The Past, Present and Future of Cannabis sativa Tissue Culture
Abstract
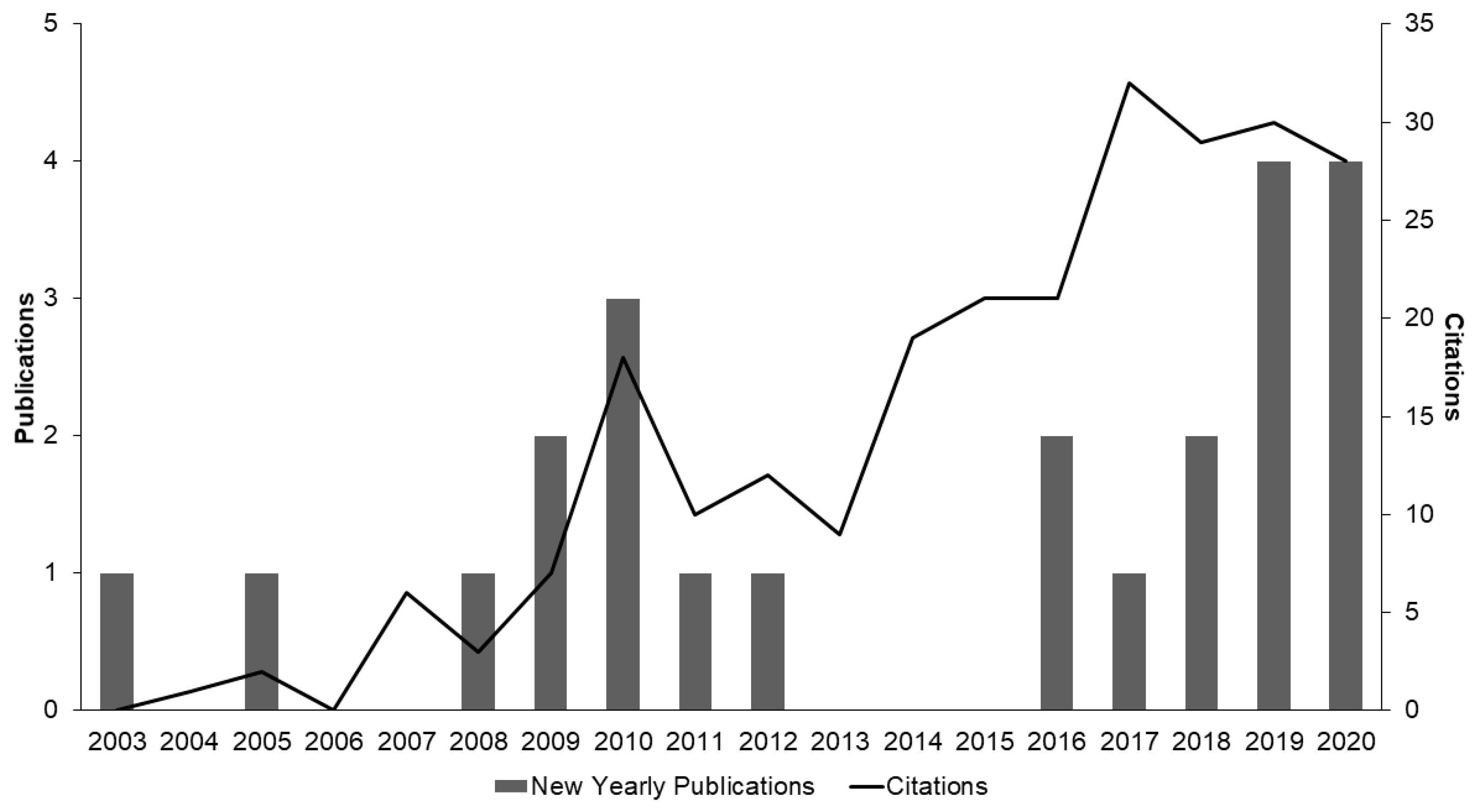
1. Introduction
2. Brief History of C. sativa in North America
3. Botany and Taxonomy of C. sativa
4. Current Production Practices
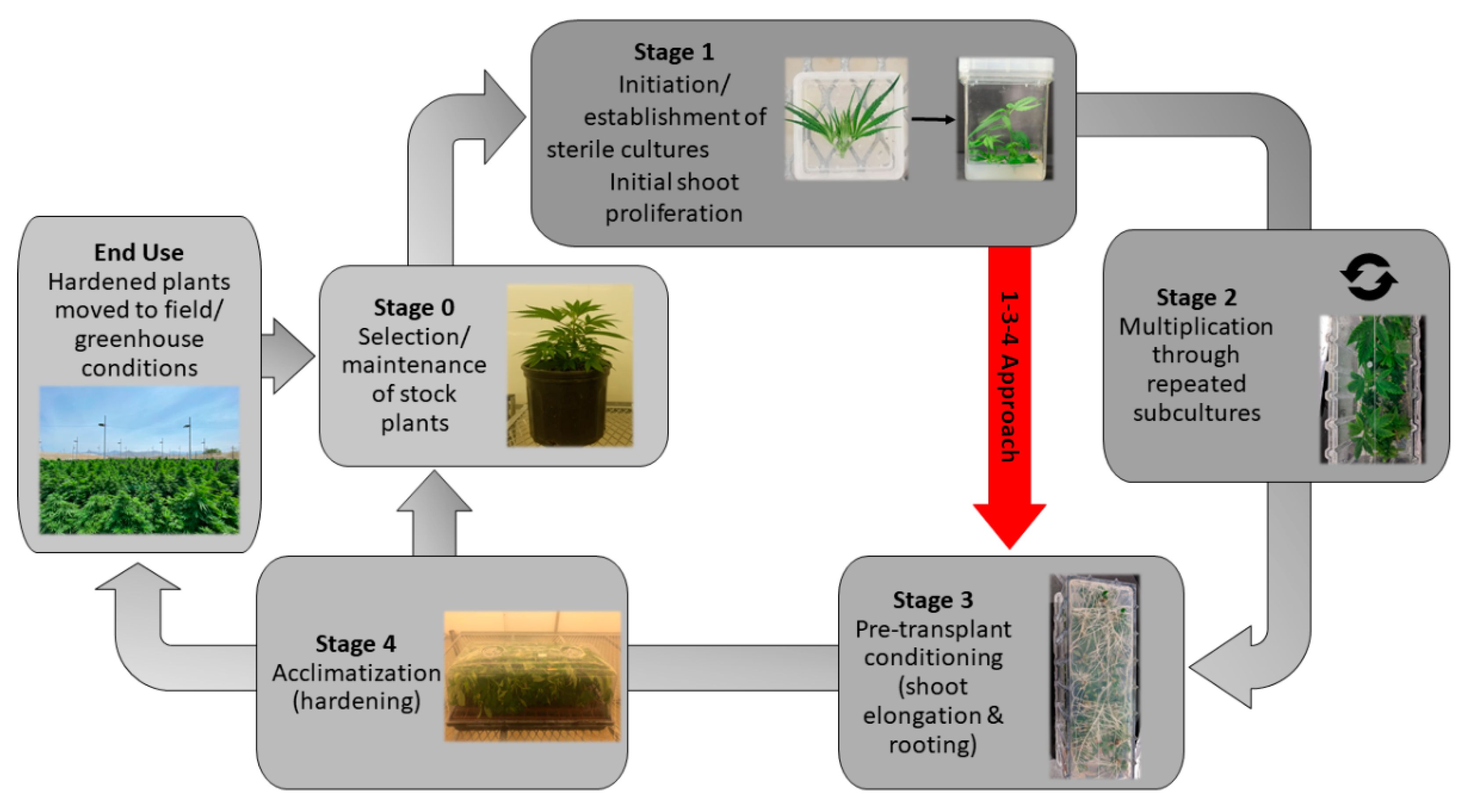
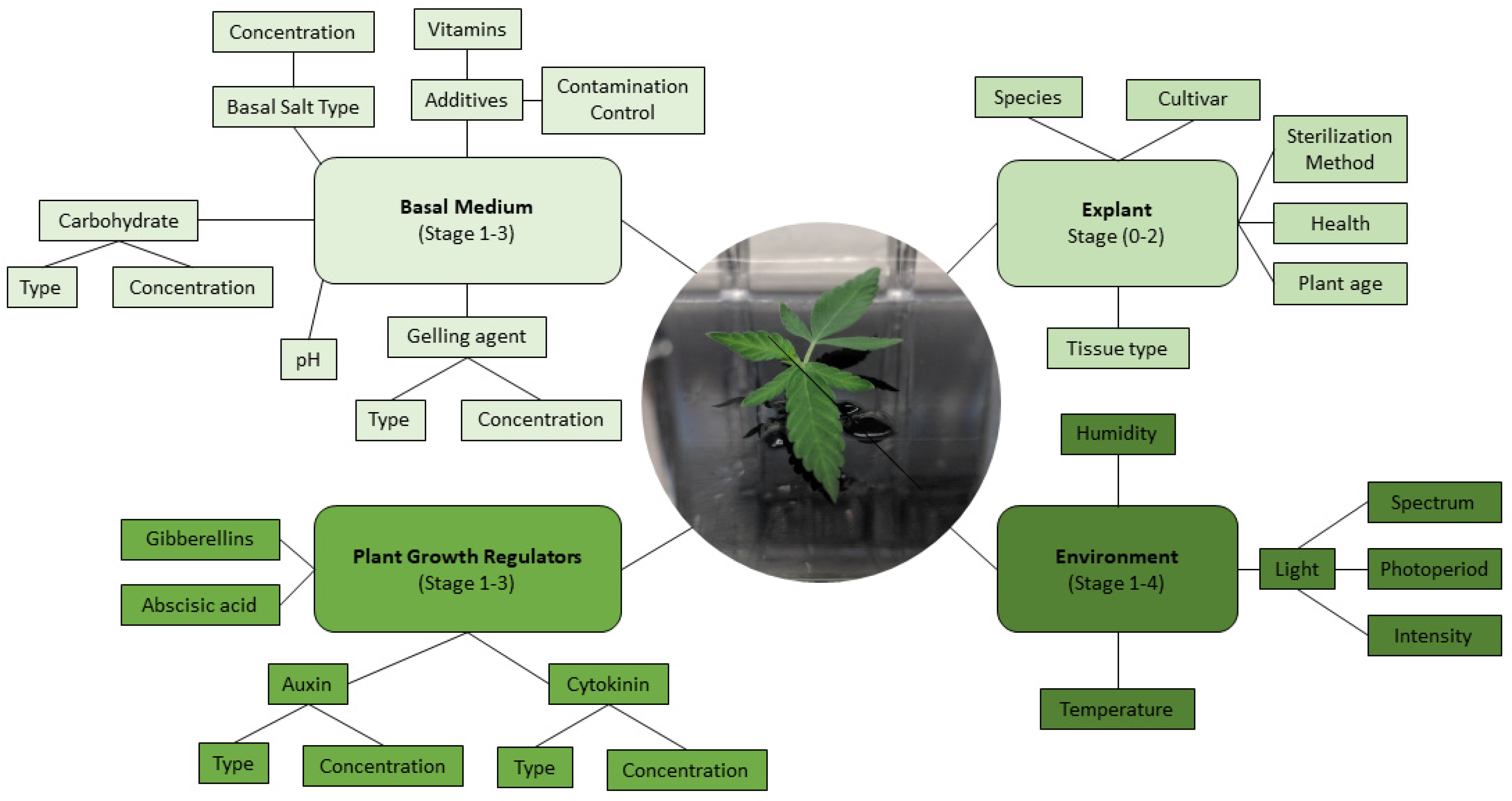
5. Micropropagation of C. sativa
Floral Reversion: An Alternate Micropropagation Approach
6. Regeneration in C. sativa
6.1. Incomplete and Ambiguously Reported Results
6.2. Genotype and Tissue Specificity
6.3. Recalcitrance to Regeneration
6.4. Lack of Reproducibility
7. Genetic Stability and Preservation
8. Future Directions
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wills, S. Cannabis use and abuse by man: An historical perspective. C. In Cannabis: The genus Cannabis; Harwood Academic: Amsterdam, Netherlands, 1998; pp. 1–27. [Google Scholar]
- Hesami, M.; Pepe, M.; Alizadeh, M.; Rakei, A.; Baiton, A.; Phineas Jones, A.M. Recent advances in cannabis biotechnology. Ind. Crops Prod. 2020, 158, 1–20. [Google Scholar] [CrossRef]
- Lata, H.; Chandra, S.; Khan, I.A.; Elsohly, M.A.; Lata, H.; Chandra, Á.S.; Khan, I.A.; Elsohly, Á.M.A.; Elsohly, M.A. Micropropagation of Cannabis sativa L.-An Update. In Cannabis Sativa L.-Botany and Biotechnology; Chandra, S., Lata, H., ElSohly, M.A., Eds.; Springer: Cham, Switzerland, 2017; pp. 285–297. [Google Scholar]
- League of Nations Convention on Psychotropic Substances. In Proceedings of the Second Opium Conference, Geneva, Switzerland, 17 June 1925; p. 52.
- Erickson, P.G.; Oscapella, E. Cannabis in Canada–A Puzzling Policy. Int. J. Drug Policy 1999, 10, 313–318. [Google Scholar] [CrossRef]
- Fischer, B.; Ala-Leppilampi, K.; Single, E.; Robins, A. Cannabis law reform in canada: Is the “saga of promise, hesitation and retreat” coming to an end?! Can. J. Criminol. Crim. Justice 2003, 45, 265–297. [Google Scholar] [CrossRef]
- Baker, D.; Pryce, G.; Giovannoni, G.; Thompson, A.J. The therapeutic potential of cannabis. Lancet Neurol. 2003, 2, 291–298. [Google Scholar] [CrossRef]
- Bridgeman, M.B.; Abazia, D.T. Medicinal cannabis: History, pharmacology, and implications for the acute care setting. Pharm. Ther. 2017, 42, 180–188. [Google Scholar]
- Cherney, J.H.; Small, E. Industrial hemp in North America: Production, politics and potential. Agronomy 2016, 6, 58. [Google Scholar] [CrossRef]
