C.; Baptista, P.; Pereira, J.A. Arbutus unedo L. leaves as source of phytochemicals with bioactive properties. Ind. Crops Prod. 2012, 37, 473–478. [Google Scholar] [CrossRef]
- de Ancos, B.; Gonzalez, E.M.; Cano, M.P. Ellagic acid, vitamin C, and total phenolic contents and radical scavenging capacity affected by freezing and frozen storage in raspberry fruit. J. Agric. Food Chem. 2000, 48, 4565–4570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Males, Z.; Plazibat, M.; Vundać, V.B.; Zuntar, I. Qualitative and quantitative analysis of flavonoids of the strawberry tree—Arbutus unedo L. (Ericaceae). Acta Pharm. 2006, 56, 245–250. [Google Scholar] [PubMed]
- Jardim, C.; Macedo, D.; Figueira, I.; Dobson, G.; McDougall, G.; Stewart, D.; Ferreira, R.; Menezes, R.; Santos, C. (Poly)phenol metabolites from Arbutus unedo leaves protect yeast from oxidative injury by activation of antioxidant and protein clearance pathways. J. Funct. Foods 2017, 32, 332–346. [Google Scholar] [CrossRef]
- Kachlicki, P.; Piasecka, A.; Stobiecki, M.; Marczak, Ł. Structural characterization of flavonoid glycoconjugates and their derivatives with mass spectrometric techniques. Molecules 2016, 21, 1494. [Google Scholar] [CrossRef] [Green Version]
- Jurica, K.; Brčić Karačonji, I.; Šegan, S.; Milojković Opsenica, D.; Kremer, D. Quantitative analysis of arbutin and hydroquinone in strawberry tree (Arbutus unedo L., Ericaceae) leaves by gas chromatography-mass spectrometry. Arh. Hig. Rada Toksikol. 2015, 66, 197–202. [Google Scholar] [CrossRef] [Green Version]
- Sagbas, H.I.; Ilhan, G.; Zitouni, H.; Anjum, M.A.; Hanine, H.; Necas, T.; Ondrasek, I.; Ercisli, S. Morphological and biochemical characterization of diverse strawberry tree (Arbutus unedo L.) genotypes from northern Turkey. Agronomy 2020, 10, 1581. [Google Scholar] [CrossRef]
- Dib, M.A.; Paolini, J.; Bendahou, M.; Varesi, L.; Allali, H.; Desjobert, J.-M.; Tabti, B.; Costa, J. Chemical composition of fatty acid and unsaponifiable fractions of leaves, stems and roots of Arbutus unedo and in vitro antimicrobial activity of unsaponifiable extracts. Nat. Prod. Commun. 2010, 5, 1085–1090. [Google Scholar] [CrossRef]
- Jurica, K.; Brčić Karačonji, I.; Kopjar, N.; Shek-Vugrovečki, A.; Cikač, T.; Benković, V. The effects of strawberry tree water leaf extract, arbutin and hydroquinone on haematological parameters and levels of primary DNA damage in white blood cells of rats. J. Ethnopharmacol. 