Abstract
Berries of Actinidia kolomikta (A. kolomikta) are known for high ascorbic acid content, but the diversity of phenolic compounds has been little studied. The present research aimed to investigate phenolic compounds and antioxidant activity in berries and leaves of twelve A. kolomikta cultivars. The UHPLC-ESI-MS/MS technique was used to determine differences among cultivars in the quantitative composition of individual phenolic compounds. Antioxidant activity was determined by DPPH• free radical scavenging and CUPRAC methods. In the present study, 13 phenolic compounds were detected in berries, whereas leaves contained 17 phenolic compounds. Flavonols were the primary class found in both berries and leaves; other identified phenolic compounds were flavan-3-ols, flavones and, phenolic acids; and dihydrochalcone phloridzin was identified in the leaves. The amount and variety of phenolic compounds in berries and leaves and antioxidant activity were found to be cultivar-dependent. The highest total content of phenolic compounds was found in the leaves of the cultivar ‘Aromatnaja’ and in the berries of the cultivar ‘VIR-2’. Results of this study have confirmed that berries and leaves of A. kolomikta could be a valuable raw material for both food and pharmaceutical industries.
1. Introduction
Quantitative and qualitative research on the various biochemical compounds derived from fruits, including berries, is becoming the basis for innovative products in the pharmaceutical, cosmetic, and food industries. Fruits and their products possess a high concentration of phenolic compounds (flavonols, phenolic acids, flavan-3-ols, flavones, and others); and the antioxidant activity of phenolic compounds in vitro and in vivo has been proven [1,2]. Individual phenolic compounds or various combinations can have a substantial effect on human health. Protection against cardiovascular diseases, inflammation, and cancer have been confirmed [3,4,5].
Studies of berries of various Actinidia Lindl. species have shown higher levels of phenolic compounds than apples, grapefruits, lemons, or peaches [6,7]. A review of studies on phenolic compounds showed that berries of different Actinidia species were characterized by both quantitative and qualitative diversity of these compounds. Biologically active compounds of species A. deliciosa (A.Chev.) C.F.Liang & A.R.Ferguson and A. chinensis Planch. have been studied in more detail using state-of-the-art equipment and modern methods [8,9,10]. In recent years, more attention has been paid to A. arguta (Siebold & Zucc.) Planch. ex Miq., A. arguta var. purpurea (Rehder) C.F.Liang ex Q.Q.Chang and their hybrid cultivars [11,12,13]. Zuo et al. found that phenolic compounds were the major contributors to the total antioxidant capacity of Actinidia berries [14].
Phenolic compounds are usually concentrated in the skin of A. arguta and A. deliciosa berries. The phenolic content in the berry skin can be up to ten times greater than that of the berry pulp [15]. A. arguta leaves were also investigated for biochemical compounds and, antioxidant and antimicrobial activity [16]. The results of this study confirmed the presence of phenolic compounds from flavan-3-ols, flavonols, and hydroxycinnamic acid classes and verified the antimicrobial activity of leaf extracts. Other authors ascertained that berries of winter-hardy Actinidia species accumulated significantly higher amounts of phenolic compounds than the world’s most widely grown cultivars of A. deliciosa and A. chinensis [12,15,17]. Eventually, phenolic compounds of A. deliciosa, A. chinensis and A. arguta berries could be highly appreciated for the health-promoting benefits of dietary consumption [17,18,19].
The quantitative and qualitative composition of phenolic compounds significantly influences Actinidia berries’ quality and taste properties [20,21,22]. The results of different studies have corroborated that phenolic compounds play an essential role in plants’ defensive reactions, i.e., environmental stress can raise the accumulation of phenolic compounds, including phenolic acids [23,24,25,26]. Actinidia kolomikta (Rupr. & Maxim.) Maxim. is the most cold-hardy species and extends north to 52°40′ N, further north than any other Actinidia species [27]. Interestingly, A. kolomikta cultivars and clones are cultivated because of their exceptional resistance to pests and diseases in Lithuania. So far, this dioecious species has been extensively investigated, and new cultivars have been selected in Russia, Poland, and Lithuania. A. kolomikta was commonly cultivated in the temperate zone of Europe due to its winter hardiness and berry harvest. The cultivation of A. kolomikta cultivars has so far been mainly of interest to amateur gardeners who have used berries for their own purposes. On the other hand, long-term investigations on A. kolomikta have confirmed pest resistance, which supports cultivation in organic plantations [28]. This study aimed to detect the diversity of phenolic compounds and antioxidant activity in berries and leaves of A. kolomikta and determine variation among cultivars.
Few studies have been published on the biochemical composition of A. kolomikta berries. Biologically active substances, namely vitamin C, total phenolic, and total flavonoid compounds, have been presented in some studies by different authors [14,17,29]. Nevertheless, A. kolomikta has not been properly assessed for its composition of biologically active compounds. Investigations on secondary metabolites in berries and leaves, including phenolic compounds, are relevant because of the growing interest in their multiple biological effects.
2. Results and Discussion
2.1. Determination of Phenolic Compounds Using UHPLC-ESI-MS/MS
The results of this study confirmed some quantitative and qualitative differences in phenolic compounds between berries and leaves of A. kolomikta. A total of 13 phenolic compounds in the berries and 17 phenolic compounds in the leaves were identified. In the berries and leaves, the major flavonols identified were kaempherol, kaempherol-3-O-glucoside, quercetin, quercitrin, isorhamnetin, and rutin (Table 1 and Table 2).

Table 1.
Content of phenolic compounds (µg/g) in leaves of different Actinidia kolomikta cultivars (1–12).

Table 2.
Content of phenolic compounds (µg/g) in berries of different Actinidia kolomikta cultivars (1–12).
