Electrically Conductive Materials: Opportunities and Challenges in Tissue Engineering
Abstract
1. Introduction
2. Conductive Materials in Tissue Engineering
2.1. Polypyrrole
2.2. Polyaniline
2.3. Poly (3, 4-ethylenedioxythiophene)
2.4. Polythiophene
2.5. Carbon
2.6. Silicon
2.7. Gold
2.8. Melanin
2.9. Calcium Titanate
3. Application of Conductive Materials in Tissue Engineering
3.1. Nerve Tissue Engineering
3.2. Cardiovascular Tissue Engineering
3.3. Bone Tissue Engineering
3.4. Muscular Tissue Engineering
4. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ECM | extracellular matrix |
| SCI | spinal cord injuries |
| NDD | neurodegenerative diseases |
| CM | cardiomyocyte |
| MI | Myocardial infarction |
| hASCs | human adipose stem cells |
| MSC | mesenchymal stem cell |
| NGF | Nerve growth factor |
| BDNF | Brain-derived neurotrophic factor |
| CP | conductive polymer |
| PPy | polypyrrole |
| PANi | polyaniline |
| XCA | xanthan hydrogels |
| PLA | poly(lactic acid) |
| PEDOT | poly (3, 4-ethylenedioxythiophene) |
| PLLA | poly(L-lactic acid) |
| PDLLA | poly (d,l-lactide) |
| CNT | carbon nanotube |
| SWNT | single wall carbon nanotube |
| MWNT | multiple wall carbon nanotube |
| CNFs | carbon nanofibers |
| 3D-GFs | three-dimensional graphene foams |
| PCL | polycaprolactone |
| AuNPs | Gold nanoparticles |
| SiNWs | Silicon nanowires |
| HA | hyaluronic acid |
| PNIPAAm | poly (N-isopropylacrylamide) |
| SGH | self-supporting graphene hydrogel |
| PABS | poly (aminobenzene sulfonic acid) |
References
- Liu, Y.; Lim, J.; Teoh, S.-H. Review: Development of clinically relevant scaffolds for vascularised bone tissue engineering. Biotechnol. Adv. 2013, 31, 688–705. [Google Scholar] [CrossRef] [PubMed]
- Farokhi, M.; Mottaghitalab, F.; Samani, S.; Shokrgozar, M.A.; Kundu, S.C.; Reis, R.L.; Fattahi, Y.; Kaplan, D.L. Silk fibroin/hydroxyapatite composites for bone tissue engineering. Biotechnol. Adv. 2018, 36, 68–91. [Google Scholar] [CrossRef] [PubMed]
- Nilforoushzadeh, M.A.; Zare, M.; Zarrintaj, P.; Alizadeh, E.; Taghiabadi, E.; Heidari-Kharaji, M.; Amirkhani, M.A.; Saeb, M.R.; Mozafari, M. Engineering the niche for hair regeneration–a critical review. Nanomed. Nanotechnol. Biol. Med. 2018, 15, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Mozafari, M. Nanotechnology and Nanomedicine: Start small, think big. Mater. Today Proc. 2018, 5, 15492–15500. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Moghaddam, A.S.; Manouchehri, S.; Atoufi, Z.; Amiri, A.; Amirkhani, M.A.; Nilforoushzadeh, M.A.; Saeb, M.R.; Hamblin, M.R.; Mozafari, M. Can regenerative medicine and nanotechnology combine to heal wounds? The search for the ideal wound dressing. Nanomedicine 2017, 12, 2403–2422. [Google Scholar] [CrossRef] [PubMed]
- Zarrintaj, P.; Jouyandeh, M.; Ganjali, M.R.; Hadavand, B.S.; Mozafari, M.; Sheiko, S.S.; Russell, A.J.; Vatankhah-Varnoosfaderani, M.; Gutiérrez, T.J.; Saeb, M.R. Thermo-sensitive polymers in medicine: A review. Eur. Polym. J. 2019, 117, 402–403. [Google Scholar] [CrossRef]
- Sefat, F.; Youseffi, M.; Khaghani, S.A.; Soon, C.F.; Javid, F. Effect of transforming growth factor-β3 on mono and multilayer chondrocytes. Cytokine 2016, 83, 118–126. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Bakhshandeh, B.; Saeb, M.R.; Sefat, F.; Rezaeian, I.; Ganjali, M.R.; Ramakrishna, S.; Mozafari, M. Oligoaniline-based Conductive Biomaterials for Tissue Engineering. Acta Biomater. 2018, 72, 16–34. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Ahmadi, Z.; Saeb, M.R.; Mozafari, M. Poloxamer-based stimuli-responsive biomaterials. Mater. Today Proc. 2018, 5, 15516–15523. [Google Scholar] [CrossRef]
- Farokhi, M.; Mottaghitalab, F.; Fatahi, Y.; Saeb, M.R.; Zarrintaj, P.; Kundu, S.C.; Khademhosseini, A. Silk fibroin scaffolds for common cartilage injuries: Possibilities for future clinical applications. Eur. Polym. J. 2019, 115, 251–267. [Google Scholar] [CrossRef]
- Fouladiha, H.; Marashi, S.-A.; Shokrgozar, M.A.; Farokhi, M.; Atashi, A. Applications of a metabolic network model of mesenchymal stem cells for controlling cell proliferation and differentiation. Cytotechnology 2018, 70, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Rahmati, M.; Pennisi, C.P.; Mobasheri, A.; Mozafari, M. Bioengineered Scaffolds for Stem Cell Applications in Tissue Engineering and Regenerative Medicine. Adv. Exp. Med. Biol. 2018, 3, 73–89. [Google Scholar]
- Touri, M.; Moztarzadeh, F.; Osman, N.A.A.; Dehghan, M.M.; Mozafari, M. 3D-printed biphasic calcium phosphate scaffolds coated with an oxygen generating system for enhancing engineered tissue survival. Mater. Sci. Eng. C 2018, 84, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Zarrintaj, P.; Manouchehri, S.; Ahmadi, Z.; Saeb, M.R.; Urbanska, A.M.; Kaplan, D.L.; Mozafari, M. Agarose-based biomaterials for tissue engineering. Carbohydr. Polym. 2018, 187, 66–84. [Google Scholar] [CrossRef] [PubMed]
- Kalantari, E.; Naghib, S.M.; Naimi-Jamal, M.R.; Aliahmadi, A.; Iravani, N.J.; Mozafari, M. Nanostructured monticellite for tissue engineering applications-Part I: Microstructural and physicochemical characteristics. Ceram. Int. 2018, 44, 12731–12738. [Google Scholar] [CrossRef]
- Naserzadeh, P.; Mortazavi, S.A.; Ashtari, K.; Salimi, A.; Farokhi, M.; Pourahmad, J. Evaluation of the toxicity effects of silk fibroin on human lymphocytes and monocytes. J. Biochem. Mol. Toxicol. 2018, 32, e22056. [Google Scholar] [CrossRef] [PubMed]
- Jazayeri, M.; Shokrgozar, M.A.; Haghighipour, N.; Bolouri, B.; Mirahmadi, F.; Farokhi, M. Effects of electromagnetic stimulation on gene expression of mesenchymal stem cells and repair of bone lesions. Cell J. 2017, 19, 34–44. [Google Scholar]
- Jazayeri, M.; Shokrgozar, M.A.; Haghighipour, N.; Mahdian, R.; Farrokhi, M.; Bonakdar, S.; Mirahmadi, F.; Abbariki, T.N. Evaluation of mechanical and chemical stimulations on osteocalcin and Runx2 expression in mesenchymal stem cells. Mol. Cell Biomech. 2015, 12, 197–213. [Google Scholar]
- Derakhshandeh, M.R.; Eshraghi, M.J.; Javaheri, M.; Khamseh, S.; Sari, M.G.; Zarrintaj, P.; Saeb, M.R.; Mozafari, M. Diamond-like carbon-deposited films: A new class of biocorrosion protective coatings. Surf. Innov. 2018, 6, 266–276. [Google Scholar] [CrossRef]
- Bassett, C.A.L.; Becker, R.O. Generation of electric potentials by bone in response to mechanical stress. Science 1962, 137, 1063–1064. [Google Scholar] [CrossRef]
- Prabhakaran, M.P.; Ghasemi-Mobarakeh, L.; Jin, G.; Ramakrishna, S. Electrospun conducting polymer nanofibers and electrical stimulation of nerve stem cells. J. Biosci. Bioeng. 2011, 112, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Ning, C.; Zhou, Z.; Tan, G.; Zhu, Y.; Mao, C. Electroactive polymers for tissue regeneration: Developments and perspectives. Prog. Polym. Sci. 2018, 81, 144–162. [Google Scholar] [CrossRef] [PubMed]
- Leppik, L.; Zhihua, H.; Mobini, S.; Parameswaran, V.T.; Eischen-Loges, M.; Slavici, A.; Helbing, J.; Pindur, L.; Oliveira, K.M.; Bhavsar, M.B. Combining electrical stimulation and tissue engineering to treat large bone defects in a rat model. Sci. Rep. 2018, 8, 6307. [Google Scholar] [CrossRef] [PubMed]
- Memic, A.; Aldhahri, M.; Tamayol, A.; Mostafalu, P.; Abdel-Wahab, M.S.; Samandari, M.; Moghaddam, K.M.; Annabi, N.; Bencherif, S.A.; Khademhosseini, A. Nanofibrous silver-coated polymeric scaffolds with tunable electrical properties. Nanomaterials 2017, 7, 63. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Wang, L.; Guo, B.; Shao, Y.; Ma, P.X. Electroactive biodegradable polyurethane significantly enhanced Schwann cells myelin gene expression and neurotrophin secretion for peripheral nerve tissue engineering. Biomaterials 2016, 87, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Zarrintaj, P.; Saeb, M.R.; Ramakrishna, S.; Mozafari, M. Biomaterials Selection for Neuroprosthetics. Curr. Opin. Biomed. Eng. 2018, 6, 99–109. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Ahmadi, Z.; Vahabi, H.; Ducos, F.; Saeb, M.R.; Mozafari, M. Polyaniline in retrospect and prospect. Mater. Today Proc. 2018, 5, 15852–15860. [Google Scholar] [CrossRef]
- Babanejad, N.; Nabid, M.R.; Farhadian, A.; Dorkoosh, F.; Zarrintaj, P.; Saeb, M.; Mozafari, M. Sustained delivery of olanzapine from sunflower oil-based polyol-urethane nanoparticles synthesized through a cyclic carbonate ring-opening reaction. IET Nanobiotechnol. 2019. [Google Scholar] [CrossRef]
- Guo, B.; Ma, P.X. Conducting polymers for tissue engineering. Biomacromolecules 2018, 19, 1764–1782. [Google Scholar] [CrossRef]
- Guo, B.; Glavas, L.; Albertsson, A.-C. Biodegradable and electrically conducting polymers for biomedical applications. Prog. Polym. Sci. 2013, 38, 1263–1286. [Google Scholar] [CrossRef]
- Hiltunen, M.; Pelto, J.; Ellä, V.; Kellomäki, M. Uniform and electrically conductive biopolymer-doped polypyrrole coating for fibrous PLA. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 1721–1729. [Google Scholar] [CrossRef] [PubMed]
- Jin, G.; Li, K. The electrically conductive scaffold as the skeleton of stem cell niche in regenerative medicine. Mater. Sci. Eng. C 2014, 45, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Collier, J.H.; Camp, J.P.; Hudson, T.W.; Schmidt, C.E. Synthesis and characterization of polypyrrole–hyaluronic acid composite biomaterials for tissue engineering applications. J. Biomed. Mater. Res. 2000, 50, 574–584. [Google Scholar] [CrossRef]
- Breukers, R.; Gilmore, K.J.; Kita, M.; Wagner, K.; Higgins, M.; Moulton, S.; Clark, G.M.; Officer, D.; Kapsa, R.; Wallace, G. Creating conductive structures for cell growth: Growth and alignment of myogenic cell types on polythiophenes. J. Biomed. Mater. Res. Part A 2010, 95, 256–268. [Google Scholar] [CrossRef] [PubMed]
- Ferris, C.J. Conducting bio-materials based on gellan gum hydrogels. Soft Matter 2009, 5, 3430–3437. [Google Scholar] [CrossRef]
- You, J.-O.; Rafat, M.; Ye, G.J.; Auguste, D.T. Nanoengineering the heart: Conductive scaffolds enhance connexin 43 expression. Nano Lett. 2011, 11, 3643–3648. [Google Scholar] [CrossRef] [PubMed]
- Anand, J.; Palaniappan, S.; Sathyanarayana, D. Conducting polyaniline blends and composites. Prog. Polym. Sci. 1998, 23, 993–1018. [Google Scholar] [CrossRef]
- Li, M.; Guo, Y.; Wei, Y.; MacDiarmid, A.G.; Lelkes, P.I. Electrospinning polyaniline-contained gelatin nanofibers for tissue engineering applications. Biomaterials 2006, 27, 2705–2715. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.Y.; Langer, R.; Ingber, D.E. Electrically conducting polymers can noninvasively control the shape and growth of mammalian cells. Proc. Natl. Acad. Sci. USA 1994, 91, 3201–3204. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Shao, J.; Wang, Y.; Zhang, P.; Chen, X.; Wei, Y. PLA-PEG-PLA and Its Electroactive Tetraaniline Copolymer as Multi-interactive Injectable Hydrogels for Tissue Engineering. Biomacromolecules 2013, 14, 1904–1912. [Google Scholar] [CrossRef]
- Huang, K.; Niu, Y.; Wang, L.J.; Liu, Y.; Chen, J.S.; Wang, R.Z. pH-Induced Cross-Linking of Dopamine-Containing Block Copolymers with Fe3+ to Form Self-Healing Hydrogels. Adv. Mater. Res. 2012, 569, 11–14. [Google Scholar] [CrossRef]
- Lu, X.; Zhang, W.; Wang, C.; Wen, T.-C.; Wei, Y. One-dimensional conducting polymer nanocomposites: Synthesis, properties and applications. Prog. Polym. Sci. 2011, 36, 671–712. [Google Scholar] [CrossRef]
- Balint, R.; Cassidy, N.J.; Cartmell, S.H. Conductive polymers: Towards a smart biomaterial for tissue engineering. Acta Biomater. 2014, 10, 2341–2353. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.; Doherty, P. A preliminary assessment of poly (pyrrole) in nerve guide studies. J. Mater. Sci. Mater. Med. 1994, 5, 429–433. [Google Scholar] [CrossRef]
- Moroder, P.; Runge, M.B.; Wang, H.; Ruesink, T.; Lu, L.; Spinner, R.J.; Windebank, A.J.; Yaszemski, M.J. Material properties and electrical stimulation regimens of polycaprolactone fumarate–polypyrrole scaffolds as potential conductive nerve conduits. Acta Biomater. 2011, 7, 944–953. [Google Scholar] [CrossRef] [PubMed]
- Mozafari, M.; Vashaee, D.; Tayebi, L.; Mehraien, M. Electroconductive nanocomposite scaffolds: A new strategy into tissue engineering and regenerative medicine. In Nanocomposites—New Trends and Developments; Ebrahimi, F., Ed.; INTECH Open Access Publisher: London, UK, 2012. [Google Scholar]
- Derakhshandeh, M.R.; Eshraghi, M.J.; Hadavi, M.M.; Javaheri, M.; Khamseh, S.; Sari, M.G.; Zarrintaj, P.; Saeb, M.R.; Mozafari, M. Diamond-like carbon thin films prepared by pulsed-DC PE-CVD for biomedical applications. Surf. Innov. 2018, 6, 167–175. [Google Scholar] [CrossRef]
- Bendrea, A.-D.; Cianga, L.; Cianga, I. Review paper: Progress in the field of conducting polymers for tissue engineering applications. J. Biomater. Appl. 2011, 26, 3–84. [Google Scholar] [CrossRef]
- Ateh, D.; Navsaria, H.; Vadgama, P. Polypyrrole-based conducting polymers and interactions with biological tissues. J. R. Soc. Interface 2006, 3, 741–752. [Google Scholar] [CrossRef]
- Runge, M.B.; Dadsetan, M.; Baltrusaitis, J.; Knight, A.M.; Ruesink, T.; Lazcano, E.A.; Lu, L.; Windebank, A.J.; Yaszemski, M.J. The development of electrically conductive polycaprolactone fumarate–polypyrrole composite materials for nerve regeneration. Biomaterials 2010, 31, 5916–5926. [Google Scholar] [CrossRef]
- Moreno, J.S.; Panero, S.; Artico, M.; Filippini, P. Synthesis and characterization of new electroactive polypyrrole–chondroitin sulphate A substrates. Bioelectrochemistry 2008, 72, 3–9. [Google Scholar] [CrossRef]
- Shi, G.; Rouabhia, M.; Wang, Z.; Dao, L.H.; Zhang, Z. A novel electrically conductive and biodegradable composite made of polypyrrole nanoparticles and polylactide. Biomaterials 2004, 25, 2477–2488. [Google Scholar] [CrossRef] [PubMed]