2018, 215, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Fiuza, S.M.; Gomes, C.; Teixeira, L.J.; Girão da Cruz, M.T.; Cordeiro, M.N.D.S.; Milhazes, N.; Borges, F.; Marques, M.P.M. Phenolic acid derivatives with potential anticancer properties—A structure-activity relationship study. Part 1: Methyl, propyl and octyl esters of caffeic and gallic acids. Bioorg. Med. Chem. 2004, 12, 3581–3589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choubey, S.; Varughese, L.R.; Kumar, V.; Beniwal, V. Medicinal importance of gallic acid and its ester derivatives: A patent review. Pharm. Pat. Anal. 2015, 4, 305–315. [Google Scholar] [CrossRef]
- Locatelli, C.; Leal, P.C.; Yunes, R.A.; Nunes, R.J.; Creczynski-Pasa, T.B. Gallic acid ester derivatives induce apoptosis and cell adhesion inhibition in melanoma cells: The relationship between free radical generation, glutathione depletion and cell death. Chem. Biol. Interact. 2009, 181, 175–184. [Google Scholar] [CrossRef]
- Oliveira, I.; Coelho, V.; Baltasar, R.; Pereira, J.A.; Baptista, P. Scavenging capacity of strawberry tree (Arbutus unedo L.) leaves on free radicals. Food Chem. Toxicol. 2009, 47, 1507–1511. [Google Scholar] [CrossRef]
- Bouzid, K.; Toumi Benali, F.; Chadli, R.; Bouzouina, M.; Bouzid, A.; Benchohra, A.; Mahmoud Dif, M. Extraction, identification and quantitative HPLC analysis of flavonoids from fruit extracts of Arbutus unedo L from Tiaret Area (Western Algeria). Eur. J. Mol. Biotechnol. 2014, 4, 160–169. [Google Scholar] [CrossRef]
- Guendouze-Bouchefa, N.; Madani, K.; Chibane, M.; Boulekbache-Makhlouf, L.; Hauchard, D.; Kiendrebeogo, M.; Stévigny, C.; Ndjolo, P.; Duez, P. Phenolic compounds, antioxidant and antibacterial activities of three Ericaceae from Algeria. Ind. Crop. Prod. 2015, 70, 459–466. [Google Scholar] [CrossRef] [Green Version]
- Orak, H.H.; Yagar, H.; Isbilir, S.S.; Demirci, A.Ş.; Gümüş, T.; Ekinci, N. Evaluation of antioxidant and antimicrobial potential of strawberry tree (Arbutus unedo L.) leaf. Food Sci. Biotechnol. 2011, 20, 1249–1256. [Google Scholar] [CrossRef]
- Samuel, A.; Brejea, R.; Domuta, C.; Bungau, S.; Cenusa, N.; Tit, D. Enzymatic indicators of soil quality. J. Environ. Prot. Ecol. 2017, 18, 871–878. [Google Scholar]
- Bungau, S.; Behl, T.; Aleya, L.; Bourgeade, P.; Aloui-Sossé, B.; Purza, A.L.; Abid, A.; Samuel, A.D. Expatiating the impact of anthropogenic aspects and climatic factors on long-term soil monitoring and management. Environ. Sci. Pollut. Res. 2021, 28, 30528–30550. [Google Scholar] [CrossRef]
- Samuel, A.D.; Tit, D.M.; Melinte, C.E.; Iovan, C.; Purza, L.; Gitea, M.; Bungau, S. Enzymological and physicochemical evaluation of the effects of soil management practices. Rev. Chim. 2017, 68, 2243–2247. [Google Scholar] [CrossRef]
- Alarcão-E-Silva, M.L.C.M.M.; Leitão, A.E.B.; Azinheira, H.G.; Leitão, M.C.A. The arbutus berry: Studies on its color and chemical characteristics at two mature stages. J. Food Compos. Anal. 2001, 14, 27–35. [Google Scholar] [CrossRef]
