Biological Evaluation of the Osteoinductive Potential of Dry Teeth after Chemical Demineralization Treatment Using the Tooth Transformer Device
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Reagents
2.3. Western Blot Analysis
2.4. Measurement of BMP-2 and Collagen Type 1
2.5. Statistical Analysis
3. Results
3.1. Evaluation of Type I Collagen Content and BMP-2 Protein
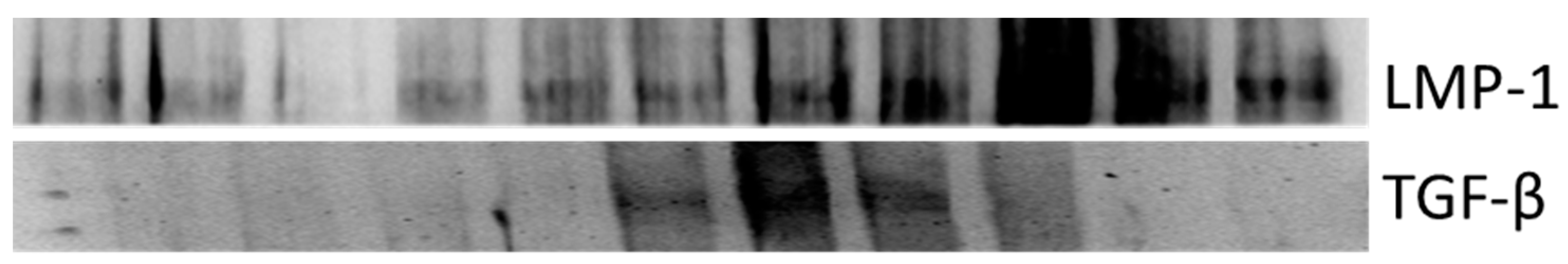
3.2. Evaluation of Mineralization Protein LIM-1 (LMP-1) and Transforming Growth Factor-β (TGF-β)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Fiske, J.; Davis, D.M.; Frances, C.; Gelbier, S. The emotional effects of tooth loss in edentulous people. Br. Dent. J. 1998, 184, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Tallgren, A. The continuing reduction of the residual alveolar ridges in complete denture wearers: A mixed-longitudinal study covering 25 years. J. Prosthet. Dent. 1972, 27, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Chappuis, V.; Araújo, M.G.; Buser, D. Clinical relevance of dimensional bone and soft tissue alterations post-extraction in esthetic sites. Periodontol. 2000 2017, 73, 73–83. [Google Scholar] [CrossRef]
- Johnson, K. A study of the dimensional changes occurring in the maxilla following tooth extraction. Aust. Dent. J. 1969, 14, 241–244. [Google Scholar] [CrossRef]
- Minetti, E.; Giacometti, E.; Gambardella, U.; Contessi, M.; Ballini, A.; Marenzi, G.; Celko, M.; Mastrangelo, F. Alveolar Socket Preservation with Different Autologous Graft Materials: Preliminary Results of a Multicenter Pilot Study in Human. Materials 2020, 13, 1153. [Google Scholar] [CrossRef] [PubMed]
- Mastrangelo, F.; Scacco, S.; Ballini, A.; Quaresima, R.; Gnoni, A.; De Vito, D.; Scarano, A.; Dipalma, G.; Gargiulo Isacco, C.; Cantore, S.; et al. A pilot study of human mesenchymal stem cells from visceral and sub-cutaneous fat tissue and their differentiation to osteogenic phenotype. Eur Rev Med Pharmacol Sci. 2019, 23, 2924–2934. [Google Scholar] [PubMed]
- Araújo, M.G.; Lindhe, J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J. Clin. Periodontol. 2005, 32, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Van der Weijden, F.; Dell’Acqua, F.; Slot, D.E. Alveolar bone dimensional changes of post-extraction sockets in humans: A systematic review. J. Clin. Periodontol. 2009, 36, 1048–1058. [Google Scholar] [CrossRef]
- Wang, B.; Feng, C.; Liu, Y.; Mi, F.; Dong, J. Recent advances in biofunctional guided bone regeneration materials for repairing defective alveolar and maxillofacial bone: A review. Jpn. Dent. Sci. Rev. 2022, 58, 233–248. [Google Scholar] [CrossRef]
- Bernard, G.W. Healing and repair of osseous defects. Dent. Clin. N. Am. 1991, 35, 469–477. [Google Scholar] [CrossRef]
- Cushing, M. Autogenous red marrow grafts: Their potential for induction of osteogenesis. J. Periodontol. 1969, 40, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Gamradt, S.C.; Lieberman, J.R. Bone graft for revision hip arthroplasty: Biology and future applications. Clin. Orthop. Relat. Res. 2003, 417, 183–194. [Google Scholar] [CrossRef] [PubMed]
- LeGeros, R.Z. Properties of osteoconductive biomaterials: Calcium phosphates. Clin. Orthop. Relat. Res. 2002, 395, 81–98. [Google Scholar] [CrossRef] [PubMed]
