Spatiotemporal Assessment of Littoral Waterbirds for Establishing Ecological Indicators of Mediterranean Coastal Lagoons
Abstract
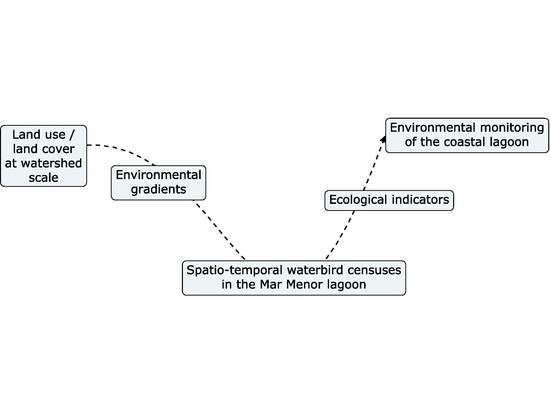
1. Introduction
2. Materials and Methods
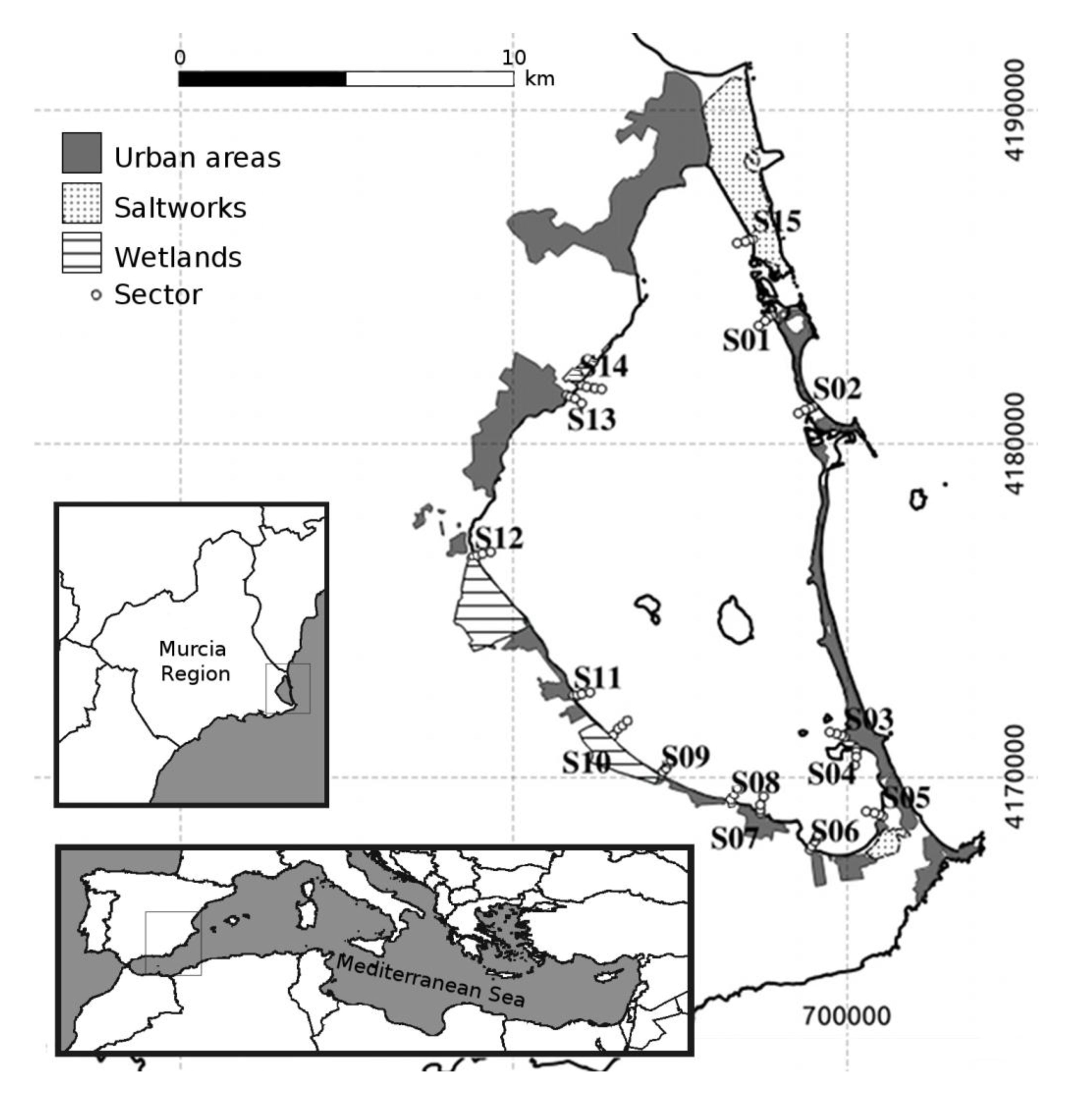
2.1. Study Area
2.2. Bird Census
2.3. Data Analyses
2.3.1. Bird Data
2.3.2. Definition of Landscape Gradients and Selection of Environmental Predictors
2.3.3. Multiple Regression Models
3. Results
3.1. Spatiotemporal Variation of Waterbird-Based Indices
3.2. Indicator Species, Community Indices, and Influential Gradients
3.3. Multiple Regression Models
4. Discussion
4.1. Spatial and Temporal Variation of Waterbird-Based Indices
4.2. Community Indices’ Models
4.3. Wintering Indicator Species’ Models
4.4. Summer Indicator Species’ Models
4.5. Management Implications
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Crutzen, P.J. Geology of mankind. Nature 2002, 415, 23. [Google Scholar]
- Brazner, J.C.; Danz, N.P.; Niemi, G.J.; Regal, R.R.; Trebitz, A.S.; Howe, R.W.; Hanowski, J.M.; Johnson, L.B.; Ciborowski, J.J.H.; Johnston, C.A.; et al. Evaluation of geographic, geomorphic and human influences on Great Lakes wetland indicators: A multi-assemblage approach. Ecol. Indic. 2007, 7, 610–635. [Google Scholar] [CrossRef]
- Danz, N.P.; Niemi, G.J.; Regal, R.R.; Hollenhorst, T.; Johnson, L.B.; Hanowski, J.M.; Axler, R.P.; Ciborowski, J.J.H.; Hrabik, T.; Brady, V.J.; et al. Integrated measures of anthropogenic stress in the U.S. Great Lakes Basin. Environ. Manag. 2007, 39, 631–647. [Google Scholar] [CrossRef] [PubMed]
- Martínez-López, J.; Carreño, M.F.; Martínez-Fernández, J.; Esteve, M.A. Wetland and landscape indices for assessing the condition of semiarid Mediterranean saline wetlands under agricultural hydrological pressures. Ecol. Indic. 2014, 36, 400–408. [Google Scholar] [CrossRef]
- Howe, R.W.; Regal, R.R.; Hanowski, J.; Niemi, G.J.; Danz, N.P.; Smith, C.R. An index of ecological condition based on bird assemblages in Great Lakes coastal wetlands. J. Gt. Lakes Res. 2007, 33, 93–105. [Google Scholar] [CrossRef]
- European Commission. Common Implementation Strategy for the Water Framework Directive (2000/60/EC); Publications Office of the European Union: Luxembourg City, Luxembourg, 2000. [Google Scholar]
- Rowan, J.S.; Carwardine, J.; Duck, R.W.; Bragg, O.M.; Black, A.R.; Cutler, M.E.J.; Soutar, I.; Boon, P.J. Development of a technique for Lake Habitat Survey (LHS) with applications for the European Union Water Framework Directive. Aquat. Conserv. Mar. Freshw. Ecosyst. 2006, 16, 637–657. [Google Scholar] [CrossRef]
