The Effects of Microgravity on the Structure and Function of Cardiomyocytes
Abstract
1. Introduction
2. Materials and Methods
3. Effects of Spaceflight and Real Microgravity on Cardiac Models
3.1. In Vitro Studies
3.1.1. Primary Cardiomyocytes
3.1.2. Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes
3.1.3. Cardiovascular Progenitor Cells and Embryonic Stem Cells
3.2. Animal Studies
3.2.1. Male C57BL/6 Mice
3.2.2. Current Insights, Limitations, and Future Directions for Animal Studies in Space
4. Effects of Simulated Microgravity Conditions on Cardiac Models
4.1. In Vitro Studies
4.1.1. Primary Cardiomyocytes
4.1.2. Stem Cell-Derived Cardiomyocytes
4.1.3. Immortalized Cardiomyocyte Cells
4.1.4. Simulated Space Radiation
4.2. Hindlimb Unloading
4.2.1. Wistar Rats
4.2.2. Sprague Dawley Rats
4.2.3. Kunming Mice
4.2.4. C57BL/6 Mice
4.2.5. Pharmacological or Therapeutic Interventions
4.2.6. Current Insights, Limitations, and Future Directions for the HU Model
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| μg | Microgravity |
| SV | Stroke volume |
| CO | Cardiac output |
| CVP | Central venous pressure |
| DBP | Diastolic blood pressure |
| MAP | Mean arterial pressure |
| SBP | Systolic blood pressure |
| HR | Heart rate |
| ANP | Atrial natriuretic peptide |
| SVR | Systemic vascular resistance |
| HRV | Heart rate variability |
| POI | Postflight orthostatic intolerance |
| HiPSC-CMs | Human-induced pluripotent stem cell-derived cardiomyocytes |
| ISS | International space station |
| CSS | Chinese space station |
| TCA | Tricarboxylic acid |
| s-μg | Simulated microgravity |
| ECM | Extracellular matrix |
| dECM | Decellularized porcine myocardial extracellular matrix |
| rGO | Reduced graphene oxide |
| CPCs | Cardiovascular progenitor cells |
| mESCs | Mouse embryonic stem cells |
| EBs | Embryoid bodies |
| AC5 | Adenylyl cyclase type 5 |
| RWV | Rotating wall vessel |
| ROS | Reactive oxygen species |
| CaMKII | Ca2+/calmodulin-dependent protein kinase II |
| HDAC4 | Histone deacetylase 4 |
| BNP | Brain natriuretic peptide |
| SSR | Simulated space radiation |
| IMJ | Intramitochondrial junction |
| IR | Ischemia–reperfusion |
| AMPK | AMP-activated protein kinase |
| PQS | Panax quinquefolium saponin |
| TRF | Time-restricted feeding |
References
- Hussain, I.; Ullah, R.; Simran, B.F.N.U.; Kaur, P.; Kumar, M.; Raj, R.; Faraz, M.; Mehmoodi, A.; Malik, J. Cardiovascular effects of long-duration space flight. Health Sci. Rep. 2024, 7, e2305. [Google Scholar] [CrossRef] [PubMed]
- Videbaek, R.; Norsk, P. Atrial distension in humans during microgravity induced by parabolic flights. J. Appl. Physiol. 1997, 83, 1862–1866. [Google Scholar] [CrossRef] [PubMed]
- Hargens, A.R.; Richardson, S. Cardiovascular adaptations, fluid shifts, and countermeasures related to space flight. Respir. Physiol. Neurobiol. 2009, 169, S30–S33. [Google Scholar] [CrossRef]
- Baran, R.; Marchal, S.; Garcia Campos, S.; Rehnberg, E.; Tabury, K.; Baselet, B.; Wehland, M.; Grimm, D.; Baatout, S. The Cardiovascular System in Space: Focus on In Vivo and In Vitro Studies. Biomedicines 2021, 10, 59. [Google Scholar] [CrossRef] [PubMed]
- Buckey, J.C.; Gaffney, F.A.; Lane, L.D.; Levine, B.D.; Watenpaugh, D.E.; Blomqvist, C.G. Central Venous Pressure in Space. N. Engl. J. Med. 1993, 328, 1853–1854. [Google Scholar] [CrossRef]
- Norsk, P.; Asmar, A.; Damgaard, M.; Christensen, N.J. Fluid shifts, vasodilatation and ambulatory blood pressure reduction during long duration spaceflight. J. Physiol. 2015, 593, 573–584. [Google Scholar] [CrossRef]
- Khine, H.W.; Steding-Ehrenborg, K.; Hastings, J.L.; Kowal, J.; Daniels, J.D.; Page, R.L.; Goldberger, J.J.; Ng, J.; Adams-Huet, B.; Bungo, M.W.; et al. Effects of Prolonged Spaceflight on Atrial Size, Atrial Electrophysiology, and Risk of Atrial Fibrillation. Circ. Arrhythmia Electrophysiol. 2018, 11, e005959. [Google Scholar] [CrossRef]