Flavonols isorhamnetin-3-O-rutinoside and galangin were identified only in leaves of the cultivars ‘Anykšta’, ‘Sentiabrskaja’, and ‘Aromatnaja’, respectively. Phloridzin (dihydrochalcone) was found in leaves of the cultivar ‘VIR-1’ (Table 1). The total content of flavonols ranged from 727.96 µg/g to 2128.08 µg/g in the leaves and from 157.21 µg/g to 589.01 µg/g in the berries of A. kolomikta cultivars (Table 3). Flavon-3-ols (+)-catechin, (−)-epicatechin, and procyanidin C1 represented the second most abundant class of phenolic compounds; but the total amount of flavon-3-ols was the most extensive, measuring on average 36.4% in the berries and 61.6% in the leaves of the total phenolics amount. Three compounds belonging to the hydroxycinnamic acids class were found in the both berries and leaves, i.e., chlorogenic, neochlorogenic, and caffeic acids. High amounts of chlorogenic and neochlorogenic acids were detected in the leaves of all cultivars. Chlorogenic acid was notably predominant in the berries, while caffeic acid was found only in the cultivar ‘Lankė’, and the berries of the cultivar ‘VIR-2’ were distinguished by neochlorogenic acid. Other authors have reported that neochlorogenic acid was the major phenolic acid in berries of A. arguta except for ‘Jumbo’, in which chlorogenic acid dominated [5]. For flavone class, acacetin was found in the leaves of some cultivars but this compound was not present in the berries at all. Small amounts of apigenin have been found in both the berries and leaves of many cultivars. It must be acknowledged that the total amount of flavones was insignificant compared to other classes of phenolic compounds and ranged from 0.28 µg/g to 8.9 µg/g and from 0.02 µg/g to 0.09 µg/g in the leaves and berries, respectively. As in our study, the total amount of flavan-3-ols was largest in berries of five cultivars of A. arguta cultivars and in two crosses of A. arguta × A. purpurea [5].

Table 3.
Summary content of phenolic compounds (µg/g) in berries and leaves of different Actinidia kolomikta cultivars.
Investigations of phenolic compounds in the berries and leaves of twelve A. kolomikta cultivars confirmed that their quantitative and qualitative composition depends on the genotype. In our study, the largest amount of total phenolic compounds was detected in the cultivar ‘VIR-2’ berries (Table 3). The highest concentration of flavan-3-ols including procyanidin C1 was found in berries of the cultivar ‘VIR-2’, 1468.27 µg/g and 1236.66 µg/g, respectively and the lowest in ‘Matovaja’, 436.11 µg/g and 316.04, respectively.
Leaves of the cultivar ‘Aromatnaja ‘were characterized by exceptionally high content of phenolic compounds (7679.72 µg/g), of which 63.7% were flavan-3-ols. The amounts of phenolic acids in the leaves of different cultivars varied greatly because the leaves of the cultivar ‘Aromatnaja’ accumulated 1193.61 µg/g of phenolic acids whereas in the leaves of the cultivar ‘Landė’ 52.97 µg/g of phenolic acids were present. Other authors have indicated that the amount of phenolic compounds in A. arguta berries was also significantly cultivar dependent [18]. Examination of A. arguta cultivars ‘Ananasnaja’, ‘Geneva’, ‘Weiki’, and ‘Issai’ showed that the berries of the cultivar ‘Ananasnaja’ accumulated the most phenolic compounds (6679.18 mg/100 g DW) [5].
To the best of our knowledge, little research has been carried out on the biochemical composition of Actinidia spp. leaves. Almeida et al. found that HPLC analysis revealed the highest amounts of phenolic acids (hydroxycinnamic acid derivatives) and flavonoids (flavan-3-ol and flavonols derivatives) in the leaves of A. arguta [16]. In our study, the leaves of A. kolomikta cultivars were characterized by significant amounts of phenolic compounds. High amounts of phenolic compounds in the leaves of A. arguta have been described by Marangi et al. [30]. Interestingly, young apical leaves were identified as having the highest content of total phenolic compounds and flavonoids, and a continuous decrease in total phenolic content with leaf maturity was detected in eight accessions of A. arguta [31].
The predominant phenolic compounds in A. deliciosa berries were flavonoids, tannins, and flavonols [29,32]. Zuo et al. summarized the results of A. chinensis, A. deliciosa, A. arguta, and A. kolomikta biochemical investigations and confirmed that the total content of phenolic compounds was the highest in the berries of A. kolomikta genotypes whereas, the amounts of flavonoids found in A. kolomikta and A. arguta did not differ significantly [14]. Comparison of different studies on the qualitative and quantitative variety of phenolic compounds has some limitations due to variation in the preparation of extracts and methods used. The classes of phenolic compounds found in the berries and leaves of A. kolomikta cultivars were essentially consistent with the data of other authors evaluating the phenolic compounds in different A. arguta genotypes [16,29].
In our study, trends of the accumulation of various valuable health-promoting phenolic compounds were similar in both the leaves and berries. Investigations have shown that the highest levels of phenolic compounds in A. kolomikta leaves and berries were found for procyanidin C1. As Neyestany reported, this compound was found to be a potent inhibitor of lipid peroxidation [9]. The evaluation of the impact of quercetin on oxidative stress and vascular function confirmed quercetin’s effects on blood [33]. In vitro and in vivo protection of cells from inflammatory injury has been reported for kaempherol–3-O-glucoside [34]. Small amounts of the flavonol galangin, and dihydrochalcone phloridzin were found in the leaves of the cultivars ‘Aromatnaja’ and ‘VIR-1’, nevertheless, their biological activity is worthy of attention and further research. In vitro studies have shown that galangin regulates glucose levels, and the effect of phloridzin on human health, especially diabetes, was also substantiated [34,35,36].
