Sustainable Processing of Floral Bio-Residues of Saffron (Crocus sativus L.) for Valuable Biorefinery Products
Abstract
1. Introduction
2. Results
2.1. Qualitative and Antioxidant Properties of Dried Tepals Extracts
2.2. Correlation Analysis
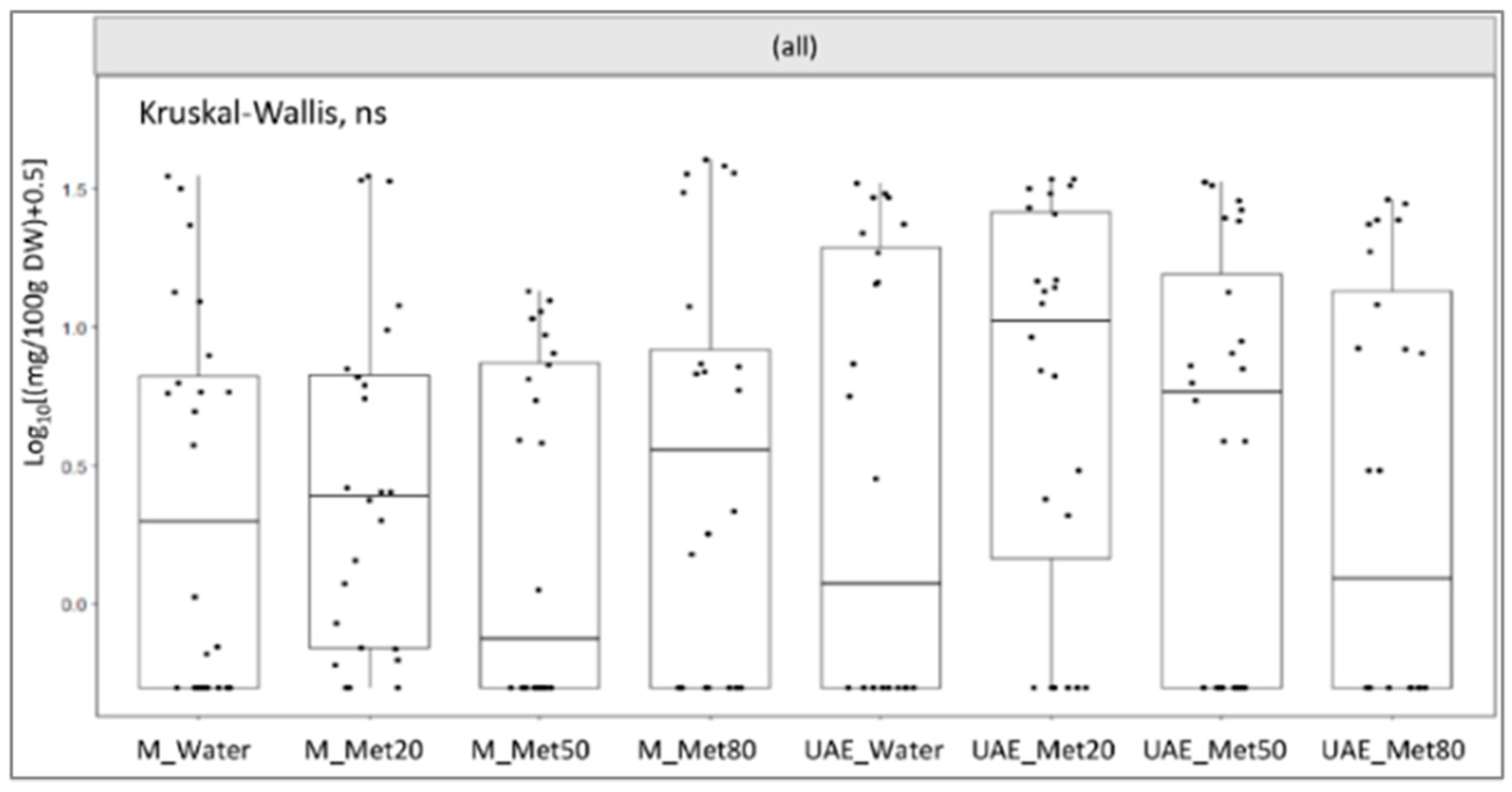
2.3. Bioactive Compounds from HPLC Analysis
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Extraction Methods
4.3. Spectrophotometric Analysis
4.3.1. Total Phenolic Content
4.3.2. Total Anthocyanin Content
4.3.3. Antioxidant Activity
- Ferric ion reducing antioxidant power (FRAP) method. The FRAP solution was produced by mixing a buffer solution at pH 3.6 (C2H3NaO2 + C2H4O2 in water), 2,4,6-tripyridyltriazine (TPTZ, 10 mM in HCl 40 mM), and FeCl3∙6H2O (20 mM). Then, 90 µL of deionized water and 900 µL of the FRAP reagent were added to 30 µL of phytoextract in each plastic tube. The samples were left at 37 °C for 30 min and the absorbance was read at 595 nm at a spectrophotometer (Cary 60 UV-Vis, Agilent Technologies, Santa Clara, CA, USA). Results were expressed as millimoles of ferrous iron equivalents per kilogram of dry weight (mmol Fe2+ Kg−1 DW).
