Chemical Constituents of the Leaves of Diospyros kaki (Persimmon)
Abstract
1. Introduction
2. Results and Discussion
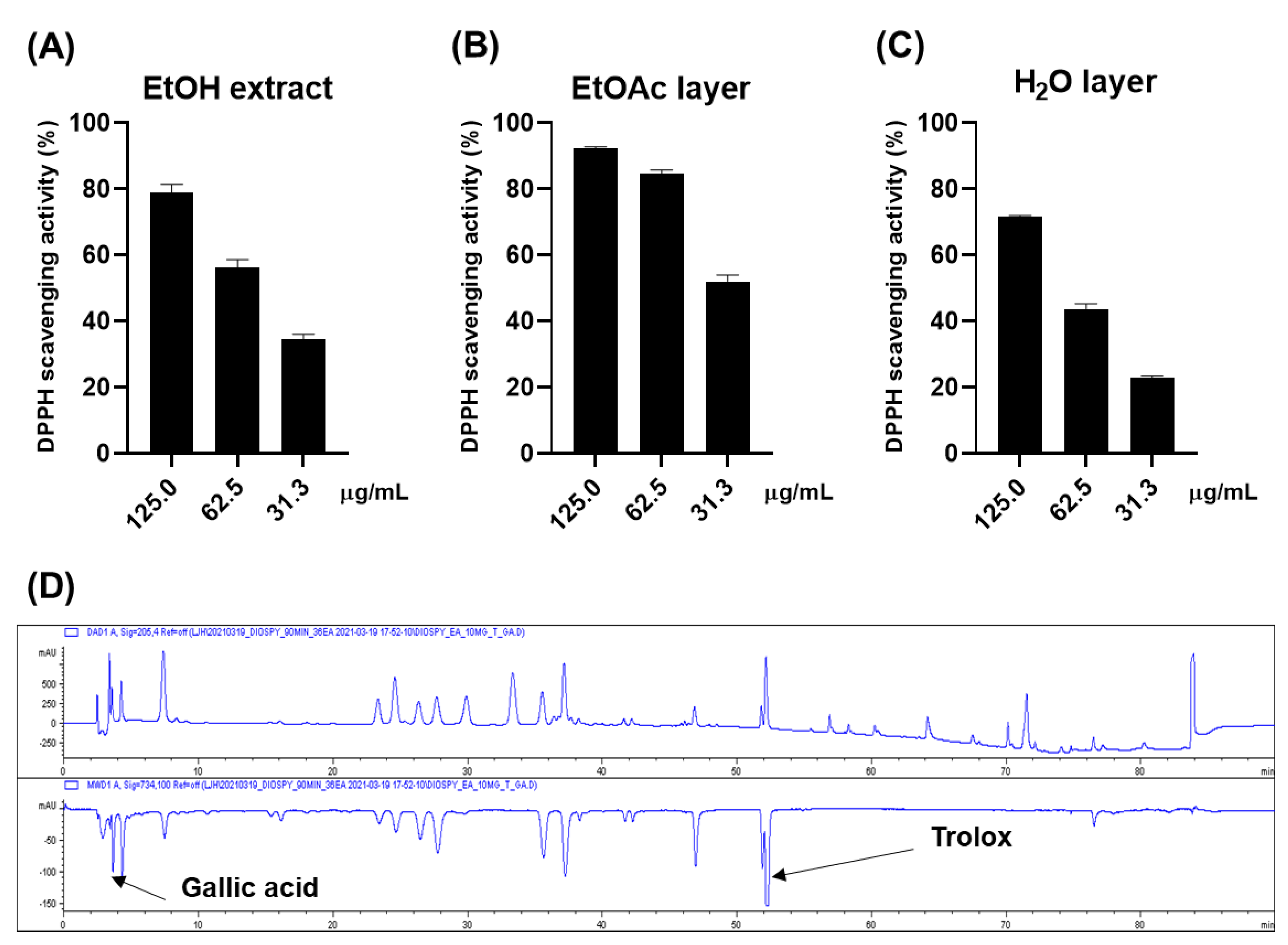
2.1. Antioxidative Effect of the Persimmon Leaves
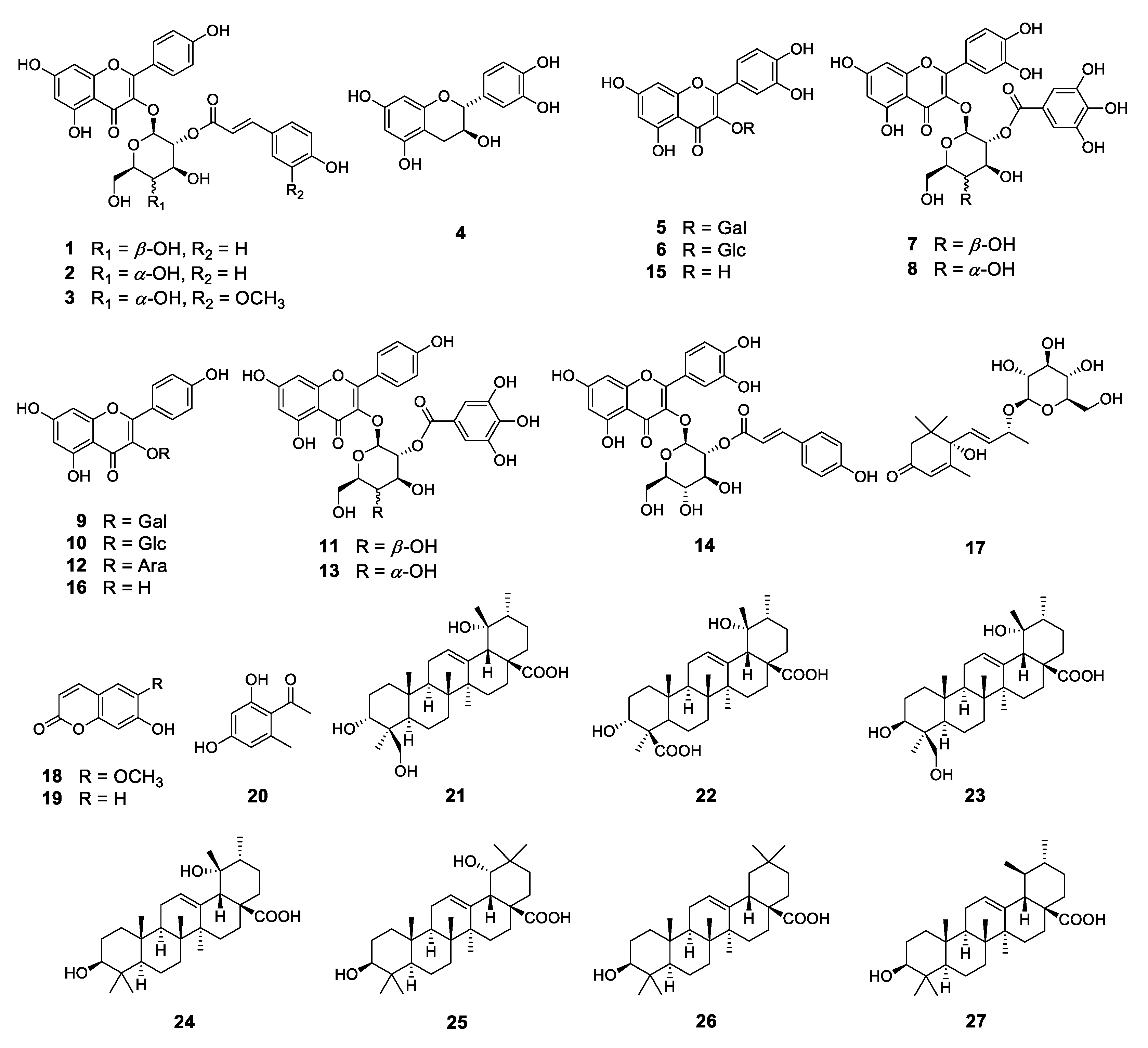
2.2. Phytochemical Investigation
2.3. Antioxidative Activities of the Isolated Compounds
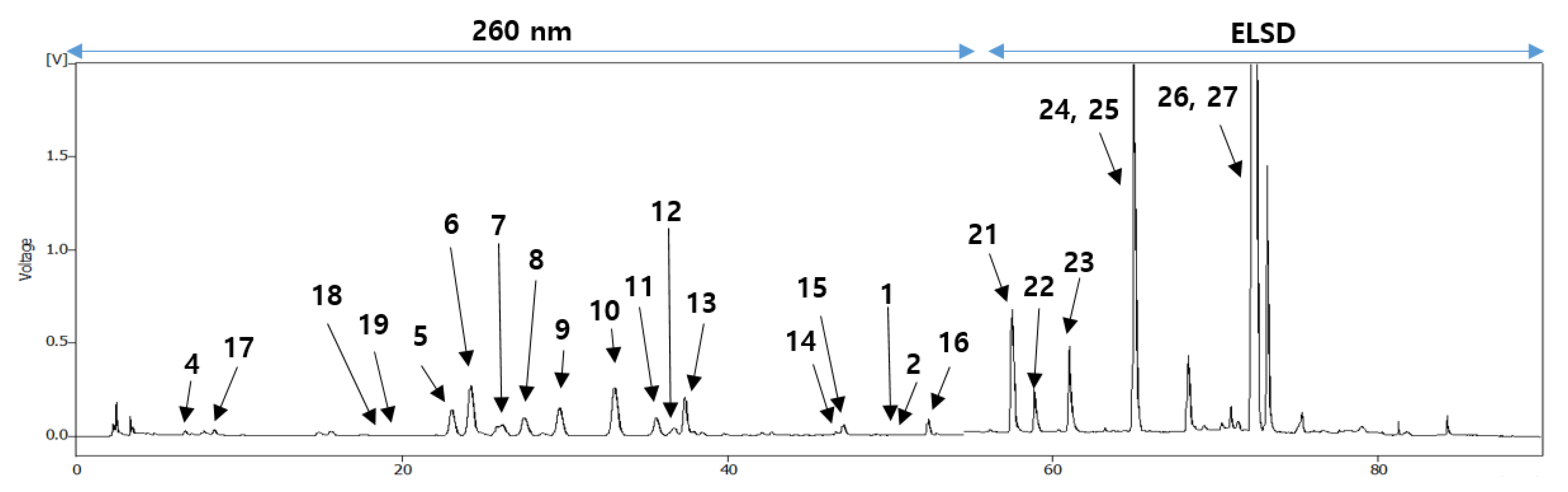
2.4. Quantitative Analysis of Isolated Compounds
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. General Experimental Procedures
4.3. Extraction and Isolation
4.3.1. kaempferol-3-O-β-d-2′′-coumaroylgalactoside (1)
4.3.2. kaempferol-3-O-β-d-2′′-feruloylglucoside (3)
4.3.3. kaempferol-3-O-β-d-2′′-galloylgalactoside (11)
4.4. Quantitative Analysis of Nine Compounds in the Persimmon Leaves
4.5. DPPH and Online HPLC-ABTS Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Xie, C.; Xie, Z.; Xu, X.; Yang, D. Persimmon (Diospyros kaki L.) leaves: A review on traditional uses, phytochemistry and pharmacological properties. J. Ethnopharmacol. 2015, 163, 229–240. [Google Scholar] [CrossRef]
- Bei, W.; Zang, L.; Guo, J.; Peng, W.; Xu, A.; Good, D.A.; Hu, Y.; Wu, W.; Hu, D.; Zhu, X. Neuroprotective effects of a standardized flavonoid extract from Diospyros kaki leaves. J. Ethnopharmacol. 2009, 126, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Sakanaka, S.; Tachibana, Y.; Okada, Y. Preparation and antioxidant properties of extracts of Japanese persimmon leaf tea (kakinoha-cha). Food Chem. 2005, 89, 569–575. [Google Scholar] [CrossRef]
- Sa, Y.S.; Kim, S.-J.; Choi, H.-S. The anticoagulant fraction from the leaves of Diospyros kaki L. has an antithrombotic activity. Arch. Pharmacal Res. 2005, 28, 667–674. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Zhang, Y.; Zhang, M.; Gu, L.; Liu, Z.; Jia, J.; Chen, X. Effects of phospholipid complexes of total flavonoids from Persimmon (Diospyros kaki L.) leaves on experimental atherosclerosis rats. J. Ethnopharmacol. 2016, 191, 245–253. [Google Scholar] [CrossRef]
