Unconventional Myosins from Caenorhabditis elegans as a Probe to Study Human Orthologues
Abstract
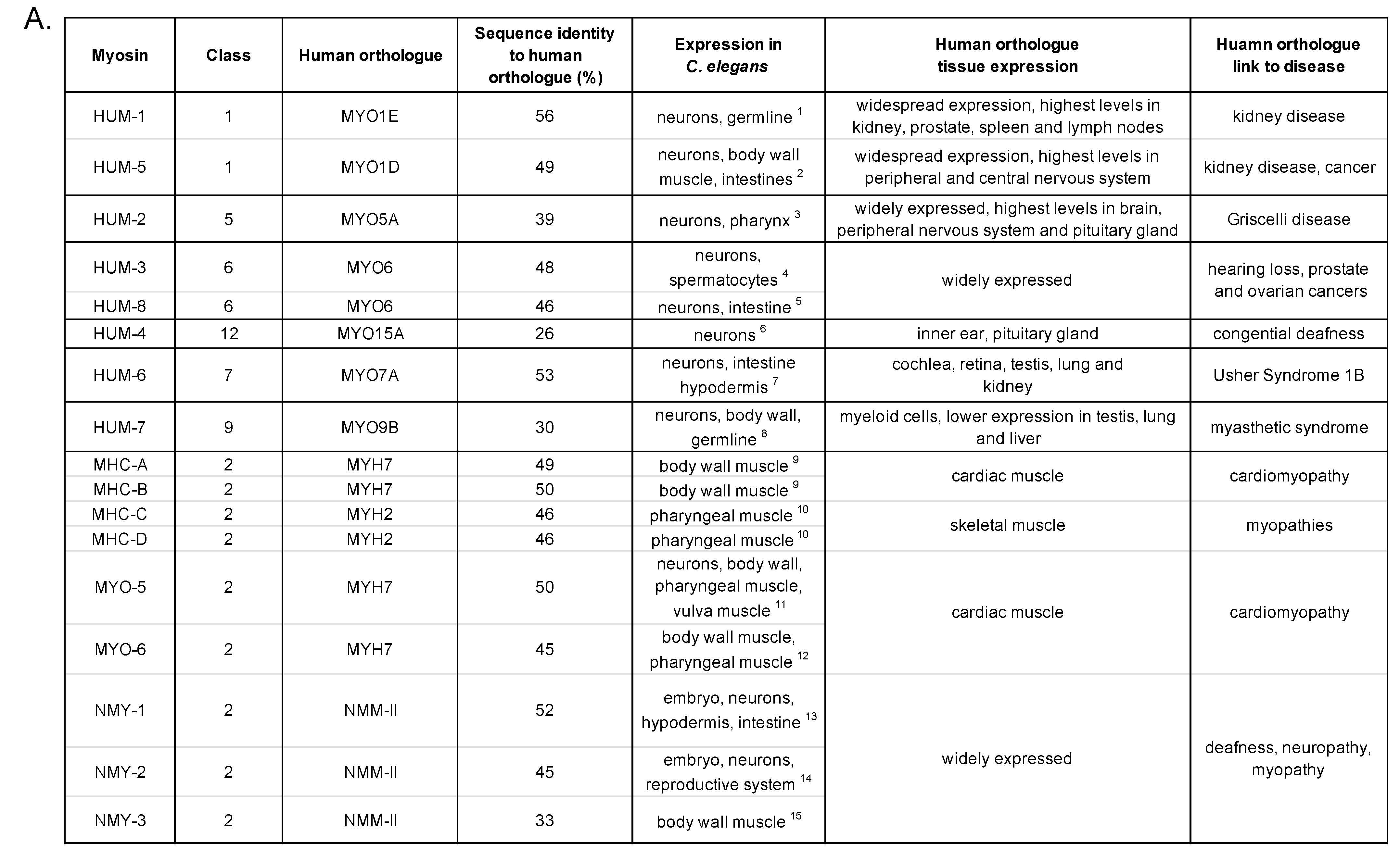
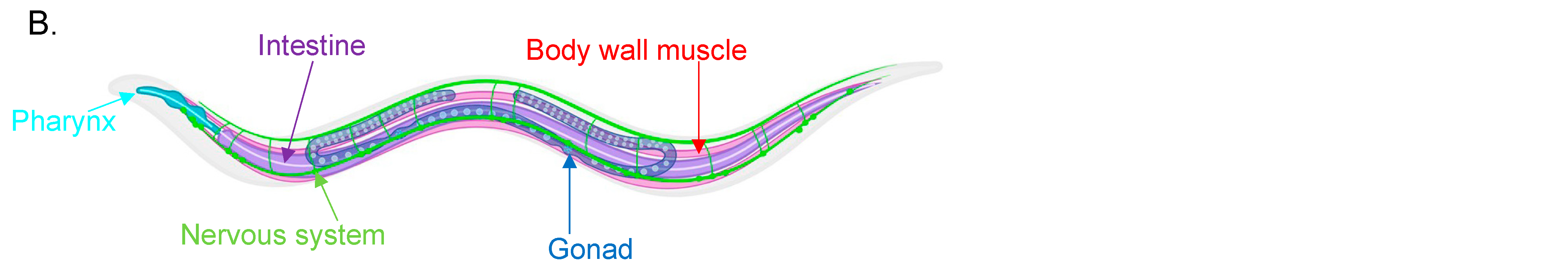
:1. Introduction
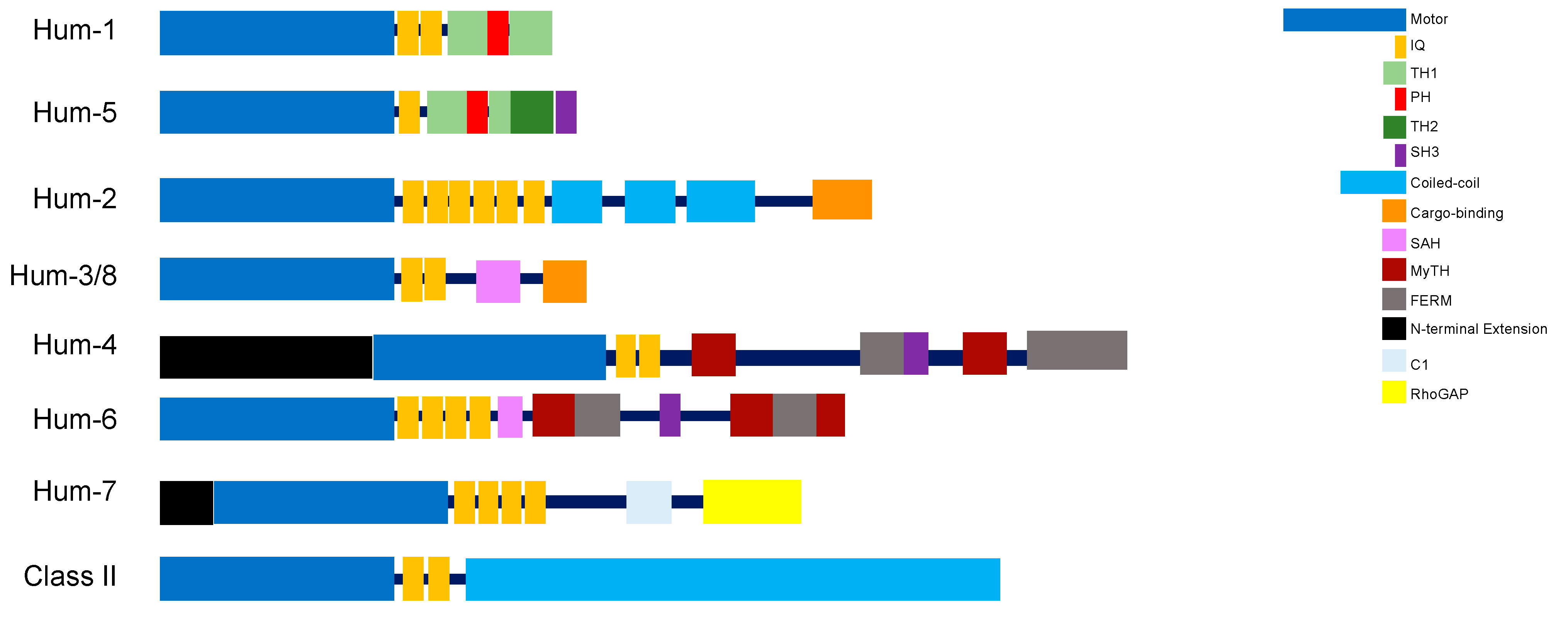
2. HUM-1 and HUM-5
3. HUM-2
4. HUM-3 and HUM-8
5. HUM-4
6. HUM-6
7. HUM-7
C. elegans Myosins of Class II
8. Conclusions and Future Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hartman, M.A.; Finan, D.; Sivaramakrishnan, S.; Spudich, J.A. Principles of Unconventional Myosin Function and Targeting. Annu. Rev. Cell Dev. Biol. 2011, 27, 133–155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bähler, M.; Rhoads, A. Calmodulin Signaling via the IQ Motif. FEBS Lett. 2002, 513, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Heissler, S.M.; Sellers, J.R. Myosin Light Chains: Teaching Old Dogs New Tricks. Bioarchitecture 2014, 4, 169–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lowey, S.; Slayter, H.S.; Weeds, A.G.; Baker, H. Substructure of the Myosin Molecule. I. Subfragments of Myosin by Enzymic Degradation. J. Mol. Biol. 1969, 42, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Weeds, A.G.; Pope, B. Studies on the Chymotryptic Digestion of Myosin. Effects of Divalent Cations on Proteolytic Susceptibility. J. Mol. Biol. 1977, 111, 129–157. [Google Scholar] [CrossRef] [PubMed]
- Szent-Györgyi, A.G. The Early History of the Biochemistry of Muscle Contraction. J. Gen. Physiol. 2004, 123, 631–641. [Google Scholar] [CrossRef]
- Krendel, M.; Mooseker, M.S. Myosins: Tails (and Heads) of Functional Diversity. Physiol. Bethesda Md 2005, 20, 239–251. [Google Scholar] [CrossRef] [Green Version]
- Hartman, M.A.; Spudich, J.A. The Myosin Superfamily at a Glance. J. Cell Sci. 2012, 125, 1627–1632. [Google Scholar] [CrossRef] [Green Version]
- Masters, T.A.; Kendrick-Jones, J.; Buss, F. Myosins: Domain Organisation, Motor Properties, Physiological Roles and Cellular Functions. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2016; Volume 235, pp. 77–122. [Google Scholar]
- Odronitz, F.; Kollmar, M. Drawing the Tree of Eukaryotic Life Based on the Analysis of 2,269 Manually Annotated Myosins from 328 Species. Genome Biol. 2007, 8, 196. [Google Scholar] [CrossRef] [Green Version]
- Thompson, R.F.; Langford, G.M. Myosin Superfamily Evolutionary History. Anat. Rec. 2002, 268, 276–289. [Google Scholar] [CrossRef]
- Brown, S.S. Myosins in Yeast. Curr. Opin. Cell Biol. 1997, 9, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Bezanilla, M.; Forsburg, S.L.; Pollard, T.D. Identification of a Second Myosin-II in Schizosaccharomyces Pombe: Myp2p Is Conditionally Required for Cytokinesis. Mol. Biol. Cell 1997, 8, 2693–2705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- May, K.M.; Win, T.Z.; Hyams, J.S. Yeast Myosin II: A New Subclass of Unconventional Conventional Myosins? Cell Motil. Cytoskeleton 1998, 39, 195–200. [Google Scholar] [CrossRef]
- Lee, W.L.; Bezanilla, M.; Pollard, T.D. Fission Yeast Myosin-I, Myo1p, Stimulates Actin Assembly by Arp2/3 Complex and Shares Functions with WASp. J. Cell Biol. 2000, 151, 789–800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Win, T.Z.; Gachet, Y.; Mulvihill, D.P.; May, K.M.; Hyams, J.S. Two Type V Myosins with Non-Overlapping Functions in the Fission Yeast Schizosaccharomyces Pombe: Myo52 Is Concerned with Growth Polarity and Cytokinesis, Myo51 Is a Component of the Cytokinetic Actin Ring. J. Cell Sci. 2001, 114, 69–79. [Google Scholar] [CrossRef]
- Adams, M.D.; Celniker, S.E.; Holt, R.A.; Evans, C.A.; Gocayne, J.D.; Amanatides, P.G.; Scherer, S.E.; Li, P.W.; Hoskins, R.A.; Galle, R.F.; et al. The Genome Sequence of Drosophila Melanogaster. Science 2000, 287, 2185–2195. [Google Scholar] [CrossRef] [Green Version]
- Goldstein, L.S.; Gunawardena, S. Flying through the Drosophila Cytoskeletal Genome. J. Cell Biol. 2000, 150, F63–F68. [Google Scholar] [CrossRef]
- Berg, J.S.; Powell, B.C.; Cheney, R.E. A Millennial Myosin Census. Mol. Biol. Cell 2001, 12, 780. [Google Scholar] [CrossRef] [Green Version]
- Coluccio, L.M. Myosins and Disease. Adv. Exp. Med. Biol. 2020, 1239, 245–316. [Google Scholar] [CrossRef]
