Untargeted Metabolomics of Rind Essential Oils Allowed to Differentiate Two Closely Related Clementine Varieties
Abstract
:1. Introduction
2. Results and Discussion
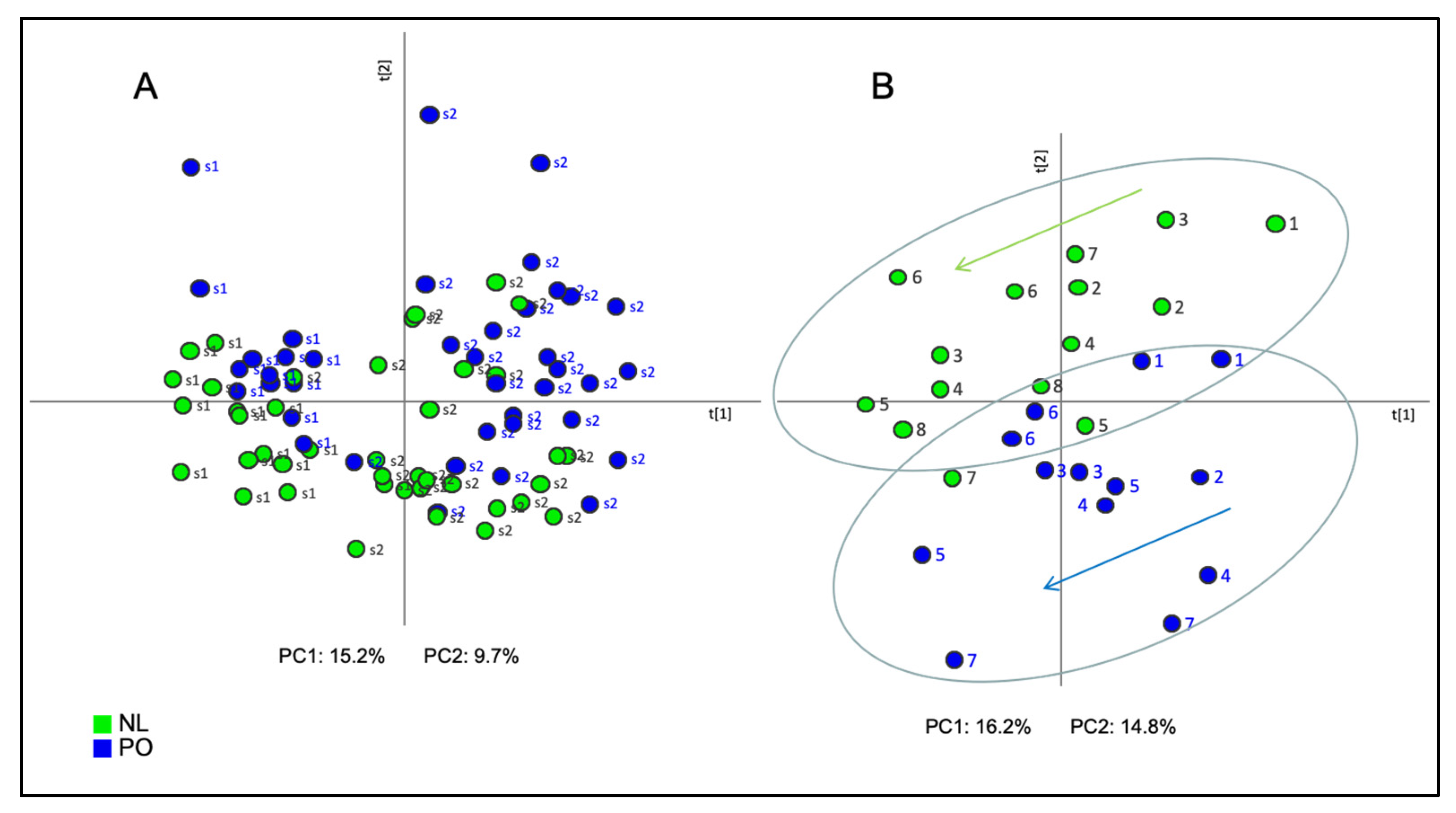
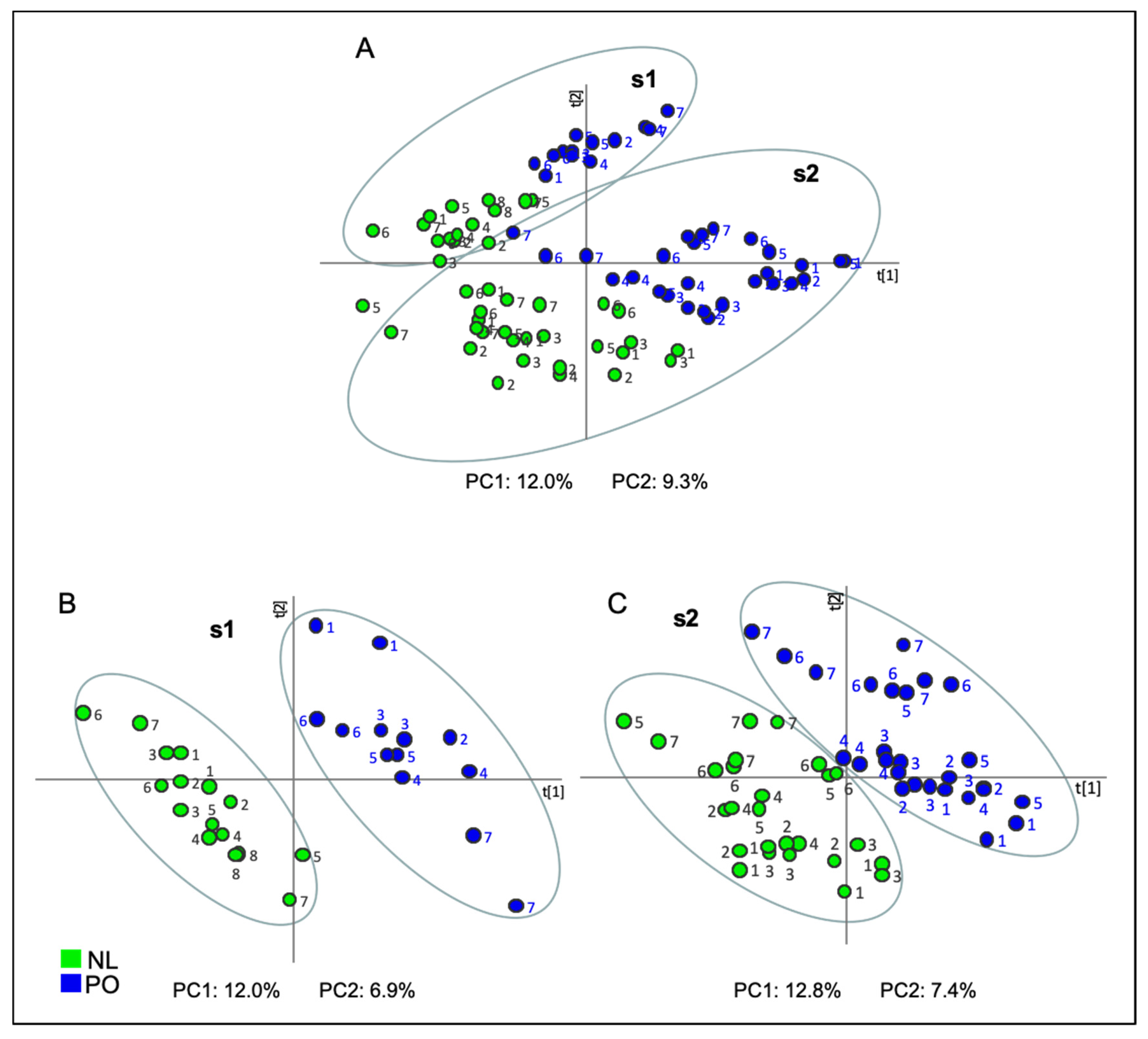
2.1. Principal Component Analysis (PCA)
2.2. Evolution of the Volatile Profile along Harvest Time
2.3. Projection to Latent Structures-Discriminant Analysis (PLS-DA)
2.4. Identification of Differential VOCs between NL and PO Essential Oil
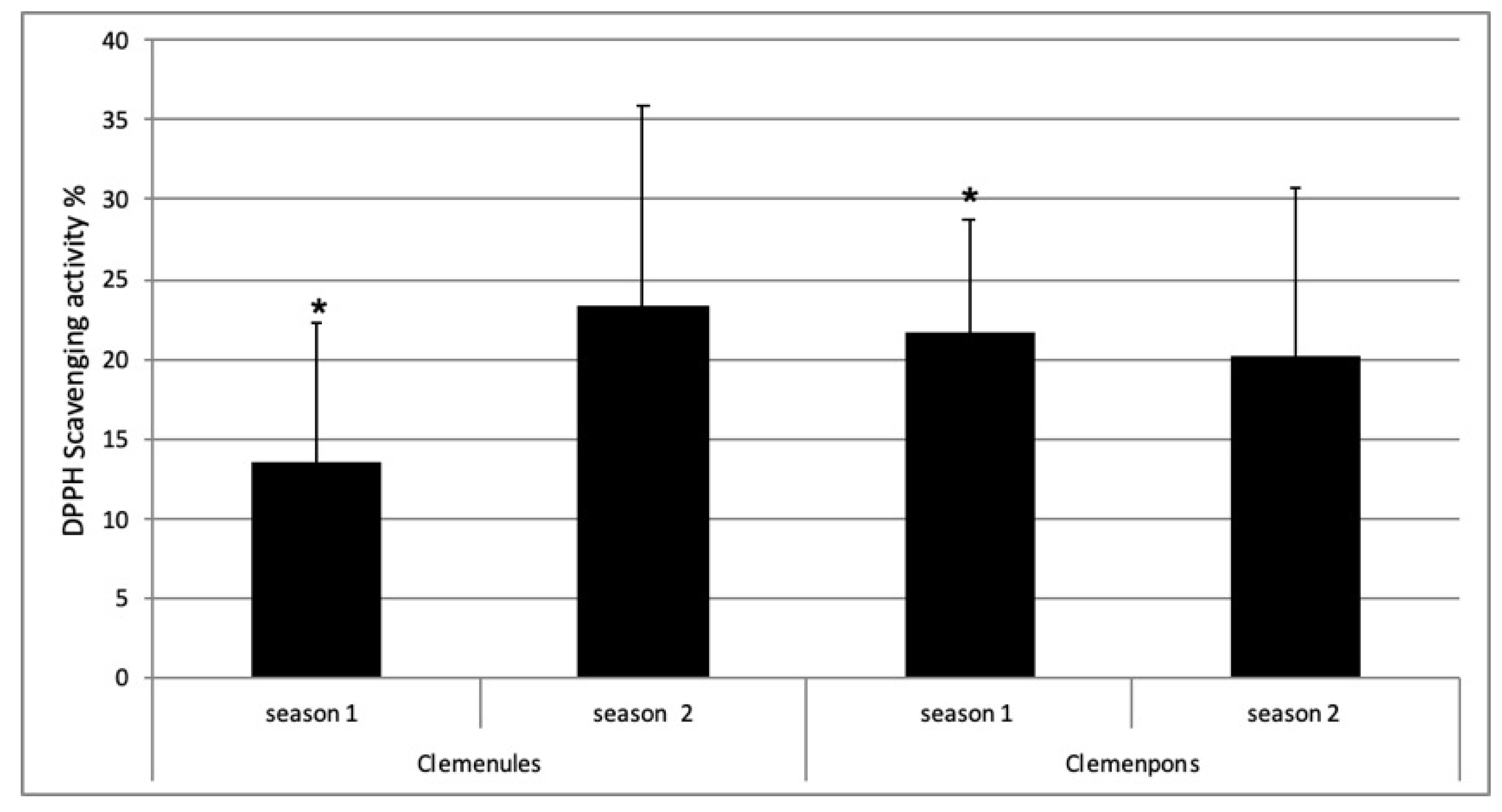
2.5. Antioxidant Activity of NL and PO EO
3. Materials and Methods
3.1. Plant Material
3.2. Sampling and EO Extraction
3.3. Analysis of the EO by GC-MS after Head Space-Solid Phase Microextraction (HS-SPME)
3.4. VOCs’ Identification and Quantification
3.5. Antioxidant Activity
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, G.A.; Terol, J.; Ibanez, V.; López-García, A.; Pérez-Román, E.; Borredá, C.; Domingo, C.; Tadeo, F.R.; Carbonell-Caballero, J.; Alonso, R.; et al. Genomics of the origin and evolution of Citrus. Nature 2018, 554, 311. [Google Scholar] [CrossRef] [Green Version]
- Ruberto, G.; Renda, A.; Piattelli, M.; Rapisarda, P.; Starrantino, A. Essential oil of two new pigmented citrus hybrids Citrus clementina × Citrus sinensis. J. Agric. Food Chem. 1997, 45, 467–471. [Google Scholar] [CrossRef]
- Merle, H.; Morón, M.; Blázquez, M.A.; Boira, H. Taxonomical contribution of essential oils in mandarins cultivars. Biochem. Syst. Ecol. 2004, 32, 491–497. [Google Scholar] [CrossRef]
- Fabroni, S.; Ruberto, G.; Rapisarda, P. Essential oil profiles of new Citrus hybrids, a tool for genetic citrus improvement. J. Essent. Oil Res. 2012, 24, 159–169. [Google Scholar] [CrossRef]
- Fanciullino, A.-L.; Tomi, F.; Luro, F.; Desjobert, J.M.; Casanova, J. Chemical variability of peel and leaf oils of mandarins. Flavour Frag. J. 2006, 21, 359–367. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Pérez-Alvárez, J.A. Chemical composition of mandarin (C. reticulata L.), grapefruit (C.paradisi L.), lemon (C. limon L.) and orange (C. sinensis L.) essential oils. J. Essent. Oil Bear. Plants 2009, 12, 236–243. [Google Scholar] [CrossRef]