- Anzai, T.; Frey, M.A.; Nogami, A. Cardiac arrhythmias during long-duration spaceflights. J. Arrhythmia 2014, 30, 139–149. [Google Scholar] [CrossRef]
- Rossum, A.C.; Wood, M.L.; Bishop, S.L.; Deblock, H.; Charles, J.B. Evaluation of Cardiac Rhythm Disturbances During Extravehicular Activity. Am. J. Cardiol. 1997, 79, 1153–1155. [Google Scholar] [CrossRef]
- Fritsch-Yelle, J.M.; Leuenberger, U.A.; D’Aunno, D.S.; Rossum, A.C.; Brown, T.E.; Wood, M.L.; Josephson, M.E.; Goldberger, A.L. An episode of ventricular tachycardia during long-duration spaceflight. Am. J. Cardiol. 1998, 81, 1391–1392. [Google Scholar] [CrossRef]
- Otsuka, K.; Cornelissen, G.; Furukawa, S.; Kubo, Y.; Hayashi, M.; Shibata, K.; Mizuno, K.; Aiba, T.; Ohshima, H.; Mukai, C. Long-term exposure to space’s microgravity alters the time structure of heart rate variability of astronauts. Heliyon 2016, 2, e00211. [Google Scholar] [CrossRef]
- Blomqvist, G.C. Regulation of the systemic circulation at microgravity and during readaptation to 1G. Med. Sci. Sports Exerc. 1996, 28, 9. [Google Scholar] [CrossRef]
- Summers, R.L.; Martin, D.S.; Meck, J.V.; Coleman, T.G. Mechanism of Spaceflight-Induced Changes in Left Ventricular Mass. Am. J. Cardiol. 2005, 95, 1128–1130. [Google Scholar] [CrossRef] [PubMed]
- Perhonen, M.A.; Franco, F.; Lane, L.D.; Buckey, J.C.; Blomqvist, C.G.; Zerwekh, J.E.; Peshock, R.M.; Weatherall, P.T.; Levine, B.D. Cardiac atrophy after bed rest and spaceflight. J. Appl. Physiol. 2001, 91, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Meck, J.V.; Reyes, C.J.; Perez, S.A.; Goldberger, A.L.; Ziegler, M.G. Marked exacerbation of orthostatic intolerance after long- vs. short-duration spaceflight in veteran astronauts. Psychosom. Med. 2001, 63, 865–873. [Google Scholar] [CrossRef]
- Pramanik, J.; Kumar, A.; Panchal, L.; Prajapati, B. Countermeasures for Maintaining Cardiovascular Health in Space Missions. Curr. Cardiol. Rev. 2023, 19, e300323215156. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, H.; Yang, Z.; Chen, Y.; Wong, D.W.-C.; Lam, W.-K. Research Trends on Astronaut Physical Training as Countermeasures: A Bibliometric Analysis from Past 30 Years. Microgravity Sci. Technol. 2024, 36, 37. [Google Scholar] [CrossRef]
- Goldstein, M.A.; Edwards, R.J.; Schroeter, J.P. Cardiac morphology after conditions of microgravity during COSMOS 2044. J. Appl. Physiol. 1992, 73, S94–S100. [Google Scholar] [CrossRef]
- Hwang, H.; Rampoldi, A.; Forghani, P.; Li, D.; Fite, J.; Boland, G.; Maher, K.; Xu, C. Space microgravity increases expression of genes associated with proliferation and differentiation in human cardiac spheres. NPJ Microgravity 2023, 9, 8. [Google Scholar] [CrossRef]
- Rampoldi, A.; Forghani, P.; Li, D.; Hwang, H.; Armand, L.C.; Fite, J.; Boland, G.; Maxwell, J.; Maher, K.; Xu, C. Space microgravity improves proliferation of human iPSC-derived cardiomyocytes. Stem Cell Rep. 2022, 17, 2272–2285. [Google Scholar] [CrossRef]
- Jha, R.; Wu, Q.; Singh, M.; Preininger, M.K.; Han, P.; Ding, G.; Cho, H.C.; Jo, H.; Maher, K.O.; Wagner, M.B.; et al. Simulated Microgravity and 3D Culture Enhance Induction, Viability, Proliferation and Differentiation of Cardiac Progenitors from Human Pluripotent Stem Cells. Sci. Rep. 2016, 6, 30956. [Google Scholar] [CrossRef] [PubMed]
- Ruyters, G.; Braun, M.; Stang, K.M. Breakthroughs in Space Life Science Research: From Apollo 16 to the ISS; Springer International Publishing: Cham, Switzerland, 2021. [Google Scholar] [CrossRef]
