Prosopis Plant Chemical Composition and Pharmacological Attributes: Targeting Clinical Studies from Preclinical Evidence
Abstract
1. Introduction
2. Prosopis Plants Phytochemical Composition
3. Traditional Medicinal Uses of Prosopis Plants
3.1. Prosopis cineraria
3.2. Prosopis juliflora
3.3. Prosopis africana
3.4. Other Prosopis Plants
4. Biological Activities Prosopis Plants
4.1. In vitro Biological Effects of Prosopis Plants
4.1.1. Prosopis juliflora
Antioxidant Effects
Antiplasmodial Effects
Antimicrobial Effects
Anthelmintic, Antiprotozoal and Antiplasmodial Effects
Autophagy and Apoptosis Effects
4.1.2. Prosopis cineraria
Antioxidant Effects
Analgesic Effects
Antiplasmodial Effects
Antimicrobial Effects
Anticancer Effects
4.1.3. Prosopis farcta
Antioxidant Effects
Antimicrobial Effects
Anticancer Effects
4.1.4. Prosopis glandulosa
Antiplasmodial Effects
Antimicrobial Effects
4.1.5. Prosopis laevigata
Antioxidant Effects
Cardioprotective Effects
4.1.6. Prosopis flexuosa
Antioxidant Effects
4.1.7. Prosopis africana
Antimicrobial Effects
Anti-Trypanosomal Effects
4.1.8. Prosopis alba and Prosopis nigra
Antioxidant Effects
4.1.9. Prosopis kuntzei and Prosopis ruscifolia
Antimicrobial Effects
4.2. In vivo Biological Effects of Prosopis Plants
4.2.1. Prosopis cineraria
Antidiabetic Effects
Wound Healing Effects
Antipyretic Effects
Spasmolytic, Bronchodilator and Vasodilator Effects
Anti-Depressive and CNS Disorders Effects
Skin Caring and Antiaging effects
Anthelmintic Effects
4.2.2. Prosopis glandulosa
Antidiabetic Effects
Inflammation and Regeneration
Antimalarial Effects
Antimicrobial Effects
4.2.3. Prosopis juliflora
Antipyretic Effects
Antimalarial Effects
Antimicrobial Effects
Anti-Dandruff Agent
4.2.4. Prosopis farcta
Neuroprotective Effects
Cardiovascular Disorders
Antimicrobial Effects
4.2.5. Prosopis ruscifolia
Antidiabetic Effects
4.2.6. Prosopis strombulifera
Antinociceptive Effect
5. Adverse Effects and Toxicological Attributes
5.1. Cytotoxicity
5.2. Fertility
5.3. Poisoning and Toxicity
5.4. Allergy
6. Pre-Clinical Effectiveness: Paving the Way for Clinical Studies
7. Food Preservative Applications of Prosopis Plants
7.1. Nutritional Attributes beyond Health Promotion
7.2. Plant Growth Inhibition
8. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sharifi-Rad, J.; Salehi, B.; Varoni, E.M.; Sharopov, F.; Yousaf, Z.; Ayatollahi, S.A.; Kobarfard, F.; Sharifi-Rad, M.; Afdjei, M.H.; Sharifi-Rad, M.; et al. Plants of the Melaleuca genus as antimicrobial agents: From farm to pharmacy. Phytother. Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Roberts, T.H.; Matthews, K.R.; Bezerra, C.F.; Morais-Braga, M.F.B.; Coutinho, H.D.M.; Sharopov, F.; Salehi, B.; Yousaf, Z.; Sharifi-Rad, M.; et al. Ethnobotany of the genus Taraxacum—Phytochemicals and antimicrobial activity. Phytother. Res. 2018, 32, 2131–2145. [Google Scholar] [CrossRef] [PubMed]
- Umair, M.; Altaf, M.; Abbasi, A.M. An ethnobotanical survey of indigenous medicinal plants in Hafizabad district, Punjab-Pakistan. PLoS ONE 2017, 12, e0177912. [Google Scholar] [CrossRef] [PubMed]
- Younis, W.; Asif, H.; Sharif, A.; Riaz, H.; Bukhari, I.A.; Assiri, A.M. Traditional medicinal plants used for respiratory disorders in Pakistan: A review of the ethno-medicinal and pharmacological evidence. Chin. Med. 2018, 13, 48. [Google Scholar]
- Ahmed, N.; Mahmood, A.; Ashraf, A.; Bano, A.; Tahir, S.S.; Mahmood, A. Ethnopharmacological relevance of indigenous medicinal plants from district Bahawalnagar, Punjab, Pakistan. J. Ethnopharmacol. 2015, 175, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Abarca, N.A.; Campos, M.G.; Reyes, J.A.A.; Jimenez, N.N.; Corral, J.H.; Valdez, L.G.S. Antioxidant activity of polyphenolic extract of monofloral honeybee-collected pollen from mesquite (Prosopis juliflora, Leguminosae). J. Food Compos. Anal. 2007, 20, 119–124. [Google Scholar] [CrossRef]
- Cattaneo, F.; Sayago, J.E.; Alberto, M.R.; Zampini, I.C.; Ordoñez, R.M.; Chamorro, V.; Pazos, A.; Isla, M.I. Anti-inflammatory and antioxidant activities, functional properties and mutagenicity studies of protein and protein hydrolysate obtained from Prosopis alba seed flour. Food Chem. 2014, 161, 391–399. [Google Scholar] [CrossRef]
- Jahromi, M.A.F.; Etemadfard, H.; Zebarjad, Z. Antimicrobial and antioxidant characteristics of volatile components and ethanolic fruit extract of Prosopis farcta (Bank & Soland.). Trends Pharm. Sci. 2018, 4, 177–186. [Google Scholar]
- Rodriguez, I.F.; Pérez, M.J.; Cattaneo, F.; Zampini, I.C.; Cuello, A.S.; Mercado, M.I.; Ponessa, G.; Isla, M.I. Morphological, histological, chemical and functional characterization of Prosopis alba flours of different particle sizes. Food Chem. 2019, 274, 583–591. [Google Scholar] [CrossRef]
- Perez, M.J.; Cuello, A.S.; ZampiniI, C.; Ordonez, R.M.; Alberto, M.R.; Quispe, C.; Schmeda-Hirschmann, G.; Isla, M.I. Polyphenolic compounds and anthocyanin content of Prosopis nigra and Prosopis albapods flour and their antioxidant and anti-inflammatory capacities. Food Res. Int. 2014, 64, 762–771. [Google Scholar] [CrossRef]
- Vasile, F.E.; Romero, A.M.; Judis, M.A.; Mattalloni, M.; Virgolini, M.B.; Mazzobre, M.F. Phenolics composition, antioxidant properties and toxicological assessment of Prosopis alba exudate gum. Food Chem. 2019. [Google Scholar] [CrossRef] [PubMed]
- Afrin, S.; Gasparrini, M.; Forbes-Hernandez, T.Y.; Reboredo-Rodriguez, P.; Mezzetti, B.; Varela-Lopez, A.; Giampieri, F.; Battino, M. Promising health benefits of the strawberry: A focus on clinical studies. J. Agric. Food Chem. 2016, 64, 4435–4449. [Google Scholar] [CrossRef] [PubMed]
- Yaseen, G.; Ahmad, M.; Sultana, S.; Alharrasi, A.S.; Hussain, J.; Zafar, M. Ethnobotany of medicinal plants in the Thar Desert (Sindh) of Pakistan. J. Ethnopharmacol. 2015, 163, 43–59. [Google Scholar] [CrossRef] [PubMed]
- Sadat-Hosseini, M.; Farajpour, M.; Boroomand, N.; Solaimani-Sardou, F. Ethnopharmacological studies of indigenous medicinal plants in the south of Kerman, Iran. J. Ethnopharmacol. 2017, 199, 194–204. [Google Scholar] [CrossRef]
- Suroowan, S.; Javeed, F.; Ahmad, M.; Zafar, M.; Noor, M.J.; Kayani, S.; Javed, A.; Mahomoodally, M.F. Ethnoveterinary health management practices using medicinal plants in South Asia—A review. Vet. Res. Commun. 2017, 41, 147–168. [Google Scholar] [CrossRef]
- Tariq, A.; Adnan, M.; Iqbal, A.; Sadia, S.; Fan, Y.; Nazar, A.; Mussarat, S.; Ahmad, M.; Olatunji, O.A.; Begum, S. Ethnopharmacology and toxicology of Pakistani medicinal plants used to treat gynecological complaints and sexually transmitted infections. S. Afr. J. Bot. 2018, 114, 132–149. [Google Scholar] [CrossRef]
- Aziz, M.A.; Adnan, M.; Khan, A.H.; Shahat, A.A.; Al-Said, M.S.; Ullah, R. Traditional uses of medicinal plants practiced by the indigenous communities at Mohmand Agency, FATA, Pakistan. J. Ethnobiol. Ethnomed. 2018, 14, 2. [Google Scholar] [CrossRef]
- Wagh, V.V.; Jain, A.K. Status of ethnobotanical invasive plants in western Madhya Pradesh, India. S. Afr. J. Bot. 2018, 114, 171–180. [Google Scholar] [CrossRef]
- Diarra, N.; van’t Klooster, C.; Togola, A.; Diallo, D.; Willcox, M.; de Jong, J. Ethnobotanical study of plants used against malaria in Sélingué subdistrict, Mali. J. Ethnopharmacol. 2015, 166, 352–360. [Google Scholar] [CrossRef]
- Catarino, L.; Havik, P.J.; Romeiras, M.M. Medicinal plants of Guinea-Bissau: Therapeutic applications, ethnic diversity and knowledge transfer. J. Ethnopharmacol. 2016, 183, 71–94. [Google Scholar] [CrossRef]
- Odoh, U.E.; Uzor, P.F.; Eze, C.L.; Akunne, T.C.; Onyegbulam, C.M.; Osadebe, P.O. Medicinal plants used by the people of Nsukka Local Government Area, south-eastern Nigeria for the treatment of malaria: An ethnobotanical survey. J. Ethnopharmacol. 2018, 218, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Salihu, T.; Olukunle, J.O.; Adenubi, O.T.; Mbaoji, C.; Zarma, M.H. Ethnomedicinal plant species commonly used to manage arthritis in North-West Nigeria. S. Afr. J. Bot. 2018, 118, 33–43. [Google Scholar] [CrossRef]
- Suárez, M.E. Medicines in the forest: Ethnobotany of wild medicinal plants in the pharmacopeia of the Wichí people of Salta province (Argentina). J. Ethnopharmacol. 2019, 231, 525–544. [Google Scholar] [CrossRef] [PubMed]
- Nasab, F.K.; Esmailpour, M. Ethno-medicinal survey on weed plants in agro-ecosystems: A case study in Jahrom, Iran. Environ. Dev. Sustain. 2018, 21, 2145–2164. [Google Scholar] [CrossRef]
- Estrada-Castillón, E.; Villarreal-Quintanilla, J.Á.; Rodríguez-Salinas, M.M.; Encinas-Domínguez, J.A.; González-Rodríguez, H.; Figueroa, G.R.; Arévalo, J.R. Ethnobotanical survey of useful species in Bustamante, Nuevo León, Mexico. Hum. Ecol. 2018, 46, 117–132. [Google Scholar] [CrossRef]
- Badri, A.M.; Garbi, M.I.; Gmaraldeen, S.M.; Magzoub, A.A.; Ibrahim, I.T.; Saleh, M.S.; Kabbashi, A.S.; Mohamed, S.G. Antioxidant activity and phytochemical screening of Prosopis juliflora leaves extract. Adv. Med. Plant Res. 2017, 5, 37–40. [Google Scholar] [CrossRef]
- Ravikumar, S.; Inbaneson, S.J.; Suganthi, P. In vitro antiplasmodial activity of ethanolic extracts of South Indian medicinal plants against Plasmodium falciparum. Asian Pac. J. Trop. Dis. 2012, 2, 180–183. [Google Scholar] [CrossRef]
- Badri, A.M.; Garbi, M.I.; Kabbashi, A.S.; Saleh, M.S.; Yousof, Y.S.; Mohammed, S.F.; Ibrahim, I.T.; Magzoub, A.A. In vitro anti-bacterial activity of Prosopis juliflora leafs extract against pathogenic bacteria. Adv. Med. Plant Res. 2016, 5, 1–4. [Google Scholar] [CrossRef]
- Tajbakhsh, S.; Barmak, A.; Vakhshiteh, F.; Gharibi, M. In vitro antibacterial activity of the Prosopis juliflora seed pods on some common pathogens. J. Clin. Diagn. Res. 2015, 9, 13–15. [Google Scholar]
- Naji, T.; Hakemi, M.; Asareh, M. Antibacterial efects of Prosopis julifora occurring in Iran. Microbes Appl. Res. 2012, 204–208. [Google Scholar] [CrossRef]
- Cáceres, A.; Menéndez, H.; Méndez, E.; Cohobón, E.; Samayoa, B.E.; Jauregui, E.; Peralta, E.; Carrillo, G. Antigonorrhoeal activity of plants used in Guatemala for the treatment of sexually transmitted diseases. J. Ethnopharmacol. 1995, 48, 85–88. [Google Scholar] [CrossRef]
- Thakur, R.; Singh, R.; Saxena, P.; Mani, A. Evaluation of antibacterial activity of Prosopis juliflora (SW.) DC. leaves. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Arya, G.; Kumari, R.M.; Sharma, N.; Gupta, N.; Kumar, A.; Chatterjee, S.; Nimesh, S. Catalytic, antibacterial and antibiofilm efficacy of biosynthesised silver nanoparticles using Prosopis juliflora leaf extract along with their wound healing potential. J. Photochem. Photobiol. B Biol. 2019, 190, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Raghavendra, M.P.; Satish, S.; Raveesha, K.A. Alkaloid extracts of Prosopis juliflora (Sw.) DC. (Mimosaceae) against Alternaria alternate. J. Biopestic. 2009, 2, 56–59. [Google Scholar]
