Phenological and Geographical Effects on Phenolic and Triterpenoid Content in Vaccinium vitis-idaea L. Leaves
Abstract
:1. Introduction
2. Results and Discussion
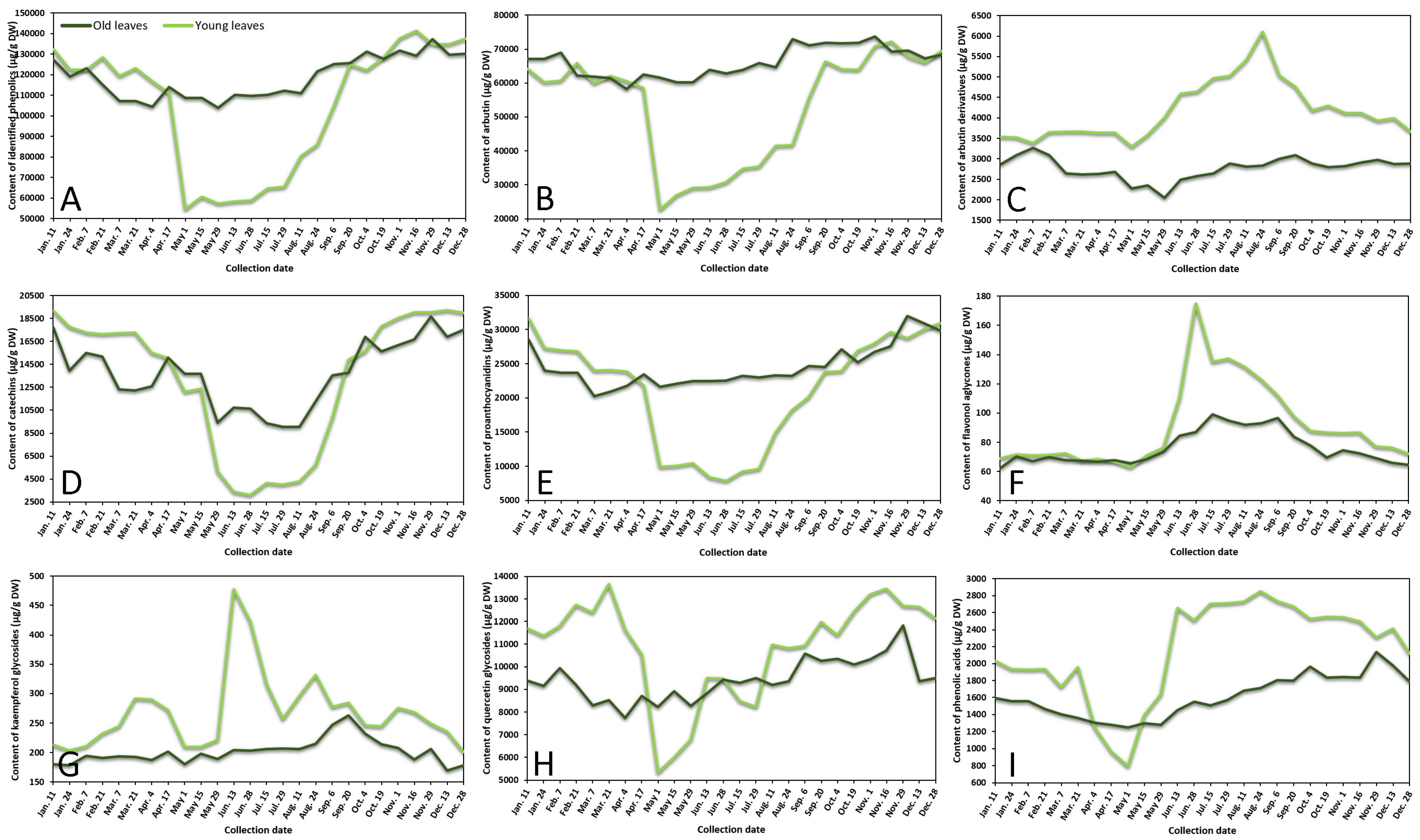
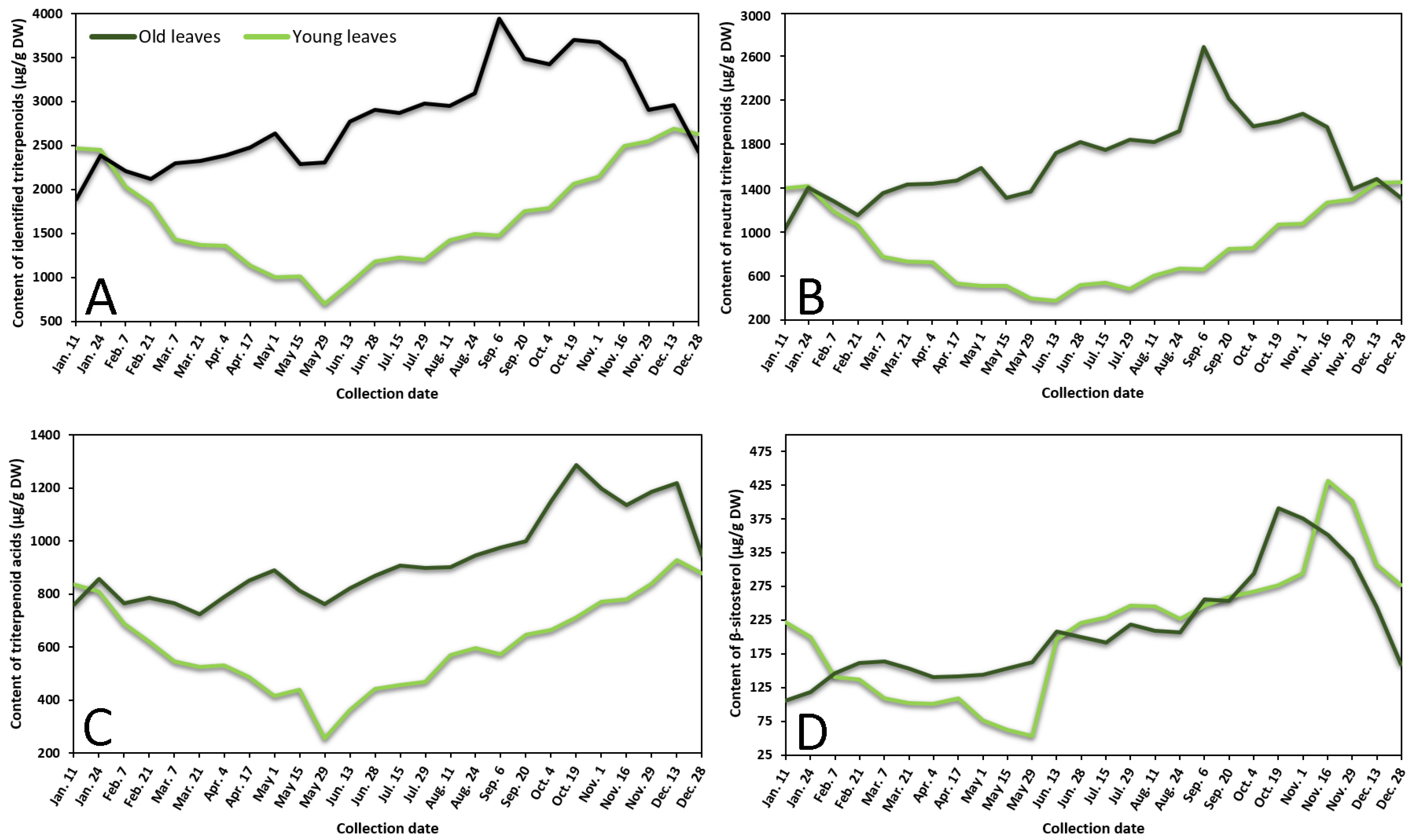
2.1. Seasonal Variation of Phenolic and Triterpenic Compounds in Lingonberry Leaves
2.1.1. Phenological Profiling by HPLC-PDA
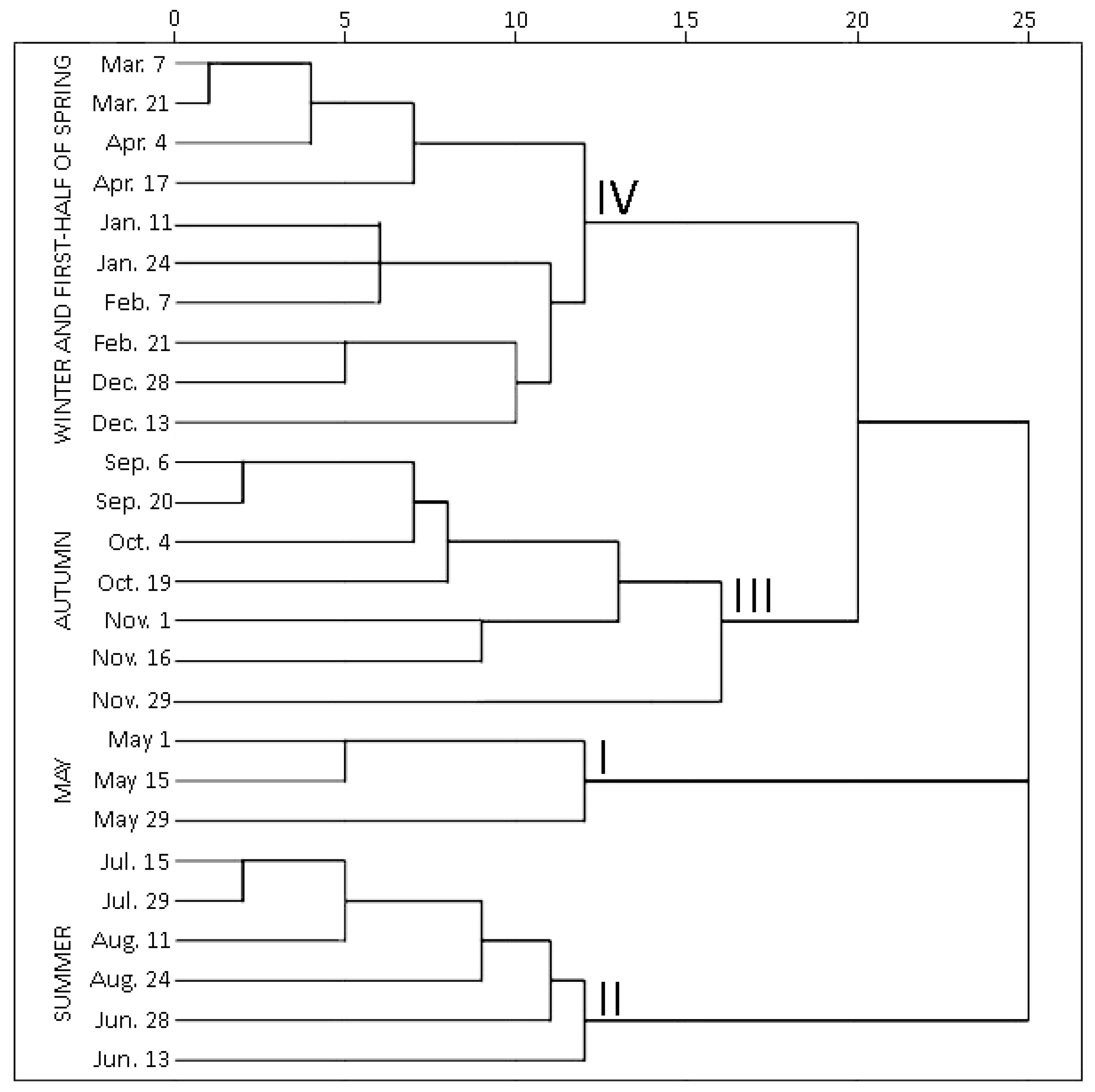
2.1.2. Hierarchical Cluster Analysis of Phenolic and Triterpenic Compounds
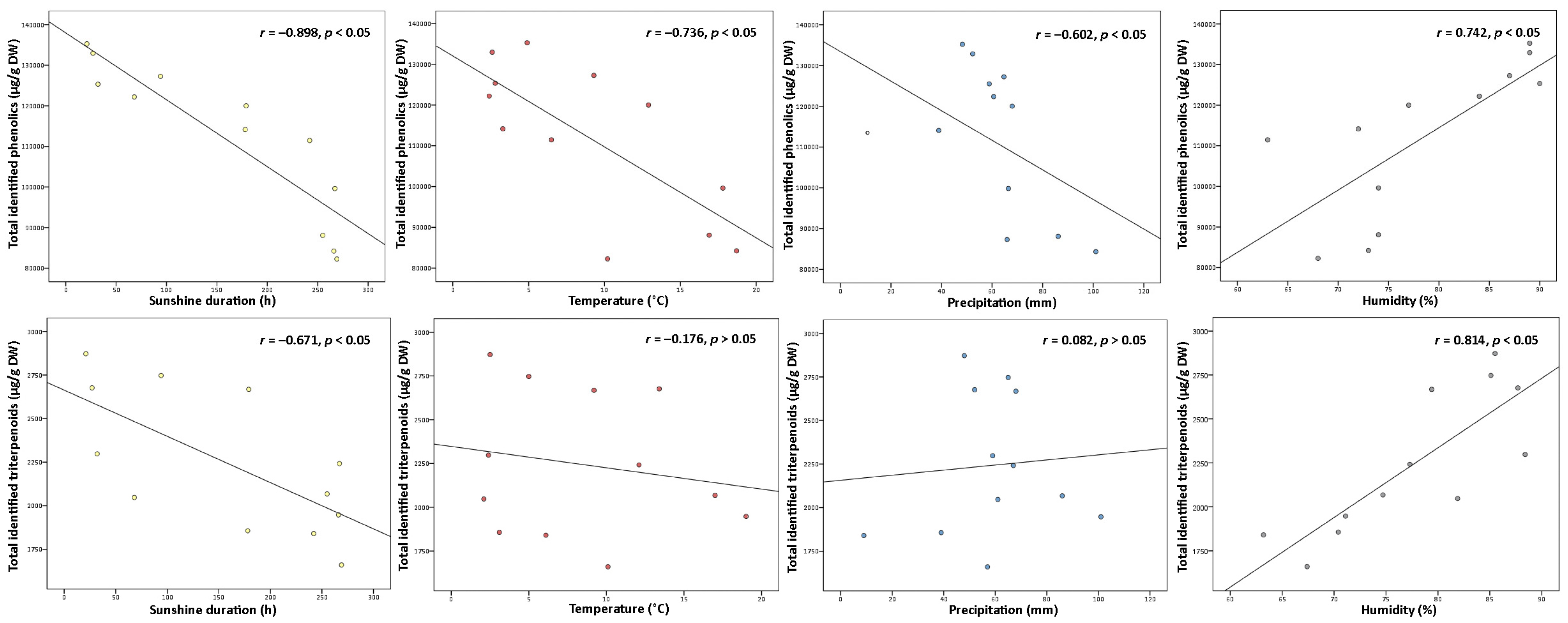
2.1.3. Environmental Factors Analysis
2.2. Geographical Variation of Phenolic and Triterpenic Compounds in Lingonberry Leaves
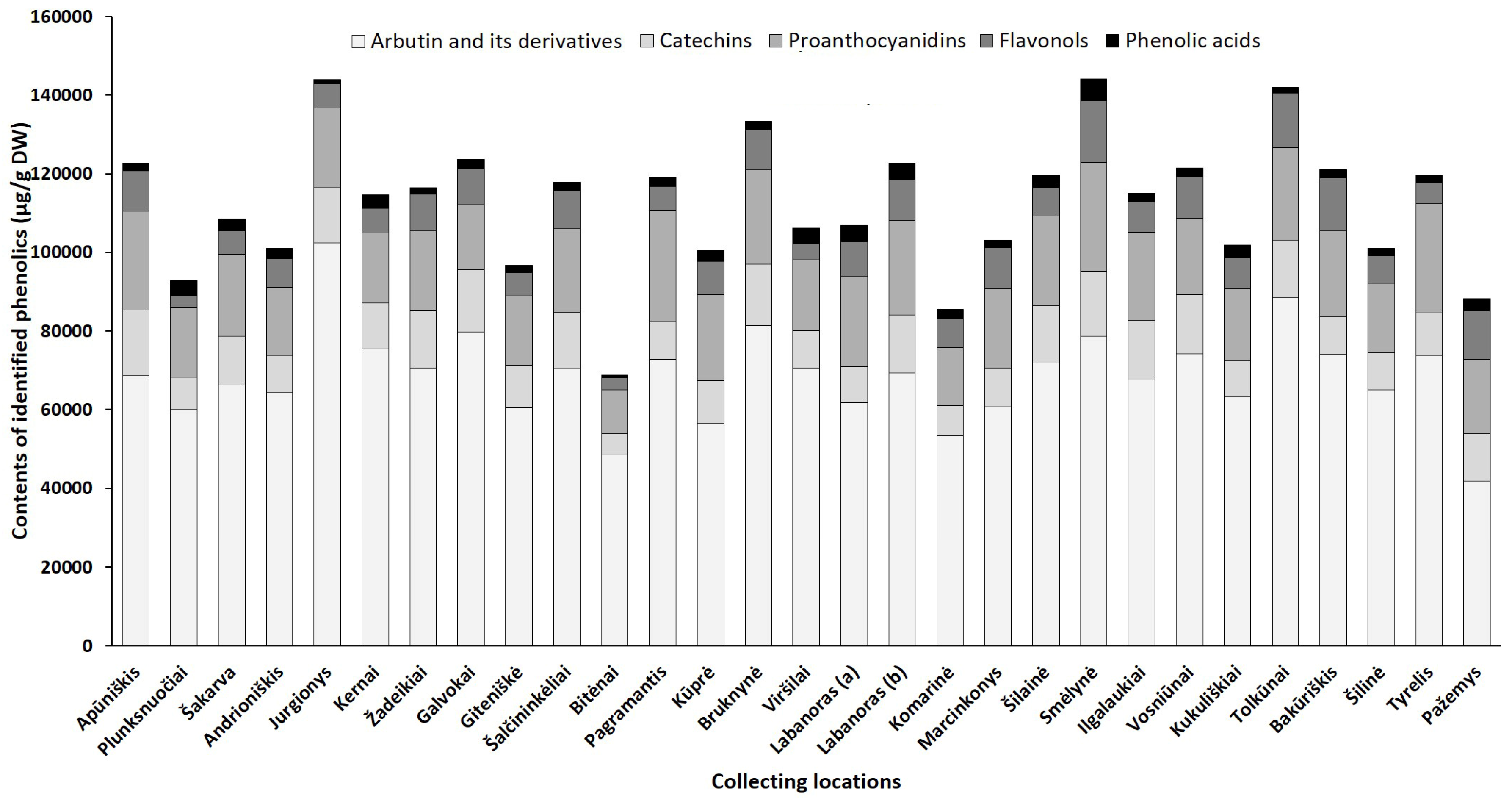
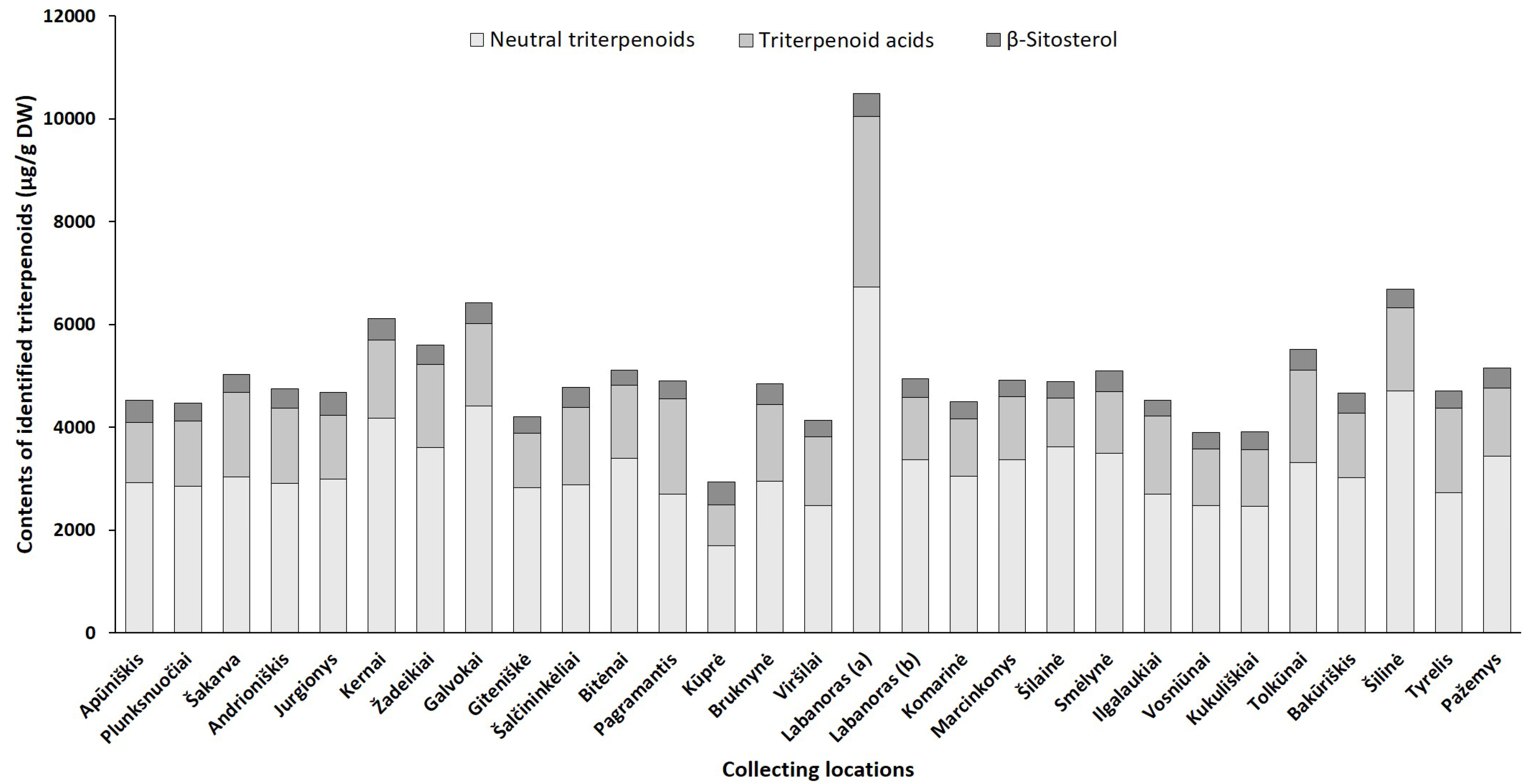
2.2.1. Phytogeographical Profiling by HPLC-PDA
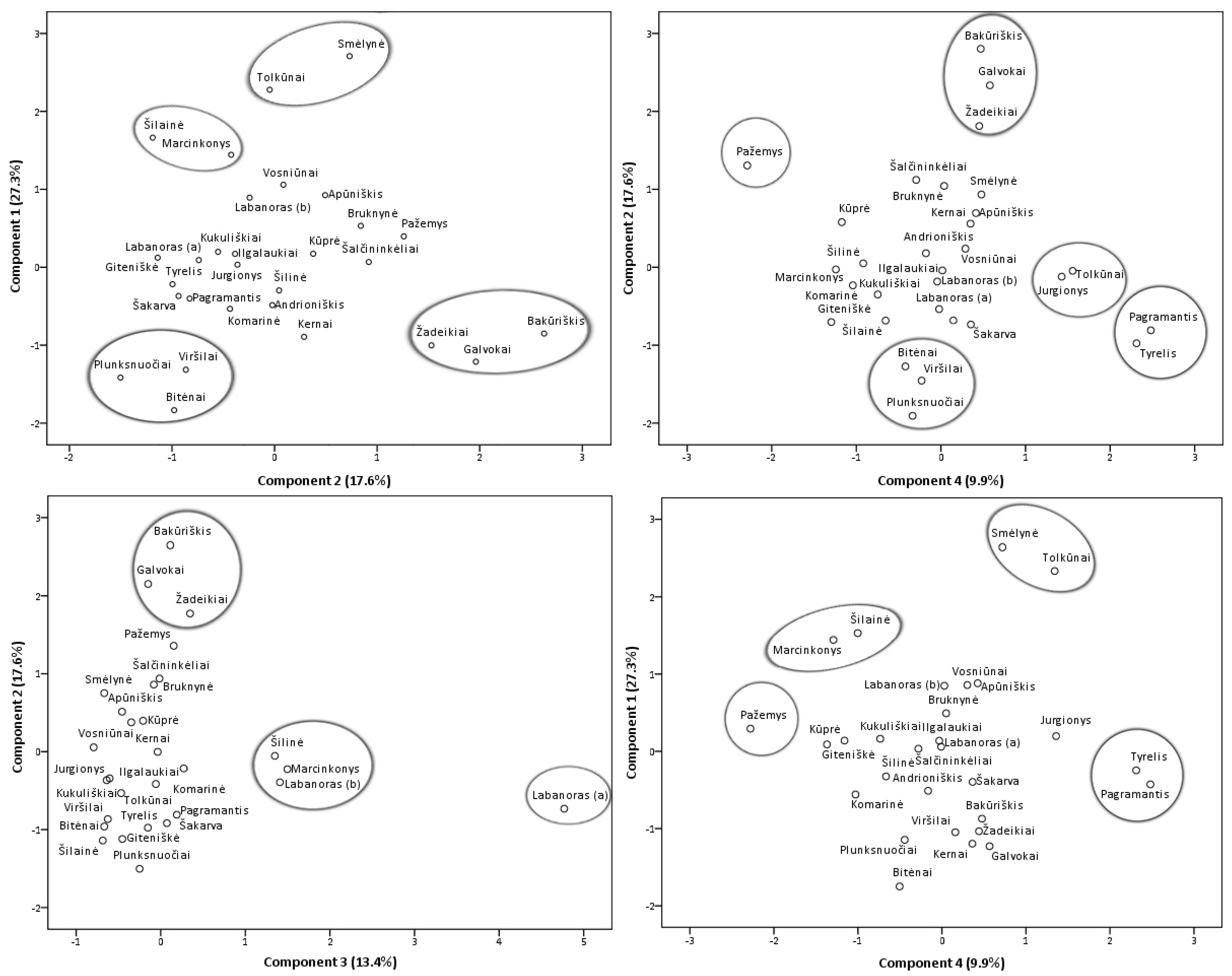
2.2.2. Principal Component Analysis of Phenolic and Triterpenic Compounds
2.2.3. Location and Soil Effects Analysis
3. Materials and Methods
3.1. Chemicals
3.2. Plant Material and Growing Conditions
3.3. Preparation of Extracts
3.4. HPLC-PDA Conditions
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
References
- Debnath, S.C.; Arigundam, U. In vitro propagation strategies of medicinally important berry crop, lingonberry (Vaccinium vitis-idaea L.). Agronomy 2020, 10, 744. [Google Scholar] [CrossRef]
- Karlsons, A.; Tomsone, S.; Lazdāne, M.; Osvalde, A. Effect of fertilization on growth of lingonberry (Vaccinium vitis-idaea L.). Agron. Res. 2021, 19, 1039–1051. [Google Scholar] [CrossRef]
- Ross, K.A.; Godfrey, D.; Fukumoto, L. The chemical composition, antioxidant activity and α-glucosidase inhibitory activity of water-extractable polysaccharide conjugates from northern manitoba lingonberry. Cogent Food Agric. 2015, 1, 1109781. [Google Scholar] [CrossRef]
- Ștefănescu, B.E.; Szabo, K.; Mocan, A.; Crişan, G. Phenolic compounds from five Ericaceae species leaves and their related bioavailability and health benefits. Molecules 2019, 24, 2046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kowalska, K.; Dembczyński, R.; Gołąbek, A.; Olkowicz, M.; Olejnik, A. ROS modulating effects of lingonberry (Vaccinium vitis-idaea L.) polyphenols on obese adipocyte hypertrophy and vascular endothelial dysfunction. Nutrients 2021, 13, 885. [Google Scholar] [CrossRef]
