Response Surface Optimization of Extraction Conditions and In Vitro Antioxidant and Antidiabetic Evaluation of an Under-Valued Medicinal Weed, Mimosa pudica
Abstract
1. Introduction
2. Materials and Methods
2.1. Box-Behnken Design
2.2. Preparation of Plant Extracts
2.3. Determination of DPPH Radical Scavenging Assay
2.4. Determination of Total Phenolic Content (TPC)
2.5. Determination of Total Flavonoid Content (TFC)
2.6. Cell Passaging and Maintenance
2.7. Cell Viability Assay (MTT Assay)
2.8. Differentiation of 3T3-L1 Cells into Mature Adipocytes
2.9. Glucose Uptake Assay
2.10. Statistical Analysis
3. Results and Discussion
3.1. Optimization of Extraction Conditions
3.1.1. Extract Yield
3.1.2. IC50 Value
3.1.3. Total Phenolic Content
3.1.4. Total Flavonoid Content
3.2. Correlation Analysis
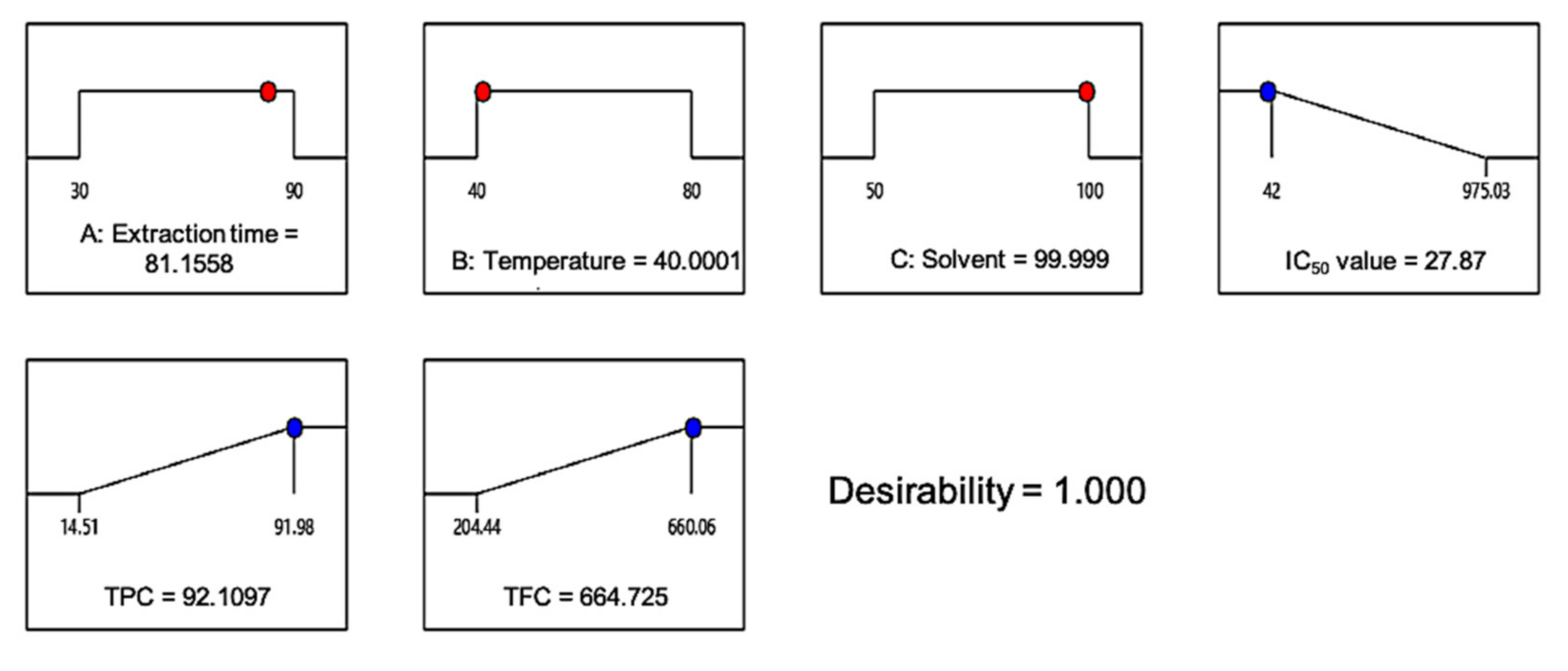
3.3. Verification of Quadratic Model
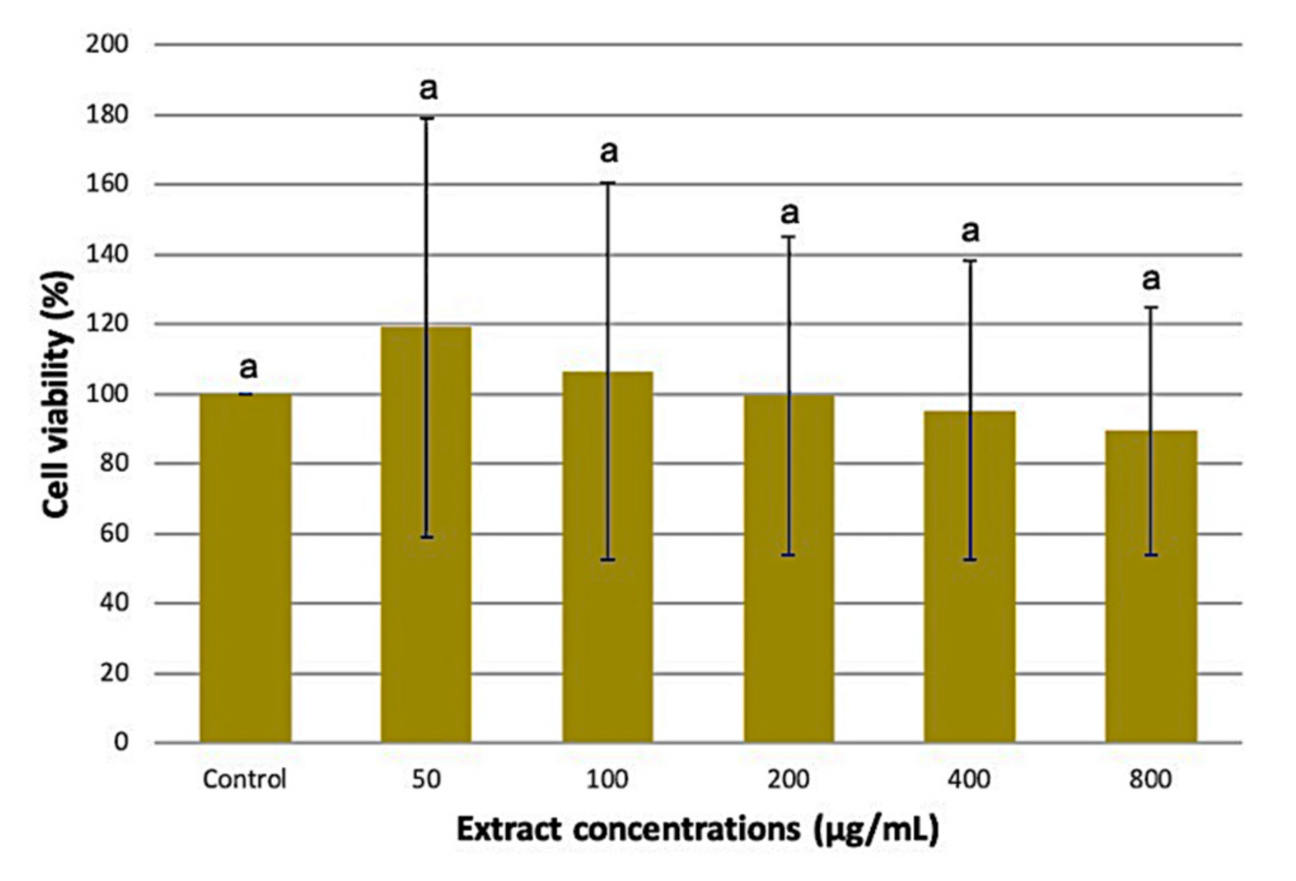
3.4. Effect of the Optimized M. pudica Extracts on 3T3-L1 Cell Viability
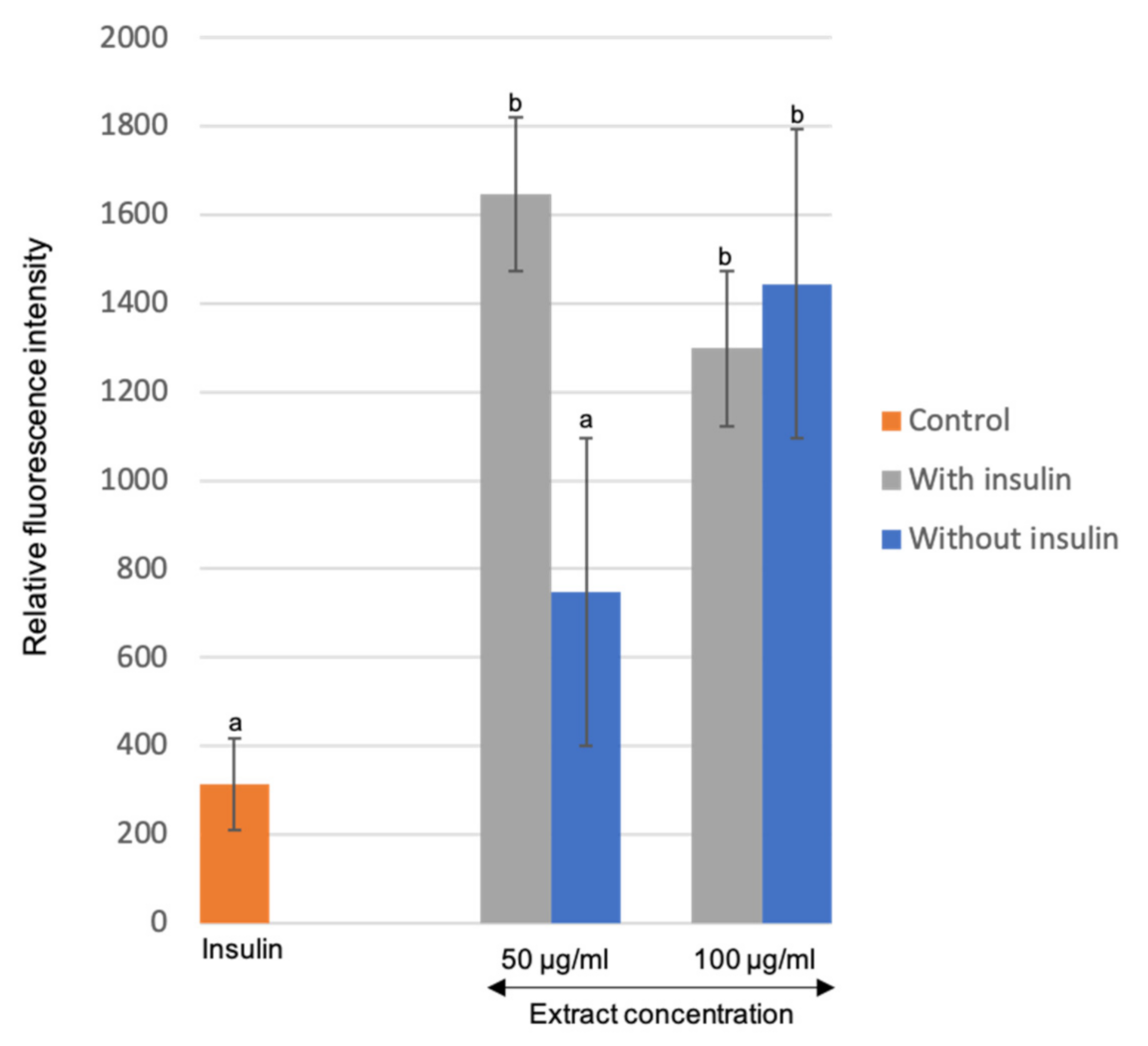
3.5. Effects of the Optimized M. Pudica Extracts on Stimulating Glucose Uptake in 3T3-L1 Adipocyte Cells
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tunna, T.S.; Ahmed, Q.U.; Uddin, A.B.M.H.; Sarker, M.Z.I. Weeds as Alternative Useful Medicinal Source: Mimosa Pudica Linn. on Diabetes Mellitus and Its Complications. Adv. Mater. Res. 2014, 995, 49–59. [Google Scholar] [CrossRef]
- Yuan, K.; Lu, J.L.; Jia, A.; Zhu, J.X. Two New C-Glycosylflavones from Mimosa Pudica. Chin. Chem. Lett. 2007, 18, 1231–1234. [Google Scholar] [CrossRef]
- Chandran, R.P.; Deepak, V.; Krishna, S.; Fathima, S.; Thaha, A.; Raj, J. Analysis of Phytochemical Constituents and Anthelmintic Activity of Leaf Extracts of Mimosa pudica L. Asian J. Biomed. Pharm. Sci. 2018, 8, 1–5. [Google Scholar] [CrossRef]
- Sriram, S.; Vaidyanathan, M.; Venkatesh, K.; Nayagam, A.A.J. GC-MS Study and Phytochemical Profiling of Mimosa Pudica Linn. J. Pharm. Res. 2011, 4, 741–742. [Google Scholar]
- Champanerkar, P.A.; Vaidya, V.V.; Shailajan, S.; Menon, S.N. A Sensitive, Rapid and Validated Liquid Chromatography-Tandem Mass Spectrometry (LC-MS-MS) Method for Determination of Mimosine in Mimosa Pudica Linn. Nat. Sci. 2010, 2, 713–717. [Google Scholar] [CrossRef]
- Ahuchaogu, A.A.; Chukwu, O.J.; Echeme, J.O. Secondary Metabolites from Mimosa Pudica: Isolation, Purification and NMR Characterization. IOSR J. Appl. Chem. 2017, 10, 15–20. [Google Scholar] [CrossRef]
- Kumar, V. Phytochemical, Pharmacological Activities and Ayurvedic Significances of Magical Plant Mimosa Pudica Linn. Mini-Rev. Org. Chem. 2021, 18, 296–312. [Google Scholar] [CrossRef]
