Chemical Composition and New Biological Activities of Essential Oil and Hydrosol of Hypericum perforatum L. ssp. veronense (Schrank) H. Lindb
Abstract
1. Introduction
2. Results and Discussion
2.1. Gas Chromatography and Mass Spectrometry (GC-MS) Analysis of the Free Volatile Compounds from Essential Oils and Hydrosol
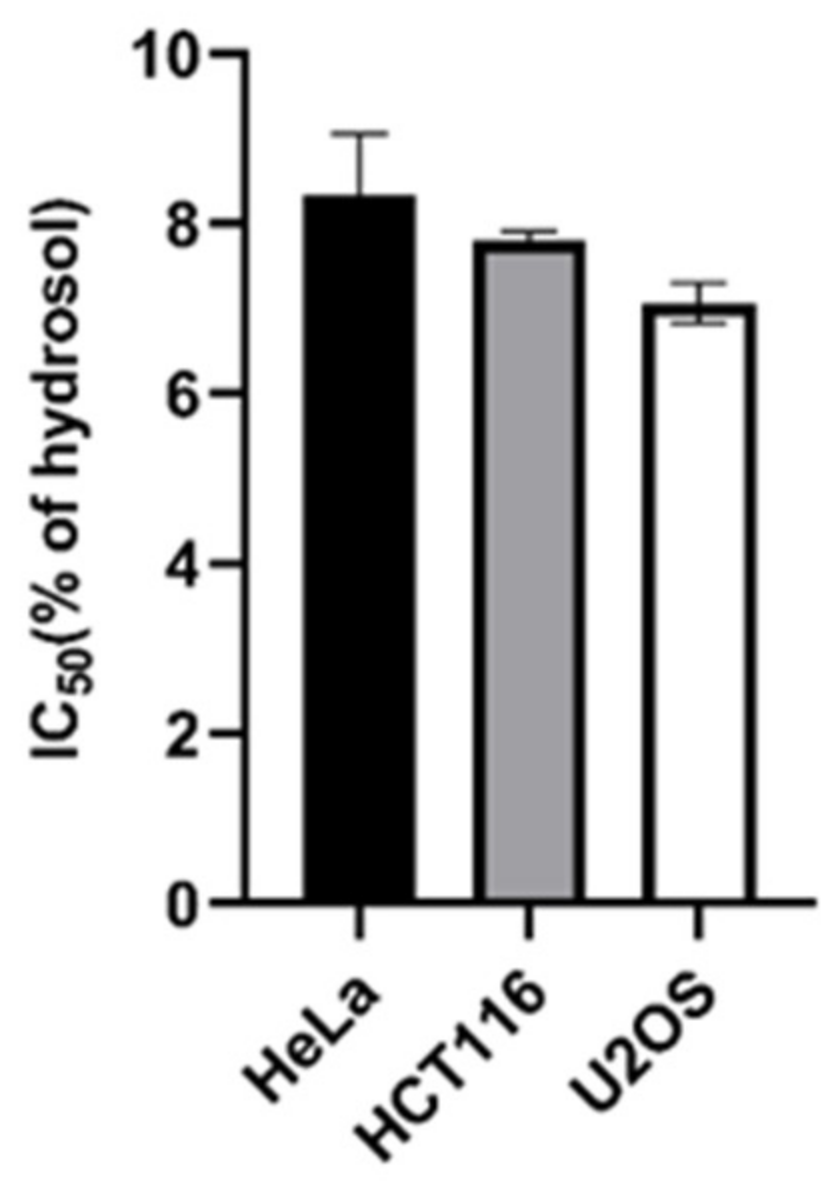
2.2. Antiproliferative Activity
2.3. Antioxidant Activity
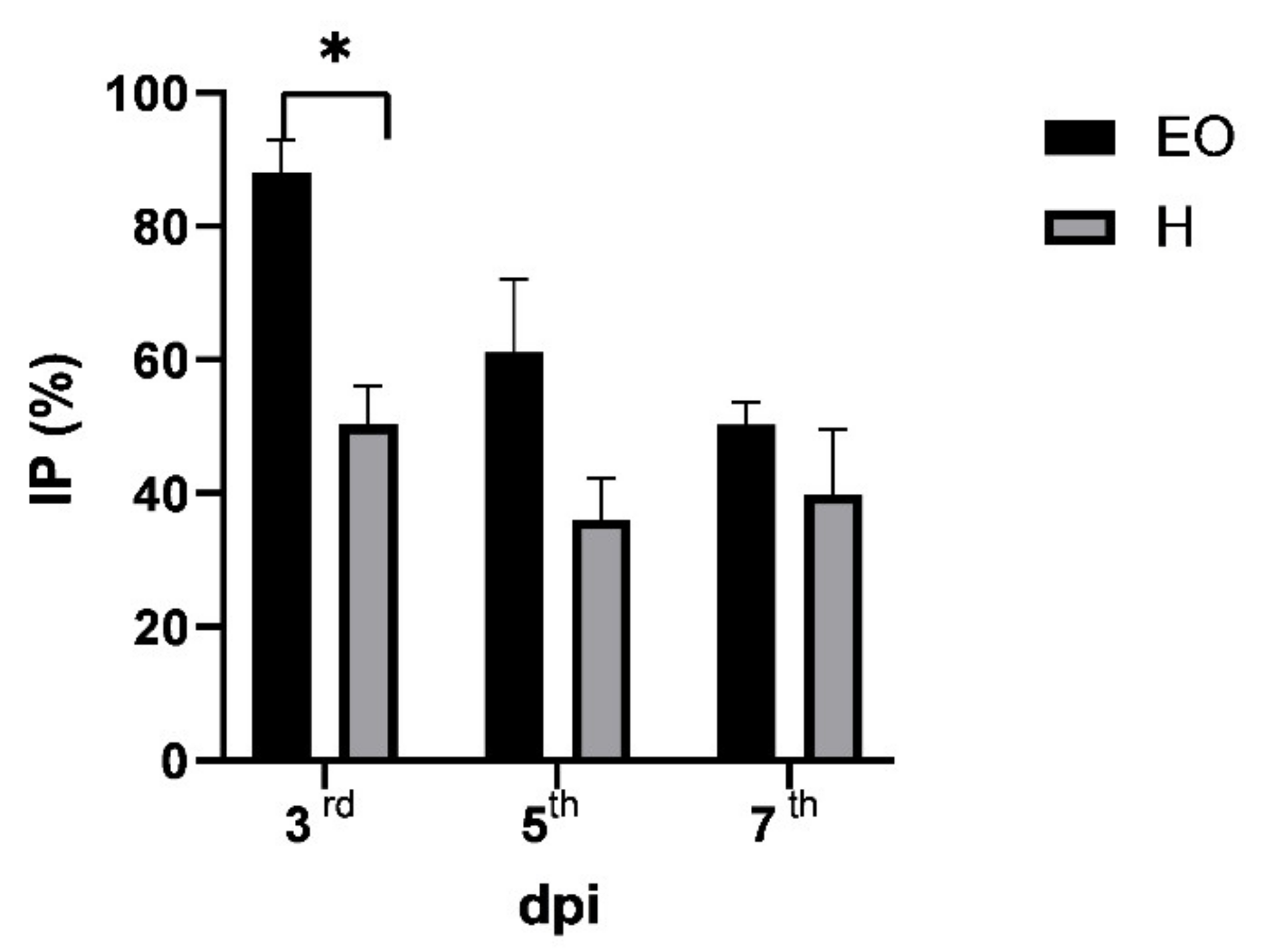
2.4. Antiphytoviral Activity
3. Materials and Methods
3.1. Herbal Material
3.2. GC and GC-MS Analyses
3.3. Antiproliferative Analysis
3.4. Antioxidant Activity
3.4.1. Oxygen Radical Absorbance Capacity Assay (ORAC)
3.4.2. Measurement of the DPPH Radical Scavenging Activity
3.5. Antiphytoviral Activity
3.5.1. Virus and Plant Hosts
3.5.2. Antiphytoviral Activity Assay
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schepetkin, I.; Özek, G.; Özek, T.; Kirpotina, L.; Khlebnikov, A.; Quinn, M. Chemical Composition and Immunomodulatory Activity of Hypericum perforatum Essential Oils. Biomolecules 2020, 10, 916. [Google Scholar] [CrossRef] [PubMed]
- Galeotti, N. Hypericum perforatum (St John’s wort) beyond depression: A therapeutic perspective for pain conditions. J. Ethnopharmacol. 2017, 200, 136–146. [Google Scholar] [CrossRef]
- Rocha, L.; Marston, A.; Potterat, O.; Kaplan, M.A.C.; Stoeckli-Evans, H.; Hostettmann, K. Antibacterial phloroglucinols and flavonoids from Hypericum brasiliense. Phytochemistry 1995, 40, 1447–1452. [Google Scholar] [CrossRef]
- Shafaghat, A. Antioxidant, Antimicrobial Activities and Fatty Acid Components of Flower, Leaf, Stem and Seed of Hypericum scabrum. Nat. Prod. Commun. 2011, 6, 1739–1742. [Google Scholar] [CrossRef]
