Oxygen: The Rate-Limiting Factor for Episodic Memory Performance, Even in Healthy Young Individuals
Abstract
1. Introduction
2. Materials and Methods
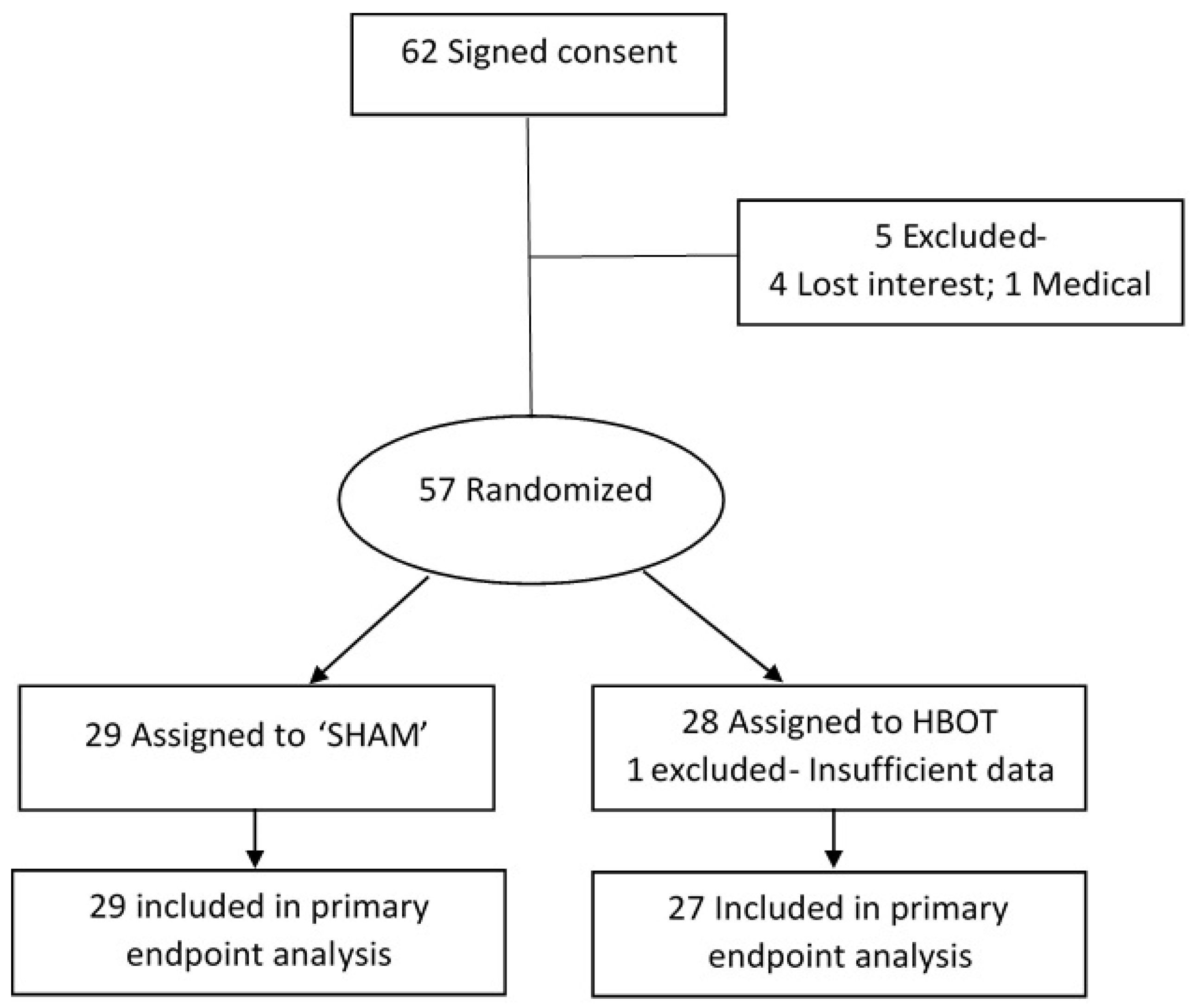
2.1. Participants
2.2. Protocol
2.2.1. Training Session
2.2.2. In-Chamber Evaluation Procedure
2.3. Outcome Measures
- Working memory:
- ○
- Auditory N-back test—In this test, the participants listened to a sequence of stimuli and were required to state whenever the presented stimulus matched the one preceding “n” steps earlier in the sequence. In the classic auditory N-back test, the participant hears a sequence that consist of letters. The N-back task captures the active part of working memory when N ≥ 2 [15]. In the current paradigm, we played a recording of six 20-digit strings read out loud at a constant pace. In the first stage, the instructions were to note whenever there was N = 1 repetition (i.e., “1”—“1”). In the second stage, the instructions were to note whenever there was N = 2 repetition (i.e., “1”—“3”—“1”). The accuracy score reflected the percentage of correctly traced repetitions out of the total repetitions from the second stage sequences. The number sequences were randomized using MATLAB version 7.10.0 (R2010a, The MathWorks Inc., Natick, MA, USA), while maintaining a rule that in each sequence there will be valid repetitions at a 1:3 probability.