- The 2,2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) method. The ABTS radical cation (ABTS·) was obtained by the reaction of 7.0 mM ABTS solution with 2.45 mM K2S2O8 solution. The solution was incubated for 12–16 h in the dark at room temperature and then diluted with distilled water up to read an absorbance of 0.70 (±0.02) at 734 nm. 500 μL of diluted ABTS· was added to 15 µL of phytoextract and, after incubation in the dark at room temperature for 10 min, the absorbance was measured at 734 nm by means of a spectrophotometer (Cary 60 UV-Vis, Agilent Technologies, Santa Clara, CA, USA). The ABTS radical-scavenging activity was calculated aswhere Abs0 is the absorbance of the control (solution without phytoextract) and Abs1 is the absorbance of the sample. The results were expressed as µmol of Trolox equivalents per gram of dry weight (μmol TE g−1 DW).[(Abs0 − Abs1/Abs0) × 100]
- The 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging method. To obtain 100 μM of DPPH radical cation (DPPH·) 2 mg of DPPH were mixed up with 50 mL of MeOH, up to have an absorbance of 1.000 (±0.005) at 515 nm. Then, 1.5 mL of diluted DPPH· was added to 20 µL of phytoextract and the reaction was left in the dark at room temperature for 30 min. The absorbance was read at 515 nm at a spectrophotometer (Cary 60 UV-Vis, Agilent Technologies, Santa Clara, CA, USA). The DPPH radical-scavenging activity was calculated aswhere Abs0 is the absorbance of the control (solution without phytoextract) and Abs1 is the absorbance of the sample. Results were expressed as μmol of Trolox equivalents per gram of dry weight (μmol TE g−1 DW).[(Abs0 − Abs1/Abs0) × 100]
4.4. HPLC Analysis
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khan, M.; Hanif, M.A.; Ayub, M.A.; Jilani, M.I.; Chatha, S.A.S. Saffron. Med. Plants South Asia 2020, 587–600. [Google Scholar] [CrossRef]
- Caser, M.; Demasi, S.; Victorino, Í.M.M.; Donno, D.; Faccio, A.; Lumini, E.; Bianciotto, V.; Scariot, V. Arbuscular Mycorrhizal Fungi Modulate the Crop Performance and Metabolic Profile of Saffron in Soilless Cultivation. Agronomy 2019, 9, 232. [Google Scholar] [CrossRef]
- Caser, M.; Victorino, Í.M.M.; Demasi, S.; Berruti, A.; Donno, D.; Lumini, E.; Bianciotto, V.; Scariot, V. Saffron Cultivation in Marginal Alpine Environments: How AMF Inoculation Modulates Yield and Bioactive Compounds. Agronomy 2018, 9, 12. [Google Scholar] [CrossRef]
- Melnyk, J.P.; Wang, S.; Marcone, M.F. Chemical and biological properties of the world’s most expensive spice: Saffron. Food Res. Int. 2010, 43, 1981–1989. [Google Scholar] [CrossRef]
- Bagur, M.J.; Salinas, G.L.A.; Jiménez-Monreal, A.M.; Chaouqi, S.; Llorens, S.; Martínez-Tomé, M.; Alonso, G.L. Saffron: An Old Medicinal Plant and a Potential Novel Functional Food. Molecules 2017, 23, 30. [Google Scholar] [CrossRef] [PubMed]
- Gresta, F.; Lombardo, G.M.; Siracusa, L.; Ruberto, G. Saffron, an alternative crop for sustainable agricultural systems. A review. Agron. Sustain. Dev. 2008, 28, 95–112. [Google Scholar] [CrossRef]
- Halevy, A.H. recent advances in control of flowering and growth habit of geophytes. Acta Hortic. 1990, 35–42. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, V.; Devi, K.; Sharma, M.; Singh, M.; Ahuja, P. State of Art of Saffron (Crocus sativus L.) Agronomy: A Comprehensive Review. Food Rev. Int. 2008, 25, 44–85. [Google Scholar] [CrossRef]
- Molina, R.; Valero, M.; Navarro, Y.; Guardiola, J.; García-Luis, A. Temperature effects on flower formation in saffron (Crocus sativus L.). Sci. Hortic. 2005, 103, 361–379. [Google Scholar] [CrossRef]