- Kaletta, T.; Hengartner, M.O. Finding Function in Novel Targets: C. Elegans as a Model Organism. Nat. Rev. Drug Discov. 2006, 5, 387–399. [Google Scholar] [CrossRef]
- O’Reilly, L.P.; Luke, C.J.; Perlmutter, D.H.; Silverman, G.A.; Pak, S.C.C. Elegans in High-Throughput Drug Discovery. Adv. Drug Deliv. Rev. 2014, 69–70, 247–253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baker, J.P.; Titus, M.A. A Family of Unconventional Myosins from the Nematode Caenorhabditis Elegans. J. Mol. Biol. 1997, 272, 523–535. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.J.; Watson, J.D.; Spencer, W.C.; O’Brien, T.; Cha, B.; Albeg, A.; Treinin, M.; Miller, D.M. Time-Lapse Imaging and Cell-Specific Expression Profiling Reveal Dynamic Branching and Molecular Determinants of a Multi-Dendritic Nociceptor in C. Elegans. Dev. Biol. 2010, 345, 18–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, S.; Schroeder, E.A.; Silva-García, C.G.; Hebestreit, K.; Mair, W.B.; Brunet, A. Mono-Unsaturated Fatty Acids Link H3K4me3 Modifiers to C. Elegans Lifespan. Nature 2017, 544, 185–190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dupuy, D.; Bertin, N.; Hidalgo, C.A.; Venkatesan, K.; Tu, D.; Lee, D.; Rosenberg, J.; Svrzikapa, N.; Blanc, A.; Carnec, A.; et al. Genome-Scale Analysis of in Vivo Spatiotemporal Promoter Activity in Caenorhabditis Elegans. Nat. Biotechnol. 2007, 25, 663–668. [Google Scholar] [CrossRef]
- Li, Q.; Marcu, D.-C.; Palazzo, O.; Turner, F.; King, D.; Spires-Jones, T.L.; Stefan, M.I.; Busch, K.E. High Neural Activity Accelerates the Decline of Cognitive Plasticity with Age in Caenorhabditis Elegans. eLife 2020, 9, e59711. [Google Scholar] [CrossRef]
- He, S.; Philbrook, A.; McWhirter, R.; Gabel, C.V.; Taub, D.G.; Carter, M.H.; Hanna, I.M.; Francis, M.M.; Miller, D.M. Transcriptional Control of Synaptic Remodeling through Regulated Expression of an Immunoglobulin Superfamily Protein. Curr. Biol. CB 2015, 25, 2541–2548. [Google Scholar] [CrossRef] [Green Version]
- Kratsios, P.; Kerk, S.Y.; Catela, C.; Liang, J.; Vidal, B.; Bayer, E.A.; Feng, W.; De La Cruz, E.D.; Croci, L.; Giacomo Consalez, G.; et al. An Intersectional Gene Regulatory Strategy Defines Subclass Diversity of C. Elegans Motor Neurons. eLife 2017, 6, 25751. [Google Scholar] [CrossRef]
- Taylor, S.R.; Santpere, G.; Weinreb, A.; Barrett, A.; Reilly, M.B.; Xu, C.; Varol, E.; Oikonomou, P.; Glenwinkel, L.; McWhirter, R.; et al. Molecular Topography of an Entire Nervous System. Cell 2021, 184, 4329–4347.e23. [Google Scholar] [CrossRef]
- Grün, D.; Kirchner, M.; Thierfelder, N.; Stoeckius, M.; Selbach, M.; Rajewsky, N. Conservation of MRNA and Protein Expression during Development of C. Elegans. Cell Rep. 2014, 6, 565–577. [Google Scholar] [CrossRef]
- Blazie, S.M.; Geissel, H.C.; Wilky, H.; Joshi, R.; Newbern, J.; Mangone, M. Alternative Polyadenylation Directs Tissue-Specific MiRNA Targeting in Caenorhabditis Elegans Somatic Tissues. Genetics 2017, 206, 757–774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lockhead, D.; Schwarz, E.M.; O’Hagan, R.; Bellotti, S.; Krieg, M.; Barr, M.M.; Dunn, A.R.; Sternberg, P.W.; Goodman, M.B. The Tubulin Repertoire of C. Elegans Sensory Neurons and Its Context-Dependent Role in Process Outgrowth. Mol. Biol. Cell 2016, 27, 3717–3728. [Google Scholar] [CrossRef] [PubMed]
- Spencer, W.C.; Zeller, G.; Watson, J.D.; Henz, S.R.; Watkins, K.L.; McWhirter, R.D.; Petersen, S.; Sreedharan, V.T.; Widmer, C.; Jo, J.; et al. A Spatial and Temporal Map of C. Elegans Gene Expression. Genome Res. 2011, 21, 325–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallace, A.G.; Raduwan, H.; Carlet, J.; Soto, M.C. The RhoGAP HUM-7/Myo9 Integrates Signals to Modulate RHO-1/RhoA during Embryonic Morphogenesis in Caenorhabditiselegans. Dev. Camb. Engl. 2018, 145, dev168724. [Google Scholar] [CrossRef] [Green Version]
- Epstein, H.F.; Thomson, J.N. Temperature-Sensitive Mutation Affecting Myofilament Assembly in Caenorhabditis Elegans. Nature 1974, 250, 579–580. [Google Scholar] [CrossRef]
- Miller, D.M.; Ortiz, I.; Berliner, G.C.; Epstein, H.F. Differential Localization of Two Myosins within Nematode Thick Filaments. Cell 1983, 34, 477–490. [Google Scholar] [CrossRef]
