Organellar DNA Polymerases in Complex Plastid-Bearing Algae
Abstract
1. Introduction
2. Materials and Methods
2.1. BLAST Search of POP Homologous Genes
2.2. Phylogenetic Analysis
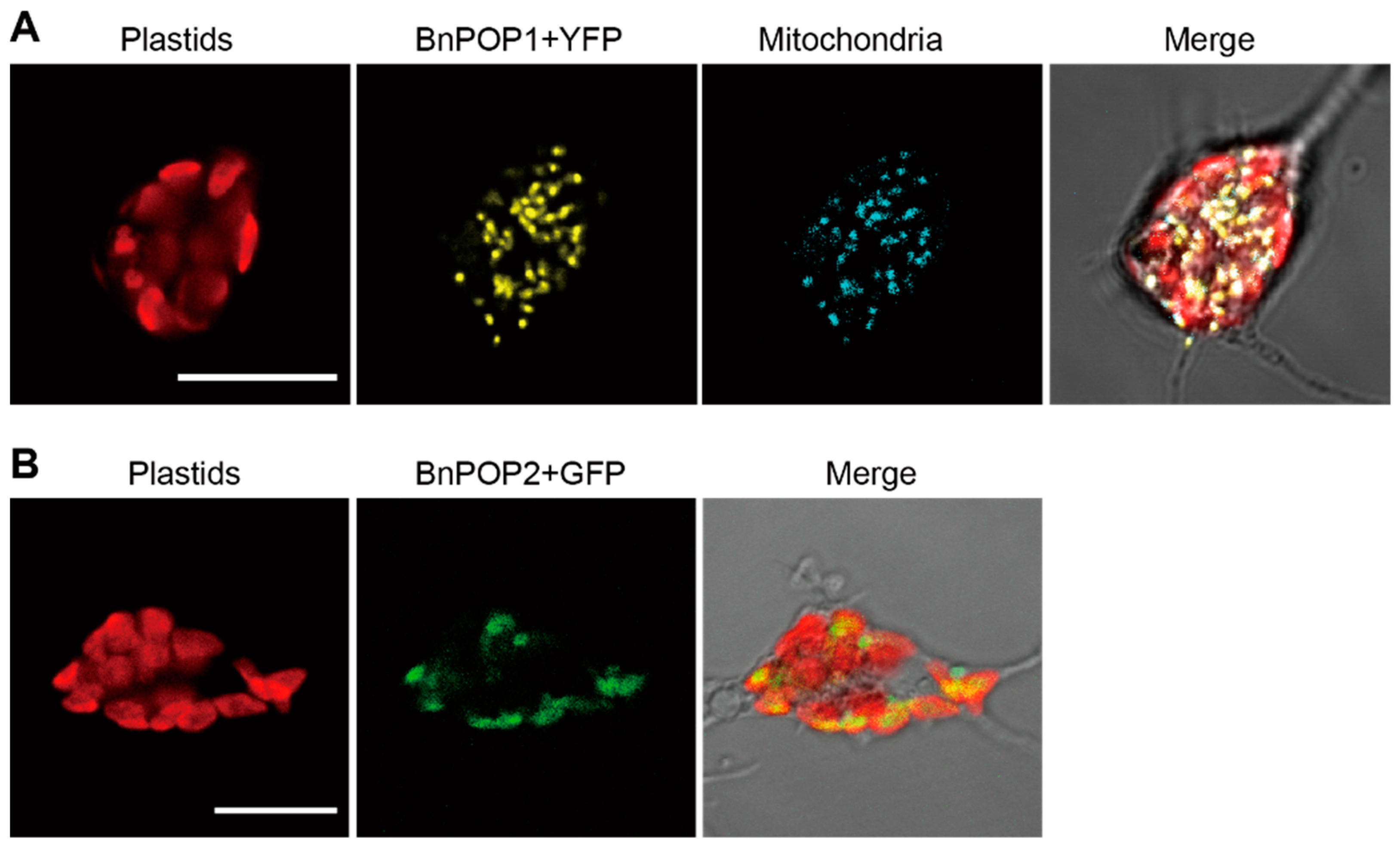
2.3. Subcellular Localization of Chlorarachniophyte POPs
2.4. In Silico Prediction of Targeting Signals and Functional Domains
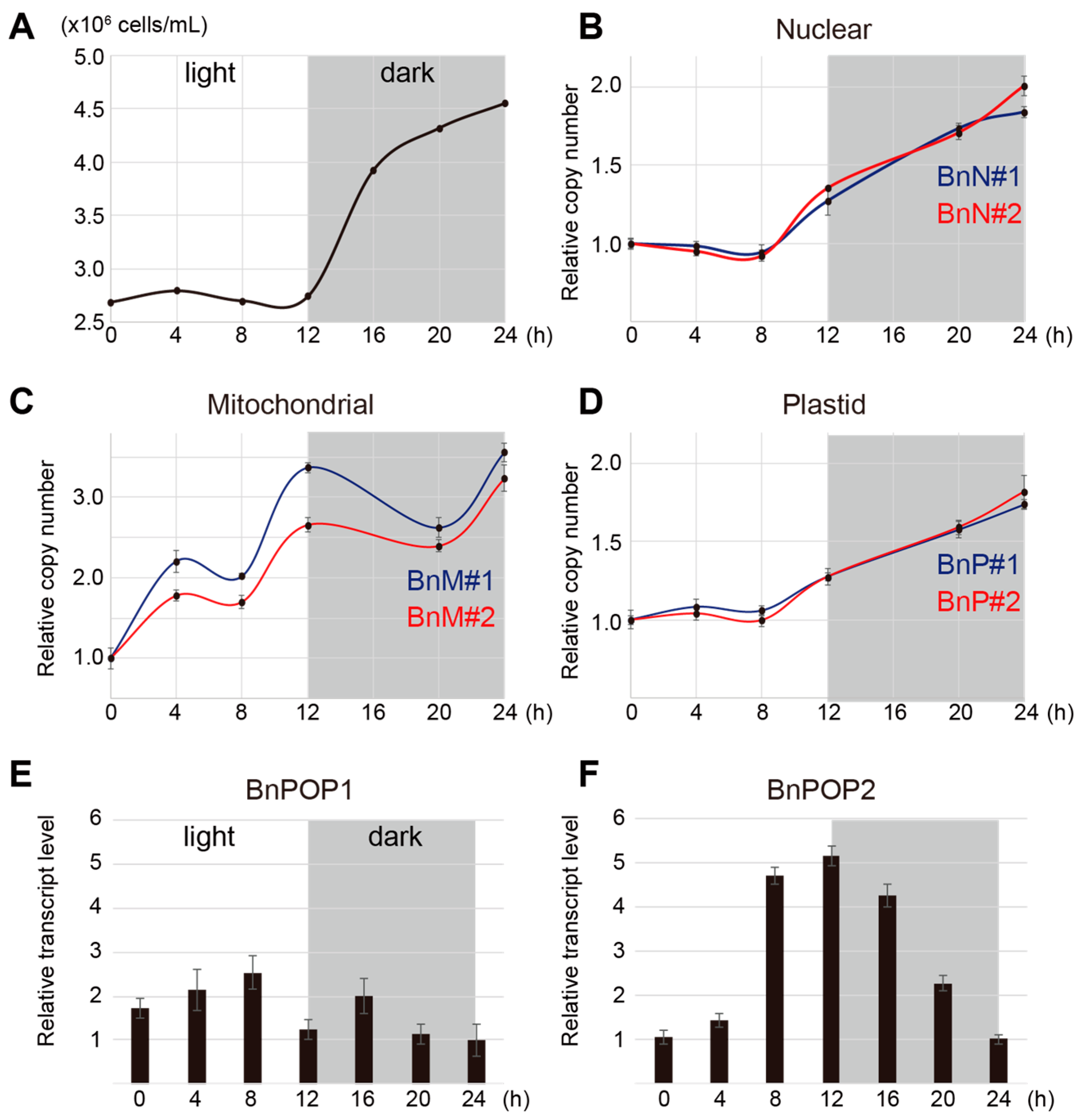
2.5. Real-time Quantitative PCR
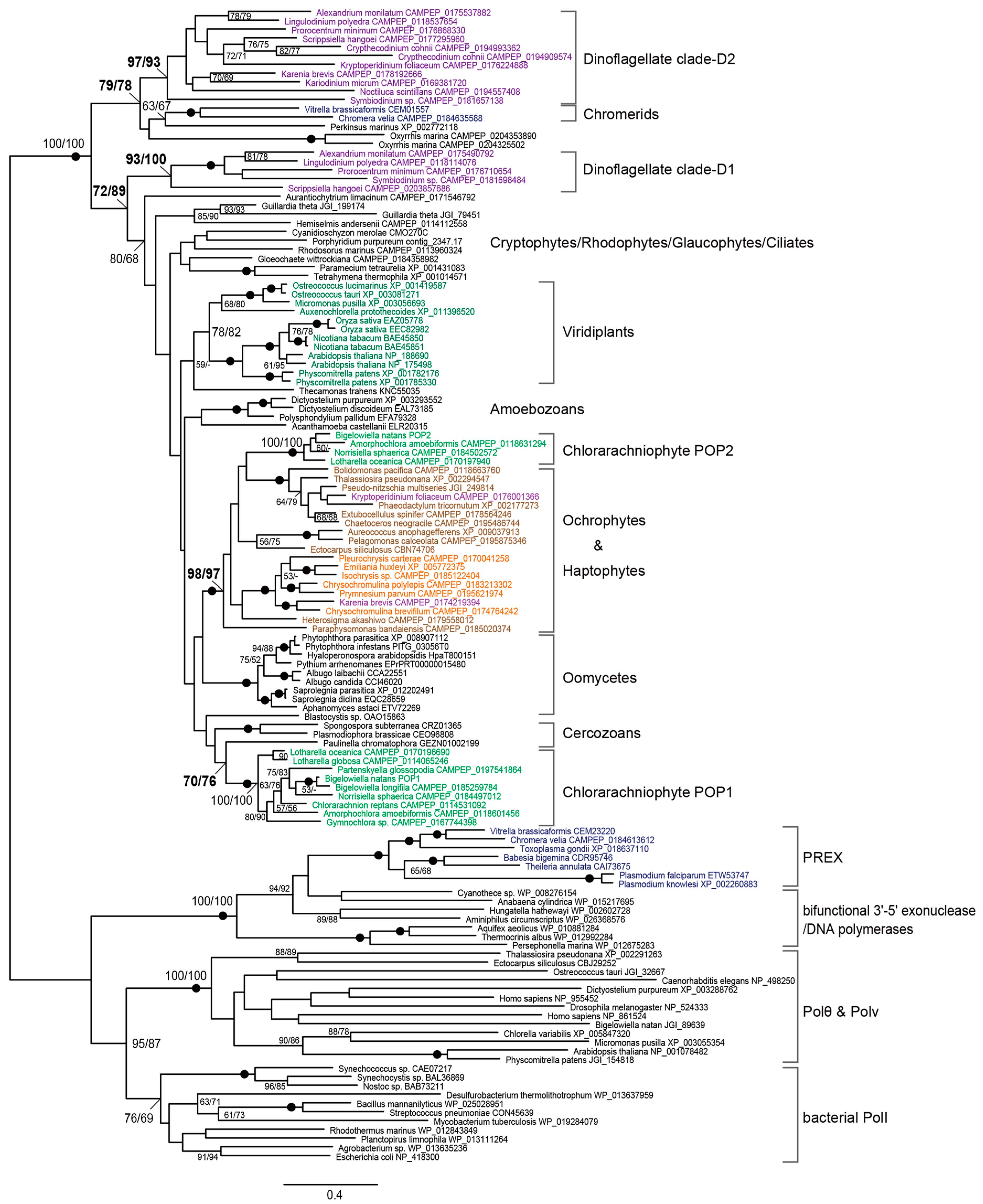
3. Results
3.1. Identification of POP Genes in Diverse Algae and Protists
3.2. Localization Experiments for Chlorarachniophyte POP Proteins
3.3. Timing of Organellar DNA Replication in B. natans
3.4. Prediction of Targeting Signals in POP Sequences
4. Discussion
4.1. Evolution of Two Distinct POPs in Chlorarachniophytes
4.2. POP Evolution in Alveolates
4.3. POP Evolution in Haptophytes and Ochrophytes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Archibald, J.M. Endosymbiosis and eukaryotic cell evolution. Curr. Biol. 2015, 25, R911–R921. [Google Scholar] [CrossRef] [PubMed]
- Price, D.C.; Chan, C.X.; Yoon, H.S.; Yang, E.C.; Qiu, H.; Weber, A.P.M.; Schwacke, R.; Gross, J.; Blouin, N.A.; Lane, C.; et al. Cyanophora paradoxa genome elucidates origin of photosynthesis in algae and plants. Science 2012, 335, 843–847. [Google Scholar] [CrossRef]
- Ponce-Toledo, R.I.; Deschamps, P.; López-García, P.; Zivanovic, Y.; Benzerara, K.; Moreira, D. An early-branching freshwater cyanobacterium at the origin of plastids. Curr. Biol. 2017, 27, 386–391. [Google Scholar] [CrossRef]
- Gould, S.B.; Waller, R.F.; McFadden, G.I. Plastid evolution. Annu. Rev. Plant Biol. 2008, 59, 491–517. [Google Scholar] [CrossRef]
- Keeling, P.J. The number, speed, and impact of plastid endosymbioses in eukaryotic evolution. Annu. Rev. Plant Biol. 2013, 64, 583–607. [Google Scholar] [CrossRef]