- Ferlemi, A.-V.; Lamari, F.N. Berry leaves: An alternative source of bioactive natural products of nutritional and medicinal value. Antioxidants 2016, 5, 17. [Google Scholar] [CrossRef]
- Páscoa, R.N.M.J.; Gomes, M.J.; Sousa, C. Antioxidant activity of blueberry (Vaccinium spp.) cultivar leaves: Differences across the vegetative stage and the application of near infrared spectroscopy. Molecules 2019, 24, 3900. [Google Scholar] [CrossRef] [Green Version]
- Vilkickyte, G.; Raudone, L.; Petrikaite, V. Phenolic fractions from Vaccinium vitis-idaea L. and their antioxidant and anticancer activities assessment. Antioxidants 2020, 9, 1261. [Google Scholar] [CrossRef]
- Alam, Z.; Roncal, J.; Peña-Castillo, L. Genetic variation associated with healthy traits and environmental conditions in Vaccinium vitis-idaea L. BMC Genom. 2018, 19, 4. [Google Scholar] [CrossRef] [Green Version]
- Bujor, O.-C.; Tanase, C.; Popa, M.E. Phenolic antioxidants in aerial parts of wild Vaccinium species: Towards pharmaceutical and biological properties. Antioxidants 2019, 8, 649. [Google Scholar] [CrossRef] [Green Version]
- Ștefănescu, B.-E.; Călinoiu, L.F.; Ranga, F.; Fetea, F.; Mocan, A.; Vodnar, D.C.; Crișan, G. Chemical composition and biological activities of the Nord-West Romanian wild bilberry (Vaccinium myrtillus L.) and lingonberry (Vaccinium vitis-idaea L.) leaves. Antioxidants 2020, 9, 495. [Google Scholar] [CrossRef]
- Szakiel, A.; Pączkowski, C.; Koivuniemi, H.; Huttunen, S. Comparison of the triterpenoid content of berries and leaves of lingonberry Vaccinium vitis-idaea L. from Finland and Poland. J. Agric. Food Chem. 2012, 60, 4994–5002. [Google Scholar] [CrossRef]
- Tian, Y.; Liimatainen, J.; Alanne, A.-L.; Lindstedt, A.; Liu, P.; Sinkkonen, J.; Kallio, H.; Yang, B. Phenolic compounds extracted by acidic aqueous ethanol from berries and leaves of different berry plants. Food Chem. 2017, 220, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Isah, T. Stress and defense responses in plant secondary metabolites production. Biol. Res. 2019, 52, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vrancheva, R.; Ivanov, I.; Dincheva, I.; Badjakov, I.; Pavlov, A. Triterpenoids and other non-polar compounds in leaves of wild and cultivated Vaccinium species. Plants 2021, 10, 94. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhu, Z.; Guo, Q.; Zhang, L.; Zhang, X. Variation in concentrations of major bioactive compounds in Prunella vulgaris L. related to plant parts and phenological stages. Biol. Res. 2012, 45, 171–175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radusiene, J.; Karpaviciene, B.; Stanius, Ž. Effect of external and internal factors on secondary metabolites accumulation in St. John’s Worth. Bot. Lith. 2012, 18, 101–108. [Google Scholar] [CrossRef]
- Li, Y.; Kong, D.; Fu, Y.; Sussman, M.R.; Wu, H. The effect of developmental and environmental factors on secondary metabolites in medicinal plants. Plant Physiol. Biochem. 2020, 148, 80–89. [Google Scholar] [CrossRef]
- Bergman, M.E.; Davis, B.; Phillips, M.A. medically useful plant terpenoids: Biosynthesis, occurrence, and mechanism of action. Molecules 2019, 24, 3961. [Google Scholar] [CrossRef] [Green Version]
- Obata, T. Metabolons in plant primary and secondary metabolism. Phytochem. Rev. 2019, 18, 1483–1507. [Google Scholar] [CrossRef]
- Verma, N.; Shukla, S. Impact of various factors responsible for fluctuation in plant secondary metabolites. J. Appl. Res. Med. Aromat. Plants 2015, 2, 105–113. [Google Scholar] [CrossRef]
- Alqahtani, A.; Tongkao-on, W.; Li, K.M.; Razmovski-Naumovski, V.; Chan, K.; Li, G.Q. Seasonal variation of triterpenes and phenolic compounds in Australian Centella asiatica (L.) Urb. Phytochem. Anal. PCA 2015, 26, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Bandzaitiene, Z.; Daubaras, R.; Labokas, J. Brukne: Vaccinium vitis-idaea L.; Botanikos Instituto Leidykla: Vilnius, Lithuania, 2007; pp. 5–36. [Google Scholar]
- Bujor, O.-C.; Ginies, C.; Popa, V.I.; Dufour, C. Phenolic compounds and antioxidant activity of lingonberry (Vaccinium vitis-idaea L.) leaf, stem and fruit at different harvest periods. Food Chem. 2018, 252, 356–365. [Google Scholar] [CrossRef]
- Bujor, O.-C.; Le Bourvellec, C.; Volf, I.; Popa, V.I.; Dufour, C. Seasonal variations of the phenolic constituents in bilberry (Vaccinium myrtillus L.) leaves, stems and fruits, and their antioxidant activity. Food Chem. 2016, 213, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Sommavilla, V.; Haidacher-Gasser, D.; Sgarbossa, M.; Zidorn, C. Seasonal variation in phenolics in leaves of Celtis australis (Cannabaceae). Biochem. Syst. Ecol. 2012, 41, 110–114. [Google Scholar] [CrossRef]