- Ardizzi, J.P.; Epstein, H.F. Immunochemical Localization of Myosin Heavy Chain Isoforms and Paramyosin in Developmentally and Structurally Diverse Muscle Cell Types of the Nematode Caenorhabditis Elegans. J. Cell Biol. 1987, 105, 2763–2770. [Google Scholar] [CrossRef] [Green Version]
- Cinar, H.; Keles, S.; Jin, Y. Expression Profiling of GABAergic Motor Neurons in Caenorhabditis Elegans. Curr. Biol. 2005, 15, 340–346. [Google Scholar] [CrossRef] [Green Version]
- Fox, R.M.; Watson, J.D.; Von Stetina, S.E.; McDermott, J.; Brodigan, T.M.; Fukushige, T.; Krause, M.; Miller, D.M. The Embryonic Muscle Transcriptome of Caenorhabditis Elegans. Genome Biol. 2007, 8, R188. [Google Scholar] [CrossRef] [Green Version]
- Gaudet, J.; Muttumu, S.; Horner, M.; Mango, S.E. Whole-Genome Analysis of Temporal Gene Expression during Foregut Development. PLoS Biol. 2004, 2, e352. [Google Scholar] [CrossRef]
- Mckay, S.J.; Johnsen, R.; Khattra, J.; Asano, J.; Baillie, D.L.; Chan, S.; Dube, N.; Fang, L.; Goszczynski, B.; Ha, E.; et al. Gene Expression Profiling of Cells, Tissues, and Developmental Stages of the Nematode C. Elegans. Cold Spring Harb. Symp. Quant. Biol. 2003, 68, 159–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piekny, A.J.; Johnson, J.-L.F.; Cham, G.D.; Mains, P.E. The Caenorhabditis Elegans Nonmuscle Myosin Genes Nmy-1 and Nmy-2 Function as Redundant Components of the Let-502/Rho-Binding Kinase and Mel-11/Myosin Phosphatase Pathway during Embryonic Morphogenesis. Dev. Camb. Engl. 2003, 130, 5695–5704. [Google Scholar] [CrossRef] [Green Version]
- Ding, S.S.; Woollard, A. Non-Muscle Myosin II Is Required for Correct Fate Specification in the Caenorhabditis Elegans Seam Cell Divisions. Sci. Rep. 2017, 7, 3524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.V.; Flavell, R.A. Myosin I: From Yeast to Human. Cell. Mol. Life Sci. 2008, 65, 2128–2137. [Google Scholar] [CrossRef] [PubMed]
- Zot, H.G.; Pollard, T.D. Motility of Myosin I on Planar Lipid Surfaces. Methods Cell Biol. 1993, 39, 51–63. [Google Scholar] [CrossRef]
- McIntosh, B.B.; Ostap, E.M. Myosin-I Molecular Motors at a Glance. J. Cell Sci. 2016, 129, 2689. [Google Scholar] [CrossRef] [Green Version]
- Bloemink, M.; Geeves, M. Shaking the Myosin Family Tree: Biochemical Kinetics Defines Four Types of Myosin Motor. Semin. Cell Dev. Biol. 2011, 22, 961–967. [Google Scholar] [CrossRef] [Green Version]
- Mooseker, M.S.; Cheney, R.E. Unconventional Myosins. Annu. Rev. Cell Dev. Biol. 1995, 11, 633–675. [Google Scholar] [CrossRef]
- Gupta, P.; Gauthier, N.C.; Cheng-Han, Y.; Zuanning, Y.; Pontes, B.; Ohmstede, M.; Martin, R.; Knölker, H.-J.; Döbereiner, H.-G.; Krendel, M.; et al. Myosin 1E Localizes to Actin Polymerization Sites in Lamellipodia, Affecting Actin Dynamics and Adhesion Formation. Biol. Open 2013, 2, 1288–1299. [Google Scholar] [CrossRef] [Green Version]
- Krendel, M.; Osterweil, E.K.; Mooseker, M.S. Myosin 1E Interacts with Synaptojanin-1 and Dynamin and Is Involved in Endocytosis. FEBS Lett. 2007, 581, 644–650. [Google Scholar] [CrossRef]
- Gauthier, N.C.; Masters, T.A.; Sheetz, M.P. Mechanical Feedback between Membrane Tension and Dynamics. Trends Cell Biol. 2012, 22, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, C.; Kinge, P.; Hutter, H. Axon Guidance Genes Identified in a Large-Scale RNAi Screen Using the RNAi-Hypersensitive Caenorhabditis Elegans Strain Nre-1(Hd20) Lin-15b(Hd126). Proc. Natl. Acad. Sci. USA 2007, 104, 834–839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huber, L.A.; Fialka, I.; Paiha, K.; Hunziker, W.; Sacks, D.B.; Bähler, M.; Way, M.; Gagescu, R.; Gruenberg, J. Both Calmodulin and the Unconventional Myosin Myr4 Regulate Membrane Trafficking along the Recycling Pathway of MDCK Cells. Traffic 2000, 1, 494–503. [Google Scholar] [CrossRef] [PubMed]
- Nambiar, R.; McConnell, R.E.; Tyska, M.J. Control of Cell Membrane Tension by Myosin-I. Proc. Natl. Acad. Sci. USA 2009, 106, 11972–11977. [Google Scholar] [CrossRef] [Green Version]
- Benesh, A.E.; Fleming, J.T.; Chiang, C.; Carter, B.D.; Tyska, M.J. Expression and Localization of Myosin-1d in the Developing Nervous System. Brain Res. 2012, 1440, 9–22. [Google Scholar] [CrossRef] [Green Version]