- Barros, L.; Carvalho, A.M.; Morais, J.S.; Ferreira, I.C.F.R. Strawberry-tree, blackthorn and rose fruits: Detailed characterisation in nutrients and phytochemicals with antioxidant properties. Food Chem. 2010, 120, 247–254. [Google Scholar] [CrossRef]
- Isbilir, S.S.; Orak, H.H.; Yagar, H.; Ekinci, N. Determination of antioxidant activities of strawberry tree (Arbutus unedo L.) flowers and fruits at different ripening stages. Acta Sci. Pol. Hortorum Cultus 2012, 11, 223–237. [Google Scholar]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for analysis of plant phenolic compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef]
- Glevitzky, I.; Dumitrel, G.A.; Glevitzky, M.; Pasca, B.; Otrisal, P.; Bungau, S.; Cioca, G.; Pantis, C.; Popa, M. Statistical analysis of the relationship between antioxidant activity and the structure of flavonoid compounds. Rev. Chim. 2019, 70, 3103–3107. [Google Scholar] [CrossRef]
- Jurica, K.; Gobin, I.; Kremer, D.; Vitali Čepo, D.; Jurišić Grubešić, R.; Brčić Karačonji, I.; Kosalec, I. Arbutin and its metabolite hydroquinone as the main factors in the antimicrobial effect of strawberry tree (Arbutus unedo L.) leaves. J. Herb. Med. 2017, 8, 17–23. [Google Scholar] [CrossRef]
- Kečkeš, S.; Gašić, U.; Veličković, T.Ć.; Milojković-Opsenica, D.; Natić, M.; Tešić, Ž. The determination of phenolic profiles of Serbian unifloral honeys using ultra-high-performance liquid chromatography/high resolution accurate mass spectrometry. Food Chem. 2013, 138, 32–40. [Google Scholar] [CrossRef]
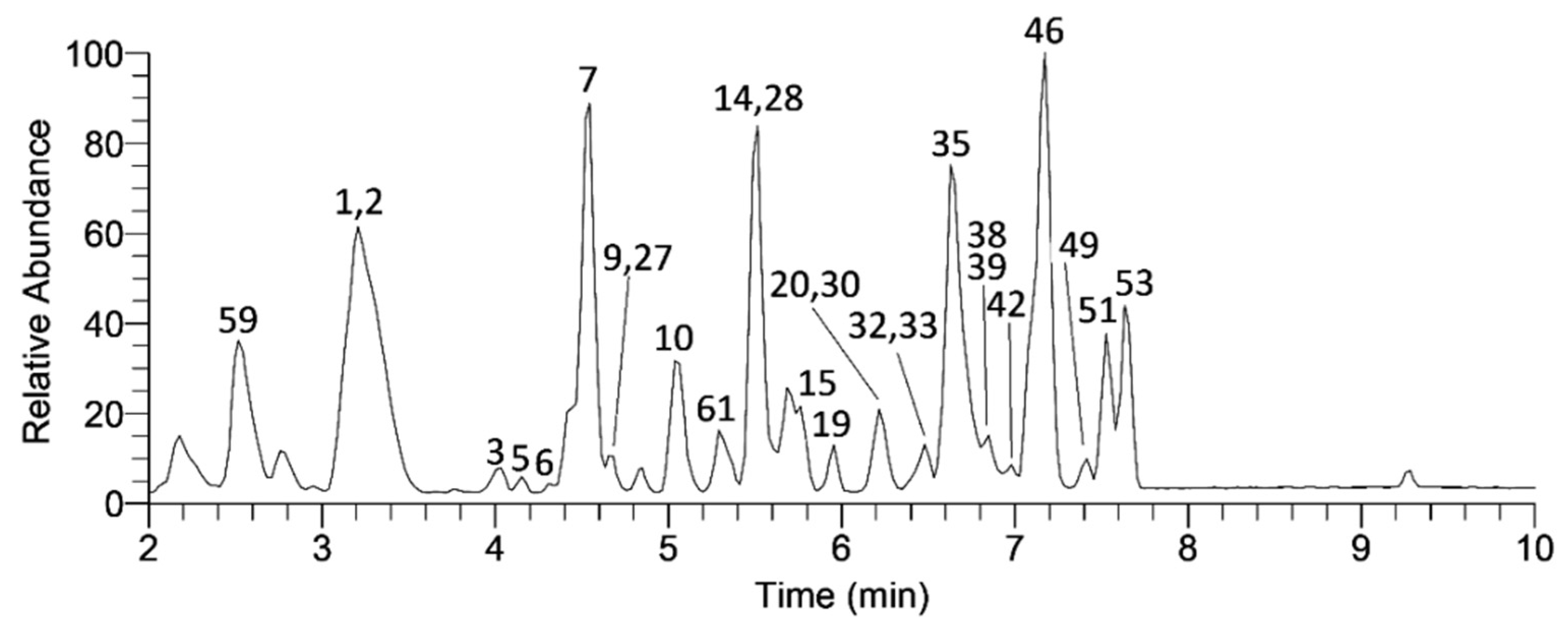
| No. | Compound Name a | tR (min) | Molecular Formula [M–H]– | Calculated Mass [M–H]– | Exact Mass [M–H]– | Δ ppm | MS2 Fragments (% Base Peak) | MS3 Fragments (% Base Peak) | MS4 Fragments (% Base Peak) |
|---|---|---|---|---|---|---|---|---|---|
| Phenolic acids and their derivatives | |||||||||
| 1 | Gallic acid F | 3.13 | C7H5O5− | 169.01425 | 169.01350 | 4.44 | 125(100) | 107(100) | - |
| 2 | Galloylquinic acid L,F | 3.21 | C14H15O10− | 343.06707 | 343.06540 | 4.87 | 191(100), 169(10) | 173(80), 127(100), 85(90) | 85(100) |
| 3 | Hydroxybenzoic acid hexoside isomer 1 L,F | 4.03 | C13H15O8− | 299.07724 | 299.07579 | 4.85 | 137(100) | 93(100) | - |
| 4 | Dihydroxybenzoic acid hexoside L,F | 4.10 | C13H15O9− | 315.07216 | 315.07065 | 4.79 | 153(100), 152(50), 109(15), 108(10) | 109(100) | 84(100), 81(60) |
| 5 | Gallic acid dihexoside L | 4.15 | C19H25O15− | 493.11989 | 493.11829 | 3.24 | 433(100), 331(20), 313(80), 283(30), 169(20) | 323(100), 161(10) | 179(20), 161(100), 143(20) |