2.2. Determination of Antioxidant Activity and Correlation with TPC
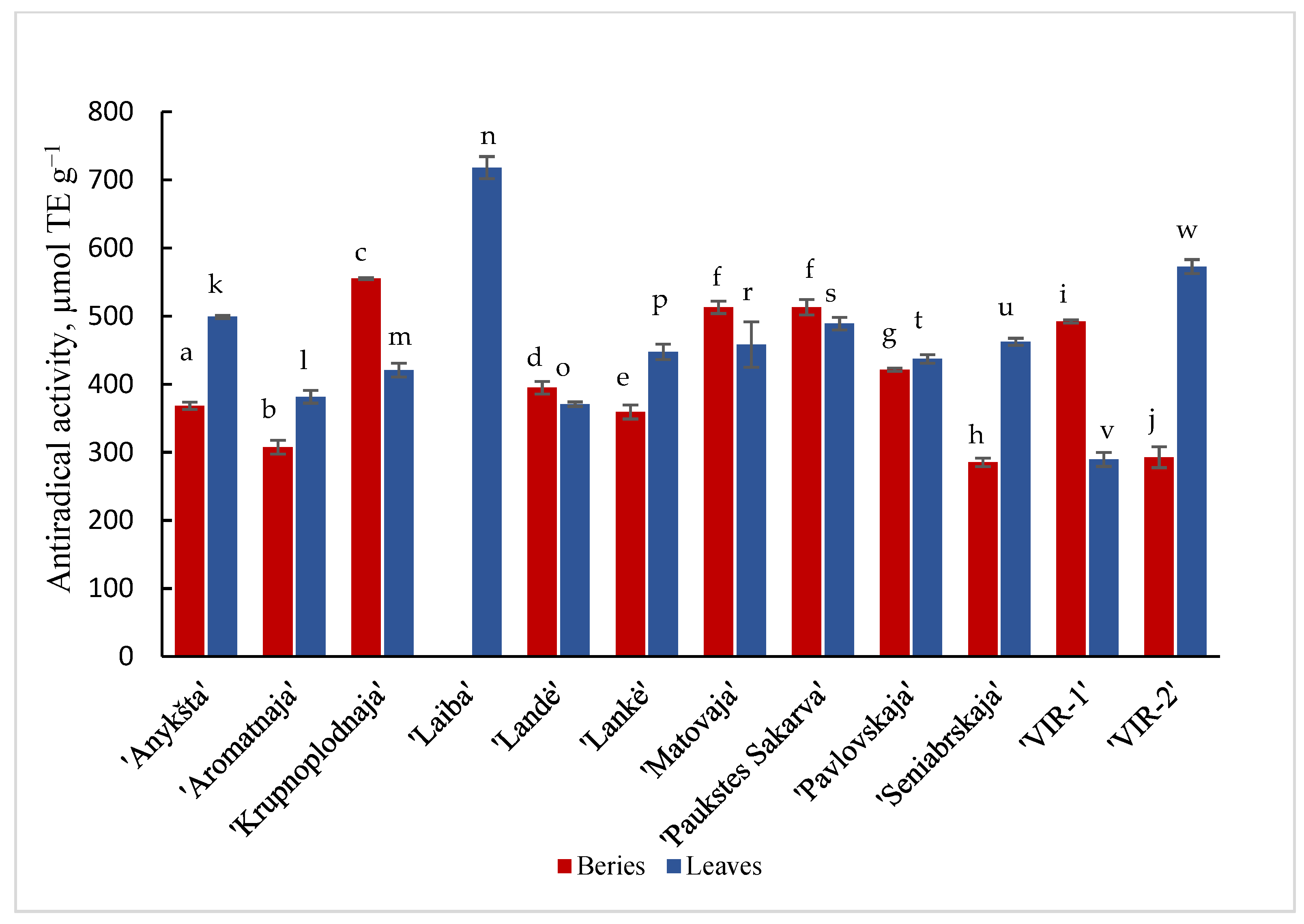
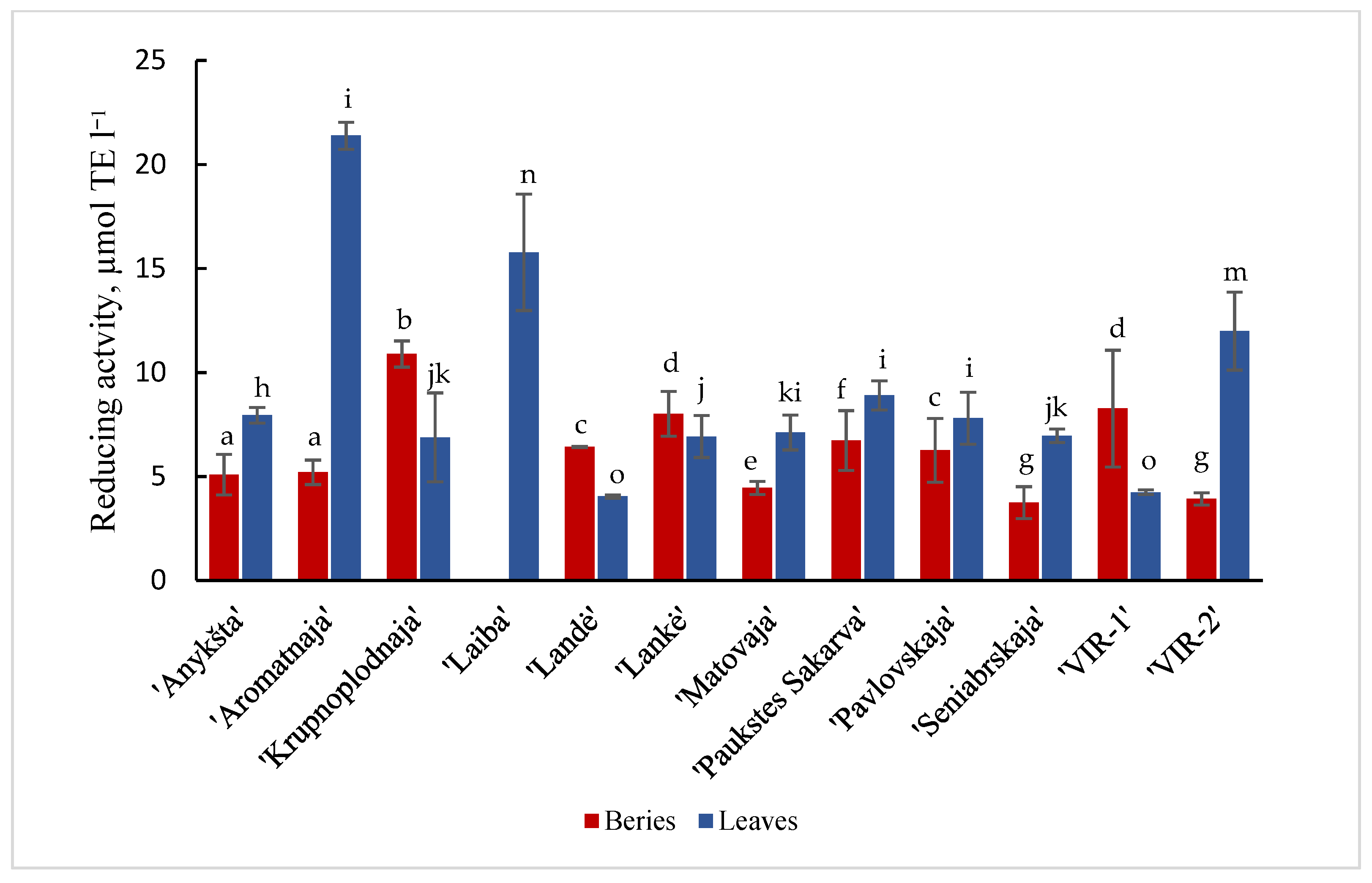
Results are shown in Figure 1 and Figure 2 of the antioxidant activity of different cultivars of A. kolomikta. Specifically, berries and leaves were taken to measure DPPH and CUPRAC antioxidant activity. Significant variation was found in the antiradical activity of the studied cultivars and the values in berries and leaves varied from 285.1 to 555.21 μmol/TE g−1 and from 370.7 to 499.1 μmol/TE g−1 (DPPH assay), respectively. These results confirm that leaves of the cultivar ‘Laiba’ and berries of the cultivar ‘Krupnoplodnaja’ had exceptionally high antiradical activity (Figure 1). The CUPRAC method determined the highest reducing activity for ‘Aromatnaja’ leaves and ‘Krupnoplodnaja’ berries. The value of the reducing activity varied from 5.09 to 10.89 μmol/TE g−1 in the berries and from 4.04 to 21.38 μmol/TE g−1 in the leaves of different cultivars as determined by the CUPRAC assay (Figure 2).