- Bueno, V.B.; Takahashi, S.H.; Catalani, L.H.; de Torresi, S.I.C.; Petri, D.F.S. Biocompatible xanthan/polypyrrole scaffolds for tissue engineering. Mater. Sci. Eng. C 2015, 52, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, R.; Chu, H.K.; Sun, D. Design and characterization of a conductive nanostructured polypyrrole-polycaprolactone coated magnesium/PLGA composite for tissue engineering scaffolds. J. Biomed. Mater. Res. Part A 2015, 103, 2966–2973. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Guo, Y.; Ma, P.X.; Guo, B. Rapid thermal responsive conductive hybrid cryogels with shape memory properties, photothermal properties and pressure dependent conductivity. J. Colloid Interface Sci. 2018, 526, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Saeb, M.R.; Zarrintaj, P. Chapter 10—Polyaniline/graphene-based nanocomposites. In Fundamentals and Emerging Applications of Polyaniline; Mozafari, M., Chauhan, N.P.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 165–175. [Google Scholar]
- Saeb, M.R.; Zarrintaj, P.; Khandelwal, P.; Chauhan, N.P.S. Chapter 2—Synthetic route of polyaniline (I): Conventional oxidative polymerization. In Fundamentals and Emerging Applications of Polyaniline; Mozafari, M., Chauhan, N.P.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 17–41. [Google Scholar]
- Zarrintaj, P.; Yazdi, M.K.; Jouyandeh, M.; Saeb, M.R. Chapter 7—PANI-based nanostructures. In Fundamentals and Emerging Applications of Polyaniline; Mozafari, M., Chauhan, N.P.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 121–130. [Google Scholar]
- Zarrintaj, P.; Khalili, R.; Vahabi, H.; Saeb, M.R.; Ganjali, M.R.; Mozafari, M. Chapter 8—Polyaniline/metal oxides nanocomposites. In Fundamentals and Emerging Applications of Polyaniline; Mozafari, M., Chauhan, N.P.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 131–141. [Google Scholar]
- Zarrintaj, P.; Yazdi, M.K.; Vahabi, H.; Moghadam, P.N.; Saeb, M.R. Towards advanced flame retardant organic coatings: Expecting a new function from polyaniline. Prog. Org. Coat. 2019, 130, 144–148. [Google Scholar] [CrossRef]
- Ahmadi, Z.; Chauhan, N.P.S.; Zarrintaj, P.; Khiabani, A.B.; Saeb, M.R.; Mozafari, M. Chapter 13—Experimental procedures for assessing electrical and thermal conductivity of polyaniline. In Fundamentals and Emerging Applications of Polyaniline; Mozafari, M., Chauhan, N.P.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 227–258. [Google Scholar]
- Zarrintaj, P.; Saeb, M.R. Chapter 5—Synthetic route of polyaniline (IV): Irradiation path. In Fundamentals and Emerging Applications of Polyaniline; Mozafari, M., Chauhan, N.P.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 91–103. [Google Scholar]
- Yazdi, M.K.; Saeidi, H.; Zarrintaj, P.; Saeb, M.R.; Mozafari, M. Chapter 9—PANI-CNT nanocomposites. In Fundamentals and Emerging Applications of Polyaniline; Mozafari, M., Chauhan, N.P.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 143–163. [Google Scholar]
- Li, L.; Ge, J.; Guo, B.; Ma, P.X. In situ forming biodegradable electroactive hydrogels. Polym. Chem. 2014, 5, 2880–2890. [Google Scholar] [CrossRef]
- Rahman, N.A.; Feisst, V.; Dickinson, M.E.; Malmström, J.; Dunbar, P.R.; Travas-Sejdic, J. Functional polyaniline nanofibre mats for human adipose-derived stem cell proliferation and adhesion. Mater. Chem. Phys. 2013, 138, 333–341. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Vahabi, H.; Saeb, M.R.; Mozafari, M. Chapter 14—Application of polyaniline and its derivatives. In Fundamentals and Emerging Applications of Polyaniline; Mozafari, M., Chauhan, N.P.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 259–272. [Google Scholar]
- Zhao, X.; Li, P.; Guo, B.; Ma, P.X. Antibacterial and conductive injectable hydrogels based on quaternized chitosan-graft-polyaniline/oxidized dextran for tissue engineering. Acta Biomater. 2015, 26, 236–248. [Google Scholar] [CrossRef]
- Gizdavic-Nikolaidis, M.; Ray, S.; Bennett, J.R.; Easteal, A.J.; Cooney, R.P. Electrospun Functionalized Polyaniline Copolymer-Based Nanofibers with Potential Application in Tissue Engineering. Macromol. Biosci. 2010, 10, 1424–1431. [Google Scholar] [CrossRef]
- Hafshejani, T.M.; Zamanian, A.; Venugopal, J.R.; Rezvani, Z.; Sefat, F.; Saeb, M.R.; Vahabi, H.; Zarrintaj, P.; Mozafari, M. Antibacterial glass-ionomer cement restorative materials: A critical review on the current status of extended release formulations. J. Control. Release 2017, 262, 317–328. [Google Scholar] [CrossRef]
- McKeon-Fischer, K.; Freeman, J. Addition of conductive elements to polymeric scaffolds for muscle tissue engineering. Nano Life 2012, 2, 1230011. [Google Scholar] [CrossRef]
- Baheiraei, N.; Yeganeh, H.; Ai, J.; Gharibi, R.; Azami, M.; Faghihi, F. Synthesis, characterization and antioxidant activity of a novel electroactive and biodegradable polyurethane for cardiac tissue engineering application. Mater. Sci. Eng. C 2014, 44, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Choi, J.H.; Kim, S.; Jang, I.; Jeong, S.; Lee, J.Y. Micropatterned conductive hydrogels as multifunctional muscle-mimicking biomaterials: Graphene-incorporated hydrogels directly patterned with femtosecond laser ablation. Acta Biomater. 2019. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Qu, J.; Zhao, X.; Zhang, M. Degradable conductive self-healing hydrogels based on dextran-graft-tetraaniline and N-carboxyethyl chitosan as injectable carriers for myoblast cell therapy and muscle regeneration. Acta Biomater. 2019, 84, 180–193. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Dong, R.; Guo, B.; Ma, P.X. Dopamine-incorporated dual bioactive electroactive shape memory polyurethane elastomers with physiological shape recovery temperature, high stretchability, and enhanced C2C12 myogenic differentiation. ACS Appl. Mater. Interfaces 2017, 9, 29595–29611. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.I.; Jun, I.D.; Choi, M.J.; Nho, Y.C.; Lee, Y.M.; Shin, H. Development of Electroactive and Elastic Nanofibers that contain Polyaniline and Poly (L-lactide-co-ε-caprolactone) for the Control of Cell Adhesion. Macromol. Biosci. 2008, 8, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Bouten, C.V.C.; Dankers, P.Y.W.; Driessen-Mol, A.; Pedron, S.; Brizard, A.M.A.; Baaijens, F.P.T. Substrates for cardiovascular tissue engineering. Adv. Drug Deliv. Rev. 2011, 63, 221–241. [Google Scholar] [CrossRef]
- Guo, B.; Finne-Wistrand, A.; Albertsson, A.-C. Facile synthesis of degradable and electrically conductive polysaccharide hydrogels. Biomacromolecules 2011, 12, 2601–2609. [Google Scholar] [CrossRef]
- Guo, B.; Finne-Wistrand, A.; Albertsson, A.-C. Electroactive Hydrophilic Polylactide Surface by Covalent Modification with Tetraaniline. Macromolecules 2012, 45, 652–659. [Google Scholar] [CrossRef]
- Zhang, X.; Qi, H.; Wang, S.; Feng, L.; Ji, Y.; Tao, L.; Li, S.; Wei, Y. Cellular responses of aniline oligomers: A preliminary study. Toxicol. Res. 2012, 1, 201–205. [Google Scholar] [CrossRef]
- Qi, H.; Liu, M.; Xu, L.; Feng, L.; Tao, L.; Ji, Y.; Zhang, X.; Wei, Y. Biocompatibility evaluation of aniline oligomers with different end-functional groups. Toxicol. Res. 2013, 2, 427–433. [Google Scholar] [CrossRef]
- Xu, C.; Huang, Y.; Yepez, G.; Wei, Z.; Liu, F.; Bugarin, A.; Tang, L.; Hong, Y. Development of dopant-free conductive bioelastomers. Sci. Rep. 2016, 6, 34451. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Ge, J.; Li, Y.; Guo, B.; Ma, P.X. Nanofibrous electroactive scaffolds from a chitosan-grafted-aniline tetramer by electrospinning for tissue engineering. RSC Adv. 2014, 4, 13652–13661. [Google Scholar] [CrossRef]
- Kim, H.; Jeong, S.-M.; Park, J.-W. Electrical switching between vesicles and micelles via redox-responsive self-assembly of amphiphilic rod− coils. J. Am. Chem. Soc. 2011, 133, 5206–5209. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Yu, M.; Chen, X.; Nie, S.; Lai, W.Y.; Su, W.; Cui, Z.; Huang, W. Ito-Free Flexible Electronics: Screen-Printed Poly (3, 4-Ethylenedioxythiophene): Poly (Styrenesulfonate) Grids as ITO-Free Anodes for Flexible Organic Light-Emitting Diodes (Adv. Funct. Mater. 11/2018). Adv. Funct. Mater. 2018, 28, 1870072. [Google Scholar] [CrossRef]
- Povlich, L.K.; Feldman, K.E.; Shim, B.S.; Martin, D.C. 1.130-Electroactive Polymeric Biomaterials. In Comprehensive Biomaterials; Ducheyne, P., Ed.; Elsevier: Oxford, UK, 2011; pp. 547–561. [Google Scholar]
- Niu, X.; Rouabhia, M.; Chiffot, N.; King, M.W.; Zhang, Z. An electrically conductive 3D scaffold based on a nonwoven web of poly (l-lactic acid) and conductive poly (3, 4-ethylenedioxythiophene). J. Biomed. Mater. Res. Part A 2015, 103, 2635–2644. [Google Scholar] [CrossRef] [PubMed]
- Abidian, M.R.; Corey, J.M.; Kipke, D.R.; Martin, D.C. Conducting-polymer nanotubes improve electrical properties, mechanical adhesion, neural attachment, and neurite outgrowth of neural electrodes. Small 2010, 6, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Malliaras, G.; Abidian, M.R. Organic bioelectronic materials and devices. Adv. Mater. 2015, 27, 7492. [Google Scholar] [CrossRef] [PubMed]
- Mehmood, U.; Al-Ahmed, A.; Hussein, I.A. Review on recent advances in polythiophene based photovoltaic devices. Renew. Sustain. Energy Rev. 2016, 57, 550–561. [Google Scholar] [CrossRef]
- Schopf, G.; Kossmehl, G. Polythiophenes-Electrically Conductive Polymers; Springer: Berlin, Germany, 1997. [Google Scholar]
- Iyoda, M.; Shimizu, H. Multifunctional π-expanded oligothiophene macrocycles. Chem. Soc. Rev. 2015, 44, 6411–6424. [Google Scholar] [CrossRef]
- Hussain, S.T.; Abbas, F.; Kausar, A.; Khan, M.R. New polyaniline/polypyrrole/polythiophene and functionalized multiwalled carbon nanotube-based nanocomposites: Layer-by-layer in situ polymerization. High Perform. Polym. 2013, 25, 70–78. [Google Scholar] [CrossRef]
- Bitounis, D.; Ali-Boucetta, H.; Hong, B.H.; Min, D.H.; Kostarelos, K. Prospects and challenges of graphene in biomedical applications. Adv. Mater. 2013, 25, 2258–2268. [Google Scholar] [CrossRef]
- Goodarzi, S.; Da Ros, T.; Conde, J.; Sefat, F.; Mozafari, M. Fullerene: Biomedical engineers get to revisit an old friend. Mater. Today 2017, 20, 460–480. [Google Scholar] [CrossRef]
- Goenka, S.; Sant, V.; Sant, S. Graphene-based nanomaterials for drug delivery and tissue engineering. J. Control. Release 2014, 173, 75–88. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Guillot, S.; Dabboue, H.; Tranchant, J.-F.; Salvetat, J.-P. Carbon nanotubes as structural nanofibers for hyaluronic acid hydrogel scaffolds. Biomacromolecules 2008, 9, 505–509. [Google Scholar] [CrossRef]
- Lau, C.; Cooney, M.J.; Atanassov, P. Conductive macroporous composite chitosan− carbon nanotube scaffolds. Langmuir 2008, 24, 7004–7010. [Google Scholar] [CrossRef]
- Alasv, N.; Mozafari, M. Graphene-Proceed with Caution: What We Know, what We don’t. J. Clin. Toxicol. 2015, 5, E122. [Google Scholar]
- Li, Y.; Samad, Y.A.; Polychronopoulou, K.; Alhassan, S.M.; Liao, K. Highly Electrically Conductive Nanocomposites Based on PolymerInfused Graphene Sponges. Sci. Rep. 2014, 4, 4652. [Google Scholar] [CrossRef]
- Menaa, F.; Abdelghani, A.; Menaa, B. Graphene nanomaterials as biocompatible and conductive scaffolds for stem cells: Impact for tissue engineering and regenerative medicine. J. Tissue Eng. Regen. Med. 2015, 9, 1321–1338. [Google Scholar] [CrossRef]
- Song, H.S.; Kwon, O.S.; Kim, J.-H.; Conde, J.; Artzi, N. 3D hydrogel scaffold doped with 2D graphene materials for biosensors and bioelectronics. Biosens. Bioelectron. 2017, 89, 187–200. [Google Scholar] [CrossRef]
- Tran, P.A.; Zhang, L.; Webster, T.J. Carbon nanofibers and carbon nanotubes in regenerative medicine. Adv. Drug Deliv. Rev. 2009, 61, 1097–1114. [Google Scholar] [CrossRef]
- Edwards, S.L.; Werkmeister, J.A.; Ramshaw, J.A. Carbon nanotubes in scaffolds for tissue engineering. Expert Rev. Med. Devices 2009, 6, 499–505. [Google Scholar] [CrossRef]
- Mozafari, M. The Critical Impact of Controlled Drug Delivery in the Future of Tissue Engineering. Trends Biomater. Artif. Organs. 2014, 28. [Google Scholar]
- Hopley, E.L.; Salmasi, S.; Kalaskar, D.M.; Seifalian, A.M. Carbon nanotubes leading the way forward in new generation 3D tissue engineering. Biotechnol. Adv. 2014, 32, 1000–1014. [Google Scholar] [CrossRef]
- MacDonald, R.A.; Voge, C.M.; Kariolis, M.; Stegemann, J.P. Carbon nanotubes increase the electrical conductivity of fibroblast-seeded collagen hydrogels. Acta Biomater. 2008, 4, 1583–1592. [Google Scholar] [CrossRef]
- Upadhyayula, V.K.K.; Gadhamshetty, V. Appreciating the role of carbon nanotube composites in preventing biofouling and promoting biofilms on material surfaces in environmental engineering: A review. Biotechnol. Adv. 2010, 28, 802–816. [Google Scholar] [CrossRef]
- Edwards, S.L.; Church, J.S.; Werkmeister, J.A.; Ramshaw, J.A. Tubular micro-scale multiwalled carbon nanotube-based scaffolds for tissue engineering. Biomaterials 2009, 30, 1725–1731. [Google Scholar] [CrossRef]
- Cardiel, J.J.; Zhao, Y.; Kim, J.-H.; Chung, J.-H.; Shen, A.Q. Electro-conductive porous scaffold with single-walled carbon nanotubes in wormlike micellar networks. Carbon 2014, 80, 203–212. [Google Scholar] [CrossRef]
- Abarrategi, A.; Gutiérrez, M.C.; Moreno-Vicente, C.; Hortigüela, M.J.; Ramos, V.; López-Lacomba, J.L.; Ferrer, M.L.; del Monte, F. Multiwall carbon nanotube scaffolds for tissue engineering purposes. Biomaterials 2008, 29, 94–102. [Google Scholar] [CrossRef]
- Correa-Duarte, M.A.; Wagner, N.; Rojas-Chapana, J.; Morsczeck, C.; Thie, M.; Giersig, M. Fabrication and biocompatibility of carbon nanotube-based 3D networks as scaffolds for cell seeding and growth. Nano Lett. 2004, 4, 2233–2236. [Google Scholar] [CrossRef]