- Yang, F.; Li, Y.; Ding, B.; Nie, J.; Wang, H.; Zhang, X.; Wang, C.; Ling, S.; Ni, C.; Dai, Z.; et al. Reduced function and disassembled microtubules of cultured cardiomyocytes in spaceflight. Chin. Sci. Bull. 2008, 53, 1185–1192. [Google Scholar] [CrossRef]
- Han, X.; Qu, L.; Yu, M.; Ye, L.; Shi, L.; Ye, G.; Yang, J.; Wang, Y.; Fan, H.; Wang, Y.; et al. Thiamine-modified metabolic reprogramming of human pluripotent stem cell-derived cardiomyocyte under space microgravity. Signal Transduct. Target. Ther. 2024, 9, 86. [Google Scholar] [CrossRef] [PubMed]
- Wnorowski, A.; Sharma, A.; Chen, H.; Wu, H.; Shao, N.-Y.; Sayed, N.; Liu, C.; Countryman, S.; Stodieck, L.S.; Rubins, K.H.; et al. Effects of Spaceflight on Human Induced Pluripotent Stem Cell-Derived Cardiomyocyte Structure and Function. Stem Cell Rep. 2019, 13, 960–969. [Google Scholar] [CrossRef]
- Acharya, A.; Brungs, S.; Lichterfeld, Y.; Hescheler, J.; Hemmersbach, R.; Boeuf, H.; Sachinidis, A. Parabolic, Flight-Induced, Acute Hypergravity and Microgravity Effects on the Beating Rate of Human Cardiomyocytes. Cells 2019, 8, 352. [Google Scholar] [CrossRef] [PubMed]
- Forghani, P.; Liu, W.; Wang, Z.; Ling, Z.; Takaesu, F.; Yang, E.; Tharp, G.K.; Nielsen, S.; Doraisingam, S.; Countryman, S.; et al. Spaceflight alters protein levels and gene expression associated with stress response and metabolic characteristics in human cardiac spheroids. Biomaterials 2025, 317, 123080. [Google Scholar] [CrossRef]
- Mair, D.B.; Tsui, J.H.; Higashi, T.; Koenig, P.; Dong, Z.; Chen, J.F.; Meir, J.U.; Smith, A.S.T.; Lee, P.H.U.; Ahn, E.H.; et al. Spaceflight-induced contractile and mitochondrial dysfunction in an automated heart-on-a-chip platform. Proc. Natl. Acad. Sci. USA 2024, 121, e2404644121. [Google Scholar] [CrossRef]
- Yang, H.; Yang, Y.; Kiskin, F.N.; Shen, M.; Zhang, J.Z. Recent advances in regulating the proliferation or maturation of human-induced pluripotent stem cell-derived cardiomyocytes. Stem Cell Res. Ther. 2023, 14, 228. [Google Scholar] [CrossRef]
- Sharma, A.; Wu, J.C.; Wu, S.M. Induced pluripotent stem cell-derived cardiomyocytes for cardiovascular disease modeling and drug screening. Stem Cell Res. Ther. 2013, 4, 150. [Google Scholar] [CrossRef]
- Kistamás, K.; Müller, A.; Muenthaisong, S.; Lamberto, F.; Zana, M.; Dulac, M.; Leal, F.; Maziz, A.; Costa, P.; Bernotiene, E.; et al. Multifactorial approaches to enhance maturation of human iPSC-derived cardiomyocytes. J. Mol. Liq. 2023, 387, 122668. [Google Scholar] [CrossRef]
- Camberos, V.; Baio, J.; Mandujano, A.; Martinez, A.F.; Bailey, L.; Hasaniya, N.; Kearns-Jonker, M. The Impact of Spaceflight and Microgravity on the Human Islet-1+ Cardiovascular Progenitor Cell Transcriptome. Int. J. Mol. Sci. 2021, 22, 3577. [Google Scholar] [CrossRef] [PubMed]
- Baio, J.; Martinez, A.F.; Bailey, L.; Hasaniya, N.; Pecaut, M.J.; Kearns-Jonker, M. Spaceflight Activates Protein Kinase C Alpha Signaling and Modifies the Developmental Stage of Human Neonatal Cardiovascular Progenitor Cells. Stem Cells Dev. 2018, 27, 805–818. [Google Scholar] [CrossRef]
- Blaber, E.A.; Finkelstein, H.; Dvorochkin, N.; Sato, K.Y.; Yousuf, R.; Burns, B.P.; Globus, R.K.; Almeida, E.A. Microgravity Reduces the Differentiation and Regenerative Potential of Embryonic Stem Cells. Stem Cells Dev. 2015, 24, 2605–2621. [Google Scholar] [CrossRef]
- Li, H.; Zhu, H.; Zhang, F.; Dong, X.; Hao, T.; Jiang, X.; Zheng, W.; Zhang, T.; Chen, X.; Wang, P.; et al. Spaceflight Promoted Myocardial Differentiation of Induced Pluripotent Stem Cells: Results from Tianzhou-1 Space Mission. Stem Cells Dev. 2019, 28, 357–360. [Google Scholar] [CrossRef]
- Menasché, P.; Vanneaux, V.; Hagège, A.; Bel, A.; Cholley, B.; Cacciapuoti, I.; Parouchev, A.; Benhamouda, N.; Tachdjian, G.; Tosca, L.; et al. Human embryonic stem cell-derived cardiac progenitors for severe heart failure treatment: First clinical case report. Eur. Heart J. 2015, 36, 2011–2017. [Google Scholar] [CrossRef]