- Alatrach, M.; Agyin, C.; Mehta, R.; Adams, J.; DeFronzo, R.A.; Abdul-Ghani, M. Glucose-Mediated Glucose Disposal at Baseline Insulin Is Impaired in IFG. J. Clin. Endocrinol. Metab. 2019, 104, 163–171. [Google Scholar] [CrossRef]
- Chaudhury, A.; Duvoor, C.; Reddy Dendi, V.S.; Kraleti, S.; Chada, A.; Ravilla, R.; Marco, A.; Shekhawat, N.S.; Montales, M.T.; Kuriakose, K.; et al. Clinical Review of Antidiabetic Drugs: Implications for Type 2 Diabetes Mellitus Management. Front. Endocrinol. 2017, 8, 6. [Google Scholar] [CrossRef]
- Liu, J.-X.; Shen, S.-N.; Tong, Q.; Wang, Y.-T.; Lin, L.-G. Honokiol Protects Hepatocytes from Oxidative Injury through Mitochondrial Deacetylase SIRT3. Eur. J. Pharmacol. 2018, 834, 176–187. [Google Scholar] [CrossRef]
- Afrisham, R.; Aberomand, M.; Ghaffari, M.A.; Siahpoosh, A.; Jamalan, M. Inhibitory Effect of Heracleum Persicum and Ziziphus Jujuba on Activity of Alpha-Amylase. J. Bot. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Giacco, F.; Brownlee, M. Oxidative Stress and Diabetic Complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef]
- Mohammadi, S.; Montasser Kouhsari, S.; Monavar Feshani, A. Antidiabetic Properties of the Ethanolic Extract of Rhus Coriaria Fruits in Rats. Daru 2010, 18, 270–275. [Google Scholar]
- Maritim, A.C.; Sanders, R.A.; Watkins, J.B. Diabetes, Oxidative Stress, and Antioxidants: A Review. J. Biochem. Mol. Toxicol. 2003, 17, 24–38. [Google Scholar] [CrossRef]
- Carocho, M.; Ferreira, I.C.F.R. A Review on Antioxidants, Prooxidants and Related Controversy: Natural and Synthetic Compounds, Screening and Analysis Methodologies and Future Perspectives. Food Chem. Toxicol. 2013, 51, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Oguntibeju, O.O. Type 2 Diabetes Mellitus, Oxidative Stress and Inflammation: Examining the Links. Int. J. Physiol. Pathophysiol. Pharmacol. 2019, 11, 45–63. [Google Scholar]
- Bayani, U.; Ajay, S.V.; Paolo, Z.; Mahajan, R.T. Oxidative Stress and Neurodegenerative Diseases: A Review of Upstream and Downstream Antioxidant Therapeutic Options. Curr. Neuropharmacol. 2009, 7, 65–74. [Google Scholar]
- Hariprasath, L.; Jegadeesh, R.; Arjun, P.; Raaman, N. In Vitro Propagation of Senecio Candicans DC and Comparative Antioxidant Properties of Aqueous Extracts of the in Vivo Plant and in Vitro-Derived Callus. S. Afr. J. Bot. 2015, 98, 134–141. [Google Scholar] [CrossRef]
- Engwa, G.A. Free Radicals and the Role of Plant Phytochemicals as Antioxidants against Oxidative Stress-Related Diseases. In Phytochemicals-Source of Antioxidants and Role in Disease Prevention; InTech: London, UK, 2018. [Google Scholar]