- Saroglou, V.; Marin, P.D.; Rancic, A.; Veljic, M.; Skaltsa, H. Composition and antimicrobial activity of the essential oil of six Hypericum species from Serbia. Biochem. Syst. Ecol. 2007, 35, 146–152. [Google Scholar] [CrossRef]
- Dell’Aica, I.; Caniato, R.; Biggin, S.; Garbisa, S. Matrix proteases, green tea, and St. John’s wort: Biomedical research catches up with folk medicine. Clin. Chim. Acta 2007, 381, 69–77. [Google Scholar] [CrossRef]
- Cakir, A.; Kordali, S.; Kilic, H.; Kaya, E. Antifungal properties of essential oil and crude extracts of Hypericum linarioides Bosse. Biochem. Syst. Ecol. 2005, 33, 245–256. [Google Scholar] [CrossRef]
- Cakir, A.; Kordali, S.; Zengin, H.; Izumi, S.; Hirata, T. Composition and antifungal activity of essential oils isolated from Hypericum hyssopifolium and Hypericum heterophyllum. Flavour Fragr. J. 2004, 19, 62–68. [Google Scholar] [CrossRef]
- Bilia, A.R.; Gallori, S.; Vincieri, F.F. St John’s wort and depression. Prescrire Int. 2004, 13, 3077–3096. [Google Scholar] [CrossRef]
- Gudžić, B.; Djokovic, D.; Vajs, V.; Palić, R.; Stojanovic, G. Composition and antimicrobial activity of the essential oil of Hypericum maculatum Crantz. Flavour Fragr. J. 2002, 17, 392–394. [Google Scholar] [CrossRef]
- Couladis, M.; Baziou, P.; Petrakis, P.V.; Harvala, C. Essential oil composition of Hypericum perfoliatum L. growing in different locations in Greece. Flavour Fragr. J. 2001, 16, 204–206. [Google Scholar] [CrossRef]
- Flora Croatica Database: 28954. Available online: https://hirc.botanic.hr/fcd/DetaljiFrame.aspx?IdVrste=28954&taxon=Hypericum+perforatum+L.+ssp.+veronense+(Schrank)+H.+Lindb (accessed on 28 April 2021).