- Behfar, A.; Yamada, S.; Crespo-Diaz, R.; Nesbitt, J.J.; Rowe, L.A.; Perez-Terzic, C.; Gaussin, V.; Homsy, C.; Bartunek, J.; Terzic, A. Guided cardiopoiesis enhances therapeutic benefit of bone marrow human mesenchymal stem cells in chronic myocardial infarction. J. Am. Coll. Cardiol. 2010, 56, 721–734. [Google Scholar] [CrossRef]
- Le, T.; Chong, J. Cardiac progenitor cells for heart repair. Cell Death Discov. 2016, 2, 16052. [Google Scholar] [CrossRef]
- Teerlink, J.R.; Metra, M.; Filippatos, G.S.; Davison, B.A.; Bartunek, J.; Terzic, A.; Gersh, B.J.; Povsic, T.J.; Henry, T.D.; Alexandre, B.; et al. Benefit of cardiopoietic mesenchymal stem cell therapy on left ventricular remodelling: Results from the Congestive Heart Failure Cardiopoietic Regenerative Therapy (CHART-1) study. Eur. J. Heart Fail. 2017, 19, 1520–1529. [Google Scholar] [CrossRef] [PubMed]
- Walls, S.; Diop, S.; Birse, R.; Elmen, L.; Gan, Z.; Kalvakuri, S.; Pineda, S.; Reddy, C.; Taylor, E.; Trinh, B.; et al. Prolonged Exposure to Microgravity Reduces Cardiac Contractility and Initiates Remodeling in Drosophila. Cell Rep. 2020, 33, 108445. [Google Scholar] [CrossRef] [PubMed]
- Davet, J.; Fagette, S.; Mani-Ponset, L.; Bayard, B.; Dumars, P.; Reiss-Bubenheim, D.; Güell, A.; Gharib, C.; Gabrion, J. Cardiac atrial natriuretic peptide (ANP) in rat dams and fetuses developed in space (NIH-R1 and NIH-R2 experiments). Life Sci. 1999, 64, 1533–1541. [Google Scholar] [CrossRef]
- Ulanova, A.; Gritsyna, Y.; Vikhlyantsev, I.; Salmov, N.; Bobylev, A.; Abdusalamova, Z.; Rogachevsky, V.; Shenkman, B.; Podlubnaya, Z. Isoform composition and gene expression of thick and thin filament proteins in striated muscles of mice after 30-day space flight. Biomed. Res. Int. 2015, 2015, 104735. [Google Scholar] [CrossRef]
- Ogneva, I.V.; Maximova, M.V.; Larina, I.M. Structure of cortical cytoskeleton in fibers of mouse muscle cells after being exposed to a 30-day space flight on board the BION-M1 biosatellite. J. Appl. Physiol. 2014, 116, 1315–1323. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.Y.; Saravia-Butler, A.; Shirazi-Fard, Y.; Leveson-Gower, D.; Stodieck, L.S.; Cadena, S.M.; Beegle, J.; Solis, S.; Ronca, A.; Globus, R.K. Validation of a New Rodent Experimental System to Investigate Consequences of Long Duration Space Habitation. Sci. Rep. 2020, 10, 2336. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Tsunematsu, T.; Jiao, Q.; Ohnuki, Y.; Mototani, Y.; Shiozawa, K.; Jin, M.; Cai, W.; Jin, H.-L.; Fujita, T.; et al. Pharmacological Stimulation of Type 5 Adenylyl Cyclase Stabilizes Heart Rate Under Both Microgravity and Hypergravity Induced by Parabolic Flight. J. Pharmacol. Sci. 2012, 119, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Okumura, S.; Tsunematsu, T.; Bai, Y.; Jiao, Q.; Ono, S.; Suzuki, S.; Kurotani, R.; Sato, M.; Minamisawa, S.; Umemura, S.; et al. Type 5 adenylyl cyclase plays a major role in stabilizing heart rate in response to microgravity induced by parabolic flight. J. Appl. Physiol. 2008, 105, 173–179. [Google Scholar] [CrossRef]
- Hamlin, R.L.; Keene, B.W. Species differences in cardiovascular physiology that affect pharmacology and toxicology. Curr. Opin. Toxicol. 2020, 23–24, 106–113. [Google Scholar] [CrossRef]
- Borst, A.G.; Van Loon, J.J.W.A. Technology and Developments for the Random Positioning Machine, RPM. Microgravity Sci. Technol. 2009, 21, 287–292. [Google Scholar] [CrossRef]
- Globus, R.K.; Morey-Holton, E. Hindlimb unloading: Rodent analog for microgravity. J. Appl. Physiol. 2016, 120, 1196–1206. [Google Scholar] [CrossRef]
