The Molecular Mechanism of Epithelial–Mesenchymal Transition for Breast Carcinogenesis
Abstract
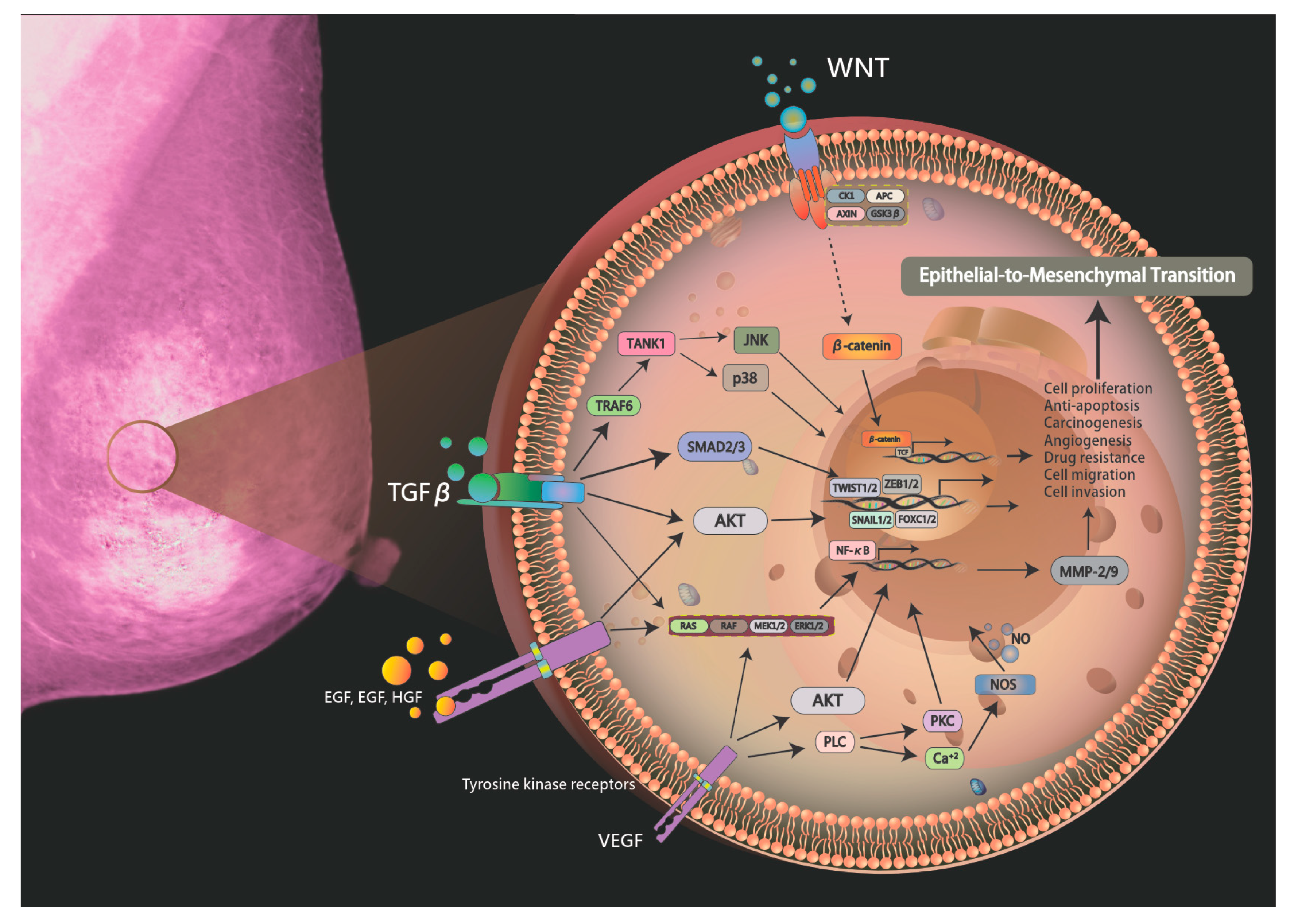
:1. Introduction
2. Epithelial-to-Mesenchymal Transition in Breast Cancer
3. The Role of the TGF-β Pathway in Breast Cancer
4. Relevant Regulatory Factors of EMT
5. Exosomes in EMT
6. Angiogenesis in EMT
7. ECM Remodeling during EMT
8. PI3K/AKT/mTOR Signaling in EMT
9. Tumor Microenvironment and EMT Formation
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barrios, C.H.; Reinert, T.; Werutsky, G. Access to High-Cost Drugs for Advanced Breast Cancer in Latin America, Particularly Trastuzumab. Ecancermedicalscience 2019, 13, 898. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.A.; Pinillos, L.; Villarreal-Garza, C.; Morante, Z.; Villaran, M.V.; Mejia, G.; Caglevic, C.; Aguilar, A.; Fajardo, W.; Usuga, F.; et al. Barriers in Latin America for the Management of Locally Advanced Breast Cancer. Ecancermedicalscience 2019, 13, 897. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.A.; Saravia, C.H.; Flores, C.; Araujo, J.M.; Martinez, D.; Schwarz, L.J.; Casas, A.; Bravo, L.; Zavaleta, J.; Chuima, B.; et al. Precision Medicine for Locally Advanced Breast Cancer: Frontiers and Challenges in Latin America. Ecancermedicalscience 2019, 13, 896. [Google Scholar] [CrossRef] [PubMed]
- Caparica, R.; Lambertini, M.; Ponde, N.; Fumagalli, D.; de Azambuja, E.; Piccart, M. Post-Neoadjuvant Treatment and the Management of Residual Disease in Breast Cancer: State of the Art and Perspectives. Ther. Adv. Med. Oncol. 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.; McCartney, A.; Risi, E.; Malorni, L.; Biganzoli, L.; Di Leo, A. Managing Advanced Hr-Positive, Her2-Negative Breast Cancer with Cdk4/6 Inhibitors in Post-Menopausal Patients: Is There a Best Sequence? Ther. Adv. Med. Oncol. 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Sorlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; et al. Gene Expression Patterns of Breast Carcinomas Distinguish Tumor Subclasses with Clinical Implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, T.O.; Hsu, F.D.; Jensen, K.; Cheang, M.; Karaca, G.; Hu, Z.; Hernandez-Boussard, T.; Livasy, C.; Cowan, D.; Dressler, L.; et al. Immunohistochemical and Clinical Characterization of the Basal-Like Subtype of Invasive Breast Carcinoma. Clin. Cancer Res. 2004, 10, 5367–5374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matos, I.; Dufloth, R.; Alvarenga, M.; Zeferino, L.C.; Schmitt, F. P63, Cytokeratin 5, and P-Cadherin: Three Molecular Markers to Distinguish Basal Phenotype in Breast Carcinomas. Virchows Arch. 2005, 447, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Van de Rijn, M.; Perou, C.M.; Tibshirani, R.; Haas, P.; Kallioniemi, O.; Kononen, J.; Torhorst, J.; Sauter, G.; Zuber, M.; Kochli, O.R.; et al. Expression of Cytokeratins 17 and 5 Identifies a Group of Breast Carcinomas with Poor Clinical Outcome. Am. J. Pathol. 2002, 161, 1991–1996. [Google Scholar] [CrossRef]
- Prat, A.; Parker, J.S.; Karginova, O.; Fan, C.; Livasy, C.; Herschkowitz, J.I.; He, X.; Perou, C.M. Phenotypic and Molecular Characterization of the Claudin-Low Intrinsic Subtype of Breast Cancer. Breast Cancer Res. 2010, 12, R68. [Google Scholar] [CrossRef] [PubMed]
- Dias, K.; Dvorkin-Gheva, A.; Hallett, R.M.; Wu, Y.; Hassell, J.; Pond, G.R.; Levine, M.; Whelan, T.; Bane, A.L. Claudin-Low Breast Cancer; Clinical & Pathological Characteristics. PLoS ONE 2017, 12, e0168669. [Google Scholar]