- Muriel, P.; Rivera-Espinoza, Y. Beneficial Drugs for Liver Diseases. J. Appl. Toxicol. 2008, 28, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.Y.; Tang, C.Y. Determination of Total Phenolic and Flavonoid Contents in Selected Fruits and Vegetables, as Well as Their Stimulatory Effects on Mouse Splenocyte Proliferation. Food Chem. 2007, 101, 140–147. [Google Scholar] [CrossRef]
- Song, H.; Zhang, Q.; Zhang, Z.; Wang, J. In Vitro Antioxidant Activity of Polysaccharides Extracted from Bryopsis Plumosa. Carbohydr. Polym. 2010, 80, 1057–1061. [Google Scholar] [CrossRef]
- Guerriero, G.; Berni, R.; Muñoz-Sanchez, J.A.; Apone, F.; Abdel-Salam, E.M.; Qahtan, A.A.; Alatar, A.A.; Cantini, C.; Cai, G.; Hausman, J.F.; et al. Production of Plant Secondary Metabolites: Examples, Tips and Suggestions for Biotechnologists. Genes 2018, 9, 309. [Google Scholar] [CrossRef]
- Najib, T.; Solgi, M.; Farazmand, A.; Heydarian, S.M.; Nasernejad, B. Optimization of Sulfate Removal by Sulfate Reducing Bacteria Using Response Surface Methodology and Heavy Metal Removal in a Sulfidogenic UASB Reactor. J. Environ. Chem. Eng. 2017, 5, 3256–3265. [Google Scholar] [CrossRef]
- Zolgharnein, J.; Shahmoradi, A.; Ghasemi, J.B. Comparative Study of Box-Behnken, Central Composite, and Doehlert Matrix for Multivariate Optimization of Pb (II) Adsorption onto Robinia Tree Leaves. J. Chemom. 2013, 27, 12–20. [Google Scholar] [CrossRef]
- Ganesan, V.; Gurumani, V.; Kunjiappan, S.; Panneerselvam, T.; Somasundaram, B.; Kannan, S.; Chowdhury, A.; Saravanan, G.; Bhattacharjee, C. Optimization and Analysis of Microwave-Assisted Extraction of Bioactive Compounds from Mimosa Pudica L. Using RSM & ANFIS Modeling. J. Food Meas. Charact. 2018, 12, 228–242. [Google Scholar]
- Sheng, Z.; Zhao, J.; Muhammad, I.; Zhang, Y. Optimization of Total Phenolic Content from Terminalia Chebula Retz. Fruits Using Response Surface Methodology and Evaluation of Their Antioxidant Activities. PLoS ONE 2018, 13, e0202368. [Google Scholar] [CrossRef]
- Mokrani, A.; Madani, K. Effect of Solvent, Time and Temperature on the Extraction of Phenolic Compounds and Antioxidant Capacity of Peach (Prunus Persica L.) Fruit. Sep. Purif. Technol. 2016, 162, 68–76. [Google Scholar] [CrossRef]
- Dirar, A.I.; Alsaadi, D.H.M.; Wada, M.; Mohamed, M.A.; Watanabe, T.; Devkota, H.P. Effects of Extraction Solvents on Total Phenolic and Flavonoid Contents and Biological Activities of Extracts from Sudanese Medicinal Plants. S. Afr. J. Bot. 2019, 120, 261–267. [Google Scholar] [CrossRef]
- Akowuah, G.A.; Ismail, Z.; Norhayati, I.; Sadikun, A. The Effects of Different Extraction Solvents of Varying Polarities on Polyphenols of Orthosiphon Stamineus and Evaluation of the Free Radical-Scavenging Activity. Food Chem. 2005, 93, 311–317. [Google Scholar] [CrossRef]