- Eddy, M. Medical Marijuana: Review and Analysis of Federal and State Policies; United States Congressional Research Service: Washington, DC, USA, 2010. [Google Scholar]
- Government of Canada. Industrial Hemp Regulations Règlement sur le Chanvre Industriel; Ministry of Justice: Ottawa, ON, Canada, 2015. [Google Scholar]
- Schwabe, A.L.; Hansen, C.J.; Hyslop, R.M.; McGlaughlin, M.E. Research grade marijuana supplied by the National Institute on Drug Abuse is genetically divergent from commercially available Cannabis. BioRxiv 2019, 7, 31–38. [Google Scholar]
- Blake, D.R.; Robson, P.; Ho, M.; Jubb, R.W.; McCabe, C.S. Preliminary assessment of the efficacy, tolerability and safety of a cannabis-based medicine (Sativex) in the treatment of pain caused by rheumatoid arthritis. Rheumatology 2006, 45, 50–52. [Google Scholar] [CrossRef]
- Nurmikko, T.J.; Serpell, M.G.; Hoggart, B.; Toomey, P.J.; Morlion, B.J.; Haines, D. Sativex successfully treats neuropathic pain characterised by allodynia: A randomised, double-blind, placebo-controlled clinical trial. Pain 2007, 133, 210–220. [Google Scholar] [CrossRef]
- Perucca, E. Cannabinoids in the treatment of epilepsy: Hard evidence at last? J. Epilepsy Res. 2017, 7, 61–76. [Google Scholar] [CrossRef]
- Barchel, D.; Stolar, O.; De-Haan, T.; Ziv-Baran, T.; Saban, N.; Fuchs, D.O.; Koren, G.; Berkovitch, M. Oral cannabidiol use in children with autism spectrum disorder to treat related symptoms and Co-morbidities. Front. Pharmacol. 2019, 9, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Bahji, A.; Meyyappan, A.C.; Hawken, E.R. Efficacy and acceptability of cannabinoids for anxiety disorders in adults: A systematic review & meta-analysis. J. Psychiatr. Res. 2020, 129, 257–264. [Google Scholar] [PubMed]
- Mark, T.; Shepherd, J.; Olson, D.; Snell, W.; Proper, S.; Thornsbury, S. Economic Viability of Industrial Hemp in the United States: A Review of State Pilot Programs; U.S. Department of Agriculture, Economic Research Service: Washington, DC, USA, 2020. [Google Scholar]
- Government of Canada. Understanding the New Access to Cannabis for Medical Purposes Regulations. Available online: https://www.canada.ca/en/health-canada/services/publications/drugs-health-products/understanding-new-access-to-cannabis-for-medical-purposes-regulations.html (accessed on 25 September 2020).