- Liao, H.; Qi, R.; Shen, M.; Cao, X.; Guo, R.; Zhang, Y.; Shi, X. Improved cellular response on multiwalled carbon nanotube-incorporated electrospun polyvinyl alcohol/chitosan nanofibrous scaffolds. Colloids Surf. B Biointerfaces 2011, 84, 528–535. [Google Scholar] [CrossRef]
- Mackle, J.N.; Blond, D.J.P.; Mooney, E.; McDonnell, C.; Blau, W.J.; Shaw, G.; Barry, F.P.; Murphy, J.M.; Barron, V. In vitro Characterization of an Electroactive Carbon-Nanotube-Based Nanofiber Scaffold for Tissue Engineering. Macromol. Biosci. 2011, 11, 1272–1282. [Google Scholar] [CrossRef]
- Vardharajula, S.; Ali, S.Z.; Tiwari, P.M.; Eroğlu, E.; Vig, K.; Dennis, V.A.; Singh, S.R. Functionalized carbon nanotubes: Biomedical applications. Int. J. Nanomed. 2012, 7, 5361. [Google Scholar]
- Serrano, M.C.; Gutiérrez, M.C.; del Monte, F. Role of polymers in the design of 3D carbon nanotube-based scaffolds for biomedical applications. Prog. Polym. Sci. 2014, 39, 1448–1471. [Google Scholar] [CrossRef]
- Hermant, M.-C.; van der Schoot, P.; Klumperman, B.; Koning, C.E. Probing the Cooperative Nature of the Conductive Components in Polystyrene/Poly (3, 4-ethylenedioxythiophene): Poly (styrene sulfonate)− Single-Walled Carbon Nanotube Composites. Acs Nano 2010, 4, 2242–2248. [Google Scholar] [CrossRef]
- Garipcan, B.; Odabas, S.; Demirel, G.; Burger, J.; Nonnenmann, S.S.; Coster, M.T.; Gallo, E.M.; Nabet, B.; Spanier, J.E.; Piskin, E. In Vitro Biocompatibility of n-Type and Undoped Silicon Nanowires. Adv. Eng. Mater. 2011, 13, B3–B9. [Google Scholar] [CrossRef]
- Tölli, M.A.; Ferreira, M.P.; Kinnunen, S.M.; Rysä, J.; Mäkilä, E.M.; Szabó, Z.; Serpi, R.E.; Ohukainen, P.J.; Välimäki, M.J.; Correia, A.M. In vivo biocompatibility of porous silicon biomaterials for drug delivery to the heart. Biomaterials 2014, 35, 8394–8405. [Google Scholar] [CrossRef]
- Tan, Y.; Richards, D.; Xu, R.; Stewart-Clark, S.; Mani, S.K.; Borg, T.K.; Menick, D.R.; Tian, B.; Mei, Y. Silicon Nanowire-Induced Maturation of Cardiomyocytes Derived from Human Induced Pluripotent Stem Cells. Nano Lett. 2015, 15, 2765–2772. [Google Scholar] [CrossRef]
- Jiang, K.; Fan, D.; Belabassi, Y.; Akkaraju, G.; Montchamp, J.-L.; Coffer, J.L. Medicinal surface modification of silicon nanowires: Impact on calcification and stromal cell proliferation. Acs Appl. Mater. Interfaces 2008, 1, 266–269. [Google Scholar] [CrossRef]
- Conde, J.; Dias, J.T.; Grazú, V.; Moros, M.; Baptista, P.V.; Jesus, M. Revisiting 30 years of biofunctionalization and surface chemistry of inorganic nanoparticles for nanomedicine. Front. Chem. 2014, 2, 48. [Google Scholar] [CrossRef]
- Sperling, R.A.; Parak, W. Surface modification, functionalization and bioconjugation of colloidal inorganic nanoparticles. Philos. Trans. R. Soc. A 2010, 368, 1333–1383. [Google Scholar] [CrossRef]
- Ravichandran, R.; Sridhar, R.; Venugopal, J.R.; Sundarrajan, S.; Mukherjee, S.; Ramakrishna, S. Gold Nanoparticle Loaded Hybrid Nanofibers for Cardiogenic Differentiation of Stem Cells for Infarcted Myocardium Regeneration. Macromol. Biosci. 2014, 14, 515–525. [Google Scholar] [CrossRef]
- McKeon-Fischer, K.D.; Freeman, J.W. Characterization of electrospun poly(L-lactide) and gold nanoparticle composite scaffolds for skeletal muscle tissue engineering. J. Tissue Eng. Regen. Med. 2011, 5, 560–568. [Google Scholar] [CrossRef]
- Sperling, R.A.; Gil, P.R.; Zhang, F.; Zanella, M.; Parak, W.J. Biological applications of gold nanoparticles. Chem. Soc. Rev. 2008, 37, 1896–1908. [Google Scholar] [CrossRef]
- Cohen-Karni, T.; Jeong, K.J.; Tsui, J.H.; Reznor, G.; Mustata, M.; Wanunu, M.; Graham, A.; Marks, C.; Bell, D.C.; Langer, R. Nanocomposite gold-silk nanofibers. Nano Lett. 2012, 12, 5403–5406. [Google Scholar] [CrossRef]
- Conde, J.; Oliva, N.; Artzi, N. Implantable hydrogel embedded dark-gold nanoswitch as a theranostic probe to sense and overcome cancer multidrug resistance. Proc. Natl. Acad. Sci. USA 2015, 112, E1278–E1287. [Google Scholar] [CrossRef]
- Conde, J.; Bao, C.; Cui, D.; Baptista, P.V.; Tian, F. Antibody–drug gold nanoantennas with Raman spectroscopic fingerprints for in vivo tumour theranostics. J. Control. Release 2014, 183, 87–93. [Google Scholar] [CrossRef]
- Gilam, A.; Conde, J.; Weissglas-Volkov, D.; Oliva, N.; Friedman, E.; Artzi, N.; Shomron, N. Local microRNA delivery targets Palladin and prevents metastatic breast cancer. Nat. Commun. 2016, 7, 12868. [Google Scholar] [CrossRef]
- Conde, J.; Oliva, N.; Zhang, Y.; Artzi, N. Local triple-combination therapy results in tumour regression and prevents recurrence in a colon cancer model. Nat. Mater. 2016, 15, 1128–1138. [Google Scholar] [CrossRef]
- Riley, P. Melanin. Int. J. Biochem. Cell Biol. 1997, 29, 1235–1239. [Google Scholar] [CrossRef]
- Kai, D.; Prabhakaran, M.P.; Jin, G.; Ramakrishna, S. Biocompatibility evaluation of electrically conductive nanofibrous scaffolds for cardiac tissue engineering. J. Mater. Chem. B 2013, 1, 2305–2314. [Google Scholar] [CrossRef]
- Bernsmann, F.; Frisch, B.; Ringwald, C.; Ball, V. Protein adsorption on dopamine–melanin films: Role of electrostatic interactions inferred from ζ-potential measurements versus chemisorption. J. Colloid Interface Sci. 2010, 344, 54–60. [Google Scholar] [CrossRef]
- Bernsmann, F.; Ersen, O.; Voegel, J.C.; Jan, E.; Kotov, N.A.; Ball, V. Melanin-Containing Films: Growth from Dopamine Solutions versus Layer-by-Layer Deposition. ChemPhysChem 2010, 11, 3299–3305. [Google Scholar] [CrossRef]
- Balachandran, U.; Odekirk, B.; Eror, N.G. Electrical conductivity in calcium titanate. J. Solid State Chem. 1982, 41, 185–194. [Google Scholar] [CrossRef]
- Figueiredo, F.M.; Kharton, V.V.; Waerenborgh, J.C.; Viskup, A.P.; Naumovich, E.N.; Frade, J.R. Influence of Microstructure on the Electrical Properties of Iron-Substituted Calcium Titanate Ceramics. J. Am. Ceram. Soc. 2004, 87, 2252–2261. [Google Scholar] [CrossRef]
- Thrivikraman, G.; Mallik, P.K.; Basu, B. Substrate conductivity dependent modulation of cell proliferation and differentiation in vitro. Biomaterials 2013, 34, 7073–7085. [Google Scholar] [CrossRef]
- Farokhi, M.; Mottaghitalab, F.; Shokrgozar, M.A.; Kaplan, D.L.; Kim, H.-W.; Kundu, S.C. Prospects of peripheral nerve tissue engineering using nerve guide conduits based on silk fibroin protein and other biopolymers. Int. Mater. Rev. 2017, 62, 367–391. [Google Scholar] [CrossRef]
- Subramanian, A.; Krishnan, U.M.; Sethuraman, S. Development of biomaterial scaffold for nerve tissue engineering: Biomaterial mediated neural regeneration. J. Biomed. Sci. 2009, 16, 108. [Google Scholar] [CrossRef]
- Ahn, H.-S.; Hwang, J.-Y.; Kim, M.S.; Lee, J.-Y.; Kim, J.-W.; Kim, H.-S.; Shin, U.S.; Knowles, J.C.; Kim, H.-W.; Hyun, J.K. Carbon-nanotube-interfaced glass fiber scaffold for regeneration of transected sciatic nerve. Acta Biomater. 2015, 13, 324–334. [Google Scholar] [CrossRef]
- Huang, J.; Hu, X.; Lu, L.; Ye, Z.; Zhang, Q.; Luo, Z. Electrical regulation of Schwann cells using conductive polypyrrole/chitosan polymers. J. Biomed. Mater. Res. A 2010, 93, 164–174. [Google Scholar] [CrossRef]
- Qi, F.; Wang, Y.; Ma, T.; Zhu, S.; Zeng, W.; Hu, X.; Liu, Z.; Huang, J.; Luo, Z. Electrical regulation of olfactory ensheathing using conductive polypyrrole/chitosan polymers. Biomaterials 2013, 34, 1799–1809. [Google Scholar] [CrossRef]
- Lee, J.Y.; Bashur, C.A.; Goldstein, A.S.; Schmidt, C.E. Polypyrrole-coated electrospun PLGA nanofibers for neural tissue applications. Biomaterials 2009, 30, 4325–4335. [Google Scholar] [CrossRef]
- Thrivikraman, G.; Madras, G.; Basu, B. Intermittent electrical stimuli for guidance of human mesenchymal stem cell lineage commitment towards neural-like cells on electroconductive substrates. Biomaterials 2014, 35, 6219–6235. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, L.; Hu, T.; Ma, P.X.; Guo, B. Conductive micropatterned polyurethane films as tissue engineering scaffolds for Schwann cells and PC12 cells. J. Colloid Interface Sci. 2018, 518, 252–262. [Google Scholar] [CrossRef]
- Guarino, V.; Alvarez-Perez, M.A.; Borriello, A.; Napolitano, T.; Ambrosio, L. Conductive PANi/PEGDA macroporous hydrogels for nerve regeneration. Adv. Healthc. Mater. 2013, 2, 218–227. [Google Scholar] [CrossRef]
- Bagher, Z.; Atoufi, Z.; Alizadeh, R.; Farhadi, M.; Zarrintaj, P.; Moroni, L.; Setayeshmehr, M.; Komeili, A.; Kamrava, S.K. Conductive hydrogel based on chitosan-aniline pentamer/gelatin/agarose significantly promoted motor neuron-like cells differentiation of human olfactory ecto-mesenchymal stem cells. Mater. Sci. Eng. C 2019, 101, 243–253. [Google Scholar] [CrossRef]
- Manouchehri, S.; Bagheri, B.; Rad, S.H.; Nezhad, M.N.; Kim, Y.C.; Park, O.O.; Farokhi, M.; Jouyandeh, M.; Ganjali, M.R.; Yazdi, M.K. Electroactive bio-epoxy incorporated chitosan-oligoaniline as an advanced hydrogel coating for neural interfaces. Prog. Org. Coat. 2019, 131, 389–396. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Bakhshandeh, B.; Rezaeian, I.; Heshmatian, B.; Ganjali, M.R. A novel electroactive agarose-aniline pentamer platform as a potential candidate for neural tissue engineering. Sci. Rep. 2017, 7, 17187. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Rezaeian, I.; Bakhshandeh, B.; Heshmatian, B.; Ganjali, M.R. Bio-conductive scaffold based on agarose-polyaniline for tissue engineering. J. Skin Stem Cell 2017, 4. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Urbanska, A.M.; Gholizadeh, S.S.; Goodarzi, V.; Saeb, M.R.; Mozafari, M. A facile route to the synthesis of anilinic electroactive colloidal hydrogels for neural tissue engineering applications. J. Colloid Interface Sci. 2018, 516, 57–66. [Google Scholar] [CrossRef]
- Atoufi, Z.; Zarrintaj, P.; Motlagh, G.H.; Amiri, A.; Bagher, Z.; Kamrava, S.K. A novel bio electro active alginate-aniline tetramer/agarose scaffold for tissue engineering: Synthesis, characterization, drug release and cell culture study. J. Biomater. Sci. Polym. Ed. 2017, 28, 1617–1638. [Google Scholar] [CrossRef]
- Alizadeh, R.; Zarrintaj, P.; Kamrava, S.K.; Bagher, Z.; Farhadi, M.; Heidari, F.; Komeili, A.; Gutiérrez, T.J.; Saeb, M.R. Conductive agarose/alginate/chitosan-based hydrogels for neural disorder therapy. Carbohydr. Polym. 2019, 115161. [Google Scholar] [CrossRef]
- Mattson, M.P.; Haddon, R.C.; Rao, A.M. Molecular functionalization of carbon nanotubes and use as substrates for neuronal growth. J. Mol. Neurosci. 2000, 14, 175–182. [Google Scholar] [CrossRef]
- Kabiri, M.; Soleimani, M.; Shabani, I.; Futrega, K.; Ghaemi, N.; Ahvaz, H.H.; Elahi, E.; Doran, M.R. Neural differentiation of mouse embryonic stem cells on conductive nanofiber scaffolds. Biotechnol. Lett. 2012, 34, 1357–1365. [Google Scholar] [CrossRef]
- Li, N.; Zhang, Q.; Gao, S.; Song, Q.; Huang, R.; Wang, L.; Liu, L.; Dai, J.; Tang, M.; Cheng, G. Three-dimensional graphene foam as a biocompatible and conductive scaffold for neural stem cells. Sci. Rep. 2013, 3, 1604. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, J.-W.; Schmidt, C.E. Neuroactive conducting scaffolds: Nerve growth factor conjugation on active ester-functionalized polypyrrole. J. R. Soc. Interface 2008, 6. [Google Scholar] [CrossRef]
- Huang, L.; Zhuang, X.; Hu, J.; Lang, L.; Zhang, P.; Wang, Y.; Chen, X.; Wei, Y.; Jing, X. Synthesis of biodegradable and electroactive multiblock polylactide and aniline pentamer copolymer for tissue engineering applications. Biomacromolecules 2008, 9, 850–858. [Google Scholar] [CrossRef]
- Richardson-Burns, S.M.; Hendricks, J.L.; Foster, B.; Povlich, L.K.; Kim, D.-H.; Martin, D.C. Polymerization of the conducting polymer poly (3, 4-ethylenedioxythiophene)(PEDOT) around living neural cells. Biomaterials 2007, 28, 1539–1552. [Google Scholar] [CrossRef]
- Stewart, E.; Kobayashi, N.R.; Higgins, M.J.; Quigley, A.F.; Jamali, S.; Moulton, S.E.; Kapsa, R.M.; Wallace, G.G.; Crook, J.M. Electrical stimulation using conductive polymer polypyrrole promotes differentiation of human neural stem cells: A biocompatible platform for translational neural tissue engineering. Tissue Eng. Part C Methods 2014, 21, 385–393. [Google Scholar] [CrossRef]
- Xie, J.; MacEwan, M.R.; Willerth, S.M.; Li, X.; Moran, D.W.; Sakiyama-Elbert, S.E.; Xia, Y. Conductive core–sheath nanofibers and their potential application in neural tissue engineering. Adv. Funct. Mater. 2009, 19, 2312–2318. [Google Scholar] [CrossRef]
- Cui, Z.; Ni, N.C.; Wu, J.; Du, G.Q.; He, S.; Yau, T.M.; Weisel, R.D.; Sung, H.W.; Li, R.K. Polypyrrole-chitosan conductive biomaterial synchronizes cardiomyocyte contraction and improves myocardial electrical impulse propagation. Theranostics 2018, 8, 2752–2764. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Lu, L.; Zhang, J.; Hu, X.; Zhang, Y.; Liang, W.; Wu, S.; Luo, Z. Electrical Stimulation to Conductive Scaffold Promotes Axonal Regeneration and Remyelination in a Rat Model of Large Nerve Defect. PLoS ONE 2012, 7, e39526. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Holzwarth, J.M.; Yan, Y.; Xu, P.; Zheng, H.; Yin, Y.; Li, S.; Ma, P.X. Conductive PPY/PDLLA conduit for peripheral nerve regeneration. Biomaterials 2014, 35, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.G.; Khaing, Z.Z.; Xin, S.; Tien, L.W.; Ghezzi, C.E.; Mouser, D.J.; Sukhavasi, R.C.; Preda, R.C.; Gil, E.S.; Kaplan, D.L. Into the groove: Instructive silk-polypyrrole films with topographical guidance cues direct DRG neurite outgrowth. J. Biomater. Sci. Polym. Ed. 2015, 26, 1327–1342. [Google Scholar] [CrossRef] [PubMed]
- Abidian, M.R.; Daneshvar, E.D.; Egeland, B.M.; Kipke, D.R.; Cederna, P.S.; Urbanchek, M.G. Hybrid conducting polymer–hydrogel conduits for axonal growth and neural tissue engineering. Adv. Healthc. Mater. 2012, 1, 762–767. [Google Scholar] [CrossRef]