- Pignatti, S. Flora d’Italia; Edagricole: Bologna, Italy, 1982; Volumes I–III. [Google Scholar]
- Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Valentine, D.H.; Walters, S.M.; Webb, D.A. Flora Europaea; Cambridge University Press: London, UK, 1968–1980; Volumes 1–5. [Google Scholar]
- Zidorn, C. Plant chemophenetics—A new term for plant chemosystematics/plant chemotaxonomy in the macro-molecular era. Phytochemistry 2019, 163, 147–148. [Google Scholar] [CrossRef]
- Djordjevic, A. Chemical composition of Hypericum perforatum L. Essential oil. Adv. Technol. 2015, 4, 64–68. [Google Scholar] [CrossRef]
- Quassinti, L.; Lupidi, G.; Maggi, F.; Sagratini, G.; Papa, F.; Vittori, S.; Bianco, A.; Bramucci, M. Antioxidant and antiproliferative activity of Hypericum hircinum L. subsp. majus (Aiton) N. Robson essential oil. Nat. Prod. Res. 2013, 27, 862–868. [Google Scholar] [CrossRef]
- França, H.S.; Rocha, L.; Fernande, C.P.; Ruiz, A.L.T.G.; de Carvalho, J.E. Antiproliferative activity of the hexanic extract and phloroglucinols from Hypericum brasiliense. Rev. Bras. Farmacogn. 2013, 23, 844–847. [Google Scholar] [CrossRef][Green Version]
- Zorzetto, C.; Sánchez-Mateo, C.C.; Rabanal, R.M.; Lupidi, G.; Petrelli, D.; Vitali, L.A.; Bramucci, M.; Quassinti, L.; Caprioli, G.; Papa, F.; et al. Phytochemical analysis and in vitro biological activity of three Hypericum species from the Canary Islands (Hypericum reflexum, Hypericum canariense and Hypericum grandifolium). Fitoterapia 2015, 100, 95–109. [Google Scholar] [CrossRef]
- Güzey, G.; Ibadova, S.; Öztürk, Y.; Öztürk, N.; Maggi, F.; Sagratini, G.; Ricciutelli, M.; Vittori, S. Antiproliferative and antioxidant effects of three Hypericum species of Turkish origin: H. perforatum, H. montbretii and H. origanifolium. Med. Aromat. Plant. Sci. Biotechnol. 2011, 5, 91–99. [Google Scholar]
- Napoli, E.; Siracusa, L.; Ruberto, G.; Carrubba, A.; Lazzara, S.; Speciale, A.; Cimino, F.; Saija, A.; Cristani, M. Phytochemical profiles, phototoxic and antioxidant properties of eleven Hypericum species—A comparative study. Phytochemistry 2018, 152, 162–173. [Google Scholar] [CrossRef]
- Ivanova, D.; Gerova, D.; Chervenkov, T.; Yankova, T. Polyphenols and antioxidant capacity of Bulgarian medicinal plants. J. Ethnopharmacol. 2005, 96, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Vuko, E.; Rusak, G.; Dunkić, V.; Kremer, D.; Kosalec, I.; Rađa, B.; Bezić, N. Inhibition of Satellite RNA Associated Cucumber Mosaic Virus Infection by Essential Oil of Micromeria croatica (Pers.) Schott. Molecules 2019, 24, 1342. [Google Scholar] [CrossRef]
- Dunkić, V.; Vuko, E.; Bezić, N.; Kremer, D.; Ruščić, M. Composition and antiviral activity of the essential oils of Eryngium alpinum and E. amethystinum. Chem. Biodivers. 2013, 10, 1894–1902. [Google Scholar] [CrossRef] [PubMed]
- Vuko, E.; Dunkić, V.; Bezić, N.; Ruščić, M.; Kremer, D. Chemical Composition and Antiphytoviral Activity of Essential Oil of Micromeria graeca. Nat. Prod. Commun. 2012, 7, 1934578X1200700. [Google Scholar] [CrossRef]
- Bezić, N.; Vuko, E.; Dunkić, V.; Ruščić, M.; Blažević, I.; Burčul, F. Antiphytoviral Activity of Sesquiterpene-Rich Essential Oils from Four Croatian Teucrium Species. Molecules 2011, 16, 8119–8129. [Google Scholar] [CrossRef] [PubMed]
- Dunkić, V.; Bezić, N.; Vuko, E. Antiphytoviral Activity of Essential Oil from Endemic Species Teucrium arduini. Nat. Prod. Commun. 2011, 6, 1934578X1100600. [Google Scholar] [CrossRef]