- Kotani, M.; Matsumoto, M.; Fujita, A.; Higa, S.; Wang, W.; Suemura, M.; Kishimoto, T.; Tanaka, T. Persimmon leaf extract and astragalin inhibit development of dermatitis and IgE elevation in NC/Nga mice. J. Allergy Clin. Immunol. 2000, 106, 159–166. [Google Scholar] [CrossRef]
- Thuong, P.T.; Lee, C.H.; Dao, T.T.; Nguyen, P.H.; Kim, W.G.; Lee, S.J.; Oh, W.K. Triterpenoids from the leaves of Diospyros kaki (persimmon) and their inhibitory effects on protein tyrosine phosphatase 1B. J. Nat. Prod. 2008, 71, 1775–1778. [Google Scholar] [CrossRef]
- Matsuo, T.; Ito, S. The chemical structure of kaki-tannin from immature fruit of the persimmon (Diospyros kaki L.). Agric. Biol. Chem. 1978, 42, 1637–1643. [Google Scholar] [CrossRef]
- Bawazeer, S.; Rauf, A. In vivo anti-inflammatory, analgesic, and sedative studies of the extract and naphthoquinone isolated from Diospyros kaki (persimmon). ACS Omega 2021, 6, 9852–9856. [Google Scholar] [CrossRef]
- Yoshimura, M.; Mochizuki, A.; Amakura, Y. Identification of phenolic constituents and inhibitory activity of persimmon calyx and shiteito against tumor cell proliferation. Chem. Pharm. Bull. 2021, 69, 32–39. [Google Scholar] [CrossRef]
- Wang, L.; Xu, M.L.; Rasmussen, S.K.; Wang, M.-H. Vomifoliol 9-O-α-arabinofuranosyl (1→ 6)-β-D-glucopyranoside from the leaves of Diospyros Kaki stimulates the glucose uptake in HepG2 and 3T3-L1 cells. Carbohydr. Res. 2011, 346, 1212–1216. [Google Scholar] [CrossRef] [PubMed]
- Hitaka, Y.; Nakano, A.; Tsukigawa, K.; Manabe, H.; Nakamura, H.; Nakano, D.; Kinjo, J.; Nohara, T.; Maeda, H. Characterization of carotenoid fatty acid esters from the peels of the persimmon Diospyros kaki. Chem. Pharm. Bull. 2013, 61, 666–669. [Google Scholar] [CrossRef]
- Simpson, D.S.; Oliver, P.L. ROS generation in microglia: Understanding oxidative stress and inflammation in neurodegenerative disease. Antioxidants 2020, 9, 743. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ren, Z.; Zhang, J.; Chuang, C.-C.; Kandaswamy, E.; Zhou, T.; Zuo, L. Role of ROS and nutritional antioxidants in human diseases. Front. Physiol. 2018, 9, 477. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.; Hwang, H.; Selvaraj, B.; Lee, J.H.; Park, W.; Ryu, S.M.; Lee, D.; Park, J.-S.; Kim, H.S.; Lee, J.W. Phenolic constituents isolated from Senna tora sprouts and their neuroprotective effects against glutamate-induced oxidative stress in HT22 and R28 cells. Bioorganic Chem. 2021, 114, 105112. [Google Scholar] [CrossRef] [PubMed]
- Romussi, G.; Bignardi, G.; Pizza, C.; De Tommasi, N. Constituents of cupuliferae, XIII: New and revised structures of acylated flavonoids from Quercus Suber L. Arch. Der Pharm. 1991, 324, 519–524. [Google Scholar] [CrossRef]
- Li, H.-Z.; Song, H.-J.; Li, H.-M.; Pan, Y.-Y.; Li, R.-T. Characterization of phenolic compounds from Rhododendron alutaceum. Arch. Pharmacal Res. 2012, 35, 1887–1893. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.; Choi, J.; Chae, H.-S.; Cho, J.Y.; Kim, Y.-D.; Htwe, K.M.; Lee, W.-S.; Chin, Y.-W.; Kim, J.; Yoon, K.D. Flavonoids from Symplocos racemosa. Molecules 2015, 20, 358–365. [Google Scholar] [CrossRef]
- Xu, J.; Wang, X.; Yue, J.; Sun, Y.; Zhang, X.; Zhao, Y. Polyphenols from acorn leaves (Quercus liaotungensis) protect pancreatic beta cells and their inhibitory activity against α-glucosidase and protein tyrosine phosphatase 1B. Molecules 2018, 23, 2167. [Google Scholar] [CrossRef]
- Botirov, E.K. Flavonoids and phenolcarboxylic acids from Lamium album. Chem. Nat. Compd. 2019, 55, 1159–1160. [Google Scholar] [CrossRef]
- Kawakami, K.; Shibukura, Y.; Kanno, T.; Furuki, T.; Aketa, S.; Hirayama, M. Identification of 2′′-galloylated flavonol 3-O-glycosides accumulating in developing leaves of Persimmon. Phytochem. Anal. 2011, 22, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Suktap, C.; Lee, H.K.; Amnuaypol, S.; Suttisri, R.; Sukrong, S. Wound healing effect of flavonoid glycosides from Afgekia mahidolae BL Burtt & Chermsir. Leaves. Rec. Nat. Prod. 2018, 12, 391–396. [Google Scholar]
- Isobe, T.; Ito, N.; Noda, Y. Minor flavonoids of Polygonum nodosum. Phytochemistry 1980, 19, 1877. [Google Scholar] [CrossRef]