| 6 | Gallic acid hexoside L,F | 4.31 | C13H15O10− | 331.06707 | 331.06540 | 5.04 | 169(100), 125(5) | 125(100) | 107(100), 83(10), 65(5) |
| 7 | Galloylshikimic acid L,F | 4.55 | C14H13O9− | 325.05651 | 325.05497 | 4.74 | 169(100), 125(15) | 125(100) | 107(100), 97(50), 81(40), 79(10), 69(5) |
| 8 | Protocatechuic acid L | 4.68 | C7H5O4− | 153.01933 | 153.01868 | 4.25 | 109(100), 107(20), 95(5) | 81(50), 79(100) | - |
| 9 | Hydroxybenzoic acid hexoside isomer 2 L,F | 4.70 | C13H15O8− | 299.07724 | 299.07590 | 4.48 | 137(100) | 93(100) | - |
| 10 | Digalloylquinic acid L,F | 5.04 | C21H19O14− | 495.07803 | 495.07611 | 3.88 | 343(100), 325(10), 301(10), 191(15), 169(5) | 191(100), 169(20) | 173(70), 127(90), 85(100) |
| 11 | Digalloylshikimic acid L,F | 5.22 | C21H17O13− | 477.06746 | 477.06525 | 4.63 | 417(10), 325(100) | 169(100), 125(15) | 125(100) |
| 12 | Coumaric acid hexoside L | 5.35 | C15H17O8− | 325.09289 | 325.09164 | 3.85 | 163(100), 119(10) | 119(100) | - |
| 13 | 5-O-Caffeoylquinic acid (Chlorogenic acid) L | 5.38 | C16H17O9− | 353.08781 | 353.08688 | 2.63 | 191(100), 179(5) | 173(75), 127(100), 111(40), 93(60), 85(90) | 109(30), 99(60), 85(100) |
| 14 | p- Hydroxybenzoic acid L,F | 5.51 | C7H5O3− | 137.02442 | 137.02390 | 3.79 | 109(10), 93(100) | 93(100) | - |
| 15 | Caffeic acidL,F | 5.73 | C9H7O4− | 179.03498 | 179.03413 | 4.75 | 135(100), 119(15), 117(10), 91(20), 59(15) | 107(30), 91(100), 89(80), 59(10) | - |
| 16 | Ellagic acid hexoside L,F | 5.84 | C20H15O13− | 463.05181 | 463.05075 | 2.29 | 302(15), 301(100), 300(50), 289(10), 273(10) | 301(70), 284(20), 257(100), 229(65), 185(30) | 229(50), 213(30), 185(100) |
| 17 | Syringic acid L,F | 5.85 | C9H9O5− | 197.04555 | 197.04462 | 4,72 | 182(100), 153(50), 138(10) | 167(100), 138(10), 123(5) | 123(100) |
| 18 | Vanillic acid L,F | 5.91 | C8H7O4− | 167.03498 | 167.03444 | 3.23 | 153(10), 152(80), 124(10), 123(100), 108(20) | 108(100) | 123(30), 80(35), 78(100) |
| 19 | Trigalloylquinic acid L | 5.93 | C28H23O18− | 647.08899 | 647.08722 | 2.74 | 495(100), 343(5) | 343(100), 325(5), 191(15) | 191(100), 169(15) |
| 20 | Trigalloylshikimic acid L | 6.22 | C28H21O17− | 629.07842 | 629.07654 | 2.99 | 477(100), 325(5) | 325(100) | 169(100), 125(15) |
| 21 | Ellagic acid pentoside L,F | 6.30 | C19H13O12− | 433.04125 | 433.04037 | 2.03 | 302(15), 301(100), 300(60) | 301(60), 300(40), 284(25), 257(100), 229(70) | 240(10), 229(100), 213(30), 201(20), 185(90) |
| 22 | p-Hydroxyphenylacetic acid L,F | 6.57 | C8H7O3− | 151.04007 | 151.03957 | 3.31 | 136(100), 95(5) | 108(25), 92(100) | 108(100) |
| 23 | p-Coumaric acid L,F | 6.69 | C9H7O3− | 163.04007 | 163.03944 | 3.86 | 149(40), 119(100) | 104(60), 77(100) | - |