- Solanki, T.; Aphalo, P.J.; Neimane, S.; Hartikainen, S.M.; Pieristè, M.; Shapiguzov, A.; Porcar-Castell, A.; Atherton, J.; Heikkilä, A.; Robson, T.M. UV-screening and springtime recovery of photosynthetic capacity in leaves of Vaccinium vitis-idaea above and below the snow pack. Plant Physiol. Biochem. 2019, 134, 40–52. [Google Scholar] [CrossRef]
- Rahajanirina, V.; Faramalala, M.; Edmond, R.; Zebrowski, C.; Leong, J.; Tsy, J.-M.; Danthu, P. Effects of harvest frequency on leaf biomass and triterpenoid content of Centella asiatica (L.) Urb from Madagascar. J. Med. Biol. Res. 2016, 2, 1–8. [Google Scholar]
- Dashbaldan, S.; Becker, R.; Pączkowski, C.; Szakiel, A. Various patterns of composition and accumulation of steroids and triterpenoids in cuticular waxes from screened Ericaceae and Caprifoliaceae berries during fruit development. Molecules 2019, 24, 3826. [Google Scholar] [CrossRef] [Green Version]
- Olofsson, J.; Ericson, L.; Torp, M.; Stark, S.; Baxter, R. Carbon balance of Arctic tundra under increased snow cover mediated by a plant pathogen. Nat. Clim. Chang. 2011, 1, 220–223. [Google Scholar] [CrossRef]
- Shitan, N. Secondary metabolites in plants: Transport and self-tolerance mechanisms. Biosci. Biotechnol. Biochem. 2016, 80, 1283–1293. [Google Scholar] [CrossRef] [Green Version]
- Murthy, H.N.; Lee, E.-J.; Paek, K.-Y. Production of secondary metabolites from cell and organ cultures: Strategies and approaches for biomass improvement and metabolite accumulation. Plant Cell Tissue Organ Cult. PCTOC 2014, 118, 1–16. [Google Scholar] [CrossRef]
- Ribeiro, D.A.; Camilo, C.J.; de Fátima Alves Nonato, C.; Rodrigues, F.F.G.; Menezes, I.R.A.; Ribeiro-Filho, J.; Xiao, J.; de Almeida Souza, M.M.; da Costa, J.G.M. Influence of seasonal variation on phenolic content and in vitro antioxidant activity of Secondatia floribunda A. DC. (Apocynaceae). Food Chem. 2020, 315, 126277. [Google Scholar] [CrossRef]