- Zeng, J.; Huang, Z.; Yin, G.; Qin, J.; Chen, X.; Gu, J. Fabrication of conductive NGF-conjugated polypyrrole–poly (l-lactic acid) fibers and their effect on neurite outgrowth. Colloids Surf. B Biointerfaces 2013, 110, 450–457. [Google Scholar] [CrossRef]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Morshed, M.; Nasr-Esfahani, M.H.; Baharvand, H.; Kiani, S.; Al-Deyab, S.S.; Ramakrishna, S. Application of conductive polymers, scaffolds and electrical stimulation for nerve tissue engineering. J. Tissue Eng. Regen. Med. 2011, 5, e17–e35. [Google Scholar] [CrossRef]
- Chiono, V.; Tonda-Turo, C. Trends in the design of nerve guidance channels in peripheral nerve tissue engineering. Prog. Neurobiol. 2015, 131, 87–104. [Google Scholar] [CrossRef]
- Sudwilai, T.; Ng, J.J.; Boonkrai, C.; Israsena, N.; Chuangchote, S.; Supaphol, P. Polypyrrole-coated electrospun poly (lactic acid) fibrous scaffold: Effects of coating on electrical conductivity and neural cell growth. J. Biomater. Sci. Polym. Ed. 2014, 25, 1240–1252. [Google Scholar] [CrossRef]
- Lee, W.; Parpura, V. Carbon nanotubes as substrates/scaffolds for neural cell growth. Prog. Brain Res. 2009, 180, 110–125. [Google Scholar]
- Zhou, K.; Thouas, G.A.; Bernard, C.C.; Nisbet, D.R.; Finkelstein, D.I.; Li, D.; Forsythe, J.S. Method to impart electro-and biofunctionality to neural scaffolds using graphene–polyelectrolyte multilayers. ACS Appl. Mater. Interfaces 2012, 4, 4524–4531. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Gao, H.; Zhu, G.; Cao, X.; Shi, X.; Wang, Y. The preparation and characterization of polycaprolactone/graphene oxide biocomposite nanofiber scaffolds and their application for directing cell behaviors. Carbon 2015, 95, 1039–1050. [Google Scholar] [CrossRef]
- Ribeiro, J.; Pereira, T.; Caseiro, A.R.; Armada-da-Silva, P.; Pires, I.; Prada, J.; Amorim, I.; Amado, S.; França, M.; Gonçalves, C. Evaluation of biodegradable electric conductive tube-guides and mesenchymal stem cells. World J. Stem Cells 2015, 7, 956. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tavangarian, F.; Li, Y. Carbon nanostructures as nerve scaffolds for repairing large gaps in severed nerves. Ceram. Int. 2012, 38, 6075–6090. [Google Scholar] [CrossRef]
- Fabbro, A.; Sucapane, A.; Toma, F.M.; Calura, E.; Rizzetto, L.; Carrieri, C.; Roncaglia, P.; Martinelli, V.; Scaini, D.; Masten, L. Adhesion to carbon nanotube conductive scaffolds forces action-potential appearance in immature rat spinal neurons. PLoS ONE 2013, 8, e73621. [Google Scholar] [CrossRef] [PubMed]
- Kabiri, M.; Oraee-Yazdani, S.; Shafiee, A.; Hanaee-Ahvaz, H.; Dodel, M.; Vaseei, M.; Soleimani, M. Neuroregenerative effects of olfactory ensheathing cells transplanted in a multi-layered conductive nanofibrous conduit in peripheral nerve repair in rats. J. Biomed. Sci. 2015, 22, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.Q.; Wang, T.; Zhao, B.; Li, J.; Jin, L. Soft Graphene Nanofibers Designed for the Acceleration of Nerve Growth and Development. Adv. Mater. 2015, 27, 6462–6468. [Google Scholar] [CrossRef] [PubMed]
- Behan, B.L.; DeWitt, D.G.; Bogdanowicz, D.R.; Koppes, A.N.; Bale, S.S.; Thompson, D.M. Single-walled carbon nanotubes alter Schwann cell behavior differentially within 2D and 3D environments. J. Biomed. Mater. Res. Part A 2011, 96, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Tosun, Z.; McFetridge, P. A composite SWNT–collagen matrix: Characterization and preliminary assessment as a conductive peripheral nerve regeneration matrix. J. Neural Eng. 2010, 7, 066002. [Google Scholar] [CrossRef] [PubMed]
- Aregueta-Robles, U.A.; Woolley, A.J.; Poole-Warren, L.A.; Lovell, N.H.; Green, R.A. Organic electrode coatings for next-generation neural interfaces. Front. Neuoeng. 2014. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Morshed, M.; Nasr-Esfahani, M.H.; Ramakrishna, S. Electrical stimulation of nerve cells using conductive nanofibrous scaffolds for nerve tissue engineering. Tissue Eng. Part A 2009, 15, 3605–3619. [Google Scholar] [CrossRef] [PubMed]
- Bhang, S.H.; Jeong, S.I.; Lee, T.J.; Jun, I.; Lee, Y.B.; Kim, B.S.; Shin, H. Electroactive Electrospun Polyaniline/Poly [(L-lactide)-co-(ε-caprolactone)] Fibers for Control of Neural Cell Function. Macromol. Biosci. 2012, 12, 402–411. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Sun, Y.; Finne-Wistrand, A.; Mustafa, K.; Albertsson, A.-C. Electroactive porous tubular scaffolds with degradability and non-cytotoxicity for neural tissue regeneration. Acta Biomater. 2012, 8, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Sirivisoot, S.; Pareta, R.; Harrison, B.S. Protocol and cell responses in three-dimensional conductive collagen gel scaffolds with conductive polymer nanofibres for tissue regeneration. Interface Focus 2014, 4, 20130050. [Google Scholar] [CrossRef] [PubMed]
- Baniasadi, H.; SA, A.R.; Mashayekhan, S. Fabrication and characterization of conductive chitosan/gelatin-based scaffolds for nerve tissue engineering. Int. J. Biol. Macromol. 2015, 74, 360–366. [Google Scholar] [CrossRef]
- Urbanchek, M.; Shim, B.; Baghmanli, Z.; Wei, B.; Schroeder, K.; Langhals, N.; Miriani, R.; Egeland, B.; Kipke, D.; Martin, D. Conduction properties of decellularized nerve biomaterials. In Proceedings of the 26th Southern Biomedical Engineering Conference SBEC 2010, College Park, MD, USA, 30 April–2 May 2010; pp. 430–433. [Google Scholar]
- Meng, X.; Stout, D.A.; Sun, L.; Beingessner, R.L.; Fenniri, H.; Webster, T.J. Novel injectable biomimetic hydrogels with carbon nanofibers and self assembled rosette nanotubes for myocardial applications. J. Biomed. Mater. Res. Part A 2013, 101, 1095–1102. [Google Scholar] [CrossRef]
- Chen, Q.-Z.; Harding, S.E.; Ali, N.N.; Lyon, A.R.; Boccaccini, A.R. Biomaterials in cardiac tissue engineering: Ten years of research survey. Mater. Sci. Eng. R. Rep. 2008, 59, 1–37. [Google Scholar] [CrossRef]
- Stout, D.A.; Basu, B.; Webster, T.J. Poly (lactic–co-glycolic acid): Carbon nanofiber composites for myocardial tissue engineering applications. Acta Biomater. 2011, 7, 3101–3112. [Google Scholar] [CrossRef]
- Zia, S.; Mozafari, M.; Natasha, G.; Tan, A.; Cui, Z.; Seifalian, A.M. Hearts beating through decellularized scaffolds: Whole-organ engineering for cardiac regeneration and transplantation. Crit. Rev. Biotechnol. 2015, 1–11. [Google Scholar] [CrossRef]
- Kai, D.; Prabhakaran, M.P.; Jin, G.; Ramakrishna, S. Polypyrrole-contained electrospun conductive nanofibrous membranes for cardiac tissue engineering. J. Biomed. Mater. Res. Part A 2011, 99, 376–385. [Google Scholar] [CrossRef]
- Mukherjee, S.; Venugopal, J.R.; Ravichandran, R.; Ramakrishna, S.; Raghunath, M. Multimodal biomaterial strategies for regeneration of infarcted myocardium. J. Mater. Chem. 2010, 20, 8819–8831. [Google Scholar] [CrossRef]
- Moura, R.M.; de Queiroz, A.A.A. Dendronized polyaniline nanotubes for cardiac tissue engineering. Artif. Organs 2011, 35, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Sepantafar, M.; Maheronnaghsh, R.; Mohammadi, H.; Rajabi-Zeleti, S.; Annabi, N.; Aghdami, N.; Baharvand, H. Stem cells and injectable hydrogels: Synergistic therapeutics in myocardial repair. Biotechnol. Adv. 2016, 34, 362–379. [Google Scholar] [CrossRef] [PubMed]
- Kharaziha, M.; Shin, S.R.; Nikkhah, M.; Topkaya, S.N.; Masoumi, N.; Annabi, N.; Dokmeci, M.R.; Khademhosseini, A. Tough and flexible CNT–polymeric hybrid scaffolds for engineering cardiac constructs. Biomaterials 2014, 35, 7346–7354. [Google Scholar] [CrossRef] [PubMed]
- Gálvez-Montón, C.; Prat-Vidal, C.; Roura, S.; Soler-Botija, C.; Bayes-Genis, A. Cardiac tissue engineering and the bioartificial heart. Rev. Española De Cardiol. 2013, 66, 391–399. [Google Scholar] [CrossRef]
- Tandon, V.; Zhang, B.; Radisic, M.; Murthy, S.K. Generation of tissue constructs for cardiovascular regenerative medicine: From cell procurement to scaffold design. Biotechnol. Adv. 2013, 31, 722–735. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.R.; Jung, S.M.; Zalabany, M.; Kim, K.; Zorlutuna, P.; Kim, S.B.; Nikkhah, M.; Khabiry, M.; Azize, M.; Kong, J. Carbon-nanotube-embedded hydrogel sheets for engineering cardiac constructs and bioactuators. ACS Nano 2013, 7, 2369–2380. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, L.; Guo, B.; Ma, P.X. Interwoven aligned conductive nanofiber yarn/hydrogel composite scaffolds for engineered 3D cardiac anisotropy. ACS Nano 2017, 11, 5646–5659. [Google Scholar] [CrossRef]
- Spearman, B.S.; Hodge, A.J.; Porter, J.L.; Hardy, J.G.; Davis, Z.D.; Xu, T.; Zhang, X.; Schmidt, C.E.; Hamilton, M.C.; Lipke, E.A. Conductive interpenetrating networks of polypyrrole and polycaprolactone encourage electrophysiological development of cardiac cells. Acta Biomater. 2015, 28, 109–120. [Google Scholar] [CrossRef]
- Liang, S.; Zhang, Y.; Wang, H.; Xu, Z.; Chen, J.; Bao, R.; Tan, B.; Cui, Y.; Fan, G.; Wang, W. Paintable and Rapidly Bondable Conductive Hydrogels as Therapeutic Cardiac Patches. Adv. Mater. 2018, 30, 1704235. [Google Scholar] [CrossRef] [PubMed]
- Borriello, A.; Guarino, V.; Schiavo, L.; Alvarez-Perez, M.; Ambrosio, L. Optimizing PANi doped electroactive substrates as patches for the regeneration of cardiac muscle. J. Mater. Sci. Mater. Med. 2011, 22, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Crowder, S.W.; Liang, Y.; Rath, R.; Park, A.M.; Maltais, S.; Pintauro, P.N.; Hofmeister, W.; Lim, C.C.; Wang, X.; Sung, H.-J. Poly (ε-caprolactone)-carbon nanotube composite scaffolds for enhanced cardiac differentiation of human mesenchymal stem cells. Nanomedicine 2013, 8, 1763–1776. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, V.; Cellot, G.; Toma, F.M.; Long, C.S.; Caldwell, J.H.; Zentilin, L.; Giacca, M.; Turco, A.; Prato, M.; Ballerini, L. Carbon nanotubes promote growth and spontaneous electrical activity in cultured cardiac myocytes. Nano Lett. 2012, 12, 1831–1838. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, V.; Cellot, G.; Toma, F.M.; Long, C.S.; Caldwell, J.H.; Zentilin, L.; Giacca, M.; Turco, A.; Prato, M.; Ballerini, L. Carbon nanotubes instruct physiological growth and functionally mature syncytia: Nongenetic engineering of cardiac myocytes. ACS Nano 2013, 7, 5746–5756. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhou, J.; Liu, Z.; Chen, J.; Lü, S.; Sun, H.; Li, J.; Lin, Q.; Yang, B.; Duan, C. A PNIPAAm-based thermosensitive hydrogel containing SWCNTs for stem cell transplantation in myocardial repair. Biomaterials 2014, 35, 5679–5688. [Google Scholar] [CrossRef] [PubMed]
- Stout, D.A.; Raimondo, E.; Marostica, G.; Webster, T.J. Growth characteristics of different heart cells on novel nanopatch substrate during electrical stimulation. Bio-Med Mater. Eng. 2014, 24, 2101–2107. [Google Scholar]
- Hsiao, C.-W.; Bai, M.-Y.; Chang, Y.; Chung, M.-F.; Lee, T.-Y.; Wu, C.-T.; Maiti, B.; Liao, Z.-X.; Li, R.-K.; Sung, H.-W. Electrical coupling of isolated cardiomyocyte clusters grown on aligned conductive nanofibrous meshes for their synchronized beating. Biomaterials 2013, 34, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Lü, S.; Jiang, X.-X.; Li, X.; Li, H.; Lin, Q.; Mou, Y.; Zhao, Y.; Han, Y.; Zhou, J.; et al. Carbon nanotubes enhance intercalated disc assembly in cardiac myocytes via the β1-integrin-mediated signaling pathway. Biomaterials 2015, 55, 84–95. [Google Scholar] [CrossRef]
- Dvir, T.; Timko, B.P.; Brigham, M.D.; Naik, S.R.; Karajanagi, S.S.; Levy, O.; Jin, H.; Parker, K.K.; Langer, R.; Kohane, D.S. Nanowired three-dimensional cardiac patches. Nat. Nanotechnol. 2011, 6, 720–725. [Google Scholar] [CrossRef]
- Zhou, J.; Chen, J.; Sun, H.; Qiu, X.; Mou, Y.; Liu, Z.; Zhao, Y.; Li, X.; Han, Y.; Duan, C. Engineering the heart: Evaluation of conductive nanomaterials for improving implant integration and cardiac function. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef]
- Noorman, M.; van der Heyden, M.A.; van Veen, T.A.; Cox, M.G.; Hauer, R.N.; de Bakker, J.M.; van Rijen, H.V. Cardiac cell–cell junctions in health and disease: Electrical versus mechanical coupling. J. Mol. Cell. Cardiol. 2009, 47, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Yazdanpanah, A.; Amoabediny, G.; Shariatpanahi, P.; Nourmohammadi, J.; Tahmasbi, M.; Mozafari, M. Synthesis and Characterization of Polylactic Acid Tubular Scaffolds with Improved Mechanical Properties for Vascular Tissue Engineering. Trends Biomater. Artif. Organs 2014, 28, 99–105. [Google Scholar]
- Rowlands, A.S.; Cooper-White, J.J. Directing phenotype of vascular smooth muscle cells using electrically stimulated conducting polymer. Biomaterials 2008, 29, 4510–4520. [Google Scholar] [CrossRef] [PubMed]
- Mihardja, S.S.; Sievers, R.E.; Lee, R.J. The effect of polypyrrole on arteriogenesis in an acute rat infarct model. Biomaterials 2008, 29, 4205–4210. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-W.; Serna, F.; Schmidt, C.E. Carboxy-endcapped conductive polypyrrole: Biomimetic conducting polymer for cell scaffolds and electrodes. Langmuir 2006, 22, 9816–9819. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, J.-W. Porous-structured Conductive Polypyrrole Cell Scaffolds. Notes 2014, 35, 293. [Google Scholar] [CrossRef]
- Bidez, P.R.; Li, S.; MacDiarmid, A.G.; Venancio, E.C.; Wei, Y.; Lelkes, P.I. Polyaniline, an electroactive polymer, supports adhesion and proliferation of cardiac myoblasts. J. Biomater. Sci. Polym. Ed. 2006, 17, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Qazi, T.H.; Rai, R.; Dippold, D.; Roether, J.E.; Schubert, D.W.; Rosellini, E.; Barbani, N.; Boccaccini, A.R. Development and characterization of novel electrically conductive PANI–PGS composites for cardiac tissue engineering applications. Acta Biomater. 2014, 10, 2434–2445. [Google Scholar] [CrossRef] [PubMed]
