Non-Pancreatic Digestive Enzymes
Abstract
1. Overview
2. Pre-Pancreatic Digestion
2.1. Pre-Pancreatic Digestion of Carbohydrates: Salivary Amylase
2.2. Pre-Pancreatic Digestion of Fats
2.2.1. Lingual Lipase
2.2.2. Gastric Lipase
2.3. Pre-Pancreatic Digestion of Proteins: Pepsin
3. Post-Pancreatic Digestion
3.1. Post-Pancreatic Digestion of Carbohydrates
3.1.1. Alpha Glucosidases
Sucrase-Isomaltase
Maltase-Glucoamylase
Trehalase
3.1.2. Lactase-Phlorizin Hydrolase
3.2. Post-Pancreatic Digestion of Fats
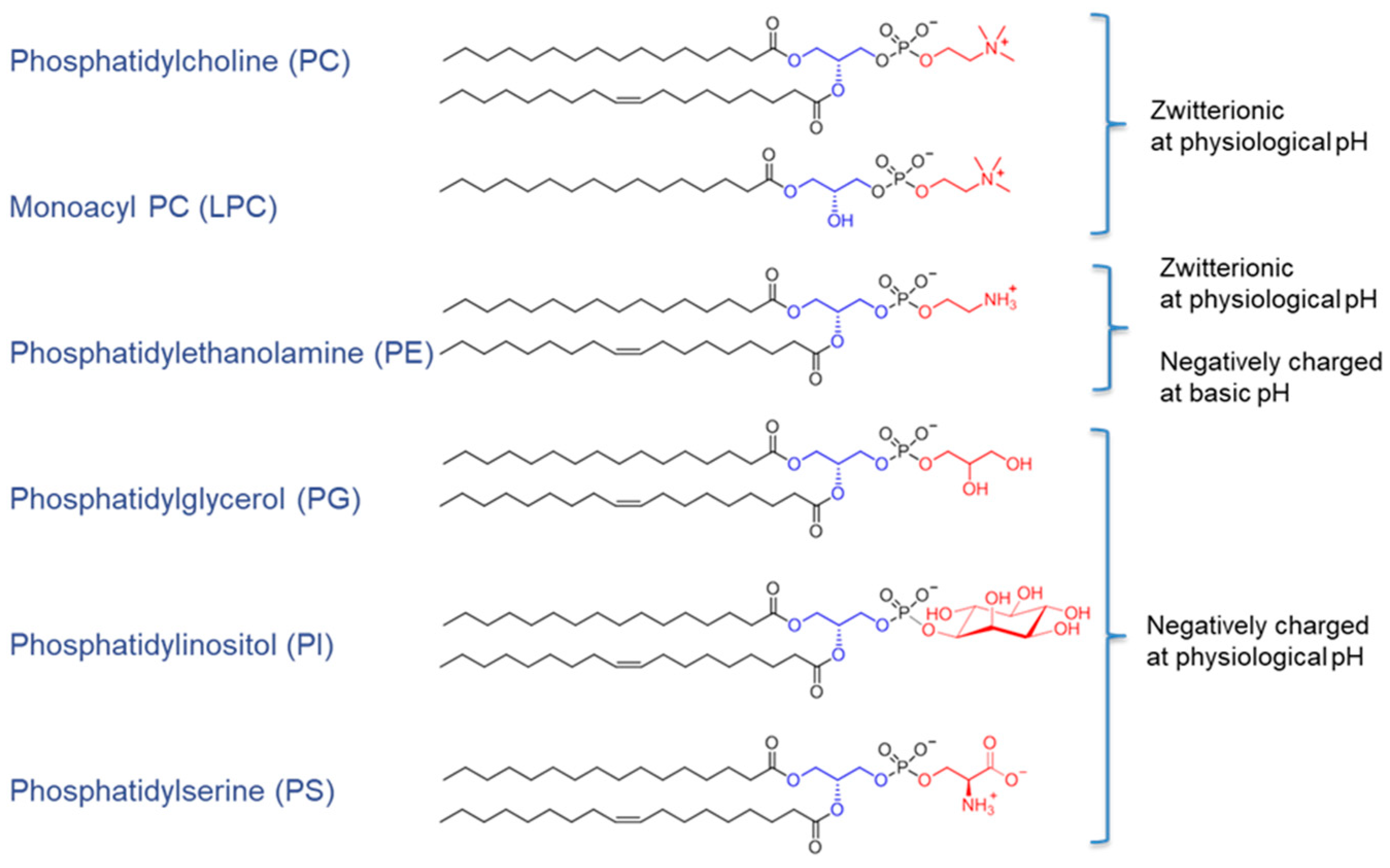
3.2.1. Glycerophospholipids
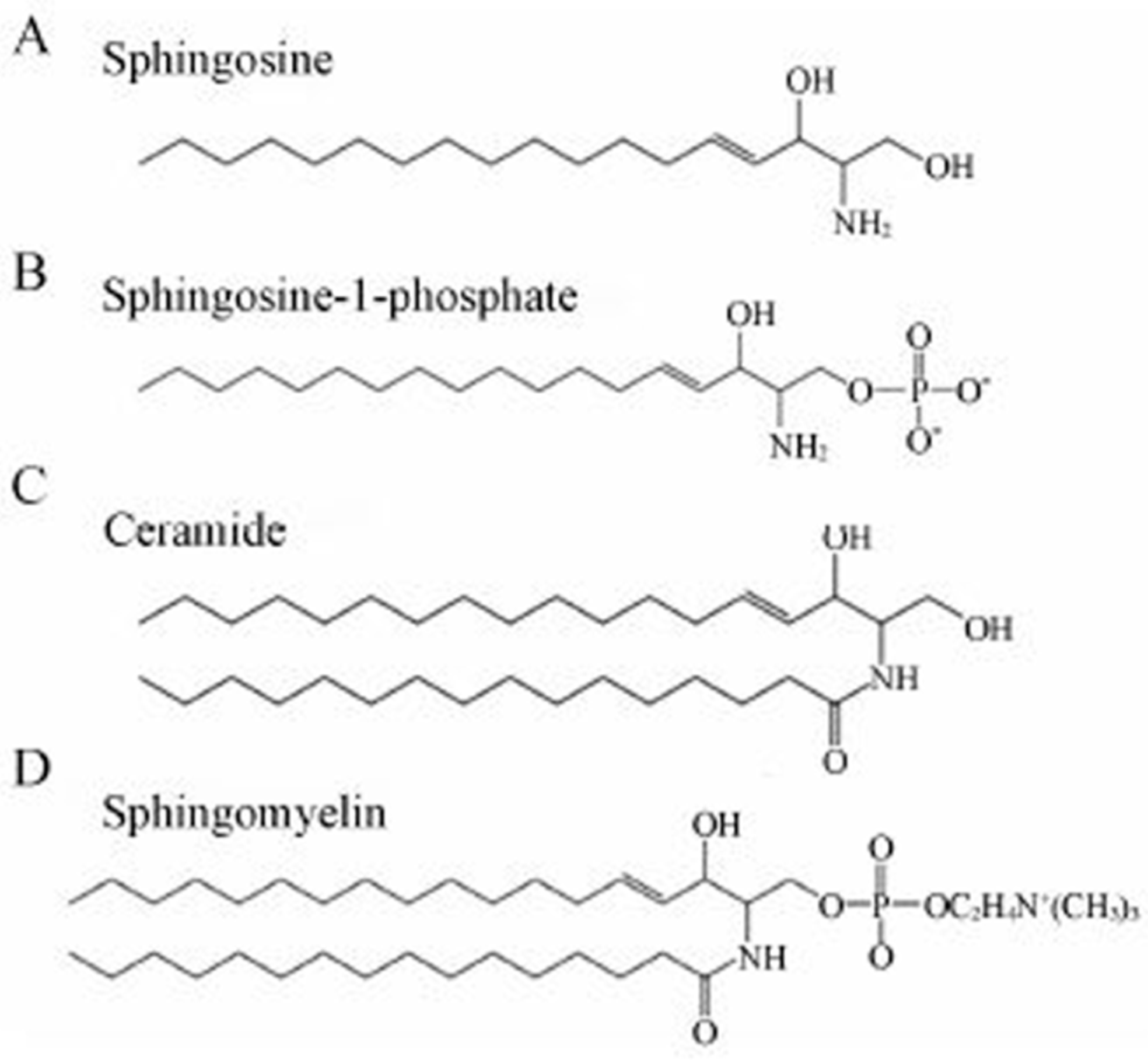
3.2.2. Sphingophospholipids
Alkaline Sphingomyelinase (Alk-SMase)
Neutral Ceramidase
3.3. Post-Pancreatic Digestion of Proteins
3.3.1. Aminopeptidase N
3.3.2. Dipeptidyl Aminopeptidase IV
3.3.3. Glutamyl Aminopeptidase (ENPEP; Aminopeptidase A)
3.3.4. Carboxypeptidases
3.3.5. Angiotensin-Converting Enzymes (ACE and ACE2)
3.3.6. A Few Endopeptidases Are Also Found in the Small Intestine
4. Clinical Relevance
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef]
- Fieker, A.; Philpott, J.; Armand, M. Enzyme replacement therapy for pancreatic insufficiency: Present and future. Clin. Exp. Gastroenterol. 2011, 4, 55–73. [Google Scholar] [CrossRef] [PubMed]
- Evans, I.D.; Haisman, D.R.; Elson, E.L.; Pasternak, C.; McConnaughey, W.B. The effect of salivary amylase on the viscosity behaviour of gelatinised starch suspensions and the mechanical properties of gelatinised starch granules. J. Sci. Food Agric. 1986, 37, 573–590. [Google Scholar] [CrossRef]
- Meites, S.; Rogols, S. Serum amylases, isoenzymes, and pancreatitis. I. Effect of substrate variation. Clin. Chem. 1968, 14, 1176–1184. [Google Scholar] [CrossRef]
- Hall, F.F.; Ratliff, C.R.; Hayakawa, T.; Culp, T.W.; Hightower, N.C. Substrate differentiation of human pancreatic and salivary alpha-amylases. Am. J. Dig. Dis. 1970, 15, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Perry, G.H.; Dominy, N.J.; Claw, K.G.; Lee, A.S.; Fiegler, H.; Redon, R.; Werner, J.; Villanea, F.A.; Mountain, J.L.; Misra, R.; et al. Diet and the evolution of human amylase gene copy number variation. Nat. Genet. 2007, 39, 1256–1260. [Google Scholar] [CrossRef] [PubMed]
- Merigo, F.; Benati, D.; Cecchini, M.P.; Cristofoletti, M.; Osculati, F.; Sbarbati, A. Amylase expression in taste receptor cells of rat circumvallate papillae. Cell Tissue Res. 2009, 336, 411–421. [Google Scholar] [CrossRef]
- Lapis, T.J.; Penner, M.H.; Lim, J. Evidence that humans can taste glucose polymers. Chem. Senses 2014, 39, 737–747. [Google Scholar] [CrossRef]
