Alpha-Synuclein mRNA Level Found Dependent on L444P Variant in Carriers and Gaucher Disease Patients on Enzyme Replacement Therapy
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Blood Samples
2.2. Biobanking
2.3. α-Synuclein mRNA Analysis
2.4. α-Synuclein Total and Oligomer Protein Concentration
2.5. Statistical Analysis
3. Results
3.1. Patients’ Characteristics
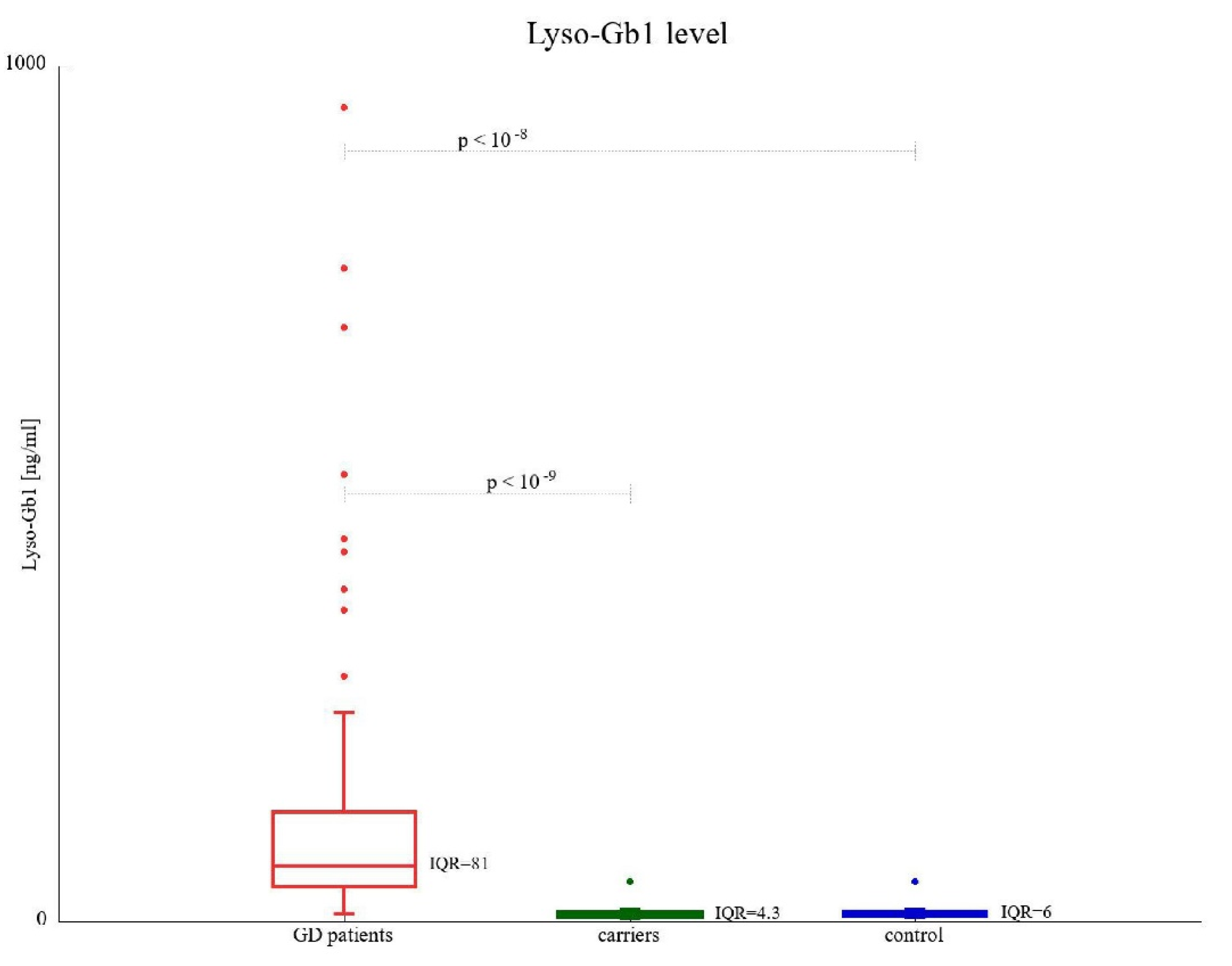
3.2. Lyso-Gb1 Concentration
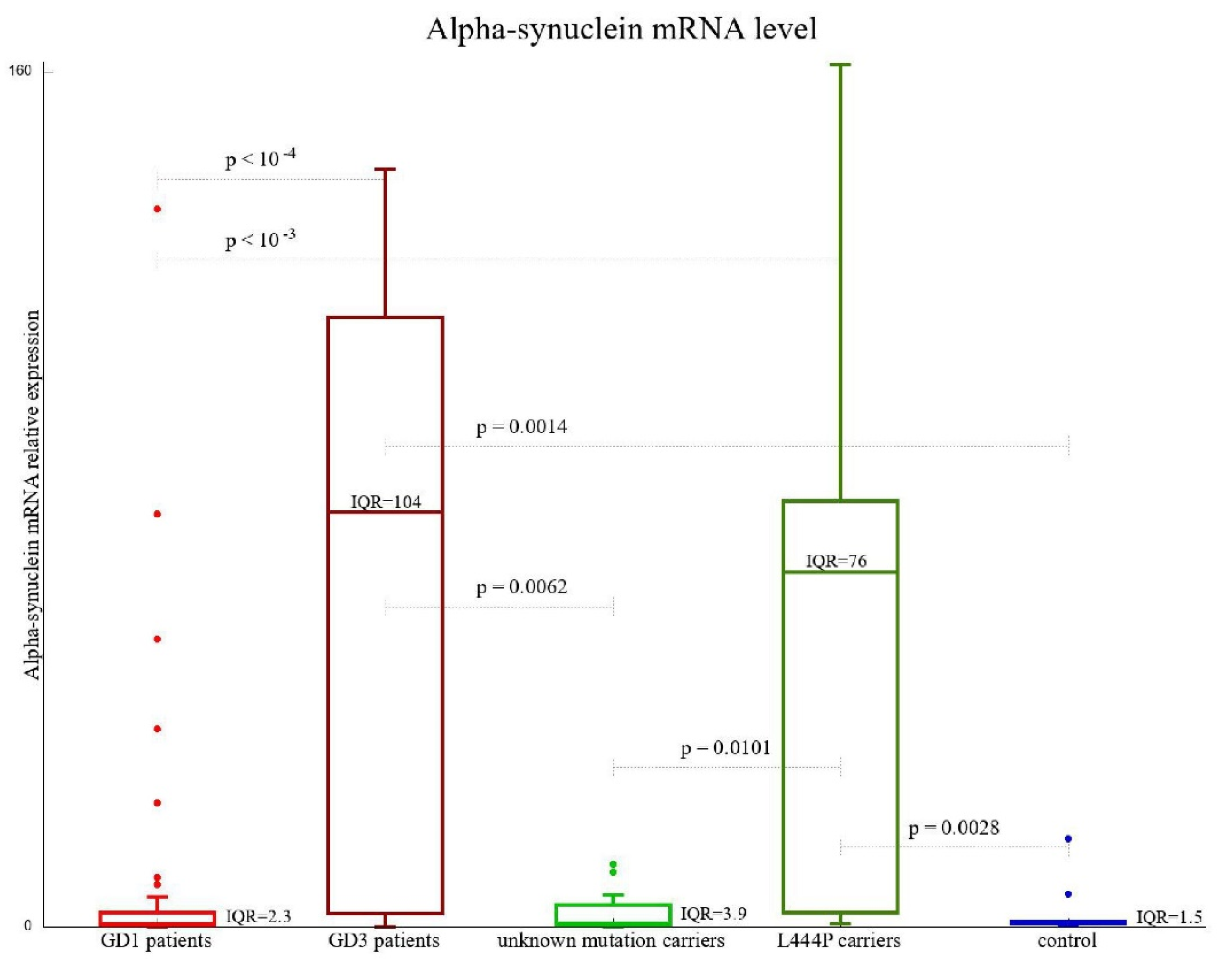
3.3. SNCA Gene Expression
3.4. α-SNCA Total Protein Level
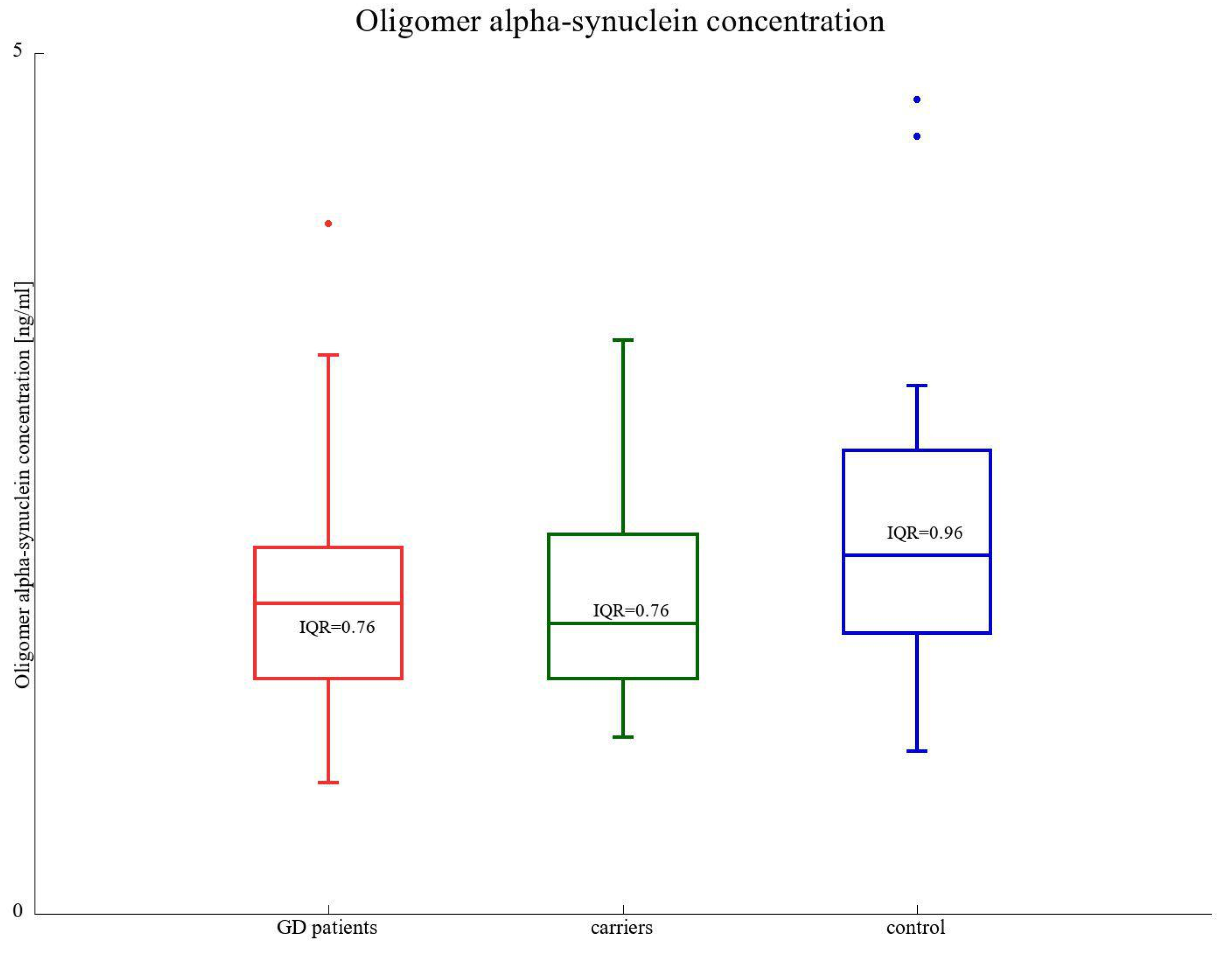
3.5. α-SNCA Oligomer Concentration
3.6. Correlations with Age
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Koprivica, V.; Stone, D.L.; Park, J.K.; Callahan, M.; Frisch, A.; Cohen, I.J.; Tayebi, N.; Sidransky, E. Analysis and Classification of 304 Mutant Alleles in Patients with Type 1 and Type 3 Gaucher Disease. Am. J. Hum. Genet. 2000, 66, 1777–1786. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, M.; Wilder, S.; Horowitz, Z.; Reiner, O.; Gelbart, T.; Beutler, E. The human glucocerebrosidase gene and pseudogene: Structure and evolution. Genomics 1989, 4, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Stirnemann, J.; Belmatoug, N.; Camou, F.; Serratrice, C.; Froissart, R.; Caillaud, C.; Levade, T.; Astudillo, L.; Serratrice, J.; Brassier, A.; et al. A Review of Gaucher Disease Pathophysiology, Clinical Presentation and Treatments. Int. J. Mol. Sci. 2017, 18, 441. [Google Scholar] [CrossRef] [PubMed]
- Brady, R.O.; Kanfer, J.N.; Bradley, R.M.; Shapiro, D. Demonstration of a deficiency of glucocerebroside-cleaving enzyme in Gaucher’s disease. J. Clin. Investig. 1966, 45, 1112–1115. [Google Scholar] [CrossRef] [PubMed]
- Sidransky, E. Gaucher disease: Insights from a rare Mendelian disorder. Discov. Med. 2012, 14, 273–281. [Google Scholar]
- Dekker, N.; van Dussen, L.; Hollak, C.E.M.; Overkleeft, H.; Scheij, S.; Ghauharali, K.; van Breemen, M.J.; Ferraz, M.J.; Groener, J.E.M.; Maas, M.; et al. Elevated plasma glucosylsphingosine in Gaucher disease: Relation to phenotype, storage cell markers, and therapeutic response. Blood 2011, 118, e118–e127. [Google Scholar] [CrossRef]
- Rolfs, A.; Giese, A.K.; Grittner, U.; Mascher, D.; Elstein, D.; Zimran, A.; Böttcher, T.; Lukas, J.; Hübner, R.; Gölnitz, U.; et al. Glucosylsphingosine Is a Highly Sensitive and Specific Biomarker for Primary Diagnostic and Follow-Up Monitoring in Gaucher Disease in a Non-Jewish, Caucasian Cohort of Gaucher Disease Patients. PLoS ONE 2013, 8, e79732. [Google Scholar] [CrossRef]