| 24 | Ellagic acid L,F | 6.77 | C14H5O8− | 300.99899 | 300.99750 | 4.95 | 284(40), 271(60), 257(100), 229(85), 185(40) | 229(100), 213(20), 185(85) | 201(100), 185(95), 157(30), 145(20), 129(10) |
| 25 | Ferulic acid L,F | 6.88 | C10H9O4− | 193.05063 | 193.04982 | 4.20 | 178(70), 149(100), 134(50) | 134(100) | 106(100) |
| 26 | Cinnamic acid L | 8.84 | C9H7O2− | 147.04515 | 147.04451 | 4.35 | 104(10), 103(100), 87(10) | 119(100) | - |
| Flavonoids and their derivatives | |||||||||
| 27 | GallocatechinL,F | 4.63 | C15H13O7− | 305.06668 | 305.06537 | 4.29 | 261(50), 221(70), 219(70), 179(100), 165(35) | 164(100), 151(40), 135(30) | 120(100), 108(20) |
| 28 | Catechin L,F | 5.50 | C15H13O6− | 289.07176 | 289.07053 | 4.25 | 271(5), 245(100), 205(40), 179(15), 125(5) | 227(30), 203(100), 187(25), 175(10), 161(20) | 188(70), 185(20), 175(100), 161(40), 157(10) |
| 29 | Myricetin 3-O-(6″-rhamnosyl)glucoside L | 6.09 | C27H29O17− | 625.14102 | 625.13920 | 2.91 | 607(15), 317(80), 316(100), 287(5), 271(15) | 287(40), 271(100), 179(30) | 271(15), 243(100) |
| 30 | Myricetin 3-O-hexoside L,F | 6.24 | C21H19O13− | 479.08311 | 479.08087 | 4.68 | 463(30), 455(10), 317(80), 316(100) | 287(30), 271(100), 179(40) | 271(15), 243(100), 227(30) |
| 31 | Myricetin 3-O-pentoside L,F | 6.40 | C20H17O12− | 449.07255 | 449.07123 | 2.94 | 318(10), 317(100), 316(80), 315(20) | 272(25), 179(100), 151(40) | 151(100) |
| 32 | Quercetin 3-O-(6″-rhamnosyl)glucoside (Rutin) L,F | 6.48 | C27H29O16− | 609.14611 | 609.14392 | 3.60 | 343(5), 301(100), 300(30), 271(10), 255(5) | 273(25), 257(20), 179(100), 151(75) | 151(100) |
| 33 | Quercetin 3-O-(6″-galloyl)hexoside L,F | 6.49 | C28H23O16− | 615.09916 | 615.09662 | 4.13 | 463(100), 301(20) | 343(5), 301(100), 300(40) | 273(15), 257(30), 229(20), 179(100), 151(90) |
| 34 | Myricetin 7-O-pentoside L,F | 6.58 | C20H17O12− | 449.07255 | 449.07108 | 3.27 | 318(10), 317(100) | 272(25), 179(100), 151(40) | 151(100) |
| 35 | Myricetin 3-O-rhamnoside (Myricitrin) L | 6.65 | C21H19O12− | 463.08820 | 463.08661 | 3.43 | 317(50), 316(100) | 287(30), 271(100), 179(40) | 271(15), 243(100), 227(30) |
| 36 | Quercetin 3-O-galactoside (Hyperoside) F | 6.68 | C21H19O12− | 463.08820 | 463.08685 | 2.92 | 301(100), 300(30) | 273(25), 257(20), 179(100), 151(75) | 151(100) |
| 37 | Catechin 3-gallate L,F | 6.78 | C22H17O10− | 441.08272 | 441.08118 | 3.49 | 331(10), 289(100), 271(10), 169(25) | 271(5), 245(100), 205(40), 179(20) | 227(20), 203(100), 187(20), 175(10), 161(20) |
| 38 | Kaempferol 7-O-(6″-rhamnosyl)glucoside L,F | 6.85 | C27H29O15− | 593.15119 | 593.14850 | 4.54 | 285(100) | 267(50), 257(100), 241(40), 229(50), 213(30) | 255(20), 239(30), 229(100), 213(30), 163(60) |
