Dithiocarbamate as a Valuable Scaffold for the Inhibition of Metallo-β-Lactmases
Abstract
1. Introduction
2. Materials and Methods
2.1. General Information
2.2. Synthesis of Inhibitors
2.3. Inhibition Studies
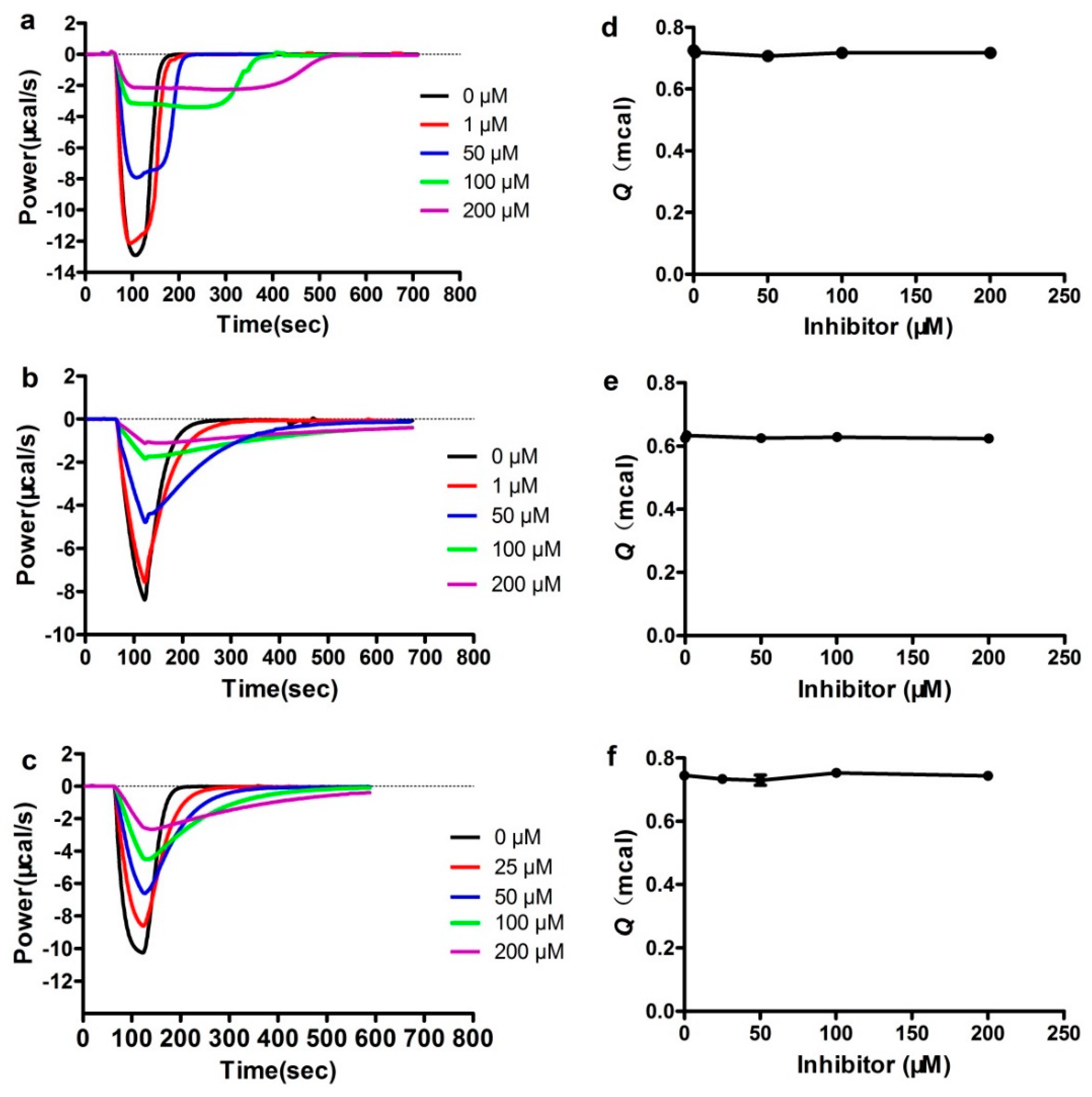
2.4. Isothermal Titration Calorimetry (ITC) Assays
2.5. MIC Determination
2.6. Docking Studies
2.7. Cytotoxicity Evaluation
3. Results and Discussion
3.1. Synthesis
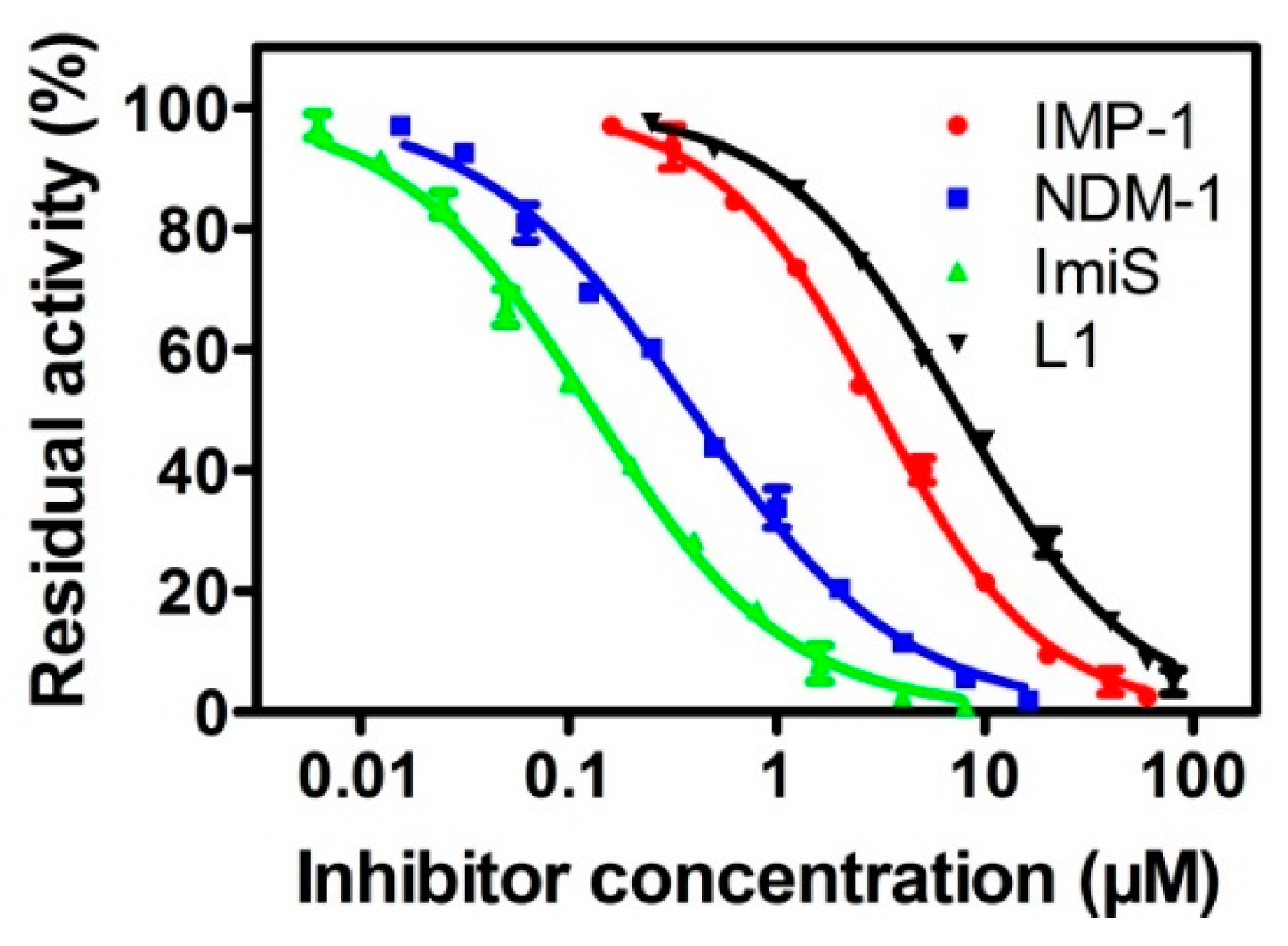
3.2. Inhibition Studies
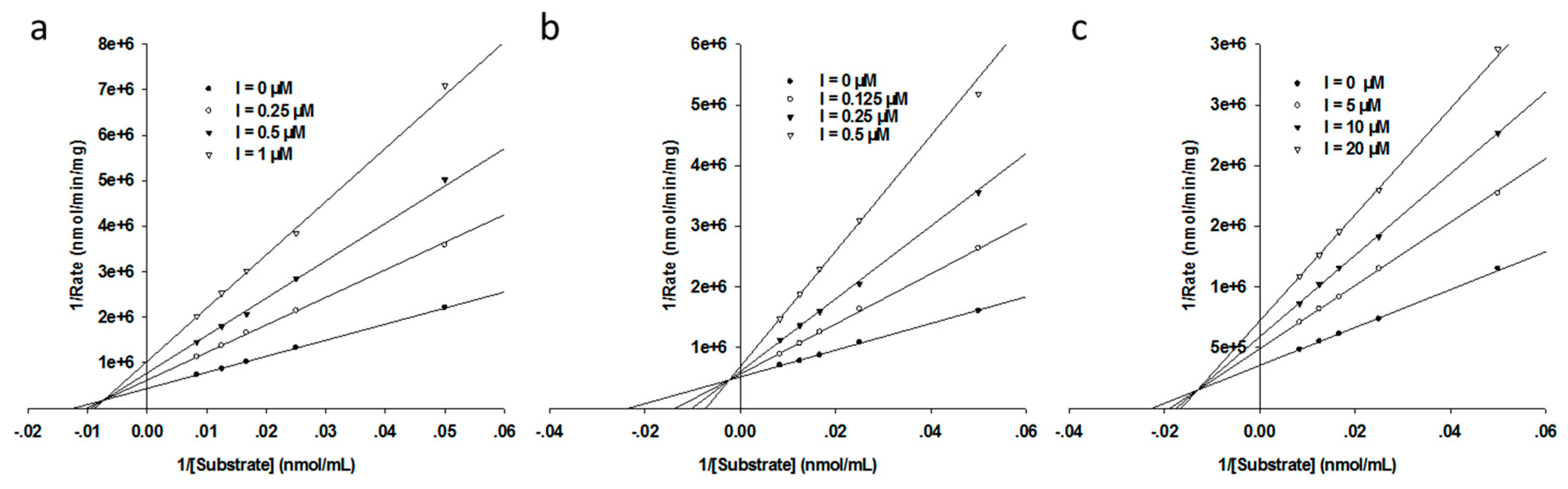
3.3. Inhibition Mode Assays
3.4. MIC Assays
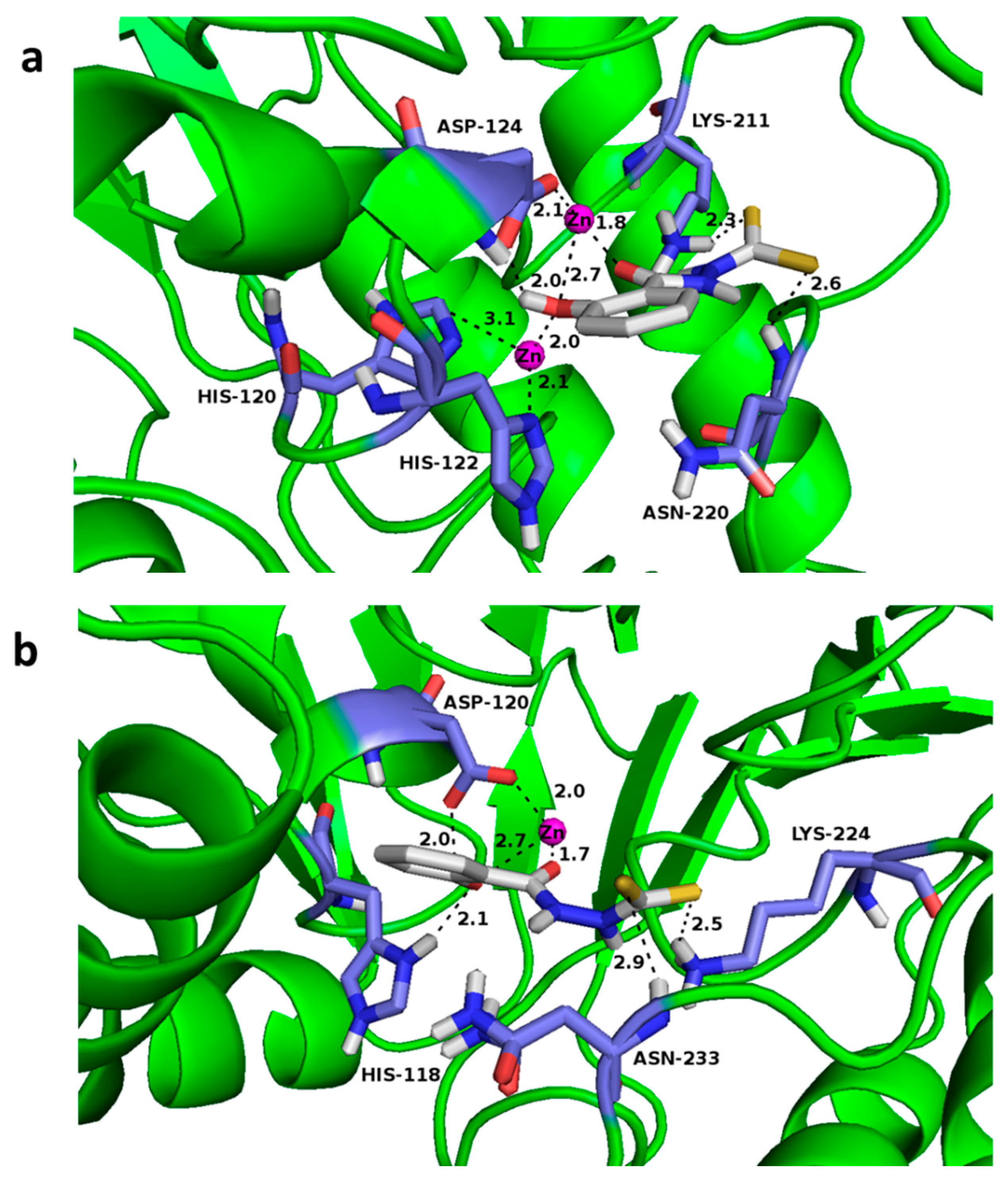
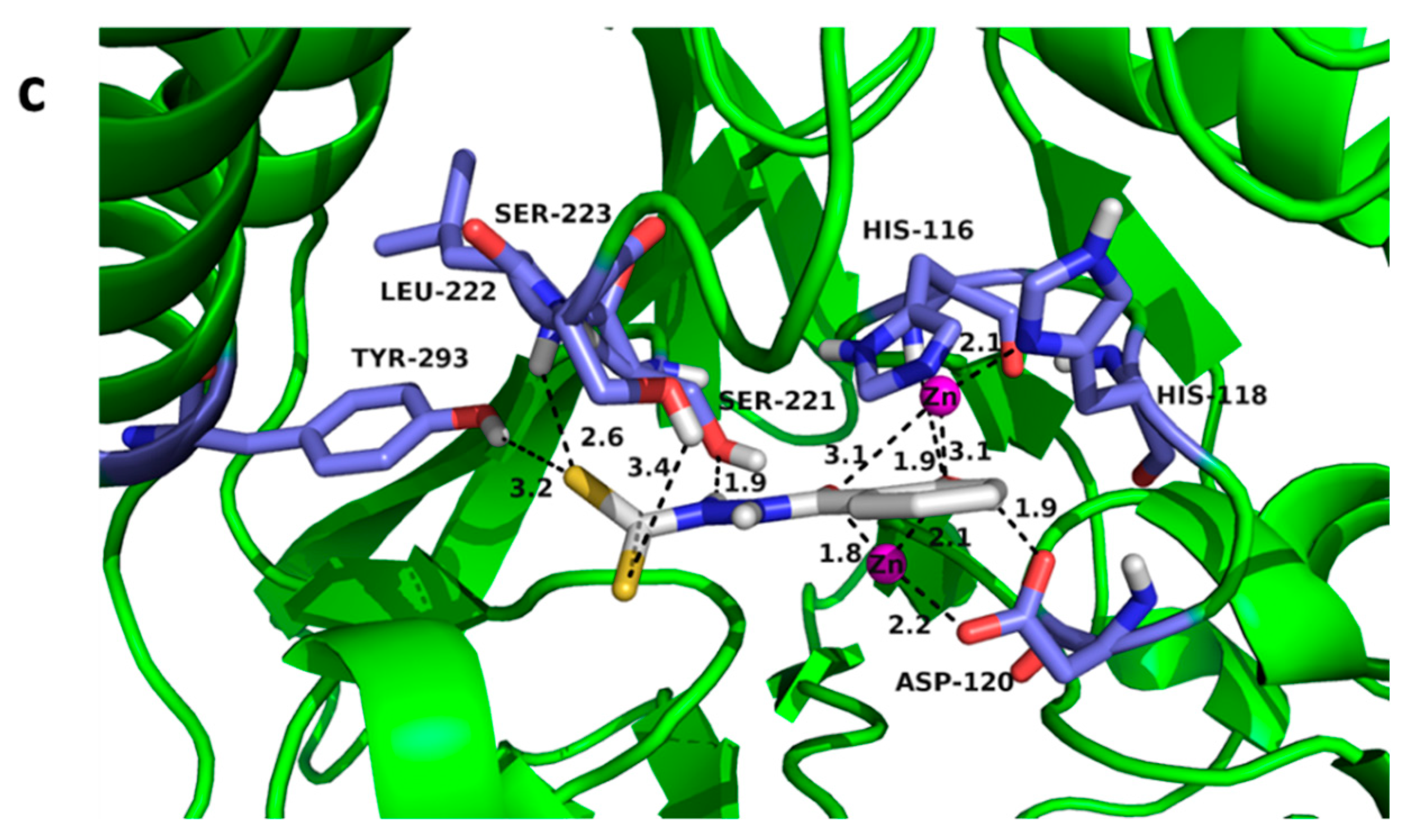
3.5. Docking Assays
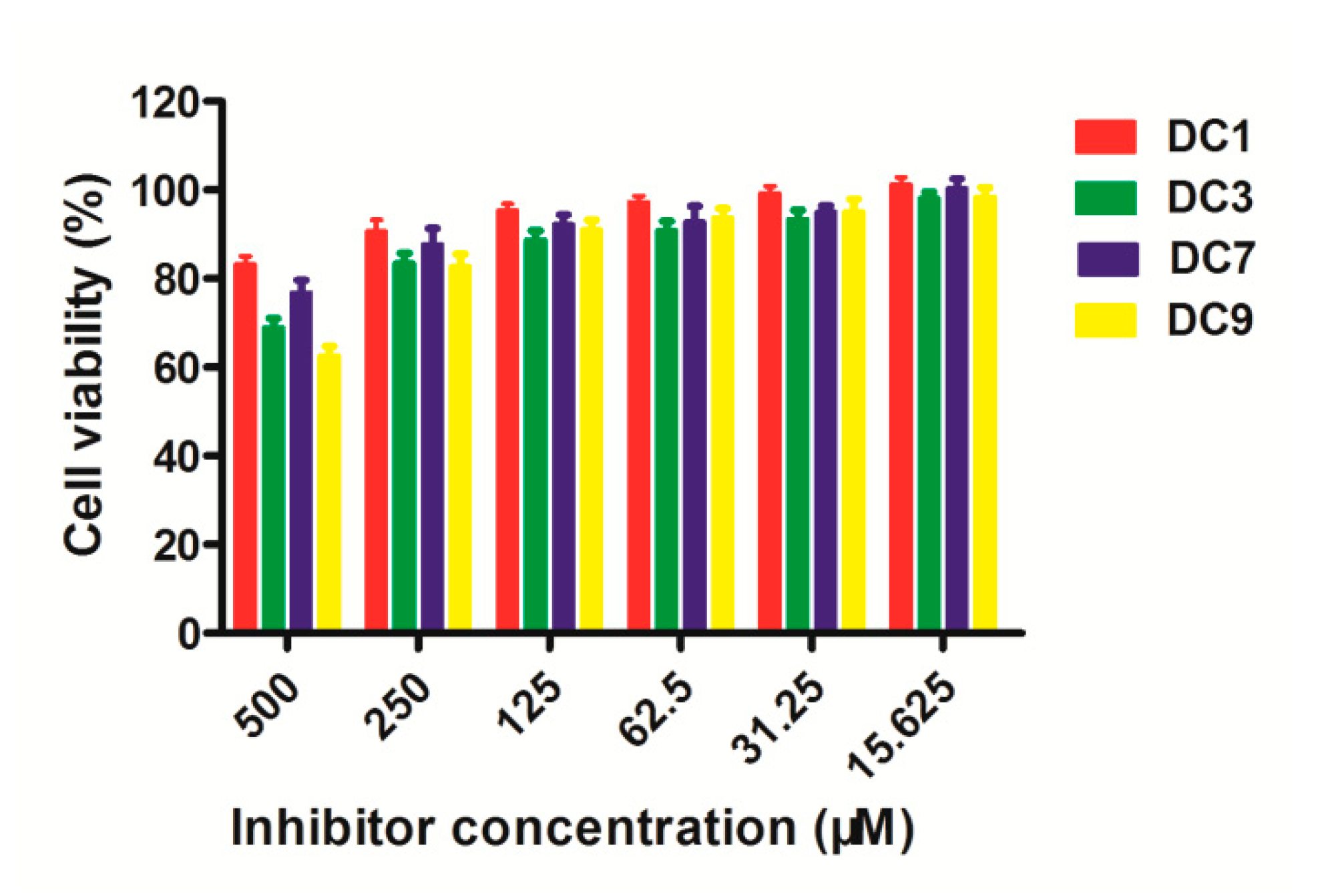
3.6. Cytotoxicity Assays
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Drawz, S.M.; Bonomo, R.A. Three decades of beta-lactamase inhibitors. Clin. Microbiol. Rev. 2010, 23, 160–201. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.F.; Meroueh, S.O.; Mobashery, S. Bacterial resistance to beta-lactam antibiotics: Compelling opportunism, compelling opportunity. Chem. Rev. 2005, 105, 395–424. [Google Scholar] [CrossRef] [PubMed]
- Bush, K. Past and Present Perspectives on β-Lactamases. Antimicrob. Agents Chemother. 2018, 62, e01076-18. [Google Scholar] [CrossRef] [PubMed]
- Ambler, R.P. The Structure of β-Lactamases. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 1980, 289, 321–331. [Google Scholar] [CrossRef]
- Crowder, M.W.; Spencer, J.; Vila, A.J. Metallo-beta-lactamases: Novel weaponry for antibiotic resistance in bacteria. Acc. Chem. Res. 2006, 39, 721–728. [Google Scholar] [CrossRef]
- Bebrone, C. Metallo-beta-lactamases (classification, activity, genetic organization, structure, zinc coordination) and their superfamily. Biochem. Pharmacol. 2007, 74, 1686–1701. [Google Scholar] [CrossRef]