- Waller, R.F.; Kořený, L. Plastid complexity in dinoflagellates: A picture of gains, losses, replacements and revisions. In Adv. Bot. Re. 2017, 84, 105–143. [Google Scholar]
- Burki, F. The Convoluted evolution of eukaryotes with complex plastids. In Adv. Bot. Res. 2017, 84, 1–30. [Google Scholar]
- Douglas, S.E.; Penny, S.L. The plastid genome of the cryptophyte alga, Guillardia theta: Complete sequence and conserved synteny groups confirm its common ancestry with red algae. J. Mol. Evol. 1999, 48, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Oudot-Le Secq, M.-P.; Grimwood, J.; Shapiro, H.; Armbrust, E.V.; Bowler, C.; Green, B.R. Chloroplast genomes of the diatoms Phaeodactylum tricornutum and Thalassiosira pseudonana: Comparison with other plastid genomes of the red lineage. Mol. Genet. Genomics 2007, 277, 427–439. [Google Scholar] [CrossRef] [PubMed]
- Christensen, A.C.; Lyznik, A.; Mohammed, S.; Elowsky, C.G.; Elo, A.; Yule, R.; Mackenzie, S. A Dual-domain, dual-targeting organellar protein presequences in Arabidopsis can use non-AUG start codons. Plant Cell 2005, 17, 2805–2816. [Google Scholar] [CrossRef]
- Mori, Y.; Kimura, S.; Saotome, A.; Kasai, N.; Sakaguchi, N.; Uchiyama, Y.; Ishibashi, T.; Yamamoto, T.; Chiku, H.; Sakaguchi, K. Plastid DNA polymerases from higher plants, Arabidopsis thaliana. Biochem. Biophys. Res. Commun. 2005, 334, 43–50. [Google Scholar] [CrossRef]
- Ono, Y.; Sakai, A.; Takechi, K.; Takio, S.; Takusagawa, M.; Takano, H. NtPolI-like1 and NtPolI-like2, bacterial DNA polymerase I homologs isolated from BY-2 cultured tobacco cells, encode DNA polymerases engaged in DNA replication in both plastids and mitochondria. Plant Cell Physiol. 2007, 48, 1679–1692. [Google Scholar] [CrossRef]
- Morley, S.A.; Nielsen, B.L. Chloroplast DNA copy number changes during plant development in organelle DNA polymerase mutants. Front. Plant Sci. 2016, 7, 57. [Google Scholar] [CrossRef]