- Mele, C.; Iatropoulos, P.; Donadelli, R.; Calabria, A.; Maranta, R.; Cassis, P.; Buelli, S.; Tomasoni, S.; Piras, R.; Krendel, M.; et al. MYO1E Mutations and Childhood Familial Focal Segmental Glomerulosclerosis. N. Engl. J. Med. 2011, 365, 295–306. [Google Scholar] [CrossRef] [Green Version]
- Al-Hamed, M.H.; Al-Sabban, E.; Al-Mojalli, H.; Al-Harbi, N.; Faqeih, E.; Al Shaya, H.; Alhasan, K.; Al-Hissi, S.; Rajab, M.; Edwards, N.; et al. A Molecular Genetic Analysis of Childhood Nephrotic Syndrome in a Cohort of Saudi Arabian Families. J. Hum. Genet. 2013, 58, 480–489. [Google Scholar] [CrossRef]
- Ouderkirk-Pecone, J.L.; Goreczny, G.J.; Chase, S.E.; Tatum, A.H.; Turner, C.E.; Krendel, M. Myosin 1e Promotes Breast Cancer Malignancy by Enhancing Tumor Cell Proliferation and Stimulating Tumor Cell De-Differentiation. Oncotarget 2016, 7, 46419–46432. [Google Scholar] [CrossRef] [Green Version]
- Ngollo, M.; Lebert, A.; Daures, M.; Judes, G.; Rifai, K.; Dubois, L.; Kemeny, J.-L.; Penault-Llorca, F.; Bignon, Y.-J.; Guy, L.; et al. Global Analysis of H3K27me3 as an Epigenetic Marker in Prostate Cancer Progression. BMC Cancer 2017, 17, 261. [Google Scholar] [CrossRef]
- Win, T.Z.; Mulvihill, D.P.; Hyams, J.S. Take Five: A Myosin Class Act in Fission Yeast. Cell Motil. Cytoskeleton 2002, 51, 53–56. [Google Scholar] [CrossRef]
- Veigel, C.; Wang, F.; Bartoo, M.L.; Sellers, J.R.; Molloy, J.E. The Gated Gait of the Processive Molecular Motor, Myosin V. Nat. Cell Biol. 2002, 4, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Veigel, C.; Schmitz, S.; Wang, F.; Sellers, J.R. Load-Dependent Kinetics of Myosin-V Can Explain Its High Processivity. Nat. Cell Biol. 2005, 7, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, O.C.; Cheney, R.E. Human Myosin-Vc Is a Novel Class V Myosin Expressed in Epithelial Cells. J. Cell Sci. 2002, 115, 991–1004. [Google Scholar] [CrossRef] [PubMed]
- Pastural, E.; Barrat, F.J.; Dufourcq-Lagelouse, R.; Certain, S.; Sanal, O.; Jabado, N.; Seger, R.; Griscelli, C.; Fischer, A.; de Saint Basile, G. Griscelli Disease Maps to Chromosome 15q21 and Is Associated with Mutations in the Myosin-Va Gene. Nat. Genet. 1997, 16, 289–292. [Google Scholar] [CrossRef] [PubMed]
- Wells, A.L.; Lin, A.W.; Chen, L.Q.; Safer, D.; Cain, S.M.; Hasson, T.; Carragher, B.O.; Milligan, R.A.; Sweeney, H.L. Myosin VI Is an Actin-Based Motor That Moves Backwards. Nature 1999, 401, 505–508. [Google Scholar] [CrossRef]
- Buss, F.; Arden, S.D.; Lindsay, M.; Luzio, J.P.; Kendrick-Jones, J. Myosin VI Isoform Localized to Clathrin-Coated Vesicles with a Role in Clathrin-Mediated Endocytosis. EMBO J. 2001, 20, 3676–3684. [Google Scholar] [CrossRef] [Green Version]
- Ménétrey, J.; Bahloul, A.; Wells, A.L.; Yengo, C.M.; Morris, C.A.; Sweeney, H.L.; Houdusse, A. The Structure of the Myosin VI Motor Reveals the Mechanism of Directionality Reversal. Nature 2005, 435, 779–785. [Google Scholar] [CrossRef] [Green Version]
- Bryant, Z.; Altman, D.; Spudich, J.A. The Power Stroke of Myosin VI and the Basis of Reverse Directionality. Proc. Natl. Acad. Sci. USA 2007, 104, 772–777. [Google Scholar] [CrossRef] [Green Version]
- De La Cruz, E.M.; Ostap, E.M.; Sweeney, H.L. Kinetic Mechanism and Regulation of Myosin VI. J. Biol. Chem. 2001, 276, 32373–32381. [Google Scholar] [CrossRef] [Green Version]
- Lister, I.; Schmitz, S.; Walker, M.; Trinick, J.; Buss, F.; Veigel, C.; Kendrick-Jones, J. A Monomeric Myosin VI with a Large Working Stroke. EMBO J. 2004, 23, 1729–1738. [Google Scholar] [CrossRef]
- Phichith, D.; Travaglia, M.; Yang, Z.; Liu, X.; Zong, A.B.; Safer, D.; Sweeney, H.L. Cargo Binding Induces Dimerization of Myosin VI. Proc. Natl. Acad. Sci. USA 2009, 106, 17320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, C.; Feng, W.; Wei, Z.; Miyanoiri, Y.; Wen, W.; Zhao, Y.; Zhang, M. Myosin VI Undergoes Cargo-Mediated Dimerization. Cell 2009, 138, 537–548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altman, D.; Sweeney, H.L.; Spudich, J.A. The Mechanism of Myosin VI Translocation and Its Load-Induced Anchoring. Cell 2004, 116, 737–749. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altman, D.; Goswami, D.; Hasson, T.; Spudich, J.A.; Mayor, S. Precise Positioning of Myosin VI on Endocytic Vesicles in Vivo. PLoS Biol. 2007, 5, 1712–1722. [Google Scholar] [CrossRef]