| 39 | Myricetin 7-O-hexuronide L | 6.87 | C21H17O14− | 493.06238 | 493.06064 | 3.53 | 317(100) | 193(15), 179(100), 151(45) | 151(100) |
| 40 | MorinL | 6.91 | C15H9O7− | 301.03538 | 301.03403 | 4.48 | 286(100), 273(90), 257(15), 207(20) | 285(10), 268(100), 257(70), 240(40), 229(15) | 240(100), 212(10) |
| 41 | Quercetin 3-O-pentoside isomer 1 L,F | 6.93 | C20H17O11− | 433.07763 | 433.07608 | 3.58 | 301(100), 300(15) | 283(20), 273(25), 257(10), 179(100), 151(75) | 151(100) |
| 42 | Kaempferol 3-O-(6″-galloyl)hexoside L | 6.98 | C28H23O15− | 599.10424 | 599.10175 | 4.16 | 447(70), 313(100), 285(50), 284(25), 271(10) | 241(20), 211(10), 169(100), 151(5), 125(15) | 125(100) |
| 43 | Naringin L,F | 7.02 | C27H31O14− | 579.17193 | 579.16919 | 4.73 | 459(100), 357(5), 313(25), 271(45), 235(10) | 441(30), 357(100), 339(30), 271(55), 235(85) | 339(100), 169(20), 151(50), 125(20) |
| 44 | Kaempferol 3-O-glucoside (Astragalin) F | 7.05 | C21H19O11− | 447.09329 | 447.09183 | 3.27 | 327(20), 285(80), 284(100), 255(10) | 255(100), 227(10) | 227(100), 211(60) |
| 45 | Quercetin 3-O-pentoside isomer 2 L,F | 7.07 | C20H17O11− | 433.07763 | 433.07645 | 2.72 | 301(100), 300(15) | 283(20), 273(25), 257(10), 179(100), 151(75) | 151(100) |
| 46 | Quercetin 3-O-rhamnoside (Quercitrin) F | 7.17 | C21H19O11− | 447.09329 | 447.09113 | 4.83 | 301(100), 300(35), 284(20) | 273(25), 257(20), 179(100), 151(75) | 151(100) |
| 47 | MyricetinL | 7.26 | C15H9O8− | 317.03029 | 317.02930 | 3.12 | 299(10), 273(35), 207(100),163(95) | 179(100), 151(15) | 151(100) |
| 48 | Kaempferol 3-O-pentoside isomer 1 L,F | 7.31 | C20H17O10− | 417.08272 | 417.08115 | 3.76 | 327(10), 285(30), 284(100), 255(10) | 255(100), 227(15) | 255(15), 227(100), 211(80), 167(15) |
| 49 | Quercetin 3-O-hexuronide L,F | 7.40 | C21H17O13− | 477.06746 | 477.06552 | 4.07 | 302(15), 301(100), 299(10) | 273(20), 257(15), 179(100), 151(80) | 151(100) |
| 50 | Quercetin 3-O-(6″-p-hydroxybenzoyl)hexosideL | 7.43 | C28H23O14− | 583.10933 | 583.10754 | 3.07 | 463(100), 433(10), 301(50), 300(25), 271(10) | 301(100), 300(40) | 273(15), 257(30), 229(20), 179(100), 151(90) |
| 51 | Kaempferol 3-O-pentoside isomer 2 L | 7.53 | C20H17O10− | 417.08272 | 417.08142 | 3.12 | 327(5), 285(100), 284(70), 255(5) | 267(50), 257(100), 241(40), 229(60), 213(30) | 255(15), 239(40), 229(100), 213(30), 163(60) |
| 52 | Kaempferol 7-O-hexuronide L | 7.58 | C21H17O12− | 461.07255 | 461.07108 | 3.19 | 285(100) | 285(100), 284(60) | 267(40), 257(100), 229(50), 213(30), 163(20) |
| 53 | Kaempferol 3-O-rhamnoside L,F | 7.63 | C21H19O10− | 431.09837 | 431.09674 | 3.78 | 285(100), 284(20) | 267(50), 257(100), 241(40), 229(50), 213(30) | 255(20), 239(30), 229(100), 213(30), 163(60) |