- Pin, K.Y.; Chuah, A.L.; Rashih, A.A.; Mazura, M.P.; Fadzureena, J.; Vimala, S.; Rasadah, M. Antioxidant and Anti-Inflammatory Activities of Extracts of Betel Leaves (Piper Betle) from Solvents with Different Polarities. J. Trop. For. Sci. 2010, 22, 448–455. [Google Scholar]
- Patro, G.; Bhattamisra, S.; Mohanty, B.; Sahoo, H. In Vitro and in Vivo Antioxidant Evaluation and Estimation of Total Phenolic, Flavonoidal Content of Mimosa pudica L. Pharmacogn. Res. 2016, 8, 22–28. [Google Scholar] [CrossRef]
- Ibrahim, M.M.; AL Sahli, A.A.A.; Alaraidh, I.A.; Al-Homaidan, A.A.; Mostafa, E.M.; EL-Gaaly, G.A. Assessment of Antioxidant Activities in Roots of Miswak (Salvadora persica) Plants Grown at Two Different Locations in Saudi Arabia. Saudi J. Biol. Sci. 2015, 22, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Khan, S.; Avula, B.; Lata, H.; Yang, M.H.; ElSohly, M.A.; Khan, I.A. Assessment of Total Phenolic and Flavonoid Content, Antioxidant Properties, and Yield of Aeroponically and Conventionally Grown Leafy Vegetables and Fruit Crops: A Comparative Study. Evid.-Based Complement. Altern. Med. 2014, 2014, 1–9. [Google Scholar] [CrossRef]
- Ijaz, S.; Shoaib Khan, H.M.; Anwar, Z.; Talbot, B.; Walsh, J.J. HPLC Profiling of Mimosa Pudica Polyphenols and Their Non-Invasive Biophysical Investigations for Anti-Dermatoheliotic and Skin Reinstating Potential. Biomed. Pharmacother. 2019, 109, 865–875. [Google Scholar] [CrossRef]
- Zhang, Z.-S.; Li, D.; Wang, L.-J.; Ozkan, N.; Chen, X.D.; Mao, Z.-H.; Yang, H.-Z. Optimization of Ethanol–Water Extraction of Lignans from Flaxseed. Sep. Purif. Technol. 2007, 57, 17–24. [Google Scholar] [CrossRef]
- Nawaz, H.; Shad, M.A.; Rehman, N.; Andaleeb, H.; Ullah, N. Effect of Solvent Polarity on Extraction Yield and Antioxidant Properties of Phytochemicals from Bean (Phaseolus vulgaris) Seeds. Braz. J. Pharm. Sci. 2020, 56, 56. [Google Scholar] [CrossRef]
- Barchan, A.; Bakkali, M.; Arakrak, A.; Pagán, R.; Laglaoui, A. The Effects of Solvents Polarity on the Phenolic Contents and Antioxidant Activity of Three Mentha Species Extracts. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 399–412. [Google Scholar]
- Aykul, S.; Martinez-Hackert, E. Determination of Half-Maximal Inhibitory Concentration Using Biosensor-Based Protein Interaction Analysis. Anal. Biochem. 2016, 508, 97–103. [Google Scholar] [CrossRef]
- Ismail-Suhaimy, N.W.; Gani, S.S.A.; Zaidan, U.H.; Halmi, M.I.E.; Bawon, P. Optimizing Conditions for Microwave-Assisted Extraction of Polyphenolic Content and Antioxidant Activity of Barleria Lupulina Lindl. Plants 2021, 10, 682. [Google Scholar] [CrossRef] [PubMed]