- ○
- Digit-span test—In this test, a list of random numbers was read out loud at the rate of one per second. Subjects were asked to write as many digits as they recalled from each string. There were two versions of this test. In the forward version, the subjects recalled the sequence in the order that it was read. In the backward version, the subjects recalled the sequence in reverse order (i.e., from the last to first number read). Standard administration of the test started with a three-digit sequence and ended after the subject made two consecutive errors [16]. In the current research, we opted to prevent ceiling effects. Therefore, the initial sequence included seven digits in the forward version and five digits in the backward version. The test followed a standard procession in which there were two trials for each sequence length, and the subjects were required to recall them all until the test ended at sequence length = 10 in the forward test and sequence length = 8 in the backward test. Two scores were given for each version of the test. The “span score” is the highest sequence length correctly recalled. The “accuracy score” is the sum of digits correctly recalled in their position, across the whole test, regardless of the accuracy of the full sequences. To construct this test, random number sequences were generated for each sequence length.
- Divided attention: In this test, the subjects were instructed to divide attention between a visual search task (i.e., searching for the cluster 1-7-4 and circling all of its occurrences, among a long string of randomized numbers) and an auditory tracing task (i.e., recognizing verbally presented words containing the Hebrew letter “מ” (mem), which is equivalent to the English letter “M”, and writing them down). The words in the test were sampled from dictation lists for children in the third grade. Words containing the letter “מ” appeared among other words in random intervals. The test ended when all of the words were verbally presented (using a prepared recording). The score is the percent of correct responses, summed up from both channels.
- Episodic memory: In this test, subjects were required to pass a version of the California verbal learning test (CVLT) [17], which is a widely used episodic memory test. The test procedure consisted of two parts: (1) immediate learning—hearing a list of 16 nouns that are drawn from four semantic categories (i.e., fruits, clothes, etc.) and recalling as many words as possible immediately afterward. The order of words in the list was randomized. The list was read for a second time (after re-randomization of words). The score for this part is the sum of words retrieved in both trials. (2) Interference—recalling words from a second list of 16 nouns and then, once again, retrieving words from the initial list (without another exposure). The score for this part is total number of words recalled from the first list after interference.
- Cognitive processing speed: In this test, subjects solved a symbol search paradigm known to be a valid measure of cognitive processing speed in healthy older adults [18]. The assignment was time-limited and lasted five minutes. For each trial, two target symbols were presented on the left side of the paper. The subjects had to decide whether one of the two targets appeared in a series of five shapes on the right (shapes could reappear on the right in different sizes and/or different orientations) (see Figure 1). The score for this test is the sum of the correct responses.