- González-Mas, M.C.; Rambla, J.L.; López-Gresa, M.P.; Blázquez, M.A.; Granell, A. Volatile Compounds in Citrus Essential Oils: A Comprehensive Review. Front. Plant Sci. 2019, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, Y. Comparative study on the volatiles in Citrus reticulata ‘Dahongpao’ peel from the same plant and their antioxidant activities. J. Essent. Oil Bear. Plants 2014, 17, 303–308. [Google Scholar] [CrossRef]
- Tao, N.; Jia, L.; Zhou, H. Anti-fungal activity of Citrus reticulata Blanco essential oil against Penicillium italicum and Penicillium digitatum. Food Chem. 2014, 153, 265–271. [Google Scholar] [CrossRef]
- Dosoky, N.S.; Setzer, W.N. Biological activities and safety of Citrus spp. essential oils. Int. J. Mol. Sci. 2018, 19, 1966. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.-L.; Yang, Z.-Y.; Fan, G.; Ren, J.-N.; Yin, K.-J.; Pan, S.-Y. Antidepressant-like effect of Citrus sinensis (L.) Osbeck essential oil and its main component limonene on mice. J. Agric. Food Chem. 2019, 67, 13817–13828. [Google Scholar] [CrossRef] [PubMed]
- Bora, H.; Kamle, M.; Kumar Mahato, D.; Tiwari, P.; Kumar, P. Citrus essential oils (CEOs) and their applications in food: An overview. Plants 2020, 9, 357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, B.; Singh, J.P.; Kaur, A.; Yadaw, M.P. Insights into chemical composition and bioactivities of citrus peel essential oils. Food Res. Int. 2021, 143, 110231. [Google Scholar] [CrossRef] [PubMed]
- Giunti, G.; Palermo, D.; Laudani, F.; Algeri, G.M.; Campolo, O.; Palmeri, V. Repellence and acute toxicity of a nano-emulsion of sweet orange essential oil toward two major stored grain insect pests. Ind. Crop Prod. 2019, 142, 111869. [Google Scholar] [CrossRef]
- Maffei, M.E.; Gertsch, J.; Appendino, G. Plant volatiles: Production, function and pharmacology. Nat. Prod. Rep. 2011, 28, 1359–1380. [Google Scholar] [CrossRef]
