Unravelling the Skin Secretion Peptides of the Gliding Leaf Frog, Agalychnis spurrelli (Hylidae)
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Skin Secretion Samples
2.2. Molecular Cloning
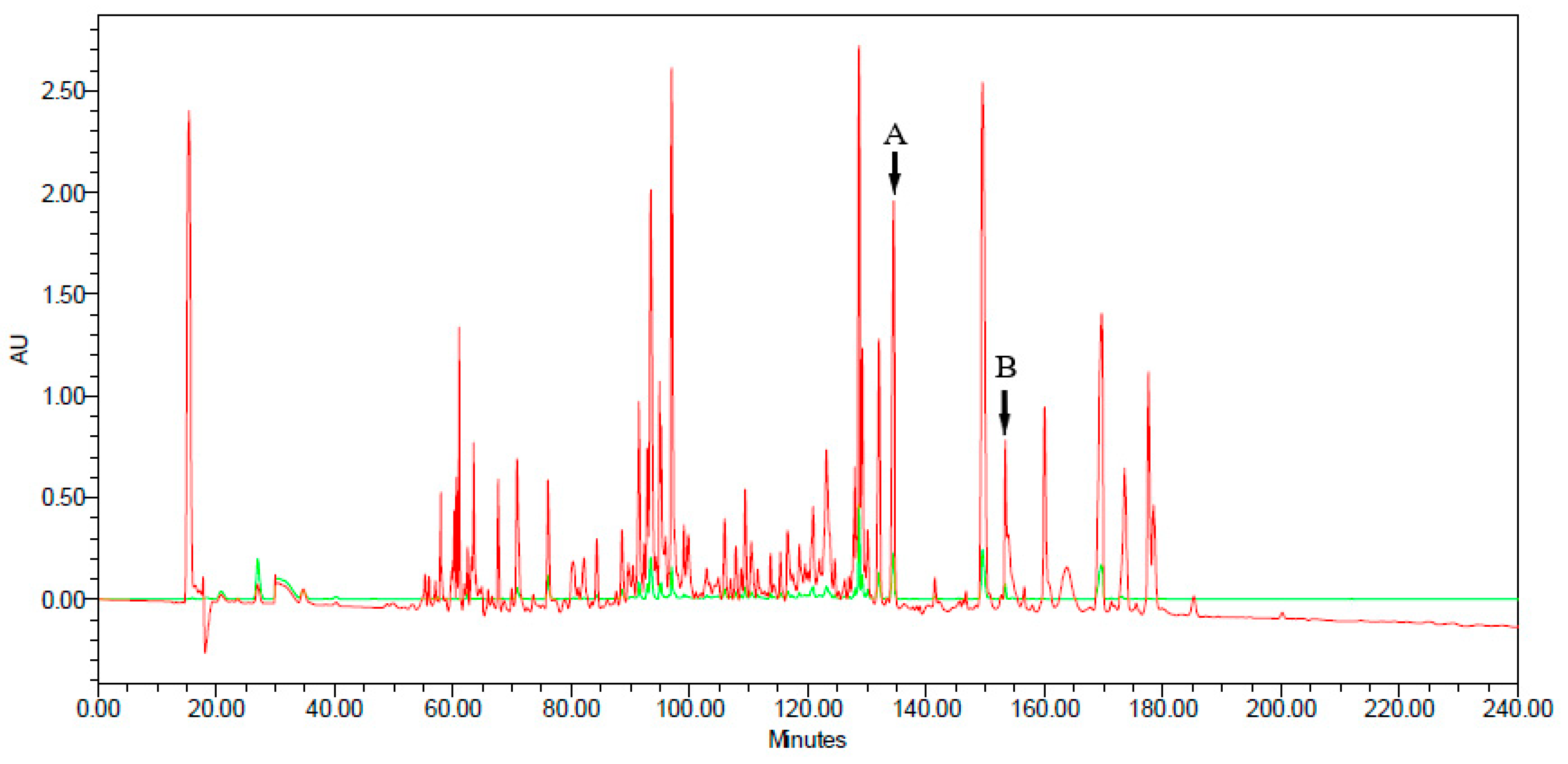
2.3. Reverse-Phase, High Performance Liquid Chromatography (HPLC) Fractionation
2.4. Tandem Mass Spectrometry
2.5. Bioinformatic Analysis
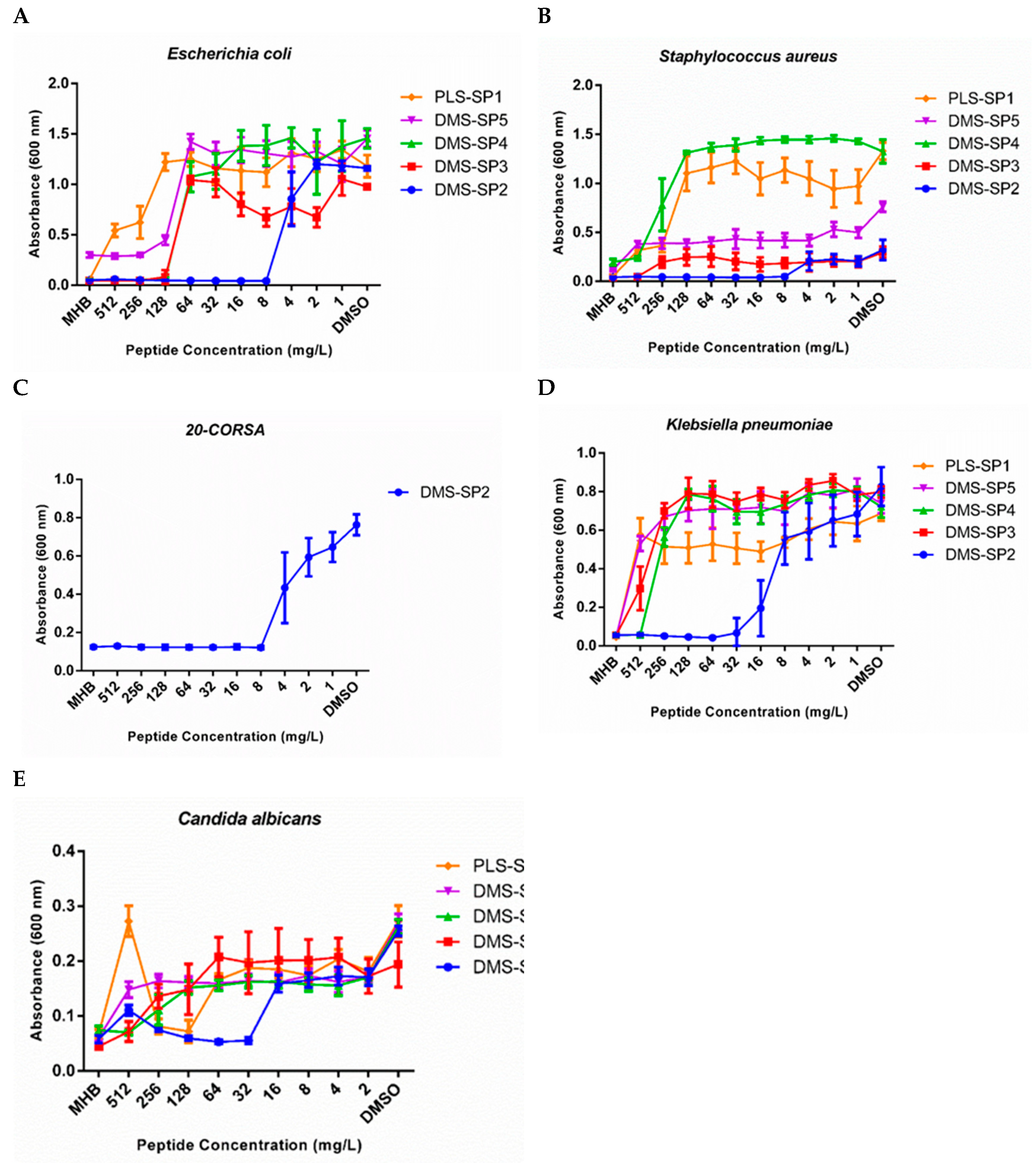
2.6. Antimicrobial Activities of the Novel Peptides Identified
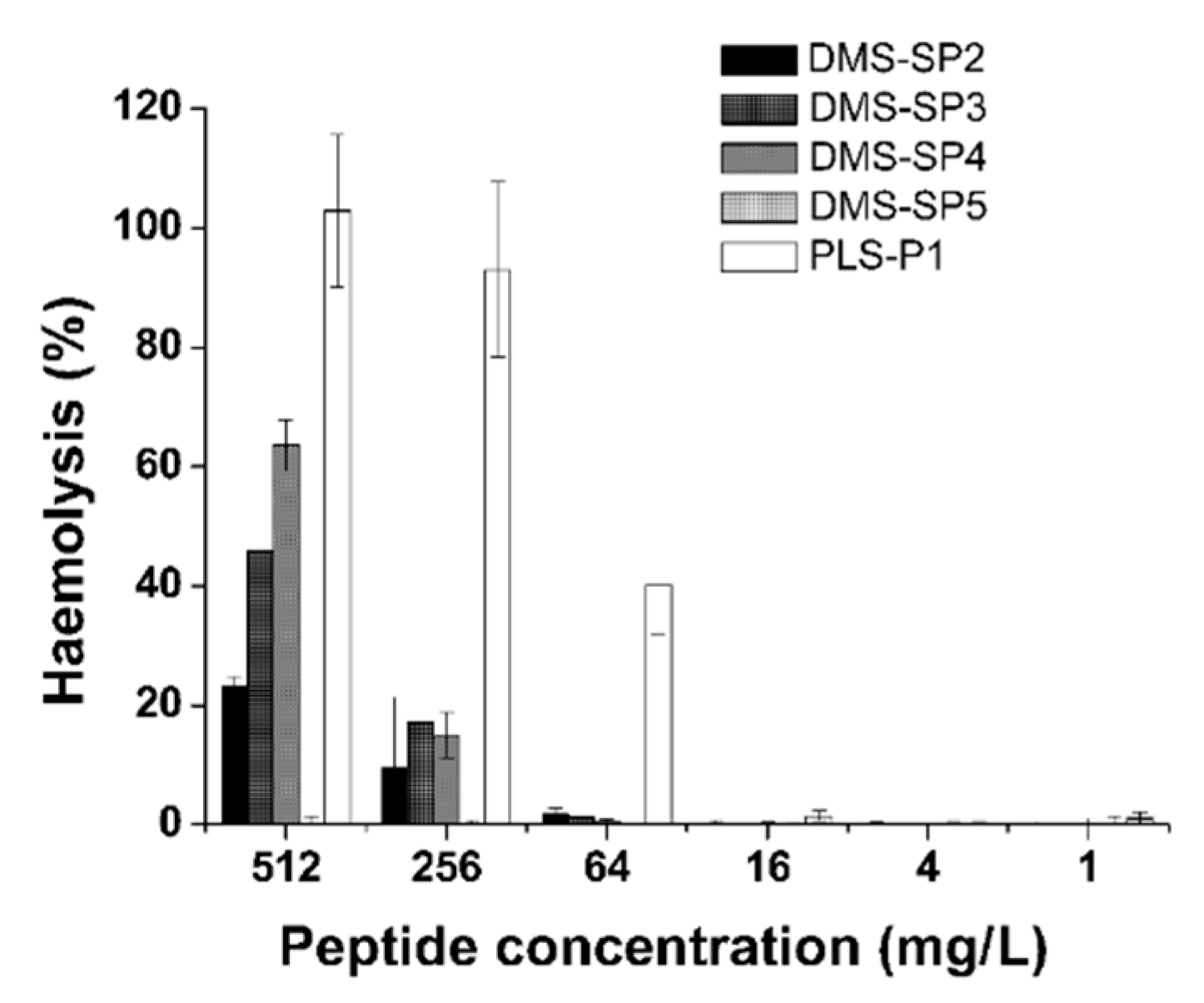
2.7. The Determination of Toxicity to Red Blood Cells
3. Results
3.1. Molecular Cloning of the Novel Peptide Precursors Encoding cDNA Sequences, and the HPLC Profile
3.1.1. Phyllokinins
3.1.2. Medusin-AS
3.1.3. Phylloseptin-AS1
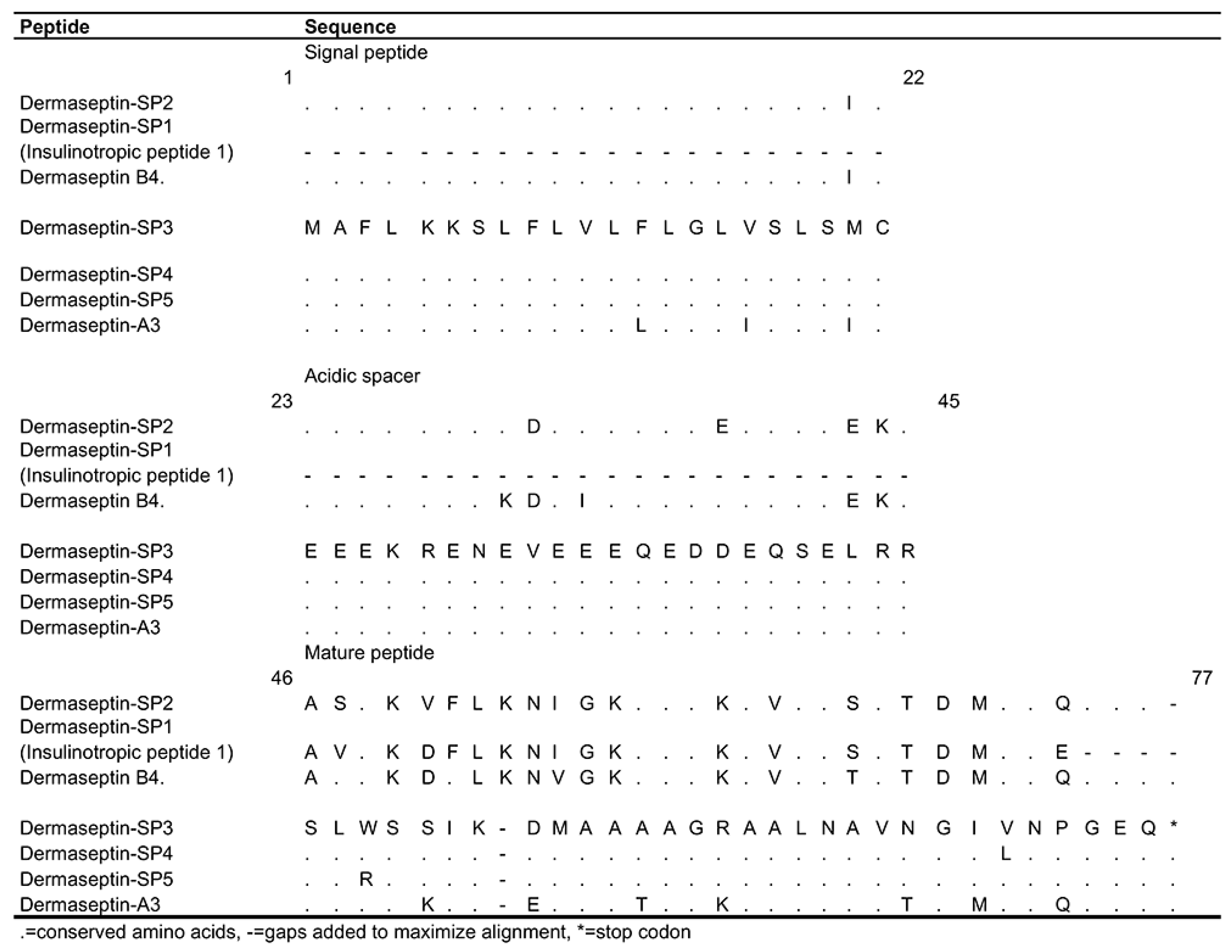
3.1.4. Dermaseptins
3.1.5. Orphan Peptides
3.2. Peptide Identification by Tandem Mass Spectrometry
3.3. The Antimicrobial and Haemolytic Activities of Synthetic Peptides Based on Amino Acid Sequences Identified and Toxicity to Red Blood Cells
3.4. Secondary Structures and Physicochemical Properties of Synthetic Peptides
4. Discussion
4.1. Phyllokinins
4.2. Medusin-AS
4.3. Phylloseptin-AS1
4.4. Dermaseptin-SP
4.5. Orphan Peptides
4.6. Tandem Mass Sequencing
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shankar, P.R.; Balasubramanium, R. Antimicrobial resistance: global report on surveillance 2014. Australas. Med J. 2014, 7, 237. [Google Scholar]
- Conlon, J.M.; Sonnevend, A. Clinical Applications of Amphibian Antimicrobial Peptides. J. Med. Sci. 2011, 4, 62–72. [Google Scholar] [CrossRef]