- Moriyama, T.; Terasawa, K.; Sato, N. Conservation of POPs, the plant organellar DNA polymerases, in eukaryotes. Protist 2011, 162, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Seow, F.; Sato, S.; Janssen, C.S.; Riehle, M.O.; Mukhopadhyay, A.; Phillips, R.S.; Wilson, R.J.M.; Barrett, M.P. The plastidic DNA replication enzyme complex of Plasmodium falciparum. Mol. Biochem. Parasitol. 2005, 141, 145–153. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Chen, C.-Y.; Doerig, C.; Henriquez, F.L.; Roberts, C.W.; Barrett, M.P. The Toxoplasma gondii plastid replication and repair enzyme complex, PREX. Parasitology 2009, 136, 747–755. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Janouškovec, J.; Tikhonenkov, D.V.; Burki, F.; Howe, A.T.; Kolísko, M.; Mylnikov, A.P.; Keeling, P.J. Factors mediating plastid dependency and the origins of parasitism in apicomplexans and their close relatives. Proc. Natl. Acad. Sci. USA 2015, 112, 10200–10207. [Google Scholar] [CrossRef]
- Filée, J.; Forterre, P.; Sen-Lin, T.; Laurent, J. Evolution of DNA polymerase families: Evidences for multiple gene exchange between cellular and viral proteins. J. Mol. Evol. 2002, 54, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Graziewicz, M.A.; Longley, M.J.; Copeland, W.C. DNA polymerase γ in mitochondrial DNA replication and repair. Chem. Rev. 2006, 106, 383–405. [Google Scholar] [CrossRef]
- Moriyama, T.; Terasawa, K.; Fujiwara, M.; Sato, N. Purification and characterization of organellar DNA polymerases in the red alga Cyanidioschyzon merolae. FEBS J. 2008, 275, 2899–2918. [Google Scholar] [CrossRef]
- O’Donnell, M. Replisome architecture and dynamics in Escherichia coli. J. Biol. Chem. 2006, 281, 10653–10656. [Google Scholar] [CrossRef]
- Gowda, A.S.P.; Moldovan, G.L.; Spratt, T.E. Human DNA polymerase ν catalyzes correct and incorrect DNA synthesis with high catalytic efficiency. J. Biol. Chem. 2015, 290, 16292–16303. [Google Scholar] [CrossRef]