- Viaroli, P.; Mistri, M.; Troussellier, M.; Guerzoni, S.; Cardoso, A.C. Structure, functions and ecosystems alterations in Southern European coastal lagoons: Preface. Hydrobiologia 2005, 550, vii–ix. [Google Scholar] [CrossRef]
- Newton, A.; Icely, J.; Cristina, S.; Brito, A.; Cardoso, A.C.; Colijn, F.; Riva, S.D.; Gertz, F.; Hansen, J.; Holmer, M.; et al. An overview of ecological status, vulnerability and future perspectives of European large shallow, semi-enclosed coastal systems, lagoons and transitional waters. Estuar. Coast. Shelf Sci. 2014, 140, 95–122. [Google Scholar] [CrossRef]
- Gregory, R.D.; Noble, D.; Field, R.; Marchant, J.; Raven, M.; Gibbons, D.W. Using birds as indicators of biodiversity. Ornis Hung. 2003, 12–13, 11–24. [Google Scholar]
- Stolen, E.D.; Breininger, D.R.; Frederick, P.C. Using waterbirds as indicators in estuarine systems: Successes and perils. In Estuarine Indicators; Bortone, S.A., Ed.; CRC Press: Boca Raton, FL, USA, 2004; pp. 409–422. [Google Scholar]
- Paillisson, J.M.; Reeber, S.; Marion, L. Bird assemblages as bio-indicators of water regime management and hunting disturbance in natural wet grasslands. Biol. Conserv. 2002, 106, 115–127. [Google Scholar] [CrossRef]
- Palacio–Núñez, J.; Verdú, J.R.; Galante, E.; Jiménez–García, D.; Olmos–Oropeza, G. Birds and fish as bioindicators of tourist disturbance in springs in semi–arid regions in Mexico: A basis for management. Anim. Biodivers. Conserv. 2007, 30, 29–41. [Google Scholar]
- Robledano Aymerich, F.; Pagán Abellán, I.; Calvo Sendín, J.F. Waterbirds and nutrient enrichment in Mar Menor Lagoon, a shallow coastal lake in southeast Spain. Lakes Reserv. Res. Manag. 2008, 13, 37–49. [Google Scholar] [CrossRef]
- Robledano, F.; Esteve, M.A.; Martinez-Fernandez, J.; Farinós, P. Determinants of wintering waterbird changes in a Mediterranean coastal lagoon affected by eutrophication. Ecol. Indic. 2011, 11, 395–406. [Google Scholar] [CrossRef]
- McKinney, R.A.; McWilliams, S.R.; Charpentier, M.A. Waterfowl-habitat associations during winter in an urban North Atlantic estuary. Biol. Conserv. 2006, 132, 239–249. [Google Scholar] [CrossRef]
- Cardoni, D.A.; Isacch, J.P.; Fanjul, M.E.; Escapa, M.; Iribarne, O.O. Relationship between anthropogenic sewage discharge, marsh structure and bird assemblages in an SW Atlantic saltmarsh. Mar. Environ. Res. 2011, 71, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Carreño, M.F.; Esteve, M.A.; Martinez, J.; Palazón, J.A.; Pardo, M.T. Habitat changes in coastal wetlands associated to hydrological changes in the water-shed. Estuar. Coast. Shelf Sci. 2008, 77, 475–483. [Google Scholar] [CrossRef]