- Herschkowitz, J.I.; Simin, K.; Weigman, V.J.; Mikaelian, I.; Usary, J.; Hu, Z.; Rasmussen, K.E.; Jones, L.P.; Assefnia, S.; Chandrasekharan, S.; et al. Identification of Conserved Gene Expression Features between Murine Mammary Carcinoma Models and Human Breast Tumors. Genome Biol. 2007, 8, R76. [Google Scholar] [CrossRef] [PubMed]
- Pernas, S.; Tolaney, S.M. Her2-Positive Breast Cancer: New Therapeutic Frontiers and Overcoming Resistance. Ther. Adv. Med. Oncol. 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.M.; Brookes, C.L.; Robson, T.; van de Velde, C.J.; Billingham, L.J.; Campbell, F.M.; Grant, M.; Hasenburg, A.; Hille, E.T.; Kay, C.; et al. Estrogen Receptor and Progesterone Receptor as Predictive Biomarkers of Response to Endocrine Therapy: A Prospectively Powered Pathology Study in the Tamoxifen and Exemestane Adjuvant Multinational Trial. J. Clin. Oncol. 2011, 29, 1531–1538. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.C.; Chu, P.Y.; Liao, W.T.; Wu, M.Y.; Tsui, K.H.; Lin, L.T.; Huang, C.H.; Chen, L.L.; Li, C.J. Glycyrrhizic Acid Induces Human Mda-Mb-231 Breast Cancer Cell Death and Autophagy Via the Ros-Mitochondrial Pathway. Oncol. Rep. 2018, 39, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Baker, D.; Dijke, P.T. Tgf-Beta-Mediated Epithelial-Mesenchymal Transition and Cancer Metastasis. Int. J. Mol. Sci. 2019, 20, 2767. [Google Scholar] [CrossRef] [PubMed]
- Micalizzi, D.S.; Ford, H.L. Epithelial-Mesenchymal Transition in Development and Cancer. Future Oncol. 2009, 5, 1129–1143. [Google Scholar] [CrossRef] [PubMed]
- Felipe Lima, J.; Nofech-Mozes, S.; Bayani, J.; Bartlett, J.M. Emt in Breast Carcinoma—A Review. J. Clin. Med. 2016, 5, 65. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.C.; Li, C.J.; Yiang, G.T.; Tsai, A.P.; Wu, M.Y. Epithelial to Mesenchymal Transition and Cell Biology of Molecular Regulation in Endometrial Carcinogenesis. J. Clin. Med. 2019, 8, 439. [Google Scholar] [CrossRef]
- Chiu, H.C.; Wu, M.Y.; Li, C.H.; Huang, S.C.; Yiang, G.T.; Yen, H.S.; Liu, W.L.; Li, C.J.; Kao, W.Y. Epithelial-Mesenchymal Transition with Malignant Transformation Leading Multiple Metastasis from Disseminated Peritoneal Leiomyomatosis. J. Clin. Med. 2018, 7, 207. [Google Scholar] [CrossRef]
- Wu, M.Y.; Li, C.J.; Yiang, G.T.; Cheng, Y.L.; Tsai, A.P.; Hou, Y.T.; Ho, Y.C.; Hou, M.F.; Chu, P.Y. Molecular Regulation of Bone Metastasis Pathogenesis. Cell. Physiol. Biochem. 2018, 46, 1423–1438. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Bueno, G.; Portillo, F.; Cano, A. Transcriptional Regulation of Cell Polarity in Emt and Cancer. Oncogene 2008, 27, 6958–6969. [Google Scholar] [CrossRef] [PubMed]
- Kent, C.N.; Reed, I.K.G. Regulation of Epithelial-Mesenchymal Transition in Endometrial Cancer: Connecting Pi3k, Estrogen Signaling, and Micrornas. Clin. Transl. Oncol. 2016, 18, 1056–1061. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Weinberg, R.A. The Basics of Epithelial-Mesenchymal Transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular Mechanisms of Epithelial-Mesenchymal Transition. Nat. Rev. Mol. Cell Biol. 2014, 15, 178–196. [Google Scholar] [CrossRef] [PubMed]
- Makker, A.; Goel, M.M. Tumor Progression, Metastasis, and Modulators of Epithelial-Mesenchymal Transition in Endometrioid Endometrial Carcinoma: An Update. Endocr. Relat. Cancer 2016, 23, R85–R111. [Google Scholar] [CrossRef] [PubMed]