- Kwai, I.U.N. Reclassifies cannabis as a less dangerous drug. New York Times, 2 December 2020; pp. 1–3. [Google Scholar]
- Chopra, I.; Chopra, N. The Use of the Cannabis Drugs in India; United Nations Office on Drugs and Crime: Vienna, Austria, 1957. [Google Scholar]
- Spitzer-Rimon, B.; Duchin, S.; Bernstein, N.; Kamenetsky, R. Architecture and florogenesis in female Cannabis sativa plants. Front. Plant Sci. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Reed, J. Morphology of Cannabis Sativa L.; State University of Iowa: Iowa City, IA, USA, 1914. [Google Scholar]
- Yang, M.Q.; van Velzen, R.V.; Bakker, F.T.; Sattarian, A.; Li, D.Z.; Yi, T.S. Molecular Phylogenetics and character evolution of Cannabaceae. Taxon 2013, 62, 473–485. [Google Scholar] [CrossRef]
- Kovalchuk, I.; Pellino, M.; Rigault, P.; Van Velzen, R.; Ebersbach, J.; Ashnest, J.R.; Mau, M.; Schranz, M.E.; Alcorn, J.; Laprairie, R.B.; et al. The genomics of Cannabis and its close relatives. Annu. Rev. Plant Biol. 2020, 71, 713–739. [Google Scholar] [CrossRef]
- Clarke, R. Botany of the genus cannabis. In Advances in Hemp Research; Ranalli, P., Ed.; Food Product Press: New York, NY, USA, 1999; pp. 1–19. [Google Scholar]
- Schilling, S.; Dowling, C.A.; Shi, J.; Ryan, L.; Hunt, D.; O’Reilly, E.; Perry, A.S.; Kinnane, O.; McCabe, P.F.; Melzer, R. The cream of the crop: Biology, breeding and applications of Cannabis sativa. Authorea 2020, 1–46. [Google Scholar] [CrossRef]
- Farag, S. Cannabinoids Production in Cannabis Sativa L.: An In Vitro Approach. Ph.D. Dissertation, Technischen Universität Dortmund, Dortmund, Germany, 2014. [Google Scholar]
- Moher, M.; Jones, M.; Zheng, Y. Photoperiodic response of In Vitro Cannabis sativa Plants. Preprints 2020. [Google Scholar] [CrossRef]
- Barcaccia, G.; Palumbo, F.; Scariolo, F.; Vannozzi, A.; Borin, M.; Bona, S. Potentials and challenges of genomics for breeding cannabis cultivars. Front. Plant Sci. 2020, 11, 1–19. [Google Scholar] [CrossRef]
- Romero, P.; Peris, A.; Vergara, K.; Matus, J.T. Comprehending and improving cannabis specialized metabolism in the systems biology era. Plant Sci. 2020, 298, 110571. [Google Scholar] [CrossRef]
- Small, E. Evolution and classification of Cannabis sativa (Marijuana, Hemp) in relation to human utilization. Bot. Rev. 2015, 81, 189–294. [Google Scholar] [CrossRef]
- Schwabe, A.L.; McGlaughlin, M.E. Genetic tools weed out misconceptions of strain reliability in Cannabis sativa: Implications for a budding industry. J. Cannabis Res. 2019, 1, 1–16. [Google Scholar] [CrossRef]
- McPartland, J.M. Cannabis systematics at the levels of family, genus, and species. Cannabis Cannabinoid Res. 2018, 3, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Sawler, J.; Stout, J.M.; Gardner, K.M.; Hudson, D.; Vidmar, J.; Butler, L.; Page, J.E.; Myles, S. The genetic structure of marijuana and hemp. PLoS ONE 2015, 10, e0133292. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Lata, H.; Elsohly, M.A. Propagation of Cannabis for clinical research: An approach towards a modern herbal medicinal products development. Front. Plant Sci. 2020, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Fetterman, P.S.; Keith, E.S.; Waller, C.W.; Guerrero, O.; Doorenbos, N.J.; Quimby, M.W. Mississippi-grown Cannabis sativa L.: Preliminary observation on chemical definition of phenotype and variations in tetrahydrocannabinol content versus age, sex, and plant part. J. Pharm. Sci. 1971, 60, 1246–1249. [Google Scholar] [CrossRef] [PubMed]
- Mudge, E.M.; Murch, S.J.; Brown, P.N. Chemometric analysis of cannabinoids: Chemotaxonomy and domestication syndrome. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Hazekamp, A.; Fischedick, J.T. Cannabis—From cultivar to chemovar. Drug Test. Anal. 2012, 4, 660–667. [Google Scholar] [CrossRef]
- Mudge, E.M.; Brown, P.N.; Murch, S.J. The terroir of Cannabis: Terpene metabolomics as a tool to understand Cannabis sativa selections. Planta Med. 2019, 85, 781–796. [Google Scholar] [CrossRef]
- De Meijer, E.P.M.; Bagatta, M.; Carboni, A.; Crucitti, P.; Moliterni, V.M.C.; Ranalli, P.; Mandolino, G. The inheritance of chemical phenotype in Cannabis sativa L. Genetics 2003, 163, 335–346. [Google Scholar]
- Van Bakel, H.; Stout, J.M.; Cote, A.G.; Tallon, C.M.; Sharpe, A.G.; Hughes, T.R.; Page, J.E. The draft genome and transcriptome of Cannabis sativa. Genome Biol. 2011, 12, 1–17. [Google Scholar] [CrossRef]
- Hillig, K.W. A chemotaxonomic analysis of terpenoid variation in Cannabis. Biochem. Syst. Ecol. 2004, 32, 875–891. [Google Scholar] [CrossRef]
- Hillig, K.W. Genetic evidence for speciation in Cannabis (Cannabaceae). Genet. Resour. Crop Evol. 2005, 52, 161–180. [Google Scholar] [CrossRef]
- Clarke, R.C.; Merlin, M.D. Classical and molecular taxonomy. In Cannabis: Evolution and Ethanobotany; University of California Press: Berkeley, CA, USA, 2013; pp. 311–331. [Google Scholar]
- McPartland, J.M.; Small, E. A classification of endangered high-THC cannabis (Cannabis sativa subsp. indica) domesticates and their wild relatives. PhytoKeys 2020, 144, 81–112. [Google Scholar] [CrossRef]
- Small, E.; Cronquist, A. A practical and natural taxonomy for Cannabis. Taxon 1976, 25, 405–435. [Google Scholar] [CrossRef]
- Small, E. Morphological variation of achenes of Cannabis. Can. J. Bot. 1975, 53, 978–987. [Google Scholar] [CrossRef]
- Small, E. American law and the species problem in Cannabis: Science and semantics. Bull. Narc. 1975, 27, 1–20. [Google Scholar]
- Potter, D.J.; Duncombe, P. The effect of electrical lighting power and irradiance on indoor-grown cannabis potency and yield. J. Forensic Sci. 2012, 57, 618–622. [Google Scholar] [CrossRef]
- Caplan, D.; Dixon, M.; Zheng, Y. Optimal rate of organic fertilizer during the flowering stage for cannabis grown in two coir-based substrates. HortScience 2017, 52, 1796–1803. [Google Scholar] [CrossRef]
- Caplan, D.; Stemeroff, J.; Dixon, M.; Zheng, Y. Vegetative propagation of cannabis by stem cuttings: Effects of leaf number, cutting position, rooting hormone and leaf tip removal. Can. J. Plant Sci. 2018, 98, 1126–1132. [Google Scholar] [CrossRef]
- Mills, E. The carbon footprint of indoor Cannabis production. Energy Policy 2012, 46, 58–67. [Google Scholar] [CrossRef]
- Vera, C.L.; Hanks, A. Hemp production in Western Canada. J. Ind. Hemp 2004, 9, 79–86. [Google Scholar] [CrossRef]
- Campbell, L.G.; Naraine, S.G.U.; Dusfresne, J. Phenotypic plasticity influences the success of clonal propagation in industrial pharmaceutical Cannabis sativa. PLoS ONE 2019, 14, e0213434. [Google Scholar] [CrossRef] [PubMed]
- Lata, H.; Chandra, S.; Techen, N.; Khan, I.A.; Elsohly, M.A. Assessment of the genetic stability of micropropagated plants of Cannabis sativa by ISSR markers. Planta Med. 2010, 76, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Fike, J.H.; Darby, H.; Johnson, B.L.; Smart, L.; Williams, D.W. Industrial Hemp in the USA: A Brief Synopsis Bt- Sustainable Agriculture Reviews 42: Hemp Production and Applications; Crini, G., Lichtfouse, E., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 89–109. [Google Scholar]
- Shukla, M.R.; Singh, A.S.; Piunno, K.; Saxena, P.K.; Jones, A.M.P. Application of 3D printing to prototype and develop novel plant tissue culture systems. Plant Methods 2017, 13, 1–10. [Google Scholar] [CrossRef]
- Kotkas, K.; Rosenberg, V. Disease eradication and propagation of the initial seed potato material in Estonia. Potato Res. 1999, 42, 577–583. [Google Scholar] [CrossRef]
- Mori, K. Production of virus-free plants by means of meristem culture. Japan Agric. Res. Q. 1971, 6, 1–7. [Google Scholar]
- Reuther, G. Propagation of disease-free Pelagonium cultivars by tissue culture. Acta Hortic. 1983, 311–320. [Google Scholar] [CrossRef]
- Punyarani, K.; Devala Devi, K.; Henary Singh, C.; Samarjit Singh, N.; Homen Singh, H.; Dikash Singh, T.; Moirangthem, S.; Sunitibala Devi, H. In Vitro production of genetically stable and virus free plantlets of Musa sp. var. Meitei Hei using male inflorescence as explant. Sci. Hortic. 2013, 164, 440–447. [Google Scholar] [CrossRef]
- Murashige, T. Plant propagtion through tissue cultures. Ann. Rev. Plant Physio 1974, 25, 135–136. [Google Scholar] [CrossRef]
- George, E.F.; Hall, M.A.; De Klerk, G.J.; Debergh, P.C. Micropropagation: Uses and methods. In Plant Propagation by Tissue Culture, 3rd ed.; Springer: Amsterdam, The Netherlands, 2008; Volume 1, pp. 29–64. [Google Scholar]
- International Atomic Energy Agency Low cost options for tissue culture technology in developing countries. In Proceedings of the Technical Meeting organized by the Joint FAO/IAEA Division of Nuclear Techniques in Food and Agriculture, Vienna, Austria, 26–30 August 2002.