- Morris, S.M.; Arden, S.D.; Roberts, R.C.; Kendrick-Jones, J.; Cooper, J.A.; Luzio, J.P.; Buss, F. Myosin VI Binds to and Localises with Dab2, Potentially Linking Receptor-Mediated Endocytosis and the Actin Cytoskeleton. Traffic 2002, 3, 331–341. [Google Scholar] [CrossRef]
- Warner, C.L.; Stewart, A.; Luzio, J.P.; Steel, K.P.; Libby, R.T.; Kendrick-Jones, J.; Buss, F. Loss of Myosin VI Reduces Secretion and the Size of the Golgi in Fibroblasts from Snell’s Waltzer Mice. EMBO J. 2003, 22, 569–579. [Google Scholar] [CrossRef] [Green Version]
- Spudich, G.; Chibalina, M.V.; Au, J.S.-Y.; Arden, S.D.; Buss, F.; Kendrick-Jones, J. Myosin VI Targeting to Clathrin-Coated Structures and Dimerization Is Mediated by Binding to Disabled-2 and PtdIns(4,5)P2. Nat. Cell Biol. 2007, 9, 176–183. [Google Scholar] [CrossRef] [Green Version]
- Tumbarello, D.A.; Waxse, B.J.; Arden, S.D.; Bright, N.A.; Kendrick-Jones, J.; Buss, F. Autophagy Receptors Link Myosin VI to Autophagosomes to Mediate Tom1-Dependent Autophagosome Maturation and Fusion with the Lysosome. Nat. Cell Biol. 2012, 14, 1024–1035. [Google Scholar] [CrossRef] [Green Version]
- Masters, T.A.; Tumbarello, D.A.; Chibalina, M.V.; Buss, F. MYO6 Regulates Spatial Organization of Signaling Endosomes Driving AKT Activation and Actin Dynamics. Cell Rep. 2017, 19, 2088–2101. [Google Scholar] [CrossRef] [Green Version]
- O’Loughlin, T.; Masters, T.A.; Buss, F. The MYO6 Interactome Reveals Adaptor Complexes Coordinating Early Endosome and Cytoskeletal Dynamics. EMBO Rep. 2018, 19, e44884. [Google Scholar] [CrossRef]
- Skop, A.R.; Liu, H.; Yates, J.; Meyer, B.J.; Heald, R. Dissection of the Mammalian Midbody Proteome Reveals Conserved Cytokinesis Mechanisms. Science 2004, 305, 61–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arden, S.D.; Puri, C.; Au, J.S.-Y.; Kendrick-Jones, J.; Buss, F. Myosin VI Is Required for Targeted Membrane Transport during Cytokinesis. Mol. Biol. Cell 2007, 18, 4750–4761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- L’Hernault, S.W.; Shakes, D.C.; Ward, S. Developmental Genetics of Chromosome I Spermatogenesis-Defective Mutants in the Nematode Caenorhabditis Elegans. Genetics 1988, 120, 435–452. [Google Scholar] [CrossRef] [PubMed]
- Kelleher, J.F.; Mandell, M.A.; Moulder, G.; Hill, K.L.; L’Hernault, S.W.; Barstead, R.; Titus, M.A. Myosin VI Is Required for Asymmetric Segregation of Cellular Components during C. Elegans Spermatogenesis. Curr. Biol. 2000, 10, 1489–1496. [Google Scholar] [CrossRef] [Green Version]
- Hu, J.; Cheng, S.; Wang, H.; Li, X.; Liu, S.; Wu, M.; Liu, Y.; Wang, X. Distinct Roles of Two Myosins in C. Elegans Spermatid Differentiation. PLoS Biol. 2019, 17, e3000211. [Google Scholar] [CrossRef] [Green Version]
- Avraham, K.B.; Hasson, T.; Steel, K.P.; Kingsley, D.M.; Russell, L.B.; Mooseker, M.S.; Copeland, N.G.; Jenkins, N.A. The Mouse Snell’s Waltzer Deafness Gene Encodes an Unconventional Myosin Required for Structural Integrity of Inner Ear Hair Cells. Nat. Genet. 1995, 11, 369–375. [Google Scholar] [CrossRef]
- Melchionda, S.; Ahituv, N.; Bisceglia, L.; Sobe, T.; Glaser, F.; Rabionet, R.; Arbones, M.L.; Notarangelo, A.; Di Iorio, E.; Carella, M.; et al. MYO6, the Human Homologue of the Gene Responsible for Deafness in Snell’s Waltzer Mice, Is Mutated in Autosomal Dominant Nonsyndromic Hearing Loss. Am. J. Hum. Genet. 2001, 69, 635–640. [Google Scholar] [CrossRef]
- Zakrzewski, P.; Lenartowska, M.; Buss, F. Diverse Functions of Myosin VI in Spermiogenesis. Histochem. Cell Biol. 2021, 155, 323–340. [Google Scholar] [CrossRef]
- Mohiddin, S.A.; Ahmed, Z.M.; Griffith, A.J.; Tripodi, D.; Friedman, T.B.; Fananapazir, L.; Morell, R.J. Novel Association of Hypertrophic Cardiomyopathy, Sensorineural Deafness, and a Mutation in Unconventional Myosin VI (MYO6). J. Med. Genet. 2004, 41, 309–314. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, H.; Cheng, W.; Hung, J.; Montell, D.; Geisbrecht, E.; Rosen, D.; Liu, J.; Naora, H. Lessons from Border Cell Migration in the Drosophila Ovary: A Role for Myosin VI in Dissemination of Human Ovarian Cancer. Proc. Natl. Acad. Sci. USA 2004, 101, 8144–8149. [Google Scholar] [CrossRef]