- Erspamer, V. Bioactive secretions of the amphibian integument. In Amphibian Biology: The Integument; Heatwole, H., Bartholameus, G., Eds.; Surrey, Beatty and Sons: Chipping-Norton, N.S.W. Australia, 1994; Volume 1, pp. 178–350. [Google Scholar]
- Azevedo Calderon, L.; Silva Ade, A.; Ciancaglini, P.; Stabeli, R.G. Antimicrobial Peptides from Phyllomedusa Frogs: From Biomolecular Diversity to Potential Nanotechnologic Medical Applications. Amino Acids 2011, 40, 29–49. [Google Scholar] [CrossRef]
- Xu, X.; Lai, R. The Chemistry and Biological Activities of Peptides from Amphibian Skin Secretions. Chem. Rev. 2015, 115, 1760–1846. [Google Scholar] [CrossRef]
- Nicolas, P.; Ladram, A. Dermaseptins. In Handbook of Biologically Active Peptides; Kastin, A.J., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2013; p. 350. [Google Scholar]
- Proaño-Bolaños, C.; Zhou, M.; Wang, L.; Coloma, L.A.; Chen, T.; Shaw, C. Peptidomic Approach Identifies Cruzioseptins, a New Family of Potent Antimicrobial Peptides in the Splendid Leaf Frog, Cruziohyla Calcarifer. J. Proteom. 2016, 146, 1–13. [Google Scholar] [CrossRef]
- Proaño-Bolaños, C.; Li, R.; Zhou, M.; Wang, L.; Xi, X.; Tapia, E.E.; Coloma, L.A.; Chen, T.; Shaw, C. Novel Kazal-Type Proteinase Inhibitors from the Skin Secretion of the Splendid Leaf Frog, Cruziohyla Calcarifer. EuPA Open Proteom. 2017, 15, 1–13. [Google Scholar] [CrossRef]
- Simmaco, M.; Mignogna, G.; Canofeni, S.; Miele, R.; Mangoni, M.L.; Barra, D. Temporins, Antimicrobial Peptides from the European Red Frog Rana Temporaria. Eur. J. Biochem. 1996, 242, 788–792. [Google Scholar] [CrossRef] [PubMed]