- Scannapieco, F.A.; Torres, G.; Levine, M.J. Salivary alpha-amylase: Role in dental plaque and caries formation. Crit. Rev. Oral. Biol. Med. 1993, 4, 301–307. [Google Scholar] [CrossRef]
- Nater, U.M.; Rohleder, N. Salivary alpha-amylase as a non-invasive biomarker for the sympathetic nervous system: Current state of research. Psychoneuroendocrinology 2009, 34, 486–496. [Google Scholar] [CrossRef]
- Ali, N.; Nater, U.M. Salivary Alpha-Amylase as a Biomarker of Stress in Behavioral Medicine. Int. J. Behav. Med. 2020, 27, 337–342. [Google Scholar] [CrossRef]
- Murray, R.D.; Kerzner, B.; Sloan, H.R.; McClung, H.J.; Gilbert, M.; Ailabouni, A. The contribution of salivary amylase to glucose polymer hydrolysis in premature infants. Pediatr. Res. 1986, 20, 186–191. [Google Scholar] [CrossRef]
- Aguirre, A.; Levine, M.J.; Cohen, R.E.; Tabak, L.A. Immunochemical quantitation of alpha-amylase and secretory IgA in parotid saliva from people of various ages. Arch. Oral. Biol. 1987, 32, 297–301. [Google Scholar] [CrossRef]
- Skude, G.; Kollberg, H. Serum isoamylases in cystic fibrosis. Acta Paediatr. Scand. 1976, 65, 145–149. [Google Scholar] [CrossRef]
- Townes, P.L.; Moore, W.D.; White, M.R. Amylase polymorphism: Studies of sera and duodenal aspirates in normal individuals and in cystic fibrosis. Am. J. Hum. Genet. 1976, 28, 378–389. [Google Scholar] [PubMed]
- Al-Manei, K.; Almotairy, N.; Bostanci, N.; Kumar, A.; Grigoriadis, A. Effect of Chewing on the Expression of Salivary Protein Composition: A Systematic Review. Proteom. Clin. Appl. 2020, 14, e1900039. [Google Scholar] [CrossRef] [PubMed]
- Abrams, C.K.; Hamosh, M.; Hubbard, V.S.; Dutta, S.K.; Hamosh, P. Lingual lipase in cystic fibrosis. Quantitation of enzyme activity in the upper small intestine of patients with exocrine pancreatic insufficiency. J. Clin. Investig. 1984, 73, 374–382. [Google Scholar] [CrossRef]
- Carriere, F.; Grandval, P.; Renou, C.; Palomba, A.; Prieri, F.; Giallo, J.; Henniges, F.; Sander-Struckmeier, S.; Laugier, R. Quantitative study of digestive enzyme secretion and gastrointestinal lipolysis in chronic pancreatitis. Clin. Gastroenterol. Hepatol. 2005, 3, 28–38. [Google Scholar] [CrossRef]
- Stern, R.C.; Eisenberg, J.D.; Wagener, J.S.; Ahrens, R.; Rock, M.; doPico, G.; Orenstein, D.M. A comparison of the efficacy and tolerance of pancrelipase and placebo in the treatment of steatorrhea in cystic fibrosis patients with clinical exocrine pancreatic insufficiency. Am. J. Gastroenterol. 2000, 95, 1932–1938. [Google Scholar] [CrossRef]