- Dunkic, V.; Bezic, N.; Vuko, E.; Cukrov, D. Antiphytoviral activity of satureja montana L. ssp. variegata (host) P. W. Ball essential oil and phenol compounds on CMV and TMV. Molecules 2010, 15, 6713–6721. [Google Scholar] [CrossRef]
- Bishop, C.D. Antiviral activity of the essential oil of melaleuca alternifolia (Maiden amp; Betche) cheel (tea tree) against tobacco mosaic virus. J. Essent. Oil Res. 1995, 7, 641–644. [Google Scholar] [CrossRef]
- Hamedi, A.; Pasdaran, A.; Zebarjad, Z.; Moein, M. A Survey on Chemical Constituents and Indications of Aromatic Waters Soft Drinks (Hydrosols) Used in Persian Nutrition Culture and Folk Medicine for Neurological Disorders and Mental Health. J. Evid. Based Complementary Altern. Med. 2017, 22, 744–752. [Google Scholar] [CrossRef]
- Maggi, F.; Ferretti, G. Essential oil comparison of hypericum perforatum l. Subsp. perforatum and subsp. Veronense (schrank) ces. From central italy. J. Essent. Oil Res. 2008, 20, 492–494. [Google Scholar] [CrossRef]
- Schwob, I.; Bessière, J.M.; Viano, J. Composition of the essential oils of Hypericum perforatum L. from southeastern France. Comptes Rendus Biol. 2002, 325, 781–785. [Google Scholar] [CrossRef]
- Smelcerovic, A.; Mimica-Dukic, N.; Djordjevic, S. Essential oil composition of hypericum perforatum l. ssp. angustifolium from South Serbia. J. Essent. Oil-Bearing Plants 2004, 7, 275–278. [Google Scholar] [CrossRef]
- Cirak, C.; Radusiene, J.; Jakstas, V.; Ivanauskas, L.; Seyis, F.; Yayla, F. Altitudinal changes in secondary metabolite contents of Hypericum androsaemum and Hypericum polyphyllum. Biochem. Syst. Ecol. 2017, 70, 108–115. [Google Scholar] [CrossRef]
- Łotocka, B.; Osińska, E. Shoot anatomy and secretory structures in Hypericum species (Hypericaceae). Bot. J. Linn. Soc. 2010, 163, 70–86. [Google Scholar] [CrossRef][Green Version]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry; Allured Publishing: Carol Stream, IL, USA, 2017. [Google Scholar]
- NIST WebBook. Available online: https://webbook.nist.gov/ (accessed on 14 May 2021).
- Keser, S.; Keser, F.; Kaygili, O.; Tekin, S.; Demir, E.; Turkoglu, I.; Turkoglu, S.; Parlak, A.E.; Yilmaz, O.; Karatepe, M.; et al. Phytochemical compounds and antiradical, antimicrobial, and cytotoxic activities of the extracts from Hypericum scabrum L. Flowers. Nat. Prod. Res. 2020, 34, 714–719. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Ting, Z.; Qiu, X.; Zhang, X.; Gan, X.; Fang, Y.; Xu, X.; Xu, R. Linalool preferentially induces robust apoptosis of a variety of leukemia cells via upregulating p53 and cyclin-dependent kinase inhibitors. Toxicology 2010, 268, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Cherng, J.M.; Shieh, D.E.; Chiang, W.; Chang, M.Y.; Chiang, L.C. Chemopreventive effects of minor dietary constituents in common foods on human cancer cells. Biosci. Biotechnol. Biochem. 2007, 71, 1500–1504. [Google Scholar] [CrossRef]
- Ravizza, R.; Gariboldi, M.B.; Molteni, R.; Monti, E. Linalool, a plant-derived monoterpene alcohol, reverses doxorubicin resistance in human breast adenocarcinoma cells. Oncol. Rep. 2008, 20, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Gomes, B.S.; Neto, B.P.S.; Lopes, E.M.; Cunha, F.V.M.; Araújo, A.R.; Wanderley, C.W.S.; Wong, D.V.T.; Júnior, R.C.P.L.; Ribeiro, R.A.; Sousa, D.P.; et al. Anti-inflammatory effect of the monoterpene myrtenol is dependent on the direct modulation of neutrophil migration and oxidative stress. Chem. Biol. Interact. 2017, 273, 73–81. [Google Scholar] [CrossRef]