- Al Saleh, S.; Al Mulla, F.; Luqmani, Y.A. Estrogen Receptor Silencing Induces Epithelial to Mesenchymal Transition in Human Breast Cancer Cells. PLoS ONE 2011, 6, e20610. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Brabletz, T.; Kang, Y.; Longmore, G.D.; Nieto, M.A.; Stanger, B.Z.; Yang, J.; Weinberg, R.A. Upholding a Role for Emt in Breast Cancer Metastasis. Nature 2017, 547, E1–E3. [Google Scholar] [CrossRef] [PubMed]
- Fischer, K.R.; Altorki, N.K.; Mittal, V.; Gao, D. Fischer Et Al. Reply. Nature 2017, 547, E5–E6. [Google Scholar] [CrossRef] [PubMed]
- Mani, S.A.; Guo, W.; Liao, M.J.; Eaton, E.N.; Ayyanan, A.; Zhou, A.Y.; Brooks, M.; Reinhard, F.; Zhang, C.C.; Shipitsin, M.; et al. The Epithelial-Mesenchymal Transition Generates Cells with Properties of Stem Cells. Cell 2008, 133, 704–715. [Google Scholar] [CrossRef] [Green Version]
- Hennessy, B.T.; Giordano, S.; Broglio, K.; Duan, Z.; Trent, J.; Buchholz, T.A.; Babiera, G.; Hortobagyi, G.N.; Valero, V. Biphasic Metaplastic Sarcomatoid Carcinoma of the Breast. Ann. Oncol. 2006, 17, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Vega, S.; Morales, A.V.; Ocana, O.H.; Valdes, F.; Fabregat, I.; Nieto, M.A. Snail Blocks the Cell Cycle and Confers Resistance to Cell Death. Genes Dev. 2004, 18, 1131–1143. [Google Scholar] [CrossRef] [PubMed]
- Medici, D.; Hay, E.D.; Olsen, B.R. Snail and Slug Promote Epithelial-Mesenchymal Transition through Beta-Catenin-T-Cell Factor-4-Dependent Expression of Transforming Growth Factor-Beta3. Mol. Biol. Cell 2008, 19, 4875–4887. [Google Scholar] [CrossRef] [PubMed]
- Stemmler, M.P.; Eccles, R.L.; Brabletz, S.; Brabletz, T. Non-Redundant Functions of Emt Transcription Factors. Nat. Cell Biol. 2019, 21, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Di Gennaro, A.; Damiano, V.; Brisotto, G.; Armellin, M.; Perin, T.; Zucchetto, A.; Guardascione, M.; Spaink, H.P.; Doglioni, C.; Snaar-Jagalska, B.E.; et al. A P53/Mir-30a/Zeb2 Axis Controls Triple Negative Breast Cancer Aggressiveness. Cell Death Differ. 2018, 25, 2165–2180. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.Q.; Ma, C.; Wang, Q.; Song, Y.; Lv, T. The Role of Twist1 in Epithelial-Mesenchymal Transition and Cancers. Tumour Biol. 2016, 37, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Jiang, S.; Cheng, Y.; Li, T.; Hu, W.; Ma, Z.; Chen, F.; Yang, Y. Foxc1 in Cancer Development and Therapy: Deciphering Its Emerging and Divergent Roles. Ther. Adv. Med. Oncol. 2017, 9, 797–816. [Google Scholar] [CrossRef] [PubMed]
- Hollier, B.G.; Tinnirello, A.A.; Werden, S.J.; Evans, K.W.; Taube, J.H.; Sarkar, T.R.; Sphyris, N.; Shariati, M.; Kumar, S.V.; Battula, V.L.; et al. Foxc2 Expression Links Epithelial-Mesenchymal Transition and Stem Cell Properties in Breast Cancer. Cancer Res. 2013, 73, 1981–1992. [Google Scholar] [CrossRef] [PubMed]
- Radisky, D.C. Defining a Role for the Homeoprotein Six1 in Emt and Mammary Tumorigenesis. J. Clin. Investig. 2009, 119, 2528–2531. [Google Scholar] [CrossRef]
- Yu, M.; Smolen, G.A.; Zhang, J.; Wittner, B.; Schott, B.J.; Brachtel, E.; Ramaswamy, S.; Maheswaran, S.; Haber, D.A. A Developmentally Regulated Inducer of Emt, Lbx1, Contributes to Breast Cancer Progression. Genes Dev. 2009, 23, 1737–1742. [Google Scholar] [CrossRef] [PubMed]
- Christofori, G. Changing Neighbours, Changing Behaviour: Cell Adhesion Molecule-Mediated Signalling During Tumour Progression. EMBO J. 2003, 22, 2318–2323. [Google Scholar] [CrossRef] [PubMed]