- Konstan, M.W.; Stern, R.C.; Trout, J.R.; Sherman, J.M.; Eigen, H.; Wagener, J.S.; Duggan, C.; Wohl, M.E.; Colin, P. Ultrase MT12 and Ultrase MT20 in the treatment of exocrine pancreatic insufficiency in cystic fibrosis: Safety and efficacy. Aliment. Pharmacol. Ther. 2004, 20, 1365–1371. [Google Scholar] [CrossRef] [PubMed]
- Wooldridge, J.L.; Heubi, J.E.; Amaro-Galvez, R.; Boas, S.R.; Blake, K.V.; Nasr, S.Z.; Chatfield, B.; McColley, S.A.; Woo, M.S.; Hardy, K.A.; et al. EUR-1008 pancreatic enzyme replacement is safe and effective in patients with cystic fibrosis and pancreatic insufficiency. J. Cyst. Fibros. 2009, 8, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Trapnell, B.C.; Maguiness, K.; Graff, G.R.; Boyd, D.; Beckmann, K.; Caras, S. Efficacy and safety of Creon 24,000 in subjects with exocrine pancreatic insufficiency due to cystic fibrosis. J. Cyst. Fibros. 2009, 8, 370–377. [Google Scholar] [CrossRef]
- Liao, T.H.; Hamosh, P.; Hamosh, M. Fat digestion by lingual lipase: Mechanism of lipolysis in the stomach and upper small intestine. Pediatr. Res. 1984, 18, 402–409. [Google Scholar] [CrossRef]
- Kulkarni, B.V.; Mattes, R.D. Lingual lipase activity in the orosensory detection of fat by humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 306, R879–R885. [Google Scholar] [CrossRef]
- Kulkarni, B.; Mattes, R. Evidence for presence of nonesterified fatty acids as potential gustatory signaling molecules in humans. Chem. Senses 2013, 38, 119–127. [Google Scholar] [CrossRef]
- DeNigris, S.J.; Hamosh, M.; Kasbekar, D.K.; Lee, T.C.; Hamosh, P. Lingual and gastric lipases: Species differences in the origin of prepancreatic digestive lipases and in the localization of gastric lipase. Biochim. Biophys. Acta 1988, 959, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Abrams, C.K.; Hamosh, M.; Lee, T.C.; Ansher, A.F.; Collen, M.J.; Lewis, J.H.; Benjamin, S.B.; Hamosh, P. Gastric lipase: Localization in the human stomach. Gastroenterology 1988, 95, 1460–1464. [Google Scholar] [CrossRef]
- Sams, L.; Paume, J.; Giallo, J.; Carriere, F. Relevant pH and lipase for in vitro models of gastric digestion. Food Funct. 2016, 7, 30–45. [Google Scholar] [CrossRef]
- Carriere, F.; Renou, C.; Ransac, S.; Lopez, V.; De Caro, J.; Ferrato, F.; De Caro, A.; Fleury, A.; Sanwald-Ducray, P.; Lengsfeld, H.; et al. Inhibition of gastrointestinal lipolysis by Orlistat during digestion of test meals in healthy volunteers. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 281, G16–G28. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, S.; Jannin, V.; Rodier, J.D.; Ritter, N.; Mahler, B.; Carriere, F. Comparative study on digestive lipase activities on the self emulsifying excipient Labrasol, medium chain glycerides and PEG esters. Biochim. Biophys. Acta 2007, 1771, 633–640. [Google Scholar] [CrossRef]