- Meini, M.R.; Llarrull, L.I.; Vila, A.J. Overcoming differences: The catalytic mechanism of metallo-β-lactamases. FEBS Lett. 2015, 589, 3419–3432. [Google Scholar] [CrossRef]
- Linciano, P.; Cendron, L.; Gianquinto, E.; Spyrakis, F.; Tondi, D. Ten Years with New Delhi Metallo-β-lactamase-1 (NDM-1): From Structural Insights to Inhibitor Design. ACS Infect. Dis. 2019, 5, 9–34. [Google Scholar] [CrossRef]
- Mohamed, M.S.; Hussein, W.M.; McGeary, R.P.; Vella, P.; Schenk, G.; Abd El-Hameed, R.H. Synthesis and kinetic testing of new inhibitors for a metallo-beta-lactamase from Klebsiella pneumonia and Pseudomonas aeruginosa. Eur. J. Med. Chem. 2011, 46, 6075–6082. [Google Scholar] [CrossRef]
- Toney, J.H.; Hammond, G.G.; Fitzgerald, P.M.D.; Sharma, N.; Balkovec, J.M.; Rouen, G.P.; Olson, S.H.; Hammond, M.L.; Greenlee, M.L.; Gao, Y.D. Succinic Acids as Potent Inhibitors of Plasmid-borne IMP-1 Metallo-β-lactamase. J. Biol. Chem. 2001, 276, 31913–31918. [Google Scholar] [CrossRef]
- Hiraiwa, Y.; Saito, J.; Watanabe, T.; Yamada, M.; Morinaka, A.; Fukushima, T.; Kudo, T. X-ray crystallographic analysis of IMP-1 metallo-β-lactamase complexed with a 3-aminophthalic acid derivative, structure-based drug design, and synthesis of 3,6-disubstituted phthalic acid derivative inhibitors. Bioorg. Med. Chem. Lett. 2014, 24, 4891–4894. [Google Scholar] [CrossRef] [PubMed]
- Simm, A.M.; Loveridge, E.J.; Crosby, J.; Avison, M.B.; Walsh, T.R.; Bennett, P.M. Bulgecin A: A novel inhibitor of binuclear metallo-beta-lactamases. Biochem. J. 2005, 387, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Toney, J.H.; Fitzgerald, P.M.; Grover-Sharma, N.; Olson, S.H.; May, W.J.; Sundelof, J.G.; Vanderwall, D.E.; Cleary, K.A.; Grant, S.K.; Wu, J.K. Antibiotic sensitization using biphenyl tetrazoles as potent inhibitors of Bacteroides fragilis metallo-beta-lactamase. Chem. Biol. 1998, 5, 185–196. [Google Scholar] [CrossRef]