- Orvisky, E.; Park, J.K.; LaMarca, M.E.; Ginns, E.I.; Martin, B.M.; Tayebi, N.; Sidransky, E. Glucosylsphingosine accumulation in tissues from patients with Gaucher disease: Correlation with phenotype and genotype. Mol. Genet. Metab. 2002, 76, 262–270. [Google Scholar] [CrossRef]
- Revel-Vilk, S.; Fuller, M.; Zimran, A. Value of Glucosylsphingosine (Lyso-Gb1) as a Biomarker in Gaucher Disease: A Systematic Literature Review. Int. J. Mol. Sci. 2020, 21, 7159. [Google Scholar] [CrossRef]
- Brooks, D.J. The early diagnosis of Parkinson’s disease. Ann. Neurol. 1998, 44, S10–S18. [Google Scholar] [CrossRef]
- Bergman, H.; Deuschl, G. Pathophysiology of Parkinson’s disease: From clinical neurology to basic neuroscience and back. Mov. Disord. 2002, 17, S28–S40. [Google Scholar] [CrossRef] [PubMed]
- Wolters, E.C. Non-motor extranigral signs and symptoms in Parkinson’s disease. Park. Relat. Disord. 2009, 15, S6–S12. [Google Scholar] [CrossRef] [PubMed]
- Halliday, G.; Lees, A.; Stern, M. Milestones in Parkinson’s disease-Clinical and pathologic features. Mov. Disord. 2011, 26, 1015–1021. [Google Scholar] [CrossRef]
- Jellinger, K.A. Neuropathology of sporadic Parkinson’s disease: Evaluation and changes of concepts. Mov. Disord. 2012, 27, 8–30. [Google Scholar] [CrossRef]
- Buddhala, C.; Loftin, S.K.; Kuley, B.M.; Cairns, N.J.; Campbell, M.C.; Perlmutter, J.S.; Kotzbauer, P.T. Dopaminergic, serotonergic, and noradrenergic deficits in Parkinson disease. Ann. Clin. Transl. Neurol. 2015, 2, 949–959. [Google Scholar] [CrossRef] [PubMed]
- Singleton, A.; Hardy, J. The Evolution of Genetics: Alzheimer’s and Parkinson’s Diseases. Neuron 2016, 90, 1154–1163. [Google Scholar] [CrossRef]
- Moore, D.J.; West, A.B.; Dawson, V.L.; Dawson, T.M. Molecular pathophysiology of Parkinson’s disease. Ann. Rev. Neurosci. 2005, 28, 57–87. [Google Scholar] [CrossRef]
- Cookson, M.R. α-Synuclein and neuronal cell death. Mol. Neurodegener. 2009, 4, 4–9. [Google Scholar] [CrossRef]
- Beach, T.G.; Adler, C.H.; Sue, L.I.; Vedders, L.; Lue, L.; White Iii, C.L.; Akiyama, H.; Caviness, J.N.; Shill, H.A.; Sabbagh, M.N.; et al. Multi-organ distribution of phosphorylated α-synuclein histopathology in subjects with Lewy body disorders. Acta Neuropathol. 2010, 119, 689–702. [Google Scholar] [CrossRef]