- Baheiraei, N.; Yeganeh, H.; Ai, J.; Gharibi, R.; Ebrahimi-Barough, S.; Azami, M.; Vahdat, S.; Baharvand, H. Preparation of a porous conductive scaffold from aniline pentamer-modified polyurethane/PCL blend for cardiac tissue engineering. J. Biomed. Mater. Res. Part A 2015, 103, 3179–3187. [Google Scholar] [CrossRef]
- Stout, D.A.; Yoo, J.; Webster, T.J. Poly Lactic-Co-Glycolic Acid Carbon Nanofiber Composite for Enhancing Cardiomyocyte Function. MRS Online Proc. Libr. Arch. 2011, mrsf10-1316-qq1306-1333. [Google Scholar] [CrossRef]
- Han, Z.; Kong, H.; Meng, J.; Wang, C.; Xie, S.; Xu, H. Electrospun aligned nanofibrous scaffold of carbon nanotubes-polyurethane composite for endothelial cells. J. Nanosci. Nanotechnol. 2009, 9, 1400–1402. [Google Scholar] [CrossRef]
- Mooney, E.; Mackle, J.N.; Blond, D.J.P.; O’Cearbhaill, E.; Shaw, G.; Blau, W.J.; Barry, F.P.; Barron, V.; Murphy, J.M. The electrical stimulation of carbon nanotubes to provide a cardiomimetic cue to MSCs. Biomaterials 2012, 33, 6132–6139. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.M.; Eng, G.; Caridade, S.G.; Mano, J.F.; Reis, R.L.; Vunjak-Novakovic, G. Electrically Conductive Chitosan/Carbon Scaffolds for Cardiac Tissue Engineering. Biomacromolecules 2014, 15, 635–643. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, R.A.; Laurenzi, B.F.; Viswanathan, G.; Ajayan, P.M.; Stegemann, J.P. Collagen–carbon nanotube composite materials as scaffolds in tissue engineering. J. Biomed. Mater. Res. Part A 2005, 74, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Pok, S.; Vitale, F.; Eichmann, S.L.; Benavides, O.M.; Pasquali, M.; Jacot, J.G. Biocompatible Carbon Nanotube–Chitosan Scaffold Matching the Electrical Conductivity of the Heart. ACS Nano 2014, 8, 9822–9832. [Google Scholar] [CrossRef] [PubMed]
- Wan, A.M.-D.; Inal, S.; Williams, T.; Wang, K.; Leleux, P.; Estevez, L.; Giannelis, E.P.; Fischbach, C.; Malliaras, G.G.; Gourdon, D. 3D Conducting Polymer Platforms for Electrical Control of Protein Conformation and Cellular Functions. J. Mater. Chem. B 2015, 3, 5040–5048. [Google Scholar] [CrossRef]
- Petrisor, B.; Lau, J.T.C. Electrical Bone Stimulation: An Overview and its Use in High Risk and Charcot Foot and Ankle Reconstructions. Foot Ankle Clin. 2005, 10, 609–620. [Google Scholar] [CrossRef]
- Björninen, M.; Siljander, A.; Pelto, J.; Hyttinen, J.; Kellomäki, M.; Miettinen, S.; Seppänen, R.; Haimi, S. Comparison of chondroitin sulfate and hyaluronic acid doped conductive polypyrrole films for adipose stem cells. Ann. Biomed. Eng. 2014, 42, 1889–1900. [Google Scholar] [CrossRef]
- Hamlehkhan, A.; Mozafari, M.; Nezafati, N.; Azami, M.; Samadikuchaksaraei, A. Novel bioactive poly (ε-caprolactone)-gelatin-hydroxyapatite nanocomposite scaffolds for bone regeneration. Key Eng. Mater. 2012, 493–494, 909–915. [Google Scholar] [CrossRef]
- Cheng, C.W.; Solorio, L.D.; Alsberg, E. Decellularized tissue and cell-derived extracellular matrices as scaffolds for orthopaedic tissue engineering. Biotechnol. Adv. 2014, 32, 462–484. [Google Scholar] [CrossRef]
- Henkel, J.; Woodruff, M.A.; Epari, D.R.; Steck, R.; Glatt, V.; Dickinson, I.C.; Choong, P.F.M.; Schuetz, M.A.; Hutmacher, D.W. Bone Regeneration Based on Tissue Engineering Conceptions—A 21st Century Perspective. Bone Res. 2013, 1, 216. [Google Scholar] [CrossRef] [PubMed]
- Baxter, F.R.; Bowen, C.R.; Turner, I.G.; Dent, A.C. Electrically active bioceramics: A review of interfacial responses. Ann. Biomed. Eng. 2010, 38, 2079–2092. [Google Scholar] [CrossRef] [PubMed]
- Griffin, M.; Bayat, A. Electrical stimulation in bone healing: Critical analysis by evaluating levels of evidence. Eplasty 2011, 11, e34. [Google Scholar]
- Cui, H.; Wang, Y.; Cui, L.; Zhang, P.; Wang, X.; Wei, Y.; Chen, X. In Vitro Studies on Regulation of Osteogenic Activities by Electrical Stimulus on Biodegradable Electroactive Polyelectrolyte Multilayers. Biomacromolecules 2014, 15, 3146–3157. [Google Scholar] [CrossRef] [PubMed]
- Shahini, A.; Yazdimamaghani, M.; Walker, K.J.; Eastman, M.A.; Hatami-Marbini, H.; Smith, B.J.; Ricci, J.L.; Madihally, S.V.; Vashaee, D.; Tayebi, L. 3D conductive nanocomposite scaffold for bone tissue engineering. Int. J. Nanomed. 2014, 9, 167. [Google Scholar]
- Liu, Y.; Cui, H.; Zhuang, X.; Wei, Y.; Chen, X. Electrospinning of aniline pentamer-graft-gelatin/PLLA nanofibers for bone tissue engineering. Acta Biomater. 2014, 10, 5074–5080. [Google Scholar] [CrossRef]
- Lyu, C.-Q.; Lu, J.-Y.; Cao, C.-H.; Luo, D.; Fu, Y.-X.; He, Y.-S.; Zou, D.-R. Induction of Osteogenic Differentiation of Human Adipose-Derived Stem Cells by a Novel Self-Supporting Graphene Hydrogel Film and the Possible Underlying Mechanism. ACS Appl. Mater. Interfaces 2015, 7, 20245–20254. [Google Scholar] [CrossRef]
- Meng, S.; Zhang, Z.; Rouabhia, M. Accelerated osteoblast mineralization on a conductive substrate by multiple electrical stimulation. J. Bone Miner. Metab. 2011, 29, 535–544. [Google Scholar] [CrossRef]
- Meng, S.; Rouabhia, M.; Zhang, Z. Electrical stimulation modulates osteoblast proliferation and bone protein production through heparin-bioactivated conductive scaffolds. Bioelectromagnetics 2013, 34, 189–199. [Google Scholar] [CrossRef]
- Whitehead, M.A.; Fan, D.; Akkaraju, G.R.; Canham, L.T.; Coffer, J.L. Accelerated calcification in electrically conductive polymer composites comprised of poly (ε-caprolactone), polyaniline, and bioactive mesoporous silicon. J. Biomed. Mater. Res. Part A 2007, 83, 225–234. [Google Scholar] [CrossRef]
- Runge, M.B.; Dadsetan, M.; Baltrusaitis, J.; Yaszemski, M.J. Electrically conductive surface modifications of three-dimensional polypropylene fumarate scaffolds. J. Biol. Regul. Homeost. Agents 2011, 25, S15. [Google Scholar] [PubMed]
- Pelto, J.; Björninen, M.; Pälli, A.; Talvitie, E.; Hyttinen, J.; Mannerström, B.; Suuronen Seppanen, R.; Kellomäki, M.; Miettinen, S.; Haimi, S. Novel polypyrrole-coated polylactide scaffolds enhance adipose stem cell proliferation and early osteogenic differentiation. Tissue Eng. Part A 2013, 19, 882–892. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Liu, Y.; Deng, M.; Pang, X.; Zhang, P.; Wang, X.; Chen, X.; Wei, Y. Synthesis of biodegradable and electroactive tetraaniline grafted poly (ester amide) copolymers for bone tissue engineering. Biomacromolecules 2012, 13, 2881–2889. [Google Scholar] [CrossRef] [PubMed]
- Newman, P.; Minett, A.; Ellis-Behnke, R.; Zreiqat, H. Carbon nanotubes: Their potential and pitfalls for bone tissue regeneration and engineering. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 1139–1158. [Google Scholar] [CrossRef] [PubMed]
- Nishida, E.; Miyaji, H.; Takita, H.; Kanayama, I.; Tsuji, M.; Akasaka, T.; Sugaya, T.; Sakagami, R.; Kawanami, M. Graphene oxide coating facilitates the bioactivity of scaffold material for tissue engineering. Jpn. J. Appl. Phys. 2014, 53, 06JD04. [Google Scholar] [CrossRef]
- Xie, X.; Hu, K.; Fang, D.; Shang, L.; Tran, S.D.; Cerruti, M. Graphene and hydroxyapatite self-assemble into homogeneous, free standing nanocomposite hydrogels for bone tissue engineering. Nanoscale 2015, 7, 7992–8002. [Google Scholar] [CrossRef] [PubMed]
- Elkhenany, H.; Amelse, L.; Lafont, A.; Bourdo, S.; Caldwell, M.; Neilsen, N.; Dervishi, E.; Derek, O.; Biris, A.S.; Anderson, D. Graphene supports in vitro proliferation and osteogenic differentiation of goat adult mesenchymal stem cells: Potential for bone tissue engineering. J. Appl. Toxicol. 2015, 35, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Sayyar, S.; Murray, E.; Thompson, B.; Chung, J.; Officer, D.L.; Gambhir, S.; Spinks, G.M.; Wallace, G.G. Processable conducting graphene/chitosan hydrogels for tissue engineering. J. Mater. Chem. B 2015, 3, 481–490. [Google Scholar] [CrossRef]
- Rajzer, I.; Rom, M.; Menaszek, E.; Pasierb, P. Conductive PANI patterns on electrospun PCL/gelatin scaffolds modified with bioactive particles for bone tissue engineering. Mater. Lett. 2015, 138, 60–63. [Google Scholar] [CrossRef]
- Sirivisoot, S.; Harrison, B.S. Skeletal myotube formation enhanced by electrospun polyurethane carbon nanotube scaffolds. Int. J. Nanomed. 2011, 6, 2483. [Google Scholar] [CrossRef]
- Bach, A.; Beier, J.; Stern-Staeter, J.; Horch, R. Skeletal muscle tissue engineering. J. Cell. Mol. Med. 2004, 8, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Bian, W.; Bursac, N. Tissue engineering of functional skeletal muscle: Challenges and recent advances. IEEE Eng. Med. Biol. Mag. 2008, 27, 109–113. [Google Scholar] [PubMed]
- Kawahara, Y.; Yamaoka, K.; Iwata, M.; Fujimura, M.; Kajiume, T.; Magaki, T.; Takeda, M.; Ide, T.; Kataoka, K.; Asashima, M. Novel electrical stimulation sets the cultured myoblast contractile function to ‘on’. Pathobiology 2007, 73, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, K.J.; Kita, M.; Han, Y.; Gelmi, A.; Higgins, M.J.; Moulton, S.E.; Clark, G.M.; Kapsa, R.; Wallace, G.G. Skeletal muscle cell proliferation and differentiation on polypyrrole substrates doped with extracellular matrix components. Biomaterials 2009, 30, 5292–5304. [Google Scholar] [CrossRef] [PubMed]
- Stern-Straeter, J.; Bach, A.; Stangenberg, L.; Foerster, V.; Horch, R.; Stark, G.; Beier, J. Impact of electrical stimulation on three-dimensional myoblast cultures-a real-time RT-PCR study. J. Cell. Mol. Med. 2005, 9, 883–892. [Google Scholar] [CrossRef]
- Jun, I.; Jeong, S.; Shin, H. The stimulation of myoblast differentiation by electrically conductive sub-micron fibers. Biomaterials 2009, 30, 2038–2047. [Google Scholar] [CrossRef]
- Wang, L.; Wu, Y.; Guo, B.; Ma, P.X. Nanofiber yarn/hydrogel core–shell scaffolds mimicking native skeletal muscle tissue for guiding 3D myoblast alignment, elongation, and differentiation. ACS Nano 2015, 9, 9167–9179. [Google Scholar] [CrossRef]
- Quigley, A.F.; Razal, J.M.; Kita, M.; Jalili, R.; Gelmi, A.; Penington, A.; Ovalle-Robles, R.; Baughman, R.H.; Clark, G.M.; Wallace, G.G. Electrical stimulation of myoblast proliferation and differentiation on aligned nanostructured conductive polymer platforms. Adv. Healthc. Mater. 2012, 1, 801–808. [Google Scholar] [CrossRef]
- Chen, M.-C.; Sun, Y.-C.; Chen, Y.-H. Electrically conductive nanofibers with highly oriented structures and their potential application in skeletal muscle tissue engineering. Acta Biomater. 2013, 9, 5562–5572. [Google Scholar] [CrossRef]
- Ku, S.H.; Lee, S.H.; Park, C.B. Synergic effects of nanofiber alignment and electroactivity on myoblast differentiation. Biomaterials 2012, 33, 6098–6104. [Google Scholar] [CrossRef]
- Li, L.; Ge, J.; Wang, L.; Guo, B.; Ma, P.X. Electroactive nanofibrous biomimetic scaffolds by thermally induced phase separation. J. Mater. Chem. B 2014, 2, 6119–6130. [Google Scholar] [CrossRef]
- Ribeiro, S.; Costa, P.; Ribeiro, C.; Sencadas, V.; Botelho, G.; Lanceros-Méndez, S. Electrospun styrene–butadiene–styrene elastomer copolymers for tissue engineering applications: Effect of butadiene/styrene ratio, block structure, hydrogenation and carbon nanotube loading on physical properties and cytotoxicity. Compos. Part B Eng. 2014, 67, 30–38. [Google Scholar] [CrossRef]
- Wang, Q.; He, W.; Huang, J.; Liu, S.; Wu, G.; Teng, W.; Wang, Q.; Dong, Y. Synthesis of water soluble, biodegradable, and electroactive polysaccharide crosslinker with aldehyde and carboxylic groups for biomedical applications. Macromol. Biosci. 2011, 11, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.G.; Lee, J.Y.; Schmidt, C.E. Biomimetic conducting polymer-based tissue scaffolds. Curr. Opin. Biotechnol. 2013, 24, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Zarrintaj, P.; Mostafapoor, F.; Milan, P.B.; Saeb, M.R. Theranostic Platforms Proposed for Cancerous Stem Cells: A Review. Curr. Stem Cell Res. Ther. 2019, 14, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Tahmasbi Rad, A.; Ali, N.; Kotturi, H.S.R.; Yazdimamaghani, M.; Smay, J.; Vashaee, D.; Tayebi, L. Conducting scaffolds for liver tissue engineering. J. Biomed. Mater. Res. Part A 2014, 102, 4169–4181. [Google Scholar] [CrossRef] [PubMed]
- Mohebbi, S.; Nezhad, M.N.; Zarrintaj, P.; Jafari, S.H.; Gholizadeh, S.S.; Saeb, M.R.; Mozafari, M. Chitosan in biomedical engineering: A critical review. Curr. Stem Cell Res. Ther. 2019, 14, 93–116. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Liu, J.; Dvir, T.; Jin, L.; Tsui, J.H.; Qing, Q.; Suo, Z.; Langer, R.; Kohane, D.S.; Lieber, C.M. Macroporous nanowire nanoelectronic scaffolds for synthetic tissues. Nat. Mater. 2012, 11, 986–994. [Google Scholar] [CrossRef] [PubMed]



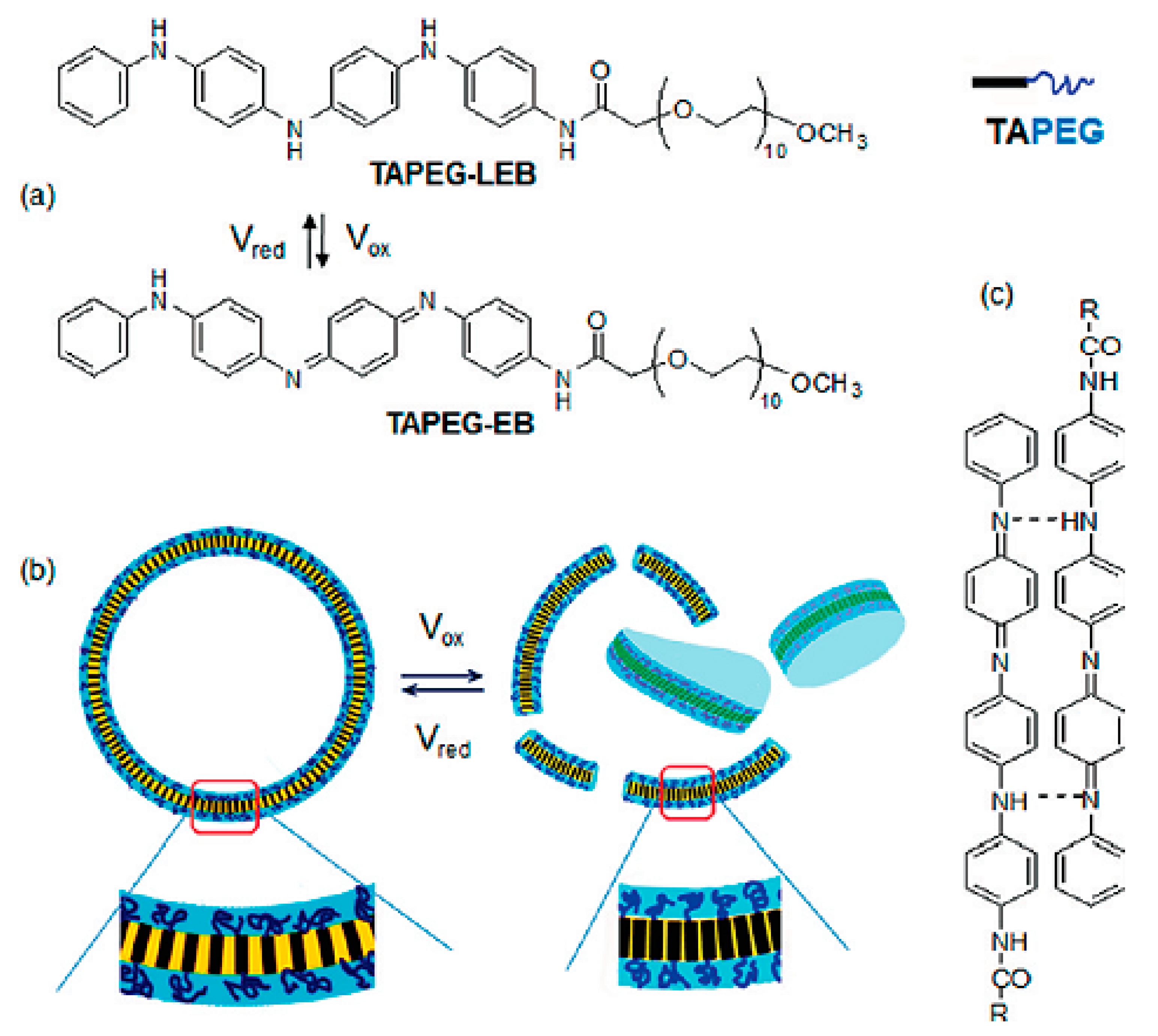

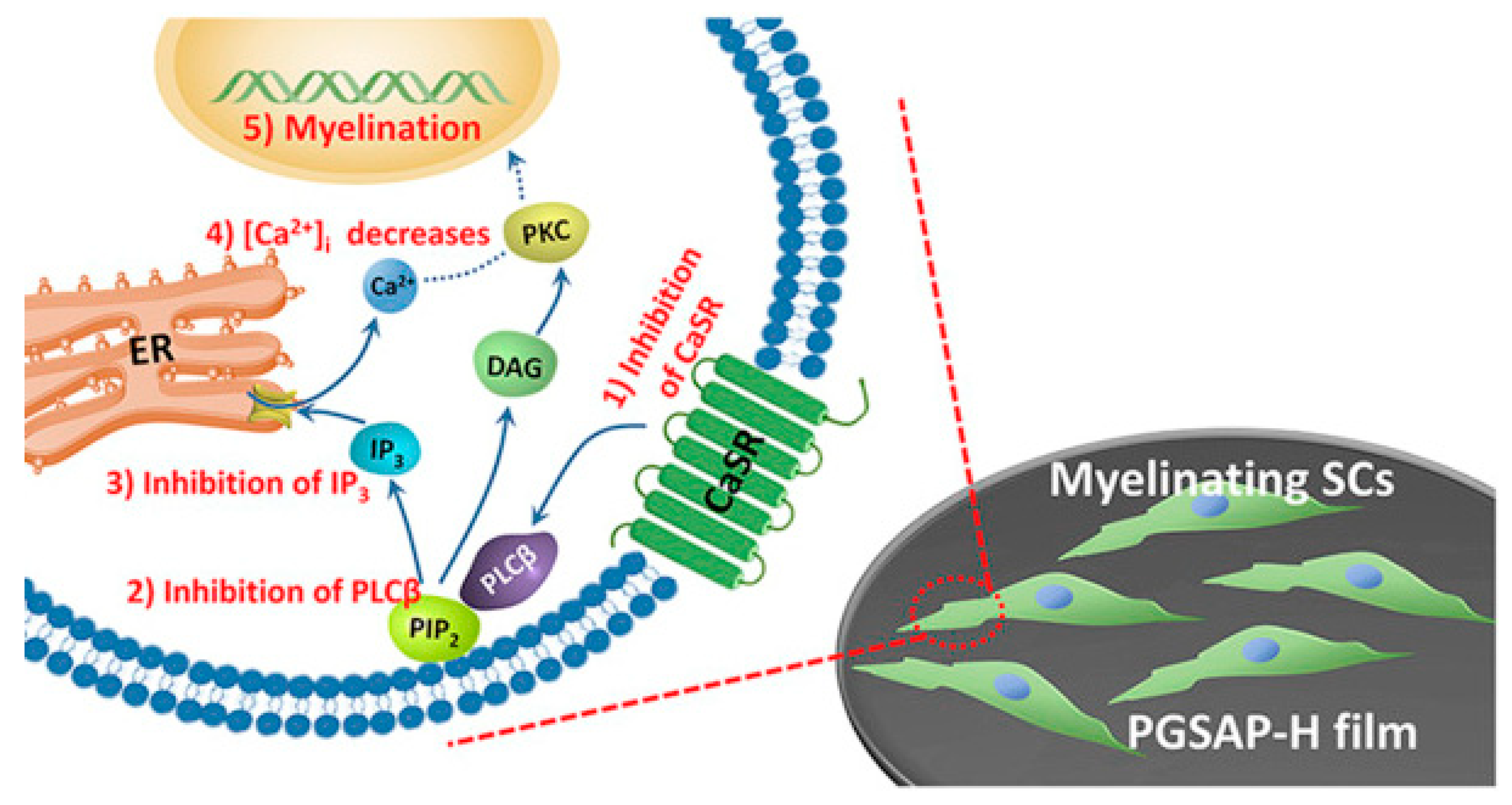
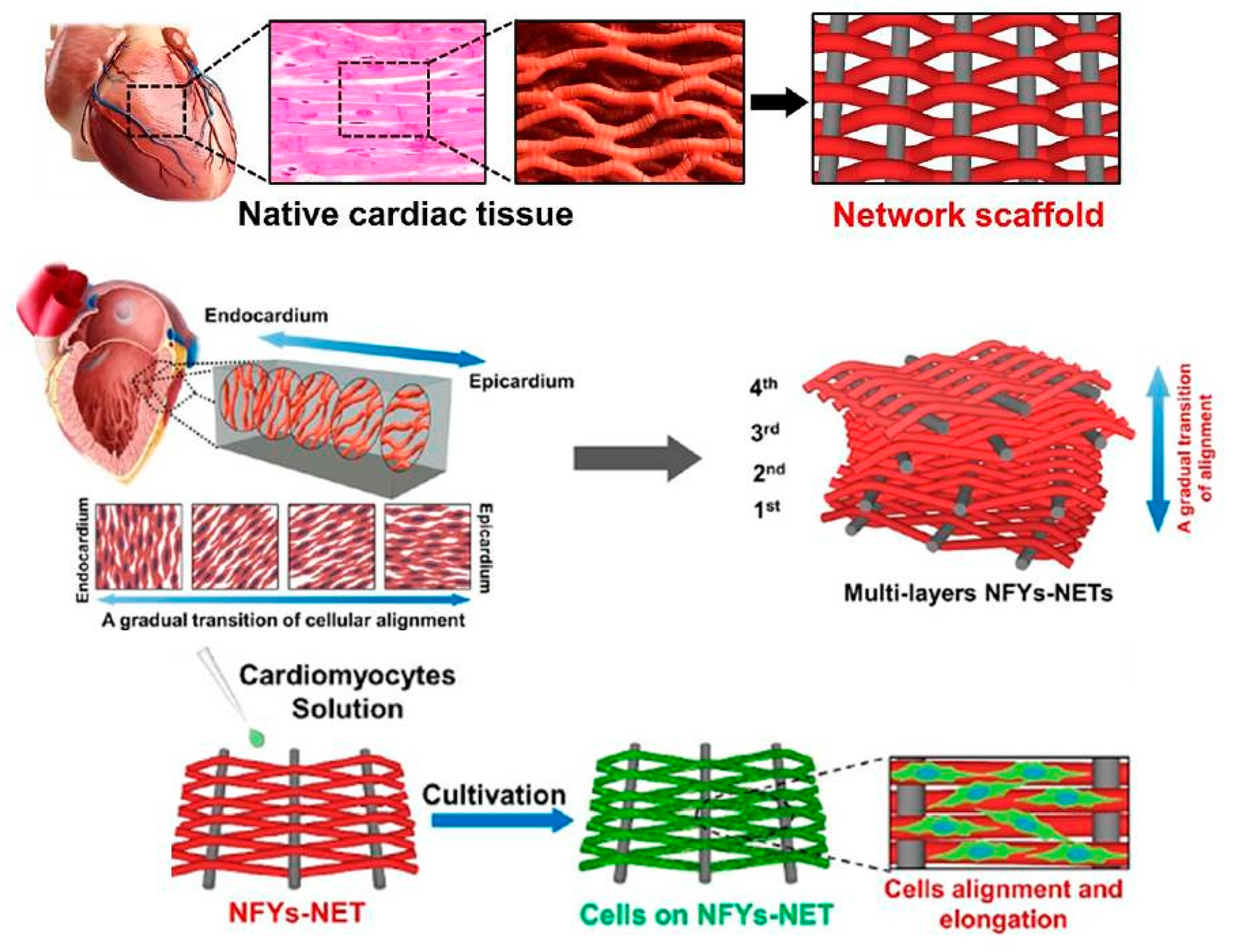



| Conductive Material | Identification Card |
|---|---|
Polyaniline | An oxidative polymer with wide ranges of conductivity, cost-effective, easy to synthesis [46]. Max conductivity = 30–200 S/cm |
Polypyrrole | Known for having a wide range of conductivity, insoluble in solvent, and quasi-unidimensional Max conductivity = 40–200 S/cm |
Poly(3,4-ethylenedioxythiophene) | A stable conductive polymer in biological condition with proper biocompatibility [46]. |
Polythiophene | Known for its proper conductivity. Max conductivity = 10–100 S/cm |
Polyacetylene | A semi-conducting polymer, which its conductivity can be enhanced using iodine vapor. Polyacetylene was difficult to synthesize and is unstable in air [43]. |
Poly (p-phenylene) | A very high thermally-stable conductive polymer mostly used in rocket nozzles. Its conductivity is 102–103 S/cm. |