- Sisay, M.; Gashaw, T. Ethnobotanical, Ethnopharmacological, and Phytochemical Studies of Myrtus communis Linn: A Popular Herb in Unani System of Medicine. J. Evidence-Based Complement. Altern. Med. 2017, 22, 1035–1043. [Google Scholar] [CrossRef]
- Paknejadi, M.; Foroohi, F.; Yousefzadi, M. Antimicrobial activities of the essential oils of five Salvia species from teheran province; Iran. J. Paramed. Sci. 2012, 3, 12–18. [Google Scholar] [CrossRef]
- Coté, H.; Boucher, M.-A.; Pichette, A.; Legault, J. Anti-Inflammatory, Antioxidant, Antibiotic, and Cytotoxic Activities of Tanacetum vulgare L. Essential Oil and Its Constituents. Medicines 2017, 4, 34. [Google Scholar] [CrossRef]
- Kang, S.H.; Kim, Y.S.; Kim, E.K.; Hwang, J.W.; Jeong, J.H.; Dong, X.; Lee, J.W.; Moon, S.H.; Jeon, B.T.; Park, P.J. Anticancer effect of thymol on AGS human gastric carcinoma cells. J. Microbiol. Biotechnol. 2015, 26, 28–37. [Google Scholar] [CrossRef]
- Nagoor Meeran, M.F.; Javed, H.; Al Taee, H.; Azimullah, S.; Ojha, S.K. Pharmacological properties and molecular mechanisms of thymol: Prospects for its therapeutic potential and pharmaceutical development. Front. Pharmacol. 2017, 8, 380. [Google Scholar] [CrossRef]
- Arab, H.-A.; Fathi, M.; Mortezai, E.; Hosseinimehr, S.J. Chemoprotective effect of thymol against genotoxicity induced by bleomycin in human lymphocytes. Pharm. Biomed. Res. 2015, 1, 26–31. [Google Scholar] [CrossRef][Green Version]
- Lee, K.P.; Kim, J.E.; Park, W.H.; Hong, H. Regulation of C6 glioma cell migration by thymol. Oncol. Lett. 2016, 11, 2619–2624. [Google Scholar] [CrossRef]
- Dai, W.; Sun, C.; Huang, S.; Zhou, Q. Carvacrol suppresses proliferation and invasion in human oral squamous cell carcinoma. OncoTargets Ther. 2016, 9, 2297–2304. [Google Scholar] [CrossRef]
- Liolios, C.C.; Gortzi, O.; Lalas, S.; Tsaknis, J.; Chinou, I. Liposomal incorporation of carvacrol and thymol isolated from the essential oil of Origanum dictamnus L. and in vitro antimicrobial activity. Food Chem. 2009, 112, 77–83. [Google Scholar] [CrossRef]
- Abid, T.A.; Al-Kafaji, B.; Karbel, H.; Naji, H. Damper Effect of Carvacrol on T47-D, a Human Breast Cancer Cell Line. Med. J. Babylon 2014, 11, 792–798. [Google Scholar]
- Arunasree, K.M. Anti-proliferative effects of carvacrol on a human metastatic breast cancer cell line, MDA-MB 231. Phytomedicine 2010, 17, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Koparal, A.T.; Zeytinoglu, M. Effects of carvacrol on a human non-small cell lung cancer (NSCLC) cell line, A549. Cytotechnology 2003, 43, 149–154. [Google Scholar] [CrossRef]
- Fan, K.; Li, X.; Cao, Y.; Qi, H.; Li, L.; Zhang, Q.; Sun, H. Carvacrol inhibits proliferation and induces apoptosis in human colon cancer cells. Anticancer Drugs 2015, 26, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.H.; Yan, F.X.; Zu, X.Y.; Wu, Y.H.; Wu, X.P.; Liao, M.C.; Deng, S.W.; Yin, L.L.; Zhuang, Y.Z. Anti-proliferative and pro-apoptotic effect of carvacrol on human hepatocellular carcinoma cell line HepG-2. Cytotechnology 2012, 64, 43–51. [Google Scholar] [CrossRef]
- Deb, D.D.; Parimala, G.; Saravana Devi, S.; Chakraborty, T. Effect of thymol on peripheral blood mononuclear cell PBMC and acute promyelotic cancer cell line HL-60. Chem. Biol. Interact. 2011, 193, 97–106. [Google Scholar] [CrossRef]