- Peter, K.V. Handbook of Herbs and Spices Vol. 1: Series in Food Science; Woodhead Publishing: Philadelphia, PA, USA, 2012; ISBN 978-0-85709-567-1. [Google Scholar]
- Mottaghipisheh, J.; Sourestani, M.M.; Kiss, T.; Horváth, A.; Tóth, B.; Ayanmanesh, M.; Khamushi, A.; Csupor, D. Comprehensive chemotaxonomic analysis of saffron crocus tepal and stamen samples, as raw materials with potential antidepressant activity. J. Pharm. Biomed. Anal. 2020, 184, 113183. [Google Scholar] [CrossRef]
- Ashktorab, H.; Soleimani, A.; Singh, G.; Amr, A.; Tabtabaei, S.; Latella, G.; Stein, U.; Akhondzadeh, S.; Solanki, N.; Gondré-Lewis, M.C.; et al. Saffron: The Golden Spice with Therapeutic Properties on Digestive Diseases. Nutrients 2019, 11, 943. [Google Scholar] [CrossRef]
- Moratalla-López, N.; Bagur, M.J.; Lorenzo, C.; Salinas, M.E.M.-N.R.; Alonso, G.L. Bioactivity and Bioavailability of the Major Metabolites of Crocus sativus L. Flower. Molecules 2019, 24, 2827. [Google Scholar] [CrossRef]
- Hosseini, A.; Razavi, B.M.; Hosseinzadeh, H. Saffron (Crocus sativus) petal as a new pharmacological target: A review. Iran. J. Basic Med. Sci 2018, 21, 1091–1099. [Google Scholar] [PubMed]
- Caser, M.; Demasi, S.; Stelluti, S.; Donno, D.; Scariot, V. Crocus sativus L. Cultivation in Alpine Environments: Stigmas and Tepals as Source of Bioactive Compounds. Agronomy 2020, 10, 1473. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef] [PubMed]
- Caser, M.; Demasi, S.; Caldera, F.; Dhakar, N.K.; Trotta, F.; Scariot, V. Activity of Ailanthus altissima (Mill.) Swingle Extract as a Potential Bioherbicide for Sustainable Weed Management in Horticulture. Agronomy 2020, 10, 965. [Google Scholar] [CrossRef]
- Demasi, S.; Caser, M.; Vanara, F.; Fogliatto, S.; Vidotto, F.; Negre, M.; Trotta, F.; Scariot, V. Ailanthone from Ailanthus altissima (Mill.) Swingle as potential natural herbicide. Sci. Hortic. 2019, 257, 108702. [Google Scholar] [CrossRef]
- Caser, M.; Chitarra, W.; D’Angiolillo, F.; Perrone, I.; Demasi, S.; Lovisolo, C.; Pistelli, L.; Pistelli, L.; Scariot, V. Drought stress adaptation modulates plant secondary metabolite production in Salvia dolomitica Codd. Ind. Crop. Prod. 2019, 129, 85–96. [Google Scholar] [CrossRef]
- Aryee, A.N.; Agyei, D.; Akanbi, T.O. Food for Oxidative Stress Relief: Polyphenols. In Encyclopedia of Food Chemistry; Elsevier: Amsterdam, The Netherlands, 2018; pp. 392–398. [Google Scholar]
- Pires, T.C.P.; Barros, L.; Santos-Buelga, C.; Ferreira, I.C. Edible flowers: Emerging components in the diet. Trends Food Sci. Technol. 2019, 93, 244–258. [Google Scholar] [CrossRef]
- Brodowska, K.M. European Journal of Biological Research Natural flavonoids: Classification, potential role, and application of flavonoid analogues. Eur. J. Biol. Res. 2017, 7, 108–123. [Google Scholar] [CrossRef]
- Santos-Buelga, C.; González-Paramás, A.M. Anthocyanins. In Encyclopedia of Food Chemistry; Elsevier: Amsterdam, The Netherlands, 2019; pp. 10–21. [Google Scholar]
- Granger, M.; Eck, P. Dietary Vitamin C in Human Health. Adv. Food Nutr. Res. 2018, 83, 281–310. [Google Scholar] [CrossRef] [PubMed]
- Akram, N.A.; Shafiq, F.; Ashraf, M. Ascorbic acid-a potential oxidant scavenger and its role in plant development and abiotic stress tolerance. Front. Plant. Sci. 2017, 8, 613. [Google Scholar] [CrossRef] [PubMed]
- Garavand, F.; Rahaee, S.; Vahedikia, N.; Jafari, S.M. Different techniques for extraction and micro/nanoencapsulation of saffron bioactive ingredients. Trends Food Sci. Technol. 2019, 89, 26–44. [Google Scholar] [CrossRef]