- Mastrangelo, F. New Implant Materials. Materials 2023, 16, 4525. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.M.; Cortez, A.L.; Moreira, R.W.; Mazzonetto, R. Complications of intraoral donor site for bone grafting prior to implant placement. Implant. Dent. 2006, 15, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Lee, J.K.; Kim, K.; Um, I.; Murata, M. Healing mechanism and clinical application of autogenous tooth bone graft material. Adv. Biomater. Sci. Biomed. Appl. 2013, 405–436. [Google Scholar] [CrossRef]
- Bono, N.; Tarsini, P.; Candiani, G. Demineralized dentin and enamel matrices as suitable substrates for bone regeneration. J. Appl. Biomater. Funct. Mater. 2017, 15, e236–e243. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Lee, J.; Kim, K.W.; Um, I.W.; Murata, M.; Ito, K. Analysis of Organic Components and Osteoinductivity in Autogenous Tooth Bone Graft Material. J. Korean Assoc. Maxillofac. Plast. Reconstr. Surg. 2013, 35, 353–359. [Google Scholar] [CrossRef]
- Boden, S.D.; Liu, Y.; Hair, G.A.; Helms, J.A.; Hu, D.; Racine, M.; Nanes, M.S.; Titus, L. LMP-1, a LIM-domain protein, mediates BMP-6 effects on bone formation. Endocrinology 1998, 139, 5125–5134. [Google Scholar] [CrossRef]
- Bessho, K.; Tanaka, N.; Matsumoto, J.; Tagawa, T.; Murata, M. Human dentin-matrix-derived bone morphogenetic protein. J. Dent. Res. 1991, 70, 171–175. [Google Scholar] [CrossRef]
- Chen, G.; Deng, C.; Li, Y.P. TGF-β and BMP signaling in osteoblast differentiation and bone formation. Int. J. Biol. Sci. 2012, 8, 272–288. [Google Scholar] [CrossRef] [PubMed]
- Grawish, M.E.; Grawish, L.M.; Grawish, H.M.; Grawish, M.M.; Holiel, A.A.; Sultan, N.; El-Negoly, S.A. Demineralized Dentin Matrix for Dental and Alveolar Bone Tissues Regeneration: An Innovative Scope Review. Tissue Eng. Regen. Med. 2022, 19, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W. Bone Induction by Demineralized Dentin Matrix in Nude Mouse Muscles. Maxillofac. Plast. Reconstr. Surg. 2014, 36, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Minetti, E.; Corbella, S.; Taschieri, S.; Canullo, L. Tooth as graft material: Histologic study. Clin. Implant. Dent. Relat. Res. 2022, 24, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Minetti, E.; Celko, M.; Contessi, M.; Carini, F.; Gambardella, U.; Giacometti, E.; Santillana, J.; Beca Campoy, T.; Schmitz, J.H.; Libertucci, M.; et al. Implants Survival Rate in Regenerated Sites with Innovative Graft Biomaterials: 1 Year Follow-Up. Materials 2021, 14, 5292. [Google Scholar] [CrossRef]
- Inchingolo, A.M.; Patano, A.; Di Pede, C.; Inchingolo, A.D.; Palmieri, G.; de Ruvo, E.; Campanelli, M.; Buongiorno, S.; Carpentiere, V.; Piras, F.; et al. Autologous Tooth Graft: Innovative Biomaterial for Bone Regeneration. Tooth Transformer® and the Role of Microbiota in Regenerative Dentistry. A Systematic Review. J. Funct. Biomater. 2023, 14, 132. [Google Scholar] [CrossRef] [PubMed]
- Patruno, A.; Pesce, M.; Grilli, A.; Speranza, L.; Franceschelli, S.; De Lutiis, M.A.; Vianale, G.; Costantini, E.; Amerio, P.; Muraro, R.; et al. mTOR Activation by PI3K/Akt and ERK Signaling in Short ELF-EMF Exposed Human Keratinocytes. PLoS ONE 2015, 10, e0139644. [Google Scholar] [CrossRef] [PubMed]
- Nasr, H.F.; Aichelmann-Reidy, M.E.; Yukna, R.A. Bone and bone substitutes. Periodontol. 2000 1999, 19, 74–86. [Google Scholar] [CrossRef]