- King, A.M.; Reid-Yu, S.A.; Wang, W.; King, D.T.; Pascale, G.D.; Strynadka, N.C.; Walsh, T.R.; Coombes, B.K.; Wright, G.D. AMA overcomes antibiotic resistance by NDM and VIM metallo-Î2-lactamases. Nature 2014, 510, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.W.; Valladares, M.H.; Adlington, R.M.; Amicosante, G.; Baldwin, J.E.; Frère, J.M.; Galleni, M.; Rossolini, G.M.; Schofield, C.J. Hydroxamate Inhibitors of Aeromonas hydrophila AE036 Metallo-B-lactamase. Bioorg. Chem. 1999, 27, 35–40. [Google Scholar] [CrossRef]
- Chiou, J.; Wan, S.; Chan, K.F.; So, P.K.; He, D.; Chan, E.W.; Chan, T.H.; Wong, K.Y.; Tao, J.; Chen, S. Ebselen as a potent covalent inhibitor of New Delhi metallo-β-lactamase (NDM-1). Chem. Commun. 2015, 51, 9543–9546. [Google Scholar] [CrossRef]
- Carta, F.; Aggarwal, M.; Maresca, A.; Scozzafava, A.; McKenna, R.; Masini, E.; Supuran, C.T. Dithiocarbamates Strongly Inhibit Carbonic Anhydrases and Show Antiglaucoma Action in Vivo. J. Med. Chem. 2012, 55, 1721–1730. [Google Scholar] [CrossRef]
- Supuran, C.T. Carbonic anhydrases: Novel therapeutic applications for inhibitors and activators. Nat. Rev. Drug Discov. 2008, 7, 168–181. [Google Scholar] [CrossRef]
- Lee, S.J.; Caboni, P.; Tomizawa, M.; Casida, J.E. Cartap hydrolysis relative to its action at the insect nicotinic channel. J. Agric. Food Chem. 2004, 52, 95–98. [Google Scholar] [CrossRef]
- Hou, X.; Ge, Z.; Wang, T.; Guo, W.; Cui, J.; Cheng, T.; Lai, C.; Li, R.; Hou, X.; Ge, Z. Dithiocarbamic acid esters as anticancer agent. Part 1: 4-Substituted-piperazine-1-carbodithioic acid 3-cyano-3,3-diphenyl-propyl esters. Bioorg. Med. Chem. Lett. 2006, 16, 4214–4219. [Google Scholar] [CrossRef]
