COVID-19 Rapid Antigen Test as Screening Strategy at Points of Entry: Experience in Lazio Region, Central Italy, August–October 2020
Abstract
1. Introduction
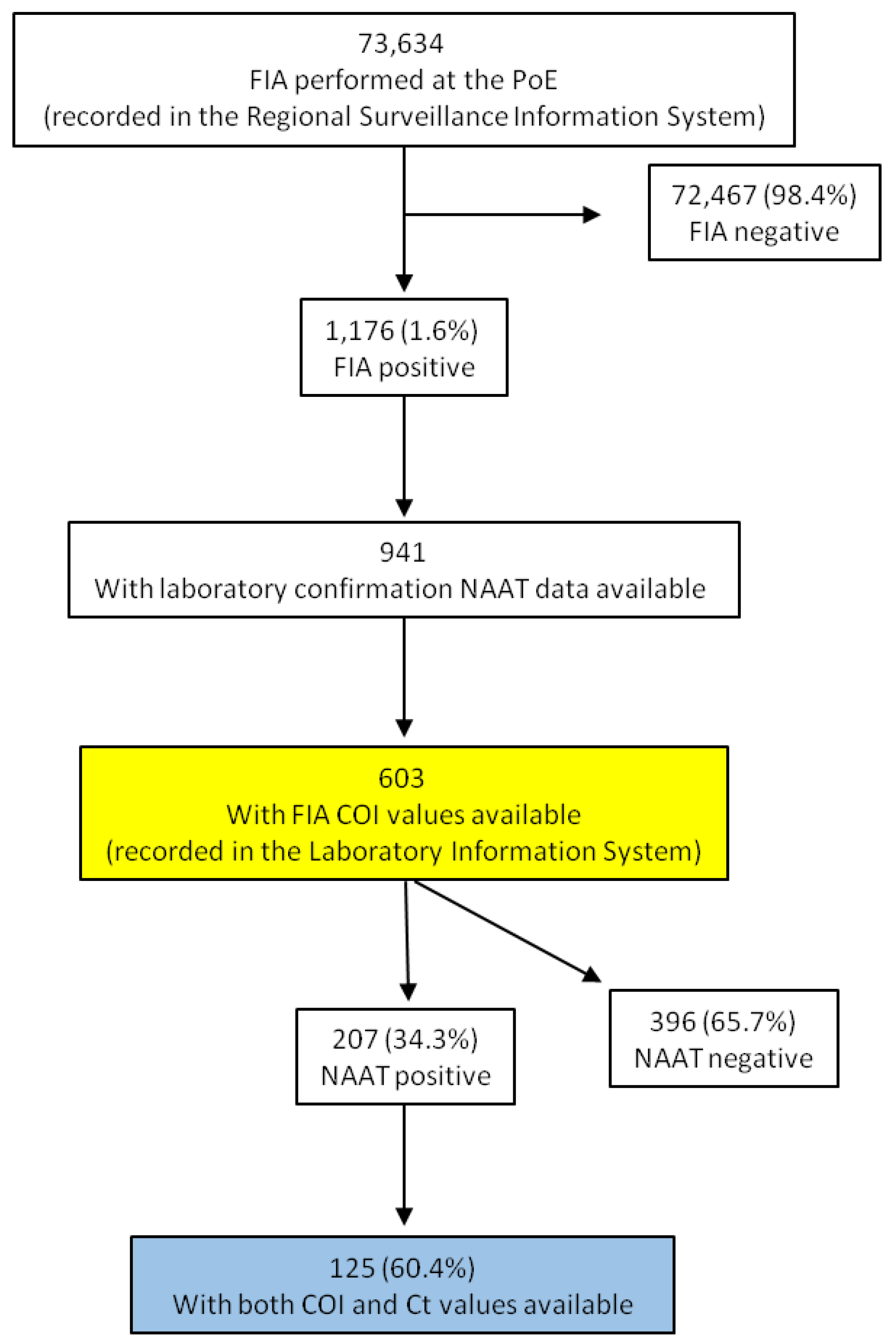
2. Materials and Methods
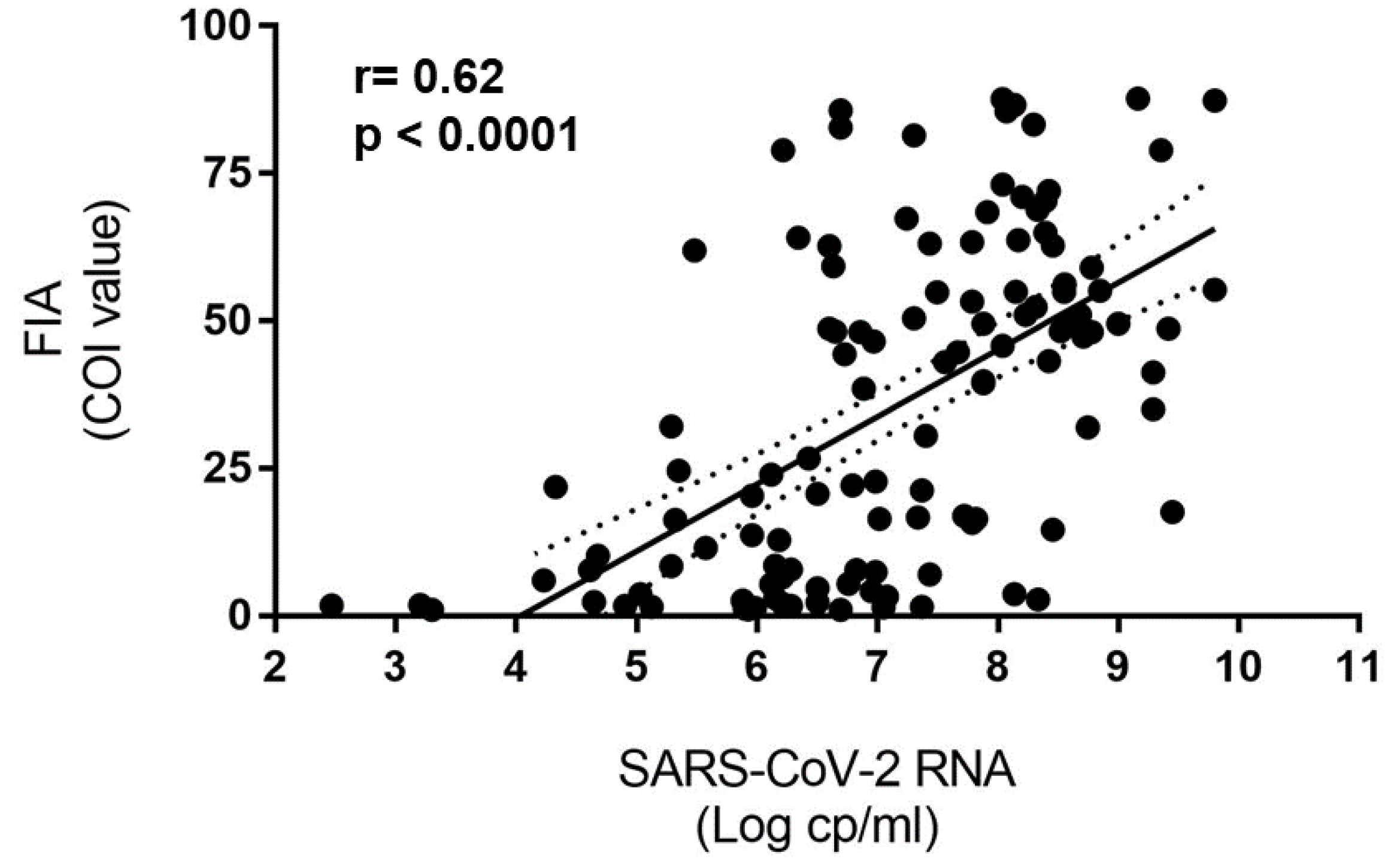
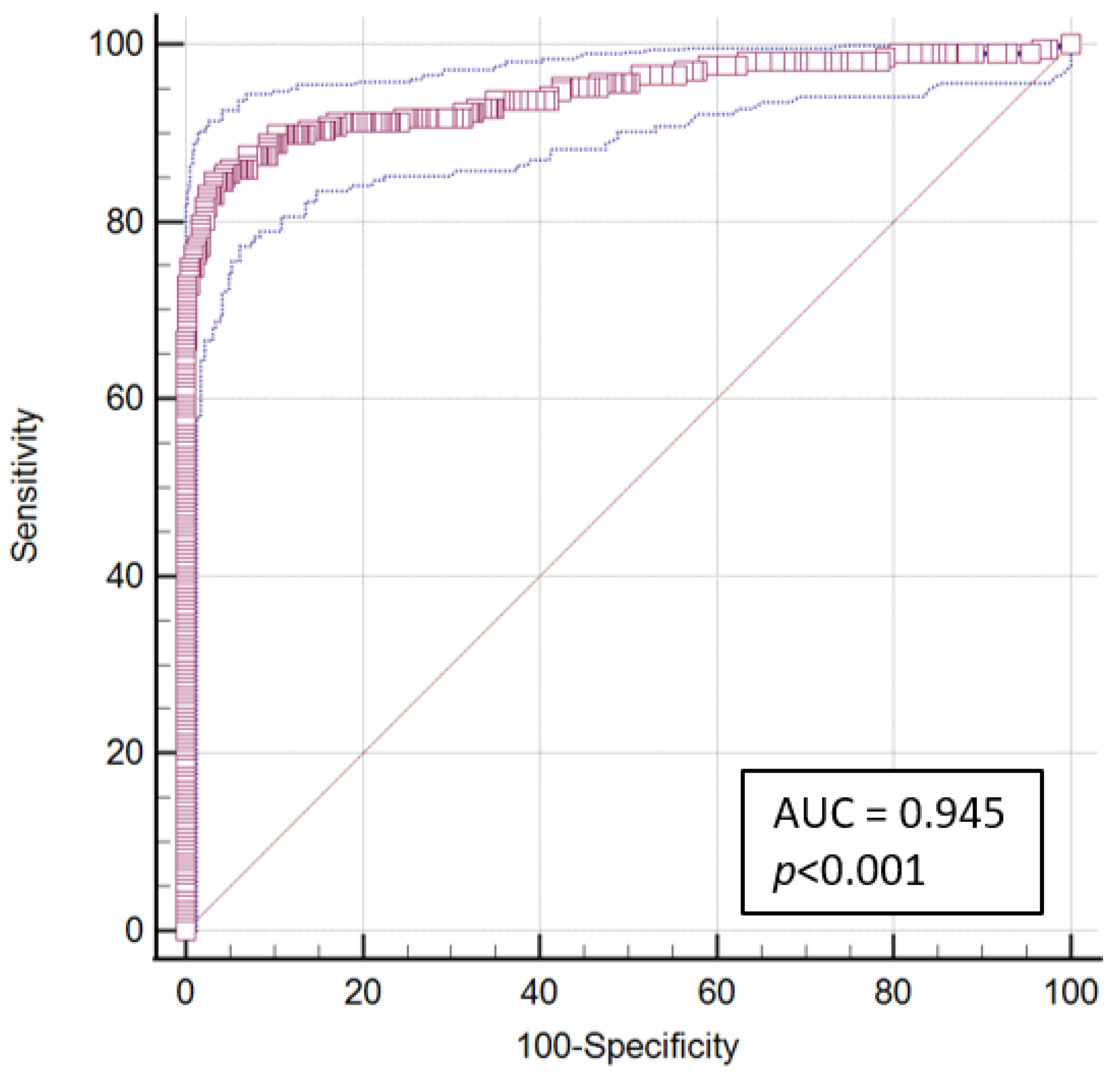
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. WHO Coronavirus Disease (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 11 February 2021).