- Dunn, T.A.; Chen, S.; Faith, D.A.; Hicks, J.L.; Platz, E.A.; Chen, Y.; Ewing, C.M.; Sauvageot, J.; Isaacs, W.B.; De Marzo, A.M.; et al. A Novel Role of Myosin VI in Human Prostate Cancer. Am. J. Pathol. 2006, 169, 1843–1854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foth, B.J.; Goedecke, M.C.; Soldati, D. New Insights into Myosin Evolution and Classification. Proc. Natl. Acad. Sci. USA 2006, 103, 3681–3686. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, F.; Takagi, Y.; Shams, A.; Heissler, S.M.; Friedman, T.B.; Sellers, J.R.; Bird, J.E. The ATPase Mechanism of Myosin 15, the Molecular Motor Mutated in DFNB3 Human Deafness. J. Biol. Chem. 2021, 296, 100243. [Google Scholar] [CrossRef] [PubMed]
- Friedman, T.B.; Liang, Y.; Weber, J.L.; Hinnant, J.T.; Barber, T.D.; Winata, S.; Arhya, I.N.; Asher, J.H. A Gene for Congenital, Recessive Deafness DFNB3 Maps to the Pericentromeric Region of Chromosome 17. Nat. Genet. 1995, 9, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Liang, Y.; Fridell, R.A.; Probst, F.J.; Wilcox, E.R.; Touchman, J.W.; Morton, C.C.; Morell, R.J.; Noben-Trauth, K.; Camper, S.A.; et al. Association of Unconventional Myosin MYO15 Mutations with Human Nonsyndromic Deafness DFNB3. Science 1998, 280, 1447–1451. [Google Scholar] [CrossRef] [Green Version]
- O’Hagan, R.; Chalfie, M.; Goodman, M.B. The MEC-4 DEG/ENaC Channel of Caenorhabditis Elegans Touch Receptor Neurons Transduces Mechanical Signals. Nat. Neurosci. 2005, 8, 43–50. [Google Scholar] [CrossRef]
- Chen, X.; Chalfie, M. Modulation of C. Elegans Touch Sensitivity Is Integrated at Multiple Levels. J. Neurosci. Off. J. Soc. Neurosci. 2014, 34, 6522–6536. [Google Scholar] [CrossRef] [Green Version]
- Kiehart, D.P.; Franke, J.D.; Chee, M.K.; Montague, R.A.; Chen, T.-L.; Roote, J.; Ashburner, M. Drosophila Crinkled, Mutations of Which Disrupt Morphogenesis and Cause Lethality, Encodes Fly Myosin VIIA. Genetics 2004, 168, 1337–1352. [Google Scholar] [CrossRef] [Green Version]
- Gibbs, D.; Diemer, T.; Khanobdee, K.; Hu, J.; Bok, D.; Williams, D.S. Function of MYO7A in the Human RPE and the Validity of Shaker1 Mice as a Model for Usher Syndrome 1B. Investig. Ophthalmol. Vis. Sci. 2010, 51, 1130–1135. [Google Scholar] [CrossRef]
- Lopes, V.S.; Gibbs, D.; Libby, R.T.; Aleman, T.S.; Welch, D.L.; Lillo, C.; Jacobson, S.G.; Radu, R.A.; Steel, K.P.; Williams, D.S. The Usher 1B Protein, MYO7A, Is Required for Normal Localization and Function of the Visual Retinoid Cycle Enzyme, RPE65. Hum. Mol. Genet. 2011, 20, 2560–2570. [Google Scholar] [CrossRef]
- Grati, M.; Kachar, B. Myosin VIIa and sans Localization at Stereocilia Upper Tip-Link Density Implicates These Usher Syndrome Proteins in Mechanotransduction. Proc. Natl. Acad. Sci. USA 2011, 108, 11476–11481. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Guan, L.; Zhan, J.; Lu, D.; Wan, J.; Zhang, H. FERM Domain-Containing Unconventional Myosin VIIA Interacts with Integrin Β5 Subunit and Regulates Avβ5-Mediated Cell Adhesion and Migration. FEBS Lett. 2014, 588, 2859–2866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weil, D.; Blanchard, S.; Kaplan, J.; Guilford, P.; Gibson, F.; Walsh, J.; Mburu, P.; Varela, A.; Levilliers, J.; Weston, M.D. Defective Myosin VIIA Gene Responsible for Usher Syndrome Type 1B. Nature 1995, 374, 60–61. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Vansant, G.; Udovichenko, I.P.; Wolfrum, U.; Williams, D.S. Myosin VIIa, the Product of the Usher 1B Syndrome Gene, Is Concentrated in the Connecting Cilia of Photoreceptor Cells. Cell Motil. Cytoskeleton 1997, 37, 240–252. [Google Scholar] [CrossRef]
- Weil, D.; Küssel, P.; Blanchard, S.; Lévy, G.; Levi-Acobas, F.; Drira, M.; Ayadi, H.; Petit, C. The Autosomal Recessive Isolated Deafness, DFNB2, and the Usher 1B Syndrome Are Allelic Defects of the Myosin-VIIA Gene. Nat. Genet. 1997, 16, 191–193. [Google Scholar] [CrossRef] [PubMed]