- Scozzafava, A.; Supuran, C.T. Carbonic anhydrase inhibitors: Synthesis of N-morpholylthiocarbonylsulfenylamino aromatic/heterocyclic sulfonamides and their interaction with isozymes I, II and IV. Bioorg. Med. Chem. Lett. 2000, 10, 1117–1120. [Google Scholar] [CrossRef]
- Horita, Y.; Takii, T.; Chiba, T.; Kuroishi, R.; Maeda, Y.; Kurono, Y.; Inagaki, E.; Nishimura, K.; Yamamoto, Y.; Abe, C. Synthesis of new sugar derivatives and evaluation of their antibacterial activities against Mycobacterium tuberculosis. Bioorg. Med. Chem. Lett. 2009, 19, 6313–6316. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, N.; Imamura, H.; Jona, H.; Kiyonaga, H.; Shimizu, A.; Moriya, M.; Sato, H.; Nakano, M.; Ushijima, R.; Nakagawa, S. Novel dithiocarbamate carbapenems with anti-MRSA activity. Bioorg. Med. Chem. 1998, 9, 1571–1578. [Google Scholar] [CrossRef]
- Len, C.; Boulognemerlot, A.; Postel, D.; Gino Ronco, A.; Villa, P.; Goubert, C.; Jeufrault, E.; Bernard Mathon, A.; Simon, H. Synthesis and Antifungal Activity of Novel Bis(dithiocarbamate) Derivatives of Glycerol. J. Agric. Food Chem. 1996, 44, 2856–2858. [Google Scholar] [CrossRef]
- Rafin, C.; Veignie, E.; Sancholle, M.; Postel, D.; Len, C.; Villa, P.; Ronco, G. Synthesis and antifungal activity of novel bisdithiocarbamate derivatives of carbohydrates against Fusarium oxysporum f. sp. lini. J. Agric. Food Chem. 2000, 48, 5283–5287. [Google Scholar] [CrossRef]
- Wang, M.M.; Chu, W.C.; Yang, Y.; Yang, Q.Q.; Qin, S.S.; Zhang, E. Dithiocarbamates: Efficient metallo-beta-lactamase inhibitors with good antibacterial activity when combined with meropenem. Bioorg. Med. Chem. Lett. 2018, 28, 3436–3440. [Google Scholar] [CrossRef]