Poly(p-phenylenevinylene) | This is the only conducting polymer with highly crystalline thin film formation ability. It can be utilized in photovoltaic devices and light-emitting diodes (LED). |
Poly-p-phenylene-sulphide | An engineering high-performance thermoplastic, opaque white to light tan in color. It can be molded, extruded, or machined to high tolerances. |
Silicon | Widely used in chemical and biological sensors and tissue engineering applications. It has controllable electrical conductivity, tunable dimensions, and convenient surface tailorability. |
Melanin | Light-absorbing polymer containing indoles and other intermediate products derived from the oxidation of tyrosine. Melanin is widely found in the animals and plants, and also known as the main pigment in the vertebrates surface structures |
| Gold | Gold has low toxicity, used in drug delivery, imaging and cancer therapy. |
| Carbon family | Carbon allotropes including graphite, diamond and carbon nanotubes show proper mechanical, electrical, thermal, optical, and structural properties [19,47]. |
| Calcium Titanate | Calcium titanium oxide is an inorganic compound (CaTiO3). |
| Conductive Material | Scaffold Type | Scaffold Material | Fabrication Method | Applying Technique | Confirmation Method | Cell Type | Electrical Stimulation/Duration | Major Outcome | Reference |
|---|---|---|---|---|---|---|---|---|---|
| PPy | Film | PPy | Synthesized in two steps by1-(2-cyanoethyl)pyrrole and NHS-1-ethyl-3-(3-dimethylaminopropyl) | Electrochemically polymerized | Enzyme-linked immunosorbent assay | PC12 | No | External electrical potential to NGF-immobilized PPy films did not cause a significant release of NGF nor reduce their neurotrophic activity | [157] |
| PPy | Ring-opening polymerization | Electrochemically polymerized | Cyclic Voltammetry | Rat C6 cells | Yes/square wave, frequency of 1 Hz, 5% duty cycle, and electrical potential of 0.1 V | Differentiation of rat neuronal pheochromocytoma PC-12 cells | [158] | ||
| PPy/chitosan | Micro emulsion polymerization | Blending | - | Schwann cells | Yes/a lateral constant potential gradient (100 mV/mm, 4 h) | Composite dramatically enhanced the expression and secretion of NGF and BDNF | [142] | ||
| PPy/poly(3,4-ethylenedioxythiophene) | Electrochemical polymerization | Coating | Cyclic Voltammetry (CV) | Neural cells | No | A RC parallel circuit must be added to the model for PEDOT + live neuron and neuron-templated PEDOT coatings. | [159] | ||
| PPy | Polymerized galvanostatically | Polymerized galvanostatically | - | hNSCs | 8 h per 24 h period for 3 days | Use of electrical stimulation to promote neuronal induction and the biocompatibility of PPy(DBS) with hNSCs | [160] | ||
| PPy/poly(ε-caprolactone)-poly(lactic acid) (PLA) | Polymerization of pyrrole on electrospun PCL or PLA | Coating | - | Dorsal root ganglia | No | The results suggest the potential use of the conductive coresheath nanofibers as scaffolds in applications such as neural tissue engineering | [161] | ||
| PPy/PCL Fumarate | Pre-formed PCLF scaffolds | polymerizing pyrrole in pre-formed PCLF scaffolds | 4-point probe method | PC12-dorsal root ganglia | Yes/DC current source | PCLF-PPy materials synthesized with NSA or DBSA support cell attachment, proliferation, neurite extension | [50] | ||
| PPy/(PCLF) | All chemicals were purchased from Fisher or Aldrich and used as is unless noted otherwise. | blending | - | PC12- | Yes/1 h/day of 10 μA 20 Hz | significant increases in percentage of neurite bearing cells, number of neurites per cell | [45] | ||
| PPy/chitosan | in-situ polymerization | in-situ preparation | Electromyography | neonatal rat cardiomyocytes | No | PPy/chitosan synchronized the contraction of physically-isolated cardiomyocytes clusters without external electrical stimulation. | [162] | ||
| PPy/chitosan | Freeze–drying method. | Mixing | Masson Trichrome Staining | Left sciatic nerve | Yes/intermittent ES (3 V, 20 Hz) | Establishing an electrical environment with electrical stimulation localized at the conductive scaffold is capable of accelerating nerve regeneration and promoting functional recovery in nerve defect in rats | [163] | ||
| PPy/poly (d,l-lactide) (PDLLA) | PDLLA was purchased from Boehringer Ingelheim (Germany) | Mixing | Cyclic Voltammetry | PC12 cells | Yes/100 mV for 2 h | When the PPY/PDLLA nerve conduit was used to repair a rat sciatic nerve defect it performed similarly to the gold standard autologous graft. | [164] | ||
| PPy/silk fibroin solution-PDMS | soft lithography technique | Coating | Cyclic Voltammetry | NGCs | No | Dorsal root ganglions adhere to the films and the grooves in the surface of the films instruct the aligned growth of processes extending from the Dorsal root ganglions | [165] | ||
| Hydrogel | PPy/PHEMA–MMA | - | Blending | EDL muscle mass and EDL maximal specific muscle force | Peroneal nerve gaps | Yes/current density of 0.5 mA cm for 30 min | The PEDOT lining may be used to facilitate future studies using electrical stimulation and/or controlled release of neurotrophins. In addition to promoting axonal growth, the conductive lining may be used as an effective interface between the electronic circuitry of neural prosthetic devices and the peripheral nervous system | [166] | |
| Nanofibers | PPy/PLLA | Electrospinning | Coating | - | Pc12 | Yes/100 mV/cm voltages through the composite fibers. | PPy-PLLA fibers could support PC12 neurite outgrowth and extension | [167] | |
| PPy/PLGA | Electrospinning | Coating | - | PC12 | No | Stimulation of the cells on aligned PPy-PLGA fibers resulted in longer neurites and more neurite-bearing cells than stimulation on random PPy-PLGA fibers, suggesting a combined effect of electrical stimulation and topographical guidance | [143] | ||
| PPy/Gelatin- poly(ε-caprolactone) | Acidic condition selectrospining | Mixing | - | C17.2-neural cells | No | The use of electrically CP very attractive for the construction of scaffolds for nerve tissue Engineering. | [168] | ||
| PPy/poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/799 po ly(e-caprolactone) (PHBHV/PCL | Electrospinning | Mixing | - | NGCs | No | The current trend of peripheral nerve tissue engineering is the design of advanced nerve guidance channels acting as physical guidance for regeneration of nerves across lesions. Nerve guidance channels should present multifunctional properties aiming to direct the sprouting of axons from the proximal nerve end, to concentrate growth factors secreted by the injured nerve ends, and to reduce the ingrowth of scar tissue into the injury site. | [169] | ||
| PPy/poly(lactic acid) | Electrospinning | Coating | Two-Point Probe | Neural progenitor cells | Yes/steady potential of 100 mV for 2 h continuing with biphasic 100 mV for 4 h per day, for three days | PPy-coated electrospun PLA fibers had a good biocompatibility with neural progenitor Cells | [170] | ||
| Carbon | Film | Carbon nanotube (CNTs) | - | - | Cyclic Voltammetry | Neural cells- glial cells- Schwann cells | No | Accumulating data support the use of CNTs as a biocompatible and permissive substrate/scaffold for neural cells and such application holds great potential in biomedicine | [171] |
| Graphene/Polyelectrolyte | Layer-by-layer (LbL) deposition | - | Probe analysis | Primary cortical neurons (PCNs) | No | Electro active scaffold modification may therefore assist in neuronal regeneration, for creating Functional and biocompatible polymer scaffolds for electrical entrainment or bio sensing applications. | [172] | ||
| Graphene | Pre-fabricated | - | Cyclic voltammetry | NSCs | Monophasic cathodic pulses with stimulation threshold was 20–30 μA | 3D-GFs can enhance the NSC differentiation towards astrocytes and especially neurons | [30] | ||
| CNT/PLDLA | Chemically tethered onto the surface | Mixing | Cyclic Voltammetry | Rat sciatic nerve | Yes/5 mA stimulus intensity, 1 Hz frequency, 1 ms duration | In vivo effect of using a CNT-interfaced scaffold in the regeneration process of a transected rat sciatic nerve strongly supports the potential use of CNT-interfaced PGFs at the Interface between the nerve conduit and peripheral neural tissues. | [140] | ||
| Graphene/PCL | Conventional electrospinning process | Mixing | - | PC L 12- mMSCs | No | Hybridization of GO nanosheets and PCL polymer dramatically enhanced the differentiation of the mMSCs and PC12-L cells into osteo- and neuro-like cells www.win | [173] | ||
| carbon nanotube (CNT)/polyvinyl alcohol (PVA) -(PVA-polypyrrole) | Casting technique to a silicone mould | Coating | - | Mesenchymal stem cellS (MSCS) | No | Results revealed that treatment with MSCs and PVA-CNTs tube-guides induced better Nerve fiber regeneration. | [174] | ||
| Nanofibers | carbon nanofiber (CNF)/PLLA | Electrospinning | Blending | - | mESC | Yes/frequency range of 1–106 Hz. | Conductive scaffold could be a useful tool for the generation of neural Tissue mimics in vitro and potentially as a scaffold for the repair of neural defects in vivo. | [155] | |
| CNF | - | - | - | Human epidermal keratinocytes (HaCaT) | No | Possibility of utilizing carbon nanostructures to repair a long gap in nerve | [175] | ||
| CNT | - | - | Using a Multiclamp 700B amplifier | Neonatal rat spinal cord | Electrophysiological recordings were acquired using a Multiclamp 700B amplifier (Molecular Devices), sampled at 10 kHz and digitized by a Digidata 1440A analog-to-digital converter. | CNT-incorporated/supported platforms trigger reparative activities involving microglia, in the absence of reactive gliosis | [176] | ||
| CNT/PLLA | Electrospinning | Mixing | Cyclic Voltammetry | OEC | No | SWNT/PLLA nanofibrous scaffolds and OEC could promote axonal outgrowth and peripheral nerve regeneration | [177] | ||
| CNT/poly(3,4-ethylenedioxythiophene)- | Electrospinning | Coating | Cyclic Voltammetry | Motor neurons | Yes/100 mV pulse electrical signal | GO sheets could be fabricated into 3D fine size nanofibers scaffold by the controlled assembly of GO sheets. The obtained G-NFs exhibited recoverable electrical conductivity, soft Acceptable physical characteristic and excellent biocompatibility and physicochemical stability. An unprecedented accelerated growth and development of the primary motor neurons was achieved by using the G-NFs for cellular electrical stimulation in a long-term culture period. | [178] | ||
| Hydrogel | Carbon nanotube | High-pressure carbon monoxide conversion synthesis | High-pressure carbon monoxide conversion synthesis | - | Schwann cell (SC)-glial | No | An electrically-conductive SWNT collagen biomaterial may be suitable for neural tissue engineering and is able to sustain populations of SC. | [179] | |
| CNT/collagen | Mixing | Coating | Four-point probe | Pc12 | No | Enhanced electrical activity and have shown positive in vitro biocompatibility results offering further evidence that SWNT-based materials have an important role in promoting neuronal regeneration. | [180] | ||
| CNT/PEDOT | - | Coating | - | - | No | Organic conductors provide safe electrical stimulation of tissue while avoiding undesirable chemical reactions and cell damage. | [181] | ||
| PANi | Nanofibers | PANi/poly (ɛ-caprolactone/gelatin) | Electrospinning | Coating | - | Nerve stem cells | 1.5 V for 15, 30, and 60 min | Conductive nanofibrous scaffolds are suitable substrates for the attachment and proliferation of nerve stem cells. | [182] |
| PANi/PLLA | Electrospinning | Blending | - | Nerve stem cells | Steady potential of 1.5 V for a period of 60 min | implication of electrical stimulation of nerve stem cells on conducting polymeric scaffolds towards neurite elongation that could be effective for nerve tissue regeneration | [21] | ||
| PANi/PLCL | Electro spinning | Blending | 4-point probe | Pc12 | No | Electro active fibers may hold Promise as a guidance scaffold for neuronal tissue engineering. | [183] | ||
| PANi/PCL | Ring-opening polymerization | Blending | 4-probe technique | HaCaT cell line | No | These scaffolds are non-cytotoxic. These degradable electroactive tubular scaffolds are good candidates for neural tissue engineering application | [184] | ||
| PANi | Oxidation of aqueous solutions of aniline by ammonium peroxydisulfate (APS) at 30 °C | Oxidation | - | Human mesenchymal stem cells | Yes/electric field (DC)was applied for 10 min, and the same sequence was repeated again over an interval of 24 h | The present work establishes the key role of intermittent and systematic delivery of electric stimuli as guidance cues in promoting neural-like differentiation of hMSCs, when grown on electroconductive Substrates. | [144] | ||
| Hydrogel | PANi/PEGDA | UV photopolymerization | Blending | 2-probes | PC12-hMSC | Yes/AC current amplitude of 0.01 mA over a frequency range from 1 to 20 × 103 Hz | PANi also improve the biological response of PC12 | [146] | |
| PANi/Collagen | Chemical synthesis | Coating | 4-point probes | PC12 | No | PANI and PEDOT nanofibres were found to be cytocompatible with both cell types and the best results were obtained with a low Concentration (0.5 wt%) of PANI. | [185] | ||
| PANi/Chitosan/Gelatin | Stirring | Mixing | 4-point probe | Schwann cells from infant human sciatic nerves | No | Porous conductive chitosan/gelatin/PAG scaffold having proper conductivity | [186] | ||
| PEDOT | - | Decellular nerve scaffolds | Electrochemical method | Coating | Cyclic voltammetry | - | No | Low concentrations of PEDOT on Decellular nerve scaffolds provided significant increases in electro active properties | [187] |
| Conductive Material | Scaffold Type | Scaffold Material | Fabrication Method | Applying Technique | Confirmation Method | Cell Type | Electrical Stimulation/Preferences | Major Outcome | Reference |