Figure 1.
DPPH• antiradical activity of berries and leaves of different cultivars of A. kolomikta. Data were expressed as mean ± SD. Different letters above columns means significant difference between berries or leaves according to Duncan’s least significant difference (LSD) procedure at 1% significance level.
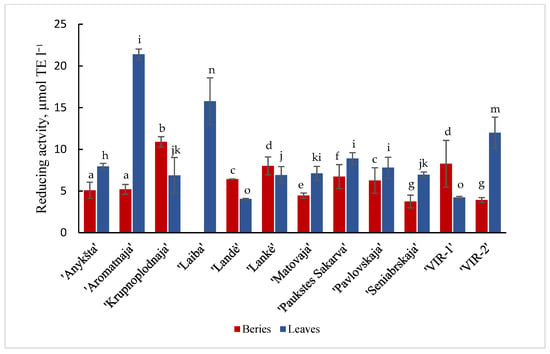
Figure 2.
CUPRAC reducing activity of berries and leaves of different cultivars of A. kolomikta. Data were expressed as mean ± SD. Different letters above columns means significant difference between berries or leaves according to Duncan’s least significant difference (LSD) procedure at 1% significance level.
As Wang et al. reported, the antioxidant potential of the commercial cultivars of A. chinensis and A. deliciosa was strongly affected by the cultivars, and the berries of A. kolomikta cultivars differed significantly in antioxidant activity [17]. This could be explained by the fact that previous studies have also confirmed exceptionally high levels of another powerful antioxidant, ascorbic acid, which is also characterized by high antioxidant potential [28].
Studies in various fruits have shown that phenolic compounds may affect antioxidant activity; and berries of Actinidia have been shown to have strong antioxidant potential compared to blueberries and cranberries. The accumulation of phenolic compounds depends on various ecological conditions and cultivation technologies [37].
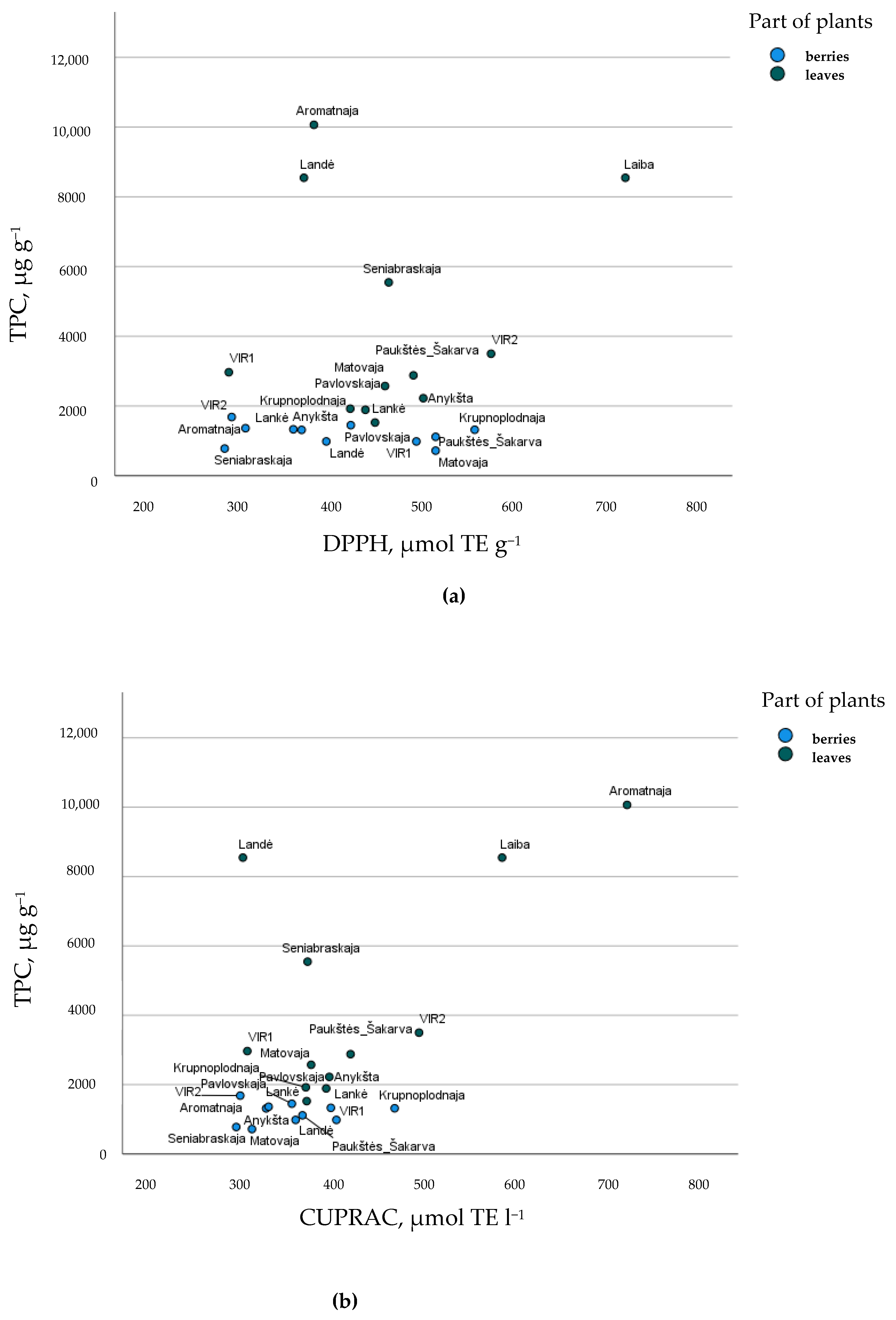
Figure 3 provides a graphical representation of the cultivars berries and leaves, according to TPC quantity and antioxidant activity. Some cultivars were shown for the uniqueness of their leaves—‘Laiba’ for DPPH antiradical activity and ‘Aromatnaja’ for CUPRAC reducing activity. This study ascertained that the berries and leaves of different cultivars of A. kolomikta accumulated a variety of phenolic compounds with high antioxidant activity from 720.64 µg/g to 1684.48 µg/g and from 1420.44 to 76,679.72, respectively. These results strongly support possible applications for pharmaceutical, nutraceutical, and food industries, and green synthesis for nanoparticles, for example, as a capping and reducing agent in nanoparticle synthesis [38].
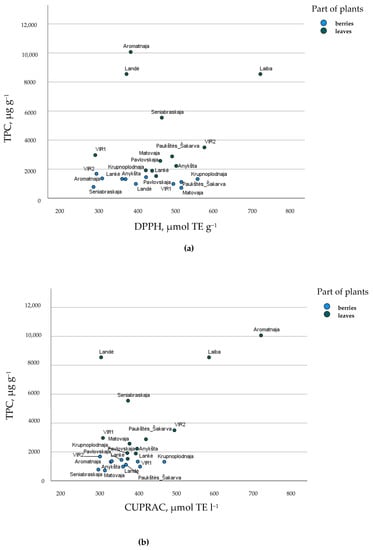
Figure 3.
Cultivar’s arrangement describing (a) total phenolic compounds (TPC, µg g−1) and DPPH, µmol TE g−1 antiradical activity and (b) total phenolic compounds (TPC, µg g−1) and CUPRAC, µmol TE g−1 reducing activity of A. kolomikta berries and leaves.
3. Materials and Methods
3.1. Plant Material
Berries and leaves were collected in the Actinidia spp. field collection of the Botanical Garden of Vytautas Magnus University. This collection was established during the implementation of the Lithuanian State Plant Genetic Resources Research Program in 1998–2018. The collection is located in the Central region of Lithuania, 76 m above sea level. The growing season lasts for approximately 180 days in this region. The species A. kolomikta. i.e., five cultivars of Lithuanian origin and seven cultivars of Russian origin were selected for investigation (Table 4).