- Hsu, S.S.; Lin, K.L.; Chou, C.T.; Chiang, A.J.; Liang, W.Z.; Chang, H.T.; Tsai, J.Y.; Liao, W.C.; Huang, F.D.; Huang, J.K.; et al. Effect of thymol on Ca2+ homeostasis and viability in human glioblastoma cells. Eur. J. Pharmacol. 2011, 670, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Llana-Ruiz-Cabello, M.; Gutiérrez-Praena, D.; Puerto, M.; Pichardo, S.; Jos, Á.; Cameán, A.M. In vitro pro-oxidant/antioxidant role of carvacrol, thymol and their mixture in the intestinal Caco-2 cell line. Toxicol. Vitr. 2015, 29, 647–656. [Google Scholar] [CrossRef]
- Ferraz, A.; Faria, D.H.; Benneti, M.N.; Brondani Da Rocha, A.; Schwartsmann, G.; Henriques, A.; Von Poser, G.L. Screening for antiproliferative activity of six southern Brazilian species of Hypericum. Phytomedicine 2005, 12, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Voutquenne, L.; Lavaud, C.; Massiot, G.; Sevenet, T.; Hadi, H.A. Cytotoxic polyisoprenes and glycosides of long-chain fatty alcohols from Dimocarpus fumatus. Phytochemistry 1999, 50, 63–69. [Google Scholar] [CrossRef]
- Benslimane, A.F.; Pouchus, Y.F.; Le Boterff, J.; Verbist, J.F.; Roussakis, C.; Monniot, F. Substances Cytotoxiqueset Antibacteriennes de L ’ ascidie aplidium antillense. J. Nat. Prod. 1988, 51, 582–583. [Google Scholar] [CrossRef] [PubMed]
- Roscetti, G.; Franzese, O.; Comandini, A.; Bonmassar, E. Cytotoxic activity of Hypericum perforatum L. on K562 erythroleukemic cells: Differential effects between methanolic extract and hypericin. Phyther. Res. 2004, 18, 66–72. [Google Scholar] [CrossRef]
- Mert Gönenç, T.; Ozturk, M.; Gürdal Türkseven, S.; Ballar Kirmizibayrak, P.; Günal, S.; Yilmaz, S. Hypericum perforatum L.: An overview of the anticancer potencies of the specimens collected from different ecological environments. Pak. J. Bot 2020, 52, 1003–1010. [Google Scholar] [CrossRef]
- Aazza, S.; Lyoussi, B.; Miguel, M.G. Antioxidant activity of some Morrocan hydrosols. J. Med. Plants Res. 2011, 5, 6688–6696. [Google Scholar] [CrossRef]
- Zardi-Bergaoui, A.; Jelassi, A.; Daami-Remadi, M.; Harzallah-Skhiri, F.; Flamini, G.; Ascrizzi, R.; Ben Jannet, H. Chemical composition and bioactivities of essential oils from Pulicaria vulgaris subsp. dentata (Sm.) Batt. growing in Tunisia. J. Essent. Oil Res. 2020, 32, 111–120. [Google Scholar] [CrossRef]
- Bentayeb, K.; Vera, P.; Rubio, C.; Nerín, C. The additive properties of Oxygen Radical Absorbance Capacity (ORAC) assay: The case of essential oils. Food Chem. 2014, 148, 204–208. [Google Scholar] [CrossRef]
- Kratchanova, M.; Denev, P.; Ciz, M.; Lojek, A.; Mihailov, A. Evaluation of antioxidant activity of medicinal plants containing polyphenol compounds. Comparison of two extraction systems. Acta Biochim. Pol. 2010, 57, 229–234. [Google Scholar] [CrossRef]
- Deans, S.G.; Ritchie, G. Antibacterial properties of plant essential oils. Int. J. Food Microbiol. 1987, 5, 165–180. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Dunkić, V.; Kosalec, I.; Kosir, I.; Potocnik, T.; Cerenak, A.; Koncic, M.; Vitali, D.; Muller, I.; Kopricanec, M.; Bezic, N.; et al. Antioxidant and antimicrobial properties of Veronica spicata L. (Plantaginaceae). Curr. Drug Targets 2015, 16, 1660–1670. [Google Scholar] [CrossRef] [PubMed]
- Fredotović, Ž.; Soldo, B.; Šprung, M.; Marijanović, Z.; Jerković, I.; Puizina, J. Comparison of organosulfur and amino acid composition between triploid onion Allium cornutum clementi ex visiani, 1842, and common onion Allium cepa L., and evidences for antiproliferative activity of their extracts. Plants 2020, 9, 98. [Google Scholar] [CrossRef] [PubMed]