- Richez-Dumanois, C.; Braut-Boucher, F.; Cosson, L.; Paris, M. Multiplication végétative In Vitro du chanvre (Cannabis sativa L.). Application à la conservation des clones sélectionnés. Agronomie 1986, 6, 487–495. [Google Scholar] [CrossRef]
- Plawuszewski, M.; Lassociński, W.; Wielgus, K. Regeneration of Polish cultivars of monoecious hemp (Cannabis sativa L.) grown In Vitro. In Renewable Resources and Plant Biotechnology; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2006; pp. 149–154. [Google Scholar]
- Wielgus, K.; Luwanska, A.; Lassocinski, W.; Kaczmarek, Z. Estimation of Cannabis sativa L. tissue culture conditions essential for callus induction and plant regeneration. J. Nat. Fibers 2008, 5, 199–207. [Google Scholar] [CrossRef]
- Chandra, S.; Lata, H.; Mehmedic, Z.; Khan, I.; ElSohly, M. Assessment of cannabinoids content in micropropagated plants of Cannabis sativa and their comparison with conventionally propagated plants and mother plant during developmental stages of growth. Planta Med. 2010, 76, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Lata, H.; Chandra, S.; Khan, I.; ElSohly, M.A. Thidiazuron-induced high-frequency direct shoot organogenesis of Cannabis sativa L. Vitr. Cell. Dev. Biol. Plant 2009, 45, 12–19. [Google Scholar] [CrossRef]
- Lata, H.; Chandra, S.; Khan, I.A.; Elsohly, M.A. Propagation through alginate encapsulation of axillary buds of Cannabis sativa L.—An important medicinal plant. Physiol. Mol. Biol. Plants 2009, 15, 79–86. [Google Scholar] [CrossRef]
- Lata, H.; Chandra, S.; Techen, N.; Khan, I.A.; ElSohly, M.A. In Vitro mass propagation of Cannabis sativa L.: A protocol refinement using novel aromatic cytokinin meta-topolin and the assessment of eco-physiological, biochemical and genetic fidelity of micropropagated plants. J. Appl. Res. Med. Aromat. Plants 2016, 3, 18–26. [Google Scholar] [CrossRef]
- Grulichova, M.; Mendel, P.; Lalge, A.B.; Slamova, N.; Trojan, V.; Vyhnanek, T.; Winkler, J.; Vaverkova, M.D.; Adamcova, D.; Dorbevic, B. Effects of different morphoregulators on growth and development of Cannabis sativa L. In Proceedings of the 24th International Phd Students Conference (Mendelnet 2017), Brno, Czech Republic, 8–9 November 2017; pp. 618–623. [Google Scholar]
- Piunno, K.; Golenia, G.; Boudko, E.A.; Downey, C.; Jones, A.M.P. Regeneration of shoots from immature and mature inflorescences of Cannabis sativa. Can. J. Plant Sci. 2019, 99, 556–559. [Google Scholar] [CrossRef]
- Smýkalová, I.; Vrbová, M.; Cvečková, M.; Plačková, L.; Žukauskaitė, A.; Zatloukal, M.; Hrdlička, J.; Plíhalová, L.; Doležal, K.; Griga, M. The effects of novel synthetic cytokinin derivatives and endogenous cytokinins on the In Vitro growth responses of hemp (Cannabis sativa L.) explants. Plant Cell Tissue Organ Cult. 2019, 139, 381–394. [Google Scholar] [CrossRef]
- Monthony, A.S.; Bagheri, S.; Zheng, Y.; Jones, A.M.P. Flower power: Floral reversion as a viable alternative to nodal micropropagation in Cannabis sativa. BioRxiv 2020. [Google Scholar] [CrossRef]
- Page, S.R.G.; Monthony, A.S.; Jones, A.M.P. Basal media optimization for the micropropagation and callogenesis of Cannabis sativa L. BioRxiv 2020, 1–23. [Google Scholar] [CrossRef]
- Wróbel, T.; Dreger, M.; Wielgus, K.; Słomski, R. Modified nodal cuttings and shoot tips protocol for rapid regeneration of Cannabis sativa L. J. Nat. Fibers 2020, 1–10. [Google Scholar] [CrossRef]
- Codesido, V.; Meyer, S.; Casano, S. Influence of media composition and genotype for successful Cannabis sativa L. In Vitro introduction. Acta Hortic. 2020, 1285, 75–80. [Google Scholar] [CrossRef]
- Mestinšek Mubi, Š.; Svetik, S.; Flajšman, M.; Murovec, J. In Vitro tissue culture and genetic analysis of two high-CBD medical Cannabis (Cannabis sativa L.) breeding lines. Genetika 2020, 52, 925–941. [Google Scholar] [CrossRef]
- Mandolino, G.; Ranalli, P. Advances in biotechnological approaches for hemp breeding and industry. In Advances in Hemp Research; CRC Press, Inc.: Cleveland, OH, USA, 1999; pp. 185–212. [Google Scholar]
- Ślusarkiewicz-Jarzina, A.; Ponitka, A.; Kaczmarek, Z. Influence of cultivar, explant source and plant growth regulator on callus induction and plant regeneration of Cannabis sativa L. Acta Biol. Crac. Ser. Bot. 2005, 47, 145–151. [Google Scholar]
- Raharjo, T.J.; Eucharia, O.; Chang, W.-T.; Verpoorte, R. Callus induction and phytochemical characterization of Cannabis sativa cell suspension cultures. Indones. J. Chem. 2006, 6, 70–74. [Google Scholar] [CrossRef]
- Flores-Sanchez, I.J.; Peč, J.; Fei, J.; Choi, Y.H.; Dušek, J.; Verpoorte, R. Elicitation studies in cell suspension cultures of Cannabis sativa L. J. Biotechnol. 2009, 143, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Lata, H.; Chandra, S.; Khan, I.A.; Elsohly, M.A. High frequency plant regeneration from leaf derived callus of high δ 9-tetrahydrocannabinol yielding Cannabis sativa L. Planta Med. 2010, 76, 1629–1633. [Google Scholar] [CrossRef]
- Movahedi, M.; Ghasemi-Omran, V.-O.; Torabi, S. The effect of different concentrations of TDZ and BA on In Vitro regeneration of Iranian cannabis (Cannabis sativa) using cotyledon and epicotyl explants. J. Plant Mol. Breed. 2015, 3, 20–27. [Google Scholar]
- Chaohua, C.; Gonggu, Z.; Lining, Z.; Chunsheng, G.; Qing, T.; Jianhua, C.; Xinbo, G.; Dingxiang, P.; Jianguang, S. A rapid shoot regeneration protocol from the cotyledons of hemp (Cannabis sativa L.). Ind. Crops Prod. 2016, 83, 61–65. [Google Scholar] [CrossRef]
- Galán-Ávila, A.; García-Fortea, E.; Prohens, J.; Herraiz, F.J. Development of a direct In Vitro plant regeneration protocol from Cannabis sativa L. Seedling explants: Developmental morphology of shoot regeneration and ploidy level of regenerated plants. Front. Plant Sci. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Monthony, A.S.; Kyne, S.T.; Grainger, C.M.; Jones, A.M.P. Recalcitrance of Cannabis sativa to de novo regeneration; a multi-genotype replication study. BioRxiv 2020. [Google Scholar] [CrossRef]
- Vardja, R.; Vardja, T. The effect of cytokinin type and concentration and the number of subcultures on the multiplication rate of some decorative plants/Tsutokiniini tuubi ja kontsentratsiooni ning umberistutuste arvu mon monede dekoratiivtaimede paljunemiskoefitsiendile. Proc. Est. Acad. Sci. Biol. 2001, 50, 22–32. [Google Scholar]
- Kumar, V.; Parvatam, G.; Ravishankar, G.A. AgNO3—A potential regulator of ethylene activity and plant growth modulator. Electron. J. Biotechnol. 2009, 12, 1–15. [Google Scholar] [CrossRef]