- Soler Aznar, J.; Soler Fayos, G.; Forner Giner, M.A.; Forner Valero, J. Criterios para Identificar Variedades y Patrones de Cítricos, 1st ed.; LLIG: Valencia, Spain, 2006; pp. 114–129. [Google Scholar]
- González-Mas, M.C.; Escriche, M.D.; Jover, S.; Bermejo, A.; Cano, A.; Gutiérrez-Suanzes, A. Estudio del aceite esencial de clementinas: Diferenciación de variedades según el perfil volátil de la corteza. Levante Agrícola 2010, 401, 185–192. [Google Scholar]
- Garcia-Sanchez, F.; Simon-Grao, S.; Gimeno, V.; Galvez-Sola, L.; Lidon, V.; Simon, I.; Hernandez, F.; Martinez-Nicolas, J.J.; Carbonell-Barrachina, A.A. Phytochemical properties and volatile composition profile of nine early maturing mandarins cultivated in South-East Spain. J. Agric. Sci. Technol. 2016, 18, 1367–1380. [Google Scholar]
- González-Mas, M.C.; Rambla, J.L.; Alamar, M.C.; Gutiérrez, A.; Granell, A. Comparative analysis of the volatile fraction of fruit juice from different Citrus species. PLoS ONE 2011, 6, e220166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirbaşlar, Ş.İ.; Gök, A.; Kirbaşlar, F.G.; Tepe, S. Volatiles in Turkish clementine (Citrus clementina Hort.) peel. J. Essent. Oil Res. 2012, 28, 96–103. [Google Scholar] [CrossRef]
- Thi Nguyen, T.-T.; Thi Tran, T.-T.; Hua, T.-M.; Diep, T.-T.; Nguyen Chau, D.-K.; Duus, F.; Ngoc Le, T. Investigation of peel and leaf essential oils of Citrus clementina Hort. ex Tan. growing in the south of Vietnam. J. Essent. Oil Res. 2012, 24, 153–157. [Google Scholar] [CrossRef]
- Boussaada, O.; Chemli, R. Seasonal variation of essential oil composition of Citrus aurantium L. var. amara. J. Essent. Oil Bear. Plants 2007, 10, 109–120. [Google Scholar] [CrossRef]
- Di Rauso Simeone, G.; Di Matteo, A.; Rao, M.A.; Di Vaio, C. Variations of peel essential oils during fruit ripening in four lemon (Citrus limon (L.) Burm. F.) cultivars. J. Sci. Food Agric. 2020, 100, 193–200. [Google Scholar] [CrossRef]