- Hazan, R.B.; Qiao, R.; Keren, R.; Badano, I.; Suyama, K. Cadherin Switch in Tumor Progression. Ann. N. Y. Acad. Sci. 2004, 1014, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Mrozik, K.M.; Blaschuk, O.W.; Cheong, C.M.; Zannettino, A.C.W.; Vandyke, K. N-Cadherin in Cancer Metastasis, Its Emerging Role in Haematological Malignancies and Potential as a Therapeutic Target in Cancer. BMC Cancer 2018, 18, 939. [Google Scholar] [CrossRef] [PubMed]
- Wendt, M.K.; Allington, T.M.; Schiemann, W.P. Mechanisms of the Epithelial-Mesenchymal Transition by Tgf-Beta. Future Oncol. 2009, 5, 1145–1168. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Hu, R.; Sullivan, C.; Swanson, R.J.; Oehninger, S.; Sun, Y.P.; Bocca, S. Mfge8 Regulates Tgf-Beta-Induced Epithelial Mesenchymal Transition in Endometrial Epithelial Cells in Vitro. Reproduction 2016, 152, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Bokhari, A.A.; Lee, L.R.; Raboteau, D.; Hamilton, C.A.; Maxwell, G.L.; Rodriguez, G.C.; Syed, V. Progesterone Inhibits Endometrial Cancer Invasiveness by Inhibiting the Tgfbeta Pathway. Cancer Prev. Res. 2014, 7, 1045–1055. [Google Scholar] [CrossRef] [PubMed]
- Izzi, L.; Attisano, L. Ubiquitin-Dependent Regulation of Tgfbeta Signaling in Cancer. Neoplasia 2006, 8, 677–688. [Google Scholar] [CrossRef]
- Viloria-Petit, A.M.; David, L.; Jia, J.Y.; Erdemir, T.; Bane, A.L.; Pinnaduwage, D.; Roncari, L.; Narimatsu, M.; Bose, R.; Moffat, J.; et al. A Role for the Tgfβ-Par6 Polarity Pathway in Breast Cancer Progression. Proc. Natl. Acad. Sci. USA 2009, 106, 14028–14033. [Google Scholar] [CrossRef]
- Liu, F.; Gu, L.N.; Shan, B.E.; Geng, C.Z.; Sang, M.X. Biomarkers for Emt and Met in Breast Cancer: An Update. Oncol. Lett. 2016, 12, 4869–4876. [Google Scholar] [CrossRef]
- Huang, F.; Shi, Q.; Li, Y.; Xu, L.; Xu, C.; Chen, F.; Wang, H.; Liao, H.; Chang, Z.; Liu, F.; et al. Her2/Egfr-Akt Signaling Switches Tgf-Β from Inhibiting Cell Proliferation to Promoting Cell Migration in Breast Cancer. Cancer Res. 2018, 78, 6073–6085. [Google Scholar] [CrossRef]
- Sundqvist, A.; Morikawa, M.; Ren, J.; Vasilaki, E.; Kawasaki, N.; Kobayashi, M.; Koinuma, D.; Aburatani, H.; Miyazono, K.; Heldin, C.H.; et al. Junb Governs a Feed-Forward Network of Tgfβ Signaling That Aggravates Breast Cancer Invasion. Nucleic Acids Res. 2018, 46, 1180–1195. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.Y.; Yiang, G.T.; Cheng, P.W.; Chu, P.Y.; Li, C.J. Molecular Targets in Hepatocarcinogenesis and Implications for Therapy. J. Clin. Med. 2018, 7, 213. [Google Scholar] [CrossRef] [PubMed]
- Eger, A.; Stockinger, A.; Park, J.; Langkopf, E.; Mikula, M.; Gotzmann, J.; Mikulits, W.; Beug, H.; Foisner, R. Beta-Catenin and Tgfbeta Signalling Cooperate to Maintain a Mesenchymal Phenotype after Foser-Induced Epithelial to Mesenchymal Transition. Oncogene 2004, 23, 2672–2680. [Google Scholar] [CrossRef]
- Brabletz, T.; Kalluri, R.; Nieto, M.A.; Weinberg, R.A. Emt in Cancer. Nat. Rev. Cancer 2018, 18, 128–134. [Google Scholar] [CrossRef]
- Onder, T.T.; Gupta, P.B.; Mani, S.A.; Yang, J.; Lander, E.S.; Weinberg, R.A. Loss of E-Cadherin Promotes Metastasis Via Multiple Downstream Transcriptional Pathways. Cancer Res. 2008, 68, 3645–3654. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Gomez, S.J.; Maziveyi, M.; Alahari, S.K. Regulation of Epithelial-Mesenchymal Transition through Epigenetic and Post-Translational Modifications. Mol. Cancer 2016, 15, 18. [Google Scholar] [CrossRef]