- van Zoggel, H.; Hamma-Kourbali, Y.; Galanth, C.; Ladram, A.; Nicolas, P.; Courty, J.; Amiche, M.; Delbe, J. Antitumor and Angiostatic Peptides from Frog Skin Secretions. Amino Acids 2012, 42, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Popovic, S.; Urban, E.; Lukic, M.; Conlon, J.M. Peptides with Antimicrobial and Anti-Inflammatory Activities that have Therapeutic Potential for Treatment of Acne Vulgaris. Peptides 2012, 34, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Conlon, J.M.; Mechkarska, M.; Abdel-Wahab, Y.H.; Flatt, P.R. Peptides from Frog Skin with Potential for Development into Agents for Type 2 Diabetes Therapy. Peptides 2018, 100, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Faivovich, J.; Haddad, C.; Baeta, D.; Jungfer, K.; Alvares, G.; Brandao, R.A.; Sheil, C.; Barrientos, L.; Barrio-Amoros, C.; Cruz, C.; et al. The Phyllogenetic Relationships of the Charismatic Poster Frogs, Phyllomedusinae (Anura, Hylidae). Cladistics 2010, 26, 227–261. [Google Scholar] [CrossRef]
- Wang, L.; Zhou, M.; McClelland, A.; Reilly, A.; Chen, T.; Gagliardo, R.; Walker, B.; Shaw, C. Novel Dermaseptin, Adenoregulin and Caerin Homologs from the Central American Red-Eyed Leaf Frog, Agalychnis Callidryas, Revealed by Functional Peptidomics of Defensive Skin Secretion. Biochimie 2008, 90, 1435–1441. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Andrade, M.H. Agalychnis Spurrelli Boulenger (Anura, Hylidae): Variación, Distribución Y Sinonimia. Pap. Avulsos Zool. 2008, 48, 103–117. [Google Scholar] [CrossRef]
- Vanhoye, D.; Bruston, F.; Nicolas, P.; Amiche, M. Antimicrobial Peptides from Hylid and Ranin Frogs Originated from a 150-Million-Year-Old Ancestral Precursor with a Conserved Signal Peptide but a Hypermutable Antimicrobial Domain. Eur. J. Biochem. 2003, 270, 2068–2081. [Google Scholar] [CrossRef]