- He, M.; He, C.-Q.; Ding, N.-Z. Abiotic stresses: General defenses of land plants and chances for engineering multistress tolerance. Front. Plant Sci. 2018, 9, 1771. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bertolucci, S.K.V.; Pereira, A.B.D.; Pinto, J.E.B.P.; Oliveira, A.B.; Braga, F.C. Seasonal variation on the contents of coumarin and kaurane-type diterpenes in Mikania laevigata and M. glomerata leaves under different shade levels. Chem. Biodivers. 2013, 10, 288–295. [Google Scholar] [CrossRef]
- Taylor, A.O. Some Effects of Photoperiod on the biosynthesis of phenylpropane derivatives in Xanthium. Plant Physiol. 1965, 40, 273–280. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Drappier, J.; Hilbert, G.; Guillaumie, S.; Dai, Z.; Geny-Denis, L.; Delrot, S.; Darriet, P.; Thibon, C.; Pieri, P. The effects of a moderate grape temperature increase on berry secondary metabolites. OENO One 2019, 53, 321–333. [Google Scholar] [CrossRef]
- Vallat, A.; Gu, H.; Dorn, S. How rainfall, relative humidity and temperature influence volatile emissions from apple trees in situ. Phytochemistry 2005, 66, 1540–1550. [Google Scholar] [CrossRef]
- Yang, L.; Wen, K.-S.; Ruan, X.; Zhao, Y.-X.; Wei, F.; Wang, Q. Response of plant secondary metabolites to environmental factors. Molecules 2018, 23, 762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirakosyan, A.; Kaufman, P.; Warber, S.; Zick, S.; Aaronson, K.; Bolling, S.; Chul Chang, S. Applied environmental stresses to enhance the levels of polyphenolics in leaves of hawthorn plants. Physiol. Plant. 2004, 121, 182–186. [Google Scholar] [CrossRef]
- Oksanen, E.; Lihavainen, J.; Keinänen, M.; Keski-Saari, S.; Kontunen-Soppela, S.; Sellin, A.; Sõber, A. Northern forest trees under increasing atmospheric humidity. In Progress in Botany; Cánovas, F.M., Lüttge, U., Matyssek, R., Pretzsch, H., Eds.; Springer International Publishing: Cham, Switzerland, 2019; Volume 80, pp. 317–336. [Google Scholar] [CrossRef]
- Migas, P.; Krauze-Baranowska, M. The significance of arbutin and its derivatives in therapy and cosmetics. Phytochem. Lett. 2015, 13, 35–40. [Google Scholar] [CrossRef]
- Scarano, A.; Chieppa, M.; Santino, A. Looking at flavonoid biodiversity in horticultural crops: A colored mine with nutritional benefits. Plants 2018, 7, 98. [Google Scholar] [CrossRef] [Green Version]