- Drasin, D.J.; Robin, T.P.; Ford, H.L. Breast Cancer Epithelial-to-Mesenchymal Transition: Examining the Functional Consequences of Plasticity. Breast Cancer Res. 2011, 13, 226. [Google Scholar] [CrossRef]
- Demirkan, B. The Roles of Epithelial-to-Mesenchymal Transition (Emt) and Mesenchymal-to-Epithelial Transition (Met) in Breast Cancer Bone Metastasis: Potential Targets for Prevention and Treatment. J. Clin. Med. 2013, 2, 264–282. [Google Scholar] [CrossRef]
- Zhang, J.; Liang, Q.; Lei, Y.; Yao, M.; Li, L.; Gao, X.; Feng, J.; Zhang, Y.; Gao, H.; Liu, D.X.; et al. Sox4 Induces Epithelial-Mesenchymal Transition and Contributes to Breast Cancer Progression. Cancer Res. 2012, 72, 4597–4608. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, N.; Tiwari, V.K.; Waldmeier, L.; Balwierz, P.J.; Arnold, P.; Pachkov, M.; Meyer-Schaller, N.; Schubeler, D.; van Nimwegen, E.; Christofori, G. Sox4 Is a Master Regulator of Epithelial-Mesenchymal Transition by Controlling Ezh2 Expression and Epigenetic Reprogramming. Cancer Cell 2013, 23, 768–783. [Google Scholar] [CrossRef] [Green Version]
- Huang, W.; Pal, A.; Kleer, C.G. On How Ccn6 Suppresses Breast Cancer Growth and Invasion. J. Cell Commun. Signal. 2012, 6, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.Y.; Ju, F.; Wang, Z.H.; Ma, X.Z.; Zhao, H. Ing5 Inhibits Epithelial-Mesenchymal Transition in Breast Cancer by Suppressing Pi3k/Akt Pathway. Int. J. Clin. Exp. Med. 2015, 8, 15498–15505. [Google Scholar] [PubMed]
- Nagaoka, K.; Fujii, K.; Zhang, H.; Usuda, K.; Watanabe, G.; Ivshina, M.; Richter, J.D. Cpeb1 Mediates Epithelial-to-Mesenchyme Transition and Breast Cancer Metastasis. Oncogene 2016, 35, 2893–2901. [Google Scholar] [CrossRef] [PubMed]
- Zhan, T.; Rindtorff, N.; Boutros, M. Wnt Signaling in Cancer. Oncogene 2017, 36, 1461–1473. [Google Scholar] [CrossRef] [PubMed]
- Scheel, C.; Eaton, E.N.; Li, S.H.; Chaffer, C.L.; Reinhardt, F.; Kah, K.J.; Bell, G.; Guo, W.; Rubin, J.; Richardson, A.L.; et al. Paracrine and Autocrine Signals Induce and Maintain Mesenchymal and Stem Cell States in the Breast. Cell 2011, 145, 926–940. [Google Scholar] [CrossRef] [Green Version]
- Davis, F.M.; Stewart, T.A.; Thompson, E.W.; Monteith, G.R. Targeting Emt in Cancer: Opportunities for Pharmacological Intervention. Trends Pharmacol. Sci. 2014, 35, 479–488. [Google Scholar] [CrossRef]
- Saitoh, M. Involvement of Partial Emt in Cancer Progression. J. Biochem. 2018, 164, 257–264. [Google Scholar] [CrossRef]
- Micalizzi, D.S.; Farabaugh, S.M.; Ford, H.L. Epithelial-Mesenchymal Transition in Cancer: Parallels between Normal Development and Tumor Progression. J. Mammary Gland Biol. Neoplasia 2010, 15, 117–134. [Google Scholar] [CrossRef]
- Hesari, A.; Moghadam, S.A.G.; Siasi, A.; Rahmani, M.; Behboodi, N.; Rastgar-Moghadam, A.; Ferns, G.A.; Ghasemi, F.; Avan, A. Tumor-Derived Exosomes: Potential Biomarker or Therapeutic Target in Breast Cancer? J. Cell. Biochem. 2018, 119, 4236–4240. [Google Scholar] [CrossRef]
- Rashed, M.H.; Bayraktar, E.; Helal, G.K.; Abd-Ellah, M.F.; Amero, P.; Chavez-Reyes, A.; Rodriguez-Aguayo, C. Exosomes: From Garbage Bins to Promising Therapeutic Targets. Int. J. Mol. Sci. 2017, 18, 538. [Google Scholar] [CrossRef]
- Harris, D.A.; Patel, S.H.; Gucek, M.; Hendrix, A.; Westbroek, W.; Taraska, J.W. Exosomes Released from Breast Cancer Carcinomas Stimulate Cell Movement. PLoS ONE 2015, 10, e0117495. [Google Scholar] [CrossRef] [PubMed]