- Zhai, L.; Zhang, Y.L.; Kang, J.S.; Oelschlaeger, P.; Xiao, L.; Nie, S.S.; Yang, K.W. Triazolylthioacetamide: A Valid Scaffold for the Development of New Delhi Metallo-beta-Lactmase-1 (NDM-1) Inhibitors. ACS Med. Chem. Lett. 2016, 7, 413–417. [Google Scholar] [CrossRef]
- King, D.T.; Worrall, L.J.; Gruninger, R.; Strynadka, N.C.J. New Delhi Metallo-β-Lactamase: Structural Insights into β-Lactam Recognition and Inhibition. J. Am. Chem. Soc. 2012, 134, 11362–11365. [Google Scholar] [CrossRef]
- Liénard, B.M.; Garau, G.; Horsfall, L.; Karsisiotis, A.I.; Damblon, C.; Lassaux, P.; Papamicael, C.; Roberts, G.C.; Galleni, M.; Dideberg, O. Structural basis for the broad-spectrum inhibition of metallo-beta-lactamases by thiols. Org. Biomol. Chem. 2008, 6, 2282–2294. [Google Scholar] [CrossRef]
- Spencer, J.; Read, J.; Sessions, R.B.; Howell, S.; Blackburn, G.M.; Gamblin, S.J. Antibiotic Recognition by Binuclear Metallo-β-Lactamases Revealed by X-ray Crystallography. J. Am. Chem. Soc. 2005, 127, 14439–14444. [Google Scholar] [CrossRef]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated Docking with Selective Receptor Flexibility. J. Comput. Chem. 2010, 30, 2785–2791. [Google Scholar] [CrossRef] [PubMed]
- Stote, R.H.; Karplus, M. Zinc binding in proteins and solution: A simple but accurate nonbonded representation. Proteins-Struct. Funct. Bioinform. 2010, 23, 12–31. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Chen, C.; Wang, W.M.; Xu, L.W.; Yang, K.W.; Oelschlaeger, P.; He, Y. Rhodanine as a Potent Scaffold for the Development of Broad-Spectrum Metallo-beta-lactamase Inhibitors. ACS Med. Chem. Lett. 2018, 9, 359–364. [Google Scholar] [CrossRef]