- Rah, B.; Shafarin, J.; Qaisar, R.; Karim, A.; Hamad, M.; Muhammad, J.S. Mouse hindlimb unloading, as a model of simulated microgravity, leads to dysregulated iron homeostasis in liver and skeletal muscle cells. Life Sci. Space Res. 2025, 45, 7–15. [Google Scholar] [CrossRef]
- Sun, H.; Ling, S.; Zhao, D.; Li, Y.; Zhong, G.; Guo, M.; Li, Y.; Yang, L.; Du, J.; Zhou, Y.; et al. Panax quinquefolium saponin attenuates cardiac remodeling induced by simulated microgravity. Phytomedicine 2019, 56, 83–93. [Google Scholar] [CrossRef]
- Xiong, J.; Li, Y.; Nie, J. Effects of simulated microgravity on nitric oxide level in cardiac myocytes and its mechanism. Sci. China C Life Sci. 2003, 46, 302–309. [Google Scholar] [CrossRef]
- Feger, B.J.; Thompson, J.W.; Dubois, L.G.; Kommaddi, R.P.; Foster, M.W.; Mishra, R.; Shenoy, S.K.; Shibata, Y.; Kidane, Y.H.; Moseley, M.A.; et al. Microgravity induces proteomics changes involved in endoplasmic reticulum stress and mitochondrial protection. Sci. Rep. 2016, 6, 34091. [Google Scholar] [CrossRef]
- Akins, R.E.; Schroedl, N.A.; Gonda, S.R.; Hartzell, C.R. Neonatal rat heart cells cultured in simulated microgravity. Vitr. Cell. Dev. Biol.-Anim. 1997, 33, 337–343. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Leung, C.M.; de Haan, P.; Ronaldson-Bouchard, K.; Kim, G.-A.; Ko, J.; Rho, H.S.; Chen, Z.; Habibovic, P.; Jeon, N.L.; Takayama, S.; et al. A guide to the organ-on-a-chip. Nat. Rev. Methods Primers 2022, 2, 33. [Google Scholar] [CrossRef]
- Liu, B.; Wang, S.; Ma, H.; Deng, Y.; Du, J.; Zhao, Y.; Chen, Y. Heart-on-a-chip: A revolutionary organ-on-chip platform for cardiovascular disease modeling. J. Transl. Med. 2025, 23, 132. [Google Scholar] [CrossRef]
- Acharya, A.; Nemade, H.; Papadopoulos, S.; Hescheler, J.; Neumaier, F.; Schneider, T.; Prasad, K.R.; Khan, K.; Hemmersbach, R.; Gusmao, E.G.; et al. Microgravity-induced stress mechanisms in human stem cell-derived cardiomyocytes. iScience 2022, 25, 104577. [Google Scholar] [CrossRef] [PubMed]
- Forghani, P.; Rashid, A.; Armand, L.C.; Wolfson, D.; Liu, R.; Cho, H.C.; Maxwell, J.T.; Jo, H.; Salaita, K.; Xu, C. Simulated microgravity improves maturation of cardiomyocytes derived from human induced pluripotent stem cells. Sci. Rep. 2024, 14, 2243. [Google Scholar] [CrossRef]
- Hashida, A.; Nakazato, T.; Uemura, T.; Liu, L.; Miyagawa, S.; Sawa, Y.; Kino-Oka, M. Effect of morphological change on the maturation of human induced pluripotent stem cell-derived cardiac tissue in rotating flow culture. Regen. Ther. 2023, 24, 479–488. [Google Scholar] [CrossRef]
- Hatzistergos, K.E.; Jiang, Z.; Valasaki, K.; Takeuchi, L.M.; Balkan, W.; Atluri, P.; Saur, D.; Seidler, B.; Tsinoremas, N.; DiFede, D.L.; et al. Simulated Microgravity Impairs Cardiac Autonomic Neurogenesis from Neural Crest Cells. Stem Cells Dev. 2018, 27, 819–830. [Google Scholar] [CrossRef]
- Shinde, V.; Brungs, S.; Henry, M.; Wegener, L.; Nemade, H.; Rotshteyn, T.; Acharya, A.; Baumstark-Khan, C.; Hellweg, C.E.; Hescheler, J.; et al. Simulated Microgravity Modulates Differentiation Processes of Embryonic Stem Cells. Cell Physiol. Biochem. 2016, 38, 1483–1499. [Google Scholar] [CrossRef]
- White, S.M.; Constantin, P.E.; Claycomb, W.C. Cardiac physiology at the cellular level: Use of cultured HL-1 cardiomyocytes for studies of cardiac muscle cell structure and function. Am. J. Physiol. Heart Circ. Physiol. 2004, 286, H823–H829. [Google Scholar] [CrossRef] [PubMed]
- Watkins, S.J.; Borthwick, G.M.; Arthur, H.M. The H9C2 cell line and primary neonatal cardiomyocyte cells show similar hypertrophic responses in vitro. Vitr. Cell. Dev. Biol.-Anim. 2011, 47, 125–131. [Google Scholar] [CrossRef]