- Martínez-López, J.; Carreño, M.F.; Palazón-Ferrando, J.A.; Martínez-Fernández, J.; Esteve, M.A. Free advanced modeling and remote sensing techniques for wetland watersheds delineation and monitoring. Int. J. Geogr. Inf. Sci. 2013, 18. [Google Scholar] [CrossRef]
- Martínez-López, J.; Carreño, M.F.; Palazón-Ferrando, J.; Martínez-Fernández, J.; Esteve, M.A. Remote sensing of plant communities as a tool for assessing the condition of semiarid Mediterranean saline wetlands in agricultural catchments. Int. J. Appl. Earth Observ. Geoinform. 2014, 26, 193–204. [Google Scholar] [CrossRef]
- Martínez-López, J.; Martínez-Fernández, J.; Naimi, B.; Carreño, M.F.; Esteve, M.A. An open-source spatio-dynamic wetland model of plant community responses to hydrological pressures. Ecol. Model. 2015, 306, 326–333. [Google Scholar] [CrossRef]
- Webb, E.B.; Smith, L.M.; Vrtiska, M.P.; Lagrange, T.G. Effects of local and landscape variables on wetland bird habitat Use during migration through the rainwater basin. J. Wildl. Manag. 2010, 74, 109–119. [Google Scholar] [CrossRef]
- Pearse, A.; Kaminski, R.; Reinecke, K.; Dinsmore, S. Local and landscape associations between wintering dabbling ducks and wetland complexes in Mississippi. Wetlands 2012, 32, 859–869. [Google Scholar] [CrossRef]
- Martínez, J.; Esteve, M.A.; Robledano, F.; Pardo, M.T.; Carreño, M.F. Aquatic birds as bioindicators of trophic changes and ecosystem deterioration in the Mar Menor lagoon (SE, Spain). Hydrobiologia 2005, 550, 221–235. [Google Scholar]
- Velasco, J.; Lloret, J.; Millán, A.; Marín, A.; Barahona, J.; Abellán, P.; Sánchez-Fernández, D. Nutrient and particulate inputs into the Mar Menor lagoon (SE Spain) from an intensive agricultural watershed. Water Air Soil Pollut. 2006, 176, 37–56. [Google Scholar] [CrossRef]
- Pardo, M.T.; Esteve, M.A.; Giménez, A.; Martínez-Fernández, J.; Carreño, M.F.; Serrano, J.; Miñano, J. Assessment of hydrological alterations on wandering beetle assemblages (coleoptera: Carabidae and Tenebrionidae) in coastal wetlands of arid Mediterranean systems. J. Arid Environ. 2008, 72, 1803–1810. [Google Scholar] [CrossRef]
- Fuller, R.J. (Ed.) Birds and Habitat: Relationships in Changing Landscapes; Cambridge University Press: Cambridge, UK, 2012. [Google Scholar]
- Ramsar Convention Secretariat. The Ramsar Convention Manual: A guide to the Convention on Wetlands (Ramsar, Iran, 1971), 6th ed.; Ramsar Convention Secretariat: Gland, Switzerland, 2013. [Google Scholar]
- Robledano, F.; Farinós, P. Waterbirds as bioindicators in coastal lagoons: Background, potential value and recent research in Mediterranean areas. In Lagoons: Biology, Management and Environmental Impact; Friedman, A.G., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2010; pp. 153–183. [Google Scholar]