- Lara, I.; Heredia, A.; Domínguez, E. Shelf life potential and the fruit cuticle: The unexpected player. Front. Plant Sci. 2019, 10, 770. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Sun, K.; Xie, L.; He, Y.; Luo, T.; Sheng, L.; Luo, Y.; Zeng, Y.; Xu, J.; Deng, X.; et al. Regulation of cuticle formation during fruit development and ripening in ‘Newhall’ navel orange (Citrus sinensis Osbeck) revealed by transcriptomic and metabolomic profiling. Plant Sci. 2016, 243, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Cornell University. Flavornet and Human Odor Space, Gas Chromatography—Olfactometry (GCO) of Natural Products; Cornell University: Ithaca, NY, USA, 2004; Available online: http://www.flavornet.org/flavornet.html (accessed on 28 June 2021).
- Njoroge, S.M.; Koaze, H.; Karanja, P.N.; Sawamura, M. Essential oil constituents of three varieties of Kenyan sweet oranges (Citrus sinensis). Flavour Frag. J. 2005, 20, 80–85. [Google Scholar] [CrossRef]
- Dugo, P.; Mondello, L. Citrus Oils: Composition, Advanced Analytical Techniques, Contaminants, and Biological Activity; CRC Press: Boca Raton, FL, USA, 2011; Volume 49. [Google Scholar]
- Chen, Y.; Wu, J.; Xu, Y.; Fu, M.; Xiao, G. Effect of second cooling on the chemical components of essential oils from orange peel (Citrus sinensis). J. Agric. Food Chem. 2014, 62, 8786–8790. [Google Scholar] [CrossRef] [PubMed]
- Lota, M.-L.; de Rocca Serra, D.; Tomi, F.; Casanova, J. Chemical variability of peel and leaf essential oils of 15 species of mandarins. Biochem. Syst. Ecol. 2001, 29, 77–104. [Google Scholar] [CrossRef]
- Ruberto, G.; Rapisarda, P. Essential oils of new pigmented citrus hybrids: Citrus sinensis L. Osbeck x C. clementina Hort. ex Tanaka. J. Food Sci. 2002, 67, 2778–2780. [Google Scholar] [CrossRef]
- Bouwmeester, H.J.; Gershenzon, J.; Konings, M.C.J.M.; Croteau, R. Biosynthesis of the monoterpenes limonene and carvone in the fruit of caraway. Plant Physiol. 1998, 117, 901–912. [Google Scholar] [CrossRef] [Green Version]