- Sudhakar, S.; Sengottaiyan, A.; Manoharan, K.; Sudhakar, C. Inhibitory effect of Prosopis juliflora on plant and human pathogens. Int. J. Adv. Sci. Eng. 2015, 1, 47–49. [Google Scholar]
- Dos Santos, E.T.; Pereira, M.L.; da Silva, C.F.; Souza-Neta, L.C.; Geris, R.; Martins, D.; Santana, A.E.; Barbosa, L.C.; Silva, H.G.; Freitas, G.C.; et al. Antibacterial activity of the alkaloid-enriched extract from Prosopis juliflora pods and its influence on in vitro ruminal digestion. Int. J. Mol. Sci. 2013, 14, 8496–8516. [Google Scholar] [CrossRef]
- Lima, G.H.; Gomes, D.C.; Santos, N.S.; Dias, Ê.R.; Botura, M.B.; Batatinha, M.J.M.; Branco, A. Prosopis juliflora Pods alkaloid-rich fraction: In vitro anthelmintic activity on goat gastrointestinal parasites and Its cytotoxicity on vero cells. Pharm. Mag. 2017, 13, 684–687. [Google Scholar]
- Briones-Labarca, V.; Muñoz, C.; Maureira, H. Effect of high hydrostatic pressure on antioxidant capacity, mineral and starch bioaccessibility of a non conventional food: Prosopis chilensis seed. Food Res. Int. 2011, 44, 875–883. [Google Scholar] [CrossRef]
- Velmurugan, V.; Arunachalam, G.; Ravichandran, V. Anthelmintic potential of Prosopis cineraria (Linn.) druce stem barks. Asian J. Plant Sci. Res. 2011, 1, 88–91. [Google Scholar]
- Saad, A.M.; Ghareeb, M.A.; Abdel-Aziz, M.S.; Madkour, H.M.F.; Khalaf, O.M.; El-Ziaty, A.K.; Abdel-Mogib, M. Chemical constituents and biological activities of different solvent extracts of Prosopis farcta growing in Egypt. J. Pharmacogn. Phytother. 2017, 9, 67–76. [Google Scholar]
- Tapia, A.; Feresin, G.E.; Bustos, D.; Astudillo, L.; Theoduloz, C.; Schmeda-Hirschmann, G. Biologically active alkaloids and a free radical scavenger from Prosopis species. J. Ethnopharmacol. 2000, 71, 241–246. [Google Scholar] [CrossRef]
- García-Andrade, M.; González-Laredo, R.F.; Rocha-Guzmán, N.E.; Gallegos-Infante, J.A.; Rosales-Castro, M.; Medina-Torres, L. Mesquite leaves (Prosopis laevigata), a natural resource with antioxidant capacity and cardioprotection potential. Ind. Crops Prod. 2013, 44, 336–342. [Google Scholar] [CrossRef]
- Kumar, A.; Yadav, S.K.; Singh, S.; Pandey, S.N. Analgesic activity of ethanolic extract of roots of Prosopis cineraria (L.) Druce. J. Appl. Pharm. Sci. 2011, 1, 158–160. [Google Scholar]
- Satish, P.V.V.; Somaiah, K.; Brahmam, P.; Rekha, N.S.; Sunita, K. Antimalarial activity of Prosopis cineraria (L) Druce against chloroquine sensitive plasmodium Falciparum 3D7 strain. Eur. J. Pharm. Med. Res. 2015, 2, 295–303. [Google Scholar]
- Rahman, A.A.; Samoylenko, V.; Jacob, M.R.; Sahu, R.; Jain, S.K.; Khan, S.I.; Tekwani, B.L.; Muhammad, I. Antiparasitic and antimicrobial indolizidines from the leaves of Prosopis glandulosa var. glandulosa. Planta Med. 2011, 77, 1639–1643. [Google Scholar] [CrossRef]
- Al-Musayeib, N.M.; Mothana, R.A.; Al-Massarani, S.; Matheeussen, A.; Cos, P.; Maes, L. Study of the in vitro antiplasmodial, antileishmanial and antitrypanosomal activities of medicinal plants from Saudi Arabia. Molecules 2012, 17, 11379–11390. [Google Scholar] [CrossRef]
- Kolapo, A.L.; Okunade, M.B.; Adejumobi, J.A.; Ogundiya, M.O. Phytochemical composition and antimicrobial activity of Prosopis africana against some selected oral pathogens. World J. Agric. Sci. 2009, 5, 90–93. [Google Scholar]
- Darogha, S.N. Phytochemical analysis and antibacterial activity of some medicinal plants against methicillin-resistant Staphylococcus aureues. Tikrit J. Pltarm. Sci. 2009, 5, 116–126. [Google Scholar]
- Moorthy, K.; Kumar, R.S. Phytochemical and antimicrobial studies of leaf extract of Prosopis glandulosa. J. Ecotoxicol. Environ. Monit. 2011, 21, 143–149. [Google Scholar]
- Solanki, D.S.; Kumar, S.; Parihar, K.; Tak, A.; Gehlot, P.; Pathak, R.; KSingh, S. Characterization of a novel seed protein of Prosopis cineraria showing antifungal activity. Int. J. Biol. Macromol. 2018, 116, 16–22. [Google Scholar] [CrossRef]
- Mustafa, K.K.; Maulud, S.Q.; Hamad, P.A. Detection of Sphingomona spaucimobilis and antibacterial activity of Prosopis farcta extracts on it Karbala. Int. J. Mod. Sci. 2018, 4, 100–106. [Google Scholar]
- Hari, P.O.; Aluru, S.; Kishore, K.A.; Navya, A.; Hari, K.O.; Bhaskar, M.; Papa, R.A.; Ravindra, R.N. Comparative evaluation of the antibacterial efficacy of P. juliflora and three commercially available mouthrinses: An in vitro study. J. Pharm. Res. 2011, 4, 2149–2151. [Google Scholar]
- Salvat, A.; Antonacci, L.; Fortunato, R.H.; Suarez, E.Y.; Godoy, H.M. Antimicrobial activity in methanolic extracts of several plant species from northern Argentina. Phytomedicine 2004, 11, 230–234. [Google Scholar] [CrossRef]
- Neghabi, H.M.; Aliahmadi, A.; Taheri, M.R.; Ghassempour, A.; Irajian, G.; Rezadoost, H.; Feizabadi, M.M. A bioassay-guided fractionation scheme for characterization of new antibacterial compounds from Prosopis cineraria aerial parts. Iran. J. Microbiol. 2016, 2, 1–7. [Google Scholar]
- Kavita, N.P.D.P.D.; Gahlawat, D.K.; Singh, L.; Anish, S. Antibacterial and phytochemical analysis of fruit extracts of Prosopis cineraria L. Int. J. Appl. Sci. Bioeng. 2017, 1, 1–7. [Google Scholar]
- Ahmad, A.; Ahmad, V.; Khalid, S.M.; Siddiqui, S.A.; Khan, K.A. Study of the antibacterial therapeutic efficacy of juliflorine, julifloricine and a benzene insoluble alkaloidal fraction of Prosopis juliflora. J. Islamic Acad. Sci. 1995, 8, 131–136. [Google Scholar]
- Jinu, U.; Gomathi, M.; Saiqa, I.; Geetha, N.; Benelli, G.; Venkatachalam, P. Green engineered biomolecule-capped silver and copper nanohybrids using Prosopis cineraria leaf extract: Enhanced antibacterial activity against microbial pathogens of public health relevance and cytotoxicity on human breast cancer cells (MCF-7). Microb. Pathog. 2017, 105, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Khatri, A.; Rathore, A.; Patil, U.K. Assessment of anthelmintic activity of the Prosopis cineraria (Linn.) Druce bark. Indian J. Nat. Prod. Res. 2011, 2, 512–514. [Google Scholar]
- Atawodi, S.E.; Ogunbusola, F. Evaluation of anti-trypanosomal properties of four extracts of leaves, stem and root barks of Prosopis africana in laboratory Animals. Biokemistri 2009, 21, 101–108. [Google Scholar] [CrossRef]
- Renganathan, S.; Sahu, S.K.; Kathiresan, K. Phytochemical and molecular dockinganalyses of Prosopis juliflora derived phytochemicals against anti-apoptotic protein BCL-2. World J. Pharm. Res. 2015, 4, 1487–1496. [Google Scholar]
- Saragusti, A.C.; Bustos, P.S.; Pierosan, L.; Cabrera, J.L.; Chiabrando, G.A.; Santos, A.R.S.; Ortega, M.G. Involvement of the L-arginine-nitric oxide pathway in the antinociception caused by fruits of Prosopis trombulifera (Lam.) Benth. J. Ethnopharmacol. 2012, 140, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.D.A.; Pitanga, B.P.S.; Nascimento, R.P.; Souza, C.S.; Coelho, P.L.C.; Menezes-Filho, N.; Silva, A.M.M.; Costa, M.F.D.; El-Bachá, R.S.; Velozo, E.S.; et al. Juliprosopine and juliprosinefrom Prosopis juliflora leaves induce mitochondrial damage and cytoplasmic vacuolation on cocultured glial cells and neurons. Chem. Res. Toxicol. 2013, 26, 1810–1820. [Google Scholar] [CrossRef] [PubMed]
- Dhivya, K.; Vengateswari, G.; Arunthirumeni, M.; Karthi, S.; Senthil, N.S.; Shivakumar, M.S. Bioprospecting of Prosopis juliflora (Sw.) DC seed pod extract effect on antioxidant and immune system of Spodopteralitura (Lepidoptera: Noctuidae). Physiol. Mol. Plant Pathol. 2018, 101, 45–53. [Google Scholar] [CrossRef]
- Silva, V.D.A.; Cuevas, C.; Muñoz, P.; Villa, M.; Ahumada-Castro, U.; Huenchuguala, S.; dos Santos, C.C.; de Araujo, F.M.; Ferreira, R.l.S.; da Silva, V.B.; et al. Autophagy protects against neural cell death induced by piperidine alkaloids present in Prosopis juliflora (Mesquite). Anais da Academia Brasileira de Ciências 2017, 89, 247–261. [Google Scholar] [CrossRef]
- Mohammad, I.S.; Khan, H.M.S.; Rasool, F. Biological potential and phytochemical evaluation of Prosopis cineraria. World Appl. Sci. J. 2013, 27, 1489–1494. [Google Scholar]
- Gupta, A.; Sharma, G.; Pandey, S.N.; Verma, B.; Pal, V.; Agrawal, S.S. Prosopis cineraria and its various therapeutic effects with special reference to diabetes: A novel approach. Int. J. Pharm. Sci. Rev. Res. 2014, 27, 328–333. [Google Scholar]
- Velmurugan, V.; Arunachalam, G.; Ravichandiran, V. In vitro antioxidant potential of stem bark of Prosopis cineraria Linn. BioMedRx 2013, 1, 140–142. [Google Scholar]
- Cardozo, M.L.; Ordoñez, R.M.; Zampini, I.C.; Cuello, A.S.; Dibenedetto, G.; Isla, M.I. Evaluation of antioxidant capacity, genotoxicity and polyphenol content of non conventional foods: Prosopis flour. Food Res. Int. 2010, 43, 1505–1510. [Google Scholar] [CrossRef]
- Soni, L.K.; Dobhal, M.P.; Arya, D.; Bhagour, K.; Parasher, P.; Gupta, R.S. In vitro and in vivo antidiabetic activity of isolated fraction of Prosopis cineraria against streptozotocin-induced experimental diabetes: A mechanistic study. Biomed. Pharmacother. 2018, 108, 1015–1021. [Google Scholar] [CrossRef]
- Jain, P.G.; Surana, S.J. Hypolipidemic activity of Prosopis cineraria L (Druce) fruit extract and molecular modeling study with farnesoid X receptor (FXR). Trop. J. Pharm. Res. 2015, 14, 1621–1628. [Google Scholar]
- Sharma, N.; Garg, V.; Paul, A. Antihyperglycemic, antihyperlipidemic and antioxidative potential of Prosopis cineraria bark. Indian J. Clin. Biochem. 2010, 25, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Purohit, A.; Ram, H. Hypolipidemic and antiatherosclerotic effects of Prosopis cineraria bark extract in experimentally induced hyperlipidemic rabbits. Asian J. Pharm.Clin. Res. 2012, 5, 106–109. [Google Scholar]
- George, C.; Lochner, A.; Huisamen, B. The efficacy of Prosopis glandulosa as antidiabetic treatment in rat models of diabetes and insulin resistance. J. Ethnopharmacol. 2011, 137, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Bublitz, C.M.; Ibarrola, D.; Oacute, N.-L.M.; Dölz, J.; Kennedy, M.L. Acute and chronic anti-hyperglycemic effect of Prosopis ruscifolia extract in normoglycemic and alloxan-induced hyperglycemic rats. J. Appl. Pharm. Sci. 2016, 6, 178–184. [Google Scholar] [CrossRef][Green Version]
- Mollashahi, M.; Tehranipour, M.; Khayyatzade, J.; Moosavi, B.Z.J. The neuroprotective effects of Prosopis farctapod aqueous and ethanol extracts on spinal cord α-motoneurons neuronal density after sciatic nerve injury in rats. Life Sci. J. 2013, 10, 293–297. [Google Scholar]