- Donno, D.; Mellano, M.G.; Prgomet, Ž.; Cerutti, A.K.; Beccaro, G.L. Phytochemical characterization and antioxidant activity evaluation of mediterranean medlar fruit (Crataegus azarolus L.): Preliminary study of underutilized genetic resources as a potential source of health-promoting compound for food supplements. J. Food Nutr. Res. 2017, 56, 18–31. [Google Scholar]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for Analysis of Plant Phenolic Compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef] [PubMed]
- Aliaño-González, M.J.; Espada-Bellido, E.; Ferreiro-González, M.; Carrera, C.; Palma, M.; Ayuso, J.; Álvarez, J.A.; Barbero, G.F. Extraction of Anthocyanins and Total Phenolic Compounds from Açai (Euterpe oleracea Mart.) Using an Experimental Design Methodology. Part 2: Ultrasound-Assisted Extraction. Agronomy 2020, 10, 326. [Google Scholar] [CrossRef]
- Zhao, L.; Fan, H.; Zhang, M.; Chitrakar, B.; Bhandari, B.; Wang, B. Edible flowers: Review of flower processing and extraction of bioactive compounds by novel technologies. Food Res. Int. 2019, 126, 108660. [Google Scholar] [CrossRef]
- Medina-Torres, N.; Ayora-Talavera, T.; Espinosa-Andrews, H.; Sánchez-Contreras, A.; Pacheco, N. Ultrasound Assisted Extraction for the Recovery of Phenolic Compounds from Vegetable Sources. Agronomy 2017, 7, 47. [Google Scholar] [CrossRef]
- Goupy, P.; Vian, M.A.; Chemat, F.; Caris-Veyrat, C. Identification and quantification of flavonols, anthocyanins and lutein diesters in tepals of Crocus sativus by ultra performance liquid chromatography coupled to diode array and ion trap mass spectrometry detections. Ind. Crop. Prod. 2013, 44, 496–510. [Google Scholar] [CrossRef]
- A Review on the Extraction Methods Use in Medicinal Plants, Principle, Strength and Limitation. Med. Aromat. Plants 2015, 4, 196. [CrossRef]
- Da Porto, C.; Natolino, A. Extraction kinetic modelling of total polyphenols and total anthocyanins from saffron floral bio-residues: Comparison of extraction methods. Food Chem. 2018, 258, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Ameer, K.; Shahbaz, H.M.; Kwon, J.-H. Green Extraction Methods for Polyphenols from Plant Matrices and Their Byproducts: A Review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 295–315. [Google Scholar] [CrossRef]
- Wali, A.F.; Alchamat, H.A.A.; Hariri, H.K.; Hariri, B.K.; Menezes, G.A.; Zehra, U.; Rehman, M.U.; Ahmad, P. Antioxidant, Antimicrobial, Antidiabetic and Cytotoxic Activity of Crocus sativus L. Petals. Appl. Sci. 2020, 10, 1519. [Google Scholar] [CrossRef]
- Khazaei, K.M.; Jafari, S.M.; Ghorbani, M.; Kakhki, A.H.; Sarfarazi, M. Optimization of Anthocyanin Extraction from Saffron Petals with Response Surface Methodology. Food Anal. Methods 2015, 9, 1993–2001. [Google Scholar] [CrossRef]
- Lahmass, I.; Lamkami, T.; Delporte, C.; Sikdar, S.; Van Antwerpen, P.; Saalaoui, E.; Megalizzi, V. The waste of saffron crop, a cheap source of bioactive compounds. J. Funct. Foods 2017, 35, 341–351. [Google Scholar] [CrossRef]
- Sánchez-Vioque, R.; Santana-Méridas, O.; Polissiou, M.; Vioque, J.; Astraka, K.; Alaiz, M.; Herraiz-Peñalver, D.; Tarantilis, P.A.; Girón-Calle, J. Polyphenol composition and in vitro antiproliferative effect of corm, tepal and leaf from Crocus sativus L. on human colon adenocarcinoma cells (Caco-2). J. Funct. Foods 2016, 24, 18–25. [Google Scholar] [CrossRef]
- Jadouali, S.M.; Atifi, H.; Mamouni, R.; Majourhat, K.; Bouzoubaâ, Z.; Laknifli, A.; Faouzi, A. Chemical characterization and antioxidant compounds of flower parts of Moroccan crocus sativus L. J. Saudi Soc. Agric. Sci. 2019, 18, 476–480. [Google Scholar] [CrossRef]