- Arigita, L.; Tamés, R.S.; González, A. 1-Methylcyclopropene and ethylene as regulators of In Vitro organogenesis in kiwi explants. Plant Growth Regul. 2003, 40, 59–64. [Google Scholar] [CrossRef]
- Galoch, E. The hormonal control of sex differentiation in dioecious plants of hemp (Cannabis sativa). Acta Soc. Bot. Pol. 1978, 47, 153–162. [Google Scholar] [CrossRef]
- Phillips, G.; Garda, M. Plant tissue culture media and practices: An overview. Vitr. Cell. Dev. Biol. Plant 2019, 55, 242–257. [Google Scholar] [CrossRef]
- Driver, J.; Kuniyuki, A. In Vitro propagation of Paradox walnut rootstock. HortScience 1984, 19, 507–509. [Google Scholar]
- Eapen, S.; George, L. Plant regeneration from peduncle segments of oil seed Brassica species: Influence of silver nitrate and silver thiosulfate. Plant Cell. Tissue Organ Cult. 1997, 51, 229–232. [Google Scholar] [CrossRef]
- Phulwaria, M.; Shekhawat, N.S. An efficient In Vitro shoot regeneration from immature inflorescence and ex vitro rooting of Arnebia hispidissima (Lehm). DC.—A red dye (Alkannin) yielding plant. Physiol. Mol. Biol. Plants 2013, 19, 435–441. [Google Scholar] [CrossRef]
- Shareefa, M.; Thomas, R.J.; Sreelekshmi, J.S.; Rajesh, M.K.; Karun, A. In Vitro regeneration of coconut plantlets from immature inflorescence. Curr. Sci. 2019, 117, 813–820. [Google Scholar] [CrossRef]
- Tooke, F.; Ordidge, M.; Chiurugwi, T.; Battey, N. Mechanisms and function of flower and inflorescence reversion. J. Exp. Bot. 2005, 56, 2587–2599. [Google Scholar] [CrossRef]
- Kavas, M.; Öktem, H.A.; Yücel, M. Factors affecting plant regeneration from immature inflorescence of two winter wheat cultivars. Biol. Plant. 2008, 52, 621–626. [Google Scholar] [CrossRef]
- Gubišová, M.; Gubiš, J.; Žofajová, A.; Mihálik, D.; Kraic, J. Enhanced In Vitro propagation of Miscanthus×giganteus. Ind. Crops Prod. 2013, 41, 279–282. [Google Scholar] [CrossRef]
- Zayed, E.M.M.; Zein El Din, A.F.M.; Manaf, H.H.; Abdelbar, O.H. Floral reversion of mature inflorescence of date palm In Vitro. Ann. Agric. Sci. 2016, 61, 125–133. [Google Scholar] [CrossRef]
- Cheng, Z.J.; Zhu, S.S.; Gao, X.Q.; Zhang, X.S. Cytokinin and auxin regulates WUS induction and inflorescence regeneration In Vitro in Arabidopsis. Plant Cell Rep. 2010, 29, 927–933. [Google Scholar] [CrossRef] [PubMed]
- Reshi, N.A.; Sudarshana, M.S.; Nandini, B.P. In Vitro propagation from inflorescence explants of Anisochilus carnosus- an ethno-medicinal herb. Int. J. Pharm. Sci. Res. 2014, 5, 2423–2427. [Google Scholar]
- Deo, P.C.; Tyagi, A.P.; Taylor, M.; Harding, R.; Becker, D. Factors affecting somatic embryogenesis and transformation in modern plant breeding. South Pac. J. Nat. Appl. Sci. 2010, 28, 27. [Google Scholar] [CrossRef]
- Florez, S.L.; Erwin, R.L.; Maximova, S.N.; Guiltinan, M.J.; Curtis, W.R. Enhanced somatic embryogenesis in Theobroma cacao using the homologous BABY BOOM transcription factor. BMC Plant Biol. 2015, 15, 1–12. [Google Scholar] [CrossRef]
- Touraev, A.; Vicente, O.; Heberle-Bors, E. Initiation of microspore embryogenesis by stress. Trends Plant Sci. 1997, 2, 297–302. [Google Scholar] [CrossRef]
- Schachtsiek, J.; Warzecha, H.; Kayser, O.; Stehle, F. Current perspectives on biotechnological cannabinoid production in plants. Planta Med. 2018, 84, 214–220. [Google Scholar] [CrossRef]
- Vergara, D.; Bidwell, L.C.; Gaudino, R.; Torres, A.; Du, G.; Ruthenburg, T.C.; Decesare, K.; Land, D.P.; Hutchison, K.E.; Kane, N.C. Compromised external validity: Federally produced Cannabis does not reflect legal markets. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef]
- Litz, R.E. Organogenesis and somatic embryogenesis. Acta Hortic. 1993, 199–206. [Google Scholar] [CrossRef]
- Pasqual, M.; Soares, J.D.R.; Rodrigues, F.A. Tissue Culture Applications for the Genetic Improvement of Plants; Elsevier Inc.: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Bairu, M.W.; Aremu, A.O.; Van Staden, J. Somaclonal variation in plants: Causes and detection methods. Plant Growth Regul. 2011, 63, 147–173. [Google Scholar] [CrossRef]
- Lata, H.; Chandra, S.; Techen, N.; Khan, I.A.; ElSohly, M.A. Molecular analysis of genetic fidelity in Cannabis sativa L. plants grown from synthetic (encapsulated) seeds following In Vitro storage. Biotechnol. Lett. 2011, 33, 2503–2508. [Google Scholar] [CrossRef] [PubMed]
- Niino, T.; Arizaga, M.V. Cryopreservation for preservation of potato genetic resources. Breed. Sci. 2015, 65, 41–52. [Google Scholar] [CrossRef][Green Version]
- Wilkinson, T.; Wetten, A.; Prychid, C.; Fay, M.F. Suitability of cryopreservation for the long-term storage of rare and endangered plant species: A case history for Cosmos atrosanguineus. Ann. Bot. 2003, 91, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Al-Qurainy, F.; Khan, S.; Nadeem, M.; Tarroum, M.; Alansi, S.; Al-Ameri, A.; Gaafar, A.-R.; Alshameri, A. Assessing genetic fidelity in regenerated plantlets of date palm cultivars after cryopreservation. Fresenius Environ. Bull. 2017, 26, 1727–1735. [Google Scholar]
- Engelmann, F. Use of biotechnologies for the conservation of plant biodiversity. In Vitro Cell. Dev. Biol. Plant 2011, 47, 5–16. [Google Scholar] [CrossRef]
- Uchendu, E.; Lata, H.; Chandra, S.; Khan, I.A.; ElSohly, M.A. Cryopreservation of shoot tips of elite cultivars of Cannabis sativa L. by droplet vitrification. Med. Cannabis Cannabinoids 2019, 2, 29–34. [Google Scholar] [CrossRef]
- Andre, C.M.; Hausman, J.F.; Guerriero, G. Cannabis sativa: The plant of the thousand and one molecules. Front. Plant Sci. 2016, 7, 1–17. [Google Scholar] [CrossRef]
- Kumari, A.; Baskaran, P.; Plačková, L.; Omámiková, H.; Nisler, J.; Doležal, K.; Van Staden, J. Plant growth regulator interactions in physiological processes for controlling plant regeneration and In Vitro development of Tulbaghia simmleri. J. Plant Physiol. 2018, 223, 65–71. [Google Scholar] [CrossRef]
- Kosakivska, I.V.; Vasyuk, V.A.; Voytenko, L.V.; Shcherbatiuk, M.M.; Romanenko, K.O.; Babenko, L.M. Endogenous phytohormones of fern Polystichum aculeatum (L.) roth gametophytes at different stages of morphogenesis In Vitro culture. Cytol. Genet. 2020, 54, 23–30. [Google Scholar] [CrossRef]
- Bidabadi, S.S.; Mohan Jain, S. Cellular, molecular, and physiological aspects of In Vitro plant regeneration. Plants 2020, 9, 702. [Google Scholar] [CrossRef] [PubMed]