- Lévy, G.; Levi-Acobas, F.; Blanchard, S.; Gerber, S.; Larget-Piet, D.; Chenal, V.; Liu, X.Z.; Newton, V.; Steel, K.P.; Brown, S.D.; et al. Myosin VIIA Gene: Heterogeneity of the Mutations Responsible for Usher Syndrome Type IB. Hum. Mol. Genet. 1997, 6, 111–116. [Google Scholar] [CrossRef] [Green Version]
- El-Amraoui, A.; Petit, C. Usher I Syndrome: Unravelling the Mechanisms That Underlie the Cohesion of the Growing Hair Bundle in Inner Ear Sensory Cells. J. Cell Sci. 2005, 118, 4593–4603. [Google Scholar] [CrossRef] [Green Version]
- Sun, Y.; Chen, J.; Sun, H.; Cheng, J.; Li, J.; Lu, Y.; Lu, Y.; Jin, Z.; Zhu, Y.; Ouyang, X.; et al. Novel Missense Mutations in MYO7A Underlying Postlingual High- or Low-Frequency Non-Syndromic Hearing Impairment in Two Large Families from China. J. Hum. Genet. 2011, 56, 64–70. [Google Scholar] [CrossRef] [Green Version]
- Adato, A.; Weil, D.; Kalinski, H.; Pel-Or, Y.; Ayadi, H.; Petit, C.; Korostishevsky, M.; Bonne-Tamir, B. Mutation Profile of All 49 Exons of the Human Myosin VIIA Gene, and Haplotype Analysis, in Usher 1B Families from Diverse Origins. Am. J. Hum. Genet. 1997, 61, 813–821. [Google Scholar] [CrossRef] [Green Version]
- Nalavadi, V.; Nyitrai, M.; Bertolini, C.; Adamek, N.; Geeves, M.A.; Bähler, M. Kinetic Mechanism of Myosin IXB and the Contributions of Two Class IX-Specific Regions. J. Biol. Chem. 2005, 280, 38957–38968. [Google Scholar] [CrossRef]
- Kambara, T.; Ikebe, M. A Unique ATP Hydrolysis Mechanism of Single-Headed Processive Myosin, Myosin IX. J. Biol. Chem. 2006, 281, 4949–4957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liao, W.; Elfrink, K.; Bähler, M. Head of Myosin IX Binds Calmodulin and Moves Processively toward the Plus-End of Actin Filaments. J. Biol. Chem. 2010, 285, 24933–24942. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanley, P.J.; Xu, Y.; Kronlage, M.; Grobe, K.; Schön, P.; Song, J.; Sorokin, L.; Schwab, A.; Bähler, M. Motorized RhoGAP Myosin IXb (Myo9b) Controls Cell Shape and Motility. Proc. Natl. Acad. Sci. USA 2010, 107, 12145–12150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elfrink, K.; Liao, W.; Pieper, U.; Oeding, S.J.; Bähler, M. The Loop2 Insertion of Type IX Myosin Acts as an Electrostatic Actin Tether That Permits Processive Movement. PLoS ONE 2014, 9, e84874. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Connor, E.; Töpf, A.; Müller, J.S.; Cox, D.; Evangelista, T.; Colomer, J.; Abicht, A.; Senderek, J.; Hasselmann, O.; Yaramis, A.; et al. Identification of Mutations in the MYO9A Gene in Patients with Congenital Myasthenic Syndrome. Brain J. Neurol. 2016, 139, 2143–2153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, M.-J.; Xu, X.-L.; Yao, G.-L.; Yu, Q.; Zhu, C.-F.; Kong, Z.-J.; Zhao, H.; Tang, L.-M.; Qin, X.-H. MYO9B Gene Polymorphisms Are Associated with the Risk of Inflammatory Bowel Diseases. Oncotarget 2016, 7, 58862–58875. [Google Scholar] [CrossRef] [Green Version]
- Li, P.; Yang, X.-K.; Wang, X.; Zhao, M.-Q.; Zhang, C.; Tao, S.-S.; Zhao, W.; Huang, Q.; Li, L.-J.; Pan, H.-F.; et al. A Meta-Analysis of the Relationship between MYO9B Gene Polymorphisms and Susceptibility to Crohn’s Disease and Ulcerative Colitis. Hum. Immunol. 2016, 77, 990–996. [Google Scholar] [CrossRef]
- Chen, Y.-Q.; Zhang, L.; Lv, X.-Y.; Wang, H.-Z. Lack of Association between MYO9B Gene Polymorphisms and Susceptibility to Coeliac Disease in Caucasians: Evidence from a Meta-Analysis. Immunol. Investig. 2016, 45, 396–405. [Google Scholar] [CrossRef]
- Kong, R.; Yi, F.; Wen, P.; Liu, J.; Chen, X.; Ren, J.; Li, X.; Shang, Y.; Nie, Y.; Wu, K.; et al. Myo9b Is a Key Player in SLIT/ROBO-Mediated Lung Tumor Suppression. J. Clin. Investig. 2015, 125, 4407–4420. [Google Scholar] [CrossRef] [Green Version]
- Vicente-Manzanares, M.; Ma, X.; Adelstein, R.S.; Horwitz, A.R. Non-Muscle Myosin II Takes Centre Stage in Cell Adhesion and Migration. Nat. Rev. Mol. Cell Biol. 2009, 10, 778–790. [Google Scholar] [CrossRef]
- Gieseler, K.; Qadota, H.; Benian, G.M. Development, Structure, and Maintenance of C. Elegans Body Wall Muscle. In WormBook; The C. elegans Research Community. Available online: http://www.wormbook.org (accessed on 21 November 2022).