- Yadav, E.; Singh, D.; Yadav, P.; Verma, A. Antioxidant and anti-inflammatory properties of Prosopis cineraria based phenolic rich ointment in wound healing. Biomed. Pharmacother. 2018, 108, 1572–1583. [Google Scholar] [CrossRef]
- Gupta, A.; Verma, S.; Gupta, A.; Jangra, M.; Pratap, R. Evaluation of Prosopis cineraria (Linn.) Druce leaves for wound healing activity in rats. Ann. Pharm. Res. 2015, 3, 70–74. [Google Scholar]
- Ahmad, M.; Jabeen, Q.; Wajid, M.; Khan, H.M.S.; Bashir, K.; Mohammad, I.S.; Aziz, M.M.; Rehman, J.U. Time and dose dependent Antipyretic investigations of ethanolic leaves and fruits extracts of Prosopis cineraria L.(Druce). J. Pharm. Altern. Med. 2013, 2, 125–134. [Google Scholar]
- Gopinath, S.M.; Reddy, J.M.; Dayanand, K.S.; Tejaswini, A.; Shankar, A. To evaluate the antipyretic activity of Prosopis juliflora ethanolicextract in brewer’s yeast induced hyperthermia in rats. J. Biotechnol. Biosaf. 2013, 1, 28–32. [Google Scholar]
- Janbaz, K.H.; Haider, S.; Imran, I.; Zia-Ul-Haq, M.; DeMartino, L.; De Feo, V. Pharmacological evaluation of Prosopis cineraria (L.) Druce in Gastrointestinal, Respiratory, and Vascular Disorders. Evid.-Based Complement. Altern. Med. 2012. [Google Scholar] [CrossRef]
- George, M.; Joseph, L.; Sharma, A. Antidepressant and skeletal muscle relaxant effects of the aqueous extract of the Prosopis cineraria. Braz. J. Pharm. Sci. 2012, 48. [Google Scholar] [CrossRef]
- Saidi, M.R.; Farzaei, M.H.; Miraghaee, S.; Babaei, A.; Mohammadi, B.; Bahrami, M.T.; Bahrami, G. Antihyperlipidemic effect of Syrian Mesquite (Prosopis farcta) root in high cholesterol diet–fed rabbits. J. Evid.-Based Integr. Med. 2016, 21, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Keshavarzi, S.; Bahrami, G.; Mohammadi, B.; Hatami, R.; Miraghaee, S. Syrian Mesquite extract improves serum lipids and liver tissue in NFALD modelled rabbits. J. Fam. Med. 2018, 16, 150–157. [Google Scholar] [CrossRef]
- Asadollahi, A.; Sarir, H.; Omidi, A.; Torbati, M.B.M. Hepatoprotective potential of Prosopis farcta beans extracts against acetaminophen-induced hepatotoxicity in wister rats. Int. J. Prev. Med. 2014, 5, 1281–1285. [Google Scholar]
- Asadollahi, K.; Abassi, N.; Afshar, N.; Alipour, M.; Asadollahi, P. Investigation of the effects of Prosopis farcta plant extract on rat’s aorta. J. Med. Plant Res. 2010, 4, 142–147. [Google Scholar]
- George, C.; Smith, C.; Isaacs, A.W.; Huisamen, B. Chronic Prosopis glandulosa treatment blunts neutrophil infiltration and enhances muscle repair after contusion injury. Nutrients 2015, 7, 815–830. [Google Scholar] [CrossRef]
- Mohammad, I.S.; Naveed, M.; Ijaz, S.; Shumzaid, M.; Hassan, S.; Muhammad, K.S.; Rasool, F.; Akhtar, N.; Ishaq, H.M.; Khan, H.M.S. Phytocosmeceutical formulation development, characterization and its in vivo investigations. Biomed. Pharmacother. 2018, 107, 806–817. [Google Scholar] [CrossRef]
- Batista, R.; Santana, C.C.; Azevedo-Santos, A.V.; Suarez-Fontes, A.M.; Ferraz, J.L.A.; Silva, L.A.M.; Vannier-Santos, M.A. In vivo antimalarial extracts and constituents of Prosopis juliflora (Fabaceae). J. Funct. Foods 2018, 44, 74–78. [Google Scholar] [CrossRef]
- Samoylenko, V.; Ashfaq, M.K.; Jacob, M.R.; Tekwani, B.L.; Khan, S.I.; Manly, S.P.; Joshi, V.C.; Walker, L.A.; Muhammad, I. Indolizidine, antiinfective and antiparasitic compounds from Prosopis glandulosa Torr. Var. glandulosa. J. Nat. Prod. 2009, 72, 92–98. [Google Scholar] [CrossRef]
- Ashfaq, M.K.; Abdel-Bakky, M.S.; Maqbool, M.T.; Samoylenko, V.; Rahman, A.A.; Muhammad, I. Efficacy of prosopilosidine from Prosopis glandulosa var. glandulosa against Cryptococcus neoformans infection in a murine model. Molecules 2018, 23, 1674. [Google Scholar] [CrossRef]
- Sorna Kumar, R.S.A.; Bharathi, R.A.; Lakshmipriya, R.; Suganya Devi, C.; Sivaranjani, A.; Banumathi, T. Formulation of antimalassezic shampoo from datura metel and Prosopis juliflora. Indo Am. J. Pharm. Res. 2017, 7, 905–908. [Google Scholar]
- Omidi, A.; Nik, H.A.; Ghazaghi, M. Prosopis farcta beans increase HDL cholesterol and decrease LDL cholesterol in ostriches (Struthiocamelus). Trop. Anim. Health Prod. 2013, 45, 431–434. [Google Scholar] [CrossRef] [PubMed]
- Prabha, D.S.; Dahms, H.U.; Malliga, P. Assessment of acute and subactue oral toxicity of ethanolic extracts of Prosopis julifolora on Rattus norvegicus. J. Herb. Med. Toxicol. 2012, 6, 61–65. [Google Scholar]
- Ahmed, S.M.; Ahmed, S.; Tasleem, F.; Hasan, M.M.; Azhar, I. Acute systemic toxicity of four Mimosaceous plants leaves in mice. IOSR J. Pharm. 2012, 2, 291–295. [Google Scholar] [CrossRef]
- Quispe, C.; Petroll, K.; Theoduloz, C.; Schmeda-Hirschmann, G. Antioxidant effect and characterization of South American Prosopis pods syrup. Food Res. Int. 2014, 56, 174–181. [Google Scholar] [CrossRef]
- Da Silva, V.D.A.; da Silva, A.M.M.; de Silva, J.H.C.; Costa, S.L. Neurotoxicity of Prosopis juliflora: From natural poisoning to mechanism of action of Its piperidine alkaloids. Neurotox. Res. 2018, 34, 878–888. [Google Scholar] [CrossRef]
- Silva, A.M.M.; Silva, A.R.; Pinheiro, A.M.; Freitas, S.R.V.B.; Silva, V.D.A.; Souza, C.S.; Hughes, J.B.; El-Bachá, R.S.; Costa, M.F.D.; Velozo, E.S.; et al. Alkaloids from Prosopis juliflora leaves induce glial activation, cytotoxicity and stimulate NO production. Toxicon 2007, 49, 601–614. [Google Scholar] [CrossRef]
- Márquez, S.R.; Hernández, E.; Solano, F.; Romero, C.; López, G.; Rojas, L.J.; Casillas, F.; Chemineau, P.; Keller, M.; Delgadillo, J.A. Mesquite (Prosopis juliflora) pod extract decreases fertility in female but not male rats. Afr. J. Pharm. Pharmacol. 2016, 10, 984–994. [Google Scholar]
- Márquez, S.R.; Rojas, L.J.; Hernández, A.; Romero, C.; López, G.; Miranda, L.; Aguilera, A.G.; Solano, F.; Hernández, E.; Chemineau, P.; et al. Comparison of the effects of mesquite pod and Leucaena extracts with phytoestrogens on the reproductive physiology and sexual behavior in the male rat. Physiol. Behav. 2016, 164, 1–10. [Google Scholar] [CrossRef]
- Farag-Mahmod, F.; Hessam, W.; Khalil, K.A. Prevalence of mesquite (Prosopis species) allergy and efficacy of conventional allergen specific Immunotherapy (ASIT) to mesquite in Egyptian patients with perennial allergic rhinitis (PAR). Allergy Asthma Clin. Immunol. 2010, 20, 106–118. [Google Scholar] [CrossRef][Green Version]
- Almeida, V.M.; Rocha, B.P.; Pfister, J.A.; Medeiros, R.M.T.; Riet-Correa, F.; Chaves, H.A.S.; Silva Filho, G.B.; Mendonça, F.S. Spontaneous poisoning by Prosopis juliflora (Leguminosae) in sheep. Pesquisa Veterinária Brasileira 2017, 37, 110–114. [Google Scholar] [CrossRef]
- Rasouli, H.; Akbari, M.; Bahdor, T. Physiological and pharmaceutical effect of fenugreek: A review. IOSR J. Pharm. 2012, 4, 49–53. [Google Scholar]
- Inoue, M.; Hayashi, S.; Craker, L.E. Role of medicinal and aromatic plants: Past, present, and future. In Pharmacognosy-Medicinal Plants; IntechOpen: London, UK, 2019. [Google Scholar]
- Shah, K.N.; Valand, P.; Nauriyal, D.S.; Joshi, C.G. Immunomodulation of IL-1, IL-6 and IL-8 cytokines by Prosopis juliflora alkaloids during bovine sub-clinical mastitis. Biotech 2018, 8, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.H.; Maletic, V.; Raison, C.L. Inflammation and its discontents: The role of cytokines in the pathophysiology of major depression. Biol. Psychiatry 2009, 65, 732–741. [Google Scholar] [CrossRef]
- Ramazani, A.; Zakeri, S.; Sardari, S.; Khodakarim, N.; Djadidt, N.D. In vitro and in vivo anti-malarial activity of Boerhavia elegans and Solanum surattense. Malar. J. 2010, 9, 124. [Google Scholar] [CrossRef]
- Gurushidhappa, U.B.; Shivajirao, P.M.; Vasudeo, N.P.; Shankar, K.S.; Nivarti, G.R. Prosopis juliflora (Sw.), DC induces apoptosis and cell cycle arrest in triple negative breast cancer cells: In vitro and in vivo investigations. Oncotarget 2018, 9, 30304. [Google Scholar]
- De Brito Damasceno, G.A.; Ferrari, M.; Giordani, R.B. Prosopis juliflora (SW) DC, an invasive specie at the Brazilian Caatinga: Phytochemical, pharmacological, toxicological and technological overview. Phytochem. Rev. 2017, 16, 309–331. [Google Scholar] [CrossRef]
- Henciya, S.; Seturaman, P.; James, A.R.; Tsai, Y.H.; Nikam, R.; Wu, Y.C.; Dahms, H.U.; Chang, F.R. Biopharmaceutical potentials of Prosopis spp. (Mimosaceae, Leguminosa). J. Food Drug Anal. 2017, 25, 187–196. [Google Scholar] [CrossRef]
- Sivaraman, T.; Rajesh, S.S.; Elango, V. In vivo studies on detoxifying actions of aqueous bark extract of Prosopis cineraria against crude venom from Indian cobra (Naja naja). Bangladesh J. Pharmacol. 2013, 8, 395–400. [Google Scholar] [CrossRef]
- Liu, Y.; Singh, D.; Nair, M.G. Pods of Khejri (Prosopis cineraria) consumed as a vegetable showed functional food properties. J. Funct. Foods 2012, 4, 116–121. [Google Scholar] [CrossRef]
- Robertson, S.; Narayanan, N.; Raj Kapoor, B. Antitumour activity of Prosopis cineraria (L.) Druce against Ehrlich ascites carcinoma-induced mice. Nat. Prod. Res. 2011, 25, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Maideen, N.M.P.; Velayutham, R.; Manavalan, G. Protective effect of Prosopis cineraria against N-nitrosodiethylamine induced liver tumor by modulating membrane bound enzymes and glycoproteins. Adv. Pharm. Bull. 2012, 2, 179. [Google Scholar]
- Ayanwuyi, L.O.; Yaro, A.H.; Abodunde, O.M. Analgesic and anti-inflammatory effects of the methanol stem bark extract of Prosopis africana. Pharm. Biol. 2010, 48, 296–299. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Siva, K.T.; Srinivasan, K.; Rajavel, R.; Vasudevan, M.; Ganesh, M.; Kamalakannan, K.; Mallika, P. Isolation of chemical constituents from Prosopis juliflora bark and anti-inflammatory activity of its methanolic extracts. J. Pharm. Res. 2009, 2, 551–556. [Google Scholar]
- Ezike, A.C.; Akah, P.A.; Okoli, C.O.; Udegbunam, S.; Okwume, N.; Okeke, C.; Iloani, O. Medicinal plants used in wound care: A study of Prosopis africana (Fabaceae) stem bark. Indian J. Pharm. Sci. 2010, 72, 334. [Google Scholar] [CrossRef] [PubMed]
- Pérez, M.J.; Zampini, I.C.; Alberto, M.R.; Isla, M.I. Prosopis nigra mesocarp fine flour, a source of phytochemicals with potential effect on enzymes linked to metabolic syndrome, oxidative stress, and inflammatory process. J. Food Sci. 2018, 83, 1454–1462. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, K.K.; Kumar, G.; Varshney, A.; Meghvansi, M.K.; Ali, S.F.; Karthik, K.; Dhama, K.; Siddiqui, S.; Kaul, R.K. Ethnopharmacological and phytopharmaceutical evaluation of Prosopis cineraria: An overview and future prospects. Curr. Drug Metab. 2018, 19, 192–214. [Google Scholar] [CrossRef]
- Al-Frayh, A.; Hasnain, S.M.; Gad-elRab, M.O.; Al-Turki, T.; Al-Mobeireek, K.; Al-Sedairy, S.T. Human sensitization to Prosopis juliflora antigen in Saudi Arabia. Ann. Saudi Med. 1999, 19, 331–336. [Google Scholar] [CrossRef]