- Lotfi, L.; Kalbasi-Ashtari, A.; Hamedi, M.; Ghorbani, F. Effects of sulfur water extraction on anthocyanins properties of tepals in flower of saffron (Crocus sativus L). J. Food Sci. Technol. 2013, 52, 813–821. [Google Scholar] [CrossRef][Green Version]
- Lotfi, L.; Kalbasi-Ashtari, A.; Hamedi, M.; Ghorbani, F. Effects of enzymatic extraction on anthocyanins yield of saffron tepals (Crocos sativus) along with its color properties and structural stability. J. Food Drug Anal. 2015, 23, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Díaz, J.; Sanchez, A.M.; Maggi, L.; Martínez-Tomé, M.; García-Diz, L.; Murcia, M.A.; Alonso, G.L. Increasing the Applications ofCrocus sativusFlowers as Natural Antioxidants. J. Food Sci. 2012, 77, C1162–C1168. [Google Scholar] [CrossRef]
- Cusano, E.; Consonni, R.; Petrakis, E.A.; Astraka, K.; Cagliani, L.R.; Polissiou, M.G. Integrated analytical methodology to investigate bioactive compounds in Crocus sativus L. flowers. Phytochem. Anal. 2018, 29, 476–486. [Google Scholar] [CrossRef]
- Jafari, S.M.; Khazaei, K.M.; Assadpour, E. Production of a natural color through microwave-assisted extraction of saffron tepal’s anthocyanins. Food Sci. Nutr. 2019, 7, 1438–1445. [Google Scholar] [CrossRef]
- Ghanbari, J.; Khajoei-Nejad, G.; Van Ruth, S.M. Effect of saffron (Crocus sativus L.) corm provenance on its agro-morphological traits and bioactive compounds. Sci. Hortic. 2019, 256, 108605. [Google Scholar] [CrossRef]
- Moratalla-López, N.; Lorenzo, C.; Chaouqi, S.; Sánchez, A.M.; Alonso, G.L. Kinetics of polyphenol content of dry flowers and floral bio-residues of saffron at different temperatures and relative humidity conditions. Food Chem. 2019, 290, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Goli, S.A.H.; Mokhtari, F.; Rahimmalek, M. Phenolic Compounds and Antioxidant Activity from Saffron (Crocus sativus L.) Petal. J. Agric. Sci. 2012, 4, 4. [Google Scholar] [CrossRef]
- Cai, Y.; Luo, Q.; Sun, M.; Corke, H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci. 2004, 74, 2157–2184. [Google Scholar] [CrossRef]
- Li, H.; Deng, Z.; Zhu, H.; Hu, C.; Liu, R.; Young, J.C.; Tsao, R. Highly pigmented vegetables: Anthocyanin compositions and their role in antioxidant activities. Food Res. Int. 2012, 46, 250–259. [Google Scholar] [CrossRef]
- Solymosi, K.; Latruffe, N.; Morantmanceau, A.; Schoefs, B. Food colour additives of natural origin. In Colour Additives for Foods and Beverages; Elsevier: Amsterdam, The Netherlands, 2015; pp. 3–34. [Google Scholar]
- Ribeiro, L.; Ribani, R.; Francisco, T.; Soares, A.; Pontarolo, R.; Haminiuk, C. Profile of bioactive compounds from grape pomace (Vitis vinifera and Vitis labrusca) by spectrophotometric, chromatographic and spectral analyses. J. Chromatogr. B 2015, 1007, 72–80. [Google Scholar] [CrossRef]
- Markakis, P. Anthocyanins as Food Additives. In Anthocyanins as Food Colors; Elsevier: Amsterdam, The Netherlands, 1982; pp. 245–253. [Google Scholar]
- Thoo, Y.Y.; Ho, S.K.; Liang, J.Y.; Ho, C.W.; Tan, C.P. Effects of binary solvent extraction system, extraction time and extraction temperature on phenolic antioxidants and antioxidant capacity from mengkudu (Morinda citrifolia). Food Chem. 2010, 120, 290–295. [Google Scholar] [CrossRef]
- Huang, H.; Sun, Y.; Lou, S.; Li, H.; Ye, X. In vitro digestion combined with cellular assay to determine the antioxidant activity in Chinese bayberry (Myrica rubra Sieb. et Zucc.) fruits: A comparison with traditional methods. Food Chem. 2014, 146, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Donno, D.; Beccaro, G.; Mellano, M.; Cerutti, A.; Bounous, G. Goji berry fruit (Lycium spp.): Antioxidant compound fingerprint and bioactivity evaluation. J. Funct. Foods 2015, 18, 1070–1085. [Google Scholar] [CrossRef]