Table 4.
List of Actinidia kolomikta cultivars investigated in the present study.
Approximately 250–300 g of berry samples were prepared per cultivar. Berries were harvested randomly from six plants at the stage of technical maturity (berries had begun to ripen and soften) and then mixed. The time of sample collection depended on the cultivar properties and continued from the last week of July to the first week of August. Berries of the ‘Laiba’ variety were not evaluated due to their insufficient quantity. All samples of leaves were collected in July. Approximately 200 g of fully developed leaves per cultivar were collected from mixed or vegetative shoots, excluding the four lower leaves at the base of the shoot and the five to six upper leaves.
3.2. Chemicals
All the solvents, reagents, and standards used were of analytical grade. The following substances were used in the study: ethanol 96% (v/v) (SC “Vilniaus degtinė”, Vilnius, Lithuania), formic acid, acetonitrile, (−)-epicatechin, (+)-catechin, procyanidin C1, isorhamnetin, acacetin, apigenin, caffeic acid, chlorogenic acid, neochlorogenic acid, phloridzin, kaempherol, kaempherol-3-O-glucoside, quercetin, galangin, quercitrin, rutin, isorhamnetin-3-O-rutinoside, sodium carbonate, gallic acid monohydrate, DPPH (2,2-diphenyl-1-picrylhydrazyl), neocuproine, ammonium acetate, copper (II) chloride dihydrate, trolox ((±)-6-hydroxy-2,5,7,8-tetramethylchromano-2-carboxylic acid) (Sigma-Aldrich, Steinheim, Germany). During the study, we used purified de-ionized water prepared with the “Milli–Q®” (“Millipore”, Bedford, MA, USA) water purification system.
3.3. Sample Preparation
Berries were stored in a freezer at −80 °C until analysis. In the next step, the berries were lyophilized at the pressure of 0.01 mbar and a condenser temperature of −85 °C in a condenser. Lyophilized berries were ground using the knife mill Grindomix GM 200 (Retsch GmbH, Haan, Germany) and samples were stored in tightly closed containers until investigation.
The dehydration level of samples was determined with a hygrometer Precisa 310 M (Precisa, Dietikon, Switzerland). For each sample, the procedure was repeated three times and averages of the drying estimates were calculated.
For preparation of ethanolic extracts, each sample of 2.5 g lyophilized powder was placed in a dark glass bottle and 40 mL of 70% of ethanol at room temperature was added. Samples were sealed and placed in an ultrasonic bath and were extracted for 10 min at 80 kHz and 1017 W. After extraction, the ethanolic extracts were centrifuged for 2 min. at 8500 rpm, at room temperature using the centrifuge Heraeus Biofuge Stratos (Heraeus Holding GmbH, Haan, Germany). The supernatants were poured off from the residues, filtered and placed in dark wide-mouthed bottles, which were kept in a refrigerator at 4 °C until analysis. Ethanolic extracts were filtered through 0.22 μm pore size membrane filters prior to investigation.