- Cattaneo, F.; Costamagna, M.S.; Zampini, I.C.; Sayago, J.; Alberto, M.R.; Chamorro, V.; Isla, M.I. Flour from Prosopis alba cotyledons: A natural source of nutrient and bioactive phytochemicals. Food Chem. 2016, 208, 89–96. [Google Scholar] [CrossRef]
- Díaz-Batalla, L.; Hernández-Uribe, J.P.; Román-Gutiérrez, A.D.; Cariño-Cortés, R.; Castro-Rosas, J.; Téllez-Jurado, A.; Gómez-Aldapa, C.A. Chemical and nutritional characterization of raw and thermal-treated flours of Mesquite (Prosopis laevigata) pods and their residual brans. CyTA J. Food 2018, 16, 444–451. [Google Scholar] [CrossRef]
- Lingbeck, J.M.; Cordero, P.; O’Bryan, C.A.; Johnson, M.G.; Ricke, S.C.; Crandall, P.G. Functionality of liquid smoke as an all-natural antimicrobial in food preservation. Meat Sci. 2014, 97, 197–206. [Google Scholar] [CrossRef]
- Toledo, R.T. Wood Smoke Components and Functional Properties. In International Smoked Seafood Conference Proceedings; Kramer, D.E., Brown, L., Eds.; Alaska Sea Grant College Program: Fairbanks, AK, USA, 2008; pp. 55–61. [Google Scholar]
- Ruto, M.C.; Ngugi, C.M.; Kareru, P.G.; Cheruiyot, K.; Rechab, S.O.; Madivoli, E.S.; Maina, E.G. Antioxidant activity and antimicrobial properties of Entada leptostachya and Prosopis juliflora extracts. J. Med. Plants Econ. Dev. 2018, 2, 1–8. [Google Scholar] [CrossRef]
- Montazeri, N.; Himelbloom, B.H.; Oliveira, A.C.M.; Leigh, M.; Crapo, C.A. Refined liquid smoke: A potential antilisterial additive to cold-smoked sockeye salmon (Oncorhynchus nerka). J. Food Prot. 2013, 76, 812–819. [Google Scholar] [CrossRef]
- Van Loo, E.J.; Abu, D.; Crandall, P.G.; Ricke, S.C. Screening of commercial and pecan shell–extracted liquid smoke agents as natural antimicrobials against foodborne pathogens. J. Food Prot. 2012, 75, 1148–1152. [Google Scholar] [CrossRef]
- Wendorff, W.L.; Wee, C. Effect of smoke and spice oils on growth of molds on oil-coated cheeses. J. Food Prot. 1997, 60, 153–156. [Google Scholar] [CrossRef]
- Montazeri, N.; Oliveira, A.C.M.; Himelbloom, B.H.; Leigh, M.; Crapo, C.A. Chemical characterization of commercial liquid smoke products. Food Sci. Nutr. 2013, 1, 102–115. [Google Scholar] [CrossRef]
- Morey, A.; Bratcher, C.L.; Singh, M.; McKee, S.R. Effect of liquid smoke as an ingredient in frankfurters on Listeria monocytogenes and quality attributes. Poult. Sci. 2012, 91, 2341–2350. [Google Scholar] [CrossRef]
- Winter, J.; Rehbein, S.; Joachim, A. Transmission of Helminths between Species of Ruminants in Austria Appears More Likely to Occur than Generally Assumed. Front. Vet. Sci. 2018, 5, 30. [Google Scholar] [CrossRef]
- Garcia-Bustos, J.F.; Sleebs, B.E.; Gasser, R.B. An appraisal of natural products active against parasitic nematodes of animals. Parasites Vectors 2019, 12, 306. [Google Scholar] [CrossRef]
- Hu, Y.; Ellis, B.L.; Yiu, Y.Y.; Miller, M.M.; Urban, J.F.; Shi, L.Z.; Aroian, R.V. An extensive comparison of the effect of anthelmintic classes on diverse nematodes. PLoS ONE 2013, 8, e70702. [Google Scholar] [CrossRef]
- Odhiambo, R.S.; Patrick, K.G.; Helen, K.L.; Gathu, N.C.; Francis, N.K.; Richard, W.W. Evaluation of in vitro ovicidal activity of ethanolic extracts of Prosopis juliflora (Sw.) DC (Fabaceae). IOSR J. Pharm. Biol. Sci. 2014, 9, 15–18. [Google Scholar] [CrossRef]
- Kalt, W. Effects of production and processing factors on major fruit and vegetable antioxidants. J. Food Sci. 2005, 70, 11–19. [Google Scholar] [CrossRef]
- Díaz-Batalla, L.; Hernández-Uribe, J.; Gutiérrez-Dorado, R.; Téllez-Jurado, A.; Castro-Rosas, J.; Pérez-Cadena, R.; Gómez-Aldapa, C. Nutritional characterization of Prosopis laevigata Legume tree (Mesquite) seed flour and the effect of extrusion cooking on its bioactive components. Foods 2018, 7, 124. [Google Scholar] [CrossRef]
- Tomas, M.; Toydemir, G.; Boyacioglu, D.; Hall, R.D.; Beekwilder, J.; Capanoglu, E. Processing black mulberry into jam: Effects on antioxidant potential and in vitro bioaccessibility. J. Sci. Food Agric. 2017, 97, 3106–3113. [Google Scholar] [CrossRef]
- Vasile, F.E.; Romero, A.M.; Judis, M.A.; Mazzobre, M.F. Prosopis alba exudate gum as excipient for improving fish oil stability in alginate–chitosan beads. Food Chem. 2016, 190, 1093–1101. [Google Scholar] [CrossRef]
- Zainal, A.S.; Abdel-Rahim, A.M.; Abu-Ali, R.M.; Radwan, S.S. Antimicrobial substance(s) in the leaf litter of the xerophyte Prosopis juliflora. Zentral Blattfür Mikrobiologie 1988, 143, 375–381. [Google Scholar] [CrossRef]
- Picariello, G.; Sciammaro, L.; Siano, F.; Volpe, M.G.; Puppo, M.C.; Mamone, G.F. Comparative analysis of C-glycosidic flavonoids from Prosopis spp. and Ceratonia siliqua seed germ flour. Food Res. Int. 2017, 99, 730–738. [Google Scholar] [CrossRef]
- Nutter, J.; Fritz, R.; Iurlina, M.O.; Saiz, A.I. Effect of Prosopis sp. honey on the growth and fermentative ability of Pediococcus pentosaceus and Lactobacillus fermentum. LWT Food Sci. Technol. 2016, 70, 309–314. [Google Scholar] [CrossRef]
- Nakano, H.; Nakajima, E.; Hiradate, S.; Fujii, Y.; Yamada, K.; Shigemori, H.; Hasegawa, K. Growth inhibitory alkaloids from mesquite (Prosopis juliflora (Sw.) DC.) leaves. Phytochemistry 2004, 65, 587–591. [Google Scholar] [CrossRef]
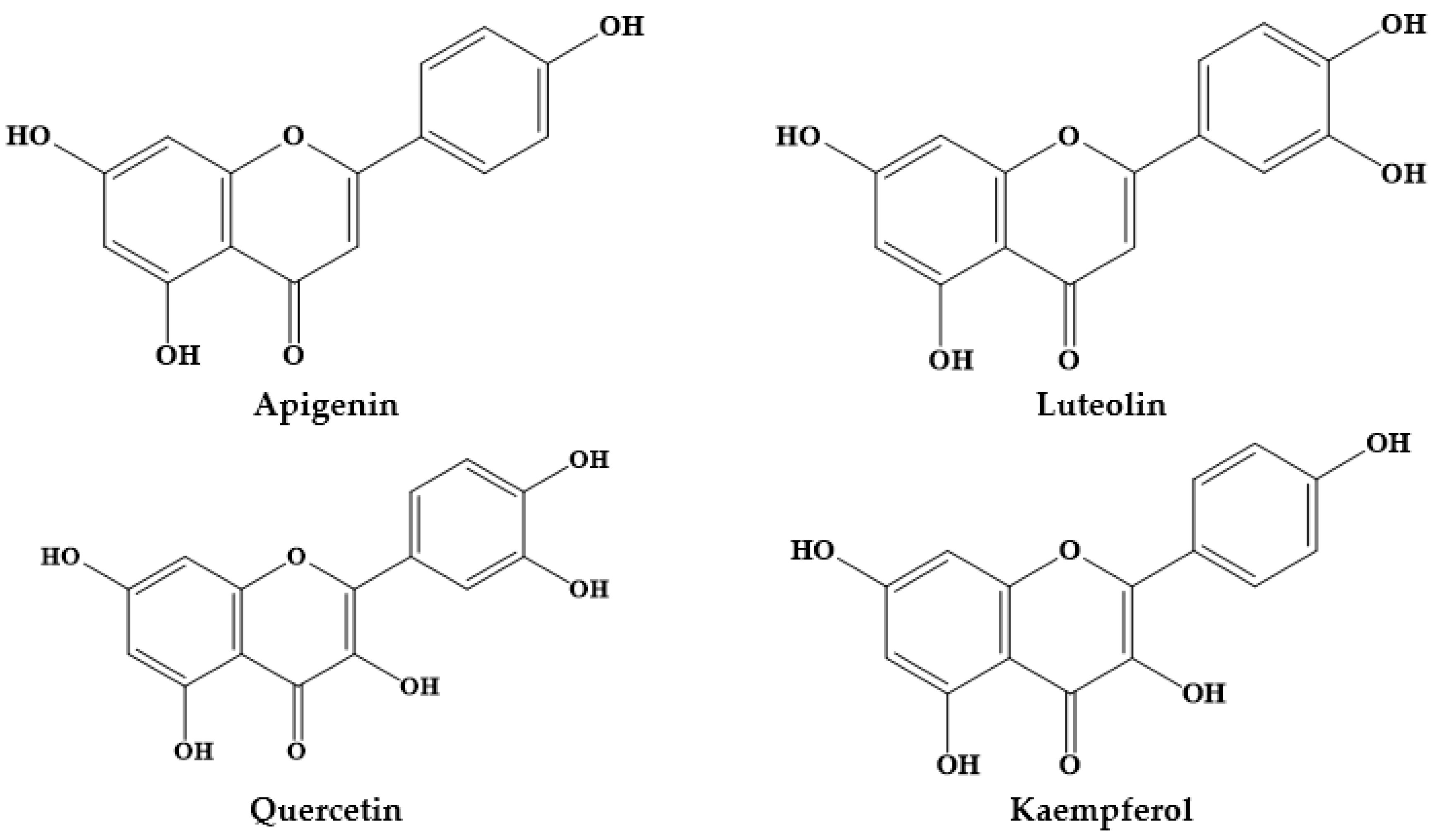
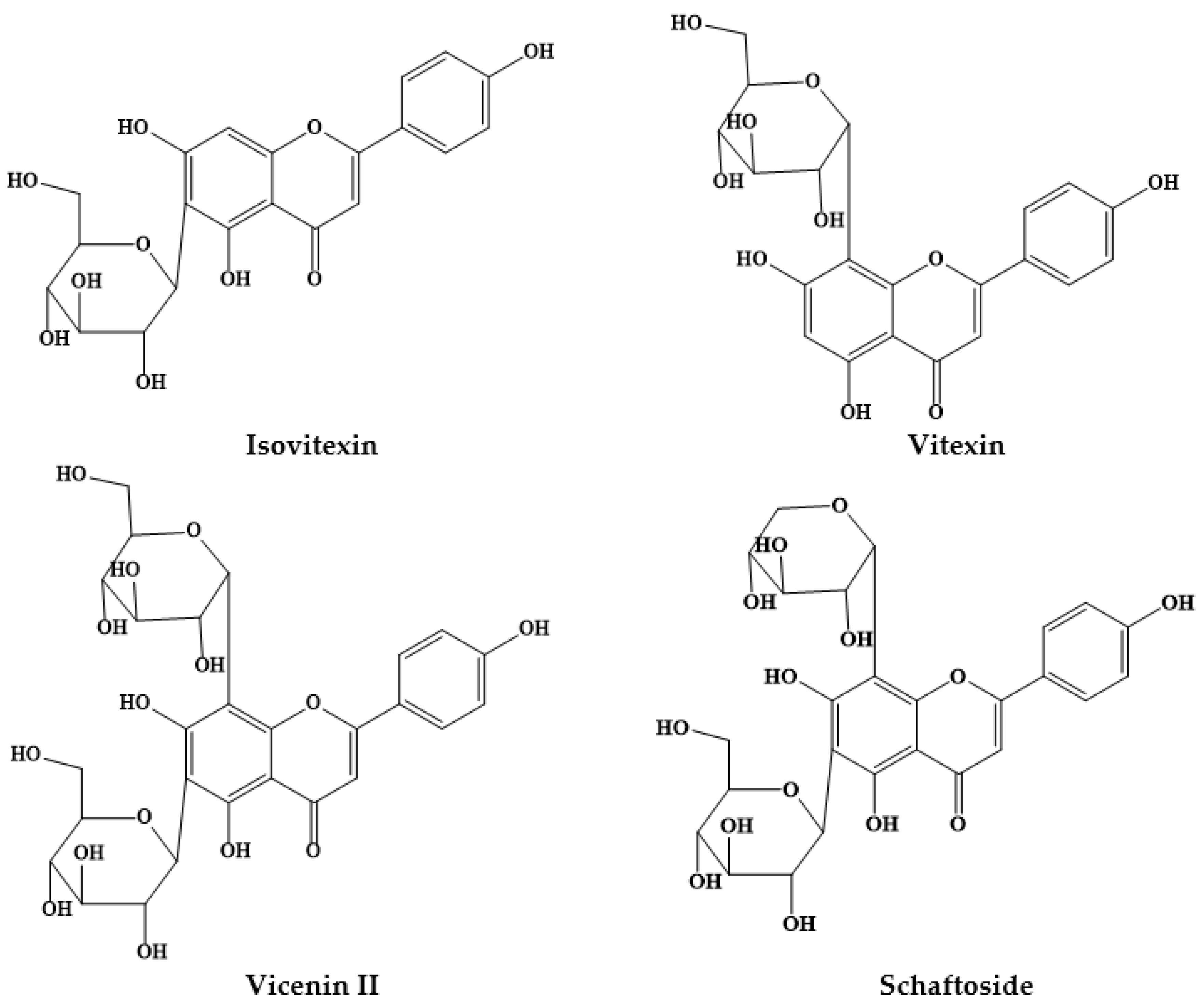
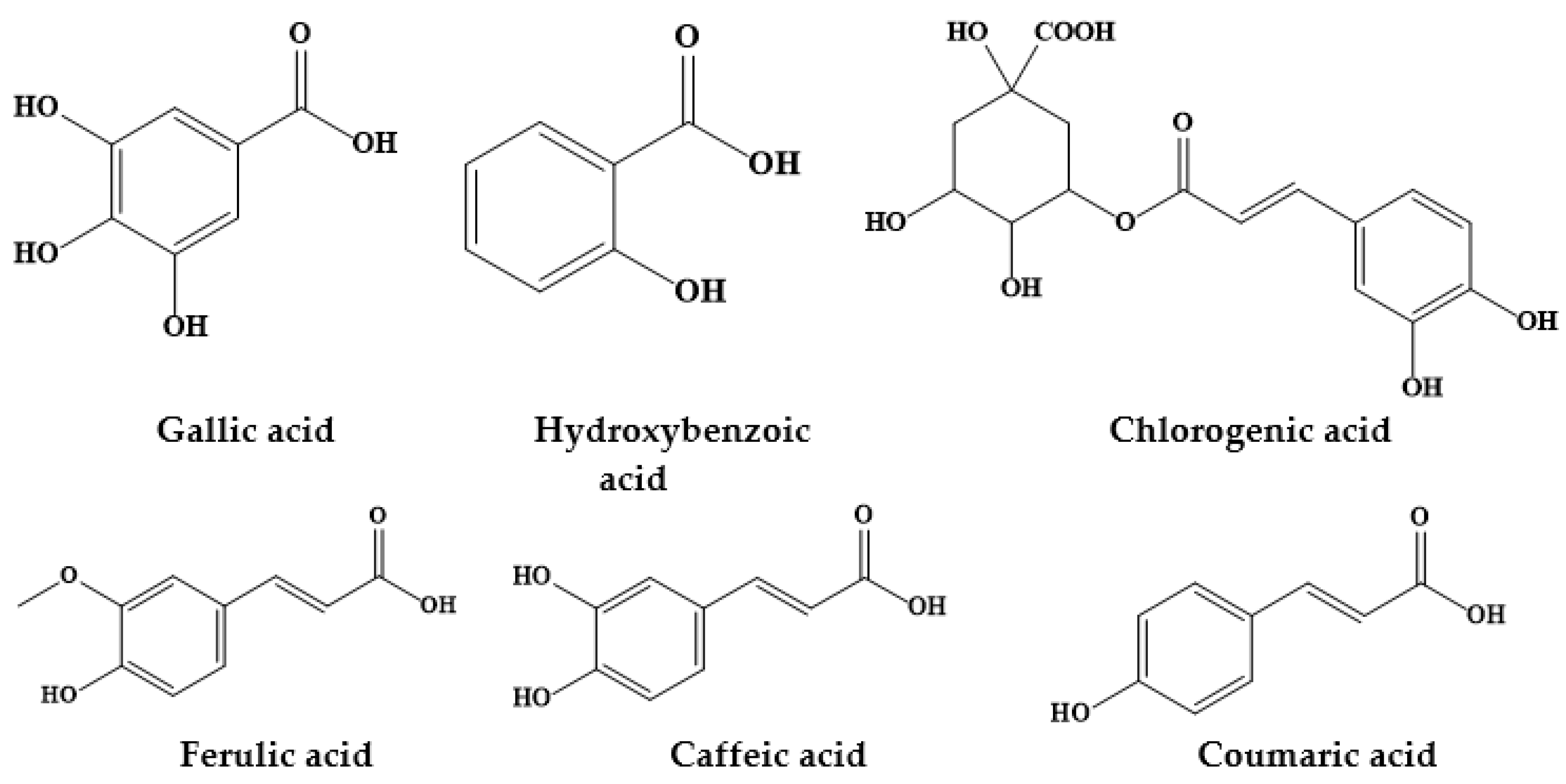
| Prosopis Plant and Part | Identified/Quantified Phytochemicals | References |
|---|---|---|
| P. alba flours | Isovitexin (1.12–0.48 μg/mg) | [9] |
| Vicenin II (1.07–0.34 μg/mg) | ||
| Vitexin (0.91–0.47 μg/mg) | ||
| Schaftoside (0.42–0.00 μg/mg) | ||
| Ferulic acid (4.01–0.28 μg/mg) | ||
| Coumaric acid (3.94–0.33 μg/mg) | ||
| P. alba pods | Q-dihexoside rhamnoside | [10] |
| Q-dihexoside | ||
| Q-methylether dihexoside | ||
| Vitexin | ||
| Q-rhamnoside hexoside | ||
| Isovitexin | ||
| Q-hexoside | ||
| K-hexoside | ||
| P. alba flour | Isoschaftoside hexoside (2.43 mg/g) | [13] |
| Schaftoside hexoside (3.33 mg/g) | ||
| Vicenin II/Isomer (0.67 mg/g) | ||
| Vicenin II/Isomer (2.34 mg/g) | ||
| Isoschaftoside (23.67 mg/g) | ||
| Schaftoside (14.86 mg/g) | ||
| Vitexin (0.46 mg/g) | ||
| Isovitexin (2.09 mg/g) | ||
| P. alba exudate gum | Ferulic acid 4-glucuronide (E) | [11] |
| Apigetrin, chrysin (E) | ||
| Chlorogenic acid (E and NE) | ||
| 3-O-feruloylquinic acid (E) | ||
| p-Coumaroylquinic acid (E) | ||
| Valoneic acid dilactone (E) | ||
| Digallic acid (E) | ||
| Ferulic acid (NE) | ||
| Esculetin derivative (NE) | ||
| 7-O-Methylapigenin (NE) | ||
| P. nigra pods | Cyanidin rhamnosyl hexoside | [10] |
| Cyanidin-3-hexoside | ||
| Peonidin-3-hexoside | ||
| Malvidin dihexoside | ||
| Cyanidin malonoyl hexoside | ||
| Petunidin-3-hexoside | ||
| Malvidin rhamnosyl hexoside | ||
| Malvidin-3-hexoside | ||
| Vicenin II | ||
| Q-dihexoside rhamnoside | ||
| Isoschaftoside | ||
| Q-dihexoside | ||
| Schaftoside | ||
| Q-hexoside rhamnose | ||
| K-hexoside rhamnoside | ||
| Isovitexin | ||
| Q-hexoside | ||
| K-hexoside | ||
| Apigenin hexoside rhamnoside | ||
| Q methyl ether hexoside rhamnoside | ||
| K-methyl ether hexoside rhamnoside | ||
| P. nigra flour | Vicenin II (0.34 μg/mg) | [14] |
| Schaftoside (0.24 μg/mg) | ||
| Isoschaftoside (0.27 μg/mg) | ||
| Isovitexin (0.81 μg/mg) | ||
| Protocatechuic acid (0.33 μg/mg) | ||
| Coumaric acid (8.16 μg/mg) | ||
| Ferulic acid (4.47 μg/mg) | ||
| P. cineraria | Protocatechuic acid (31.65 mg/g) Chlorogenic acid (22.31 mg/g) | [15,16,17] |
| Caffeic acid (6.02 mg/g) | ||
| Ferulic acid (9.24 mg/g) | ||
| Prosogerin A, B, C and D | ||
| β-sitosterol | ||
| Hentriacontane | ||
| Rutin | ||
| Gallic acid | ||
| Patulitrin | ||
| Luteolin | ||
| Spicigerin | ||
| P. laevigata | Gallic acid (8–25 mg/100 g) | [18] |
| Coumaric acid (335–635 mg/100 g) | ||
| Catechin (162.5 mg/100g) | ||
| Gallocatechin (340–648 mg/100 g) | ||
| Epicatechin gallate (10–71 mg/100 g) | ||
| Rutin (222.4–256.1 mg/100 g) | ||
| Morin (236.5 mg/100 g) | ||
| Naringenin (20 mg/100 g) | ||
| Luteolin (13 mg/100 g) | ||
| P. juliflora | 4′-O-Methylgallocatechin | [19,20] |
| (+)-catechins | ||
| (-)-mesquitol | ||
| Apigenin | ||
| Luteolin | ||
| Apigenin-6,8-di-C-glycoside | ||
| Chrysoeriol 7-O-glucoside | ||
| Luteolin 7-O-glucoside | ||
| Kaempferol 3-O-methyl ether | ||
| Quercitin 3-O-methyl ether | ||
| Isoharmentin 3-O-glucoside | ||
| Isoharmentin 3-O-rutinoside | ||
| Quercitin 3-O-rutinoside | ||
| Quercitin 3-O-diglycoside | ||
| P. glandulosa | Gallic acid (8.203 mg/g) | [21] |
| Hydroxybenzoic acid (1.797 mg/g) | ||
| Pyrocatechol (5.538 mg/g) | ||
| Caffeic acid (0.295 mg/g) | ||
| Ferulic acid (0.466 mg/g) | ||
| Quercetin (0.045 mg/g) |
| Scientific Name | Location | Local Name | Parts Used | Administration | Disease(s) Treated/Bioactive Effects | References |
|---|---|---|---|---|---|---|
| P. africana | Sélingué subdistrict, Mali | Guele | Bark trunk | Oral, Bath | Malaria | [19] |
| Guinea-Bissau | Tentera, Buiengué, Bussagan, Coquengue karbon, Késeg-késeg, Paucarvão, Pócarvão, Pó-de-carbom, Po-di-carvom, Tchelem, Tchalem-ai, tchela, Tchelangadje, Tchelem, Bal-tencali, Culengô, Culim-ô, Djandjam-ô, Quéssem-quéssem, Djeiha, Ogea | Leaves, bark, roots | Unspecified | Pains, pregnancy (childbirth, breastfeeding, diseases of the newborn), skin inflammations (wounds, burns) | [20] | |
| Nsukka Local Government Area, South-eastern Nigeria | Ugba | Leaf | Oral | Malaria | [21] | |
| North-West Nigeria | Kirya, Ko-hi | Roots | Oral | Analgesic, anti-inflammatory | [22] | |
| P. alba | Wichí people of Salta province, Argentina | Jwaayukw, Algarrobo blanco | Resin | Oral | Conjunctivitis, post-abortion pain | [23] |
| P. cineraria | Bahawalnagar, Punjab, Pakistan | Drucey | Leaves, stem | Oral | Spasm, diabetes, liver infection, diarrhea, removal of bladder and pancreas stone, fever, flu | [5] |
| Topical | Rheumatism | |||||
| Thar Desert (Sindh), Pakistan | Gujjo | Fruit | Oral | Tonic for body, leucorrhea | [13] | |
| South of Kerman, Iran | Kahour | Fruit | Topical | Asthma, skin rash | [14] | |
| Pakistan | Unspecified | Flower | Oral | Rheumatism | [15] | |
| Hafizabad district, Punjab, Pakistan | Jhand | Leaf, bark, stem, flower, fruit | Oral, topical, eye drop | Liver tonic, boils and blisters, scorpion bite, pancreatic stone, leucorrhoea, chronic dysentery, cataract | [3] | |
| Pakistan | Unspecified | Fruit, pods | Unspecified | Asthma | [4] | |
| Pakistan | Jandi, Kanda, Kandee, Jhand | Leaves, Bark, Flowers, Pods and wood | Oral | Menstrual disorders, contraceptive, prevention of abortion | [16] | |
| P. farcta | Jahrom, Iran | Kourak | Fruit | Oral | Constipation, febrifuge | [24] |