- Raal, A.; Arak, E.; Orav, A. The content and composition of the essential oil found in Carum carvi L. commercial fruits obtained from different countries. J. Essent. Oil Res. 2012, 24, 1. [Google Scholar] [CrossRef] [Green Version]
- Duetz, W.A.; Bouwmeester, H.; van Beilen, J.B.; Witholt, B. Biotransformation of limonene by bacteria, fungi, yeasts, and plants. Appl. Microbiol. Biotechnol. 2003, 61, 269–277. [Google Scholar] [CrossRef]
- Li, J.L.; Hong, P.; Jiang, Z.D.; Yang, Y.F.; Du, X.P.; Sun, H.; Wu, L.M.; Ni, H.; Chen, F. Water accelerated transformation of d-limonene induced by ultraviolet irradiation an air exposure. Food Chem. 2018, 239, 434–441. [Google Scholar] [CrossRef]
- Chisholm, M.G.; Wilson, M.A.; Gaskey, G.M. Characterization of aroma volatiles in key lime essential oils (Citrus aurantifolia Swingle). Flavour Frag. J. 2003, 18, 106–115. [Google Scholar] [CrossRef]
- Chisholm, M.G.; Jell, J.A.; Cass, D.M., Jr. Characterization of the major odorants found in the peel oil of Citrus reticulata Blanco cv. clementine using gas chromatography-olfactometry. Flavour Frag. J. 2003, 18, 275–281. [Google Scholar] [CrossRef]
- Miyazawa, N.; Fujita, A.; Kubota, K. Aroma character impact compounds in Kinokuni mandarin orange (Citrus kinokuni) compared with satsuma mandarin orange (Citrus unshiu). Biosci. Biotechnol. Biochem. 2010, 74, 835–842. [Google Scholar] [CrossRef]
- Högnadóttir, Á.; Rouseff, R.L. Identification of aroma active compounds in orange essence oil using gas chromatography-olfactometry and gas chromatography-mass spectrometry. J. Chromatogr. A 2003, 998, 201–211. [Google Scholar] [CrossRef]
- Frasinetti, S.; Caltavuturo, L.; Cini, M.; Della Croce, C.M. Antibacterial and antioxidant activity of essential oils from Citrus spp. J. Essent. Oil Res. 2011, 23, 27–31. [Google Scholar] [CrossRef]