- Rubin, R. The challenges of expanding rapid tests to curb COVID-19. JAMA 2020, 324, 1813–1815. [Google Scholar] [CrossRef] [PubMed]
- Mina, M.J.; Parker, R.; Larremore, D.B. Rethinking Covid-19 test sensitivity—A strategy for containment. N. Engl. J. Med. 2020, 383, e120. [Google Scholar] [CrossRef] [PubMed]
- Vandenberg, O.; Martiny, D.; Rochas, O.; van Belkum, A.; Kozlakidis, Z. Considerations for diagnostic COVID-19 tests. Nat. Rev. Microbiol. 2020, 19, 171–183. [Google Scholar] [CrossRef] [PubMed]
- WHO. Antigen-Detection in the Diagnosis of SARS-CoV-2 Infection Using Rapid Immunoassays: Interim Guidance. Available online: https://apps.who.int/iris/handle/10665/334253 (accessed on 11 September 2020).
- Fomsgaard, A.S.; Rosenstierne, M.W. An alternative workflow for molecular detection of SARS-CoV-2—Escape from the NA extraction kit-shortage, Copenhagen, Denmark, March 2020. Eurosurveillance 2020, 25, 2000398. [Google Scholar] [CrossRef] [PubMed]
- Mak, G.C.; Cheng, P.K.; Lau, S.S.; Wong, K.K.; Lau, C.S.; Lam, E.T.; Chan, R.C.; Tsang, D.N. Evaluation of rapid antigen test for detection of SARS-CoV-2 virus. J. Clin. Virol. 2020, 129, 104500. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.K.C.; Lam, W. Laboratory testing for the diagnosis of COVID-19. Biochem. Biophys. Res. Commun. 2021, 538, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Krüger, L.J.; Gaeddert, M.; Köppel, L.; Brümmer, L.E.; Gottschalk, C.; Miranda, I.B.; Schnitzler, P.; Kraeusslich, H.G.; Lindner, A.; Nikolai, O.; et al. Evaluation of the accuracy, ease of use and limit of detection of novel, rapid, antigen-detecting point-of-care diagnostics for SARS-CoV-2. medRxiv 2020. [Google Scholar] [CrossRef]
- Nalumansi, A.; Lutalo, T.; Kayiwa, J.; Watera, C.; Balinandi, S.; Kiconco, J.; Nakaseegu, J.; Olara, D.; Odwilo, E.; Serwanga, J.; et al. Field evaluation of the performance of a SARS-CoV-2 antigen rapid diagnostic test in Uganda using nasopharyngeal samples. Int. J. Infect. Dis. 2020, 104, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Istituto Superiore di Sanità. Nota tecnica ad interim. Test di laboratorio per SARS-CoV-2 e loro uso in sanità pubblica; Ministero della Salute—Istituto Superiore di Sanità: Rome, Italy, 2020.
- Center for Disease Control and Prevention (CDC). Interim Guidance for Rapid Antigen Testing for SARS-CoV-2. Available online: https://www.cdc.gov/coronavirus/2019-ncov/lab/resources/antigen-tests-guidelines.html (accessed on 11 February 2021).
- Ministero della Salute. Disposizioni Attuative del Decreto-Legge 25 Marzo 2020, n. 19, Recante Misure Urgenti per Fronteggiare L’Emergenza Epidemiologica da COVID-19, e del Decreto-Legge 16 Maggio 2020, n. 33, Recante Ulteriori Misure Urgenti per Fronteggiare l’Emergenza Epidemiologica da COVID-19, 20A02717; Ministero della Salute: Rome, Italy, 2020.