- Coon, E.A.; Singer, W. Synucleinopathies. Continuum 2020, 26, 72–92. [Google Scholar] [CrossRef]
- Hallacli, E.; Kayatekin, C.; Nazeen, S.; Wang, X.H.; Sheinkopf, Z.; Sathyakumar, S.; Sarkar, S.; Jiang, X.; Dong, X.; Di Maio, R.; et al. The Parkinson’s disease protein alpha-synuclein is a modulator of processing bodies and mRNA stability. Cell 2022, 185, 2035–2056.e33. [Google Scholar] [CrossRef]
- Polymeropoulos, M.H.; Lavedan, C.; Leroy, E.; Ide, S.E.; Dehejia, A.; Dutra, A.; Dutra, A.; Pike, B.; Root, H.; Rubenstein, J.; et al. Mutation in the α-Synuclein Gene Identified in Families with Parkinson’s Disease. Science 1997, 276, 2045–2047. [Google Scholar] [CrossRef] [PubMed]
- Braak, H.; Del Tredici, K.; Rüb, U.; de Vos, R.A.I.; Jansen Steur, E.N.; Braak, E. Staging of brain pathology related to sporadic Par-kinson’s disease. Neurobiol. Aging 2003, 24, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, U.; Singh, S.; Pal, S.; Agrawal, B.K.; Saini, R.V.; Chakrabarti, S. Alpha-synuclein as a Biomarker of Parkinson’s Disease: Good, but Not Good Enough. Front. Aging Neurosci. 2021, 13, 702639. [Google Scholar] [CrossRef] [PubMed]
- Weinreb, P.H.; Zhen, W.; Poon, A.W.; Conway, K.A.; Lansbury, P.T. NACP, a protein implicated in Alzheimer’s disease and learning, is natively unfolded. Biochemistry 1996, 35, 13709–13715. [Google Scholar] [CrossRef]
- Pfefferkorn, C.M.; Jiang, Z.; Lee, J.C. Biophysics of α-synuclein membrane interactions. Biochim. Biophys. Acta 2012, 1818, 162–171. [Google Scholar] [CrossRef]
- Uversky, V.N. Amyloidogenesis of Natively Unfolded Proteins. Curr. Alzheimer Res. 2008, 5, 260–287. [Google Scholar] [CrossRef]
- Rosenbloom, B.; Balwani, M.; Bronstein, J.M.; Kolodny, E.; Sathe, S.; Gwosdow, A.R.; Taylor, J.S.; Cole, J.A.; Zimran, A.; Weinreb, N.J. The incidence of Parkinsonism in patients with type 1 Gaucher disease: Data from the ICGG Gaucher Registry. Blood Cells Mol. Dis. 2011, 46, 95–102. [Google Scholar] [CrossRef]
- Duran, R.; Mencacci, N.E.; Angeli, A.V.; Shoai, M.; Deas, E.; Houlden, H.; Mehta, A.; Hughes, D.; Cox, T.M.; Deegan, P.; et al. The glucocerobrosidase E326K variant predisposes to Parkinson’s disease, but does not cause Gaucher’s disease. Mov. Disord. 2012, 28, 232–236. [Google Scholar] [CrossRef]