- Wood, R.D.; Doublié, S. DNA polymerase θ (POLQ), double-strand break repair, and cancer. DNA Repair 2016, 44, 22–32. [Google Scholar] [CrossRef]
- Moriyama, T.; Tajima, N.; Sekine, K.; Sato, N. Localization and phylogenetic analysis of enzymes related to organellar genome replication in the unicellular rhodophyte Cyanidioschyzon merolae. Genome Biol. Evol. 2014, 6, 228–237. [Google Scholar] [CrossRef]
- Keeling, P.J.; Burki, F.; Wilcox, H.M.; Allam, B.; Allen, E.E.; Amaral-Zettler, L.A.; Armbrust, E.V.; Archibald, J.M.; Bharti, A.K.; Bell, C.J.; et al. The Marine Microbial Eukaryote Transcriptome Sequencing Project (MMETSP): Illuminating the functional diversity of eukaryotic life in the oceans through transcriptome sequencing. PLoS Biol. 2014, 12, e1001889. [Google Scholar] [CrossRef]
- Suzuki, S.; Ishida, K.; Hirakawa, Y. Diurnal transcriptional regulation of endosymbiotically derived genes in the chlorarachniophyte Bigelowiella natans. Genome Biol. Evol. 2016, 8, 2672–2682. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Capella-Gutiérrez, S.; Silla-Martínez, J.M.; Gabaldón, T. trimAl: A tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 2009, 25, 1972–1973. [Google Scholar] [CrossRef]
- Nguyen, L.-T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef]
- Wang, H.C.; Minh, B.Q.; Susko, E.; Roger, A.J. Modeling site heterogeneity with posterior mean site frequency profiles accelerates accurate phylogenomic estimation. Syst. Biol. 2018, 67, 216–235. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; Von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef]
- Hirakawa, Y.; Nagamune, K.; Ishida, K. Protein targeting into secondary plastids of chlorarachniophytes. Proc. Natl. Acad. Sci. USA 2009, 106, 12820–12825. [Google Scholar] [CrossRef]