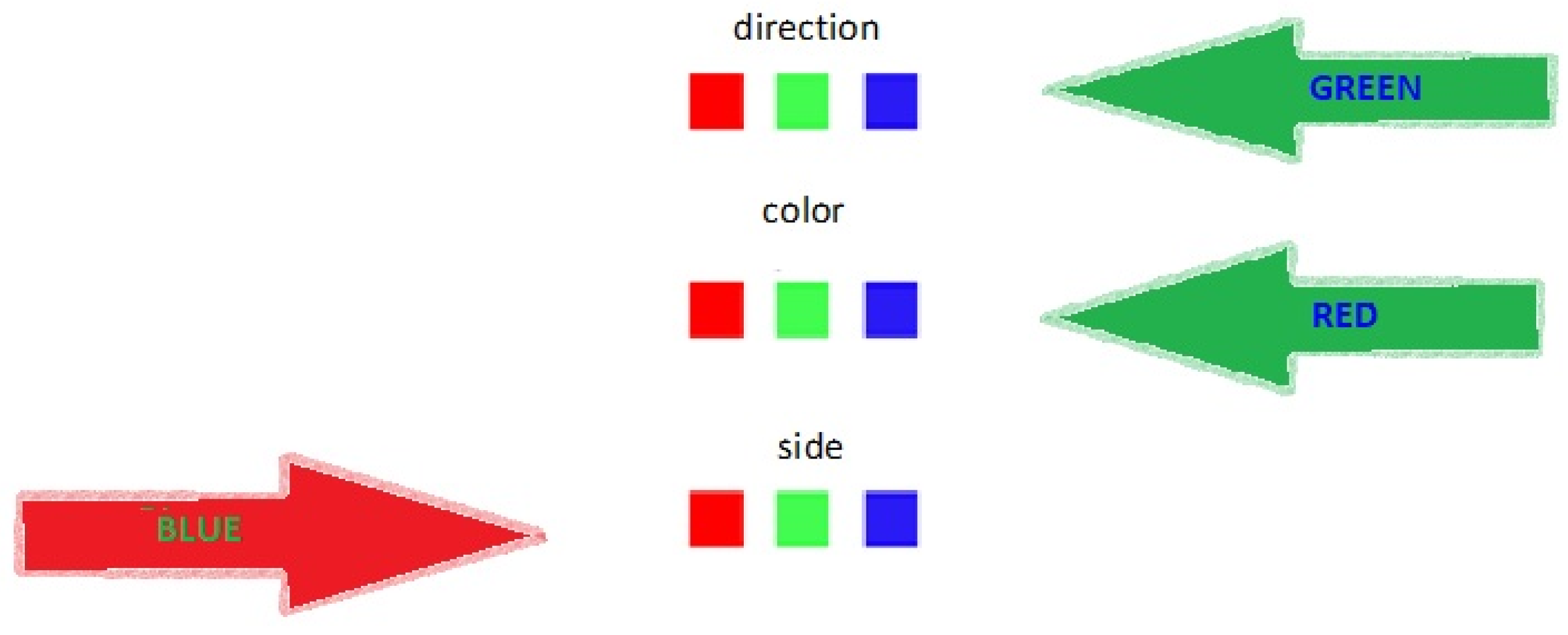
- Cognitive flexibility: In this test, subjects had to solve an augmented Stroop-like paradigm. Stroop tasks require subjects to inhibit their response to an attribute of the stimuli to attend to a competing attribute of the stimuli. The traditional Stroop-test requires the subject to neglect the content attribute of a word stimulus (e.g., “green”) to attend to the color attribute of the word (e.g., red font). The Stroop test is known to measure “response inhibition” [19]. In the current version of the test, subjects were shown colored arrows, either on the left or the right side of the page, either pointing to the left or to the right, with a “color name” written in the middle. The “color name” font color contradicted the color of the arrow (see Figure 2). The subjects had to attend either to the side of the page the arrow was on (“side”), to the direction the arrow pointed (“direction”), or to the color of the font of the word written on the arrow (“color”). The instructions changed intermittently between trials, and the test was timed (five minutes). The target score was the sum of correct responses.