- Tylki-Szymańska, A.; Vellodi, A.; El-Beshlawy, A.; Cole, J.A.; Kolodny, E. Neuronopathic Gaucher disease: Demographic and clinical features of 131 patients enrolled in the International Collaborative Gaucher Group Neurological Outcomes Subregistry. J. Inherit. Metab. Dis. 2010, 33, 339–346. [Google Scholar] [CrossRef]
- Halperin, A.; Elstein, D.; Zimran, A. Increased incidence of Parkinson disease among relatives of patients with Gaucher disease. Blood Cells Mol. Dis. 2006, 36, 426–428. [Google Scholar] [CrossRef] [PubMed]
- Maor, G.; Filocamo, M.; Horowitz, M. ITCH regulates degradation of mutant glucocerebrosidase: Implications to Gaucher disease. Hum. Mol. Genet. 2013, 22, 1316–1327. [Google Scholar] [CrossRef]
- Taguchi, Y.V.; Liu, J.; Ruan, J.; Pacheco, J.; Zhang, X.; Abbasi, J.; Keutzer, J.; Mistry, P.K.; Chandra, S.S. Glucosylsphingosine promotes alpha-synuclein pathology in mutant GBA-associated Parkinson’s disease. J. Neurosci. 2017, 37, 9617–9631. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.-H.; Xu, K.; Sun, Y.; Liou, B.; Quinn, B.; Li, R.-H.; Xue, L.; Zhang, W.; Setchell, K.D.; Witte, D.; et al. Multiple pathogenic proteins implicated in neuronopathic Gaucher disease mice. Hum. Mol. Genet. 2014, 23, 3943–3957. [Google Scholar] [CrossRef] [PubMed]
- Aflaki, E.; Borger, D.K.; Moaven, N.; Stubblefield, B.K.; Rogers, S.A.; Patnaik, S.; Schoenen, F.J.; Westbroek, W.; Zheng, W.; Sullivan, P.; et al. A new glucocerebrosidase chaperone reduces alpha-synuclein and glycolipid levels in iPSC-derived dopaminergic neurons from patients with Gaucher disease and Parkinsonism. J. Neurosci. 2016, 36, 7441–7452. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple hypothesis testing. J. R. Stat. Soc. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Dinur, T.; Zimran, A.; Becker-Cohen, M.; Arkadir, D.; Cozma, C.; Hovakimyan, M.; Oppermann, S.; Demuth, L.; Rolfs, A.; Revel-Vilk, S. Long Term Follow-Up of 103 Untreated Adult Patients with Type 1 Gaucher Disease. J. Clin. Med. 2019, 8, 1662. [Google Scholar] [CrossRef]
- Do, J.; McKinney, C.; Sharma, P.; Sidransky, E. Glucocerebrosidase and its relevance to Parkinson disease. Mol. Neurodegener. 2019, 14, 36. [Google Scholar] [CrossRef]
- Sidransky, E.; Nalls, M.A.; Aasly, J.O.; Aharon-Peretz, J.; Annesi, G.; Barbosa, E.R.; Bar-Shira, A.; Berg, D.; Bras, J.; Brice, A.; et al. Multicenter analysis of glucocerebrosidase mutations in Parkinson’s disease. N. Engl. J. Med. 2009, 361, 1651–1661. [Google Scholar] [CrossRef]