- Gargouri, Y.; Pieroni, G.; Lowe, P.A.; Sarda, L.; Verger, R. Human gastric lipase. The effect of amphiphiles. Eur. J. Biochem. 1986, 156, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Bernback, S.; Blackberg, L.; Hernell, O. The complete digestion of human milk triacylglycerol in vitro requires gastric lipase, pancreatic colipase-dependent lipase, and bile salt-stimulated lipase. J. Clin. Investig. 1990, 85, 1221–1226. [Google Scholar] [CrossRef] [PubMed]
- Gargouri, Y.; Pieroni, G.; Riviere, C.; Lowe, P.A.; Sauniere, J.F.; Sarda, L.; Verger, R. Importance of human gastric lipase for intestinal lipolysis: An in vitro study. Biochim. Biophys. Acta 1986, 879, 419–423. [Google Scholar] [CrossRef]
- MacGregor, I.L.; Martin, P.; Meyer, J.H. Gastric emptying of solid food in normal man and after subtotal gastrectomy and truncal vagotomy with pyloroplasty. Gastroenterology 1977, 72, 206–211. [Google Scholar] [CrossRef]
- Mayer, E.A.; Thompson, J.B.; Jehn, D.; Reedy, T.; Elashoff, J.; Meyer, J.H. Gastric emptying and sieving of solid food and pancreatic and biliary secretion after solid meals in patients with truncal vagotomy and antrectomy. Gastroenterology 1982, 83, 184–192. [Google Scholar] [CrossRef]
- Al Massadi, O.; Tschop, M.H.; Tong, J. Ghrelin acylation and metabolic control. Peptides 2011, 32, 2301–2308. [Google Scholar] [CrossRef]
- Campos, L.A.; Sancho, J. The active site of pepsin is formed in the intermediate conformation dominant at mildly acidic pH. FEBS Lett. 2003, 538, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Hamuro, Y.; Coales, S.J.; Molnar, K.S.; Tuske, S.J.; Morrow, J.A. Specificity of immobilized porcine pepsin in H/D exchange compatible conditions. Rapid Commun. Mass. Spectrom. 2008, 22, 1041–1046. [Google Scholar] [CrossRef]
- Conigrave, A.D.; Franks, A.H.; Brown, E.M.; Quinn, S.J. L-amino acid sensing by the calcium-sensing receptor: A general mechanism for coupling protein and calcium metabolism? Eur. J. Clin. Nutr. 2002, 56, 1072–1080. [Google Scholar] [CrossRef]
- Taylor, I.L.; Byrne, W.J.; Christie, D.L.; Ament, M.E.; Walsh, J.H. Effect of individual l-amino acids on gastric acid secretion and serum gastrin and pancreatic polypeptide release in humans. Gastroenterology 1982, 83, 273–278. [Google Scholar] [CrossRef]
- Hooton, D.; Lentle, R.; Monro, J.; Wickham, M.; Simpson, R. The Secretion and Action of Brush Border Enzymes in the Mammalian Small Intestine. Rev. Physiol. Biochem. Pharmacol. 2015, 168, 59–118. [Google Scholar] [CrossRef]