|---|---|---|---|---|---|---|---|---|---|
| PPy | Film | PPy | electrochemically polymerized | Electrochemically polymerized | 4-probe technique | HUVEC | No | This bioactive conductive platform provides a functional surface capable of tethering biomolecules that direct cell behavior without the drawback of reduced conductivity. | [217] |
| PPy | Electrochemical synthesis of PPy film then doped with HA and coated with collagen IV followed by Matrigel | Electrochemical | Cyclic voltammetry | Vascular smooth muscle cell | Yes/50 mA sinusoidal, 0.05, 5 and 500 Hz for 24 and 96 h | Vascular smooth muscle cells (VSMCs) cultured on a CP substrate and subject to electrical stimulation not only exhibit enhanced proliferation but can be simultaneously encouraged to increase contractile protein expression. | [215] | ||
| PCL/PPy | Heat-pressed | Chemical polymerization | 4-probe technique | Artrial myocyte | No | When CMs were cultured on the electrically-conductive PPy-PCL, more cells were observed to have peripheral localization of the gap junction protein connexin-43 (Cx43). Importantly, the velocity of calcium wave propagation was faster, and calcium transient duration was shorter for CM monolayers on PPy-PCL relative to cells on PCL. | [210] | ||
| Nanofibers | PCL/gelatin/PPy | Elecrospinning | Co-elecrospinning | Surface resistivity by standard four-probe method | Rabbit Cardiomyocyte | No | By increasing the concentration of PPy in the composite, the average fiber diameters reduced, and the tensile modulus increased. In addition, this scaffold promote cell attachment, proliferation, interaction, and expression of cardiac-specific proteins. | [192] | |
| Porous scaffold | PPy | Electropolymerization deposition within a sacrificial agarose gel template | Electropolymerization | 4-probe technique | HUVEC | No | The porous-structured PPy increased the viability of HUVECs. The higher viability of HUVECs on the porous-structured PPy can be attributed to the increased surface area and three-dimensionally hollow micro-/nanostructures, which can facilitate cell distribution and adhesion. | [218] | |
| Hydrogel | Alginate/PPy | Bivalent crosslinking | Blending | 4-probe technique | HUVEC | No | Addition of PPy mediated cell attachment and proliferation. local injection of polymer blend in to the infarct zone yielded significantly higher levels of arteriogenesis at 5 weeks post-treatment. Also, this scaffold significantly enhanced infiltration of myofibroblasts into the infarct area. | [216] | |
| PANi | Film | PANi | Heating and vacuum drying | Heating and vacuum drying | Surface resistivity by standard four-probe method | H9c2 | No | Both conductive and non-conductive form of PANi improve cell attachment and proliferation in comparison with Polystyrene plate. | [219] |
| PCL | Solvent casting | Blending and nanoneedles | picometer/voltage source meter | hMSC and cardiomyocyte-like cell | No | PANi short fibers provide a more efficient transfer of electric signal. this electrically conductive environment are able to stimulate the cell differentiation to cardiomyocites, for a successful use in the myocardium muscle regeneration. | [203] | ||
| PU containing aniline pentamer/PCL | Casting | PU containing aniline pentamer | 4-probe technique | HUVEC and L929 | No | Bioelectroactive polyurethane is a platform substrate to study the effect of electrical signals on cell activities and to direct desirable cell function for tissue engineering applications. | [71] | ||
| poly(glycerol-sebacate)/PANi | Solvent casting | Blending | 4-probe technique | C2C12 | No | The novel PANi–PGS composites, being able to maintain their electrical conductivity, not only have the potential to modulate cellular function but, when fabricated in 3-D porous scaffolds, also hold promise to serve as carrier and delivery vehicles of functional cells to the myocardial infarct. | [220] | ||
| Nanofibers | Gelatin/PANi | Elecrospinning | Co- elecrospinning | Potentiostat/Galvanostat | H9c2 | No | By increasing the amount of PANi the average fiber size reduced and the tensile modulus increased. This scaffold supported cell attachment and proliferation. | [38] | |
| Nanofibers contained PANi nanotubes modified by polyglycerol dendrimers | Electrospinning | Electrospinning | - | Cardiac myocytes and CHO cell | No | The PGLD-coated PANINTs showed noncytotoxic effects to Chinese hamster ovary cells. It was observed that the application of microcurrent stimulates the differentiation of cardiac cells cultured on these scaffolds. | [194] | ||
| Composite nanofibers of PANi and PLGA | Electrospinning | Co-elecrospinning | 4-probe technique | Neonatal cardiomyocyte | No | During incubation, the adhered CMs became associated with each other and formed isolated cell clusters; the cells within each cluster elongated and aligned their morphology along the major axis of the fibrous mesh. | [209] | ||
| Porous scaffold | PU containing aniline pentamer/PCL | Compression molding | PU containing aniline pentamer | 4-probe technique | Neonatal cardiomyocyte | No | Scaffolds supported neonatal CMs adhesion and growth, showing more extensive effect on the expression of the cardiac genes involved in muscle contraction and relaxation and cytoskeleton alignment. | [221] | |
| Carbon | Film | PLGA-CNF composite | Heating and vacuum drying | Blending | Multimeter | Cardiomyocyte and neuron | No | PLGA:CNF materials are conductive, and that the conductivity is related to the amounts of CNF. Also, CM and neuron density increases with greater amounts of CNF in PLGA. | [190] |
| PLGA-CNF composite | Heating and vacuum drying | Blending | Multimeter | Human cardiomyocyte | No | Results showed that PLGA:CNF materials were conductive. Furthermore, results indicated that CM density increased with greater amounts of CNFs of 200nm in diameter in PLGA. | [222] | ||
| PLGA-CNF composite | Heating and vacuum drying | Blending | - | Aortic endothelial, 3T3 and cardiomyocyte | Yes/rectangular, 2 nm, 5 V/cm, 1 Hz for 24, 72 and 120 h | This composite can stimulate CM growth and activity while hindering fibroblast and endothelial cell growth. During continuous electrical stimulation, CM cell density increased in comparison to its static counterparts after 24, 72 and 120 h. A minor rise in Troponin I excretion in electrical stimulation compared to static conditions indicated nominal CM cell function during cell experiments. | [208] | ||
| MWNT | Deposition/drying to have a thin film | Deposition/drying | - | Neonatal rat ventricular myocyte | No | The proliferative capacity of CMs on carbon nanotubes was significantly higher. Cells grown on CNT substrates displayed values of resting potential more negative than those of gelatin cells. | [205] | ||
| MWNT | Deposition/drying to have a thin film | Deposition/drying | AFM with a Veeco Nano Scope V in tapping mode | Neonatal rat ventricular myocyte | No | CNT scaffolds promote CM growth and maturation by altering the gene expression program, implementing the cell electrophysiological properties and improving networking and maturation of functional syncytia. | [206] | ||
| Collagen/SWNT | Deposition/Vacuum drying | Dispersion | Multimeter | Neonatal cardiomyocyte | No | The addition of CNTs remarkably increased intercalated disc related protein expression and enhanced ID assembly and functionality. Notably, CNTs remarkably accelerated gap junction format ion via activation of the b1-integrin-mediated FAK/ERK/GATA 4 pathway. | [210] | ||
| Nanofibers | PU/MWNT | Electrospinning | Co-elecrospinning | - | HUVEC | No | Experimental results indicated that the nanofibrous scaffold of MWNT/PU exhibited promotional influence on the cell proliferation. It was also observed that the scaffold possessed an advantage of supporting Endothelial Cells migrating and aggregating along the axis of the aligned nanofibers. It was also demonstrated that the endothelial cells growing on the scaffold expressed non-thrombogenic phenotype with low tissue factor released. | [223] | |
| PLA/SWNT | Electrospinning | Co-elecrospinning | Electrical resistance measure | hMSC | Yes/0.15 V/cm for 2 ms duration at a frequency of 1 Hz | After electrical stimulation the cells reoriented perpendicular to the direction of the current and adopted an elongated morphology. An upregulation in a range of cardiac markers was detected. | [224] | ||
| poly(e-caprolactone)/MWNT | Electrospinning | Co-elecrospinning | Electrochemical impedance spectroscopy | hMSC | Yes/500 v/m and 5 ms pulse width at 1 Hz, for 10 min per day for 4 days | hMSC differentiation can be enhanced by either culturing in electrically conductive, carbon nanotube-containing composite scaffolds without electrical stimulation in the presence of 5-azacytidine, or extrinsic electrical stimulation in nonconductive poly(e-caprolactone) scaffolds without CNT and azacytidine. | [204] | ||
| poly(glycerolsebacate)/gelatin/CNT | Electrospinning | Co-elecrospinning | Electrochemical Impedance spectrometry | Cardiomyocyte | No | Aligned CNT-PG scaffold exhibited superior mechanical properties with enhanced CM beating properties. | [196] | ||
| Porous scaffold | Chitosan/CNF | Precipitation | Dispersion | Picoammeter -Voltage source | Neonatal rat cardiomyocytes | No | Incorporation of CNFs into porous chitosan scaffolds improved the properties of cardiac tissue constructs, presumably through enhanced transmission of electrical signals between the cells. | [225] | |
| Collagen/SWNT | Gelation at 37 °C | Dispersion | - | Rat aortic smooth muscle cells (RASMC) | No | Cell viability in all constructs was consistently above 85% at both Day 3 and Day 7. | [226] | ||
| Hydrogel | pHEMA/rosette nanotubes/CNF | Radical polymerization | Dispersion | Multimeter | Cardiomyocyte | No | CM density increased after 4 h, 1 day, and 3 days with greater amounts of CNFs and greater amounts of RNTs in pHEMA. In fact, wettability, conductivity, and surface nanoroughness become greater with greater amounts of CNFs and RNTs. | [188] | |
| Gelatin/SWNT | Glutaraldehyde crosslinking | Dispersion | Multimeter | Neonatal rat cardiomyocytes | No | SWCNTs could provide cellular microenvironment in vitro favorable for cardiac contraction and the expression of electrochemical associated proteins. Upon implantation into the infarct hearts in rats, this scaffold structurally integrated with the host myocardium. | [212] | ||
| Gelatin methacrylate/CNT | Photopolymerization under UV light | Dispersion | Electrochemical Impedance spectrometry | Neonatal rat cardiomyocytes | No | myocardial tissues cultured on 50 µm thick CNTGelMA showed 3 times higher spontaneous synchronous beating rates and 85% lower excitation threshold, compared to those cultured on pristine GelMA hydrogels. | [199] | ||
| Poly (N-isopropylacrylamide) modified with SWNT | In situ forming (gelled above 32 °C) | Dispersion | Electrochemistry workstation | Brown adipose-derived stem cell | No | In vitro study showed that the PNIPAAm/SWNTs hydrogel demonstrated significantly higher bioactivities to encapsulated brown adipose -derived stem cells compared with onefold PNIPAAm hydrogel, including promoting cell adhesion and proliferation. When used as carrier for intramyocardial delivery of BASCs after MI, the PNIPAAm/SWNTs hydrogel significantly enhanced the engraftment of seeding cells in infarct myocardium and augmented their therapeutic efficacies in MI. | [207] | ||
| gelatin/chitosan/SWNT | Lyophilization | Dispersion | Voltage sensitive dye and an ionoptix system | Ventricular myocyte | No | These engineered tissues achieve excitation conduction velocities similar to native myocardial tissue and could function as a full-thickness patch for several cardiovascular defect repair procedures. | [227] | ||
| Au | Nanofibers | Polyvinyl alcohol/Bovine serum albumin/Au nanoparticles | Electrospinning | Dispersion | - | hMSC | No | AuNPs loaded nanofibrous scaffolds facilitates the functional differentiation of MSCs. | [123] |
| Hydrogel | HEMA/Au nanoparticles | Photopolymerization under UV light | Dispersion | Electrometer | Neonatal rat cardiomyocyte | No | Neonatal rat CMs exhibited increased expression of connexin 43 on hybrid scaffolds relative to HEMA with or without electrical stimulation. | [36] | |
| Alginate/Au nanowire | Bivalent crosslinking | Dispersion | Electrochemical Impedance spectrometry | Neonatal rat cardiomyocyes and fibroblasts | No | Incorporating gold nanowires within alginate scaffolds can bridge the electrically resistant pore walls of alginate and improve electrical communication between adjacent cardiac cells. So thicker and better aligned tissue grown on this scaffold and cells in these tissues contracted synchronously. | [211] | ||
| melanin | Nanofibers | Poly(L-lactide-co-3-caprolactoe)/gelatin/melanin | Electrospinning | Co-elecrospinning | 4-probe technique | Human cardiac myocyte | Yes/rectangular, 150 ms, 1 V/cm, 1 Hz for 4 and 8 days | Conductive nanofibers containing 10% melanin promote cell interaction with expression of cardiac-specific proteins compared to other scaffolds. Electrical stimulation through the scaffolds showed enhanced cell proliferation and the expression of connexin-43. | [132] |
| silicon | Hydrogel | Agarose/silicon nanowire | Casting | Dispersion | - | Rat neonatal cardiac cells and human induced pluripotent stem cell | No | Incorporation of a trace amount of electrically conductive silicon nanowires (e-SiNWs) in otherwise scaffold-free cardiac spheroids can form an electrically conductive network, leading to synchronized and significantly enhanced contraction, resulting in significantly more advanced cellular structural and contractile maturation. | [119] |