- Lardon, R.; Geelen, D. Natural variation in plant pluripotency and regeneration. Plants 2020, 9, 1261. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.X.; Xu, Y.P.; Liu, J.Q.; Luo, X.; Zhang, S.S.; Zheng, L.W.; Guo, D.P. Cytokinins affect shoot regeneration and modulate the expression of IPT and CKX genes of In Vitro cultured Eleocharis dulcis (Brum.f.) Trin. J. Hortic. Sci. Biotechnol. 2020, 1–8. [Google Scholar] [CrossRef]
- Li, F.; Liu, S.; Zeng, M. An efficient micropropagation protocol for Monochasma savatieri Franch. ex Maxim through seed germination and direct shoot regeneration. In Vitro Cell. Dev. Biol. Plant 2020. [Google Scholar] [CrossRef]
- Liu, Y.; Lu, J.; Zhu, H.; Li, L.; Shi, Y.; Yin, X. Efficient culture protocol for plant regeneration from cotyledonary petiole explants of Jatropha curcas L. Biotechnol. Biotechnol. Equip. 2016, 30, 907–914. [Google Scholar] [CrossRef]
- Hou, J.; Mao, Y.; Su, P.; Wang, D.; Chen, X.; Huang, S.; Ni, J.; Zhao, W.; Wu, L. A high throughput plant regeneration system from shoot stems of Sapium sebiferum Roxb., a potential multipurpose bioenergy tree. Ind. Crops Prod. 2020, 154, 112653. [Google Scholar] [CrossRef]
- Zhang, J.-j.; Yang, Y.-s.; Lin, M.-f.; Li, S.-q.; Tang, Y.; Chen, H.-b.; Chen, X.-y. An efficient micropropagation protocol for direct organogenesis from leaf explants of an economically valuable plant, drumstick (Moringa oleifera Lam.). Ind. Crops Prod. 2017, 103, 59–63. [Google Scholar]
- Dutta Gupta, S.; Agarwal, A. Artificial lighting system for plant growth and development: Chronological advancement, working principles, and comparative assessment. In Light Emitting Diodes for Agriculture; Springer: Singapore, 2017; pp. 1–25. [Google Scholar]
- Dos Reis Oliveira, T.; Aragão, V.P.M.; Moharana, K.C.; Fedosejevs, E.; do Amaral, F.P.; Sousa, K.R.; Thelen, J.J.; Venâncio, T.M.; Silveira, V.; Santa-Catarina, C. Light spectra affect the In Vitro shoot development of Cedrela fissilis Vell. (Meliaceae) by changing the protein profile and polyamine contents. Biochim. Biophys. Acta Proteins Proteom. 2020, 1868, 140529. [Google Scholar] [CrossRef]
- Sivanesan, I.; Park, S.W. Optimizing factors affecting adventitious shoot regeneration, In Vitro flowering and fruiting of Withania somnifera (L.) Dunal. Ind. Crops Prod. 2015, 76, 323–328. [Google Scholar] [CrossRef]
- Kaur, A.; Kumar, A. The effect of gelling agent, medium pH and silver nitrate on adventitious shoot regeneration in Solanum tuberosum. bioRxiv 2020, 1–15. [Google Scholar] [CrossRef]
- Yaseen, M.; Ahmad, T.; Sablok, G.; Standardi, A.; Hafiz, I.A. Review: Role of carbon sources for In Vitro plant growth and development. Mol. Biol. Rep. 2013, 40, 2837–2849. [Google Scholar] [CrossRef] [PubMed]
- Del Castello, F.; Nejamkin, A.; Cassia, R.; Correa-Aragunde, N.; Fernández, B.; Foresi, N.; Lombardo, C.; Ramirez, L.; Lamattina, L. The era of nitric oxide in plant biology: Twenty years tying up loose ends. Nitric Oxide 2019, 85, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Tiburcio, A.F.; Alcázar, R. Potential applications of polyamines in agriculture and plant biotechnology. Polyam. Methods Mol. Biol. 2018, 1694, 489–508. [Google Scholar]
- Tariq, A.; Ilyas, S.; Naz, S. Nanotechnology and plant tissue culture. In Nanoagronomy; Springer International Publishing: Cham, Switzerland, 2020; pp. 23–35. [Google Scholar]
- Gupta, K.J.; Hancock, J.T.; Petrivalsky, M.; Kolbert, Z.; Lindermayr, C.; Durner, J.; Barroso, J.B.; Palma, J.M.; Brouquisse, R.; Wendehenne, D.; et al. Recommendations on terminology and experimental best practice associated with plant nitric oxide research. New Phytol. 2020, 225, 1828–1834. [Google Scholar] [CrossRef]
- Mukherjee, S. Insights into nitric oxide–melatonin crosstalk and N-nitrosomelatonin functioning in plants. J. Exp. Bot. 2019, 70, 6035–6047. [Google Scholar] [CrossRef]
- Xu, X.-T.; Jin, X.; Liao, W.-B.; Dawuda, M.M.; Li, X.-P.; Wang, M.; Niu, L.-J.; Ren, P.-J.; Zhu, Y.-C. Nitric oxide is involved in ethylene-induced adventitious root development in cucumber (Cucumis sativus L.) explants. Sci. Hortic. 2017, 215, 65–71. [Google Scholar] [CrossRef]
- Arun, M.; Naing, A.H.; Jeon, S.M.; Ai, T.N.; Aye, T.; Kim, C.K. Sodium nitroprusside stimulates growth and shoot regeneration in chrysanthemum. Hortic. Environ. Biotechnol. 2017, 58, 78–84. [Google Scholar] [CrossRef]
- Pradhan, N.; Singh, P.; Dwivedi, P.; Pandey, D.K. Evaluation of sodium nitroprusside and putrescine on polyethylene glycol induced drought stress in Stevia rebaudiana Bertoni under In Vitro condition. Ind. Crops Prod. 2020, 154, 112754. [Google Scholar] [CrossRef]
- Pandey, S.; Sundararajan, S.; Ramalingam, S.; Pant, B. Effects of sodium nitroprusside and growth regulators on callus, multiple shoot induction and tissue browning in commercially important Valeriana jatamansi Jones. Plant Cell Tissue Organ Cult. 2020, 142, 653–660. [Google Scholar] [CrossRef]
- Hesami, M.; Tohidfar, M.; Alizadeh, M.; Daneshvar, M.H. Effects of sodium nitroprusside on callus browning of Ficus religiosa: An important medicinal plant. J. For. Res. 2020, 31, 789–796. [Google Scholar] [CrossRef]
- Ajithan, C.; Vasudevan, V.; Sathish, D.; Sathish, S.; Krishnan, V.; Manickavasagam, M. The influential role of polyamines on the In Vitro regeneration of pea (Pisum sativum L.) and genetic fidelity assessment by SCoT and RAPD markers. Plant Cell Tissue Organ Cult. 2019, 139, 547–561. [Google Scholar] [CrossRef]
- Sundararajan, S.; Sivakumar, H.P.; Nayeem, S.; Rajendran, V.; Subiramani, S.; Ramalingam, S. Influence of exogenous polyamines on somatic embryogenesis and regeneration of fresh and long-term cultures of three elite indica rice cultivars. Cereal Res. Commun. 2020. [Google Scholar] [CrossRef]
- Sathish, D.; Theboral, J.; Vasudevan, V.; Pavan, G.; Ajithan, C.; Appunu, C.; Manickavasagam, M. Exogenous polyamines enhance somatic embryogenesis and Agrobacterium tumefaciens-mediated transformation efficiency in sugarcane (Saccharum spp. hybrid). In Vitro Cell. Dev. Biol. Plant 2020, 56, 29–40. [Google Scholar] [CrossRef]
- Erland, L.A.E.; Yasunaga, A.; Li, I.T.S.; Murch, S.J.; Saxena, P.K. Direct visualization of location and uptake of applied melatonin and serotonin in living tissues and their redistribution in plants in response to thermal stress. J. Pineal Res. 2019, 66, 1–10. [Google Scholar] [CrossRef]
- Kim, D.H.; Gopal, J.; Sivanesan, I. Nanomaterials in plant tissue culture: The disclosed and undisclosed. RSC Adv. 2017, 7, 36492–36505. [Google Scholar] [CrossRef]