| P. glandulosa Torr | Bustamante, Nuevo León, Mexico | Mezquite | Inflorescences | Oral | Stomach pain | [25] |
| P. juliflora | Thar Desert (Sindh), Pakistan | Devi | Leaves, Gum | Oral | Painkiller, boils opening, eye inflammation, body tonic, muscular pain | [13] |
| Hafizabad district, Punjab, Pakistan | Mosquit pod | Whole plant, Flower, Stem, Leaves, Bark | Oral, topical, and as toothbrush | Galactagogue, kidney stones, toothache, breast cancer, asthma, boils | [3] | |
| Pakistan | Unspecified | Xerophytic shrub | Unspecified | Asthma, cough | [4] | |
| Mohmand Agency, FATA, Pakistan | Kikrye | Leaves | Oral | Lactation, expectorant | [17] | |
| Western Madhya Pradesh, India | Reuja | Stem bark | Oral | Asthma | [18] | |
| P. nigra | Wichí people of Salta province, Argentina | Wosochukw, Algarrobo negro | Resin | Oral | Ocular trauma, conjunctivitis | [23] |
| P. ruscifolia | Wichí people of Salta province, Argentina | Atek, Vinal | Leaves | Oral | Conjunctivitis, stomachache, pimples/rash, scabies, callouses, fever, birth/postpartum pains, diarrhoea, pediculosis, otitis | [23] |
| P. spicigera | Pakistan | Unspecified | Bark, leaves, flowers | Unspecified | Asthma | [4] |
| Species | Model | Plant Part | Formulation/Dosage | Result | Ref. |
|---|---|---|---|---|---|
| Antioxidant | |||||
| P. alba | In vitro method | Edible pods | The sugar-free polyphenolic extracts of and obtained from edible pods and anthocyanins enriched extracts | Polyphenolic extracts of P. alba exhibited activity against a pro-inflammatory enzyme | [10] |
| P. chilensis | In vitro method | Seeds | Seeds were pressurized at 500 MPa during 2, 4, 8 and 10 min. | Antioxidant activity, mineral and starch content and bioaccessibility of samples were significantly affected by the processing and digestion conditions. All treatments enhanced the bioaccessibility of the antioxidant activity (IC50), minerals (dialysis and solubility) and starch (resistant and digestible) as compared to the untreated sample | [38] |
| P. cineraria | In vitro method | Stem bark | Methanolic extract of was analyzed and compared with ascorbic acid as reference 10.52 μg/mL (y = 0.4992x + 101.25, 0.9921) and result is 193.54 μg/mL | MPCL can be used as easily an accessible source of natural antioxidants and as a possible food supplement or in pharmaceutical industry | [39] |
| P. farcta | In vitro method | Aerial part | Oven dried material was grounded into powder (1.5 kg). Powdered materials were soaked in MeOH for 72 h followed by filtration and evaporation. Resulting crude extract was further used for solvent extraction using n-hexane, methylene chloride, ethyl acetate and n-butanol. | P. farcta inhibited ABTS radical in 83.1, 82.0, 87.2 and 87.0%, respectively, for the n-hexane, methylene chloride, ethyl acetate and n-butanol extracts, respectively, when compared to ascorbic acid (89.2%) | [40] |
| P. flexuosa | In vitro method | DNA binding effect was found mainly in the basic fraction. The alkaloids tryptamine as well as piperidine and phenethylamine derivatives were isolated from the basic extracts. | At 0.50 mg/mL, DNA binding activities ranged from 28% for tryptamine to 0–27% for the phenethylamine and 47–54% for the piperidine derivatives. Tryptamine and 2-β-methyl-3-β-hydroxy-6-β-piperidinedodecanol displayed moderate inhibition (27–32%) of β-glucosidase at 100 μg/mL. The exudate of P. flexuosa displayed a strong free radical scavenger effect in the DPPH discoloration assay, with the main active constituent identified being catechin | [41] | |
| P. juliflora | In vitro method | Leaves | Ethanol extract of was investigated for antioxidant activity using in vitro DPPH assay. | Better antioxidant activity (61.55 ± 1.02 RSA %) was found as compared to the control of propyl gallate levels (88 ± 0.07 RSA %) | [26] |
| P. laevigata | In vitro method | Leaves | Leaves were extracted with aqueous acetone (70%) and the polar extract was purified in Sep-Pak® Cartridges and used for evaluation of their fractions. | Significant variations were stated to antioxidant activity among fractions and crude extracts using scavenging hydroxyl and DPPH radical assays | [42] |
| P. nigra | In vitro method | Edible pods | The sugar-free polyphenolic extracts of and obtained from edible pods and anthocyanins enriched extracts | Extract demonstrated antioxidant activity. Polyphenolic extracts exhibited activity against a pro-inflammatory enzyme | [10] |
| Analgesic | |||||
| P. cineraria | In vitro hot-plate method and tail-immersion methods | Roots | Ethanolic extract doses (200 and 300 mg/kg, orally) were selected to study the analgesic activity. | Extracts prevented analgesic property for hot plate and tail immersion method. P. cineraria roots extract at 200 mg/kg was comparatively more effective at higher dose (300 mg/kg body weight) using both assays | [43] |
| Cardioprotective | |||||
| P. laevigata | In vitro method | Leaves | Acetone extracts and purified fractions were dewaxed with petroleum ether and extracted with aqueous acetone (70%); the polar extract was purified and their fractions analyzed | Significant variations among fractions and crude extracts were found in antioxidant capacity by scavenging hydroxyl and DPPH assays. Purified fractions displayed antihypertensive activity, preventing angiotensin converting enzyme and low-density lipoprotein oxidation | [42] |
| Antiplasmodial | |||||
| P. cineraria | Chloroquine (CQ)-sensitive Plasmodium falciparum 3D7 strain and cytotoxicity against THP-1 cell line | Leaves, stem, flowers and roots | Extraction with methanol, chloroform, hexane, ethyl acetate and aqueous sequentially. These extracts were tested in vitro against laboratory adopted P. falciparum 3D7 strain. The crude extracts were also tested for their cytotoxicity against THP-1 cell line. | Ethyl acetate extract of leaf, stem, flower and root and chloroform extract of root showed IC50 values from 5 to 50 μg/mL with good antimalarial activity. Chloroform extracts of leaf, stem, flower and the aqueous extracts of stem, flower and root showed IC50 values of 50–100 μg/mL. The ethyl acetate extract of flower (IC50 = 27.33 μg/mL) showed excellent antimalarial effects. All extracts were non-toxic to THP-1 cells | [44] |
| P. glandulosa | In vitro method | Leaves | Two new indolizidine alkaloid, named Δ1,6-juliprosopine and juliprosine were isolated. The structures of these compounds were elucidated using a combination of NMR and MS. | Compound juliprosine showed potent antiplasmodial activity against P. falciparum D6 and W2 strains (IC50 = 170 and 150 ng/mL, respectively), while Δ1,6-juliprosopine was found to be less active (IC50 = 560 and 600 ng/mL). Both compounds were devoid of VERO cells toxicity up to 23,800 ng/mL. When tested against macrophage cultures, the tertiary bases (Δ1,6-juliprosopine, juliprosine) were found to be more potent than quaternary salts, with IC50 values between 0.8 and 1.7 μg/mL and 3.1– 6.0 μg/mL, respectively | [45] |
| P. juliflora | Plasmodium falciparum | Leaf, bark and flower | The filter sterilized ethanol extracts at 100, 50, 25, 12.5, 6.25 and 3.125 μg/mL doses | Leaf, bark and flower extracts of P. juliflora showed IC50 values >100 μg/mL. Significant antiplasmodial activity was stated between the concentrations and time of exposure. No chemical injury was found in erythrocytes incubated with the ethanolic extract | [27] |
| Antimicrobial | |||||
| P. juliflora | Plasmodium falciparum | MRC-5 cells | Methanol extracted materials screened in vitro against erythrocytic schizonts of intracellular amastigotes of Leishmania infantum and Trypanosoma cruzi and free promastigotes of T. brucei. The criterion for activity was an IC50 <10 μg/mL (<5 μg/mL for T. brucei) and a selectivity index of >4. | Antiplasmodial activity was found to the extracts of P. juliflora | [46] |
| P. juliflora | In vitro method | Leaves | Aqueous, petroleum ether, benzene, chloroform, methanol and ethanol extracts and alkaloid extract studied using poisoned food technique against Alternaria alternata | Aqueous extract recorded significant antifungal activity at 24%. Among different solvent extracts screened, methanol and ethanol extract displayed significantly higher antifungal effects. Methanol extract fractionation leads to the isolation of alkaloid extract with highly significant antifungal activity against the test fungus (minimum inhibitory activity of 1000 ppm). The antifungal activity of alkaloid extract at 2000 ppm or even lower dose was highly effective than the synthetic fungicides. | [34] |
| P. africana | In vitro method | Root and stem | Aqueous and ethanol extract was investigated against various microorganisms (C. albicans, S. mutans and S. saprophyticus) | Ethanol and aqueous extracts of plant parts revealed inhibitory effects on the growth of microorganisms. For both aqueous and ethanol extracts, the inhibitory effect of the stem extract on C. albicans was significantly higher than that exhibited by the root extracts. Ethanol extract exhibited a significant higher inhibitory effect on C. albicans when compared to water extract. The inhibitory effects produced by the aqueous and ethanol extracts on Streptococcus mutans and Staphylococcus saprophyticus did not differ. The effects produced by the stem and root extracts on S. mutans and S. saprophyticus were not significantly different. | [47] |
| P. farcta | In vitro method | Leaf | Aqueous extract and ethanolic extract for potential antibacterial activity against MRSA isolates | MIC/IBC of aqueous and ethanolic extracts of P. farcta was 100, 125 mg/mL and 25, 112.5. mg/mL respectively | [48] |
| P. juliflora | In vitro method | Leaf | Silver nanoparticles (AgNPs) synthesized using an aqueous extract | Concentration-dependent activity was shown against E. coli and P. aeruginosa. Most of the studied microorganisms showed sensitivity to methanolic extract (inhibition zone ranging from 12 to 41 mm). The largest inhibition zone was detected against to P. aeruginosa (41 mm) and L. monocytogenes (33 mm) using 100 mg/mL | [28] |
| P. glandulosa | In vitro method | Leaves | Ethanolic extract against 17 microorganisms using disc diffusion method | Ethanolic extract exhibited moderate-to-high inhibitory activity against bacteria and fungi. The maximum inhibitory activity was showed against C. neoformans (30.6 mm), C. albicans (20.0 mm), S. epidermidis (21.8 mm), S. aureus (17.4 mm), Shigella flexneri (19.8 mm), Proteus vulgaris (18.0 mm) and Vibrio parahaemolyticus (15.8 mm) | [49] |