- Hirakawa, Y.; Burki, F.; Keeling, P.J. Dual targeting of aminoacyl-tRNA synthetases to the mitochondrion and complex plastid in chlorarachniophytes. J. Cell Sci. 2012, 125, 6176–6184. [Google Scholar] [CrossRef]
- Hirakawa, Y.; Kofuji, R.; Ishida, K. Transient transformation of a chlorarachniophyte alga, Lotharella amoebiformis (chlorarachniophyceae), with uidA and egfp reporter genes. J. Phycol. 2008, 44, 814–820. [Google Scholar] [CrossRef]
- Emanuelsson, O.; Brunak, S.; von Heijne, G.; Nielsen, H. Locating proteins in the cell using TargetP, SignalP and related tools. Nat. Protoc. 2007, 2, 953–971. [Google Scholar] [CrossRef]
- Small, I.; Peeters, N.; Legeai, F.; Lurin, C. Predotar: A tool for rapidly screening proteomes for N-terminal targeting sequences. Proteomics 2004, 4, 1581–1590. [Google Scholar] [CrossRef]
- Petsalaki, E.I.; Bagos, P.G.; Litou, Z.I.; Hamodrakas, S.J. PredSL: A Tool for the N-terminal sequence-based prediction of protein subcellular localization. Geno. Prot. Bioinfo. 2006, 4, 48–55. [Google Scholar] [CrossRef]
- Krogh, A.; Larsson, B.; von Heijne, G.; Sonnhammer, E.L. Predicting transmembrane protein topology with a hidden Markov model: Application to complete genomes. J. Mol. Biol. 2001, 305, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Hirakawa, Y.; Burki, F.; Keeling, P.J. Nucleus- and nucleomorph-targeted histone proteins in a chlorarachniophyte alga. Mol. Microbiol. 2011, 80, 1439–1449. [Google Scholar] [CrossRef]
- Hirakawa, Y.; Ishida, K. Polyploidy of endosymbiotically derived genomes in complex algae. Genome Biol. Evol. 2014, 6, 974–980. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Moriyama, T.; Sato, N. The plant and protist organellar DNA replication enzyme POP showing up in place of DNA polymerase gamma may be a suitable antiprotozoal drug target. In The Mechanisms of DNA Replication; Stuart, D., Ed.; InTech: Rijeka, Croatia, 2013; chapter 11. [Google Scholar]