| PEDOT | Porous scaffold | poly(styrenesulfonate)/PEDOT | Ice-templating | Physical crosslinking | Electrochemical transistor | 3T3 cell | No | The scaffolds support the growth of mouse fibroblasts for 7 days, and are able to electrically control cell adhesion and pro-angiogenic capability. These 3D matrix-mimicking platforms offer precise control of protein conformation and major cell functions, over large volumes and long cell culture times. | [228] |
| Conductive Material | Type | Composition | Fabrication Method | Applying Technique | Confirmation Method | Cell Type | Electrical Stimulation/Duration | Major Out Come | Reference |
|---|---|---|---|---|---|---|---|---|---|
| PPy | Film | PPy/Polypropylene fumarate | Rapid prototyping technique | Coating | - | Osteoblasts | No | Osteoblasts maintained their phenotype on PPF scaffolds with and without coatings. Thus, these Scaffolds could be appropriate candidates for our future in vivo studies | [243] |
| PPy/PLLA/Heparin | Water-in-oil emulsion | Blending | - | Osteoblasts-like Saos-2 cells | Yes/200 mV/mm 6h per day | The electrical stimulation was able to promote osteoblast adhesion and growth, resulting in significantly higher calcium and phosphate content in the mineral deposition of the electrically stimulated membranes. | [240] | ||
| PPy/PLA | Extruding | Coating | - | Human adipose stem cells | Yes/DC voltage repeated at a frequency of 1 or 100 Hz, ES for 4 h/day. | The alkaline phosphatase (ALP) activity of the hASCs was generally higher in PLA-PPy seeded scaffolds | [244] | ||
| PPy/poly(L-lactide) | Chemical synthesis | Blending | - | Saos-2 cells | Yes/Four potential intensitieswere applied to the conductive membranes, that is 100, 200, 300, and 400 mV/mm. The cells were stimulated for 2, 4, 6 and 8 h at each ES intensity | This work demonstrated that important osteoblast markers can be modulated with specific electrical stimulation parameters mediated through CPs substrates, providing a unique strategy for bone tissue engineering. | [241] | ||
| PPy/Hyaluronic acid or chondroitin sulfate | Chemical synthesis | Coating | - | Human adipose stem cells | Yes/Samples were stimulated for 4 h a day for 14 days with a biphasic electric current (BEC) of ± 0.2 V amplitude, 2.5 ms pulse width and 100 Hz pulse repetitionFrequency. | PPy–chondroitin sulfatein particular is a potential osteogenic scaffold Coating for bone tissue engineering. | [230] | ||
| Carbon | Film | CNT/PCL | Salt-leaching technique | Coating | - | Human kidney fibroblasts cells | Yes/Varying voltages (0.5 and 0.7 V) were applied in a cathodic direction to the scaffold in medium | Materials of this type of composition have potential merit as a biomaterial | [242] |
| CNT/poly(ester amide)s | Chemical synthesis | Mixing | Cyclic Voltammetry | Preosteoblastic MC3T3-E1 | Yes/The square wave, frequency of 50 Hz, 50% duty cycle, And electrical potential of 0.2 V was adopted in the experiment. | The PEA-g-TA copolymers stimulated by pulsed electrical signal could serve to promote the differentiation of MC3T3-E1 cells | [245] | ||
| Carbon nanotube | - | - | - | Osteoblasts, chondrocytes smooth muscle cells and fibroblasts | No | Studies are evaluated with an emphasis on understanding the mechanisms through which 17 carbon nanotubes interact with biological systems. | [246] | ||
| Graphene/collagen | Chemical synthesis | Coating | - | MC3T3-E1 | No | Scaffolds modified with a suitable concentration of GO are useful as a bioactive material for tissue engineering | [247] | ||
| Graphene/citrate-stabilized HA hydrocolloids | Self-assembly | mixing | Cyclic Voltammetry | Mouse multipotent mesenchymal stromal cells | No | The resulting graphene-HA gels are highly porous, strong, electrically conductive and Biocompatible, making them promising scaffolds for bone tissue engineering. This method can be applied to produce a variety of free-standing 3D graphene-based nanocomposites with Unprecedented homogeneity. | [248] | ||
| Graphene/TCP | Oxidation | Coating | - | BMMSCs | No | Combination of graphene and goat mesenchymal stemcells provides a promising construct for bone tissue engineering. | [249] | ||
| Hydrogel | Graphene/Chitosan | Chemical synthesis | - | Cyclic Voltammetry | L-929 | No | These chitosan-graphene composites show great promise for use as conducting substrates for the growth of electro-responsive cells in tissue engineering. | [250] | |
| PANi | PANi/PGA-g-TA/PLL-g-TA | Ring-opening polymerization | Coating | - | Preosteoblastic MC3T3-E1 | Yes/The square wave, frequency of 100 Hz, 50% duty cycle, and electrical potential of 0.5 V were adopted | The comprehensive effects through coupling electroactive scaffolds with electrical stimulus are better to develop bioelectric strategies to control cell functions for bone regeneration | [236] | |
| PANi/PLLA | Electrospinning | Coating | Cyclic Voltammetry | Preosteoblastic MC3T3-E1 | Yes/The square wave with frequency of 50 Hz, electrical potential of 0.2 Vand 50% duty cycle was adopted. The samples were stimulated for 2 h every day, respectively. | Biodegradable and electroactive AP-g-GA/PLLA nanofibers had potential application in vivo as bone repair scaffold. | [238] | ||
| PANi/PCL/gelatin composite | Electrospining | Mixing | Cyclic Voltammetry | - | No | By incorporation of conductive PANi and bioactive particles and drugs (osteogenon, calcium phosphate nanoparticles) within an electrospun PCL/gelatin composite scaffold, we have obtained a biocompatible, bioactive, hybrid scaffold system, which provides an electrically conductive environment. | [251] | ||
| PEDOT | Film | PEDOT/poly(3,4-ethylenedioxythiophene) poly(4-styrene sulfonate) | Chemical synthesis | Blending | - | Human mesenchymal stem cells | No | Conductive scaffolds are not only structurally more favorable for bone tissue engineering, but also can be a step forward in combining the tissue engineering techniques with the method of enhancing the bone healing by electrical stimuli. | [237] |
| Conductive Material | Scaffold Type | Scaffold Material | Fabrication Method | Applying Technique | Confirmation Method | Cell Type | Electrical Stimulation/Preferences | Major Outcome | Reference |
|---|---|---|---|---|---|---|---|---|---|
| PANi | Nanofibrous | Poly(L-lactide-co-e-caprolactone)/PANi | Electrospinning | Mixing | 4-probe technique | Human dermal fibroblasts, NIH-3T3 and C2C12 myoblasts | Yes/DC 0–200 mA | Addition of PANi to scaffold causes improved cell attachment. In addition, the growth of NIH-3T3 fibroblasts is enhanced under the stimulation of various direct current flows. The incorporation of PANi improved the metabolic activity of all cell types treated in a concentration-dependent manner. | [75] |
| Poly(L-lactide-co-3-caprolactone)/PANi | Electrospinning | Mixing | 4-probe technique | C2C12 myoblasts | No | The prepared PLC L/PANi fibers showed no significant difference in fiber diameter or contact angle, regardless of the incorporation of PANi.after 4 days of culture, the number of cells positive for sarcomeric myosin was 3.6-times greater on the electrically conductive fibers. | [258] | ||
| Polycap rolactone/PANi | Electrospinning | Mixing | Potentiostat/galvanostat | C2C12 myoblasts | No | Myosin heavy chain expression, multinucleate myotube formation, and the expression of differentiation specific genes, the differentiation of myoblasts on PCL/PANi nanofibers was strongly dependent on both nanofiber alignment and PANi concentration. | [262] | ||
| polycap rolactone/PANi | Electrospinning | Mixing | 4-probe technique | C2C12 myoblasts | No | The aligned nanofibers could guide myoblast orientation and promote myotube formation. In addition, electrically conductive nanofibers further enhanced myotube maturation compared with non-conductive scaffolds. | [261] | ||
| Chitosan grafted aniline tetramer | Electrospinning | Mixing | Cyclic voltammetry | C2C12 myoblasts | No | The chitosan grafted aniline tetramer substrates and their degradation products are not cytotoxic and could improve the cell adhesion and proliferation of C2C12 myoblasts compared to chitosan. | [82] | ||
| Tetraaniline-polylactide | Thermally induced phase separation | Blending | Cyclic voltammetry | C2C12 myoblasts | No | These electroactive degradable materials are nontoxic and enhance the adhesion and proliferation of the C2C12 myoblast cells compared to polylactide, probably because of the more proteins adsorbed on the electroactive materials than that of polylactide. The electroactive materials significantly improved the cell proliferation of C2C12 myoblasts under ES. | [263] | ||
| Hydrogel | Gelatin–graft–polyaniline | Crosslinking by genipin | Grafting | Cyclic voltammetry | C2C12 myoblasts and MSC | No | The conductivity of this insitu forming degradable hydrogel in the swollen is proportional to PANi content in the materials. The non-cytotoxicity of the hydrogels was confirmed via cell adhesion and proliferation. | [64] | |
| Chitosan-graft-PANi/Oxidized Dextran | In situ forming | Grafting | Cyclic voltammetry | MSC and C2C12 myoblasts | No | This anitibacterial hydrogel had a fast mass loss in the first 5.5 weeks with a linear degradation trend. After that time, the degradation speed of the hydrogels went slowly and still showed linear degradation kinetics. in addition, with the increase of PANi content, the dead cell were less and they exhibited a higher cell proliferation. Furthermore, this hydrogel could form in short time by injection in vivo. | [67] | ||
| PPy | Film | PPy doped with ECM components | Galvanostatically deposition | Galvanostatically deposition | Gamray Impedance system | Skeletal muscle myoblasts | No | Polymer films including PPy/HA and PPy/para-toluene sulphonic acid showed good support for myoblast proliferation but were poor in terms of adhesion and differentiation. In contrast, PPy/poly (2-methoxy-5 aniline sulphonic acid) supported a lower degree of proliferation, but good cell adhesion and differentiation. | [256] |
| PPy/MWNT | Aerogel sheet | MWNT | Deposition | Deposition | - | Murine primary muscle cell | Yes/0.125 mA/cm2 biopolar square wave, 10 Hz, 8 h per day for 3 days | Application of electrical stimulation to myoblasts on nanostructured MWNT/para-toluene sulphonic acid doped PPy platforms led to significant enhancements in total myo-nuclear density and myoblast differentiation. | [260] |
| carbon | Nanofibrous | Polyurethane/CNT | Electrospinning | Mixing | 4-probe technique | C2C12 myoblasts | Yes/22 V/cm, 20 Hz, biphasic, twice a day on the last 2 days of culture | After electrical stimulation, the number of multinucleated myotubes on the electrospun polyurethane CNT scaffolds was significantly larger than that on nonconductive electrospun polyurethane scaffolds. In the absence of electrical stimulation, myoblasts also differentiated on the electrospun polyurethane CNT scaffolds, as evidenced by expression of Myf-5 and myosin heavy chains. | [252] |
| Styrene/butadiene/styrene/CNT | Electrospinning | Mixing | slope of I–V curves measured with an automated Keithley 487 picoammeter/voltage source | C2C12 myoblasts | No | With the introduction of CNT in the polymers, the Styrene/butadiene/styrene samples proved to be cytotoxic contrarily to styrene–ethylene/butylene–styrene samples. The styrene–ethylene/butylene–styrene composites are thus a suitable candidate for biomedical applications, including the development of scaffold membranes for tissue engineering applications. | [264] | ||
| PTh | Film | Poly(octanoic acid 2-thiophen-3-yl-ethyl ester) | Spin coating | Spin coating | Cyclic voltammetry | Primary skeletal muscle myoblasts and C2C12 myoblasts | No | The polymer films supported the proliferation and differentiation of both primary and transformed skeletal muscle myoblasts. In addition, aligned electrospun fibers formed from the polymers provided scaffolds for the guided differentiation of linearly aligned primary myotubes, suggesting their suitability as three-dimensional substrates for the in vitro engineering of skeletal muscle tissue. | [34] |
| Au | Nanofibrous | Poly(L-lactic acid)/Au nanoparticles | Electrospinning | Mixing | Measuring electrical resistance | Rat primary muscle cell | No | The first cell study showed low cell proliferation on the Au–PLLA scaffolds; however, the second cell study showed that this was not due to Au Nps toxicity. Instead, low cell proliferation may be a marker for myotube differentiation and fusion. By electrospinning higher amounts of Au Nps with PLLA, a conductive, biocompatible and biodegradable scaffold can be manufactured for skeletal muscle tissue engineering that could possibly use lower voltages to increase myotube formation. | [124] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saberi, A.; Jabbari, F.; Zarrintaj, P.; Saeb, M.R.; Mozafari, M. Electrically Conductive Materials: Opportunities and Challenges in Tissue Engineering. Biomolecules 2019, 9, 448. https://doi.org/10.3390/biom9090448
Saberi A, Jabbari F, Zarrintaj P, Saeb MR, Mozafari M. Electrically Conductive Materials: Opportunities and Challenges in Tissue Engineering. Biomolecules. 2019; 9(9):448. https://doi.org/10.3390/biom9090448
Chicago/Turabian StyleSaberi, Azadeh, Farzaneh Jabbari, Payam Zarrintaj, Mohammad Reza Saeb, and Masoud Mozafari. 2019. "Electrically Conductive Materials: Opportunities and Challenges in Tissue Engineering" Biomolecules 9, no. 9: 448. https://doi.org/10.3390/biom9090448
APA StyleSaberi, A., Jabbari, F., Zarrintaj, P., Saeb, M. R., & Mozafari, M. (2019). Electrically Conductive Materials: Opportunities and Challenges in Tissue Engineering. Biomolecules, 9(9), 448. https://doi.org/10.3390/biom9090448