| P. cineraria | In vitro method | Antifungal protein (38.6 kDa) from P. cineraria extract was purified using ammonium sulphate precipitation, ion exchange chromatography and gel filtration | Purified protein exerted antifungal activity against post-harvest fruit fungal pathogens Lasiodiplodia theobromae and Aspergillus fumigatus | [50] | |
| P. farcta | In vitro method | Aerial part | Oven dried material was grounded into powder (1.5 kg), soaked in MeOH for 72 h followed by filtration and evaporation. Resulting crude extract was further used for solvent extraction using n-hexane, methylene chloride, ethyl acetate and n-butanol. | n-hexane and methylene chloride extract exhibited moderate antimicrobial activities against Shigella spp., E. coli and Proteus vulgaris for n-hexane and Erwinia spp., E. coli and S. epidermis for methylene chloride. Ethyl acetate displayed higher antimicrobial activities against Shigella spp., E. coli, and C. albicans. Likewise, n-butanol extract showed higher activity against Shigella spp., Erwinia spp., E. coli, P. vulgaris, S. epidermis and C. albicans | [40] |
| P. juliflora | In vitro method | Acetone, chloroform, diethyl ether, methanol, ethanol and DMSO extract of P. juliflora was investigated for their antimicrobial activities. The extracts and the tetracycline as control were tested in vitro against 6 bacterial species and 4 fungal species by well diffusion method. E. coli, B. subtilis, S. marcescens, K. pneumoniae, S. aureus, P. fluorescens, P. tromiformis var. occidentalis, P. foedans, F. oxysporum and P. variotii were used | DMSO extract displayed the better antibacterial activity against E. coli (21 mm), S. marcescens (16 mm), S. aureus (17.9 mm), P. fluorescens (16.5 mm), P. mycesvariotii (13.2 mm) and P. leptostromiformis (11 mm). Methanol extract showed the better activity against B. subtilis (23 mm) and P. foedans (16 mm). Ethanol extract exhibited better activity against K. pneumoniae (11 mm); however, no extract displayed activity against the fungi F. oxysporum | [35] | |
| P. juliflora | In vitro method | Pods | Antimicrobial activity of alkaloid-enriched extracts from P. juliflora pods | Basic chloroformic extract (main constituents were juliprosopine, prosoflorine and juliprosine) exhibited antibacterial activity against Micrococcus luteus (MIC = 25 μg/mL), S. aureus (MIC = 50 μg/mL) and S. mutans (MIC = 50 μg/mL). The extract reduced gas production as efficiently as monensin after 36 h fermentation, revealing positive influence on gas production during ruminal digestion | [36] |
| P. farcta | In vitro method | Pods | Six isolates of Sphingomonas paucimobilis were isolated from 120 hospital workers hands in Erbil city/Iraq by using VITEK2 Compact system, then further confirmed by PCR technique and by detecting specific gene TDP-glucose pyrophosphorylase (320bp) for S. paucimobilis ATCC 31,461 and all local isolates. | The minimum inhibitory concentration (MIC) of P. farcta pods extracts against S. paucimobilis isolate (S.p4) was 1000 μg/mL for methanol and ethanol extracts and 1200 μg/mL of watery extract | [51] |
| P. juliflora | In vitro method | Comparative assessment of antibacterial activity of crude extract of P. juliflora with commercially available mouthrinses on oral and periodontal organisms | P. juliflora activity was highest in comparison with the other commercial mouthrinses against selected microbes | [52] | |
| P. juliflora | In vitro method | Seed pods | Methanol extract of P. juliflora at varying doses (0.05, 0.1, 0.2, 0.3, 0.4 mg mL) against S. aureus, Micrococcus luteus, Bacillus cereus, Shigella sonee, P. aeruginosa and E. coli | P. aeruginosa was the maximum resistant and Micrococcus luteus the less resistant to the extract | [30] |
| P. juliflora | In vitro method | Seed pods | In vitro antibacterial activity of the P. juliflora seed pods extract was screened against S. aureus, S. epidermidis, E. coli and P. aeruginosa | P. juliflora seed pods extract demonstrated antibacterial activity against all four test organisms. MIC of the extract was 0.312 mg/mL and 0.078 mg/mL, respectively for S. aureus and S. epidermidis, and 1.25 mg/mL for both E. coli and P. aeruginosa | [29] |
| P. kuntzei and P. ruscifolia | In vitro method | Dry extracts, dissolved in DMSO, were tested for inhibition of microbial growth via microplate assay with an oxidation-reduction dye. | P. kuntzei and P. ruscifolia exhibited MIC values ranging from to 0.08–0.5 mg dry matter/mL. All extracts at 2 × MIC were able to inhibit bacterial growth effectively, and were able to reduce the initial number of viable counts (A. balansae, G. decorticans, P. dubium, G. spinosa, P. kuntzei and B. sarmientoi) by at least one order of magnitude in 10 h | [53] | |
| P. cineraria | In vitro method | Aerial parts | Organic extract was prepared via maceration in methanol, followed by the fractionation using hexane and ethyl acetate. | The best antibacterial activities were detected to the ethyl acetate fraction. The effective antibacterial constituents of the plant were two substances with molecular weight of 348 and 184 Dalton (MIC values <125 to 62.5 μg/mL) | [54] |
| P. cineraria | In vitro method | Pods | Chloroform and benzene extracts | The antimicrobial property was examined by disc diffusion assay against three gram-positive (B. subtilis, S. aureus, M. smegmatis) and three gram-negative (P. aeruginosa, K. pneumoniae, and E. coli). Chloroform pods extract was found effective against K. pneumoniae while benzene found effective against K. pneumoniae, E. coli and B. subtilis | [55] |
| P. cineraria | In vitro method | The antimicrobial alkaloids, juliflorine, julifloricine and benzene insoluble alkaloidal fraction of P. juliflora, were studied for their therapeutic efficacy after topical application in produced superficial skin infection. Infection was produced by rubbing freshly isolated Staphylococcus aureus from human clinical specimen onto 9 cm2 shaved skin. | Juliflorine was effective on Staphylococcal skin infection. Juliforine at 0.5, 1, and 2.5% were found to heal 25, 50 and 100% lesions in two weeks and microbiological efficacy was found to be 16.66, 33.33, 58.33 and 91.66% with 0.1, 0.5, 1 and 2.5% of juliflorine. Julifloricine was less effective when compared with juliflorine and the benzene insoluble alkaloidal mixture was comparatively more effective than juliflorine. Healing was slightly faster with the mixture. Both juliflorine and the mixture exhibited effectiveness at 2.5% concentration, however these were also found toxic. Gentamicin was found superior to the alkaloids in artificially produced skin infection | [56] | |
| P. juliflora | Crude extracts were with three different solvents and examined for preliminary antibacterial activity | Varying degrees of growth inhibition were shown by all fractions. The highest antibacterial activity was observed for aqueous fractions as compared to solvent fractions. | [32] | ||
| P. cineraria | Human pathogens | Leaf | Characterization of silver (PcAgNPs) and copper nanoparticles (PcCuNPs)was performed using P. cineraria leaf extract synthesized using microwave irradiation | The bioengineered silver and copper nanohybrids showed enhanced antimicrobial activity against Gram-positive and Gram-negative MDR human pathogens. | [57] |
| Anthelmintic, Antiprotozoal and Anti-trypanosomal | |||||
| P. juliflora | Haemonchus contortus isolated from naturally infected sheep | Roots and leaves | Roots and leaves of P. juliflora were extracted with ethanol and evaluated for anthelmintic activity according to standard procedures. | In larval mortality assay, all microencapsulated extracts of P. juliflora (leaves and roots) induced over 50% mortality at the highest concentration used (2 mg/mL). Albendazole required a maximum concentration of 0.25 mg/mL to induce 100% larval mortality. There was a significant difference in larval mortality compared to that of egg hatchability. There was a marked difference in mean percentage of adult mortality of H. contortus at different concentrations and ratios. All assays showed dose-dependent response. | [37] |
| P. juliflora | Goat gastrointestinal nematodes | pods | In vitro anthelmintic activity of the alkaloid containing fraction | High ovicidal activity was recorded with IC and IC values at 1.1 and 1.43 mg/mL for alkaloid rich fraction. This fraction also exhibited low larvicidal activity and high toxic effect | [37] |
| P. cineraria | Pheretimaposthuma | Bark | Extract in methanol is prepared and used for screening | Time required for the paralysis and death was recorded. Methanol extract was more potent than petroleum ether and aqueous extracts | [58] |
| P. africana | In vitro method | Leaves, stem bark and roots | Petroleum ether, chloroform, methanol and aqueous extracts, obtained by cold extraction | All solvent extracts showed strong in vitro anti-trypanosomal activity at both 2 and 4 mg/mL | [59] |
| Apoptosis | |||||
| P. juliflora | BCL2 protein using molecular docking approach | Five bioactive compounds, namely 2-pentadecanone; butyl 2-ethylhexyl phthalate; methyl 10-methylheptadecanoate; methyl oleate; and phorbol-12, 13- Dihexanoate were identified using GC-MS analysis | Phorbol-12,13-dihexanoate showed best docking score of −15.644 Kcal/mol, followed by methyl oleate (−13.191 Kcal/mol) | [60] | |
| Antinociceptive | |||||
| P. strombulifera | In vitro J774A.1 macrophage-derived cell line | Fruits | Fruit extract at varying concentrations in different solvent system | Chloroform (100 μg/mL) produced inhibition of LPS-induced NO production, which was not observed with ethanol and ethyl acetate at the same concentration. NO production inhibition by chloroform (10–100 μg/mL) was dose-dependent, (IC50 = 39.8 (34.4–46.1) μg/mL, and chloroform significantly inhibited LPS-induced iNOS expression in J774A.1 cells | [61] |
| Anticancer | |||||
| P. farcta | Cell lines namely; HepG-2, HeLa, PC3 and MCF-7. | Aerial part | Oven dried material was grounded into powder (1.5 kg), soaked in MeOH for 72 h followed by filtration and evaporation. Resulting crude extract was further used for solvent extraction using n-hexane, methylene chloride, ethyl acetate and n-butanol. | n-butanol extract showed the highest activity against MCF-7 cell line (IC50 = 5.6 μg/mL) compared to 5-fluorouracil (IC50 = 5.4 μg/mL), while ethyl acetate showed the highest activity against Hela cell line (IC50 = 6.9 μg/mL) compared to 5-fluorouracil (IC50 = 4.8 μg/mL) | [40] |
| P. cineraria | Breast cancer cells (MCF-7) | Leaf | Characterization of silver (PcAgNPs) and copper nanoparticles (PcCuNPs) was performed using P. cineraria leaf extract synthesized using microwave irradiation | MTT assay results indicated that CuNPs show potential cytotoxic effect followed by AgNPs against MCF-7 cancer cell line. IC50 values were 65.27, 37.02 and 197.3 for PcAgNPs, PcCuNPs and P. cineraria leaf extracts, respectively | [57] |