- Fredotovíc, Ž.; Šprung, M.; Soldo, B.; Ljubenkov, I.; Budić-Leto, I.; Bilušić, T.; Cikeš-Čulić, V.; Puizina, J. Chemical composition and biological activity of allium cepa L. and Allium × cornutum (Clementi ex Visiani 1842) methanolic extracts. Molecules 2017, 22, 448. [Google Scholar] [CrossRef]
- Nazlić, M.; Kremer, D.; Grubešić, R.J.; Soldo, B.; Vuko, E.; Stabentheiner, E.; Ballian, D.; Bogunić, F.; Dunkić, V. Endemic veronica saturejoides vis. Ssp. saturejoides–chemical composition and antioxidant activity of free volatile compounds. Plants 2020, 9, 1646. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Mensor, L.L.; Menezes, F.S.; Leitão, G.G.; Reis, A.S.; dos Santos, T.C.; Coube, C.S.; Leitão, S.G. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phyther. Res. 2015, 15, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Payet, B.; Sing, A.S.C.; Smadja, J. Assessment of antioxidant activity of cane brown sugars by ABTS and DPPH radical scavenging assays: Determination of their polyphenolic and volatile constituents. J. Agric. Food Chem. 2005, 53, 10074–10079. [Google Scholar] [CrossRef] [PubMed]
- Yen, G.C.; Pin-Der, D. Scavenging Effect of Methanolic Extracts of Peanut Hulls on Free-Radical and Active-Oxygen Species. J. Agric. Food Chem. 1994, 42, 629–632. [Google Scholar] [CrossRef]
| Component | RIs | RI a | RIs | RI b | EO (Yield in %) | H (Yield in %) |
|---|---|---|---|---|---|---|
| Monoterpene hydrocarbons | 24.59 | 15.38 | ||||
| α-Pinene * | 932 | 938 | 1027 | 1025 | 16.58 ± 0.01 a | 8.69 ± 0.01 b |
| Camphene * | 946 | 962 | 1053 | 1048 | 0.59 ± 0.01 a | 0.29 ± 0.05 b |
| Sabinene | 969 | 971 | 1110 | 1095 | 1.22 ± 0.01 | - |
| β-Pinene | 974 | 982 | 1113 | 1109 | 3.67 ± 0.01 a | 0.34 ± 0.01 b |
| Myrcene | 988 | 992 | 1167 | 1167 | 0.26 ± 0.02 b | 2.55 ± 0.01 a |
| α-Terpinene | 1014 | 1016 | 1179 | 1182 | 0.79 ± 0.01 | - |
| p-Cymene | 1020 | 1021 | 1265 | 1268 | 0.17 ± 0.01 | - |
| Limonene | 1024 | 1032 | 1196 | 1204 | 0.65 ± 0.01 | 2.63 ± 0.01 a |
| (Z)-β-Ocimene * | 1032 | 1052 | 1224 | 1218 | - | 0.88 ± 0.02 |
| γ-Terpinene | 1054 | 1057 | 1238 | 1255 | 0.66 ± 0.01 | - |
| Oxygenated monoterpenes | 10.84 | 25.7 | ||||
| Linalool * | 1095 | 1099 | 1547 | 1548 | 0.45 ± 0.01 b | 4.56 ± 0.01 a |
| β-Thujone | 1112 | 1121 | 1435 | 1438 | 3.24 ± 0.01 | - |
| Camphor | 1141 | 1151 | 1515 | 1499 | 0.97 ± 0.01 b | 2.17 ± 0.01 a |
| Pinocarvone | 1160 | 1160 | 1543 | 1540 | 0.68 ± 0.01 | - |
| Borneol * | 1165 | 1176 | 1700 | 1719 | 0.55 ± 0.01 | - |
| Terpinen-4-ol | 1174 | 1184 | 1610 | 1611 | 1.26 ± 0.01 | - |
| α-Terpineol | 1186 | 1186 | 1661 | 1646 | 0.37 ± 0.02 b | 3.57 ± 0.01 a |
| Myrtenol | 1194 | 1197 | 1776 | 1782 | 0.89 ± 0.01 b | 12.33 ± 0.0 a |
| Verbenone | 1204 | 1204 | 1720 | 1705 | - | 0.69 ± 0.02 |
| β-Cyclocitral | 1217 | 1223 | 1610 | 1629 | 0.15 ± 0.01 b | 2.38 ± 0.01 a |
| Linalyl acetate | 1254 | 1252 | 1553 | 1553 | 0.61 ± 0.03 | - |
| Bornyl acetate | 1287 | 1285 | 1572 | 1570 | 0.94 ± 0.01 | - |
| Piperitone oxide | 1366 | 1366 | - | - | 0.75 ± 0.01 | - |
| Sesquiterpenehydrocarbons | 17.27 | 5.95 | ||||
| α-Copaene | 1374 | 1377 | 1482 | 1484 | 0.23 ± 0.01 b | 0.84 ± 0.01 a |
| β-Bourbonene | 1387 | 1383 | 1500 | 1508 | 0.73 ± 0.05 | - |
| (E)-Caryophyllene * | 1417 | 1424 | 1598 | 1585 | 9.52 ± 0.01 a | 2.25 ± 0.01 b |
| allo-Aromadendrene | 1458 | 1465 | 1660 | 1662 | - | 1.56 ± 0.01 |
| Germacrene D | 1484 | 1481 | 1708 | 1692 | 1.83 ± 0.01 | - |
| Viridiflorene | 1496 | 1496 | 1698 | 1697 | 1.67 ± 0.01 a | 0.37 ± 0.03 b |