- Wechselberger, C. Cloning of cDNAs Encoding New Peptides of the Dermaseptin-Family. Biochim. Biophys. Acta 1998, 1388, 279–283. [Google Scholar] [CrossRef]
- Marenah, L.; Shaw, C.; Orr, D.F.; McClean, S.; Flatt, P.R.; Abdel-Wahab, Y.H.A. Isolation and Characterisation of an Unexpected Class of Insulinotropic Peptides in the Skin of the Frog Agalychnis Litodryas. Regul. Pept. 2004, 120, 33–38. [Google Scholar] [CrossRef]
- Meneses, E.P.; Villa-Hernandez, O.; Hernandez-Orihuela, L.; Castro-Franco, R.; Pando, V.; Aguilar, M.B.; Batista, C.V. Peptidomic Analysis of the Skin Secretions of the Frog Pachymedusa Dacnicolor. Amino Acids 2011, 40, 113–122. [Google Scholar] [CrossRef]
- Conlon, J.M.; Woodhams, D.C.; Raza, H.; Coquet, L.; Leprince, J.; Jouenne, T.; Vaudry, H.; Rollins-Smith, L.A. Peptides with Differential Cytolytic Activity from Skin Secretions of the Lemur Leaf Frog Hylomantis Lemur (Hylidae: Phyllomedusinae). Toxicon 2007, 50, 498–506. [Google Scholar] [CrossRef]
- Abdel-Wahab, Y.H.; Power, G.J.; Flatt, P.R.; Woodhams, D.C.; Rollins-Smith, L.A.; Conlon, J.M. A Peptide of the Phylloseptin Family from the Skin of the Frog Hylomantis Lemur (Phyllomedusinae) with Potent in Vitro and in Vivo Insulin-Releasing Activity. Peptides 2008, 29, 2136–2143. [Google Scholar] [CrossRef]
- Xi, X.; Li, R.; Jiang, Y.; Lin, Y.; Wu, Y.; Zhou, M.; Xu, J.; Wang, L.; Chen, T.; Shaw, C. Medusins: A New Class of Antimicrobial Peptides from the Skin Secretions of Phyllomedusine Frogs. Biochimie 2013, 95, 1288–1296. [Google Scholar] [CrossRef]