- Liu, C.; Zhong, G.; Zhou, Y.; Yang, Y.; Tan, Y.; Li, Y.; Gao, X.; Sun, W.; Li, J.; Jin, X.; et al. Alteration of calcium signalling in cardiomyocyte induced by simulated microgravity and hypergravity. Cell Prolif. 2020, 53, e12783. [Google Scholar] [CrossRef]
- Forte, G.; Carotenuto, F.; Pagliari, S.; Romano, R.; Cossa, P.; Pagliari, F.; Fiaccavento, R. Effects of physical factors on cardiac stem cells. J. Mol. Cell. Cardiol. 2007, 42, S92–S93. [Google Scholar] [CrossRef]
- Guarnieri, S.; Morabito, C.; Bevere, M.; Lanuti, P.; Mariggiò, M.A. A Protective Strategy to Counteract the Oxidative Stress Induced by Simulated Microgravity on H9C2 Cardiomyocytes. Oxid. Med. Cell Longev. 2021, 2021, 9951113. [Google Scholar] [CrossRef]
- Kwon, O.; Tranter, M.; Jones, W.K.; Sankovic, J.M.; Banerjee, R.K. Differential translocation of nuclear factor-kappaB in a cardiac muscle cell line under gravitational changes. J. Biomech. Eng. 2009, 131, 064503. [Google Scholar] [CrossRef]
- Beheshti, A.; McDonald, J.T.; Miller, J.; Grabham, P.; Costes, S.V. GeneLab Database Analyses Suggest Long-Term Impact of Space Radiation on the Cardiovascular System by the Activation of FYN Through Reactive Oxygen Species. Int. J. Mol. Sci. 2019, 20, 661. [Google Scholar] [CrossRef]
- Heselich, A.; Frieß, J.L.; Ritter, S.; Benz, N.P.; Layer, P.G.; Thielemann, C. High LET radiation shows no major cellular and functional effects on primary cardiomyocytes in vitro. Life Sci. Space Res. 2018, 16, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Tavakol, D.N.; Nash, T.R.; Kim, Y.; He, S.; Fleischer, S.; Graney, P.L.; Brown, J.A.; Liberman, M.; Tamargo, M.; Harken, A.; et al. Modeling and countering the effects of cosmic radiation using bioengineered human tissues. Biomaterials 2023, 301, 122267. [Google Scholar] [CrossRef] [PubMed]
- Holy, X.; Zerath, E.; Francois, A.; Facy, P.; Malouvier, A.; Nogues, C. Changes in rat atrial ANF granules induced by hindlimb suspension. Regul. Pept. 1994, 54, 417–428. [Google Scholar] [CrossRef]
- Kamkin, A.G.; Mitrokhin, V.M.; Kamkina, O.V.; Kazansky, V.E.; Rodina, A.S.; Zolotareva, A.D.; Zolotarev, V.I.; Sutyagin, P.V.; Mladenov, M.I.; Shenkman, B.S.; et al. Simulated Microgravity Changes the Number of Mechanically Gated and Mechanosensitive Ion Channels Genes Transcripts in Rat Ventricular Cardiomyocytes. Dokl. Biochem. Biophys. 2023, 512, 251–255. [Google Scholar] [CrossRef]
- Mitrokhin, V.M.; Kamkina, O.V.; Kamkin, A.G.; Rodina, A.S.; Zolotareva, A.D.; Zolotarev, V.I.; Kazansky, V.E.; Gorbacheva, L.R.; Bilichenko, A.S.; Shileiko, S.A.; et al. Simulated Microgravity and Hypergravity Affect the Expression Level of Soluble Guanylate Cyclase, Adenylate Cyclase, and Phosphodiesterase Genesin Rat Ventricular Cardiomyocytes. Bull. Exp. Biol. Med. 2024, 176, 359–362. [Google Scholar] [CrossRef]
- Ogneva, I.V.; Mirzoev, T.M.; Biryukov, N.S.; Veselova, O.M.; Larina, I.M. Structure and functional characteristics of rat’s left ventricle cardiomyocytes under antiorthostatic suspension of various duration and subsequent reloading. J. Biomed. Biotechnol. 2012, 2012, 659869. [Google Scholar] [CrossRef]
- Varenik, E.N.; Lipina, T.V.; Shornikova, M.V.; Krasnov, I.B.; Chentzov, Y.S. The structure of the rat left ventricular myocardium in models of microgravity and artificial gravitation. Dokl. Biol. Sci. 2010, 433, 286–288. [Google Scholar] [CrossRef]
- Varenik, E.N.; Lipina, T.V.; Shornikova, M.V.; Krasnov, I.B.; Chentsov, Y.S. Electron microscope analysis of cardiomyocytes in the rat left ventricle under simulation of weightlessness effects and artificial gravitation. Biol. Bull. Russ. Acad. Sci. 2012, 39, 221–228. [Google Scholar] [CrossRef]
- Lipina, T.V.; Shornikova, M.V.; Chentsov, Y.S. Morphometric analysis of cardiomyocytes of the rat heart left ventricle under simulated weightlessness. Dokl. Biol. Sci. 2003, 392, 464–466. [Google Scholar] [CrossRef]