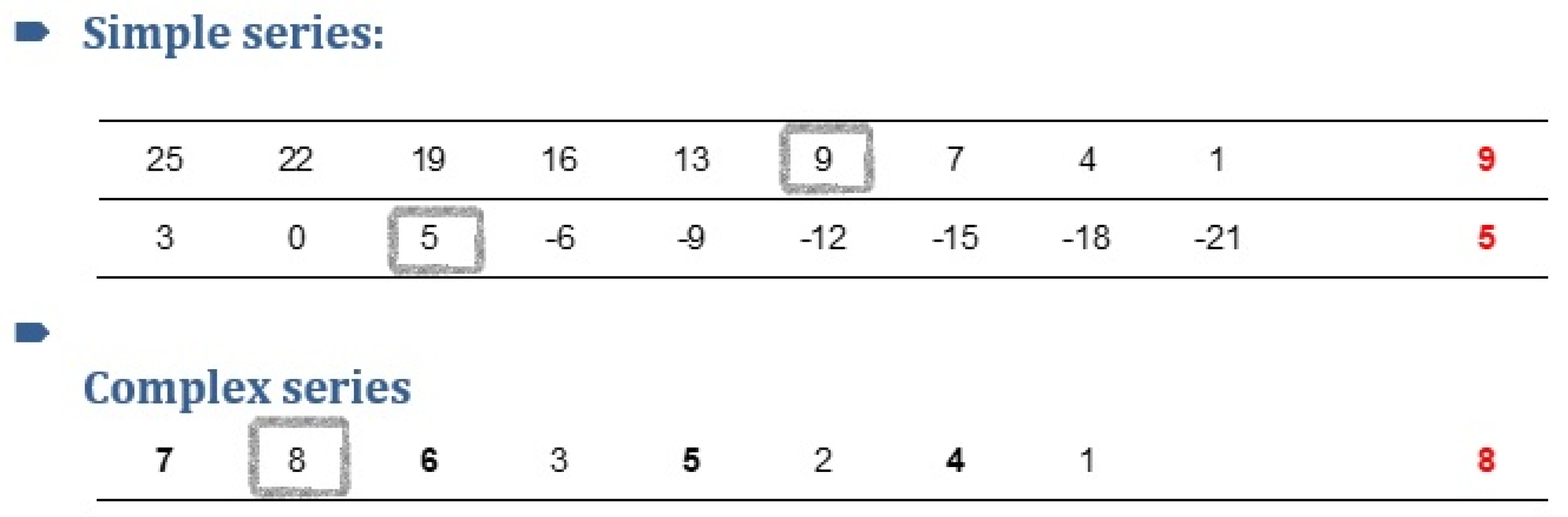
- Executive function (problem-solving): In this test, participants were required to solve an arithmetic series and trace irregular elements embedded in them, a task that is known to reflect executive functions (problem-solving) [20]. In the simple series part, subjects were given three minutes to solve up to 27 series. In the complex series part, they were given three minutes to solve up to 27 complex series, each composed of two interlocked series (see Figure 3). The score comprised the number of correct responses in each part.
2.4. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Oxford University Press. Lexico. Available online: https://www.lexico.com (accessed on 17 August 2020).