- Dahmoune, F.; Nayak, B.; Moussi, K.; Remini, H.; Madani, K. Optimization of Microwave-Assisted Extraction of Polyphenols from Myrtus communis L. Leaves. Food Chem. 2015, 166, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Tunna, T.S.; Zaidul, I.S.M.; Ahmed, Q.U.; Ghafoor, K.; Al-Juhaimi, F.Y.; Uddin, M.S.; Hasan, M.; Ferdous, S. Analyses and Profiling of Extract and Fractions of Neglected Weed Mimosa Pudica Linn. Traditionally Used in Southeast Asia to Treat Diabetes. S. Afr. J. Bot. 2015, 99, 144–152. [Google Scholar] [CrossRef]
- Weremfo, A.; Adulley, F.; Adarkwah-Yiadom, M. Simultaneous Optimization of Microwave-Assisted Extraction of Phenolic Compounds and Antioxidant Activity of Avocado (Persea Americana Mill.) Seeds Using Response Surface Methodology. J. Anal. Methods Chem. 2020, 2020, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Liyana-Pathirana, C.M.; Shahidi, F. Antioxidant and Free Radical Scavenging Activities of Whole Wheat and Milling Fractions. Food Chem. 2007, 101, 1151–1157. [Google Scholar] [CrossRef]
- Guido, L.F.; Moreira, M.M. Techniques for Extraction of Brewer’s Spent Grain Polyphenols: A Review. Food Bioprocess Technol. 2017, 10, 1192–1209. [Google Scholar] [CrossRef]
- Viacava, G.E.; Roura, S.I.; Agüero, M.V. Optimization of Critical Parameters during Antioxidants Extraction from Butterhead Lettuce to Simultaneously Enhance Polyphenols and Antioxidant Activity. Chemom. Intell. Lab. Syst. 2015, 146, 47–54. [Google Scholar] [CrossRef]
- Ogbole, O.O.; Segun, P.A.; Adeniji, A.J. In Vitro Cytotoxic Activity of Medicinal Plants from Nigeria Ethnomedicine on Rhabdomyosarcoma Cancer Cell Line and HPLC Analysis of Active Extracts. BMC Complementary Altern. Med. 2017, 17, 494. [Google Scholar] [CrossRef] [PubMed]
- Mousinho, N.M.H.D.C.; van Tonder, J.J.; Steenkamp, V. In Vitro Anti-Diabetic Activity of Sclerocarya Birrea and Ziziphus Mucronata. Nat. Prod. Commun. 2013, 8, 1279–1284. [Google Scholar] [PubMed]
- Chaves, J.O.; de Souza, M.C.; da Silva, L.C.; Lachos-Perez, D.; Torres-Mayanga, P.C.; da Fonseca Machado, A.P.; Forster-Carneiro, T.; Vázquez-Espinosa, M.; González-de-Peredo, A.V.; Barbero, G.F.; et al. Extraction of Flavonoids from Natural Sources Using Modern Techniques. Front. Chem. 2020, 8, 8. [Google Scholar] [CrossRef]
- Zhang, J.; Yuan, K.; Zhou, W.; Zhou, J.; Yang, P. Studies on the Active Components and Antioxidant Activities of the Extracts of Mimosa Pudica Linn. from Southern China. Pharmacogn. Mag. 2011, 7, 35–39. [Google Scholar]
- Zhang, H.-F.; Yang, X.-H.; Wang, Y. Microwave Assisted Extraction of Secondary Metabolites from Plants: Current Status and Future Directions. Trends Food Sci. Technol. 2011, 22, 672–688. [Google Scholar] [CrossRef]