| Bicyclogermacrene | 1500 | 1500 | 1734 | 1718 | 1.47 ± 0.01 a | 0.54 ± 0.01 b |
| β-Bisabolene | 1505 | 1494 | 1728 | 1729 | 0.85 ± 0.01 a | 0.39 ± 0.01 b |
| δ-Cadinene | 1522 | 1517 | 1757 | 1745 | 0.97 ± 0.01 | - |
| Oxygenated sesquiterpenes | 12.51 | 7.54 | ||||
| Spathulenol | 1577 | 1577 | 2106 | 2101 | 2.28 ± 0.01 b | 4.93 ± 0.01 a |
| Caryophyllene oxide * | 1582 | 1581 | 1954 | 1955 | 7.69 ± 0.01 a | 0.76 ± 0.01 b |
| γ-Eudesmol | 1630 | 1632 | 2166 | 2135 | 0.87 ± 0.01 a | 0.47 ± 0.02 b |
| α-Cadinol | 1652 | 1655 | 2210 | 2208 | 0.67 ± 0.05 | - |
| α-Bisabolol | 1685 | 1688 | 2168 | 2116 | 0.76 ± 0.02 | - |
| α-Bisabolol oxide | 1749 | 1748 | - | 2511 | 0.24 ± 0.02 b | 1.38 ± 0.01 a |
| Phenolic compounds | 1.69 | 13.35 | ||||
| Thymol * | 1289 | 1290 | 2198 | 2198 | 0.14 ± 0.01 b | 3.48 ± 0.01 a |
| Carvacrol * | 1298 | 1298 | 2239 | 2239 | 1.37 ± 0.01 b | 9.87 ± 0.01 a |
| Eugenol * | 1356 | 1370 | 2173 | 2175 | 0.18 ± 0.03 | - |
| Aliphatic compounds | 13.86 | 4.29 | ||||
| n-Nonane | 900 | 900 | - | 1011 | 13.59 ± 0.01 a | 4.29 ± 0.01 b |
| 1-Octen-3-ol | 974 | 974 | 1442 | 1452 | 0.27 ± 0.01 | - |
| Diterpenes | 0.26 | - | ||||
| Phytol | 1942 | 1964 | 2622 | 2622 | 0.26 ± 0.03 | - |
| Hydrocarbons | 4.23 | - | ||||
| Hexadecanoic acid | 1959 | 1959 | - | 2476 | 1.28 ± 0.01 | - |
| Docosane * | 2200 | 2200 | 2200 | 2200 | 0.57 ± 0.02 | - |
| Hexacosane * | 2600 | 2600 | 2600 | 2600 | 1.59 ± 0.01 | - |
| Heptacosane * | 2700 | 2700 | 2700 | 2700 | 0.79 ± 0.01 | - |
| Total identified (%) | 85.25 | 72.21 |
| H. perforatum ssp. veronense | ||
|---|---|---|
| Antioxidant Assay | Essential Oil | Hydrosol |
| ORAC (Trolox eq) | 2347.65 ± 119.28 a | 240.34 ± 7.59 b |
| DPPH (% inhibition) | 44.03 ± 0.74 a | 11.88 ± 1.4 b |
| DPPH (IC50) | 23.07 ± 0.49 | - |
| dpi | LLN ± SD | LLN ± SD | ||
|---|---|---|---|---|
| 3rd | C | 6.05 ± 2.19 a | C | 9.65 ± 3.77 a |
| EO | 0.72 ± 0.33 b | H | 4.93 ± 2.29 b | |
| 5th | C | 16.64 ± 3.25 a | C | 19.00 ± 3.24 a |
| EO | 6.24 ± 0.64 b | H | 12.05 ± 1.60 b | |
| 7th | C | 19.38 ± 6.05 a | C | 28.55 ± 3.82 a |
| EO | 9.50 ± 2.32 b | H | 17.34 ± 4.50 b | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vuko, E.; Dunkić, V.; Ruščić, M.; Nazlić, M.; Mandić, N.; Soldo, B.; Šprung, M.; Fredotović, Ž. Chemical Composition and New Biological Activities of Essential Oil and Hydrosol of Hypericum perforatum L. ssp. veronense (Schrank) H. Lindb. Plants 2021, 10, 1014. https://doi.org/10.3390/plants10051014
Vuko E, Dunkić V, Ruščić M, Nazlić M, Mandić N, Soldo B, Šprung M, Fredotović Ž. Chemical Composition and New Biological Activities of Essential Oil and Hydrosol of Hypericum perforatum L. ssp. veronense (Schrank) H. Lindb. Plants. 2021; 10(5):1014. https://doi.org/10.3390/plants10051014
Chicago/Turabian StyleVuko, Elma, Valerija Dunkić, Mirko Ruščić, Marija Nazlić, Nela Mandić, Barbara Soldo, Matilda Šprung, and Željana Fredotović. 2021. "Chemical Composition and New Biological Activities of Essential Oil and Hydrosol of Hypericum perforatum L. ssp. veronense (Schrank) H. Lindb" Plants 10, no. 5: 1014. https://doi.org/10.3390/plants10051014
APA StyleVuko, E., Dunkić, V., Ruščić, M., Nazlić, M., Mandić, N., Soldo, B., Šprung, M., & Fredotović, Ž. (2021). Chemical Composition and New Biological Activities of Essential Oil and Hydrosol of Hypericum perforatum L. ssp. veronense (Schrank) H. Lindb. Plants, 10(5), 1014. https://doi.org/10.3390/plants10051014