- Zauner, A.; Daugherty, W.P.; Bullock, M.R.; Warner, D.S. Brain oxygenation and energy metabolism: Part I-biological function and pathophysiology. Neurosurgery 2002, 51, 289–301. [Google Scholar]
- Bandettini, A.P. Neuronal or Hemodynamic? Grappling with the Functional MRI Signal. Brain Connect. 2014, 4, 487–498. [Google Scholar] [CrossRef]
- Huppert, F. Memory impairment associated with chronic hypoxia. Thorax 1982, 37, 858–860. [Google Scholar] [CrossRef] [PubMed]
- Stuss, D.T.; Peterkin, I.; Guzman, D.A.; Guzman, C.; Troyer, A.K. Chronic obstructive pulmonary disease: Effects of hypoxia on neurological and neuropsychological measures. J. Clin. Exp. Neuropsychol. 1997, 19, 515–524. [Google Scholar] [CrossRef]
- Areza-Fegyveres, R.; Kairalla, R.A.; Carvalho, C.R.; Nitrini, R. Cognition and chronic hypoxia in pulmonary diseases. Dement. Neuropsychol. 2010, 4, 14–22. [Google Scholar] [CrossRef]
- Kida, M.; Imai, A. Cognitive performance and event-related brain potentials under simulated high altitudes. J. Appl. Physiol. 1993, 74, 1735–1741. [Google Scholar] [CrossRef]
- Shukitt-Hale, B.; Stillman, M.J.; Welch, D.I.; Levy, A.; Devine, J.A.; Lieberman, H.R. Hypobaric hypoxia impairs spatial memory in an elevation-dependent fashion. Behav. Neural Boil. 1994, 62, 244–252. [Google Scholar] [CrossRef]
- Zani, A.; Tumminelli, C.; Proverbio, A.M. Electroencephalogram (EEG) Alpha Power as a Marker of Visuospatial Attention Orienting and Suppression in Normoxia and Hypoxia. An Exploratory Study. Brain Sci. 2020, 10, 140. [Google Scholar] [CrossRef]
- Malle, C.; Quinette, P.; Laisney, M.; Bourrilhon, C.; Boissin, J.; Desgranges, B.; Eustache, F.; Piérard, C. Working memory impairment in pilots exposed to acute hypobaric hypoxia. Aviat. Space Environ. Med. 2013, 84, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Ochi, G.; Kanazawa, Y.; Hyodo, K.; Suwabe, K.; Shimizu, T.; Fukuie, T.; Byun, K.; Soya, H. Hypoxia-induced lowered executive function depends on arterial oxygen desaturation. J. Physiol. Sci. 2018, 68, 847–853. [Google Scholar] [CrossRef]
- Scholey, A.; Moss, M.; Wesnes, K. Oxygen and cognitive performance: The temporal relationship between hyperoxia and enhanced memory. Psychopharmacology 1998, 140, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.-C.; Kwon, J.-H.; Lee, H.-W.; Tack, G.-R.; Lee, B.; Yi, J.-H.; Lee, S.-Y. Effects of high concentration oxygen administration onn-back task performance and physiological signals. Physiol. Meas. 2007, 28, 389–396. [Google Scholar] [CrossRef]
- Vadas, D.; Kalichman, L.; Hadanny, A.; Efrati, S. Hyperbaric Oxygen Environment Can Enhance Brain Activity and Multitasking Performance. Front. Integr. Neurosci. 2017, 11, 25. [Google Scholar] [CrossRef]
- Jaeggi, S.M.; Buschkuehl, M.; Perrig, W.J.; Meier, B. The concurrent validity of the N -back task as a working memory measure. Memory 2010, 18, 394–412. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, H.L.; Benton, A.L. Revised administration and scoring of the Digit Span Test. J. Consult. Psychol. 1957, 21, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Delis, D.C.; Freeland, J.; Kramer, J.H.; Kaplan, E. Integrating clinical assessment with cognitive neuroscience: Construct validation of the California Verbal Learning Test. J. Consult. Clin. Psychol. 1988, 56, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Liebel, S.W.; Uraina, S.C.; Xu, X.; Hannah, H.R.-J.; Brittany, E.H.; Nicolette, F.S.; Donald, L.; Beth, A.J.; Lawrence, H.S. An FMRI-compatible Symbol Search task. J. Int. Neuropsychol. Soc. 2015, 21, 231–238. [Google Scholar] [CrossRef]
- Simon, J.R.; Sudalaimuthu, P. Effects of S-R mapping and response modality on performance in a Stroop Task. J. Exp. Psychol. Hum. Percept. Perform. 1979, 5, 176–187. [Google Scholar] [CrossRef]