- Robledano, F.; Esteve, M.A.; Farinós, P.; Carreño, M.F.; Martıínez-Fernández, J. Terrestrial birds as indicators of agricultural-induced changes and associated loss in conservation value of Mediterranean wetlands. Ecol. Indic. 2010, 10, 274–286. [Google Scholar] [CrossRef]
- EC. Biodiversity strategy for 2020: Summaries of EU legislation. European Commission 2014: Brussels, 2014. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=URISERV:ev0029 (accessed on 24 July 2017).
- Pérez-Ruzafa, A.; Gilabert, J.; Gutiérrez, J.M.; Fernández, A.I.; Marcos, C.; Sabah, S. Evidence of a planktonic food web response to changes in nutrient input dynamics in the Mar Menor coastal lagoon, Spain. Hydrobiologia 2002, 475–476, 359–369. [Google Scholar] [CrossRef]
- Marín-Guirao, L.; Marín, A.; Lloret, J.; Martínez, E.; García Fernández, A. Effects of mining wastes on a seagrass ecosystem: Metal acumulation and bioavailability, seagrass dynamics and associated community structure. Mar. Environ. Res. 2005, 60, 317–337. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Rogel, J.; Jiménez-Cárceles, F.J.; Roca, M.J.; Ortiz, R. Changes in soils and vegetation in a Mediterranean coastal salt marsh impacted by human activities. Estuar. Coast. Shelf Sci. 2007, 73, 510–526. [Google Scholar] [CrossRef]
- García-Pintado, J.; Martínez-Mena, M.; Barberá, G.G.; Albaladejo, J.; Castillo, V.M. Anthropogenic nutrient sources and loads from a Mediterranean catchment into a coastal lagoon: Mar Menor, Spain. Sci. Total Environ. 2007, 373, 220–239. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Ruzafa, A.; Marcos, C.; Pérez-Ruzafa, I.M.; Barcala, E.; Hegazi, M.I.; Quispe, J. Detecting changes resulting from human pressure in a naturally quick-changing and heterogeneous environment: Spatial and temporal scales of variability in coastal lagoons. Estuar. Coast. Shelf Sci. 2007, 75, 175–188. [Google Scholar] [CrossRef]
- García-Sánchez, M.; Pérez-Ruzafa, I.M.; Marcos, C.; Pérez-Ruzafa, A. Suitability of benthic macrophyte indices (EEI, E-MaQI and BENTHOS) for detecting anthropogenic pressures in a Mediterranean coastal lagoon (Mar Menor, Spain). Ecol. Indic. 2012, 19, 48–60. [Google Scholar] [CrossRef]
- Hernández, V.; Robledano, F. La Comunidad de aves Ccuáticas del Mar Menor (Murcia, SE España): Aproximación a su Respuesta a las Modificaciones Ambientales en la Laguna. In Actas XII Jornadas Ornitológicas Españolas; Instituto de Estudios Almerienses: Almería, Spain, 1997; Available online: http://dialnet.unirioja.es/descarga/articulo/2244582.pdf (accessed on 24 July 2017).