| 54 | Quercetin 3-O-(6″-p-coumaroyl) hexoside L | 7.85 | C30H25O14− | 609.12498 | 609.12268 | 3.78 | 463(100), 301(20) | 301(100), 300(25) | 273(25), 257(20), 179(100) |
| 55 | Kaempferol 3-O-(6″-p-coumaroyl)hexoside L | 8.29 | C30H25O13− | 593.13006 | 593.12769 | 4.00 | 447(15), 307(10), 285(100) | 257(100), 241(50), 229(35), 213(40), 151(90) | 256(10), 239(25), 229(100), 213(20), 163(35) |
| 56 | Quercetin L,F | 8.62 | C15H9O7− | 301.03538 | 301.03391 | 4.88 | 271(50), 255(20), 179(100), 151(80), 107(5) | 151(100) | 107(100), 83(10) |
| 57 | Naringenin L | 9.35 | C15H11O5− | 271.06120 | 271.06030 | 3.32 | 225(5), 177(10), 151(100) | 107(100) | 65(100) |
| 58 | Kaempferol L | 9.51 | C15H9O6− | 285.04046 | 285.03909 | 4.81 | 255(100), 227(10) | 211(100), 195(5), 167(15) | 211(40), 137(100) |
| Other phenolic compounds | |||||||||
| 59 | Arbutin L,F | 2.52 | C12H15O7− | 271.08233 | 271.08148 | 3.14 | 193(5), 161(100), 113(10), 109(35), 101(5) | 143(10), 129(20), 113(50), 101(100), 71(50) | - |
| 60 | Aesculin L | 5.06 | C15H15O9− | 339.07216 | 339.07114 | 3.01 | 177(100) | 177(5), 149(10), 133(100), 105(10), 89(5) | 89(100) |
| 61 | Galloylarbutin L,F | 5.30 | C19H19O11− | 423.09329 | 423.09213 | 2.74 | 313(100), 169(45) | 169(100), 125(25) | 125(100) |
| 62 | Digalloylarbutin L | 6.04 | C26H23O15− | 575.10424 | 575.10211 | 3.70 | 423(100) | 313(100), 261(95), 211(5), 16930), 151(25) | 169(100), 151(5), 125(20) |
| 63 | Vanillin L,F | 6.83 | C8H7O3− | 151.04007 | 151.03960 | 3.11 | 136(100) | 108(25), 92(100) | 108(65), 79(65), 69(100), 51(30) |
| 64 | Coniferyl aldehyde L,F | 7.80 | C10H9O3− | 177.05572 | 177.05485 | 4.91 | 163(10), 162(100) | 134(100), 133(40), 120(20), 106(30) | 106(100), 65(80) |
| Phenolic Compounds | Leaf (mg/kg dw) | Fruit (mg/kg dw) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Mali Lošinj | Koločep | Mali Lošinj | Koločep | ||||||
| Water | MeOH | Water | MeOH | Water | MeOH | Water | MeOH | ||
| 1 | Gallic_acid | - | - | - | - | 21.04 | 19.30 | 23.95 | 25.80 |
| 2 | Gallocatechin | 64.21 | 97.77 | 121.11 | 211.60 | 86.08 | 90.25 | 74.63 | 91.08 |
| 3 | Protocatechuic acid | 1.68 | 2.47 | 1.27 | 1.69 | - | - | - | - |
| 4 | Aesculin | 2.56 | 5.88 | 1.95 | 3.97 | - | - | - | - |
| 5 | Chlorogenic acid | 1.95 | 1.59 | - | 1.11 | - | - | - | - |
| 6 | Catechin | 47.73 | 79.25 | 57.85 | 102.95 | 33.17 | 61.70 | 20.64 | 48.33 |
| 7 | p-Hydroxybenzoic acid | 27.08 | 22.17 | 16.76 | 16.21 | 5.85 | 2.35 | 0.68 | 4.93 |
| 8 | Caffeic acid | 4.31 | 5.75 | 2.61 | 4.07 | 4.83 | 2.68 | 2.55 | 0.41 |
| 9 | Syringic acid | 2.67 | 1.27 | 0.66 | 0.88 | - | - | - | - |