- Liotti, F.M.; Menchinelli, G.; Lalle, E.; Palucci, I.; Marchetti, S.; Colavita, F. Performance of a novel diagnostic assay for rapid SARS-CoV-2 antigen detection in nasopharynx samples. Clin. Microbiol. Infect. 2020, 27, 487–488. [Google Scholar] [CrossRef] [PubMed]
- Colavita, F.; Lapa, D.; Carletti, F.; Lalle, E.; Messina, F.; Rueca, M. Virological characterization of the first two COVID-19 patients diagnosed in Italy: Phylogenetic analysis, virus shedding profile from different body sites and antibody response kinetics. Open Forum Infect. Dis. 2020, 7, ofaa403. [Google Scholar] [CrossRef] [PubMed]
- Cerutti, F.; Burdino, E.; Milia, M.G.; Allice, T.; Gregori, G.; Bruzzone, B.; Ghisetti, V. Urgent need of rapid tests for SARS CoV-2 antigen detection: Evaluation of the SD-Biosensor antigen test for SARS-CoV-2. J. Clin. Virol. 2020, 132, 104654. [Google Scholar] [CrossRef] [PubMed]
- Porte, L.; Legarraga, P.; Vollrath, V.; Aguilera, X.; Munita, J.M.; Araos, R. Evaluation of a novel antigen-based rapid detection test for the diagnosis of SARS-CoV-2 in respiratory samples. Int. J. Infect. Dis. 2020, 99, 328–333. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. COVID-19 Strategic Preparedness and Response Plan Operational Planning Guidelines to Support Country Preparedness and Response. 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/covid-19-sprp-unct-guidelines.pdf?sfvrsn=81ff43d8_4 (accessed on 11 February 2021).
- Pettengill, M.A.; McAdam, A.J. Can we test our way out of the COVID-19 pandemic? J. Clin. Microbiol. 2020, 58, e02225-20. [Google Scholar] [CrossRef] [PubMed]
| COI | RT-PCR Positive/N (% TP) | % FP by FIA |
|---|---|---|
| ≥1 | 207/603 (34.3%) | 65.7% |
| ≥3 | 186/228 (81.6%) | 18.4% |
| ≥5 | 175/188 (93.1%) | 6.9% |
| ≥8 | 159/165 (96.4%) | 3.6% |
| ≥10 | 152/154 (98.7%) | 1.3% |
| ≥15 | 138/139 (99.3%) | 0.7% |
| ≥20 | 127/127 (100%) | 0.0% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Colavita, F.; Vairo, F.; Meschi, S.; Valli, M.B.; Lalle, E.; Castilletti, C.; Fusco, D.; Spiga, G.; Bartoletti, P.; Ursino, S.; et al. COVID-19 Rapid Antigen Test as Screening Strategy at Points of Entry: Experience in Lazio Region, Central Italy, August–October 2020. Biomolecules 2021, 11, 425. https://doi.org/10.3390/biom11030425
Colavita F, Vairo F, Meschi S, Valli MB, Lalle E, Castilletti C, Fusco D, Spiga G, Bartoletti P, Ursino S, et al. COVID-19 Rapid Antigen Test as Screening Strategy at Points of Entry: Experience in Lazio Region, Central Italy, August–October 2020. Biomolecules. 2021; 11(3):425. https://doi.org/10.3390/biom11030425
Chicago/Turabian StyleColavita, Francesca, Francesco Vairo, Silvia Meschi, Maria Beatrice Valli, Eleonora Lalle, Concetta Castilletti, Danilo Fusco, Giuseppe Spiga, Pierluigi Bartoletti, Simona Ursino, and et al. 2021. "COVID-19 Rapid Antigen Test as Screening Strategy at Points of Entry: Experience in Lazio Region, Central Italy, August–October 2020" Biomolecules 11, no. 3: 425. https://doi.org/10.3390/biom11030425
APA StyleColavita, F., Vairo, F., Meschi, S., Valli, M. B., Lalle, E., Castilletti, C., Fusco, D., Spiga, G., Bartoletti, P., Ursino, S., Sanguinetti, M., Di Caro, A., Vaia, F., Ippolito, G., & Capobianchi, M. R. (2021). COVID-19 Rapid Antigen Test as Screening Strategy at Points of Entry: Experience in Lazio Region, Central Italy, August–October 2020. Biomolecules, 11(3), 425. https://doi.org/10.3390/biom11030425