- Wan, C.; Yuan, T.; Cirello, A.L.; Seeram, N.P. Antioxidant and α-glucosidase inhibitory phenolics isolated from highbush blueberry flowers. Food Chem. 2012, 135, 1929–1937. [Google Scholar] [CrossRef]
- Liao, C.-R.; Kuo, Y.-H.; Ho, Y.-L.; Wang, C.-Y.; Yang, C.-S.; Lin, C.-W.; Chang, Y.-S. Studies on cytotoxic constituents from the leaves of Elaeagnus oldhamii Maxim. in non-small cell lung cancer A549 cells. Molecules 2014, 19, 9515–9534. [Google Scholar] [CrossRef]
- Wu, Z.-G.; Wei, W.; Xu, H.-Y.; Zheng, L.-L.; Ma, C.-M.; Wang, Y.-C. Constituents from the leaves of Tetraena mongolica and their protective activity in HEK 293t cells damaged by CdCl2. J. Nat. Prod. 2019, 82, 2707–2712. [Google Scholar] [CrossRef]
- Kang, Y.-F.; Liu, C.-M.; Kao, C.-L.; Chen, C.-Y. Antioxidant and anticancer constituents from the leaves of Liriodendron tulipifera. Molecules 2014, 19, 4234–4245. [Google Scholar] [CrossRef]
- Kwon, J.; Hiep, N.T.; Kim, D.-W.; Hong, S.; Guo, Y.; Hwang, B.Y.; Lee, H.J.; Mar, W.; Lee, D. Chemical constituents isolated from the root bark of Cudrania tricuspidata and their potential neuroprotective effects. J. Nat. Prod. 2016, 79, 1938–1951. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Mu, Y.; An, Q.; Xun, J.; Ma, J.; Wu, W.; Xu, M.; Xu, J.; Han, L.; Huang, X. Total synthesis and anti-inflammatory evaluation of violacin A and its analogues. Bioorganic Chem. 2020, 94, 103420. [Google Scholar] [CrossRef]
- Su, B.-N.; Kang, Y.-H.; Pinos, R.E.; Santarsiero, B.D.; Mesecar, A.D.; Soejarto, D.D.; Fong, H.H.; Pezzuto, J.M.; Kinghorn, A.D. Isolation and absolute stereochemistry of coussaric acid, a new bioactive triterpenoid from the stems of Coussarea brevicaulis. Phytochemistry 2003, 64, 293–302. [Google Scholar] [CrossRef]
- Nakatani, M.; Miyazaki, Y.; Iwashita, T.; Naoki, H.; Hase, T. Triterpenes from Ilex rotunda fruits. Phytochemistry 1989, 28, 1479–1482. [Google Scholar] [CrossRef]
- Lee, T.H.; Juang, S.H.; Hsu, F.L.; Wu, C.Y. Triterpene acids from the leaves of Planchonella duclitan (Blanco) Bakhuizan. J. Chin. Chem. Soc. 2005, 52, 1275–1280. [Google Scholar] [CrossRef]