- Sedraoui, S.; Badr, A.; Barba, M.G.M.; Doyen, A.; Tabka, Z.; Desjardins, Y. Optimization of the Ultrahigh-Pressure–Assisted Extraction of Phenolic Compounds and Antioxidant Activity from Palm Dates (Phoenix dactylifera L.). Food Anal. Methods 2020, 13, 1556–1569. [Google Scholar] [CrossRef]
- Jagetia, G.C.; Vanlalhruaii, F. Anticancer Potential of Mimosa Pudica Linn. Lajwanti in Cultured Dalton’s Ascites Lymphoma Cells. Int. J. Complementary Altern. Med. 2020, 13, 91–94. [Google Scholar]
- Satoh, T. Molecular Mechanisms for the Regulation of Insulin-Stimulated Glucose Uptake by Small Guanosine Triphosphatases in Skeletal Muscle and Adipocytes. Int. J. Mol. Sci. 2014, 15, 18677–18692. [Google Scholar] [CrossRef] [PubMed]
- Sunday, R.M.; Obuotor, E.M.; Kumar, A. Antioxidant and Antidiabetic Properties of Mimosa Pudica Seeds in Streptozotocin-Induced Diabetic Wistar Rats. Asian J. Biotechnol. 2019, 12, 1–8. [Google Scholar] [CrossRef][Green Version]
- Parasuraman, S.; Ching, T.H.; Leong, C.H.; Banik, U. Antidiabetic and Antihyperlipidemic Effects of a Methanolic Extract of Mimosa Pudica (Fabaceae) in Diabetic Rats. Egypt. J. Basic Appl. Sci. 2019, 6, 137–148. [Google Scholar] [CrossRef]
- Rajendiran, D.; Khan, H.B.H.; Packirisamy, S.; Gunasekaran, K. Dose Dependent Antidiabetic Effect of Mimosa Pudica Leaves Extract in Type 2 Diabetic Rat Model. Pharma Innov. J. 2019, 8, 1–4. [Google Scholar]
- Yupparach, P.; Konsue, A. Hypoglycemic and Hypolipidemic Activities of Ethanolic Extract from Mimosa pudica L. in Normal and Streptozotocin-Induced Diabetic Rats. Pharmacogn. J. 2017, 9, 834–837. [Google Scholar] [CrossRef]
- Ha, D.T.; Nam Trung, T.; Bich Thu, N.; Van On, T.; Hai Nam, N.; Van Men, C.; Thi Phuong, T.; Bae, K. Adlay Seed Extract (Coix Lachryma-Jobi L.) Decreased Adipocyte Differentiation and Increased Glucose Uptake in 3T3-L1 Cells. J. Med. Food 2010, 13, 1331–1339. [Google Scholar] [CrossRef] [PubMed]
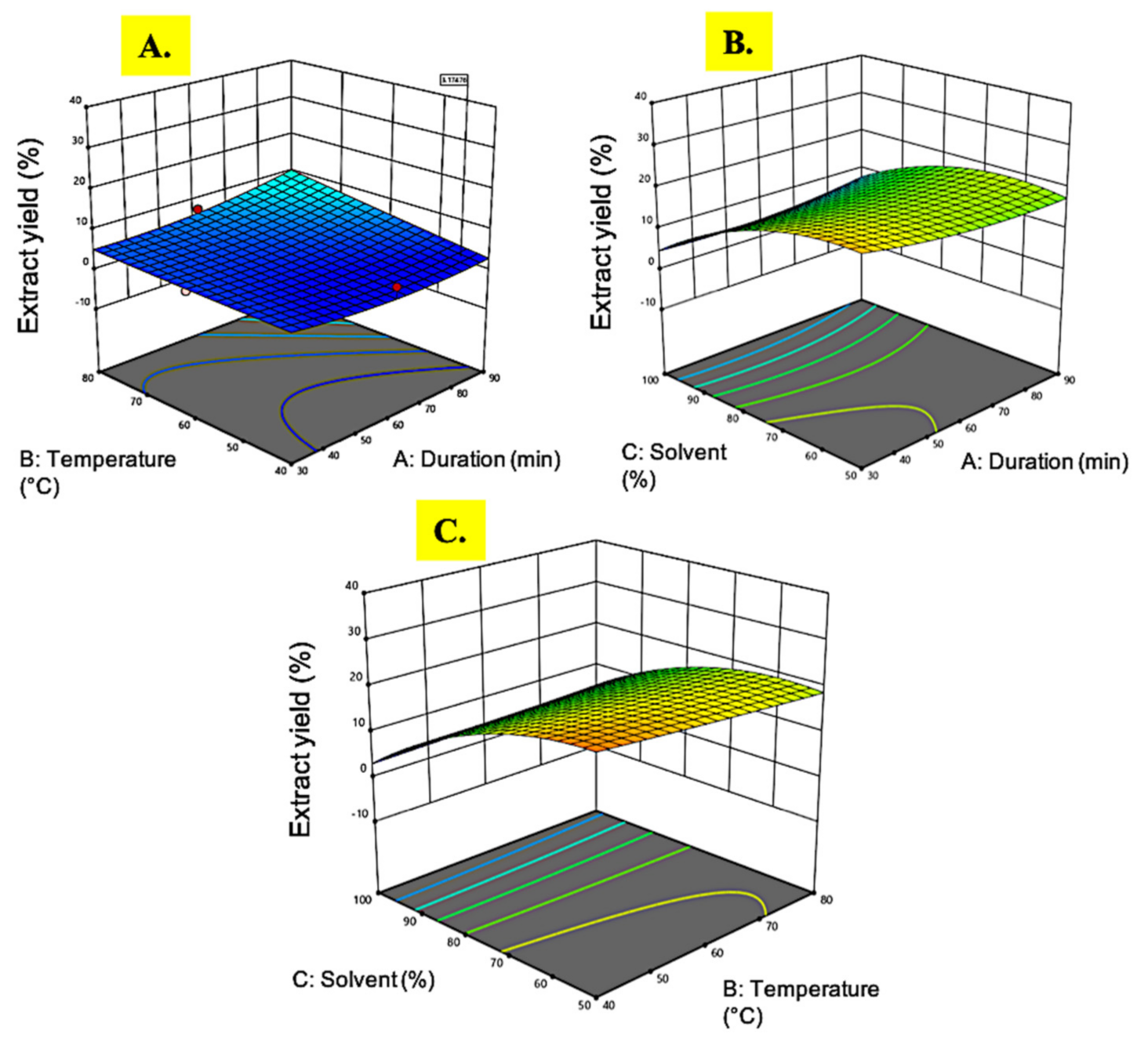
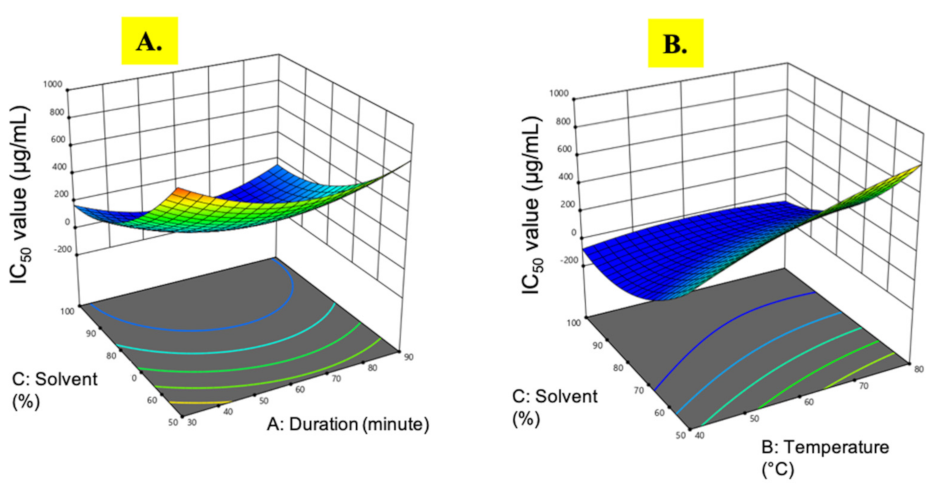
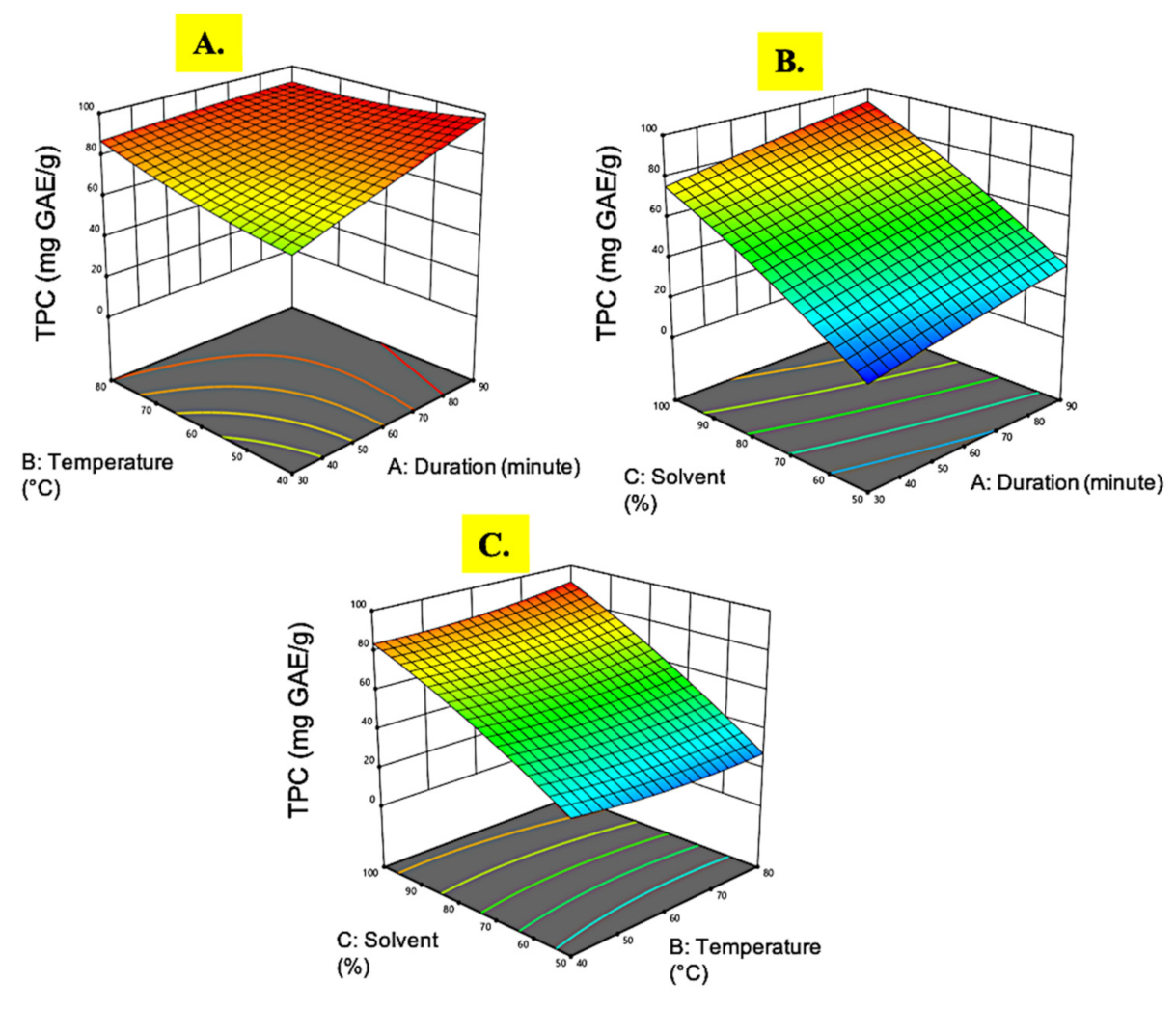
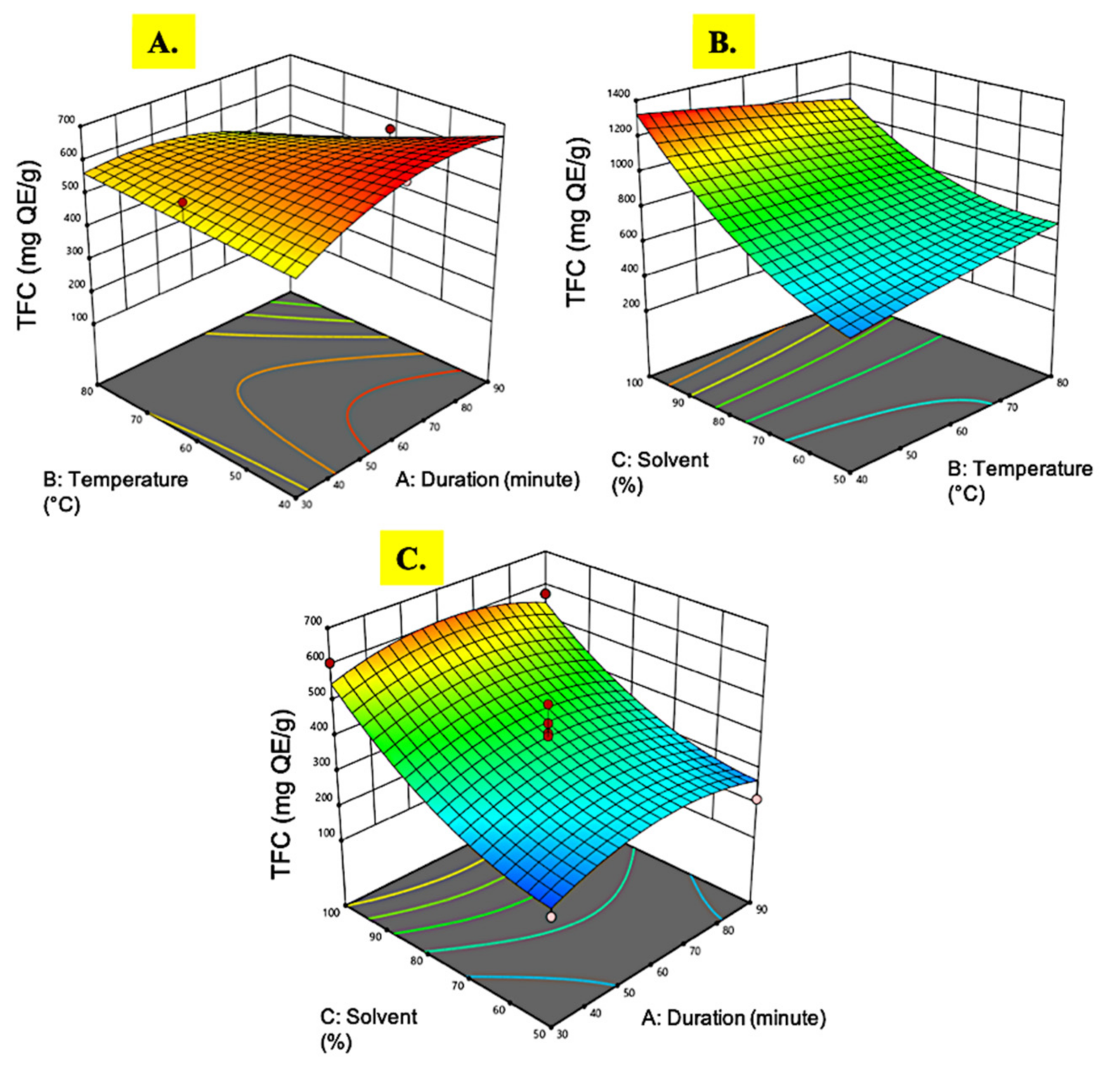
| Run | Extraction Time (min) | Temperature (°C) | Ethanol (%) | Extract Yield (%) | DPPH (IC50, µg/mL) | Phenolic Content (mg GAE/g) | Flavonoid Content (mg QE/g) |
|---|---|---|---|---|---|---|---|
| A | B | C | Y1 | Y2 | Y3 | Y4 | |
| 1 | 30 | 60 | 100 | 4.0 | 72.0 | 76.03 | 606.31 |
| 2 | 30 | 60 | 50 | 25.9 | 975.03 | 14.51 | 209.44 |
| 3 | 60 | 40 | 100 | 5.0 | 42.0 | 85.87 | 660.06 |
| 4 | 30 | 80 | 75 | 17.0 | 424.0 | 62.79 | 415.69 |
| 5 | 60 | 60 | 75 | 13.6 | 106.5 | 42.39 | 260.69 |
| 6 | 60 | 80 | 100 | 7.3 | 45.0 | 80.07 | 476.31 |
| 7 | 90 | 40 | 75 | 15.4 | 73.2 | 68.9 | 385.69 |
| 8 | 60 | 60 | 75 | 23.1 | 283.5 | 59.06 | 436.31 |
| 9 | 60 | 60 | 75 | 13.9 | 58.67 | 65.42 | 400.06 |