- Okoye, N.N.; Ajaghaku, D.L.; Okeke, H.N.; Ilodigwe, E.E.; Nworu, C.S.; Okoye, F.B.C. beta-Amyrin and alpha-amyrin acetate isolated from the stem bark of Alstonia boonei display profound anti-inflammatory activity. Pharm. Biol. 2014, 52, 1478–1486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, H.S.; Han, J.Y.; Choi, Y.E. Identification of triterpenes and functional characterization of oxidosqualene cyclases involved in triterpene biosynthesis in lettuce (Lactuca sativa). Plant Sci. 2020, 301, 110656. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Shang, X.; Ding, H.; Cao, Y.; Fang, S. Natural variations in flavonoids and triterpenoids of Cyclocarya paliurus leaves. J. For. Res. 2020, 32, 805–814. [Google Scholar] [CrossRef]
- Iwanycki Ahlstrand, N.; Havskov Reghev, N.; Markussen, B.; Bruun Hansen, H.C.; Eiriksson, F.F.; Thorsteinsdóttir, M.; Rønsted, N.; Barnes, C.J. Untargeted metabolic profiling reveals geography as the strongest predictor of metabolic phenotypes of a cosmopolitan weed. Ecol. Evol. 2018, 8, 6812–6826. [Google Scholar] [CrossRef] [PubMed]
- Vilkickyte, G.; Motiekaityte, V.; Vainoriene, R.; Liaudanskas, M.; Raudone, L. Development, validation, and application of UPLC-PDA method for anthocyanins profiling in Vaccinium L. berries. J. Berry Res. 2021, in press. [Google Scholar] [CrossRef]
- Guo, L.; Wang, S.; Zhang, J.; Yang, G.; Zhao, M.; Ma, W.; Zhang, X.; Li, X.; Han, B.; Chen, N.; et al. Effects of ecological factors on secondary metabolites and inorganic elements of Scutellaria baicalensis and analysis of geoherblism. Sci. China Life Sci. 2013, 56, 1047–1056. [Google Scholar] [CrossRef] [Green Version]
- Ullah, N.; Khurram, M.; Amin, M.; Khan, T.; Khayyam, S.; Farhat, A.; Khan, F.; Najeeb, U.; Ullah, S. Impact of geographical locations on Mentha spicata antibacterial activities. J. Med. Plants Res. 2012, 6, 1201–1206. [Google Scholar] [CrossRef]
- Vyas, P.; Curran, N.; Igamberdiev, A.; Debnath, S. Antioxidant properties of lingonberry (Vaccinium vitis-idaea L.) leaves within a set of wild clones and cultivars. Can. J. Plant Sci. 2015, 95, 663–669. [Google Scholar] [CrossRef] [Green Version]
- Hawkesford, M.; Horst, W.; Kichey, T.; Lambers, H.; Schjoerring, J.; Møller, I.S.; White, P. Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Academic Press: Cambridge, MA, USA, 2012; pp. 135–189. [Google Scholar] [CrossRef]