- Chavda, S.; Levin, L. Human Studies of Vertical and Horizontal Alveolar Ridge Augmentation Comparing Different Types of Bone Graft Materials: A Systematic Review. J. Oral Implantol. 2018, 44, 74–84. [Google Scholar] [CrossRef]
- Goldberg, V.M.; Stevenson, S. Natural history of autografts and allografts. Clin. Orthop. Relat. Res. 1987, 225, 7–16. [Google Scholar] [CrossRef]
- Murata, M.; Akazawa, T.; Mitsugi, M.; Um, I.W.; Kim, K.W.; Kim, Y.K. Human Dentin as Novel Biomaterial for Bone Regeneration. Biomater.-Phys. Chem. [CrossRef]
- Bhaskar, S.N.; Orban, B.J. Orban’s Oral Histology and Embryology, 11th ed.; Mosby Year Book: Saint Louis, MO, USA, 1991. [Google Scholar]
- Urist, M.R.; Strates, B.S. Bone morphogenetic protein. J. Dent. Res. 1971, 50, 1392–1406. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Urist, M.R. Bovine tooth-derived bone morphogenetic protein. J. Dent. Res. 1989, 68, 1069–1074. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Kim, S.G.; Byeon, J.H.; Lee, H.J.; Um, I.U.; Lim, S.C.; Kim, S.Y. Development of a novel bone grafting material using autogenous teeth. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 109, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Li, X.; Qi, Y.; Ma, X.; Qiao, S.; Cai, H.; Zhao, B.C.; Jiang, H.B.; Lee, E.S. Comparison of Autogenous Tooth Materials and Other Bone Grafts. Tissue Eng. Regen. Med. 2021, 18, 327–341. [Google Scholar] [CrossRef]
- Gual-Vaqués, P.; Polis-Yanes, C.; Estrugo-Devesa, A.; Ayuso-Montero, R.; Mari-Roig, A.; López-López, J. Autogenous teeth used for bone grafting: A systematic review. Med. Oral Patol. Oral Cir. Bucal 2018, 23, e112–e119. [Google Scholar] [CrossRef] [PubMed]
- Reis-Filho, C.R.; Silva, E.R.; Martins, A.B.; Pessoa, F.F.; Gomes, P.V.; de Araújo, M.S.; Miziara, M.N.; Alves, J.B. Demineralised human dentine matrix stimulates the expression of VEGF and accelerates the bone repair in tooth sockets of rats. Arch. Oral Biol. 2012, 57, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Gomes, M.F.; da Silva dos Anjos, M.J.; de Oliveira Nogueira, T.; Catanzaro Guimarães, S.A. Autogenous demineralized dentin matrix for tissue engineering applications: Radiographic and histomorphometric studies. Int. J. Oral Maxillofac. Implant. 2002, 17, 488–497. [Google Scholar]
- Gomes, M.F.; Abreu, P.P.; Morosolli, A.R.; Araújo, M.M.; Goulart, M.d. Densitometric analysis of the autogenous demineralized dentin matrix on the dental socket wound healing process in humans. Braz. Oral Res. 2006, 20, 324–330. [Google Scholar] [CrossRef]
- Pang, K.M.; Um, I.W.; Kim, Y.K.; Woo, J.M.; Kim, S.M.; Lee, J.H. Autogenous demineralized dentin matrix from extracted tooth for the augmentation of alveolar bone defect: A prospective randomized clinical trial in comparison with anorganic bovine bone. Clin. Oral Implant. Res. 2017, 28, 809–815. [Google Scholar] [CrossRef]
- Kim, Y.K.; Kim, S.G.; Bae, J.H.; Um, I.W.; Oh, J.S.; Jeong, K.I. Guided bone regeneration using autogenous tooth bone graft in implant therapy: Case series. Implant. Dent. 2014, 23, 138–143. [Google Scholar] [CrossRef]