- Kabeya, Y.; Miyagishima, S. Chloroplast DNA replication is regulated by the redox state independently of chloroplast division in Chlamydomonas reinhardtii. Plant Physiol. 2013, 161, 2102–2112. [Google Scholar] [CrossRef]
- Ishida, K.; Green, B.R. Second- and third-hand chloroplasts in dinoflagellates: Phylogeny of oxygen-evolving enhancer 1 (PsbO) protein reveals replacement of a nuclear-encoded plastid gene by that of a haptophyte tertiary endosymbiont. Proc. Natl. Acad. Sci. USA 2002, 99, 9294–9299. [Google Scholar] [CrossRef] [PubMed]
- Howe, C.J.; Nisbet, R.E.R.; Barbrook, A.C. The remarkable chloroplast genome of dinoflagellates. J. Exp. Bot. 2008, 59, 1035–1045. [Google Scholar] [CrossRef] [PubMed]
- Gabrielsen, T.M.; Minge, M.A.; Espelund, M.; Tooming-Klunderud, A.; Patil, V.; Nederbragt, A.J.; Otis, C.; Turmel, M.; Shalchian-Tabrizi, K.; Lemieux, C.; et al. Genome evolution of a tertiary dinoflagellate plastid. PLoS One 2011, 6, e19132. [Google Scholar] [CrossRef] [PubMed]
- Gile, G.H.; Moog, D.; Slamovits, C.H.; Maier, U.-G.; Archibald, J.M. Dual organellar targeting of aminoacyl-tRNA synthetases in diatoms and cryptophytes. Genome Biol. Evol. 2015, 7, 1728–1742. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hirakawa, Y.; Watanabe, A. Organellar DNA Polymerases in Complex Plastid-Bearing Algae. Biomolecules 2019, 9, 140. https://doi.org/10.3390/biom9040140
Hirakawa Y, Watanabe A. Organellar DNA Polymerases in Complex Plastid-Bearing Algae. Biomolecules. 2019; 9(4):140. https://doi.org/10.3390/biom9040140
Chicago/Turabian StyleHirakawa, Yoshihisa, and Arisa Watanabe. 2019. "Organellar DNA Polymerases in Complex Plastid-Bearing Algae" Biomolecules 9, no. 4: 140. https://doi.org/10.3390/biom9040140
APA StyleHirakawa, Y., & Watanabe, A. (2019). Organellar DNA Polymerases in Complex Plastid-Bearing Algae. Biomolecules, 9(4), 140. https://doi.org/10.3390/biom9040140