- Marín-Guirao, L.; César, A.; Marín, A.; Lloret, J.; Vita, R. Establishing the ecological quality status of soft-bottom mining-impacted coastal water bodies in the scope of the Water Framework Directive. Mar. Pollut. Bull. 2005, 50, 374–387. [Google Scholar] [CrossRef] [PubMed]
- Lloret, J.; Marín, A.; Marín-Guirao, L.; Velasco, J. Changes in macrophytes distribution in a hypersaline coastal lagoon associated with the development of intensively irrigated agriculture. Ocean Coast. Manag. 2005, 48, 828–842. [Google Scholar] [CrossRef]
- Pérez-Ruzafa, A.; Fernández, A.; Marcos, C.; Gilabert, J.; Quispe, J.; García-Charton, J.A. Spatial and temporal variations of hydrological conditions, nutrients and chlorophyll a in a Mediterranean coastal lagoon (Mar Menor, Spain). Hydrobiologia 2005, 550, 11–27. [Google Scholar] [CrossRef]
- Salas, F.; Marcos, C.; Neto, J.M.; Patrício, J.; Pérez-Ruzafa, A.; Marques, J.C. User-friendly guide for using benthic ecological indicators in coastal and marine quality assessment. Ocean Coast. Manag. 2006, 49, 308–331. [Google Scholar] [CrossRef]
- Pérez-Ruzafa, A.; Marcos, C. Fisheries in coastal lagoons: An assumed but poorly researched aspect of the ecology and functioning of coastal lagoons. Estuar. Coast. Shelf Sci. 2012, 110, 15–31. [Google Scholar] [CrossRef]
- Farinós, P.; Robledano, F. Structure and distribution of the waterbird community in the Mar Menor coastal lagoon (SE Spain) and relationships with environmental gradients. Waterbirds 2010, 33, 479–493. [Google Scholar]
- Farinós, P.; Robledano, F.; Perona, C.; Soto, A.J. Lagoons as a Waterbird Habitat: Response of Communities to Human Impact and Management accross Space and Time Scales. In Lagoons: Habitats and Species, Human Impacts and Ecological Effects; Mwinyihija, M., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2013; pp. 57–108. [Google Scholar]
- Taft, O.W.; Haig, S.M. Importance of wetland landscape structure to shorebirds wintering in an agricultural valley. Landsc. Ecol. 2006, 21, 169–184. [Google Scholar] [CrossRef]
- Esteve, M.A.; Martínez, J. Mar Menor-Spain. DITTY Project. EC “Energy, Environment and Sustainable Development Programme”. Available online: http://cordis.europa.eu/project/rcn/69066_en.html (accessed on 24 July 2017).
- Carreño, M.F.; Martínez-López, J.; Palazón, J.A.; González, J.C.; Esteve, M.A.; Martínez, J. Aplicación de nuevas técnicas para la clasificación supervisada de imágenes Landsat en la determinación de usos del suelo: Por píxel y por mancha. Mapp. Interact. 2011, Marzo-abril, 68–73. [Google Scholar]
- Perez-Ruzafa, A.; Marcos-Diego, C.; Ros, J.D. Environmental and biological changes related to recent human activities in the Mar Menor (SE of Spain). Mar. Pollut. Bull. 1991, 23, 747–751. [Google Scholar] [CrossRef]
- Van Roomen, M.; Koffijberg, K.; Noordhuis, R.; Soldaat, L. Long-term waterbird monitoring in The Netherlands: A tool for policy and management. In Waterbirds around the World; The Stationery Office: Edinburgh, UK, 2006. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Clarke, K.R. Non-parametric multivariate analyses of changes in community structure. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER V6: User Manual/Tutorial; PRIMER-E: Plymouth, UK, 2006. [Google Scholar]
- GRASS Development Team. Geographic Resources Analysis Support System (GRASS) Software, Version 6.4. Open Source Geospatial Foundation, 2010. Available online: http://www.primer-e.com/ (accessed on 24 July 2017).
- Martínez-López, J.; Carreño, M.F.; Palazón, J.A.; Esteve, M.A.; Martínez-Fernández, J. Wetland-Watershed Modelling and Assessment: GIS Methods for Establising Multiscale Indicators. In Wetlands: Ecology, Management and Conservation; Baranyai, A., Benkô, D., Eds.; Nova Science Publishers, Inc. Hauppauge: New York, NY, USA, 2012; pp. 231–250. [Google Scholar]
- Carreño, M.F. Seguimiento de los Cambios de Usos y su Influencia en las Comunidades y Hábitats Naturales en la Cuenca del Mar Menor, 1988–2009, con el uso de SIG y Teledetección”. Ph.D. Thesis, Universidad de Murcia, Murcia, Spain, 2015. [Google Scholar]
- Carreño, M.F.; Palazon, J.A.; Martínez-López, J. 1988–2009 time-series of land-use/land-cover maps for the Mar Menor/Campo de Cartagena watershed by means of supervised classification of Landsat images. Int. J. Geogr. Inf. Sci. 2015. [Google Scholar] [CrossRef]
- Coreau, A.; Martin, J.L. Multi-scale study of bird species distribution and of their response to vegetation change: A Mediterranean example. Landsc. Ecol. 2007, 22, 747–764. [Google Scholar] [CrossRef]
- McCune, B.; Mefford, M.J. Multivariate Analysis of Ecological Data; versión 4.20; MjM Software: Gleneden Beach, OR, USA, 1999. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. A practical Information-Theoretic Approach. In Model Selection and Multimodel Inference, 2nd ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Birdlife International. Birds in Europe. Population Estimates, Trends and Conservation Status. Available online: https://www.eea.europa.eu/data-and-maps/indicators/red-list-index-for-european-species/birdlife-international-2004a-birds-in (accessed on 24 July 2017).