- Date, K.; Satoh, A.; Iida, K.; Ogawa, H. Pancreatic alpha-Amylase Controls Glucose Assimilation by Duodenal Retrieval through N-Glycan-specific Binding, Endocytosis, and Degradation. J. Biol. Chem. 2015, 290, 17439–17450. [Google Scholar] [CrossRef]
- Amiri, M.; Naim, H.Y. Characterization of Mucosal Disaccharidases from Human Intestine. Nutrients 2017, 9, 1106. [Google Scholar] [CrossRef]
- Skovbjerg, H. Immunoelectrophoretic studies on human small intestinal brush border proteins--the longitudinal distribution of peptidases and disaccharidases. Clin. Chim. Acta 1981, 112, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Danialifar, T.F.; Chumpitazi, B.P.; Mehta, D.I.; Di Lorenzo, C. Genetic and acquired sucrase-isomaltase deficiency: A clinical review. J. Pediatr. Gastroenterol. Nutr. 2024, 78, 774–782. [Google Scholar] [CrossRef]
- Zheng, T.; Eswaran, S.; Photenhauer, A.L.; Merchant, J.L.; Chey, W.D.; D’Amato, M. Reduced efficacy of low FODMAPs diet in patients with IBS-D carrying sucrase-isomaltase (SI) hypomorphic variants. Gut 2020, 69, 397–398. [Google Scholar] [CrossRef]
- Gericke, B.; Amiri, M.; Scott, C.R.; Naim, H.Y. Molecular pathogenicity of novel sucrase-isomaltase mutations found in congenital sucrase-isomaltase deficiency patients. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Ellestad-Sayed, J.J.; Haworth, J.C.; Hildes, J.A. Disaccharide malabsorption and dietary patterns in two Canadian Eskimo communities. Am. J. Clin. Nutr. 1978, 31, 1473–1478. [Google Scholar] [CrossRef]
- Uhrich, S.; Wu, Z.; Huang, J.Y.; Scott, C.R. Four mutations in the SI gene are responsible for the majority of clinical symptoms of CSID. J. Pediatr. Gastroenterol. Nutr. 2012, 55 (Suppl. 2), S34–S35. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Hamaker, B.R. Maltase Has Most Versatile alpha-Hydrolytic Activity Among the Mucosal alpha-Glucosidases of the Small Intestine. J. Pediatr. Gastroenterol. Nutr. 2018, 66 (Suppl. 3), S7–S10. [Google Scholar] [CrossRef]
- El-Chammas, K.; Williams, S.E.; Miranda, A. Disaccharidase Deficiencies in Children with Chronic Abdominal Pain. JPEN J. Parenter. Enter. Nutr. 2017, 41, 463–469. [Google Scholar] [CrossRef]
- Viswanathan, L.; Rao, S.S. Intestinal Disaccharidase Deficiency in Adults: Evaluation and Treatment. Curr. Gastroenterol. Rep. 2023, 25, 134–139. [Google Scholar] [CrossRef]
- Oesterreicher, T.J.; Nanthakumar, N.N.; Winston, J.H.; Henning, S.J. Rat trehalase: cDNA cloning and mRNA expression in adult rat tissues and during intestinal ontogeny. Am. J. Physiol. 1998, 274, R1220–R1227. [Google Scholar] [CrossRef][Green Version]