- Stojkovska, I.; Krainc, D.; Mazzulli, J.R. Molecular mechanisms of α-synuclein and GBA1 in Parkinson’s disease. Cell Tissue Res. 2018, 373, 51–60. [Google Scholar] [CrossRef]
- Argyriou, A.; Dermentzaki, G.; Papasilekas, T.; Moraitou, M.; Stamboulis, E.; Vekrellis, K.; Michelakakis, H.; Stefanis, L. Increased dimerization of alpha-synuclein in erythrocytes in Gaucher disease and aging. Neurosci. Lett. 2012, 528, 205–209. [Google Scholar] [CrossRef]
- Pchelina, S.; Nuzhnyi, E.; Emelyanov, A.; Boukina, T.; Usenko, T.; Nikolaev, M.; Salogub, G.; Yakimovskii, A.; Zakharova, E.Y. Increased plasma oligomeric alpha-synuclein in patients with lysosomal storage diseases. Neurosci. Lett. 2014, 583, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Mazzulli, J.R.; Xu, Y.H.; Sun, Y.; Knight, A.L.; McLean, P.J.; Caldwell, G.A.; Sidransky, E.; Grabowski, G.A.; Krainc, D. Gaucher disease glucocerebrosidase and α-synuclein form a bidirectional pathogenic loop in synucleinopathies. Cell 2011, 146, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Lerche, S.; Wurster, I.; Roeben, B.; Zimmermann, M.; Riebenbauer, B.; Deuschle, C.; Hauser, A.K.; Schulte, C.; Berg, D.; Maetzler, W.; et al. Parkinson’s Disease: Glucocerebrosidase 1 Mutation Severity Is Associated with CSF Alpha-synuclein Profiles. Mov. Disord. 2020, 35, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Chiba-Falek, O.; Lopez, G.J.; Nussbaum, R.L. Levels of alpha-synuclein mRNA in sporadic Parkinson disease patients. Mov. Disord. 2006, 21, 1703–1708. [Google Scholar] [CrossRef] [PubMed]
- Bras, J.; Paisan-Ruiz, C.; Guerreiro, R.; Ribeiro, M.H.; Morgadinho, A.; Januario, C.; Sidransky, E.; Oliveira, C.; Singleton, A. Complete screening for glucocerebrosidase mutations in Parkinson disease patients from Portugal. Neurobiol. Aging 2009, 30, 1515–1517. [Google Scholar] [CrossRef] [PubMed]
- Kalinderi, K.; Bostantjopoulou, S.; Paisan-Ruiz, C.; Katsarou, Z.; Hardy, J.; Fidani, L. Complete screening for glucocerebrosidase mutations in Parkinson disease patients from Greece. Neurosci. Lett. 2009, 452, 87–89. [Google Scholar] [CrossRef]
- Mao, X.-Y.; Burgunder, J.-M.; Zhang, Z.-J.; An, X.-K.; Zhang, J.-H.; Yang, Y.; Li, T.; Wang, Y.-C.; Chang, X.-L.; Peng, R. Association between GBA L444P mutation and sporadic Parkinson’s disease from Mainland China. Neurosci. Lett. 2010, 469, 256–259. [Google Scholar] [CrossRef]