- Menck, K.; Scharf, C.; Bleckmann, A.; Dyck, L.; Rost, U.; Wenzel, D.; Dhople, V.M.; Siam, L.; Pukrop, T.; Binder, C.; et al. Tumor-Derived Microvesicles Mediate Human Breast Cancer Invasion through Differentially Glycosylated Emmprin. J. Mol. Cell Biol. 2015, 7, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.M.; O’Neill, P.; Jin, W.; Foty, R.; Medina, D.J.; Xu, Z.; Lomas, M.; Arndt, G.M.; Tang, Y.; Nakada, M.; et al. Extracellular Matrix Metalloproteinase Inducer (Cd147) Confers Resistance of Breast Cancer Cells to Anoikis through Inhibition of Bim. J. Biol. Chem. 2006, 281, 9719–9727. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.; Tian, H.; Li, X.; Jin, D.; Kong, J.; Yang, C.; Yang, X.; Lu, Y.; Luo, Y.; Lin, B.; et al. Clinical Application of a Microfluidic Chip for Immunocapture and Quantification of Circulating Exosomes to Assist Breast Cancer Diagnosis and Molecular Classification. PLoS ONE 2017, 12, e0175050. [Google Scholar] [CrossRef] [PubMed]
- Sankpal, N.V.; Fleming, T.P.; Sharma, P.K.; Wiedner, H.J.; Gillanders, W.E. A Double-Negative Feedback Loop between Epcam and Erk Contributes to the Regulation of Epithelial-Mesenchymal Transition in Cancer. Oncogene 2017, 36, 3706–3717. [Google Scholar] [CrossRef] [PubMed]
- Conigliaro, A.; Cicchini, C. Exosome-Mediated Signaling in Epithelial to Mesenchymal Transition and Tumor Progression. J. Clin. Med. 2018, 8, 26. [Google Scholar] [CrossRef] [PubMed]
- Perrot-Applanat, M.; Di Benedetto, M. Autocrine Functions of Vegf in Breast Tumor Cells: Adhesion, Survival, Migration and Invasion. Cell Adhes. Migr. 2012, 6, 547–553. [Google Scholar] [CrossRef]
- Liang, L.; Yue, Z.; Du, W.; Li, Y.; Tao, H.; Wang, D.; Wang, R.; Huang, Z.; He, N.; Xie, X.; et al. Molecular Imaging of Inducible Vegf Expression and Tumor Progression in a Breast Cancer Model. Cell Physiol. Biochem. 2017, 42, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Luo, G.; Tang, H.; Cheng, C.; Wang, P. Prognostic Significance of High Vegf-C Expression for Patients with Breast Cancer: An Update Meta Analysis. PLoS ONE 2016, 11, e0165725. [Google Scholar] [CrossRef]
- Eroglu, A.; Ersoz, C.; Karasoy, D.; Sak, S. Vascular Endothelial Growth Factor (Vegf)-C, Vegf-D, Vegfr-3 and D2-40 Expressions in Primary Breast Cancer: Association with Lymph Node Metastasis. Adv. Clin. Exp. Med. 2017, 26, 245–249. [Google Scholar]
- Radisky, E.S.; Radisky, D.C. Matrix Metalloproteinases as Breast Cancer Drivers and Therapeutic Targets. Front. Biosci. (Landmark Ed.) 2015, 20, 1144–1163. [Google Scholar] [CrossRef] [PubMed]
- Zucker, S.; Hymowitz, M.; Rollo, E.E.; Mann, R.; Conner, C.E.; Cao, J.; Foda, H.D.; Tompkins, D.C.; Toole, B.P. Tumorigenic Potential of Extracellular Matrix Metalloproteinase Inducer. Am. J. Pathol. 2001, 158, 1921–1928. [Google Scholar] [CrossRef] [Green Version]
- Tester, A.M.; Waltham, M.; Oh, S.J.; Bae, S.N.; Bills, M.M.; Walker, E.C.; Kern, F.G.; Stetler-Stevenson, W.G.; Lippman, M.E.; Thompson, E.W. Pro-Matrix Metalloproteinase-2 Transfection Increases Orthotopic Primary Growth and Experimental Metastasis of Mda-Mb-231 Human Breast Cancer Cells in Nude Mice. Cancer Res. 2004, 64, 652–658. [Google Scholar] [CrossRef] [PubMed]
- Yousef, E.M.; Tahir, M.R.; St-Pierre, Y.; Gaboury, L.A. Mmp-9 Expression Varies According to Molecular Subtypes of Breast Cancer. BMC Cancer 2014, 14, 609. [Google Scholar] [CrossRef]