- Babbar, N.; Oberoi, H.S.; Uppal, D.S.; Patil, R.T. Total phenolic content and antioxidant capacity of extracts obtained from six important fruit residues. Food Res. Int. 2011, 44, 391–396. [Google Scholar] [CrossRef]
- Terpinc, P.; Čeh, B.; Ulrih, N.P.; Abramovič, H. Studies of the correlation between antioxidant properties and the total phenolic content of different oil cake extracts. Ind. Crop. Prod. 2012, 39, 210–217. [Google Scholar] [CrossRef]
- Kalinowska, M.; Gryko, K.; Wróblewska, A.M.; Jabłońska-Trypuć, A.; Karpowicz, D. Phenolic content, chemical composition and anti-/pro-oxidant activity of Gold Milenium and Papierowka apple peel extracts. Sci. Rep. 2020, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bilek, S.E. The effects of time, temperature, solvent: Solid ratio and solvent composition on extraction of total phenolic compound from dried olive (Olea europaea L.) leaves. GIDA 2010, 35, 411–416. [Google Scholar]
- Safaiee, P.; Taghipour, A.; Vahdatkhoram, F.; Movagharnejad, K. Extraction of phenolic compounds from Mentha aquatica: The effects of sonication time, temperature and drying method. Chem. Pap. 2019, 73, 3067–3073. [Google Scholar] [CrossRef]
- Liu, Z.; Esveld, E.; Vincken, J.-P.; Bruins, M.E. Pulsed Electric Field as an Alternative Pre-treatment for Drying to Enhance Polyphenol Extraction from Fresh Tea Leaves. Food Bioprocess. Technol. 2019, 12, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, R.; Ahmed, H.; Dixit, R.; Dharamveer; Saraf, S. Crocus sativus L.: A comprehensive review. Pharmacogn. Rev. 2010, 4, 200–208. [Google Scholar] [CrossRef]
- Donno, D.; Mellano, M.; Prgomet, Z.; Beccaro, G. Advances in Ribes x nidigrolaria Rud. Bauer & A. Bauer fruits as potential source of natural molecules: A preliminary study on physico-chemical traits of an underutilized berry. Sci. Hortic. 2018, 237, 20–27. [Google Scholar] [CrossRef]
- Laufenberg, G.; Kunz, B.; Nystroem, M. Transformation of vegetable waste into value added products:(A) the upgrading concept;(B) practical implementations. Bioresour. Technol. 2003, 87, 167–198. [Google Scholar] [CrossRef]
- Philippini, R.R.; Martiniano, S.E.; Ingle, A.P.; Marcelino, P.R.F.; Silva, G.M.; Barbosa, F.G.; Dos Santos, J.C.; Silva, S.S. Agroindustrial Byproducts for the Generation of Biobased Products: Alternatives for Sustainable Biorefineries. Front. Energy Res. 2020, 8, 1–23. [Google Scholar] [CrossRef]
- Motola, V.; De Bari, I.; Pierro, N.; Giocoli, A. Bioeconomy and Biorefining Strategies in the EU Member States and Beyond—Reference Year 2018; IEA Bioenergy: Rome, Italy, 2018; ISBN 978-1-910154-60-1. [Google Scholar]
- European Commission. A Sustainable Bioeconomy for Europe: Strengthening the Connection between Economy, Society and the Environment; European Commission: Brussels, Belgium, 2018; ISBN 9789279941450. [Google Scholar]
- Bruusgaard-Mouritsen, M.A.; Johansen, J.D.; Zachariae, C.; Kirkeby, C.S.; Garvey, L.H. Natural ingredients in cosmetic products—A suggestion for a screening series for skin allergy. Contact Dermat. 2020, 83, 251–270. [Google Scholar] [CrossRef]
| Extraction | TPC (mgGAE 100 g−1 DW) | TAC (mgG3G 100 g−1 DW) | FRAP (mmolFe2+ Kg−1 DW) | ABTS (μmolTE g−1 DW) | DPPH (μmolTE g−1 DW) | |
|---|---|---|---|---|---|---|
| M | Water | 1142.27 ± 43.52 | 345.04 ± 132.47 a,b | 571.54 ± 3.21 a | 13.82 ± 0.72 | 15.56 ± 2.29 |
| M | Met20 | 1123.53 ± 59.86 | 268.13 ± 26.76 a,b | 506.73 ± 13.85 b,c | 14.20 ± 0.60 | 17.83 ± 2.46 |