- Schachat, F.; Garcea, R.L.; Epstein, H.F. Myosins Exist as Homodimers of Heavy Chains: Demonstration with Specific Antibody Purified by Nematode Mutant Myosin Affinity Chromatography. Cell 1978, 15, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Waterston, R.H. The Minor Myosin Heavy Chain, MhcA, of Caenorhabditis Elegans Is Necessary for the Initiation of Thick Filament Assembly. EMBO J. 1989, 8, 3429–3436. [Google Scholar] [CrossRef] [PubMed]
- Venolia, L.; Waterston, R.H. The Unc-45 Gene of Caenorhabditis Elegans Is an Essential Muscle-Affecting Gene with Maternal Expression. Genetics 1990, 126, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Venolia, L.; Ao, W.; Kim, S.; Kim, C.; Pilgrim, D. Unc-45 Gene OfCaenorhabditis Elegans Encodes a Muscle-Specific Tetratricopeptide Repeat-Containing Protein. Cell Motil. Cytoskeleton 1999, 42, 163–177. [Google Scholar] [CrossRef]
- Barral, J.M.; Bauer, C.C.; Ortiz, I.; Epstein, H.F. Unc-45 Mutations in Caenorhabditis Elegans Implicate a CRO1/She4p-like Domain in Myosin Assembly. J. Cell Biol. 1998, 143, 1215–1225. [Google Scholar] [CrossRef]
- Ao, W.; Pilgrim, D. Caenorhabditis Elegans Unc-45 Is a Component of Muscle Thick Filaments and Colocalizes with Myosin Heavy Chain B, but Not Myosin Heavy Chain a. J. Cell Biol. 2000, 148, 375–384. [Google Scholar] [CrossRef]
- Etheridge, L.; Diiorio, P.; Sagerström, C.G. A Zebrafish Unc-45 -Related Gene Expressed during Muscle Development. Dev. Dyn. 2002, 224, 457–460. [Google Scholar] [CrossRef]
- Price, M.G.; Landsverk, M.L.; Barral, J.M.; Epstein, H.F. Two Mammalian UNC-45 Isoforms Are Related to Distinct Cytoskeletal and Muscle-Specific Functions. J. Cell Sci. 2002, 115, 4013–4023. [Google Scholar] [CrossRef] [Green Version]
- Geach, T.J.; Zimmerman, L.B. Paralysis and Delayed Z-Disc Formation in the Xenopus Tropicalis Unc45b Mutant Dicky Ticker. BMC Dev. Biol. 2010, 10, 75. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.F.; Hauenstein, A.V.; Fleming, J.K.; Gasper, W.C.; Engelke, V.; Sankaran, B.; Bernstein, S.I.; Huxford, T. X-ray Crystal Structure of the UCS Domain-Containing UNC-45 Myosin Chaperone from Drosophila Melanogaster. Structure 2011, 19, 397–408. [Google Scholar] [CrossRef]
- Gazda, L.; Pokrzywa, W.; Hellerschmied, D.; Löwe, T.; Forné, I.; Mueller-Planitz, F.; Hoppe, T.; Clausen, T. The Myosin Chaperone UNC-45 Is Organized in Tandem Modules to Support Myofilament Formation in C. Elegans. Cell 2013, 152, 183–195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colegrave, M.; Peckham, M. Structural Implications of β-Cardiac Myosin Heavy Chain Mutations in Human Disease. Anat. Rec. 2014, 297, 1670–1680. [Google Scholar] [CrossRef] [PubMed]
- Benian, G.M.; Epstein, H.F. Caenorhabditis Elegans Muscle: A Genetic and Molecular Model for Protein Interactions in the Heart. Circ. Res. 2011, 109, 1082–1095. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Várkuti, B.H.; Yang, Z.; Kintses, B.; Erdélyi, P.; Bárdos-Nagy, I.; Kovács, A.L.; Hári, P.; Kellermayer, M.; Vellai, T.; Málnási-Csizmadia, A. A Novel Actin Binding Site of Myosin Required for Effective Muscle Contraction. Nat. Struct. Mol. Biol. 2012, 19, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Dahl-Halvarsson, M.; Pokrzywa, M.; Rauthan, M.; Pilon, M.; Tajsharghi, H. Myosin Storage Myopathy in C. Elegans and Human Cultured Muscle Cells. PLoS ONE 2017, 12, e0170613. [Google Scholar] [CrossRef] [Green Version]
- Tajsharghi, H.; Darin, N.; Rekabdar, E.; Kyllerman, M.; Wahlström, J.; Martinsson, T.; Oldfors, A. Mutations and Sequence Variation in the Human Myosin Heavy Chain IIa Gene (MYH2). Eur. J. Hum. Genet. 2005, 13, 617–622. [Google Scholar] [CrossRef]
- Volpatti, J.R.; Endo, Y.; Knox, J.; Groom, L.; Brennan, S.; Noche, R.; Zuercher, W.J.; Roy, P.; Dirksen, R.T.; Dowling, J.J. Identification of Drug Modifiers for RYR1-Related Myopathy Using a Multi-Species Discovery Pipeline. eLife 2020, 9, e52946. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, C.A.; Behbehani, R.; Buss, F. Unconventional Myosins from Caenorhabditis elegans as a Probe to Study Human Orthologues. Biomolecules 2022, 12, 1889. https://doi.org/10.3390/biom12121889
Johnson CA, Behbehani R, Buss F. Unconventional Myosins from Caenorhabditis elegans as a Probe to Study Human Orthologues. Biomolecules. 2022; 12(12):1889. https://doi.org/10.3390/biom12121889
Chicago/Turabian StyleJohnson, Chloe A, Ranya Behbehani, and Folma Buss. 2022. "Unconventional Myosins from Caenorhabditis elegans as a Probe to Study Human Orthologues" Biomolecules 12, no. 12: 1889. https://doi.org/10.3390/biom12121889