| Toxicity | |||||
| P. juliflora | Neurons and glial cells | leaves | Total alkaloid extract (TAE) and one alkaloid fraction (F32) at concentrations between 0.3 and 45 μg/mL were tested for 24 h on neuron/glial cell primary cocultures. | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide test revealed that TAE and F32 were cytotoxic to cocultures (IC50 were 31.07 and 7.362 μg/mL, respectively). Exposure to a subtoxic concentration of TAE or F32 (0.3–3 μg/mL) induced vacuolation and disruption of the astrocyte monolayer and neurite network, ultrastructural changes, characterized by formation of double-membrane vacuoles, and mitochondrial damage, associated with changes in β-tubulin III and glial fibrillary acidic protein expression. Microglial proliferation was also observed in cultures exposed to TAE or F32, with increasing levels of OX-42-positive cells | [62] |
| P. juliflora | Spodopteralitura larvae | Seed pod | A significant increase in the total hemocyte count was found. P. juliflora seed pod hexane extract was effective in producing lepidopteran larval mortality may be due to the presence of 9-Octadecyne | [63] | |
| P. juliflora | Neuron/glial cell co-culture | Leaves | A total extract (TAE) of alkaloids and fraction (F32) composed mainly of juliprosopine | TAE (30 μg/mL) and F32 (7.5 μg/mL) reduced ATP levels and led to changes in mitochondrial membrane potential at 12 h exposure. TAE and F32 induced caspase-9 activation, nuclear condensation and neuronal death at 16 h exposure. After 4 h, they induced autophagy characterized by decreases of P62 protein level, increase LC3II expression and increase GFP-LC3 cells number | [64] |
| Species | Model | Plant Part | Formulation/Dosage | Result | Ref. |
|---|---|---|---|---|---|
| Antidiabetic | |||||
| P. cineraria | STZ-induced diabetic rats | Stem bark | Chloroform fraction of species was orally administrated to STZ-induced diabetic rats at the doses of 50 and 100 mg/kg b.w for 21 days | Significant reduction in blood glucose, glycosylated hemoglobin levels and body weight, liver glycogen content and serum insulin level restoration, in a concentration-dependent manner. Decrease in serum lipid profile markers and elevation in HDL after administration, also evidencing protective effects in diabetes-associated complications | [69] |
| P. cineraria | Hyperlipidemic rats | Fruits | Extract was obtained by pulverizing whole dried fruits and extracting with 70% ethanol. | Decrease in serum cholesterol, triglyceride, VLDL and LDL levels. At 400 and 600 mg/kg, the extract significantly reduced serum cholesterol, triglyceride, VLDL, LDL and atherogenic index and these results are almost equivalent to those of drug simvastatin | [70] |
| P. cineraria | Male Swiss albino mice | Bark | Dried material was powdered followed by Soxhlet extraction with 50% aqueous ethanol and concentrated. Oven dried material was suspended in 20% tween 20 in normal saline for further experimentation. | Reduced blood glucose level, enhanced hepatic glycogen content and maintained body weight and lipid-profile attributes towards near normal range. Reduced antioxidant enzymes activity and concentration of non-enzymatic antioxidants, thereby decreasing the oxidative damage in the tissues of diabetic animals and hence indicating the anti-diabetic and antioxidant efficacy of the extract | [71] |
| P. cineraria | Hyperlipidemic rabbits | Bark | Rabbits were orally supplemented with high fat diet and cholesterol powder (500 mg/Kg body weight per day in 5 mL of coconut oil orally for 15 days) to create a hypolipidemic model. | Decreased serum total cholesterol, LDL, triglyceride, VLDL and also ischemic indices (TC/LDL and LDL/HDL). The Prevented the atherogenic changes in aorta. Toxicity profile parameters remained under normal ranges | [72] |
| P. glandulosa | Male Wistar rats (type 1 diabetic model) | Half of each group of animals was placed on treatment (100 mg/kg/day) for 8 weeks and the remaining animals served as age-matched controls. | Enhanced insulin levels, with a significant decrease in blood glucose levels. Increased small β-cells level in pancreas. Reduced fasting glucose levels and improved IPGTT. Increased and insulin-stimulated glucose uptake by cardiomyocytes | [73] | |
| P. ruscifolia | Alloxan-induced diabetic rats | Aerial part | Different animal groups were administered with a single dose of water, extract (100 mg/Kg), tolbutamide (100 mg/Kg, p.o.) or insulin (5 IU/kg, i.p.). Normoglycemic rats were also treated with hydroalcoholic extract (100 mg/kg, p.o.). | No evidence of acute toxicity. Blood glucose levels were significantly (p < 0.01) decreased with a single oral dose (100 mg/Kg) after 24 h. Blood glucose levels decreased significantly with administration of plant extract during 28 days | [74] |
| Neuroprotective | |||||
| P. farcta | Male Wistar rats | Pod | Aqueous extract of P. farcta injections (25, 50, 75 mg/kg, ip, 2 time) and (compression + ethanol extract of P. farcta injections (25, 50, 75 mg/kg, i.p., 2 time) (N = 8). | Comparative assessment of neuronal density of compression and control groups exhibited marked variations. A meaning full variation was recorded between compression and all treatment groups | [75] |
| Wound healing | |||||
| P. cineraria | Rats using excision and incision wound model | Ethyl acetate, chloroform and butanol fractions of species hydroethanolic extract were assessed for their antioxidant activity using in vitro method | Butanol fraction found most active fraction against free radicals among all. Butanol fractions possess significant anti-inflammatory, anti-collagenase and anti-elastase activities. Application of butanol fraction ointment for 16 consecutive days on the dorsal wound area of rats confirmed the faster wound repairing process, higher hydroxyproline content, reduction in epithelialization period and inflammatory markers in blood as compared to control group | [76] | |
| P. cineraria | Male albino wistar rats | Leaves | Wound excised rats administered with ethanolic extract for 13 days period. | Decrease in wound area as compared to control | [77] |
| Antipyretic | |||||
| P. cineraria | Brewer’s-yeast induced pyrexia in albino rat | Leaves and fruits | At a dose of 200 and 300 mg/kg of body weight was investigated. | Reduced hyperpyrexia to a significant level as compared to standard control. Lowered the rectal temperature of rats than fruits extract at 200 mg/kg while at dose of 300 mg/kg both leaves and fruit extract significantly decrease pyrexia. | [78] |
| P. juliflora | Male rats | Twenty-four male rats were randomly allotted to four groups (6 animals per group) and food was deprived off for 48 h water provided but before 24 h of experiment, water also withheld. Group 1 was treated with water for injection (100 mL/kg). Group 2 treated with Paracetamol (150 mg/kg p.o dissolved in water for injection). Group 3 and 4 were treated with ethanol extract of P. juliflora (250 and 300 mg/kg p.o respectively). Temperature maintained at ± 3 °C, for 0 to 4 h of interval at the dose of 250 mg/kg | Decreased the rectal temperature at 3 h and at dose 500 mg/kg. Reduced the rectal temperature at 2, 3 and 4 h in comparison with vehicle control. | [79] | |
| Spasmolytic, bronchodilator, and vasodilator activities | |||||
| P. cineraria | In vivo method | Stem bark | The extract at 3–10 mg/mL doses | The extract caused relaxation of the spontaneous as well as K+ (80 mM)-stimulated contractions at tissue bath concentrations of 3–10 mg/mL in isolated rabbit jejunum preparations. Extract displayed nonspecific relaxant effect on carbachol (1 μM)- and K+ (80 mM)-induced contractions in isolated rabbit tracheal preparations. | [80] |
| Depression and CNS disorder | |||||
| P. cineraria | Mice | Leaf | Antidepressant effect was evaluated using Forced swim test (FST). The immobility periods of control and treated mice were recorded. | Leaf extract (200 mg/kg) significantly decreased the duration of immobility time in FST. The efficacy of tested extract was comparable to that of imipramine | [81] |
| Cardiovascular disorder | |||||
| P. farcta | Rabbit | Root | The study evaluated the efficacy of aqueous extract of P. farcta root on experimental atherosclerosis development in rabbits with high cholesterol diet–induced hypercholesterolemia. | Serum lipid parameters were significantly increased in the high cholesterol diet groups in comparison with the normal control group. Treatment with P. farcta root decreased total cholesterol, triglyceride, high-density lipoprotein, low-density lipoprotein, and very low-density lipoprotein levels compared to high cholesterol diet rabbits | [82] |
| P. farcta | Rabbits | Root | High cholesterol diet–induced NAFLD in rabbits as experimental model. Male rabbits randomly divided into 4 groups namely, control (fed by standard pellet) and other groups were received 2% cholesterol amounts daily. Rabbits were fed with high cholesterol diet till the serum cholesterol level reached 1800 mg/dl, then, they were treated daily with distilled water, and 0.6 mg/kg Simvastatin, or 500 mg/kg/day P. farcta root extracts orally by gavage for 30 days. | Serum lipid parameters and enzymes were significantly enhanced in the high cholesterol diet groups in comparison with the normal control group. Histopathological findings revealed that large lipid vacuoles were formed in hepatocytes. Treatment with P. farcta root significantly improved rabbit lipid profile and reduced liver injury. | [83] |
| P. farcta | Wistar albino rats | Beans | Thirty-six male Wistar albino rats weighing 220 ± 30 g were distributed into six groups. Two groups were pretreated with extract (50 and 75 mg/kg) for 7 days before administration of acetaminophen (600 mg/kg). Two were given acetaminophen or extract (50 and 75 mg/kg) alone, and the control received normal saline. | Extract at both doses significantly attenuated total cholesterol, triglyceride, high-density lipoprotein, low-density lipoprotein, and very low-density lipoprotein levels to near normal | [84] |
| P. farcta | Rat’s thoracic aorta | In vivo method | P. farcta plant extract was firstly prepared. Then 2 cm of rat’s thoracic aorta was dissected and was divided into 4 pieces of 5 mm. After contraction of these pieces by phenylephrine (1 μm), different dosages of plant extraction (0.5, 1 and 2 mg/mL) were examined and the effect of plant extract on rat’s aorta with and without endothelium layer was measured. Different dosages of P. farcta extract (1 and 2 mg/mL) at the presence and absence of L- NAME (a nitric oxide synthetase inhibitor) was examined. | P. farcta extract showed a dose-dependent relaxing effect on contracted aorta. The relaxing effect of plant extract on aorta with endothelium was more significant than that on aorta without endothelium in the different dosages. The relaxing effect of plant extract in the presence of L- NAME was decreased significantly. The relaxing effect of plant extract was more than that by acetylcholine. | [85] |