- Gudmand-Hoyer, E.; Fenger, H.J.; Skovbjerg, H.; Kern-Hansen, P.; Madsen, P.R. Trehalase deficiency in Greenland. Scand. J. Gastroenterol. 1988, 23, 775–778. [Google Scholar] [CrossRef] [PubMed]
- Murray, I.A.; Coupland, K.; Smith, J.A.; Ansell, I.D.; Long, R.G. Intestinal trehalase activity in a UK population: Establishing a normal range and the effect of disease. Br. J. Nutr. 2000, 83, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Savilahti, E.; Launiala, K.; Kuitunen, P. Congenital lactase deficiency. A clinical study on 16 patients. Arch. Dis. Child. 1983, 58, 246–252. [Google Scholar] [CrossRef]
- Wanes, D.; Husein, D.M.; Naim, H.Y. Congenital Lactase Deficiency: Mutations, Functional and Biochemical Implications, and Future Perspectives. Nutrients 2019, 11, 461. [Google Scholar] [CrossRef] [PubMed]
- Antonowicz, I.; Lebenthal, E. Developmental pattern of small intestinal enterokinase and disaccharidase activities in the human fetus. Gastroenterology 1977, 72, 1299–1303. [Google Scholar] [CrossRef]
- Sterchi, E.E.; Mills, P.R.; Fransen, J.A.; Hauri, H.P.; Lentze, M.J.; Naim, H.Y.; Ginsel, L.; Bond, J. Biogenesis of intestinal lactase-phlorizin hydrolase in adults with lactose intolerance. Evidence for reduced biosynthesis and slowed-down maturation in enterocytes. J. Clin. Investig. 1990, 86, 1329–1337. [Google Scholar] [CrossRef]
- Heyman, M.B.; Committee on Nutrition. Lactose intolerance in infants, children, and adolescents. Pediatrics 2006, 118, 1279–1286. [Google Scholar] [CrossRef]
- Van Hoogevest, P.; Tiemessen, H.; Metselaar, J.M.; Drescher, S.; Fahr, A. The Use of Phospholipids to Make Pharmaceutical Form Line Extensions. Eur. J. Lipid Sci. Technol. 2021, 124, 2000297. [Google Scholar] [CrossRef]
- Richmond, B.L.; Boileau, A.C.; Zheng, S.; Huggins, K.W.; Granholm, N.A.; Tso, P.; Hui, D.Y. Compensatory phospholipid digestion is required for cholesterol absorption in pancreatic phospholipase A(2)-deficient mice. Gastroenterology 2001, 120, 1193–1202. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.; Duan, R.D. Pancreatic and mucosal enzymes in choline phospholipid digestion. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 316, G425–G445. [Google Scholar] [CrossRef] [PubMed]
- Rigtrup, K.M.; McEwen, L.R.; Said, H.M.; Ong, D.E. Retinyl ester hydrolytic activity associated with human intestinal brush border membranes. Am. J. Clin. Nutr. 1994, 60, 111–116. [Google Scholar] [CrossRef]
- Duan, R.D.; Cheng, Y.; Hansen, G.; Hertervig, E.; Liu, J.J.; Syk, I.; Sjostrom, H.; Nilsson, A. Purification, localization, and expression of human intestinal alkaline sphingomyelinase. J. Lipid Res. 2003, 44, 1241–1250. [Google Scholar] [CrossRef]