| M | Met50 | 1106.45 ± 9.17 | 300.39 ± 15.02 a,b | 535.83 ± 10.30 a,b | 14.62 ± 0.29 | 24.52 ± 2.55 |
| M | Met80 | 1166.96 ± 33.15 | 249.13 ± 11.97 a,b | 511.72 ± 22.49 a,b,c | 14.29 ± 0.32 | 24.17 ± 1.53 |
| UAE | Water | 1150.63 ± 11.23 | 413.30 ± 137.16 a | 556.90 ± 11.91 a,b | 12.76 ± 0.81 | 23.55 ± 3.60 |
| UAE | Met20 | 1113.27 ± 46.11 | 178.39 ± 34.03 b | 460.05 ± 35.55 c | 13.39 ± 1.46 | 19.35 ± 4.83 |
| UAE | Met50 | 1066.89 ± 26.36 | 277.09 ± 49.06 a,b | 506.68 ± 21.80 b,c | 15.10 ± 0.38 | 24.58 ± 1.46 |
| UAE | Met80 | 1153.49 ± 22.74 | 231.70 ± 30.19 a,b | 513.67 ± 21.12 a,b,c | 14.34 ± 0.81 | 21.03 ± 1.81 |
| p | ns | 0.01413 * | 0.0002608 *** | ns | ns | |
| TPC | TAC | FRAP | ABTS | |
|---|---|---|---|---|
| M | ||||
| FRAP | ns | ns | / | |
| ABTS | ns | ns | ns | / |
| DPPH | ns | ns | ns | ns |
| UAE | ||||
| FRAP | ns | 0.86 *** | / | ns |
| ABTS | −0.69 * | ns | ns | / |
| DPPH | −0.45 * | ns | ns | ns |
| Extractions | Cinnamic Acids | Benzoic Acids | Flavonols | Catechins | Vitamin C | ||||
|---|---|---|---|---|---|---|---|---|---|
| Ferulic Acid | Ellagic Acid | Hyperoside | Isoquercitrin | Quercitrin | Rutin | Epicatechin | |||
| M | Water | 0.00 ± 0.00 b | 7.67 ± 3.69 a,b,c | 4.35 ± 1.04 c | 0.31 ± 0.22 c,d | 0.00 ± 0.00 b | 8.52 ± 3.91 c | 0.00 ± 0.00 b | 29.61 ± 6.05 a |
| M | Met20 | 1.83 ± 0.31 a | 4.43 ± 4.15 c,d | 5.61 ± 0.52 a,b,c | 0.22 ± 0.12 c,d | 6.33 ± 5.27 a | 0.32 ± 0.31 d | 0.00 ± 0.00 b | 33.72 ± 0.89 a |
| M | Met50 | 9.65 ± 2.62 a | 0.00 ± 0.00 e | 5.85 ± 4.31 b,c | 4.36 ± 3.49 a,b,c | 9.27 ± 3.47 a | 0.00 ± 0.00 d | 0.00 ± 0.00 b | 0.00 ± 0.00 b |
| M | Met80 | 0.00 ± 0.00 b | 1.32 ± 0.33 d,e | 23.93 ± 15.51 a,b,c | 7.82 ± 3.09 a | 6.53 ± 0.29 a | 37.61 ± 2.22 a | 0.00 ± 0.00 b | 0.00 ± 0.00 b |
| UAE | Water | 0.00 ± 0.00 b | 8.53 ± 8.45 b,c,d | 11.58 ± 4.09 a,b,c | 0.00 ± 0.00 d | 0.00 ± 0.00 b | 28.24 ± 4.83 a,b | 0.00 ± 0.00 b | 26.68 ± 4.71 a |
| UAE | Met20 | 0.00 ± 0.00 b | 26.74 ± 10.80 a,b | 9.68 ± 6.77 a,b,c | 6.46 ± 5.03 a,b | 0.00 ± 0.00 b | 13.46 ± 10.25 b,c | 16.62 ± 15.89 a | 29.17 ± 2.31 a |
| UAE | Met50 | 0.00 ± 0.00 b | 28.39 ± 4.32 a | 27.26 ± 4.29 a | 5.57 ± 1.90 a,b,c | 7.07 ± 5.12 a | 7.24 ± 1.35 c | 0.00 ± 0.00 b | 0.00 ± 0.00 b |
| UAE | Met80 | 0.00 ± 0.00 b | 23.51 ± 5.11 a,b | 24.77 ± 2.25 a,b | 0.00 ± 0.00 d | 0.00 ± 0.00 b | 9.10 ± 2.17 c | 4.22 ± 2.90 a | 0.00 ± 0.00 b |
| p | 0.001802 ** | 2.235 × 10−7 *** | 0.004662 ** | 0.005466 ** | 0.005407 ** | 1.452 × 10−10 *** | 0.001995 ** | 0.003143 ** | |
| Tepals State | Extraction Methods | Solvents | Bioactive Compounds | References |
|---|---|---|---|---|
| Fresh | Maceration, UAE | Water and methanol at 20%, 50% and 80% | Phenolic content (ellagic acid; hyperoside; rutin; epicatechin); Anthocyanin content | [15] |
| Dried | Soxhlet extraction | Hexane; dichloromethane; ethanol | Phenolic and Flavonoid content | [37] |
| Dried | Maceration | Ethanol 25%, 50%, 75% | Anthocyanin content | [38] |
| Dried | Maceration | Ethyl acetate; methanol | Phenolic and Flavonoid content | [39] |
| Dried | Maceration | Ethanol 70% | Phenolic content (kaempferol 3-O-sophoroside-7-O-glycoside, quercetin 3,4-di-O-glycoside, kaempferol di-glycoside, kaempferol 3-O-glycoside) | [40] |
| Dried | Maceration | Methanol | Phenolic and Flavonoid content | [41] |
| Dried | Maceration | Acidified (HCl) ethanol; sulfur water solution | Phenolic and Anthocyanin content (cyanidin 3,5-diglucosides; pelargonidin 3- and 5-glucosides; delphinidin di-glucosides; pelargonidin 3-glucosides; petunidin) | [42] |