- Katunina, A.I.; Gershtein, E.S.; Ermilova, V.D.; Tereshkina, I.V.; Nazarenko, A.Y.; Tyleuova, A.A.; Dvorova, E.K.; Karabekova, Z.K.; Gritskevich, M.V.; Berezov, T.T. Matrix Metalloproteinases 2, 7, and 9 in Tumors and Sera of Patients with Breast Cancer. Bull. Exp. Biol. Med. 2011, 151, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Teng, X.D.; Guo, X.X.; Li, Z.G.; Han, J.G.; Yao, L. Expression of Tissue Levels of Matrix Metalloproteinases and Their Inhibitors in Breast Cancer. Breast 2013, 22, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Remacle, A.; McCarthy, K.; Noel, A.; Maguire, T.; McDermott, E.; O’Higgins, N.; Foidart, J.M.; Duffy, M.J. High Levels of Timp-2 Correlate with Adverse Prognosis in Breast Cancer. Int. J. Cancer 2000, 89, 118–121. [Google Scholar] [CrossRef]
- Papa, A.; Pandolfi, P.P. The Pten(-)Pi3k Axis in Cancer. Biomolecules 2019, 9, 153. [Google Scholar] [CrossRef]
- Schild, C.; Wirth, M.; Reichert, M.; Schmid, R.M.; Saur, D.; Schneider, G. Pi3k Signaling Maintains C-Myc Expression to Regulate Transcription of E2f1 in Pancreatic Cancer Cells. Mol. Carcinog. 2009, 48, 1149–1158. [Google Scholar] [CrossRef]
- Kang, S.; Bader, A.G.; Vogt, P.K. Phosphatidylinositol 3-Kinase Mutations Identified in Human Cancer Are Oncogenic. Proc. Natl. Acad. Sci. USA 2005, 102, 802–807. [Google Scholar] [CrossRef]
- Carnero, A.; Paramio, J.M. The Pten/Pi3k/Akt Pathway in Vivo, Cancer Mouse Models. Front. Oncol. 2014, 4, 252. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Angulo, A.M.; Ferrer-Lozano, J.; Stemke-Hale, K.; Sahin, A.; Liu, S.; Barrera, J.A.; Burgues, O.; Lluch, A.M.; Chen, H.; Hortobagyi, G.N.; et al. Pi3k Pathway Mutations and Pten Levels in Primary and Metastatic Breast Cancer. Mol. Cancer Ther. 2011, 10, 1093–1101. [Google Scholar] [CrossRef] [PubMed]
- Khalil, B.D.; Hsueh, C.; Cao, Y.; Saab, W.F.A.; Wang, Y.; Condeelis, J.S.; Bresnick, A.R.; Backer, J.M. Gpcr Signaling Mediates Tumor Metastasis Via Pi3kbeta. Cancer Res. 2016, 76, 2944–2953. [Google Scholar] [CrossRef] [PubMed]
- Fresno Vara, J.A.; Casado, E.; de Castro, J.; Cejas, P.; Belda-Iniesta, C.; Gonzalez-Baron, M. Pi3k/Akt Signalling Pathway and Cancer. Cancer Treat. Rev. 2004, 30, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Laughner, E.; Taghavi, P.; Chiles, K.; Mahon, P.C.; Semenza, G.L. Her2 (Neu) Signaling Increases the Rate of Hypoxia-Inducible Factor 1alpha (Hif-1alpha) Synthesis: Novel Mechanism for Hif-1-Mediated Vascular Endothelial Growth Factor Expression. Mol. Cell Biol. 2001, 21, 3995–4004. [Google Scholar] [CrossRef] [PubMed]
- Li, C.J.; Liao, W.T.; Wu, M.Y.; Chu, P.Y. New Insights into the Role of Autophagy in Tumor Immune Microenvironment. Int. J. Mol. Sci. 2017, 18, 1566. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Xiong, G.; Trinkle, C.; Xu, R. Integrated Extracellular Matrix Signaling in Mammary Gland Development and Breast Cancer Progression. Histol. Histopathol. 2014, 29, 1083–1092. [Google Scholar] [PubMed]
- Lopez, J.I.; Kang, I.; You, W.K.; McDonald, D.M.; Weaver, V.M. In Situ Force Mapping of Mammary Gland Transformation. Integ.r Biol. 2011, 3, 910–921. [Google Scholar] [CrossRef]
- Walker, C.; Mojares, E.; Del Rio Hernandez, A. Role of Extracellular Matrix in Development and Cancer Progression. Int. J. Mol. Sci. 2018, 19, 3028. [Google Scholar] [CrossRef]
- Jung, H.Y.; Fattet, L.; Yang, J. Molecular Pathways: Linking Tumor Microenvironment to Epithelial-Mesenchymal Transition in Metastasis. Clin. Cancer Res. 2015, 21, 962–968. [Google Scholar] [CrossRef]