- Hertervig, E.; Nilsson, A.; Bjork, J.; Hultkrantz, R.; Duan, R.D. Familial adenomatous polyposis is associated with a marked decrease in alkaline sphingomyelinase activity: A key factor to the unrestrained cell proliferation? Br. J. Cancer 1999, 81, 232–236. [Google Scholar] [CrossRef]
- Sjoqvist, U.; Hertervig, E.; Nilsson, A.; Duan, R.D.; Ost, A.; Tribukait, B.; Lofberg, R. Chronic colitis is associated with a reduction of mucosal alkaline sphingomyelinase activity. Inflamm. Bowel Dis. 2002, 8, 258–263. [Google Scholar] [CrossRef]
- Lundgren, P.; Nilsson, A.; Duan, R.D. Distribution and properties of neutral ceramidase activity in rat intestinal tract. Dig. Dis. Sci. 2001, 46, 765–772. [Google Scholar] [CrossRef]
- Curtis, K.J.; Gaines, H.D.; Kim, Y.S. Protein digestion and absorption in rats with pancreatic duct occlusion. Gastroenterology 1978, 74, 1271–1276. [Google Scholar] [CrossRef]
- Mamone, G.; Picariello, G. Optimized extraction and large-scale proteomics of pig jejunum brush border membranes for use in in vitro digestion models. Food Res. Int. 2023, 164, 112326. [Google Scholar] [CrossRef]
- Mina-Osorio, P. The moonlighting enzyme CD13: Old and new functions to target. Trends Mol. Med. 2008, 14, 361–371. [Google Scholar] [CrossRef]
- Rohrborn, D.; Wronkowitz, N.; Eckel, J. DPP4 in Diabetes. Front. Immunol. 2015, 6, 386. [Google Scholar] [CrossRef]
- Holmes, R.S.; Spradling-Reeves, K.D.; Cox, L.A. Mammalian Glutamyl Aminopeptidase Genes (ENPEP) and Proteins: Comparative Studies of a Major Contributor to Arterial Hypertension. J. Data Min. Genom. Proteom. 2017, 8, 2. [Google Scholar] [CrossRef]
- Garcia-Guerrero, M.C.; Garcia-Pardo, J.; Berenguer, E.; Fernandez-Alvarez, R.; Barfi, G.B.; Lyons, P.J.; Aviles, F.X.; Huber, R.; Lorenzo, J.; Reverter, D. Crystal structure and mechanism of human carboxypeptidase O: Insights into its specific activity for acidic residues. Proc. Natl. Acad. Sci. USA 2018, 115, E3932–E3939. [Google Scholar] [CrossRef]
- Stevens, B.R.; Phillips, M.I.; Fernandez, A. Ramipril inhibition of rabbit (Oryctolagus cuniculus) small intestinal brush border membrane angiotensin converting enzyme. Comp. Biochem. Physiol. C Comp. Pharmacol. Toxicol. 1988, 91, 493–497. [Google Scholar] [CrossRef]
- Jaworska, K.; Koper, M.; Ufnal, M. Gut microbiota and renin-angiotensin system: A complex interplay at local and systemic levels. Am. J. Physiol. Gastrointest. Liver Physiol. 2021, 321, G355–G366. [Google Scholar] [CrossRef]