| 10 | Vanillic acid | 7.96 | 7.12 | 4.40 | 3.71 | 1.74 | 2.54 | 1.64 | 1.52 |
| 11 | Rutin | 93.39 | 106.03 | 29.93 | 33.75 | 2.29 | 2.93 | 0.88 | 1.94 |
| 12 | p-Hydroxyphenylacetic acid | 4.35 | 5.36 | 4.63 | 6.57 | - | - | - | - |
| 13 | Hyperoside | 1149.54 | 1512.94 | 635.10 | 876.61 | 37.06 | 47.90 | 13.86 | 23.73 |
| 14 | p-Coumaric acid | 32.83 | 31.38 | 10.11 | 11.41 | - | - | - | - |
| 15 | Catechin gallate | 34.48 | 73.70 | 40.67 | 97.14 | - | 63.73 | - | - |
| 16 | Ferulic acid | 4.85 | 4.26 | 2.55 | 3.08 | - | - | - | - |
| 17 | Myricetin | 1.36 | 1.78 | - | 1.49 | - | - | - | - |
| 18 | Quercetin | 61.80 | 124.91 | 41.28 | 82.44 | 89.74 | - | - | - |
| 19 | Naringenin | 3.70 | 4.39 | - | 4.18 | - | - | - | - |
| 20 | Kaempferol | 15.15 | 35.50 | 10.63 | 22.26 | - | - | - | - |
| Sample | Location | TPC | RSA | ||||
|---|---|---|---|---|---|---|---|
| (mg GAE/g dw *) | EC50 ** (mg/L) | μmol TE/g dw * | |||||
| Water | Methanol | Water | Methanol | Water | Methanol | ||
| Leaf | Mali Lošinj | 67.07 ± 1.92 | 85.30 ± 1.81 | 62.53 ± 1.13 | 45.12 ± 0.98 | 408.92 ± 2.68 | 430.70 ± 2.11 |
| Koločep | 91.56 ± 0.45 | 104.74 ± 1.67 | 57.94 ± 1.50 | 38.23 ± 1.47 | 430.98 ± 0.84 | 428.36 ± 2.19 | |
| Fruit | Mali Lošinj | 18.73 ± 1.41 | 25.86 ± 0.72 | 265.67 ± 1.81 | 177.49 ± 1.18 | 90.55 ± 0.49 | 104.04 ± 0.80 |
| Koločep | 16.78 ± 0.91 | 20.38 ± 0.36 | 294.85 ± 1.94 | 256.72 ± 2.50 | 74.30 ± 0.09 | 97.54 ± 0.88 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brčić Karačonji, I.; Jurica, K.; Gašić, U.; Dramićanin, A.; Tešić, Ž.; Milojković Opsenica, D. Comparative Study on the Phenolic Fingerprint and Antioxidant Activity of Strawberry Tree (Arbutus unedo L.) Leaves and Fruits. Plants 2022, 11, 25. https://doi.org/10.3390/plants11010025
Brčić Karačonji I, Jurica K, Gašić U, Dramićanin A, Tešić Ž, Milojković Opsenica D. Comparative Study on the Phenolic Fingerprint and Antioxidant Activity of Strawberry Tree (Arbutus unedo L.) Leaves and Fruits. Plants. 2022; 11(1):25. https://doi.org/10.3390/plants11010025
Chicago/Turabian StyleBrčić Karačonji, Irena, Karlo Jurica, Uroš Gašić, Aleksandra Dramićanin, Živoslav Tešić, and Dušanka Milojković Opsenica. 2022. "Comparative Study on the Phenolic Fingerprint and Antioxidant Activity of Strawberry Tree (Arbutus unedo L.) Leaves and Fruits" Plants 11, no. 1: 25. https://doi.org/10.3390/plants11010025
APA StyleBrčić Karačonji, I., Jurica, K., Gašić, U., Dramićanin, A., Tešić, Ž., & Milojković Opsenica, D. (2022). Comparative Study on the Phenolic Fingerprint and Antioxidant Activity of Strawberry Tree (Arbutus unedo L.) Leaves and Fruits. Plants, 11(1), 25. https://doi.org/10.3390/plants11010025