- Lipina, T.V.; Shornikova, M.V.; Chentsov, Y.S. Electron microscopic study of left ventricular cardiomyocyte mitochondriom in rats subjected to head-down tilt. Bull. Exp. Biol. Med. 2004, 137, 291–293. [Google Scholar] [CrossRef]
- Pogodina, L.S.; Shornikova, M.V.; Chentsov, Y.S. Adaptive Responses of Secretory Cardiomyocytes of the Right Atrium during Simulation of Microgravity Effects by Long-Term and Repeated Antiorthostatic Tail Suspension of Rats. Biol. Bull. 2004, 31, 4–14. [Google Scholar] [CrossRef]
- Dunlap, A.W.; Thomason, D.B.; Menon, V.; Hofmann, P.A. Decreased Ca2+ sensitivity of isometric tension in skinned cardiac myocytes from tail-suspended rats. J. Appl. Physiol. 1996, 80, 1612–1617. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.B.; Zhang, L.F.; Jin, J.P. A proteolytic NH2-terminal truncation of cardiac troponin I that is up-regulated in simulated microgravity. J. Biol. Chem. 2001, 276, 15753–15760. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Zhang, S.-M.; Zhang, Q.-Y.; Fan, R.; Li, J.; Guo, H.-T.; Bi, H.; Wang, Y.-M.; Hu, Y.-Z.; Zheng, Q.-J.; et al. Modulation of intracellular calcium transient in response to beta-adrenoceptor stimulation in the hearts of 4-wk-old rats during simulated weightlessness. J. Appl. Physiol. 2010, 108, 838–844. [Google Scholar] [CrossRef][Green Version]
- Yin, W.; Liu, J.-C.; Fan, R.; Sun, X.-Q.; Ma, J.; Feng, N.; Zhang, Q.Y.; Yin, Z.; Zhang, S.-M.; Guo, H.-T.; et al. Modulation of {beta}-adrenoceptor signaling in the hearts of 4-wk simulated weightlessness rats. J. Appl. Physiol. 2008, 105, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Zhang, L.; Xu, P.-T.; Li, Q.; Sheng, J.-J.; Wang, Y.-Y.; Chen, Y.; Zhang, L.; Yu, Z. Nuclear translocation of calpain-2 regulates propensity toward apoptosis in cardiomyocytes of tail-suspended rats. J. Cell Biochem. 2011, 112, 571–580. [Google Scholar] [CrossRef]
- Lu, Y.-M.; Jiao, B.; Lee, J.; Zhang, L.; Yu, Z.-B. Simulated microgravity increases myocardial susceptibility to ischemia-reperfusion injury via a deficiency of AMP-activated protein kinase. Can. J. Physiol. Pharmacol. 2017, 95, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, X.-C.; Chen, Y.-F.; Wang, C.-L.; Chen, L.; Jiang, M.-Y.; Liu, X.; Zhang, X.; Feng, Y.; Xu, J. Loss and recovery of myocardial mitochondria in mice under different tail suspension time: Apoptosis and mitochondrial fission, fusion and autophagy. Exp. Physiol. 2023, 108, 1189–1202. [Google Scholar] [CrossRef] [PubMed]
- Jung, A.S.; Harrison, R.; Lee, K.H.; Genut, J.; Nyhan, D.; Brooks-Asplund, E.M.; Shoukas, A.A.; Hare, J.M.; Berkowitz, D.E. Simulated microgravity produces attenuated baroreflex-mediated pressor, chronotropic, and inotropic responses in mice. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, H600–H607. [Google Scholar] [CrossRef]
- Respress, J.L.; Gershovich, P.M.; Wang, T.; Reynolds, J.O.; Skapura, D.G.; Sutton, J.P.; Miyake, C.Y.; Wehrens, X.H. Long-term simulated microgravity causes cardiac RyR2 phosphorylation and arrhythmias in mice. Int. J. Cardiol. 2014, 176, 994–1000. [Google Scholar] [CrossRef]
- Liang, L.; Li, H.; Cao, T.; Qu, L.; Zhang, L.; Fan, G.-C.; Greer, P.A.; Li, J.; Jones, D.L.; Peng, T. Calpain activation mediates microgravity-induced myocardial abnormalities in mice via p38 and ERK1/2 MAPK pathways. J. Biol. Chem. 2020, 295, 16840–16851. [Google Scholar] [CrossRef]
- Ling, S.; Li, Y.; Zhong, G.; Zheng, Y.; Xu, Q.; Zhao, D.; Sun, W.; Jin, X.; Li, H.; Li, J.; et al. Myocardial CKIP-1 Overexpression Protects from Simulated Microgravity-Induced Cardiac Remodeling. Front. Physiol. 2018, 9, 40. [Google Scholar] [CrossRef]