- Mimaki, Y.; Fukushima, M.; Yokosuka, A.; Sashida, Y.; Furuya, S.; Sakagami, H. Triterpene glycosides from the roots of Sanguisorba officinalis. Phytochemistry 2001, 57, 773–779. [Google Scholar] [CrossRef]
- Fattorusso, E.; Lanzotti, V.; Taglialatela-Scafati, O.; Cicala, C. The flavonoids of leek, Allium porrum. Phytochemistry 2001, 57, 565–569. [Google Scholar] [CrossRef]
- Yoshida, T.; Saito, T.; Kadoya, S. New acylated flavonol glucosides in Allium tuberosum Rottler. Chem. Pharm. Bull. 1987, 35, 97–107. [Google Scholar] [CrossRef][Green Version]
- Zehl, M.; Braunberger, C.; Conrad, J.; Crnogorac, M.; Krasteva, S.; Vogler, B.; Beifuss, U.; Krenn, L. Identification and quantification of flavonoids and ellagic acid derivatives in therapeutically important Drosera species by LC–DAD, LC–NMR, NMR, and LC–MS. Anal. Bioanal. Chem. 2011, 400, 2565–2576. [Google Scholar] [CrossRef] [PubMed]
- Hossain, A.; Moon, H.K.; Kim, J.-K. Antioxidant properties of Korean major persimmon (Diospyros kaki) leaves. Food Sci. Biotechnol. 2018, 27, 177–184. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, J.; Lu, X.; Zhang, L.; Zhang, Y. Evaluation to the antioxidant activity of total flavonoids extract from persimmon (Diospyros kaki L.) leaves. Food Chem. Toxicol. 2011, 49, 2689–2696. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.-W.; Qiao, J.-W.; Sun, X.; Gao, P.-Y.; Li, L.-Z.; Liu, Q.-B.; Sun, B.; Wu, D.-L.; Song, S.-J. Secoiridoids and lignans from the leaves of Diospyros kaki Thunb. with antioxidant and neuroprotective activities. J. Funct. Foods 2016, 24, 183–195. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Said, A.; Tundis, R.; Hawas, U.W.; Rashed, K.; Menichini, F.; Frega, N.G.; Menichini, F. Antioxidant and antiproliferative activity of Diospyros lotus L. extract and isolated compounds. Plant Foods Hum. Nutr. 2009, 64, 264. [Google Scholar] [CrossRef]