- Minamizato, T.; Koga, T.; Takashi, I.; Nakatani, Y.; Umebayashi, M.; Sumita, Y.; Ikeda, T.; Asahina, I. Clinical application of autogenous partially demineralized dentin matrix prepared immediately after extraction for alveolar bone regeneration in implant dentistry: A pilot study. Int. J. Oral Maxillofac. Surg. 2018, 47, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Dłucik, R.; Orzechowska-Wylęgała, B.; Dłucik, D.; Puzzolo, D.; Santoro, G.; Micali, A.; Testagrossa, B.; Acri, G. Comparison of clinical efficacy of three different dentin matrix biomaterials obtained from different devices. Expert Rev. Med. Devices 2023, 20, 313–327. [Google Scholar] [CrossRef] [PubMed]
- Pietrzak, W.S.; Ali, S.N.; Chitturi, D.; Jacob, M.; Woodell-May, J. BMP depletion occurs during prolonged acid demineralization of bone: Characterization and implications for graft preparation. Cell Tissue Bank. 2009, 12, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Zeng, J.; Yao, A.; Tu, Q.; Li, J.; Yan, L.; Tang, Z. BMP2-loaded hollow hydroxyapatite microspheres exhibit enhanced osteoinduction and osteogenicity in large bone defects. Int J Nanomed. 2015, 10, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Ren, B.; Betz, V.M.; Thirion, C.; Salomon, M.; Jansson, V.; Müller, P.E.; Betz, O.B. Osteoinduction within BMP-2 transduced muscle tissue fragments with and without a fascia layer: Implications for bone tissue engineering. Gene Ther. 2019, 26, 16–28. [Google Scholar] [CrossRef]
- Pan, H.; Li, X.; Wang, J.; Zhang, K.; Yang, H.; Li, Z.; Zheng, Z.; Liu, H. LIM Mineralization Protein-1 Enhances Bone Morphogenetic Protein-2-Mediated Osteogenesis Through Activation of ERK1/2 MAPK Pathway and Upregulation of Runx2 Transactivity. J Bone Miner Res 2015, 30, 1523–1535. [Google Scholar] [CrossRef]
- Franceschi, R.T.; Xiao, G.; Jiang, D.; Gopalakrishnan, R.; Yang, S.; Reith, E. Multiple signaling pathways converge on the Cbfa1/Runx2 transcription factor to regulate osteoblast differentiation. Connect Tissue Res 2003, 44 (Suppl. S1), 109–116. [Google Scholar] [CrossRef]

| Title 1 | pg/mL |
|---|---|
| COL-I | 3.64 ± 1.78 |
| BMP-2 | 0.60 ± 0.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franceschelli, S.; Lagioia, R.; De Cecco, F.; Minetti, E.; Ballini, A.; Panella, V.; Speranza, L.; Grilli, A.; Mastrangelo, F. Biological Evaluation of the Osteoinductive Potential of Dry Teeth after Chemical Demineralization Treatment Using the Tooth Transformer Device. Biomolecules 2023, 13, 1727. https://doi.org/10.3390/biom13121727
Franceschelli S, Lagioia R, De Cecco F, Minetti E, Ballini A, Panella V, Speranza L, Grilli A, Mastrangelo F. Biological Evaluation of the Osteoinductive Potential of Dry Teeth after Chemical Demineralization Treatment Using the Tooth Transformer Device. Biomolecules. 2023; 13(12):1727. https://doi.org/10.3390/biom13121727
Chicago/Turabian StyleFranceschelli, Sara, Rosalba Lagioia, Federica De Cecco, Elio Minetti, Andrea Ballini, Valeria Panella, Lorenza Speranza, Alfredo Grilli, and Filiberto Mastrangelo. 2023. "Biological Evaluation of the Osteoinductive Potential of Dry Teeth after Chemical Demineralization Treatment Using the Tooth Transformer Device" Biomolecules 13, no. 12: 1727. https://doi.org/10.3390/biom13121727
APA StyleFranceschelli, S., Lagioia, R., De Cecco, F., Minetti, E., Ballini, A., Panella, V., Speranza, L., Grilli, A., & Mastrangelo, F. (2023). Biological Evaluation of the Osteoinductive Potential of Dry Teeth after Chemical Demineralization Treatment Using the Tooth Transformer Device. Biomolecules, 13(12), 1727. https://doi.org/10.3390/biom13121727