- Pan, J.; Li, J.; Li, J.; Ling, S.; Du, R.; Xing, W.; Li, Y.; Sun, W.; Li, Y.; Fan, Y.; et al. miR-199a-3p mitigates simulated microgravity-induced cardiac remodeling by targeting MEF2C. FASEB J. 2025, 39, e70331. [Google Scholar] [CrossRef]
- Zhong, G.; Zhao, D.; Li, J.; Liu, Z.; Pan, J.; Yuan, X.; Xing, W.; Zhao, Y.; Ling, S.; Li, Y. WWP1 Deficiency Alleviates Cardiac Remodeling Induced by Simulated Microgravity. Front. Cell Dev. Biol. 2021, 9, 739944. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Yuan, W.; Qu, L.; Li, H.; Zhang, L.; Fan, G.-C.; Peng, T. Administration of losartan preserves cardiomyocyte size and prevents myocardial dysfunction in tail-suspended mice by inhibiting p47(phox) phosphorylation, NADPH oxidase activation and MuRF1 expression. J. Transl. Med. 2019, 17, 279. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Cao, T.; Ding, W.; Liang, L.; Fan, G.-C.; Qu, L.; Peng, T. Pharmacological inhibition of Rac1 attenuates myocardial abnormalities in tail-suspended mice. J. Cardiovasc. Transl. Res. 2022, 15, 805–815. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-P.; Xing, C.-Y.; Zhang, J.-X.; Zhou, J.-H.; Li, Y.-C.; Yang, H.-Y.; Zhang, P.; Zhang, W.; Huang, Y.; Long, J.; et al. Time-restricted feeding alleviates cardiac dysfunction induced by simulated microgravity via restoring cardiac FGF21 signaling. FASEB J. 2020, 34, 15180–15196. [Google Scholar] [CrossRef]
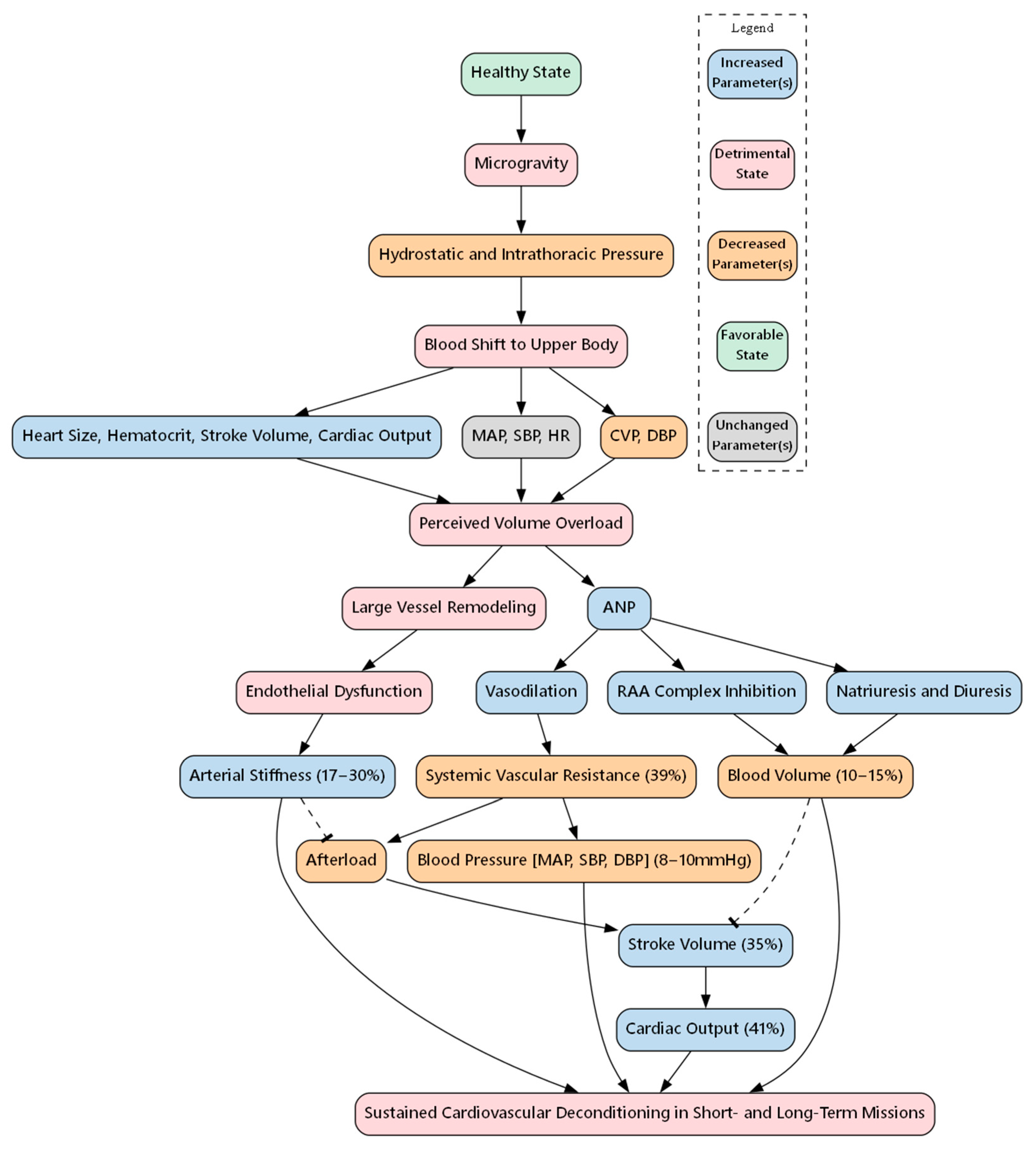
 , disturbing influence. Parts of the figure were drawn using pictures from Biorender.com and Servier Medical Art.
, disturbing influence. Parts of the figure were drawn using pictures from Biorender.com and Servier Medical Art.
 , disturbing influence. Parts of the figure were drawn using pictures from Biorender.com and Servier Medical Art.
, disturbing influence. Parts of the figure were drawn using pictures from Biorender.com and Servier Medical Art.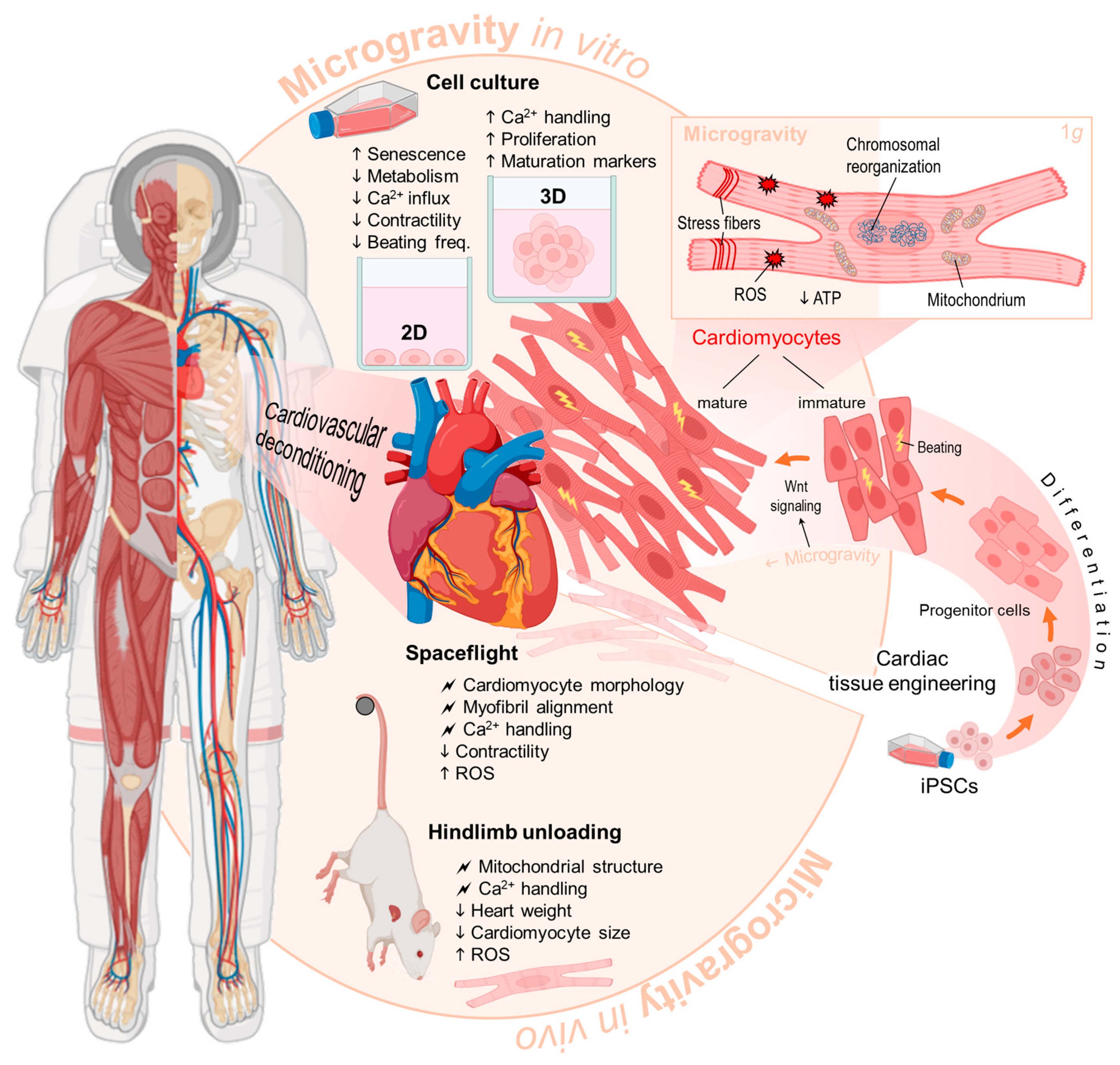
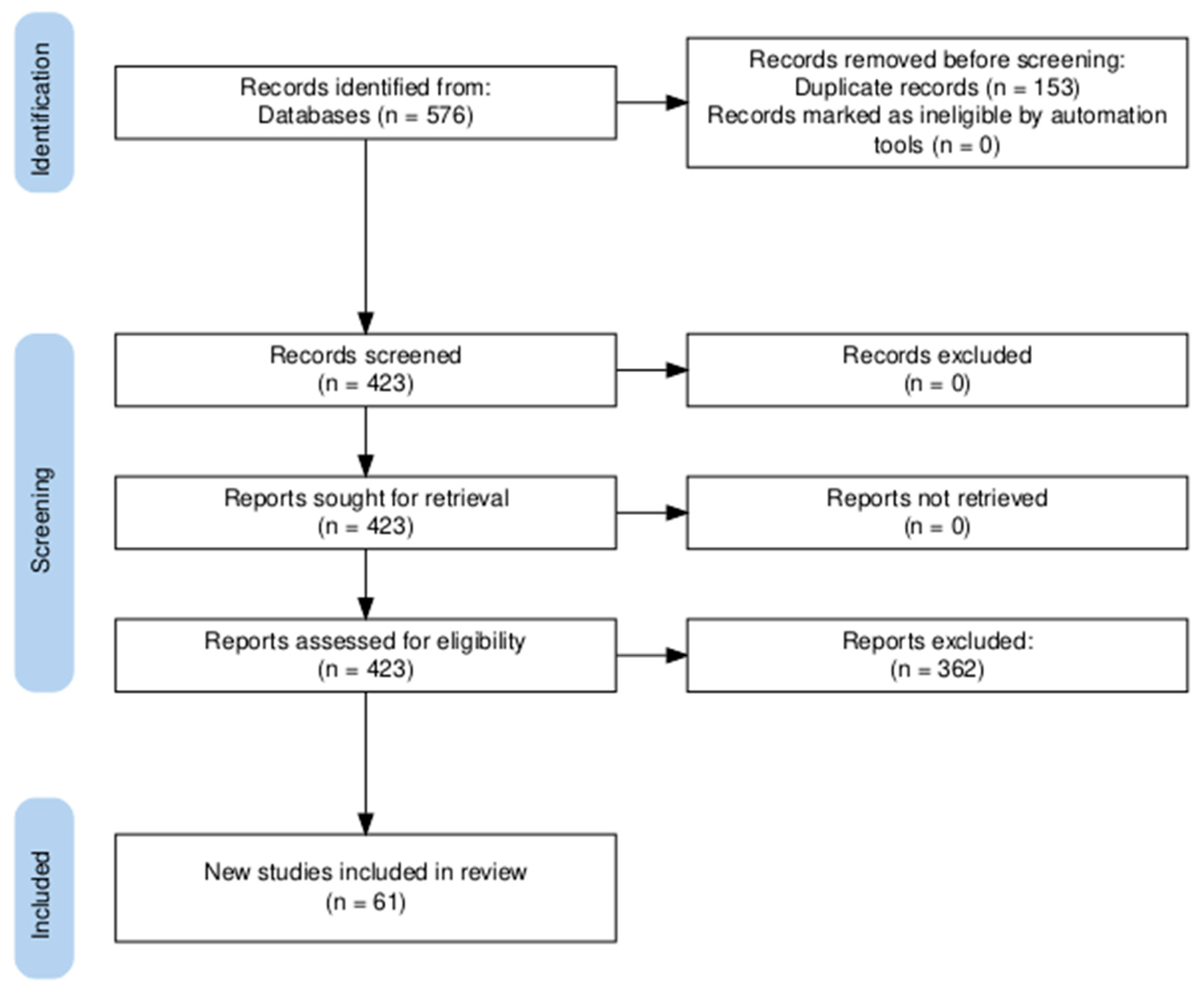
| Database | Search Strategy | No. of Articles |
|---|---|---|
| PUBMED | Cardiomyocytes AND (microgravity OR spaceflight) | 150 |
| Scopus | Cardiomyocytes AND (microgravity OR spaceflight) | 47 |
| Embase | Cardiomyocytes AND (microgravity OR spaceflight) | 336 |
| Google Scholar | allintitle: Cardiomyocytes microgravity OR spaceflight OR weightlessness | 23 |
| BIOSIS Previews | Cardiomyocytes AND (microgravity OR spaceflight) | 20 |
| Total | 576 |
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Studies involving cardiomyocytes exposed to microgravity (real or simulated) or spaceflight conditions | Studies not involving cardiomyocytes |
| Articles published in peer-reviewed journals | Studies not involving microgravity or spaceflight conditions |
| Studies reporting outcomes regarding cardiac cell structure, function, OMICs, and other relevant endpoints | Review articles, editorials, and commentaries |
| Publications written in English | Articles not written in English |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Torres, L.F.; Grimm, D.; Krüger, M. The Effects of Microgravity on the Structure and Function of Cardiomyocytes. Biomolecules 2025, 15, 1261. https://doi.org/10.3390/biom15091261
González-Torres LF, Grimm D, Krüger M. The Effects of Microgravity on the Structure and Function of Cardiomyocytes. Biomolecules. 2025; 15(9):1261. https://doi.org/10.3390/biom15091261
Chicago/Turabian StyleGonzález-Torres, Luis Fernando, Daniela Grimm, and Marcus Krüger. 2025. "The Effects of Microgravity on the Structure and Function of Cardiomyocytes" Biomolecules 15, no. 9: 1261. https://doi.org/10.3390/biom15091261
APA StyleGonzález-Torres, L. F., Grimm, D., & Krüger, M. (2025). The Effects of Microgravity on the Structure and Function of Cardiomyocytes. Biomolecules, 15(9), 1261. https://doi.org/10.3390/biom15091261