- Masuda, T.; Iritani, K.; Yonemori, S.; Oyama, Y.; Takeda, Y. Isolation and antioxidant activity of galloyl flavonol glycosides from the seashore plant, Pemphis acidula. Biosci. Biotechnol. Biochem. 2001, 65, 1302–1309. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.-P.; He, C.-H. Simultaneous quantification of three major bioactive triterpene acids in the leaves of Diospyros kaki by high-performance liquid chromatography method. J. Pharm. Biomed. Anal. 2006, 41, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Zhao, M.; Li, H. Method development and validation for simultaneous determination of six flavonoids in rat eyes after oral administration of Diospyros kaki leaves extract by UPLC-MS/MS. Chem. Pharm. Bull. 2020, 69, 218–221. [Google Scholar] [CrossRef] [PubMed]

| Number of Carbon | 1 | 3 | ||
|---|---|---|---|---|
| δH Multi (J in Hz) | δC | δH Multi (J in Hz) | δC | |
| 2 | 158.1 | 158.6 | ||
| 134.9 | 134.8 | |||
| 4 | 179.2 | 179.0 | ||
| 5 | 163.1 | 163.0 | ||
| 6 | 6.16 d (1.5) | 101.2 | 6.11 d (2.0) | 100.6 |
| 7 | 167.7 | 168.3 | ||
| 8 | 6.34 s | 95.1 | 6.29 d (2.0) | 95.2 |
| 9 | 158.5 | 158.1 | ||
| 10 | 105.3 | 105.2 | ||
| 1′ | 122.7 | 122.8 | ||
| 2′,6′ | 8.00 d (8.5) | 132.1 | 7.98 d (9.0) | 132.1 |
| 3′,5′ | 6.87 d (8.5) | 116.3 | 6.88 d (9.0) | 116.2 |
| 4′ | 161.6 | 161.6 | ||
| 1′′ | 5.57 d (8.0) | 100.4 | 5.64 d (8.0) | 100.7 |
| 2′′ | 5.36 dd (10.0, 8.0) | 74.3 | 5.03 dd (9.0, 8.0) | 75.8 |
| 3′′ | 3.75 dd (10.5, 3.5) | 73.4 | 3.64 t (9.0) | 76.3 |
| 4′′ | 3.89 d (3.5) | 70.5 | 3.41 t (10.0) | 71.5 |
| 5′′ | 3.55 t (6.0) | 77.4 | 3.29 m | 78.8 |
| 6′′ | 3.67 m | 62.0 | 3.78 dd (12.0, 2.0) | 62.5 |
| 3.61 m | ||||
| 1′′′ | 127.2 | 127.8 | ||
| 2′′′ | 7.45 d (8.5) | 131.2 | 7.18 d (1.5) | 111.7 |
| 3′′′ | 6.81 d (8.5) | 116.8 | 149.4 | |
| 4′′′ | 161.3 | 150.7 | ||
| 5′′′ | 6.81 d (8.5) | 116.8 | 6.81 d (8.5) | 116.5 |
| 6′′′ | 7.45 d (8.5) | 131.2 | 7.07 dd (8.5, 1.5) | 124.1 |
| 7′′′ | 7.65 d (15.5) | 146.9 | 7.66 d (16.0) | 147.2 |
| 8′′′ | 6.35 d (16.0) | 115.2 | 6.37 d (16.0) | 115.5 |
| 9′′′ | 168.7 | 168.4 | ||
| 3′′′-OCH3 | 3.91 s | 56.4 | ||
| No | Content (%) | Linear Range (μg/mL) | Regression Equation | r2 | Precision (% RSD) | Accuracy | ||