- Bryce, S.A.; Hughes, R.M.; Kaufmann, P.R. Development of a bird integrity index: Using bird assemblages as indicators of riparian condition. Environ. Manag. 2002, 30, 294–310. [Google Scholar] [CrossRef] [PubMed]
- Tománková, I.; Boland, H.; Reid, N.; Fox, A.D. Assessing the extent to which temporal changes in waterbird community composition are driven by either local, regional or global factors. Aquat. Conserv. Mar. Freshw. Ecosyst. 2013, 23, 343–355. [Google Scholar] [CrossRef]
- Lopes, R.J.; Múrias, T.; Cabral, J.A.; Marques, J.C. A ten year study of variation, trends and seasonality of a shorebird community in the Mondego Estuary, Portugal. Waterbirds 2005, 28, 8–18. [Google Scholar] [CrossRef]
- Rendón, M.A.; Green, A.J.; Aguilera, E.; Almaraz, P. Status, distribution and long-term changes in the waterbird community wintering in Doñana, south–west Spain. Biol. Conserv. 2008, 141, 1371–1388. [Google Scholar] [CrossRef]
- Liordos, V. Foraging Guilds of Waterbirds Wintering in a Mediterranean Coastal Wetland. Zool. Stud. 2010, 49, 311–323. [Google Scholar]
- Athearn, N.D.; Takekawa, J.Y.; Bluso-Demers, J.D.; Shinn, J.M.; Arriana, B.L.; Robinson-Nilsen, C.W.; Strong, C.M. Variability in habitat value of commercial salt production ponds: Implications for waterbird management and tidal marsh restoration planning. Hydrobiologia 2012, 697, 139–155. [Google Scholar] [CrossRef]
- Verdiell-Cubedo, D.; Oliva Paterna, F.J.; Egea Serrano, A.; Torralva Forero, M. Population biology and habitat associations of benthic fish species in the shallow areas of a Mediterranean coastal lagoon (SE Iberian Peninsula). Sci. Mar. 2008, 72, 319–328. [Google Scholar]
- Verdiell-Cubedo, D.; Torralva, M.; Andreu-Soler, A.; Oliva-Paterna, F. Effects of Shoreline Urban Modification on habitat structure and fish community in littoral areas of a Mediterranean Coastal Lagoon (Mar Menor, Spain). Wetlands 2012, 32, 631–641. [Google Scholar] [CrossRef]
- Pescador, M.; Díaz, S.; Peris, S. Abundances of waterbird species on lakes in Argentine Patagonia as a function of season, lake size and the presence of mink. Hydrobiología 2012, 697, 111–125. [Google Scholar] [CrossRef]
- Sebastián-González, E.; Sánchez-Zapata, J.; Botella, F. Agricultural ponds as alternative habitat for waterbirds: Spatial and temporal patterns of abundance and management strategies. Eur. J. Wildl. Res. 2010, 56, 11–20. [Google Scholar] [CrossRef]
- Rosique, M.J. Recopilación Y Análisis De Los Trabajos Existentes Sobre El Mar Menor; Unpublished Technical Document; Centro Oceanográfico de Murcia (IEO): Murcia, Spain, 2000. [Google Scholar]
- Infante, O.; Fuente, U.; Atienza, J.C. Las Áreas Importantes Para La Conservación De Las Aves En España; SEO/BirdLife: Madrid, Spain, 2011. [Google Scholar]
- Farinós, P.; Robledano, F.; Quispe-Becerra, J.I.; Marcos, C.; Pérez-Ruzafa, A. The waterbird community of the Mar Menor coastal lagoon (SE Spain): Spatial relationships with major environmental gradients and trophic web components. In Proceedings of the 4th European Conference on Coastal Lagoon Research, Montpellier, France, 30 November 2009. [Google Scholar]
- Blumstein, D.T.; Anthony, L.L.; Harcourt, R.; Ross, G. Testing a key assumption of wildlife buffer zones: Is flight initiation distance a species-specific trait? Biol. Conserv. 2003, 110, 97–100. [Google Scholar] [CrossRef]