- Mitsui, J.; Mizuta, I.; Toyoda, A.; Ashida, R.; Takahashi, Y.; Goto, J.; Fukuda, Y.; Date, H.; Iwata, A.; Yamamoto, M.; et al. Mutations for Gaucher Disease Confer High Susceptibility to Parkinson Disease. Arch. Neurol. 2009, 66, 571–576. [Google Scholar] [CrossRef]
- Sato, C.; Morgan, A.; Lang, A.E.; Salehi-Rad, S.; Kawarai, T.; Meng, Y.; Ray, P.N.; Farrer, L.A.; George-Hyslop, P.S.; Rogaeva, E. Analysis of the glucocerebrosidase gene in Parkinson’s disease. Mov. Disord. 2005, 20, 367–370. [Google Scholar] [CrossRef]
- Sun, Q.-Y.; Guo, J.-F.; Wang, L.; Yu, R.-H.; Zuo, X.; Yao, L.-Y.; Pan, Q.; Xia, K.; Tang, B.-S. Glucocerebrosidase Gene L444P mutation is a risk factor for Parkinson’s disease in Chinese population. Mov. Disord. 2010, 25, 1005–1011. [Google Scholar] [CrossRef]
- Tan, E.-K.; Tong, J.; Fook-Chong, S.; Yih, Y.; Wong, M.-C.; Pavanni, R.; Zhao, Y. Glucocerebrosidase Mutations and Risk of Parkinson Disease in Chinese Patients. Arch. Neurol. 2007, 64, 1056–1058. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-R.; Chen, C.-M.; Chao, C.-Y.; Ro, L.-S.; Lyu, R.-K.; Chang, K.-H.; Lee-Chen, G.-J. Glucocerebrosidase gene mutation is a risk factor for early onset of Parkinson disease among Taiwanese. J. Neurol. Neurosurg. Psychiatry 2007, 78, 977–979. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, S.G.; Eblan, M.J.; Gutti, U.; Hruska, K.S.; Stubblefield, B.K.; Goker-Alpan, O.; LaMarca, M.E.; Sidransky, E. Glucocerebrosidase mutations in Chinese subjects from Taiwan with sporadic Parkinson disease. Mol. Genet. Metab. 2007, 91, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Neumann, J.; Bras, J.; Deas, E.; O’Sullivan, S.S.; Parkkinen, L.; Lachmann, R.H.; Li, A.; Holton, J.; Guerreiro, R.; Paudel, R.; et al. Glucocerebrosidase mutations in clinical and pathologically proven Parkinson’s disease. Brain 2009, 132, 1783–1794. [Google Scholar] [CrossRef]
- Spitz, M.; Rozenberg, R.; Pereira Lda, V.; Reis Barbosa, E. Association between Parkinson’s disease and glucocerebrosidase mutations in Brazil. Parkinsonism Relat. Disord. 2008, 14, 58–62. [Google Scholar] [CrossRef]
- De Marco, E.V.; Annesi, G.; Tarantino, P.; Rocca, F.E.; Provenzano, G.; Civitelli, D.; Cirò Candiano, I.C.; Annesi, F.; Carrideo, S.; Condino, F.; et al. Glucocerebrosidase gene mutations are associated with Parkinson’s disease in southern Italy. Mov. Disord. 2008, 23, 460–463. [Google Scholar] [CrossRef]