- Chen, T.; Farragher, S.; Bjourson, A.J.; Orr, D.F.; Rao, P.; Shaw, C. Granular Gland Transcriptomes in Stimulated Amphibian Skin Secretions. Biochem. J. 2003, 371, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Bjourson, A.J.; Orr, D.F.; Kwok, H.; Rao, P.; Ivanyi, C.; Shaw, C. Unmasking Venom Gland Transcriptomes in Reptile Venoms. Anal. Biochem. 2002, 311, 152–156. [Google Scholar] [CrossRef]
- Proano-Bolanos, C.; Zhou, M.; Chen, T.; Shaw, C. Skin Secretion Transcriptome Remains in Chromatographic Fractions Suitable for Molecular Cloning. Anal. Biochem. 2019, 564–565, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Pinkse, M.; Evaristo, G.; Pieterse, M.; Yu, Y.; Verhaert, P. MS Approaches to Select Peptides with Post-Translational Modifications from Amphibian Defense Secretions Prior to Full Sequence Elucidation. EuPA Open Proteom. 2014, 5, 32–40. [Google Scholar] [CrossRef]
- Rates, B.; Silva, L.P.; Ireno, I.C.; Leite, F.S.; Borges, M.H.; Bloch, C., Jr.; De Lima, M.E.; Pimenta, A.M. Peptidomic Dissection of the Skin Secretion of Phasmahyla Jandaia (Bokermann and Sazima, 1978) (Anura, Hylidae, Phyllomedusinae). Toxicon 2011, 57, 35–52. [Google Scholar] [CrossRef]
- Thompson, A.H.; Bjourson, A.J.; Orr, D.F.; Shaw, C.; McClean, S. A Combined Mass Spectrometric and cDNA Sequencing Approach to the Isolation and Characterization of Novel Antimicrobial Peptides from the Skin Secretions of Phyllomedusa Hypochondrialis Azurea. Peptides 2007, 28, 1331–1343. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic Local Alignment Search Tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- ThermoFisher Scientific. Vector Advance NTI® 11.5. Available online: https://www.thermofisher.com/ec/en/home/life-science/cloning/vector-nti-software/vector-nti-advance-software.html (accessed on 18 September 2019).