| VARIABLE ACRONYM | DESCRIPTION |
|---|---|
| Spatial and Temporal Factors | |
| STATION | Sampling stations (S1 to S15) |
| BAND | Different distance to shore (B1 = 100 m, B2 = 100 ‒ 250 m, B3 = 250 ‒ 500 m, B4 = 500 ‒ 1000 m) |
| YEAR | Consecutive years of sampling (year 1 = annual cycle from October 2006 to October 2007; year 2 = annual cycle from October 2007 to October 2008; year 3 and year 4 correspond to the wintering periods of 2009/2010 and 2010/2011 respectively) |
| Distance to Disturbing or Shelter Elements | |
| DMMI | Distance to nearest lagoon island |
| DMEI | Distance to nearest Mediterranean island |
| DCAN | Distance to communication channel (Estacio Channel) with the Mediterranean Sea |
| DALB | Distance to main permanent discharge focus (Albujón ephemeral river mouth) |
| DURB | Distance to nearest urban centre |
| DHARB | Distance to nearest harbour |
| DAIR | Distance to airport (San Javier Airport) |
| DCSAL | Distance to nearest saltpan (industrial area with shelter role for waterbirds) |
| DCWET | Distance to nearest natural wetland (protected area with shelter role for waterbirds) |
| Land Cover (Measured at 100 m, 1000 m, and Sub-Basin Scale) | |
| NDW | Natural dense wooded (Acacia sp., Pinus sp.) |
| NCW | Natural clear wooded (Acacia sp., Pinus sp.) |
| NDS | Natural dense scrub (saline steppe and saltmarsh scrub) |
| NCS | Natural clear scrub (saline steppe and saltmarsh scrub) |
| DCW | Dry arboreal crops |
| DCS | Dry herbaceous crops |
| ICW | Irrigated arboreal crops (orchards) |
| ICS | Irrigated herbaceous and horticultural crops |
| UNP | Unproductive (urban areas) |
| WBS | Water bodies (different pond types) |
| Annual | Winter | Summer | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Variables | Season | Year | Sampling Station | Band | Post Hoc “Band” | Year | Sampling Station | Band | Post Hoc “Band” |
| Total Bird Use | <2.26 × 10−16 | 6.30 × 10−3 | 0.00078 | 4.30 × 10−3 | B1, B4 vs. B2, B3 | n.s. | 0.04 | 1.88 ×10−7 | B1 vs. B2 to B4 |
| Richness | <2.26 × 10−16 | 2.55 × 10−2 | 0.0013 | 6.94 × 10−8 | B1 vs. B2 to B4 | 0.0411 | n.s. | 6.39 × 10−12 | B1 vs. B2 to B4 |
| Shannon–Wiener Diversity | <2.26 × 10−16 | 2.76 × 10−2 | 0.012 | 0.00087 | B1 vs. B2 to B4 | n.s. | n.s. | 1.0 × 10−13 | B1 vs. B2 to B4 |
| Group | Stations | Description | Indicator Species | IV Significance | Season | Influential Gradient |
|---|---|---|---|---|---|---|
| 1 | S15 | Natural area exposed to oceanic (=Mediterranean) influence. Patches of Pinus halepensis and water bodies (saltpans); absence of structural disturbing elements of anthropogenic origin | Larus genei | 0.021 | Winter | PC2-b100, PC3-b100, PC1_r1000, PC2_r1000, PC2-dist, PC3-dist |
| 2 | S1, S2 | Semi-natural area exposed to oceanic influence. Incipient urbanization; close to disturbing structural anthropogenic elements | Sterna sandvicensis | 0.035 | Winter | PC1-r1000, PC2-dist |
| 3 | S13, S14 | Transitional area (urban-agricultural) of internal shore, influenced by land-based disturbing functional elements (point and diffuse effluent discharges); close to important disturbing structural anthropogenic elements (e.g., airport) | Fulica atra | 0.036 | Summer | PC1-r1000, PC1-dist |