3.4. Determination of Total Phenolic Content and Quantitative Composition by UHPLC-ESI-MS/MS Technique
All the spectrophotometric measurements were carried out with the M550 UV/Vis spectrophotometer (Spectronic CamSpec, Garforth, England, UK).
Separation of phenolic compounds was performed with the Acquity H-class UHPLC system (Waters, Santa Clara, CA, USA) equipped with a triple quadrupole tandem mass spectrometer (Xevo, Waters, USA) with an electrospray ionization source (ESI) to obtain MS/MS data using a previously described and validated UHPLC-ESI-MS/MS technique [2]. A YMC Triart C18 (100 × 2.0 mm; 1.9 μm) column (YMC Europe GmbH, Dislanken, Germany) was used for analysis. Column temperature was maintained at 40 °C. Gradient elution was performed with a mobile phase consisting of 0.1% formic acid water solution (solvent A) and acetonitrile (solvent B) with the flow rate set to 0.5 mL min−1. Injection volume was 10 µL. Linear gradient profile was applied as follows for solvent A: initially 95% for 1 min; to 70% over 4 min; 50% over 7 min; 95% over 2 min. Negative electrospray ionization was applied for analysis: capillary voltage −2 kV, source temperature 150 °C, desolvation temperature 400 °C, desolvation gas flow 700 L h−1, cone gas flow 20 L h−1. Each compound of interest had a specific collision energy and cone voltage selected. The selected mass spectrometry parameters for this method were presented in our previous work [2]. The evaluation of the selectivity of the method for peak identification and purity was based on the comparison of the retention times and MS spectra of the analytes with those of the standard compounds. The limit of detection (LOD) and limit of quantification (LOQ) of the analytes were assessed by comparing the peak height to the baseline noise. The signal-to-noise ratio was 3:1 for a limit of detection and it was 10:1 for a limit of quantification. The determined LOD varied from 0.4 ng/mL to 32.51 ng/mL and the determined LOQ varied from 1.2 ng/mL to 100.25 ng/mL. Calibration curves were obtained by plotting the peak areas of analytical standards. The estimated determination coefficients (R2) of calibration curves was ≥ 0.96. Content of the phenolic compound was expressed as µg/g DW. Samples of chromatograms were presented in the Supplementary Materials, Figures S1 and S2.
3.5. Determination of Antioxidant Activity
3.5.1. DPPH• Free Radical Scavenging Assay
The DPPH• free radical scavenging activity was determined using the method proposed by Brand–Williams et al. [39,40]. DPPH• solution in 96.3% v/v ethanol (3 mL, 6 × 10−5 M) was mixed with 10 μL of the ethanol extract of A. kolomikta fruits or leaves. A decrease in absorbance was determined at a wavelength of 515 nm after keeping the samples for 30 min in the dark. Trolox was used as the standard for the calibration curve and the DPPH• values were expressed as µmol of trolox equivalents (µmol TE) per g DM of extract.
3.5.2. CUPRAC Assay
CUPRAC solution included copper (II) chloride (0.01 M in water), ammonium acetate buffer solution (0.001 M, pH = 7), and neocuproine (0.0075 M in ethanol) (ratio 1:1:1). A volume of 3 mL of freshly prepared CUPRAC reagent was mixed with 10 μL of Actinidia fruits or leaves extract. An increase in absorbance was recorded after 30 min at a wavelength of 450 nm [30]. CUPRAC assay measurements and calculations were performed using trolox calibration curves and were expressed as µmol of the trolox equivalent (TE) per one gram of dry weight.
3.6. Statistical Analysis
Mean values and standard deviations were calculated using MS Excel 2020 (Redmond, WA, USA). One-way analysis of the variance (ANOVA) along with the post hoc Duncan’s test was employed for statistical analysis using SPSS Statistics version 27.0.0.0 (IBM, Armonk, NY, USA) software. Moreover, a custom chart builder was implemented using a scatter plot with a grouping variable and point ID label.
4. Conclusions
Investigations of A. kolomikta cultivars revealed a range of phenolic compounds. Both berries and leaves showed the presence of flavan-3-ols, phenolic acids, flavones, and flavonols. This indicates that A. kolomikta is a promising health promoting source of biochemical compounds with antioxidant activity. Berries of the cultivar ‘VIR-2′ and leaves of the cultivar ‘Aromatnaja’ accumulated the largest amounts of total phenolic compounds. There were significant differences amongst the cultivars studied but the cultivar ‘VIR-2’ as a potential donor of valuable biochemical properties in the breeding of cultivars with higher amounts of phenolic compounds in berries, was confirmed. For the first time the composition of the phenolic compounds of leaves from different of A. kolomikta cultivars were comprehensively evaluated, and both DPPH• and CUPRAC technologies demonstrated strong antioxidant potential. This study confirmed high levels of different classes of phenolic compounds in the leaves, thus it is important to further investigate their potential applications for the food or pharmaceutical industries.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/plants11020147/s1, Figure S1: Chromatogram of leaves of the cultivar ‘Landė’; Figure S2: Chromatogram of berries of the cultivar ‘Landė’.
Author Contributions
Conceptualization, review and editing L.Č.; writing—original draft preparation P.Š.; methodology and investigations M.L.; formal analysis V.Ž.; methodology and supervision P.V.; visualization and software J.V.; resources and data curation R.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in the article.