- Tundis, R.; Loizzo, M.R.; Bonesi, M.; Menichini, F.; Mastellone, V.; Colica, C.; Menichini, F. Comparative study of the antioxidant capacity and cholinesterase inhibitory activity of Citrus aurantifolia Swingle, C. aurantium L., and C. bergamia Risso and Poit. peel essential oils. J. Food Sci. 2012, 77, H40–H46. [Google Scholar] [CrossRef] [PubMed]
- Smeriglio, A.; Alloisio, S.; Raimondo, F.M.; Denaro, M.; Xiao, J.; Cornara, L.; Trombetta, D. Essential oil of Citrus lumia Risso: Phytochemical profile, antioxidant properties and activity on the central nervous system. Food Chem. Toxicol. 2018, 119, 407–416. [Google Scholar] [CrossRef]
- Choi, H.-S.; Song, H.S.; Ukeda, H.; Sawamura, M. Radical-scavenging activities of Citrus essential oils and their components: Detection using 1,1-diphenyl-2-picrylhydrazyl. J. Agric. Food Chem. 2000, 48, 4156–4161. [Google Scholar] [CrossRef]
- Denkova-Kostova, R.; Teneva, D.; Tomova, T.; Goranov, B.; Denkova, Z.; Shopska, V.; Slavchev, A.; Hristova-Ivanova, Y. Chemical composition, antioxidant and antimicrobial activity of essential oils from tangerine (Citrus reticulata L.), grapefruit (Citrus paradisi L.), lemon (Citrus lemon L.) and cinnamon (Cinnamomum zeylanicum Blume). Z. Naturforsch. 2021, 76, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Cao, S.; Sun, J.; Lu, D.; Zhong, B.; Chun, J. The chemical compositions, and antibacterial and antioxidant activities of four types of Citrus essential oils. Molecules 2021, 26, 3412. [Google Scholar] [CrossRef]
- Lamine, M.; Rahali, F.Z.; Hammami, M.; Mliki, A. Correlative metabolite profiling approach to understand antioxidant and antimicrobial activities from Citrus essential oils. Int. J. Food Sci. Technol. 2019, 54, 2615–2623. [Google Scholar] [CrossRef]
- Boudries, H.; Loupassaki, S.; Ladjal Ettoumi, Y.; Souagui, S.; Bachir Bey, M.; Nabet, N.; Chikhoune, A.; Madani, K.; Chibane, M. Chemical profile, antimicrobial and antioxidant activities of Citrus reticulata and Citrus clementina (L.) essential oils. Int. Food Res. J. 2017, 24, 1782–1792. Available online: http://www.ifrj.upm.edu.my/24%20(04)%202017/(56).pdf (accessed on 28 June 2021).