| 4 | S10, S12 | Inland shore with natural scrub (saline steppe, saltmarsh), influenced by land-based disturbing functional elements (point and diffuse effluent discharges); absence of disturbing structural anthropogenic elements. | Podiceps cristatus | 0.003 | Winter | PC1-b100, PC3-b100, PC2-r1000, PC1-dist |
| Sterna albifrons | 0.005 | Summer | ||||
| Larus michaellis | 0.034 | Summer | ||||
| Eggretta garzetta | 0.008 | Summer |
| Index | Season | Sampling Stations | Value | Influential Gradient |
|---|---|---|---|---|
| Bird Use | Winter | S12, S2 | Max | PC1b100, PC2r1000 |
| S4 | Min | PC1dist, PC2b100 | ||
| Summer | S12, S10 | Max | PC1b100, PC2r1000 | |
| S4 | Min | PC1dist, PC2b100 | ||
| Richness | Winter | S12, S1 | Max | PC2b100 |
| S3, S4 | Min | PC1r1000, PC3r1000, PC1dist | ||
| Shannon Diversity | Winter | S11, S13 | Max | PC1dist |
| S9, S3 | Min | PC1b100 |
| MODEL | Explained Deviance | Factor Classes Observations |
|---|---|---|
| Winter | ||
| Total Bird Use~Band + Year | 20.41% | (−)B2 (−)B3 (−)B4 |
| (−)Year2 (−)Year3 (−)Year4 | ||
| Richness~Band + Year + Ndsb100 − Ndsb1002 − Icwb100 | 39.92% | (−)B2 (−)B3 (−)B4 |
| (+)Year2 (+)Year3 (+)Year4 | ||
| Shannon Diversity~Band + Year | 9.16% | (−)B2 (−)B3 (−)B4 |
| (+)Year2 (+)Year3 (+)Year4 | ||
| Use of Podiceps cristatus~Band + Year + Ndsr1000 − Dmmi+ Dmmi2 | 57.42% | (+)B2 (+)B3 (+)B4 |
| (−)Year2 (+)Year3 (+)Year4 | ||
| Use of Larus genei~Band + Year + Ndwr1000 + Ncsb100 – Ncsb1002 | 81.8% | (−)B2 (−)B3 (−)B4 |
| (−)Year2 (−)Year3 (+)Year4 | ||
| Use of Sterna sandvicensis~Band + Year − Dcan | 44.39% | (−)B2 (−)B3 (−)B4 |
| (+)Year2 (+)Year3 (−)Year4 | ||
| Summer | ||
| Total Bird Use~Band + Year + Dcsal + Wbsr1000 − Wbsr10002 | 51.25% | (−)B2 (−)B3 (−)B4 |
| (+)Year2 | ||
| Use of Fulica atra~Band − Year + Ndsr1000 − Icsr1000 + Icsr10002 | 44.16% | (−)B2 (−)B3 (−)B4 |
| (+)Year2 | ||
| Use of Sterna albifrons~Band + Year − Dcwet + Dmmi + Ncsb100 | 89.53% | (−)B2 (−)B3 (−)B4 |
| (−)Year2 | ||
| Use of Larus michaellis~Band + Year + Ncsb100 | 36.98% | (−)B2 (−)B3 (−)B4 |
| (+)Year2 | ||
| Use of Eggretta garzetta~Band + Year + Ndsb100 + Ncsr1000 | 57.76% | (−)B2 (−)B3 (−)B4 |
| (−)Year2 | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farinós-Celdrán, P.; Robledano-Aymerich, F.; Carreño, M.F.; Martínez-López, J. Spatiotemporal Assessment of Littoral Waterbirds for Establishing Ecological Indicators of Mediterranean Coastal Lagoons. ISPRS Int. J. Geo-Inf. 2017, 6, 256. https://doi.org/10.3390/ijgi6080256
Farinós-Celdrán P, Robledano-Aymerich F, Carreño MF, Martínez-López J. Spatiotemporal Assessment of Littoral Waterbirds for Establishing Ecological Indicators of Mediterranean Coastal Lagoons. ISPRS International Journal of Geo-Information. 2017; 6(8):256. https://doi.org/10.3390/ijgi6080256
Chicago/Turabian StyleFarinós-Celdrán, Pablo, Francisco Robledano-Aymerich, María Francisca Carreño, and Javier Martínez-López. 2017. "Spatiotemporal Assessment of Littoral Waterbirds for Establishing Ecological Indicators of Mediterranean Coastal Lagoons" ISPRS International Journal of Geo-Information 6, no. 8: 256. https://doi.org/10.3390/ijgi6080256
APA StyleFarinós-Celdrán, P., Robledano-Aymerich, F., Carreño, M. F., & Martínez-López, J. (2017). Spatiotemporal Assessment of Littoral Waterbirds for Establishing Ecological Indicators of Mediterranean Coastal Lagoons. ISPRS International Journal of Geo-Information, 6(8), 256. https://doi.org/10.3390/ijgi6080256