- Mahendran, D.; Geetha, N.; Venkatachalam, P. Role of silver nitrate and silver nanoparticles on tissue culture medium and enhanced the plant growth and development. In In Vitro Plant Breeding towards Novel Agronomic Traits; Springer: Singapore, 2019; pp. 59–74. [Google Scholar]
- Nalci, O.B.; Nadaroglu, H.; Pour, A.H.; Gungor, A.A.; Haliloglu, K. Effects of ZnO, CuO and γ-Fe3O4 nanoparticles on mature embryo culture of wheat (Triticum aestivum L.). Plant Cell Tissue Organ Cult. 2019, 136, 269–277. [Google Scholar] [CrossRef]
- Sarmast, M.K.; Niazi, A.; Salehi, H.; Abolimoghadam, A. Silver nanoparticles affect ACS expression in Tecomella undulata In Vitro culture. Plant Cell Tissue Organ Cult. 2015, 121, 227–236. [Google Scholar] [CrossRef]
- Kumar, V.; Guleria, P.; Kumar, V.; Yadav, S.K. Gold nanoparticle exposure induces growth and yield enhancement in Arabidopsis thaliana. Sci. Total Environ. 2013, 461–462, 462–468. [Google Scholar] [CrossRef]
- Sharma, P.; Bhatt, D.; Zaidi, M.G.H.; Saradhi, P.P.; Khanna, P.K.; Arora, S. Silver nanoparticle-mediated enhancement in growth and antioxidant status of Brassica juncea. Appl. Biochem. Biotechnol. 2012, 167, 2225–2233. [Google Scholar] [CrossRef]
- Hesami, M.; Jones, A.M.P. Application of artificial intelligence models and optimization algorithms in plant cell and tissue culture. Appl. Microbiol. Biotechnol. 2020, 104, 9449–9485. [Google Scholar] [CrossRef] [PubMed]
- Nezami-Alanagh, E.; Garoosi, G.A.; Landín, M.; Gallego, P.P. Combining DOE with neurofuzzy logic for healthy mineral nutrition of pistachio rootstocks In Vitro culture. Front. Plant Sci. 2018, 9, 1–12. [Google Scholar] [CrossRef]
- Prasad, A.; Prakash, O.; Mehrotra, S.; Khan, F.; Mathur, A.K.; Mathur, A. Artificial neural network-based model for the prediction of optimal growth and culture conditions for maximum biomass accumulation in multiple shoot cultures of Centella asiatica. Protoplasma 2017, 254, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Jamshidi, S.; Yadollahi, A.; Arab, M.M.; Soltani, M.; Eftekhari, M.; Sabzalipoor, H.; Sheikhi, A.; Shiri, J. Combining gene expression programming and genetic algorithm as a powerful hybrid modeling approach for pear rootstocks tissue culture media formulation. Plant Methods 2019, 15, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Hesami, M.; Naderi, R.; Tohidfar, M. Introducing a hybrid artificial intelligence method for high-throughput modeling and optimizing plant tissue culture processes: The establishment of a new embryogenesis medium for chrysanthemum, as a case study. Appl. Microbiol. Biotechnol. 2020, 104, 10249–10263. [Google Scholar] [CrossRef]



| Source | Explant (Response) | C. sativa Type (#CVs Responded/Used) | Best Media | Best Results | Stages Reported |
|---|---|---|---|---|---|
| Richez-Dumanois et al., 1986 [66] | Apical and axillary nodes (SM) | Fiber-type (2/2) | SM: MS + 0.5 μM BAP + 0.1 μM IBA | SM: 2 shoots/explant (apical meristem), % response N.S. | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: Y |
| Rooting: MS + 0.2% activated charcoal + 10 μM IBA | Rooting: 47.7% response | ||||
| Lata et al., 2009a [70] | Axillary nodes (SM and rooting) | Drug-type (1/1) | SM: MS + 0.5 μM TDZ | SM: 12.6 shoots/explant 100% response | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: Y |
| Rooting: ½ MS + 2.5 μM IBA + 0.05% activated charcoal | Rooting: 4.8 roots/explant 95% response | ||||
| Lata et al., 2009b [71] | Alginate encapsulated axillary nodes (Shoot induction and rooting) | Drug-type (1/1) | Shoot induction: MS + 0.5 μM TDZ + 0.075% PPM | Shoot induction: 11.8 shoots/explant (90 days; avg. 30 explants) | Stage 0: Y Stage 1: Y Stage 2: Y Stage 3: Y Stage 4: Y |
| Rooting: (1:1) sterile fertilome: coco natural growth medium + MS + 0.5% PPM | Rooting: 100% conversion from encapsulation (90 days) | ||||
| Lata et al., 2016 [72] | Axillary nodes (SM and rooting) | Drug-type (1/1) | SM: MS + 2 μM mT | SM: 13.4 shoots/explant 100% response | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: Y |
| Rooting: MS + 2 μM mT | Rooting: 13.8 roots/explant 96% response | ||||
| Grulichova et al., 2017 [73] | Shoot tips (SM) | Fiber-type (2/2) | SM: MS + 0.54 μM NAA + 1.78 μM BAP a | SM: Shoots/explant N.S. % response N.S. | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: N Stage 4: N |
| Piunno et al., 2019 [74] | Immature and mature inflorescences (shoot induction and rooting) | Drug-type (2/3) | Shoot induction: MS + 10 µM TDZ | Shoot induction: 4 shoots/floral cluster % response N.S. | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: Y |
| Rooting: MS + 1.86 µM kinetin + 0.54 µM NAA a | Rooting: Describes ‘most’ cultures as rooting. | ||||
| Smýkalová et al., 2019 [75] | Shoot apex, isolated apical meristem, and cotyledonary nodes from seedlings (SM, shoot development, and rooting) | Fiber-type (1/1) | SM: IMB4 + 6.97 µM KIN + 0.81 µM BAP9THP + 0.11 mM adenine hemisulphate a | SM: 4.4 shoots/explant (isolated meristems) ~96% response | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: N |
| Shoot development: ½ MS no PGRs | Shoot development: N.S. | ||||
| Rooting: ½ MS + 0.20 µM NAA a | Rooting: 50% response | ||||
| Monthony et al., 2020a [76] | Single and pairs of florets (floral reversion and rooting) | Drug-type (2/2) | Floral reversion: DKW w/vitamins + 1 µM mT | Floral reversion: Estimated 18.2 explants derived from one In Vitro flowering plant 81% response | Stage 0: N Stage 1: N Stage 2: Y Stage 3: Y Stage 4: Y |
| Rooting: DKW w/vitamins | Rooting: 44% rooted | ||||
| Page et al., 2020 [77] | Axillary nodes (SM) | Drug-type (4/5) | SM: DKW + 0.5 μM TDZ | SM: 2.23 shoots/explant 80% response | Stage 0: N Stage 1: N Stage 2: Y Stage 3: N Stage 4: N |
| Wróbel et al., 2020 [78] | Shoot tips and nodes from axillary branches (SM and rooting) | Fiber-type (1/1) | SM: ½ MS + 2.85 μM IAA a | SM: 2.5 shoots/explant 70% response | Stage 0: Y Stage 1: Y Stage 2: Y Stage 3: Y Stage 4: Y |
| Rooting: ½ MS + 2.85 μM IAA a | Rooting: 74.6% rooted | ||||
| Codesido et al., 2020 [79] | Axillary nodes (SM) | Drug-type (6/6) | SM: Formula βH media | SM: Shoots/explant N.S. 58% response | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: N Stage 4: N |
| Mestinšek Mubi et al., 2020 [80] | Axillary nodes (SM) | Drug-type b (2/2) | SM: MS+ 2.07 µM mT a | SM: 1.78 shoots/explant 97.8% response | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: Y |
| Rooting: MS + no PGRs | Rooting: % response N.S. |
| Source | Explant (Response) | C. sativa Type (#CVs Responded/ Used) | Optimal Media | Optimal Results | Stages Reported |