- Yang, H.; Aitha, M.; Hetrick, A.M.; Richmond, T.K.; Tierney, D.L.; Crowder, M.W. Mechanistic and spectroscopic studies of metallo-β-lactamase NDM-1. Biochemistry 2012, 51, 3839–3847. [Google Scholar] [CrossRef] [PubMed]
- Aitha, M.; Marts, A.R.; Bergstrom, A.; Møller, A.J.; Moritz, L.; Turner, L.; Nix, J.C.; Bonomo, R.A.; Page, R.C.; Tierney, D.L. Biochemical, mechanistic, and spectroscopic characterization of metallo-β-lactamase VIM-2. Biochemistry 2014, 53, 7321–7331. [Google Scholar] [CrossRef] [PubMed]
- Crawford, P.A.; Sharma, N.; Chandrasekar, S.; Sigdel, T.; Walsh, T.R.; Spencer, J.; Crowder, M.W. Over-expression, purification, and characterization of metallo-beta-lactamase ImiS from Aeromonas veronii bv. sobria. Protein Expr. Purif. 2004, 36, 272–279. [Google Scholar] [CrossRef]
- Crowder, M.; Walsh, T.L.; Pettit, M.; Spencer, J. Overexpression, purification, and characterization of the cloned metallo-beta-lactamase L1 from Stenotrophomonas maltophilia. Antimicrob. Agents Chemother. 1998, 42, 921–926. [Google Scholar] [CrossRef]
- Cadag, E.; Vitalis, E.; Lennox, K.P.; Zhou, C.L.E.; Zemla, A.T. Computational analysis of pathogen-borne metallo β-lactamases reveals discriminating structural features between B1 types. BMC Res. Notes 2012, 5, 96. [Google Scholar] [CrossRef]
- Yuan, Q.; He, L.; Ke, H. A Potential Substrate Binding Conformation of β-Lactams and Insight into the Broad Spectrum of NDM-1 Activity. Antimicrob. Agents Chemother. 2012, 56, 5157–5163. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Wang, W.M.; Oelschlaeger, P.; Chen, C.; Lei, J.E.; Lv, M.; Yang, K.W. Real-Time Monitoring of NDM-1 Activity in Live Bacterial Cells by Isothermal Titration Calorimetry: A New Approach To Measure Inhibition of Antibiotic-Resistant Bacteria. ACS Infect. Dis. 2018, 4, 1671–1678. [Google Scholar] [CrossRef]