- Becker-Cohen, M.; Zimran, A.; Dinur, T.; Tiomkin, M.; Cozma, C.; Rolfs, A.; Arkadir, D.; Shulman, E.; Manor, O.; Paltiel, O.; et al. A Comprehensive Assessment of Qualitative and Quantitative Prodromal Par-kinsonian Features in Carriers of Gaucher Disease-Identifying Those at the Greatest Risk. Int. J. Mol. Sci. 2022, 23, 12211. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, L.; Xiong, J.; Zhang, X.; Chen, Z.; Yu, L.; Chen, C.; Huang, J.; Zhang, Z.; Mohmed, A.A.; et al. Glucocerebrosidase L444P mutation confers genetic risk for Parkinson’s disease in central China. Behav. Brain Funct. 2012, 8, 57. [Google Scholar] [CrossRef]
- Barbour, R.; Kling, K.; Anderson, J.P.; Banducci, K.; Cole, T.; Diep, L.; Fox, M.; Goldstein, J.M.; Soriano, F.; Seubert, P.; et al. Red Blood Cells Are the Major Source of Alpha-Synuclein in Blood. Neurodegener. Dis. 2008, 5, 55–59. [Google Scholar] [CrossRef]
- Zanzoni, A.; Marchese, D.; Agostini, F.; Bolognesi, B.; Cirillo, D.; Botta-Orfila, M.; Livi, C.M.; Rodriguez-Mulero, S.; Tartaglia, G.G. Principles of self-organization in biological pathways: A hypothesis on the autogenous association of alpha-synuclein. Nucleic Acids Res. 2013, 41, 9987–9998. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.P.; Kim, D.; Kim, S.; Kim, S.; Karuppagounder, S.S.; Kwon, S.-H.; Lee, S.; Kam, T.-I.; Lee, S.; Ham, S.; et al. α-Synuclein accumulation and GBA deficiency due to L444P GBA mutation contributes to MPTP-induced parkinsonism. Mol. Neurodegener. 2018, 13, 1. [Google Scholar] [CrossRef] [PubMed]

| Number of Patients | Common Variant Name | DNA Nucleotide Change | Protein Change |
|---|---|---|---|
| 28 | N370S/L444P | c.1226A>G c.1448T>C | p.Asn409Ser p.Leu483Pro |
| 6 | N370S/ others | c.1226A>G | p.Asn409Ser |
| 2 | N370S/N370S | c.1226A>G | p.Asn409Ser |
| 2 | G377S/G377S | c.1246G>A | p.Gly416Ser |
| 1 | N370S/ G377S | c.1226A>G c.1246G>A | p.Asn409Ser p.Gly416Ser |
| 1 | N370S/84GG | c.1226A>G c.84dupG | p.Asn409Ser p.Leu29AlafsTer18 |
| 1 | N370S/R496H | c.1226A>G c.1604G>A | p.Asn409Ser p.Arg535His |
| 1 | R433S/R433S | c.1416A>T | p.Arg444Ser |
| 1 | D438N/R87W | c.1312G>A c.259C>T | p.Asp438Asn p.Arg87Trp |
| 1 | D448G/R202X | c.1343A>G c.604C>T | p.Asp448Gly p.Arg202X |