|---|---|---|---|---|---|
| Mandolino and Ranalli, 1999 [81] | Leaf, hypocotyl, cotyledon, and root (Callogenesis and shoot regeneration) | Fiber-type (1/12) | Callogenesis/ shoot regeneration: MS + B5 vitamins + 13.57–45.24 µM 2,4-D + 0.04–0.44 µM BAPa | Callogenesis/shoot regeneration: One tested cultivar occasionally gave rise to organogenic callus from hypocotyl tissue. % regeneration N.S. | Stage 0: N Stage 1: Y Stage 2: N Stage 3: Y Stage 4: N |
| Ślusarkiewicz-Jarzina et al., 2005 [82] | Juvenile leaves, petioles, internodes, and axillary nodes (Callus induction, shoot induction, and rooting) | Fiber-type (5/5) | Callus induction: MS + dicamba (9.05 and 13.57 µM a) | Callus induction: 52.3% (5 CV Average; petioles) | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: Y |
| Shoot induction: MS + dicamba (9.05 and 13.57 µM a) | Shoot induction: 2.5% (cv. Silesia; petioles) | ||||
| Rooting: MS + 0.57 µM IAA + 0.54 µM NAA a | Rooting: 69.9% plantlets formed roots | ||||
| Plawuszewski et al., 2006 [67] | Axillary nodes (Direct organogenesis) Stems and roots (Indirect somatic embryogenesis) | Fiber-type (3/3) | Callus induction: DARIAind+ + NAA + BAP (concentrations N.S.) | Callus induction: % callusing N.S. | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: N |
| Shoot proliferation: DARIApro + NAA + BAP (concentrations N.S.) | Shoot proliferation: Adventitious shoot formation from axillary nodes and somatic embryo formation from stem tissue reported % N.S. | ||||
| Somatic embryogenesis: DARIApro+ + NAA + BAP (concentrations N.S.) | Somatic embryogenesis: N.S. | ||||
| Rooting: DARIAroot + IAA (concentrations N.S.) | Rooting: N.S. | ||||
| Raharjo et al., 2006 [83] | Leaves, flowers, and seedling roots, stems, and shoots (Callogenesis, callus suspension cultures) | Drug-type (0/1) | Callogenesis: MS + 0.56 mM mesoinositol + 29.65 µM thiamine diHCl + 4.86 pyridoxine HCl + 8.12 µM nicotinic acid + 4.52 µM 2,4-D a | Callogenesis: Statistical analysis N.S. Callusing was greatest using flowers and seedling shoots | Stage 0: Y Stage 1: Y Stage 2: Y Stage 3: N Stage 4: N |
| Suspension culture (2 steps): Step 1: MS (as above, aqueous; 2 weeks) Step 2: B5 media + 9.05 µM 2,4-D + 2.85 µM IAA + 2.69 µM NAA + 5.12 µM potassium a | Suspension culture: Continued callus growth, no regeneration | ||||
| Wielgus et al., 2008 [68] | Cotyledons, axillary nodes, and roots (Callus induction, shoot induction, and rooting) | Fiber-type (3/3) | Callus induction: DARIA (ind+) + 4.65 µM kinetin + 0.27 µM NAA a | Callus induction: Best morphogenic callus induction: stem explants (all cultivars) Statistical analysis N.S. | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: N |
| Shoot induction: DARIA (pro+) + 0.89 µM BAP + 0.16 µM NAA a | Shoot induction: 15.56% with cotyledon explants (cv. Beniko) | ||||
| Rooting: DARIA (root+) + 11.42 µM IAA a | Rooting: Statistical analysis N.S. | ||||
| Flores-Sanchez et al., 2009 [84] | Leaves (Callus suspension cultures and somatic embryogenesis) | Drug-type (1/1) | Suspension culture: MS + B5 vitamins + 4.52 µM 2,4-D + 4.65 µM kinetin | Suspension culture: Growth rate N.S. | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: N Stage 4: N |
| Somatic embryogenesis: Media composition N.S. | Somatic embryogenesis: Number of embryos N.S | ||||
| Lata et al., 2010 [85] | Juvenile leaves (Callogenesis, shoot induction, and rooting) | Drug-type (1/1) | Callogenesis: MS + 0.5 μM NAA + 1 μM TDZ | Callogenesis: 93.3% response | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: Y |
| Shoot induction: MS + 0.5 μM TDZ | Shoot induction: 12.3 shoots/explant 96.6% response | ||||
| Rooting: ½ MS + 2.5 μM IBA | Rooting: 10 roots/explant 96.6% response | ||||
| Farag, 2014 [28] | Juvenile leaves (Callogenesis and shoot regeneration) | Drug-type (1/1) | Callogenesis: B5 + 2.69 µM NAA + 22.20 µM BAP + 0.11 mM adenine hemisulfate a | Callogenesis: 50% callusing response | Stage 0: Y Stage 1: Y Stage 2: Y Stage 3: Y Stage 4: Y |
| Shoot regeneration: B5 + 1.44 µM GA3a | Shoot regeneration: 8.5 shoots/callus % regeneration N.S. | ||||
| Rooting: B5 + 8.56 µM IAA a | Rooting: 2.75 roots/explant 100% response | ||||
| Movahedi et al., 2015 [86] | Cotyledons and epicotyls (callogenesis+ shoot regeneration, rooting) | Drug-type (1/1) | Callogenesis/shoot regeneration: MS + 8.88 µM BAP + 2.46 µM IBA | Callogenesis/shoot regeneration: ~2 shoots/epicotyl % response N.S. | Stage 0: Y Stage 1: Y Stage 2: Y Stage 3: Y Stage 4: Y |
| Rooting: MS + 0.49 μM IBA a | Rooting: % response N.S. | ||||
| Chaohua et al., 2016 [87] | Cotyledon (callogenesis + shoot regeneration, rooting) | Fiber-type (8/8) | Callogenesis/shoot regeneration: MS + 1.82 μM TDZ + 1.07 μM NAA a | Callogenesis/shoot regeneration: 3 shoots/explant (3-day-old cotyledons) 51.7% regeneration | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: Y |
| Rooting: ½ MS + 2.46–9.84 μM IBA a | Rooting: 80% response | ||||
| Galán-Ávila et al., 2020 [88] | Hypocotyl, cotyledon and first two true leaves (direct organogenesis and rooting) | Fiber-type (5/5) | Organogenesis: MS + 1.82 μM TDZ + 1.07 μM NAA a | Organogenesis: 1.49 shoots/hypocotyl 54.17% response | Stage 0: Y Stage 1: Y Stage 2: N Stage 3: Y Stage 4: Y |
| Rooting: MS + 1.82 μM TDZ + 1.07 μM NAA a | Rooting: ~18% rooted | ||||
| Monthony et al., 2020b [89] | Young leaves (callus induction and shoot regeneration) | Drug-type (10/10) | Callogenesis: MS + 0.5 μM NAA + 1 μM TDZ | Callogenesis: 100% response across all 10 cultivars | Stage 0: N Stage 1: N Stage 2: Y Stage 3: N Stage 4: N |
| Shoot regeneration: MS + 0.5 μM TDZ | Shoot regeneration: Not achieved |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monthony, A.S.; Page, S.R.; Hesami, M.; Jones, A.M.P. The Past, Present and Future of Cannabis sativa Tissue Culture. Plants 2021, 10, 185. https://doi.org/10.3390/plants10010185
Monthony AS, Page SR, Hesami M, Jones AMP. The Past, Present and Future of Cannabis sativa Tissue Culture. Plants. 2021; 10(1):185. https://doi.org/10.3390/plants10010185
Chicago/Turabian StyleMonthony, Adrian S., Serena R. Page, Mohsen Hesami, and Andrew Maxwell P. Jones. 2021. "The Past, Present and Future of Cannabis sativa Tissue Culture" Plants 10, no. 1: 185. https://doi.org/10.3390/plants10010185
APA StyleMonthony, A. S., Page, S. R., Hesami, M., & Jones, A. M. P. (2021). The Past, Present and Future of Cannabis sativa Tissue Culture. Plants, 10(1), 185. https://doi.org/10.3390/plants10010185