| 10 | 90 | 60 | 100 | 3.1 | 51.5 | 91.98 | 574.44 |
| 11 | 60 | 80 | 50 | 14.6 | 689.0 | 20.82 | 357.56 |
| 12 | 60 | 60 | 75 | 16.2 | 82.5 | 69.96 | 490.06 |
| 13 | 60 | 60 | 75 | 14.0 | 100.2 | 49.66 | 408.81 |
| 14 | 30 | 40 | 75 | 17.3 | 170.5 | 34.36 | 204.44 |
| 15 | 90 | 60 | 50 | 17.9 | 819.92 | 31.38 | 214.44 |
| 16 | 90 | 80 | 75 | 21.5 | 358.0 | 70.52 | 335.06 |
| 17 | 60 | 40 | 50 | 18.6 | 220.0 | 39.71 | 341.94 |
| Extract Yield | IC50 Value | ||||||||
| Sources | df | Sum of Squares | Mean Square | F-Value | p-Value | Sum of Squares | Mean Square | F-Value | p-Value |
| Model | 9 | 563.34 | 62.59 | 3.67 | 0.0501 | 1.259 × 106 | 1.574 × 105 | 10.22 | 0.0018 |
| A-Extraction time | 1 | 4.96 | 4.96 | 0.2912 | 0.6062 | 14,357.50 | 14,357.50 | 0.9317 | 0.3627 |
| B-Temperature | 1 | 2.10 | 2.10 | 0.1233 | 0.7358 | 1.276 × 105 | 1.276 × 105 | 8.28 | 0.0206 |
| C-Ethanol | 1 | 414.72 | 414.72 | 24.34 | 0.0017 | 7.772 × 105 | 7.772 × 105 | 50.43 | 0.0001 |
| Interaction | |||||||||
| AB | 1 | 10.24 | 10.24 | 0.6010 | 0.4636 | *et | *et | *et | *et |
| AC | 1 | 12.60 | 12.60 | 0.7397 | 0.4182 | 4529.96 | 4529.96 | 0.2939 | 0.6025 |
| BC | 1 | 9.92 | 9.92 | 0.5824 | 0.4703 | 54,289.00 | 54,289.00 | 3.52 | 0.0974 |
| Square | |||||||||
| A2 | 1 | 9.41 | 9.41 | 0.5524 | 0.4815 | 1.370 × 105 | 1.370 × 105 | 8.89 | 0.0176 |
| B2 | 1 | 0.0885 | 0.0885 | 0.0052 | 0.9446 | 10623.70 | 10,623.70 | 0.6894 | 0.4305 |
| C2 | 1 | 102.34 | 102.34 | 6.01 | 0.0441 | 1.260 × 105 | 1.260 × 105 | 8.17 | 0.0212 |
| Residual | 7 | 119.26 | 17.04 | 1.233 × 105 | 15,410.79 | ||||
| Lack of Fit | 3 | 54.77 | 18.26 | 1.13 | 0.4364 | 91,008.99 | 22,752.25 | 2.82 | 0.1697 |
| Pure Error | 4 | 64.49 | 16.12 | 32,277.34 | 8069.34 | ||||
| R2 | 0.8253 | 0.9108 | |||||||
| Adequate precision | 6.4440 | 10.4044 | |||||||
| TPC | TFC | ||||||||
| Sources | df | Sum of Squares | Mean Square | F-Value | p-Value | Sum of Squares | Mean Square | F-Value | p-Value |
| Model | 9 | 7469.68 | 829.96 | 6.24 | 0.0123 | 2.369 × 105 | 29608.63 | 4.13 | 0.0304 |
| A-Extraction time | 1 | 705.02 | 705.02 | 5.30 | 0.0548 | 679.88 | 679.88 | 0.0949 | 0.7659 |
| B-Temperature | 1 | 3.59 | 3.59 | 0.0270 | 0.8741 | 7.05 | 7.05 | 0.0010 | 0.9757 |
| C-Ethanol | 1 | 6471.01 | 6471.01 | 48.6 | 0.0002 | 1.781 × 105 | 1.781 × 105 | 24.87 | 0.0011 |
| Interaction | |||||||||
| AB | 1 | 179.69 | 179.69 | 1.35 | 0.2831 | 17145.28 | 17145.28 | 2.39 | 0.1604 |
| AC | 1 | 0.2116 | 0.2116 | 0.0016 | 0.9693 | 339.85 | 339.85 | 0.0475 | 0.8330 |
| BC | 1 | 42.84 | 42.84 | 0.3222 | 0.5880 | 9937.10 | 9937.10 | 1.39 | 0.2727 |
| Square | |||||||||
| A2 | 1 | 1.77 | 1.77 | 0.0133 | 0.9113 | 15,736.65 | 15736.65 | 2.20 | 0.1765 |