- Yi, F.; Jin, R.; Sun, J.; Ma, B.; Bao, X. Evaluation of mechanical-pressed essential oil from Nanfeng mandarin (Blanco Cv. Kinokuni) as a food preservative based on antimicrobial and antioxidant activities. Food Sci Technol. 2018, 95, 346–353. [Google Scholar] [CrossRef]
- Alparsian, Y.; Baygar, T. Effect of chitosan film coating combined with orange peel essential oil on the shelf life of deepwater pink shrimp. Food Bioprocess Technol. 2017, 10, 842–853. [Google Scholar] [CrossRef]
- Abdel-Aziz, M.M.; Emam, T.M.; Elsherbiny, E.A. Effects of mandarin (Citrus reticulata) peel essential oil as a natural antibiofilm agent against Aspergillus niger in onion bulbs. Postharvest Biol. Technol. 2019, 156, 110959. [Google Scholar] [CrossRef]
- Durmus, M. The effects of nanoemulsions bases on citrus essential oils (orange, mandarin, grapefruit, and lemon) on the shelf life of rainbow trout (Oncorhynchus mykiss) fillets at 4 ± 2 °C. J. Food Saf. 2020, 40, e12718. [Google Scholar] [CrossRef]
- Plazas, M.; López-Gresa, M.P.; Vilanova, S.; Torres, C.; Hurtado, M.; Gramazio, P.; Andújar, I.; Herráiz, F.J.; Bellés, J.M.; Prohens, J. Diversity and relationships in key traits for functional and apparent quality in a collection of eggplant: Fruit phenolics content, antioxidant activity, polyphenol oxidase activity, and browning. J. Agric. Food Chem. 2013, 61, 8871–8879. [Google Scholar] [CrossRef]
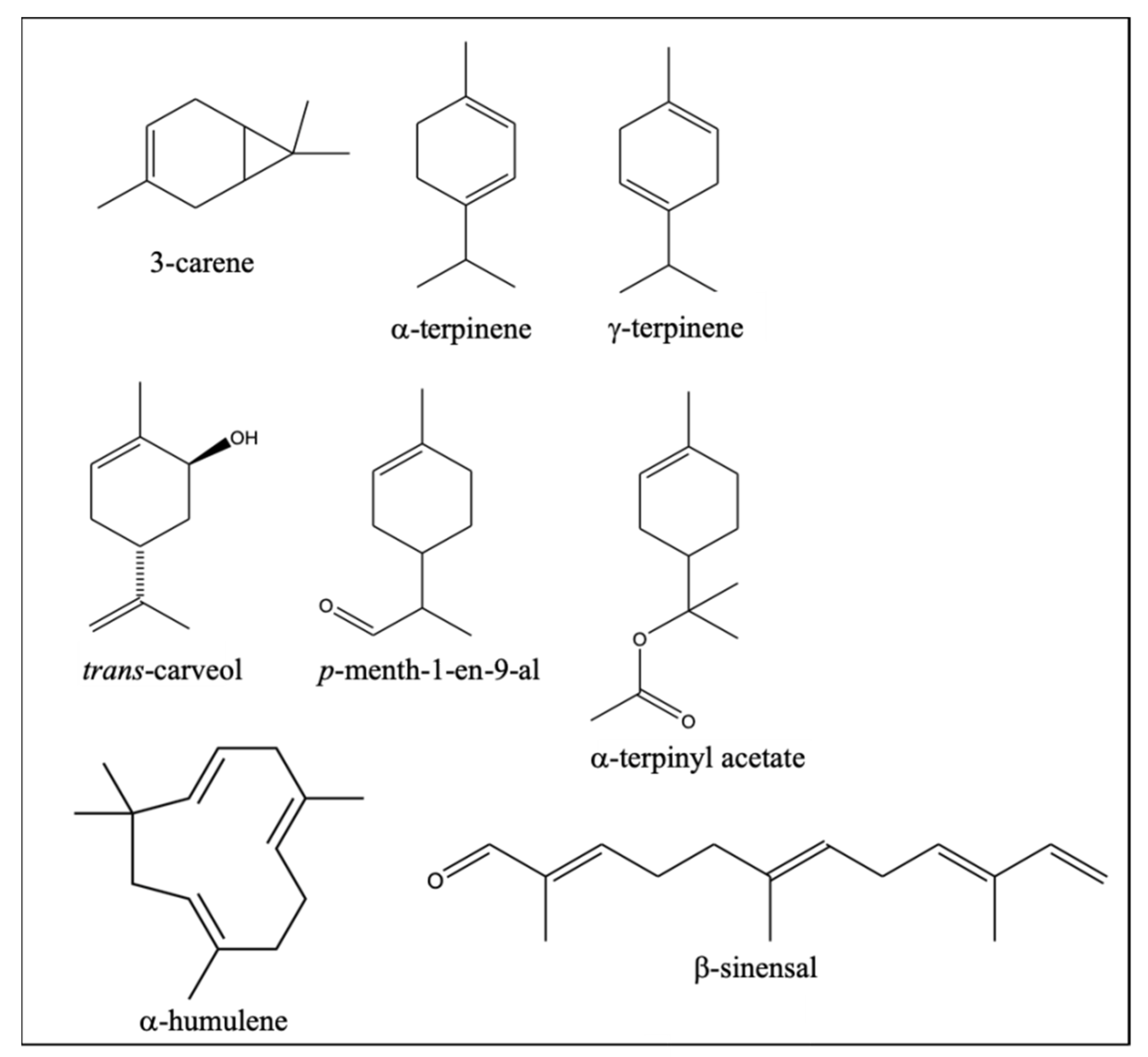
| Compound | LRI a | Compound Type | Smell [26] | Ratio PO/NL b |
|---|---|---|---|---|
| 3-carene | 1024 | MTH | lemon | 1.44 ± 0.36 b |
| α-terpinene | 1030 | MTH | lemon | 1.26 ± 0.29 b |
| γ-terpinene | 1070 | MTH | turpentine | 1.43 ± 0.39 b |
| unidentified | 1138 | OMT | - | 2.79 ± 1.89 b |
| unidentified | 1161 | OMT | - | 3.32 ± 2.79 b |
| trans-carveol | 1238 | OMT | caraway (anise) | 2.11 ± 0.79 b |
| p-menth-1-en-9-al c | 1242 | OMT | - | 1.91 ± 0.80 b |
| unidentified | 1311 | OMT | - | 2.03 ± 0.97 b |