- Petersen, T.N.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 4.0: Discriminating Signal Peptides from Transmembrane Regions. Nat. Methods 2011, 8, 785–786. [Google Scholar] [CrossRef]
- Rozenski, J. Peptide Mass Calculator V3.2. Available online: http://rna.rega.kuleuven.be/masspec/pepcalc.htm (accessed on 18 September 2019).
- Bachem. Peptide Calculator. Available online: https://www.bachem.com/service-support/peptide-calculator/ (accessed on 18 September 2019).
- Gautier, R.; Douguet, D.; Antonny, B.; Drin, G. HELIQUEST: A Web Server to Screen Sequences with Specific Alpha-Helical Properties. Bioinformatics 2008, 24, 2101–2102. [Google Scholar] [CrossRef]
- Geourjon, C.; Deleage, G. SOPMA: Significant Improvements in Protein Secondary Structure Prediction by Consensus Prediction from Multiple Alignments. Comput. Appl. Biosci. 1995, 11, 681–684. [Google Scholar] [CrossRef] [PubMed]
- Bordoli, L.; Kiefer, F.; Arnold, K.; Benkert, P.; Battey, J.; Schwede, T. Protein Structure Homology Modelling using SWISS-MODEL Workspace. Nat. Protoc. 2009, 4, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Amiche, M.; Ladram, A.; Nicolas, P. A Consistent Nomenclature of Antimicrobial Peptides Isolated from Frogs of the Subfamily Phyllomedusinae. Peptides 2008, 29, 2074–2082. [Google Scholar] [CrossRef] [PubMed]
- Charpentier, S.; Amiche, M.; Mester, J.; Vouille, V.; Le Caer, J.P.; Nicolas, P.; Delfour, A. Structure, Synthesis, and Molecular Cloning of Dermaseptins B, a Family of Skin Peptide Antibiotics. J. Biol. Chem. 1998, 273, 14690–14697. [Google Scholar] [CrossRef]
- Raja, Z.; Andre, S.; Piesse, C.; Sereno, D.; Nicolas, P.; Foulon, T.; Oury, B.; Ladram, A. Structure, Antimicrobial Activities and Mode of Interaction with Membranes of Novel [Corrected] Phylloseptins from the Painted-Belly Leaf Frog, Phyllomedusa Sauvagii. PloS ONE 2013, 8, e70782. [Google Scholar] [CrossRef]
- Cuesta, S.; Gallegos, F.; Arias, J.; Pilaquinga, F.; Blasco-Zuniga, A.; Proano-Bolanos, C.; Rivera, M.; Meneses, L. Molecular Modeling of Four Dermaseptin-Related Peptides of the Gliding Tree Frog Agalychnis Spurrelli. J. Mol. Model 2019, 25, 260. [Google Scholar] [CrossRef]
- Reimer, L.G.; Stratton, C.W.; Reller, B. Minimum Inhibitory and Bactericidal Concentrations of 44 Antimicrobial Agents Against Three Standard Control Strains in Broth with and without Human Serum. Antimicrob. Agents Chemother. 1981, 19, 1050–1055. [Google Scholar] [CrossRef]
- Conlon, J.M.; Jouenne, T.; Cosette, P.; Cosquer, D.; Vaudry, H.; Taylor, C.K.; Abel, P.W. Bradykinin-Related Peptides and Tryptophyllins in the Skin Secretions of the most Primitive Extant Frog, Ascaphus Truei. Gen. Comp. Endocrinol. 2005, 143, 193–199. [Google Scholar] [CrossRef]
- Li, L.; Vaali, K.; Paakkari, I.; Vapaatalo, H. Involvement of Bradykinin B1 and B2 Receptors in Relaxation of Mouse Isolated Trachea. Br. J. Pharmacol. 1998, 123, 1337–1342. [Google Scholar] [CrossRef][Green Version]