| Inflammation and regeneration | |||||
| P. glandulosa | Gastrocnemius muscle of rats | The gastrocnemius muscle of rats was subjected to mass-drop injury and muscle samples collected after 1-, 3 h, 1- and 7 days post-injury. Rats were treated with P. glandulosa (100 mg/kg/day) either for 8 weeks prior to injury (up until day 7 post-injury), only post-injury, or with topically applied diclofenac post-injury (0.57 mg/kg). | Chronic P. glandulosa and diclofenac treatment was associated with neutrophil response suppression to contusion injury, however only chronic P. glandulosa treatment facilitated more effective muscle recovery, while diclofenac treatment had inhibitory effects on repair, despite effective inhibition of neutrophil response. | [86] | |
| Skin caring and antiaging | |||||
| P. cineraria | Stem, leaves, and bark | Performance of 2% bark extract loaded emulsion formulation was determined by using non-invasive probe cutometer and elastometer with comparison to base formulation | Bark extract did not induce any toxicity or apoptosis, when incubated with HaCat cells. Moreover, the formulation (size 3 μm) decreased the skin melanin, erythema and sebum contents up to 2.1-,2.7-and 79%, while increased the skin hydration and elasticity up to 2-folds and 22% as compared to the base, respectively. Owing to enhanced therapeutic effects the phytocosmetic formulation proved to be a potential skin whitening, moisturizer, anti-acne, anti-wrinkle, anti-aging therapy and could actively induce skin rejuvenation and resurfacing | [87] | |
| Antimalarial | |||||
| P. juliflora | Murine model | Leaves and pods | Alkaloid-enriched extracts from (BCE) of P. juliflora, as well as FACB pure constituents (as formate salts), were obtained and assayed against Plasmodium berghei NK65 infection in mice via oral supplementation. | Alkaloid-enriched extracts from leaves and pods showed remarkable antimalarial activity with little parasitemia inhibition at the 2 mg/kg dose. Julifloridine was weakly active, but juliprosopine caused a parasitemia inhibition at the 2 mg/kg dose similar to that recorded for chloroquine at 50 mg/kg | [88] |
| Anti-trypanosomal properties | |||||
| P. africana | In vivo method | Leaves, stem bark and roots | petroleum ether, chloroform, methanol and aqueous extracts, obtained by cold extraction from the | Only the methanolic extract of leaves displayed promising anti-trypanosomal effect at 200 mg/kg dose | [59] |
| Antinociceptive | |||||
| P. strombulifera | Formalin-induced pain test in mice | Fruits | Fruit extract at varying concentrations in different solvent system | Chloroform (300 mg/kg), in contrast to ethanol and ethyl acetate extract, caused significant inhibition of the in vivo nociceptive response. Moreover, chloroform (100–1000 mg/kg, p.o.) produced a dose-dependent inhibition of neurogenic and inflammatory phases of the formalin test with inhibition values (at 600 mg/kg) of 42 ± 7 and 62 ± 7%, respectively. Antinociception was significantly attenuated by i.p. treatment of mice with l-arginine (600 mg/kg) | [61] |
| Species | Model | Plant Part | Formulation/Dosage | Result | Ref. |
|---|---|---|---|---|---|
| Cytotoxicity | |||||
| P. juliflora | Wistar rats | In an acute toxicity study P. juliflora extract was administered orally at doses ranging from 50 to 500 mg kg−1 and the animals were observed for any toxic symptoms for 72 h. In a subacute toxicity study ethanolic extracts of P. juliflora were tested at a dose of 200 mg kg−1 orally once daily for 30 days. | No changes in hematological, biochemical, renal and liver function parameters were stated in experimental animals of in this sub-chronic study when compared to control animals | [93] | |
| P. juliflora | Cattle and goats | Leaves | Cattle and goats experimentally intoxicated presents neurotoxic damage in the central nervous system | Histologic lesions were mainly characterized by vacuolation and loss of neurons in trigeminal motor nuclei. Mitochondrial damage in neurons and gliosis was reported in trigeminal nuclei of intoxicated cattle | [96] |
| P. cineraria | Swiss albino mice | Leaves | Different doses of extract were used for screening. | The extracts of investigated plants are relatively safe at the dose of 100 mg/kg b.w. | [94] |
| P. juliflora | Astrocyte primary cultures derived from the cortex of newborn Wistar rats | In vivo method | TAE and seven alkaloidal fractions, at concentrations ranging 0.03–30 μg/ml | TAE and fractions F29/30, F31/33, F32 and F34/35 were cytotoxic to astrocytes, with EC50 values for the most toxic compounds, TAE, F31/33 and F32, being respectively, 2.87 2.82 and 3.01 μg/mL. Astrocytes exposed to 3 μg/mL TAE, F29/30 or F31/33 developed compact cell body with many processes overexpressing GFAP. Treatment with 30 μg/mL TAE and fractions, induced cytotoxicity characterized by a strong cell body contraction, very thin and long processes and condensed chromatin. Also, the proportion of OX-42 positive cells was increased in cultures treated with 30 μg/mL TAE or F29/30, F31/33, F32 and F34/35, with values raging from 7.27 to 28.74%. Moreover, incubation with 3 μg/mL F32, 30 μg/mL TAE, F29/30, F31/33 or F34/35 induced accumulation of nitrite in culture medium indicating induction of NO production | [97] |
| P. glandulosa | Mice | 2,3-Dihydro-1H-indolizinium alkaloid-prosopilosidine (PPD) was studied against C. neoformans in a murine model of cryptococcosis. Mice were infected via the tail vein with live C. neoformans. Twenty-four hours post-infection, the mice were administered with PPD once a day (i.p.) or twice a day (bid) orally, or with amphotericin B (Amp B) intraperitoneally (IP), or with fluconazole (Flu) orally for 5 days | PPD showed potent in vivo activity against C. neoformans at 0.0625 mg/kg by eliminating ~76% of the organisms compared to ~83% with Amp B (1.5 mg/kg). In addition, PPD was equally efficacious, but less toxic, at either 0.125 or 0.0625 mg/kg compared to Amp B (1.5 mg/kg) when it was administered bid (twice a day) i.p. When tested by an oral route, PPD (10 mg/kg) showed potent activity in this murine model of cryptococcosis with ~82% of organisms eliminated from the brain tissue, whereas Flu (15 mg/kg) reduced ~90% of the infection. | [90] | |
| Fertility | |||||
| Mesquite | Female and male rats | Pod | Female and male rats’ group as vehicle, mesquite pod extract, DAI and E2 were administered subcutaneously for 30 days. | These extracts disrupted both female and male sexual behavior in a similar way to DAI, but less than E2. Mesquite pod extract increased the number of days in estrus and decreased lordosis intensity during proestrus. Mesquite pod extract-treated males showed lower testicular and glandular weights, as well as decreased sperm motility, viability and count. In females treated with mesquite pod extract, the number of pups was lower than in control females, and 10 to 20% of pups were dead. These effects were similar to those with DAI-treatment. Despite the lower sperm quality, the fertility of mesquite pod extract- and DAI-treated males seem not to be disrupted, as they could impregnate control females | [98] |
| Mesquite | Male rat | Pod | The following treatments were given to groups of intact male rats: vehicle; mesquite pod extract; E; DAI; GEN. | Mesquite pod extracts disrupt male sexual behavior in a similar way to DAI and GEN, but less than E. The main disruptor of sexual behavior was E, however after 40 and 50 days of administration, extracts and phytoestrogens disrupted sexual behavior in a similar way to E. The extracts also increased testicular germ cell apoptosis, decreased sperm quality, testicular weight, and testosterone levels, as phytoestrogens did, although these effects were less than those caused by estradiol. Number of seminiferous tubules increased in extracts-treated groups in a similar way to phytoestrogens groups, and E caused the greatest effect. Testicular atrophy was only observed in estradiol-treated males. Testosterone declined in males of all experimental groups compared with control. Mesquite pod extracts cause effects similar to those of phytoestrogens in male rat reproduction, these effects were lower than those caused by E | [99] |
| Allergy | |||||
| Mesquite | PAR patients | Pollen | Patients demonstrating a positive PST response to mesquite only were used for mesquite conventional subcutaneous ASIT | 86/200 patients displayed a positive PST response to mesquite allergen, of them, 38 were positive to mesquite allergen only. Remarkable attenuation in symptom and medication scores were recorded in 24/38 patients 4 months post-ASIT initiation | [100] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharifi-Rad, J.; Kobarfard, F.; Ata, A.; Ayatollahi, S.A.; Khosravi-Dehaghi, N.; Jugran, A.K.; Tomas, M.; Capanoglu, E.; Matthews, K.R.; Popović-Djordjević, J.; et al. Prosopis Plant Chemical Composition and Pharmacological Attributes: Targeting Clinical Studies from Preclinical Evidence. Biomolecules 2019, 9, 777. https://doi.org/10.3390/biom9120777
Sharifi-Rad J, Kobarfard F, Ata A, Ayatollahi SA, Khosravi-Dehaghi N, Jugran AK, Tomas M, Capanoglu E, Matthews KR, Popović-Djordjević J, et al. Prosopis Plant Chemical Composition and Pharmacological Attributes: Targeting Clinical Studies from Preclinical Evidence. Biomolecules. 2019; 9(12):777. https://doi.org/10.3390/biom9120777
Chicago/Turabian StyleSharifi-Rad, Javad, Farzad Kobarfard, Athar Ata, Seyed Abdulmajid Ayatollahi, Nafiseh Khosravi-Dehaghi, Arun Kumar Jugran, Merve Tomas, Esra Capanoglu, Karl R. Matthews, Jelena Popović-Djordjević, and et al. 2019. "Prosopis Plant Chemical Composition and Pharmacological Attributes: Targeting Clinical Studies from Preclinical Evidence" Biomolecules 9, no. 12: 777. https://doi.org/10.3390/biom9120777
APA StyleSharifi-Rad, J., Kobarfard, F., Ata, A., Ayatollahi, S. A., Khosravi-Dehaghi, N., Jugran, A. K., Tomas, M., Capanoglu, E., Matthews, K. R., Popović-Djordjević, J., Kostić, A., Kamiloglu, S., Sharopov, F., Choudhary, M. I., & Martins, N. (2019). Prosopis Plant Chemical Composition and Pharmacological Attributes: Targeting Clinical Studies from Preclinical Evidence. Biomolecules, 9(12), 777. https://doi.org/10.3390/biom9120777