|---|---|---|---|---|---|---|---|---|
| Intraday | Interday | Intraday | Interday | |||||
| 4 | 0.33 | 31.25–500 | y = 0.0014x + 0.0045 | 0.999 | 0.9 ± 0.2 | 0.9 ± 0.4 | 101.2 ± 0.3 | 102.3 ± 0.8 |
| 5 | 0.26 | 31.25–500 | y = 0.0117x + 0.1248 | 0.999 | 1.1 ± 0.5 | 1.1 ± 0.7 | 101.3 ± 1.2 | 100.5 ± 1.5 |
| 6 | 0.65 | 31.25–500 | y = 0.0118x + 0.1177 | 0.999 | 1.4 ± 0.7 | 1.1 ± 0.8 | 100.0 ± 2.5 | 99.4 ± 1.6 |
| 7 | 0.16 | 31.25–500 | y = 0.0066x + 0.0548 | 0.998 | 1.9 ± 0.2 | 1.5 ± 0.7 | 101.2 ± 2.7 | 101.9 ± 0.2 |
| 8 | 0.48 | 31.25–500 | y = 0.0113x + 0.0362 | 0.999 | 2.0 ± 1.5 | 1.3 ± 0.6 | 99.5 ± 1.4 | 101.3 ± 3.4 |
| 11 | 0.31 | 31.25–500 | y = 0.0070x + 0.0433 | 0.998 | 1.1 ± 0.5 | 1.8 ± 0.8 | 100.3 ± 1.8 | 100.7 ± 1.0 |
| 13 | 0.58 | 31.25–500 | y = 0.0097x + 0.1832 | 0.999 | 1.6 ± 0.3 | 1.3 ± 0.6 | 99.7 ± 1.8 | 100.2 ± 1.3 |
| 15 | 0.01 | 6.25–100 | y = 0.0128x + 0.0076 | 0.999 | 1.6 ± 0.9 | 1.3 ± 0.8 | 102.9 ± 1.0 | 102.0 ± 1.0 |
| 16 | 0.38 | 6.25–100 | y = 0.0126x – 0.0045 | 0.999 | 1.0 ± 0.8 | 1.7 ± 0.2 | 102.2 ± 0.8 | 101.8 ± 0.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, J.; Park, J.-E.; Lee, J.-S.; Lee, J.-H.; Hwang, H.; Jung, S.-H.; Kwon, H.-C.; Jang, D.-S. Chemical Constituents of the Leaves of Diospyros kaki (Persimmon). Plants 2021, 10, 2032. https://doi.org/10.3390/plants10102032
Kwon J, Park J-E, Lee J-S, Lee J-H, Hwang H, Jung S-H, Kwon H-C, Jang D-S. Chemical Constituents of the Leaves of Diospyros kaki (Persimmon). Plants. 2021; 10(10):2032. https://doi.org/10.3390/plants10102032
Chicago/Turabian StyleKwon, Jaeyoung, Jeong-Eun Park, Jin-Su Lee, Jung-Hwan Lee, Hoseong Hwang, Sang-Hoon Jung, Hak-Cheol Kwon, and Dae-Sik Jang. 2021. "Chemical Constituents of the Leaves of Diospyros kaki (Persimmon)" Plants 10, no. 10: 2032. https://doi.org/10.3390/plants10102032
APA StyleKwon, J., Park, J.-E., Lee, J.-S., Lee, J.-H., Hwang, H., Jung, S.-H., Kwon, H.-C., & Jang, D.-S. (2021). Chemical Constituents of the Leaves of Diospyros kaki (Persimmon). Plants, 10(10), 2032. https://doi.org/10.3390/plants10102032