- Galli, G.L.; Skovgaard, N.; Abe, A.S.; Taylor, E.W.; Conlon, J.M.; Wang, T. Cardiovascular Actions of Rattlesnake Bradykinin ([Val1,Thr6]Bradykinin) in the Anesthetized South American Rattlesnake Crotalus Durissus Terrificus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R456–465. [Google Scholar] [CrossRef][Green Version]
- Wang, T.; Axelsson, M.; Jensen, J.; Conlon, J.M. Cardiovascular Actions of Python Bradykinin and Substance P in the Anesthetized Python, Python Regius. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 279, R531–R538. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.W. Molecular Mechanism of Antimicrobial Peptides: The Origin of Cooperativity. Biochim. Biophys. Acta 2006, 1758, 1292–1302. [Google Scholar] [CrossRef] [PubMed]
- Giangaspero, A.; Sandri, L.; Tossi, A. Amphipathic Alpha Helical Antimicrobial Peptides. A Systematic Study of the Effects of Structural and Physical Properties on Biological Activity. Eur. J. Biochem. 2001, 268, 5589–5600. [Google Scholar] [CrossRef] [PubMed]
- Epand, R.M.; Vogel, H.J. Diversity of Antimicrobial Peptides and their Mechanisms of Action. Biochim. Biophys. Acta 1999, 1462, 11–28. [Google Scholar] [CrossRef]
- Epand, R.M.; Walker, C.; Epand, R.F.; Magarvey, N.A. Molecular Mechanisms of Membrane Targeting Antibiotics. Biochim. Biophys. Acta 2016, 1858, 980–987. [Google Scholar] [CrossRef]
- Hollmann, A.; Martinez, M.; Maturana, P.; Semorile, L.C.; Maffia, P.C. Antimicrobial Peptides: Interaction with Model and Biological Membranes and Synergism with Chemical Antibiotics. Front. Chem. 2018, 6, 204. [Google Scholar] [CrossRef]
- Almeida, J.R.; Mendes, B.; Lancellotti, M.; Marangoni, S.; Vale, N.; Passos, O.; Ramos, M.J.; Fernandes, P.A.; Gomes, P.; Da Silva, S.L. A Novel Synthetic Peptide Inspired on Lys49 Phospholipase A2 from Crotalus Oreganus Abyssus Snake Venom Active Against Multidrug-Resistant Clinical Isolates. Eur. J. Med. Chem. 2018, 149, 248–256. [Google Scholar] [CrossRef]
- van der Weerden, N.L.; Bleackley, M.R.; Anderson, M.A. Properties and Mechanisms of Action of Naturally Occurring Antifungal Peptides. Cell Mol. Life Sci. 2013, 70, 3545–3570. [Google Scholar] [CrossRef]
- Bondaryk, M.; Staniszewska, M.; Zielinska, P.; Urbanczyk-Lipkowska, Z. Natural Antimicrobial Peptides as Inspiration for Design of a New Generation Antifungal Compounds. J. Fungi 2017, 3, 46. [Google Scholar] [CrossRef]



| Peptide | Precursor Sequence | Identify by | Coverage % | # Peptides Fragments | # AAs | LCQ MW [Da] | Theoretical MW [Da] | Score |
|---|---|---|---|---|---|---|---|---|
| [Ser6, Val10, Asp11)-phyllokinin | RPPGFSPFRVD | mc,ms2 | 100 | 19 | 11 | 1273.66 | 1274.44 | 13.80 |
| [Thr6, Val10, Asp11)-phyllokinin | RPPGFTPFRVD | mc,ms2 | 100 | 15 | 11 | 1287.67 | 1288.47 | 8.08 |
| Medusin-AS | LLGMIPLAISAISALSKL-NH2 | mc,ms2 | 100 | 50 | 19 | 1810.10 | 1809.12 | 77.05 |
| Phylloseptin-SP1 | FLSLIPHVISAIPHVVNALSNL-NH2 | mc,ms2 | 100 | 60 | 23 | 2353.35 | 2352.37 | 59.59 |
| Dermaseptin-SP2 | ASWKVFLKNIGKAAGKAVLNSVTDMVNQ-NH2 | mc,ms2 | 100 | 83 | 31 | 2988.62 | 2987.64 | 32.49 |
| Dermaseptin-SP3 | SLWSSIKDMAAAAGRAALNAVNGIVNP-NH2 | mc,ms2 | 100 | 103 | 30 | 2696.41 | 2695.42 | 47.66 |