- Baba, T.; Hirose, D.; Sasaki, N.; Watanabe, N.; Kobayashi, N.; Kurashige, Y.; Karimi, F.; Ban, T. Mycorrhizal formation and diversity of endophytic fungi in hair roots of Vaccinium oldhamii Miq. in Japan. Microbes Environ. 2016, 31, 186–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paal, T. Lingonberry (Vaccinium vitis-idaea L.) research in Estonia: An overview. Acta Hortic. 2006, 715, 203–217. [Google Scholar] [CrossRef]
- Šimala, D. Some experiments on the ecological cultivation of the lingonberry (Vaccinium vitis-idaea L.) in a mountainous region of Slovakia. J. Fruit Ornam. Plant Res. 2004, 12, 167–174. [Google Scholar]
- Machado, R.M.A.; Serralheiro, R.P. Soil salinity: Effect on vegetable crop growth. management practices to prevent and mitigate soil salinization. Horticulturae 2017, 3, 30. [Google Scholar] [CrossRef]
- Shrivastava, P.; Kumar, R. Soil Salinity: A serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J. Biol. Sci. 2015, 22, 123–131. [Google Scholar] [CrossRef] [Green Version]
- Vilkickyte, G.; Raudone, L. Optimization, validation and application of HPLC-PDA methods for quantification of triterpenoids in Vaccinium vitis-idaea L. Molecules 2021, 26, 1645. [Google Scholar] [CrossRef] [PubMed]
| Forest | Location Parameters | Soil Parameters | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Latitude, ° | Longitude, ° | Altitude, m | pHKCl | N, mg/L | P, mg/L | K, mg/L | Ca, mg/L | Mg, mg/L | Cl, mg/L | EC, mS/cm | |
| Žadeikiai | 56.01 | 24.47 | 52 | 3.0 | 6 | 2 | 36 | 12 | 5 | 18 | 0.17 |
| Vosniūnai | 55.90 | 24.69 | 77 | 3.1 | 27 | 7 | 65 | 41 | 14 | 18 | 0.67 |
| Galvokai | 56.06 | 24.89 | 80 | 3.1 | 40 | 3 | 29 | 19 | 7 | 23 | 0.38 |
| Viršilai | 56.09 | 25.31 | 102 | 3.1 | 28 | 7 | 50 | 55 | 8 | 18 | 0.64 |
| Plunksnuočiai | 56.08 | 25.49 | 107 | 3.4 | 16 | 2 | 32 | 15 | 5 | 18 | 0.36 |
| Ilgalaukiai | 55.99 | 25.43 | 107 | 2.9 | 30 | 8 | 51 | 57 | 8 | 19 | 0.47 |
| Apūniškis | 56.01 | 25.52 | 110 | 4.2 | 116 | 6 | 48 | 60 | 23 | 18 | 1.10 |
| Bakūriškis | 56.06 | 25.68 | 123 | 2.9 | 39 | 10 | 66 | 61 | 15 | 23 | 0.83 |
| Andrioniškis | 55.60 | 25.04 | 111 | 3.6 | 50 | 10 | 91 | 24 | 6 | 18 | 0.56 |
| Pažemys | 55.66 | 26.00 | 171 | 3.5 | 114 | 3 | 87 | 23 | 9 | 23 | 0.94 |