- Celenza, G.; Vicario, M.; Bellio, P.; Linciano, P.; Perilli, M.; Oliver, A.; Blazquez, J.; Cendron, L.; Tondi, D. Phenylboronic Acid Derivatives as Validated Leads Active in Clinical Strains Overexpressing KPC-2: A Step against Bacterial Resistance. ChemMedChem 2018, 13, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Wayne, P.A. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically: Approved Standard, 9th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012; M07-A9. [Google Scholar]
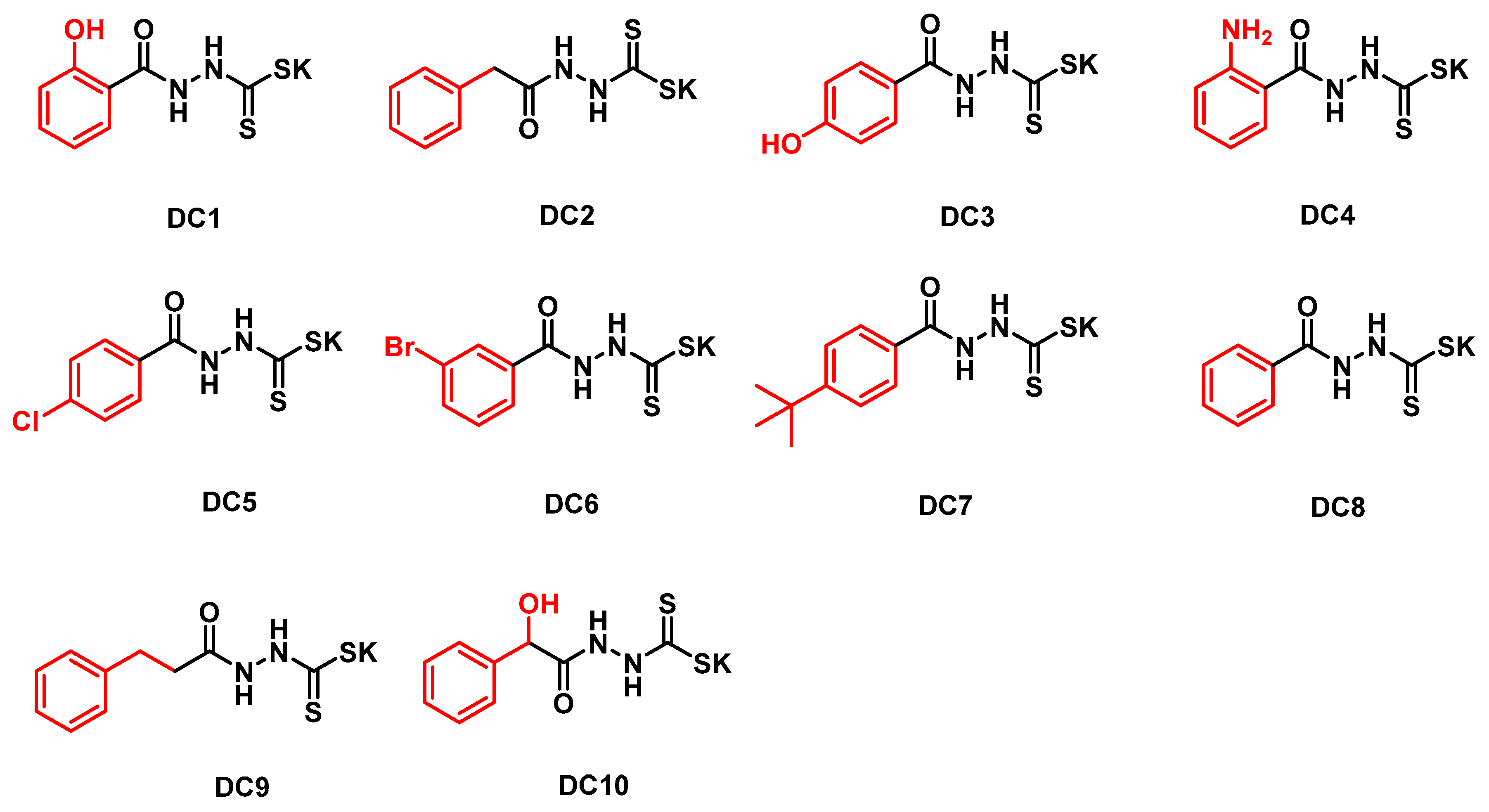
| Compd. | B1 | B2 | B3 | ||
|---|---|---|---|---|---|
| NDM-1 a | VIM-2 a | IMP-1 a | ImiS b | L1 a | |
| DC1 | 0.38 ± 0.01 | 9.31 ± 0.20 | 3.16 ± 0.10 | 0.21 ± 0.01 | 7.17 ± 0.10 |
| DC2 | 5.64 ± 0.10 | >200.00 | 166.22 ± 2.70 | 2.50 ± 0.10 | 86.81 ± 1.6 |
| DC3 | 67.12 ± 0.90 | 82.76 ± 1.10 | >200.00 | >200.00 | >200.00 |
| DC4 | 11.39 ± 0.40 | 139.07 ± 2.05 | 102.78 ± 1.45 | 1.08 ± 0.05 | 176.54 ± 2.80 |
| DC5 | 91.31 ± 1.70 | >200.00 | >200.00 | 128.82 ± 2.10 | >200.00 |
| DC6 | 15.25 ± 0.90 | >200.00 | >200.00 | 2.56 ± 0.05 | 154.37 ± 3.20 |
| DC7 | 0.52± 0.01 | 63.46 ± 1.30 | 96.38 ± 1.20 | >200.00 | 190.79 ± 4.00 |
| DC8 | 0.53 ± 0.01 | 3.07 ± 0.10 | 0.85 ± 0.01 | 0.67 ± 0.01 | 25.81 ± 0.12 |
| DC9 | 62.66 ± 1.05 | 115.20 ± 3.10 | 86.16 ± 0.90 | 156.23 ± 2.20 | >200.00 |
| DC10 | 1.52 ± 0.05 | 5.25 ± 0.20 | 2.16 ± 0.05 | 0.82 ± 0.01 | 11.68 ± 0.6 |
| A a | |||||
| Compd.\conc. | 8 | 16 | 32 | 64 | 128 |
| DC1 | 8 | 8 | 4 | 4 | 1 |
| DC2 | 16 | 8 | 8 | 8 | 4 |
| DC3 | 16 | 16 | 16 | 16 | 16 |
| DC4 | 8 | 8 | 8 | 8 | 4 |
| DC5 | 16 | 16 | 16 | 16 | 16 |
| DC6 | 16 | 16 | 16 | 16 | 16 |
| DC7 | 16 | 16 | 16 | 16 | 16 |
| DC8 | 16 | 8 | 8 | 8 | 8 |
| DC9 | 16 | 16 | 16 | 16 | 16 |
| DC10 | 16 | 8 | 8 | 8 | 4 |
| B a | |||||