Acknowledgments
The authors thank staff of the Institute of the Pharmaceutical Technologies of the Lithuanian University of Health Sciences for the opportunity to conduct the research. Special thanks to Master of Biological Sciences Sigita Bogačiovienė for her assistance in accomplishment of experiments.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Skrovankova, S.; Sumczynski, D.; Mlcek, J.; Jurikova, T.; Sochor, J. Bioactive Compounds and Antioxidant Activity in Different Types of Berries. Int. J. Mol. Sci. 2015, 16, 24673–24706. [Google Scholar] [CrossRef]
- González-Burgos, E.; Liaudanskas, M.; Viškelis, J.; Žvikas, V.; Janulis, V.; Gómez-Serranillos, M.P. Antioxidant Activity, Neuroprotective Properties and Bioactive Constituents Analysis of Varying Polarity Extracts from Eucalyptus globulus Leaves. J. Food Drug Anal. 2018, 26, 1293–1302. [Google Scholar] [CrossRef]
- Olas, B. Berry Phenolic Antioxidants—Implications for Human Health? Front. Pharmacol. 2018, 9, 78. [Google Scholar] [CrossRef] [PubMed]
- Sinelli, N.; Spinardi, A.; Egidio, V.D.; Mignani, I.; Casiraghi, E. Evaluation of Quality and Nutraceutical Content of Blueberries (Vaccinium corymbosum L.) by near and Mid-Infrared Spectroscopy. Postharvest Biol. Technol. 2008, 50, 31–36. [Google Scholar] [CrossRef]
- Wojdyło, A.; Nowicka, P.; Oszmiański, J.; Golis, T. Phytochemical Compounds and Biological Effects of Actinidia Fruits. J. Funct. Foods 2017, 30, 194–202. [Google Scholar] [CrossRef]
- Inil, L.; Sungbin, I.; Cheng-Ri, J.; Jin, H.H.; Youn-Sup, C.; Moo-Yeol, B.; Dae-Ok, K. Effect of Maturity Stage at Harvest on Antioxidant Capacity and Total Phenolics in Kiwifruits (Actinidia Spp.) Grown in Korea. Hortic. Environ. Biotechnol. 2015, 56, 841–848. [Google Scholar] [CrossRef]
- An, X.; Lee, S.G.; Kang, H.; Heo, H.J.; Cho, Y.S.; Kim, D.O. Antioxidant and Anti-Inflammatory Effects of Various Cultivars of Kiwi Berry (Actinidia arguta) on Lipopolysaccharide-Stimulated RAW 264.7 Cells. J. Microbiol. Biotechnol. 2016, 26, 1367–1374. [Google Scholar] [CrossRef]
- Henare, S.J. The Nutritional Composition of Kiwifruit (Actinidia spp.). In Nutritional Composition of Fruit Cultivars; Simmonds, S.J.M., Preedy, R.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 337–370. ISBN 978-0-12-408117-8. [Google Scholar]
- Yong-Seo, P.; Jacek, N.; Kann, V.; Hanna, L.; Maria, L.; Dinorah, B.; Alina, N.; Simon, T.; Shela, G. Bioactive Compounds and the Antioxidant Capacity in New Kiwi Fruit Cultivars. Food Chem. 2014, 165, 354–361. [Google Scholar] [CrossRef]
- Tavarini, S.; Degl’Innocenti, E.; Remorini, D.; Massai, R.; Guidi, L. Antioxidant Capacity, Ascorbic Acid, Total Phenols and Carotenoids Changes during Harvest and after Storage of Hayward Kiwifruit. Food Chem. 2008, 107, 282–288. [Google Scholar] [CrossRef]
- Latocha, P.; Wołosiak, R.; Worobiej, E.; Krupa, T. Clonal Differences in Antioxidant Activity and Bioactive Constituents of Hardy Kiwifruit (Actinidia arguta) and Its Year-to-Year Variability. J. Sci. Food Agric. 2013, 93, 1412–1419. [Google Scholar] [CrossRef]
- Leontowicz, H.; Leontowicz, M.; Latocha, P.; Jesion, I.; Park, Y.S.; Katrich, E.; Barasch, D.; Nemirovski, A.; Gorinstein, S. Bioactivity and Nutritional Properties of Hardy Kiwi Fruit Actinidia arguta in Comparison with Actinidia deliciosa ‘Hayward’ and Actinidia Eriantha ‘Bidan’. Food Chem. 2016, 196, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Dong, E.J.; Seon, K.P.; Chang, H.P.; Tae, W.S.; Heo, H. Nutritional Compositions of Three Traditional Actinidia (Actinidia arguta) Cultivars Improved in Korea. J. Korean Soc. Food Sci. Nutr. 2014, 43, 1942–1947. [Google Scholar] [CrossRef]
- Zuo, L.-L.; Wang, Z.-Y.; Fan, Z.-L.; Tian, S.-Q.; Liu, J.-R. Evaluation of Antioxidant and Antiproliferative Properties of Three Actinidia (Actinidia kolomikta, Actinidia arguta, Actinidia chinensis) Extracts in Vitro. Int. J. Mol. Sci. 2012, 13, 5506–5518. [Google Scholar] [CrossRef]
- Latocha, P.; Łata, B.; Stasiak, A. Phenolics, Ascorbate and the Antioxidant Potential of Kiwiberry vs. Common Kiwifruit: The Effect of Cultivar and Tissue Type. J. Funct. Foods 2015, 19, 155–163. [Google Scholar] [CrossRef]
- Almeida, D.; Pinto, D.; Santos, J.; Vinha, A.F.; Palmeira, J.; Ferreira, H.N.; Rodrigues, F.; Oliveira, M.B.P. Kiwifruit Leaves (Actinidia arguta): An Extraordinary Source of Value-Added Compounds for Food Industry. Food Chem. 2018, 259, 113–121. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, C.; Li, J.; Liang, Y.; Yang, R.; Liu, J.; Ma, Z.; Wu, L. Evaluation of Biochemical Components and Antioxidant Capacity of Different Kiwifruit (Actinidia spp.) Genotypes Grown in China. Biotechnol. Biotechnol. Equip. 2018, 32, 558–565. [Google Scholar] [CrossRef]
- Latocha, P.; Krupa, T.; Wołosiak, R.; Worobiej, E.; Wilczak, J. Antioxidant Activity and Chemical Difference in Fruit of Different Actinidia sp. Int. J. Food Sci. Nutr. 2010, 61, 381–394. [Google Scholar] [CrossRef]
- Baranowska-Wójcik, E.; Szwajgier, D. Characteristics and Pro-Health Properties of Mini Kiwi (Actinidia arguta). Hortic. Environ. Biotechnol. 2019, 60, 217–225. [Google Scholar] [CrossRef]