| Giteniškė | 55.60 | 26.13 | 173 | 4.1 | 7 | 0.2 | 20 | 9 | 4 | 18 | 0.19 |
| Smėlynė | 55.40 | 26.15 | 187 | 3.9 | 60 | 6 | 64 | 26 | 5 | 15 | 0.14 |
| Šakarva | 55.31 | 26.06 | 149 | 3.6 | 6 | 2 | 31 | 10 | 4 | 23 | 1.68 |
| Labanoras (a,b) | 55.16 | 25.81 | 165 | 3.3 | 12 | 0.9 | 20 | 12 | 4 | 18 | 0.27 |
| Šalčininkėliai | 54.38 | 25.38 | 171 | 3.1 | 73 | 17 | 101 | 24 | 9 | 20 | 0.90 |
| Bruknynė | 54.35 | 25.39 | 189 | 3.2 | 80 | 17 | 113 | 26 | 11 | 20 | 0.95 |
| Jurgionys | 54.46 | 24.50 | 139 | 3.3 | 89 | 28 | 116 | 25 | 18 | 22 | 1.11 |
| Tolkūnai | 54.27 | 24.41 | 145 | 3.8 | 70 | 18 | 88 | 31 | 12 | 19 | 0.52 |
| Marcinkonys | 54.07 | 24.44 | 137 | 3.2 | 9 | 1 | 26 | 12 | 4 | 12 | 0.21 |
| Šilainė | 54.08 | 23.71 | 134 | 2.9 | 11 | 2 | 30 | 17 | 6 | 23 | 0.29 |
| Bitėnai | 55.06 | 22.05 | 22 | 3.2 | 27 | 4 | 36 | 13 | 6 | 17 | 0.99 |
| Šilinė | 55.19 | 22.31 | 39 | 3.3 | 57 | 12 | 45 | 13 | 6 | 18 | 0.25 |
| Komarinė | 55.19 | 22.45 | 47 | 2.9 | 48 | 6 | 56 | 22 | 8 | 18 | 0.62 |
| Pagramantis | 55.39 | 22.22 | 89 | 3.2 | 105 | 12 | 76 | 32 | 15 | 20 | 0.85 |
| Tyrelis | 55.32 | 22.17 | 59 | 3.2 | 166 | 10 | 96 | 49 | 16 | 20 | 0.97 |
| Kūprė | 55.58 | 22.82 | 145 | 3.8 | 8 | 0.4 | 15 | 9 | 4 | 23 | 0.22 |
| Kukuliškiai | 55.78 | 21.09 | 34 | 4.8 | 52 | 7 | 61 | 10 | 13 | 20 | 1.70 |
| Kernai | 56.23 | 21.49 | 44 | 4.0 | 185 | 13 | 113 | 76 | 27 | 23 | 1.13 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilkickyte, G.; Raudone, L. Phenological and Geographical Effects on Phenolic and Triterpenoid Content in Vaccinium vitis-idaea L. Leaves. Plants 2021, 10, 1986. https://doi.org/10.3390/plants10101986
Vilkickyte G, Raudone L. Phenological and Geographical Effects on Phenolic and Triterpenoid Content in Vaccinium vitis-idaea L. Leaves. Plants. 2021; 10(10):1986. https://doi.org/10.3390/plants10101986
Chicago/Turabian StyleVilkickyte, Gabriele, and Lina Raudone. 2021. "Phenological and Geographical Effects on Phenolic and Triterpenoid Content in Vaccinium vitis-idaea L. Leaves" Plants 10, no. 10: 1986. https://doi.org/10.3390/plants10101986
APA StyleVilkickyte, G., & Raudone, L. (2021). Phenological and Geographical Effects on Phenolic and Triterpenoid Content in Vaccinium vitis-idaea L. Leaves. Plants, 10(10), 1986. https://doi.org/10.3390/plants10101986