| Dried | Maceration, Enzyme-assisted extraction | Acidified (HCl) ethanol; Enzymatic water (Pectinex) | Antocyanin content (cyanidin 3,5-diglycosides; pelargonidin 3- and 3,5-glycosides; delphinidin 3-glycosides; petunidin) | [43] |
| Freeze-dried | Maceration, UAE | Acidified (HCl) methanol; Acidified (HCl) ethanol 70%; Methanol 98% in formic acid | Phenolic, Flavonoid, Anthocyanin, and Crocetin esters content | [44] |
| Dried | Maceration, UAE | Acidified (HCl) deuterated methanol; Trifluoroacetic acid in acetonitrile 50% | Flavonoids (kaempferol di-hexoside; kaempferol 3-O-glucoside; kaempferol 3,4′-di-O-glucoside; kaempferol 3-O-β-sophoroside; kaempferol tri-hexoside; kaempferol di-hexosides; quercetin 3,4′-di-O-glucoside; isorhamnetin 3,4′-di-O-glucoside); Anthocyanins (delphinidin 3,5-di-O-β-glucoside; delphinidin 3-O-glucoside; petunidin 3,5-di-O-β-glucoside; petunidin 3-O-glucoside) | [45] |
| Dried | UAE | Ethanol 50% | Flavonoids (quercetin 3-O-sophoroside; kaempferol 3-O-sophoroside; kaempferol 3-O-glucoside) | [46] |
| Freeze-dried | Maceration, UAE | Methanol 50%; n-hexane; Methanol/KOH 20% | Flavonoid content (kaempferol 3-O-sophoroside-7-O-glucoside; kaempferol 3,7-O-diglucoside; quercetin 3,7-O-diglucoside; isorhamnetin 3,7-O-diglucoside; kaempferol 3-O-sophoroside; isorhamnetin 3-O-sophoroside; quercetin 3-O-glucoside; kaempferol 3-O-rutinoside; isorhamnetin 3-O-rutinoside; kaempferol 3-O-glucoside; kaempferol 3-O-(6??- acetyl-glycoside)-7-O-glycoside; isorhamnetin 3-O-glucoside; kaempferol 3-O-sophoroside-7-O-rhamnoside; kaempferol 3-O-(6??- acetyl-galactoside) or 3-O-(6??-acetyl- glucoside); kaempferol 3-O-(6??- acetyl-galactoside) or 3-O-(6??-acetyl- glucoside); quercetin 3-O-glucoside-7-O-rhamnoside; isorhamnetin 3-O-glucoside-7-O-rhamnoside; kaempferol 3-O-glucoside-7-O-rhamnoside); Anthocyanin content (delphinidin 3,7-O-diglucoside; petunidin 3,7-O-diglucoside; delphinidin 3-O-glucoside; petunidin 3-O-glucoside; malvidin O-glucoside); Lutein diesters | [32] |
| Dried | Microwave-assisted extraction | Acidified (HCl) ethanol | Anthocyanin content | [46] |
| Methods | Classes of Interest | Stationary Phase | Mobile Phase | Wavelength (nm) |
|---|---|---|---|---|
| A | Cinnamic acids, Flavonols | KINETEX—C18 column (4.6 × 150 mm2, 5 μm) | A: 10 mM KH2PO4/H3PO4, pH = 2.8 B: CH3CN | 330 |
| B | Benzoic acids, catechins | KINETEX—C18 column (4.6 × 150 mm2, 5 μm) | A: H2O/CH3OH/HCOOH (5:95:0.1 v/v/v), pH = 2.5 B: CH3OH/HCOOH (100:0.1 v/v) | 280 |
| C | Vitamin C | KINETEX—C18 column (4.6 × 150 mm2, 5 μm) | A: 5 mM C16H33N(CH3Br/50 mM KH2PO4, pH = 2.5 B: CH3OH | 261, 348 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stelluti, S.; Caser, M.; Demasi, S.; Scariot, V. Sustainable Processing of Floral Bio-Residues of Saffron (Crocus sativus L.) for Valuable Biorefinery Products. Plants 2021, 10, 523. https://doi.org/10.3390/plants10030523
Stelluti S, Caser M, Demasi S, Scariot V. Sustainable Processing of Floral Bio-Residues of Saffron (Crocus sativus L.) for Valuable Biorefinery Products. Plants. 2021; 10(3):523. https://doi.org/10.3390/plants10030523
Chicago/Turabian StyleStelluti, Stefania, Matteo Caser, Sonia Demasi, and Valentina Scariot. 2021. "Sustainable Processing of Floral Bio-Residues of Saffron (Crocus sativus L.) for Valuable Biorefinery Products" Plants 10, no. 3: 523. https://doi.org/10.3390/plants10030523
APA StyleStelluti, S., Caser, M., Demasi, S., & Scariot, V. (2021). Sustainable Processing of Floral Bio-Residues of Saffron (Crocus sativus L.) for Valuable Biorefinery Products. Plants, 10(3), 523. https://doi.org/10.3390/plants10030523