| Dermaseptin-SP4 | SLWSSIKDMAAAAGRAALNAVNGILNP-NH2 | mc,ms2 | 100 | 90 | 30 | 2710.42 | 2709.44 | 13.3 |
| Dermaseptin-SP5 | SLRSSIKDMAAAAGRAALNAVNGIVNP-NH2 | mc,ms2 | 100 | 75 | 30 | 2666.43 | 2665.44 | 14,13 |
| Orphan peptide SP-1 | IIGMIPDLISMISKL-NH2 | mc,ms2 | 100 | 84 | 17 | 1642.94 | 1641.96 | 108.95 |
| .rphan peptide SP-2 | FLFLPFIGRRK | mc,ms2 | 100 | 31 | 11 | 1392.84 | 1393.74 | 22.05 |
| Orphan peptide SP-3 | FLLPPFFGRKK | mc | 0 | * | 11 | * | 1349.69 | * |
| Synthetic Peptide. | Sequence | MIC μM (mg/L) | |||||
|---|---|---|---|---|---|---|---|
| E. coli ATCC 25922 | S. aureus ATCC 25923 | S. aureus Oxacillin Resistant (ORSA Strain 20) | K. pneumoniae Clinical Isolate | C. albicans | Accession Number | ||
| Dermaseptin-SP2 | ASWKVFLKNIGKAAGKAVLNSVTDMVNQ-NH2 | 2.68 (8) * | 2.68 (8) * | 2.68 (8) | 10.71 (32) | 10.71 (32) * | MK532480 |
| Dermaseptin-SP3 | SLWSSIKDMAAAAGRAALNAVNGIVNP-NH2 | 47.50 (128) * | 189.98 (512) * | ND | >189.98 (>512) | >189.98 (>512) * | MK532481 |
| Dermaseptin-SP4 | SLWSSIKDMAAAAGRAALNAVNGILNP-NH2 | 47.25 (128) * | 189.00 (512) * | ND | 189.00 (512) | >189.00 (>512) * | MK532482 |
| Dermaseptin-SP5 | SLRSSIKDMAAAAGRAALNAVNGIVNP-NH2 | 96.06 (256) * | >192.12 (>512) * | ND | >189.00 (>512) | 384.24 (1024) * | MK532483 |
| Dermaseptin-B4 | ALWKDILKNVGKAAGKAVLNTVTDMVNQ-NH2 | 5.00 ** | 3.00 ** | ND | ND | ND | P81486 |
| Dermaseptin-S1 | ALWKTMLKKLGTMALHAGKAALGAAADTISQGTQ | 46.00 ** | 60.00 ** | ND | ND | ND | AJ564794.1 |
| Phyloseptin-SP1 | FLSLIPHVISAIPHVVNALSNL-NH2 | >217.69 (>512) | >217.69 (>512) | ND | >189.00 (>512) | >217.69 (>512) | MK532479 |
| Phyloseptin-S1 | FLSLIPHIVSGVASIAKHFG | 70.00 *** | 6.25 *** | ND | ND | ND | AM903077.1 |
| Ampicillin | 6.0 **** | 0.1 **** | |||||
| Dermaseptin-SP2 | ASWKVFLKNIGKAAGKAVLNSVTDMVNQ-NH2 | 2.68 (8) * | 2.68 (8) * | 2.68 (8) | 10.71 (32) | 10.71 (32) * | MK532480 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Proaño-Bolaños, C.; Blasco-Zúñiga, A.; Almeida, J.R.; Wang, L.; Llumiquinga, M.A.; Rivera, M.; Zhou, M.; Chen, T.; Shaw, C. Unravelling the Skin Secretion Peptides of the Gliding Leaf Frog, Agalychnis spurrelli (Hylidae). Biomolecules 2019, 9, 667. https://doi.org/10.3390/biom9110667
Proaño-Bolaños C, Blasco-Zúñiga A, Almeida JR, Wang L, Llumiquinga MA, Rivera M, Zhou M, Chen T, Shaw C. Unravelling the Skin Secretion Peptides of the Gliding Leaf Frog, Agalychnis spurrelli (Hylidae). Biomolecules. 2019; 9(11):667. https://doi.org/10.3390/biom9110667
Chicago/Turabian StyleProaño-Bolaños, Carolina, Ailín Blasco-Zúñiga, José Rafael Almeida, Lei Wang, Miguel Angel Llumiquinga, Miryan Rivera, Mei Zhou, Tianbao Chen, and Chris Shaw. 2019. "Unravelling the Skin Secretion Peptides of the Gliding Leaf Frog, Agalychnis spurrelli (Hylidae)" Biomolecules 9, no. 11: 667. https://doi.org/10.3390/biom9110667
APA StyleProaño-Bolaños, C., Blasco-Zúñiga, A., Almeida, J. R., Wang, L., Llumiquinga, M. A., Rivera, M., Zhou, M., Chen, T., & Shaw, C. (2019). Unravelling the Skin Secretion Peptides of the Gliding Leaf Frog, Agalychnis spurrelli (Hylidae). Biomolecules, 9(11), 667. https://doi.org/10.3390/biom9110667