- Cheynier, V. Phenolic Compounds: From Plants to Foods. Phytochem. Rev. 2012, 11, 153–177. [Google Scholar] [CrossRef]
- Pérez-Burillo, S.; Oliveras, M.J.; Quesada, J.; Rufián-Henares, J.A.; Pastoriza, S. Relationship between Composition and Bioactivity of Persimmon and Kiwifruit. Food Res. Int. 2018, 105, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.G.; Beppu, K.; Kataoka, I. Varietal Differences in Phenolic Content and Astringency in Skin and Flesh of Hardy Kiwifruit Resources in Japan. Sci. Hortic. 2009, 120, 551–554. [Google Scholar] [CrossRef]
- Babenko, L.; Smirnov, O.; Romanenko, K.O.; Trunova, O.; Kosakivska, I.V. Phenolic Compounds in Plants: Biogenesis and Functions. Ukr. Biochem. J. 2019, 19, 5–18. [Google Scholar] [CrossRef]
- Król, A.; Amarowicz, R.; Weidner, S. Changes in the Composition of Phenolic Compounds and Antioxidant Properties of Grapevine Roots and Leaves (Vitis vinifera L.) under Continuous of Long-Term Drought Stress. Acta Physiol. Plant. 2014, 36, 1491–1499. [Google Scholar] [CrossRef]
- Rehman, F.; Khan, F.; Badruddin, S. Role of Phenolics in Plant Defense against Insect Herbivory. In Chemistry of Phytopotentials: Health, Energy and Environmental Perspective; Khemani, L., Srivastava, M., Srivastava, S., Eds.; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar] [CrossRef]
- Weidner, S.; Karamać, M.; Amarowicz, R.; Szypulska, E.; Gołgowska, A. Changes in Composition of Phenolic Compounds and Antioxidant Properties of Vitis amurensis Seeds Germinated under Osmotic Stress. Acta Physiol. Plant. 2007, 29, 283–290. [Google Scholar] [CrossRef]
- Huang, H. Kiwifruit: The Genus Actinidia; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 45–167. ISBN 978-0-12-803066-0. [Google Scholar]
- Cesoniene, L. Content of Biochemical Components in Berries of Kolomikta Kiwi (Actinidia kolomikta). Acta Hortic. Regiotect. 2007, 10, 24–27. [Google Scholar]
- Latocha, P. The Nutritional and Health Benefits of Kiwiberry (Actinidia arguta)—A Review. Plant Foods Hum. Nutr. 2017, 72, 325–334. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Demirata, B.; Özyürek, M.; Çelik, S.E.; Bektaşoğlu, B.; Berker, K.I.; Özyurt, D. Comparative Evaluation of Various Total Antioxidant Capacity Assays Applied to Phenolic Compounds with the CUPRAC Assay. Molecules 2007, 12, 1496–1547. [Google Scholar] [CrossRef]
- Marangi, F.; Pinto, D.; de Francisco, L.; Alves, R.C.; Puga, H.; Sut, S.; Dall’Acqua, S.; Rodrigues, F.; Oliveira, M.B.P. Hardy Kiwi Leaves Extracted by Multi-Frequency Multimode Modulated Technology: A Sustainable and Promising by-Product for Industry. Food Res. Int. 2018, 112, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Changhua, T.; Zhigang, W.; Xiuli, F.; Muhammad, I.; Liu, C. Identification of Bioactive Compounds in Leaves and Fruits of Actinidia arguta Accessions from Northeastern China and Assessment of Their Antioxidant Activity with a Radical-Scavenging Effect. Biotechnol. Biotechnol. Equip. 2021, 35, 593–607. [Google Scholar] [CrossRef]
- Suray, P.F.; Benadé, A.J.S.; Speake, B. Natural Antioxidants in Land- and Marine-Based Wild-Type Food. In Wild-Type Food in Health Promotion and Disease Prevention: The Columbus Concept; De Meester, F., Watson, R.R., Eds.; Humana Press: Totowa, NJ, USA, 2008; ISBN 978-1-58829-668-9. [Google Scholar]
- Dabeek, W.M.; Marra, M.V. Dietary Quercetin and Kaempferol: Bioavailability and Potential Cardiovascular-Related Bioactivity in Humans. Nutrients 2019, 11, 2288. [Google Scholar] [CrossRef] [PubMed]
- Christian, G.; Heidi, H.; Karl, S. Phloridzin: Biosynthesis, Distribution and Physiological Relevance in Plants. Phytochemistry 2010, 71, 838–843. [Google Scholar] [CrossRef]
- Kalhotra, P.; Chittepu, V.C.S.R.; Osorio-Revilla, G.; Gallardo-Velázquez, T. Discovery of Galangin as a Potential DPP-4 Inhibitor That Improves Insulin-Stimulated Skeletal Muscle Glucose Uptake: A Combinational Therapy for Diabetes. Int. J. Mol. Sci. 2019, 20, 1228. [Google Scholar] [CrossRef] [PubMed]
- Mikami-Konishide, I.; Murakami, S.; Nakanishi, K.; Takahashi, Y.; Yamaguchi, M.; Shioya, T.; Watanabe, J.; Hino, A. Antioxidant capacity and polyphenol content of extracts from crops cultivated in Japan, and the effect of cultivation environment. Food Sci. Technol. Res. 2013, 19, 69–79. [Google Scholar] [CrossRef][Green Version]
- Balciunaitiene, A.; Viskelis, P.; Viskelis, J.; Streimikyte, P.; Liaudanskas, M.; Bartkiene, E.; Zavistanaviciute, P.; Zokaityte, E.; Starkute, V.; Ruzauskas, M.; et al. Green Synthesis of Silver Nanoparticles Using Extract of Artemisia absinthium L., Humulus lupulus L. and Thymus vulgaris L., Physico-Chemical Characterization, Antimicrobial and Antioxidant Activity. Process 2021, 9, 1304. [Google Scholar] [CrossRef]
- Bobinaitė, R.; Viškelis, P.; Venskutonis, P.R. Variation of Total Phenolics, Anthocyanins, Ellagic Acid and Radical Scavenging Capacity in Various Raspberry (Rubus spp.) Cultivars. Food Chem. 2012, 132, 1495–1501. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT—Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).