- Jing, Y.; Han, Z.; Zhang, S.; Liu, Y.; Wei, L. Epithelial-Mesenchymal Transition in Tumor Microenvironment. Cell Biosci. 2011, 1, 29. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, A.; Hassanian, S.M.; Khazaei, M.; Hasanzadeh, M.; Shahidsales, S.; Maftouh, M.; Ferns, G.A.; Avan, A. The Therapeutic Potential of Targeting Tumor Microenvironment in Breast Cancer: Rational Strategies and Recent Progress. J. Cell. Biochem. 2018, 119, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Julien, S.; Puig, I.; Caretti, E.; Bonaventure, J.; Nelles, L.; van Roy, F.; Dargemont, C.; de Herreros, A.G.; Bellacosa, A.; Larue, L. Activation of Nf-Kappab by Akt Upregulates Snail Expression and Induces Epithelium Mesenchyme Transition. Oncogene 2007, 26, 7445–7456. [Google Scholar] [CrossRef] [PubMed]
- Chua, H.L.; Bhat-Nakshatri, P.; Clare, S.E.; Morimiya, A.; Badve, S.; Nakshatri, H. Nf-Kappab Represses E-Cadherin Expression and Enhances Epithelial to Mesenchymal Transition of Mammary Epithelial Cells: Potential Involvement of Zeb-1 and Zeb-2. Oncogene 2007, 26, 711–724. [Google Scholar] [CrossRef] [PubMed]
| Family | Transcription Factor | Role | Ref. |
|---|---|---|---|
| Zinc-finger domain | SNAIL | Snail blocks the cell cycle and confers resistance to cell death. | [32] |
| SLUG | Downregulation of E-cadherin expression occurs during the EMT, a process also exploited by invasive cancer cells. | [33] | |
| ZEB1 | Represses E-cadherin promoter and induces EMT by recruiting SMARCA4/BRG1. | [34] | |
| ZEB2 | ZEB2 protein is involved in chemical signaling pathways that regulate early growth and development. | [35] | |
| bHLH | TWIST1 | Overexpression of TWIST1 induces EMT, a key process in the metastasis formation of cancer. | [36] |
| FOX | FOXC1 | FOXC1 partially promotes tumor metastasis by regulating EMT programs to support microvascular invasion, thereby increasing angiogenesis. | [37] |
| FOXC2 | Transcriptional activator that are upregulated in breast cancer. | [38] | |
| Homeobox | SIX1 | Six1 can promote the metastasis of human tumors, and the increased expression of Six1 can be used as an indicator for predicting breast cancer metastasis. | [39] |
| LBX1 | LBX1 is upregulated in the unfavorable estrogen receptor (ER)/progesterone (PR)/HER2 triple-negative basal-like subtype. | [40] | |
| cadherin | E-cadherin | E-cadherin an active suppressor of invasion and growth of many epithelial cancers. | [41,42] |
| N-cadherin | It is dependent on its association with the actin-cytoskeleton and is mediated through interactions with catenin proteins. | [43] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, C.-J.; Chu, P.-Y.; Yiang, G.-T.; Wu, M.-Y. The Molecular Mechanism of Epithelial–Mesenchymal Transition for Breast Carcinogenesis. Biomolecules 2019, 9, 476. https://doi.org/10.3390/biom9090476
Li C-J, Chu P-Y, Yiang G-T, Wu M-Y. The Molecular Mechanism of Epithelial–Mesenchymal Transition for Breast Carcinogenesis. Biomolecules. 2019; 9(9):476. https://doi.org/10.3390/biom9090476
Chicago/Turabian StyleLi, Chia-Jung, Pei-Yi Chu, Giou-Teng Yiang, and Meng-Yu Wu. 2019. "The Molecular Mechanism of Epithelial–Mesenchymal Transition for Breast Carcinogenesis" Biomolecules 9, no. 9: 476. https://doi.org/10.3390/biom9090476
APA StyleLi, C.-J., Chu, P.-Y., Yiang, G.-T., & Wu, M.-Y. (2019). The Molecular Mechanism of Epithelial–Mesenchymal Transition for Breast Carcinogenesis. Biomolecules, 9(9), 476. https://doi.org/10.3390/biom9090476