| 1 | R48W/84GG | c.259C>T c.84dupG | p.Arg87Trp p.Leu29AlafsTer18 |
| 1 | R48W/R48W | c.259C>T | p.Arg87Trp |
| 1 | 84GG/ Other | c.84dupG | p.Leu29AlafsTer18 |
| Healthy Controls | GBA1 Mutation Carriers | GD1 | GD3 | |
|---|---|---|---|---|
| n | 16 | 19 | 47 | 18 |
| Sex, F/M | 10/6 | 14/5 | 28/19 | 12/8 |
| Age in years, median (range) | 44.5 | 52 (13–64) | 40 (9–71) | 30 (22–62) |
| Lyso-Gb1 (ng/mL), median (range) | 8.49 (4.82–46.73) | 7.51 (4.26–46.73) | 71.5 (22.4–950.9) | 64.55 (9.14–693.8) |
| SNCA mRNA level (2−ΔcT), median (range) | 0.83 (0.08–16.38) | 4.16 (0.02–161.42) | 0.47 (0.01–134.42) | 77.48 (0.04–141.72) |
| Total α-SNCA concentration (ng/mL), median (range) | 1.34 (0.16–37.48) | 0.53 (0.16–21.76) | 0.16 (0.16–20) | 0.92 (0.16–20) |
| Oligomer α-SNCA concentration (ng/mL), median (range) | 2.08 (0.96–4.73) | 1.69 (1.03–3.33) | 1.76 (0.76–4.01) | 1.857(0.76–3.03) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dubiela, P.; Szymańska-Rożek, P.; Eljaszewicz, A.; Lipiński, P.; Hasiński, P.; Giersz, D.; Walewska, A.; Tynecka, M.; Moniuszko, M.; Tylki-Szymańska, A. Alpha-Synuclein mRNA Level Found Dependent on L444P Variant in Carriers and Gaucher Disease Patients on Enzyme Replacement Therapy. Biomolecules 2023, 13, 644. https://doi.org/10.3390/biom13040644
Dubiela P, Szymańska-Rożek P, Eljaszewicz A, Lipiński P, Hasiński P, Giersz D, Walewska A, Tynecka M, Moniuszko M, Tylki-Szymańska A. Alpha-Synuclein mRNA Level Found Dependent on L444P Variant in Carriers and Gaucher Disease Patients on Enzyme Replacement Therapy. Biomolecules. 2023; 13(4):644. https://doi.org/10.3390/biom13040644
Chicago/Turabian StyleDubiela, Paweł, Paulina Szymańska-Rożek, Andrzej Eljaszewicz, Patryk Lipiński, Piotr Hasiński, Dorota Giersz, Alicja Walewska, Marlena Tynecka, Marcin Moniuszko, and Anna Tylki-Szymańska. 2023. "Alpha-Synuclein mRNA Level Found Dependent on L444P Variant in Carriers and Gaucher Disease Patients on Enzyme Replacement Therapy" Biomolecules 13, no. 4: 644. https://doi.org/10.3390/biom13040644
APA StyleDubiela, P., Szymańska-Rożek, P., Eljaszewicz, A., Lipiński, P., Hasiński, P., Giersz, D., Walewska, A., Tynecka, M., Moniuszko, M., & Tylki-Szymańska, A. (2023). Alpha-Synuclein mRNA Level Found Dependent on L444P Variant in Carriers and Gaucher Disease Patients on Enzyme Replacement Therapy. Biomolecules, 13(4), 644. https://doi.org/10.3390/biom13040644