| B2 | 1 | 26.18 | 26.18 | 0.1969 | 0.6706 | *et | *et | *et | *et |
| C2 | 1 | 42.42 | 42.42 | 0.3191 | 0.5898 | 16,597.46 | 16597.46 | 2.32 | 0.1664 |
| Residual | 7 | 930.61 | 132.94 | 57,291.98 | 7161.50 | ||||
| Lack of Fit | 3 | 420.62 | 140.21 | 1.10 | 0.4463 | 28,381.17 | 7095.29 | 0.9817 | 0.5069 |
| Pure Error | 4 | 509.99 | 127.50 | 28,910.80 | 7227.70 | ||||
| R2 | 0.8892 | 0.8052 | |||||||
| Adequate precision | 8.5551 | 6.6924 | |||||||
| A: Extraction Time | B: Temperature | C: Ethanol Concentration | Crude Extract Yield | IC50 Value | TPC | TFC | |
|---|---|---|---|---|---|---|---|
| A: Extraction time | |||||||
| B: Temperature | |||||||
| C: Ethanol concentration | |||||||
| Extract Yield | −0.085 | 0.055 | −0.779 | 0.612 | −0.658 | −0.749 | |
| IC50 value | −0.102 | 0.304 | −0.750 | 0.612 | −0.743 | −0.600 | |
| TPC | 0.290 | 0.021 | 0.878 | −0.658 | −0.743 | 0.832 | |
| TFC | 0.048 | −0.005 | 0.778 | −0.749 | −0.600 | 0.832 |
| IC50 Value, µg/mL | TPC, mg GAE/g | TFC, mg QE/g | |
|---|---|---|---|
| Predicted | 27.87 | 92.11 | 664.73 |
| Actual | 51.02 ± 2.07 | 68.5 ± 3.15 | 659.85 ± 31.46 |
| Similarity percentage, % | 54.63 | 74.36 | 99.27 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baharuddin, N.S.; Roslan, M.A.M.; Bawzer, M.A.M.; Mohamad Azzeme, A.; Rahman, Z.A.; Khayat, M.E.; Rahman, N.A.A.; Sobri, Z.M. Response Surface Optimization of Extraction Conditions and In Vitro Antioxidant and Antidiabetic Evaluation of an Under-Valued Medicinal Weed, Mimosa pudica. Plants 2021, 10, 1692. https://doi.org/10.3390/plants10081692
Baharuddin NS, Roslan MAM, Bawzer MAM, Mohamad Azzeme A, Rahman ZA, Khayat ME, Rahman NAA, Sobri ZM. Response Surface Optimization of Extraction Conditions and In Vitro Antioxidant and Antidiabetic Evaluation of an Under-Valued Medicinal Weed, Mimosa pudica. Plants. 2021; 10(8):1692. https://doi.org/10.3390/plants10081692
Chicago/Turabian StyleBaharuddin, Nor Saffana, Muhamad Aidilfitri Mohamad Roslan, Mohsen Ahmed Mohammed Bawzer, Azzreena Mohamad Azzeme, Zuraida Ab Rahman, Mohd Ezuan Khayat, Nor Aini Abdul Rahman, and Zulfazli M. Sobri. 2021. "Response Surface Optimization of Extraction Conditions and In Vitro Antioxidant and Antidiabetic Evaluation of an Under-Valued Medicinal Weed, Mimosa pudica" Plants 10, no. 8: 1692. https://doi.org/10.3390/plants10081692
APA StyleBaharuddin, N. S., Roslan, M. A. M., Bawzer, M. A. M., Mohamad Azzeme, A., Rahman, Z. A., Khayat, M. E., Rahman, N. A. A., & Sobri, Z. M. (2021). Response Surface Optimization of Extraction Conditions and In Vitro Antioxidant and Antidiabetic Evaluation of an Under-Valued Medicinal Weed, Mimosa pudica. Plants, 10(8), 1692. https://doi.org/10.3390/plants10081692