| Compd.\conc. | 8 | 16 | 32 | 64 | 128 |
| DC1 | 32 | 16 | 16 | 8 | 8 |
| DC2 | 64 | 64 | 64 | 32 | 32 |
| DC3 | 128 | 128 | 128 | 128 | 128 |
| DC4 | 64 | 64 | 32 | 32 | 16 |
| DC5 | 128 | 128 | 128 | 128 | 128 |
| DC6 | 128 | 128 | 128 | 128 | 128 |
| DC7 | 128 | 32 | 16 | 8 | 8 |
| DC8 | 64 | 64 | 32 | 32 | 16 |
| DC9 | 128 | 128 | 128 | 128 | 64 |
| DC10 | 128 | 128 | 128 | 32 | 16 |
| C a | |||||
| Compd.\conc. | 16 | 32 | 64 | 128 | 256 |
| DC1 | 128 | 128 | 64 | 32 | 8 |
| DC2 | 128 | 128 | 128 | 128 | 64 |
| DC3 | 128 | 128 | 128 | 128 | 128 |
| DC4 | 128 | 128 | 128 | 128 | 32 |
| DC5 | 128 | 128 | 128 | 128 | 128 |
| DC6 | 128 | 128 | 128 | 128 | 128 |
| DC7 | 128 | 128 | 128 | 128 | 128 |
| DC8 | 128 | 128 | 128 | 64 | 32 |
| DC9 | 128 | 128 | 128 | 128 | 128 |
| DC10 | 128 | 128 | 128 | 128 | 64 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ge, Y.; Xu, L.-W.; Liu, Y.; Sun, L.-Y.; Gao, H.; Li, J.-Q.; Yang, K. Dithiocarbamate as a Valuable Scaffold for the Inhibition of Metallo-β-Lactmases. Biomolecules 2019, 9, 699. https://doi.org/10.3390/biom9110699
Ge Y, Xu L-W, Liu Y, Sun L-Y, Gao H, Li J-Q, Yang K. Dithiocarbamate as a Valuable Scaffold for the Inhibition of Metallo-β-Lactmases. Biomolecules. 2019; 9(11):699. https://doi.org/10.3390/biom9110699
Chicago/Turabian StyleGe, Ying, Li-Wei Xu, Ya Liu, Le-Yun Sun, Han Gao, Jia-Qi Li, and Kewu Yang. 2019. "Dithiocarbamate as a Valuable Scaffold for the Inhibition of Metallo-β-Lactmases" Biomolecules 9, no. 11: 699. https://doi.org/10.3390/biom9110699
APA StyleGe, Y., Xu, L.-W., Liu, Y., Sun, L.-Y., Gao, H., Li, J.-Q., & Yang, K. (2019). Dithiocarbamate as a Valuable Scaffold for the Inhibition of Metallo-β-Lactmases. Biomolecules, 9(11), 699. https://doi.org/10.3390/biom9110699