- Penninger, J.M.; Grant, M.B.; Sung, J.J.Y. The Role of Angiotensin Converting Enzyme 2 in Modulating Gut Microbiota, Intestinal Inflammation, and Coronavirus Infection. Gastroenterology 2021, 160, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Holzinger, A.; Maier, E.M.; Buck, C.; Mayerhofer, P.U.; Kappler, M.; Haworth, J.C.; Moroz, S.P.; Hadorn, H.B.; Sadler, J.E.; Roscher, A.A. Mutations in the proenteropeptidase gene are the molecular cause of congenital enteropeptidase deficiency. Am. J. Hum. Genet. 2002, 70, 20–25. [Google Scholar] [CrossRef]
- Green, N.; Chan, C.; Ooi, C.Y. The gastrointestinal microbiome, small bowel bacterial overgrowth, and microbiome modulators in cystic fibrosis. Pediatr. Pulmonol. 2024, 59 (Suppl. 1), S70–S80. [Google Scholar] [CrossRef] [PubMed]
- Caporilli, C.; Gianni, G.; Grassi, F.; Esposito, S. An Overview of Short-Bowel Syndrome in Pediatric Patients: Focus on Clinical Management and Prevention of Complications. Nutrients 2023, 15, 2341. [Google Scholar] [CrossRef] [PubMed]
- Goulet, O.; Abi Nader, E.; Pigneur, B.; Lambe, C. Short Bowel Syndrome as the Leading Cause of Intestinal Failure in Early Life: Some Insights into the Management. Pediatr. Gastroenterol. Hepatol. Nutr. 2019, 22, 303–329. [Google Scholar] [CrossRef] [PubMed]
- Drucker, D.J. Glucagon-like peptide 2. J. Clin. Endocrinol. Metab. 2001, 86, 1759–1764. [Google Scholar] [CrossRef] [PubMed]
| Carbohydrates | Fats | Proteins | |
|---|---|---|---|
| Pre-pancreatic |
|
|
|
| Post pancreatic |
|
|
|
| Enzyme | Carbohydrate Subunits | Bond Cleaved | Examples of Foods Providing Substrate | |
|---|---|---|---|---|
| Products of amylase digestion: | ||||
| Isomaltose | Isomaltase | Glucose+glucose | α1,6 | Wheat, cornmeal |
| Maltose | Maltase II | Glucose+glucose | Primarily α1,4 but also α-1,2, α-1,3 and α-1,6 | |
| Limit dextrin | Glucoamylase | Glucose polymer with isomaltose branch | α1,4; cleaves from the non-reducing end | |
| Dietary disaccharides: | ||||
| Sucrose | Sucrase | Glucose+fructose | α1,2 | Some fruits and vegetables |
| Trehalose | Trehelase | Glucose+glucose | α1,1 | Mushrooms, baker’s yeast |
| Lactose | Lactase | Glucose+galactose | β1,4 | Dairy products |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borowitz, D. Non-Pancreatic Digestive Enzymes. Biomolecules 2025, 15, 1259. https://doi.org/10.3390/biom15091259
Borowitz D. Non-Pancreatic Digestive Enzymes. Biomolecules. 2025; 15(9):1259. https://doi.org/10.3390/biom15091259
Chicago/Turabian StyleBorowitz, Drucy. 2025. "Non-Pancreatic Digestive Enzymes" Biomolecules 15, no. 9: 1259. https://doi.org/10.3390/biom15091259
APA StyleBorowitz, D. (2025). Non-Pancreatic Digestive Enzymes. Biomolecules, 15(9), 1259. https://doi.org/10.3390/biom15091259