| α-terpinyl acetate | 1363 | OMT | wax | 1.68 ± 0.51 b |
| unidentified | 1423 | OMT | - | 1.71 ± 0.55 b |
| unidentified | 1498 | STH | - | 1.13 ± 0.15 b |
| α-humulene | 1502 | STH | wood | 1.12 ± 0.18 b |
| unidentified | 1529 | STH | - | 1.62 ± 0.49 b |
| unidentified | 1548 | STH | - | 1.43 ± 0.41 b |
| β-sinensal c | 1712 | OST | sweet | 1.83 ± 0.45 b |
| Compound | LRI a (Min) | Compound Type | Smell [26,36,37,38] | Ratio PO/NL b |
|---|---|---|---|---|
| 1-octanol | 1071 | alcohol | moss, mushroom | 0.91± 0.32 b |
| 1-decanol | 1275 | alcohol | fatty | 0.83 ± 0.38 b |
| dodecanal | 1415 | aldehyde | fatty, waxy | 0.72 ± 0.22 b |
| tetradecanal c | 1620 | aldehyde | fatty, wood, waxy, floral | 0.61 ± 0.19 b |
| hexadecanal c | 1823 | aldehyde | wood | 0.65 ± 0.31 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Mas, M.d.C.; Rambla, J.L.; Gómez-Cadenas, A.; Blázquez, M.A.; López-Gresa, M.P.; Granell, A. Untargeted Metabolomics of Rind Essential Oils Allowed to Differentiate Two Closely Related Clementine Varieties. Plants 2021, 10, 1789. https://doi.org/10.3390/plants10091789
González-Mas MdC, Rambla JL, Gómez-Cadenas A, Blázquez MA, López-Gresa MP, Granell A. Untargeted Metabolomics of Rind Essential Oils Allowed to Differentiate Two Closely Related Clementine Varieties. Plants. 2021; 10(9):1789. https://doi.org/10.3390/plants10091789
Chicago/Turabian StyleGonzález-Mas, María del Carmen, José L. Rambla, Aurelio Gómez-Cadenas, María Amparo Blázquez, María Pilar López-Gresa, and Antonio Granell. 2021. "Untargeted Metabolomics of Rind Essential Oils Allowed to Differentiate Two Closely Related Clementine Varieties" Plants 10, no. 9: 1789. https://doi.org/10.3390/plants10091789
APA StyleGonzález-Mas, M. d. C., Rambla, J. L., Gómez-Cadenas, A., Blázquez, M. A., López-Gresa, M. P., & Granell, A. (2021). Untargeted Metabolomics of Rind Essential Oils Allowed to Differentiate Two Closely Related Clementine Varieties. Plants, 10(9), 1789. https://doi.org/10.3390/plants10091789