- Jenks, K.M.; De Moor, J.; Van Lieshout, E.C.D.M. Arithmetic difficulties in children with cerebral palsy are related to executive function and working memory. J. Child Psychol. Psychiatry 2009, 50, 824–833. [Google Scholar] [CrossRef]
- Cabeza, R.; Nyberg, L. Neural bases of learning and memory: Functional neuroimaging evidence. Curr. Opin. Neurol. 2000, 13, 415–421. [Google Scholar] [CrossRef]
- Allen, T.A.; Fortin, N.J. The evolution of episodic memory. Proc. Natl. Acad. Sci. USA 2013, 110, 10379–10386. [Google Scholar] [CrossRef]
- Nyberg, L. Functional brain imaging of episodic memory decline in ageing. J. Intern. Med. 2016, 281, 65–74. [Google Scholar] [CrossRef]
- Moscovitch, M.; Cabeza, R.; Winocur, G.; Nadel, L. Episodic Memory and Beyond: The Hippocampus and Neocortex in Transformation. Annu. Rev. Psychol. 2016, 67, 105–134. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Han, Z.; Huang, S.; Bai, R.; Ge, X.; Chen, F.; Lei, P. Intermittent hypoxia caused cognitive dysfunction relate to miRNAs dysregulation in hippocampus. Behav. Brain Res. 2017, 335, 80–87. [Google Scholar] [CrossRef]
- Ingraham, J.P.; Forbes, M.E.; Riddle, D.R.; Sonntag, W.E. Aging reduces hypoxia-induced microvascular growth in the rodent hippocampus. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2008, 63, 12–20. [Google Scholar] [CrossRef]
- Dunwiddie, T.V. Age-related differences in the in vitro rat hippocampus. Development of inhibition and the effects of hypoxia. Dev. Neurosci. 1981, 4, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Isarida, T. Influences of environmental-context changes on rehearsal effects in episodic memory. Jpn. J. Psychol. 1992, 63, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Ousdal, O.T.; Kaufmann, T.; Kolskår, K.; Vik, A.; Wehling, E.; Lundervold, A.J.; Lundervold, A.; Westlye, L.T. Longitudinal stability of the brain functional connectome is associated with episodic memory performance in aging. Hum. Brain Mapp. 2019, 41, 697–709. [Google Scholar] [CrossRef]
- Habib, R.; Nyberg, L.; Nilsson, L.-G. Cognitive and Non-Cognitive Factors Contributing to the Longitudinal Identification of Successful Older Adults in the Betula Study. Aging Neuropsychol. Cogn. 2007, 14, 257–273. [Google Scholar] [CrossRef]
- Efrati, S.; Hadanny, A.; Daphna-Tekoah, S.; Bechor, Y.; Tiberg, K.; Pik, N.; Suzin, G.; Lev-Wiesel, R. Recovery of Repressed Memories in Fibromyalgia Patients Treated With Hyperbaric Oxygen–Case Series Presentation and Suggested Bio-Psycho-Social Mechanism. Front. Psychol. 2018, 9, 848. [Google Scholar] [CrossRef]
- Moss, M.; Scholey, A.; Wesnes, K. Oxygen administration selectively enhances cognitive performance in healthy young adults: A placebo-controlled double-blind crossover study. Psychopharmacology 1998, 138, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Suzin, G.; Ravona-Springer, R.; Ash, E.L.; Davelaar, E.J.; Usher, M. Differences in Semantic Memory Encoding Strategies in Young, Healthy Old and MCI Patients. Front. Aging Neurosci. 2019, 11, 306. [Google Scholar] [CrossRef]

| Domain | Test | Primary Outcome Measure |
|---|---|---|
| Working memory | N-back | Percentage accuracy |
| Digit span | Span score; accuracy score | |
| Divided attention | Multi-task | Percentage accuracy |
| Episodic memory | Word lists | Sum immediate retrieval; retrieval after interference |
| Cognitive processing speed | Symbol search | Sum accuracy |
| Cognitive flexibility | Stroop | Sum accuracy |
| Problem solving | Arithmetic | Sum accuracy |
| HBOT (N = 27) | SHAM (N = 29) | p Value | |
|---|---|---|---|
| Mean Age (Stdev) | 27.92 ± 4.77 | 26.96 ± 3.95 | 0.41 |
| Years of Education (Stdev) | 15.36 ± 2.11 | 14.64 ± 1.7 | 0.178 |
| Gender (% Females) | 59% | 45% | 0.28 |
| Cognitive Domain | HBOT (N = 27) | SHAM (N = 29) | t-Test (p) | FDR (p) | Effect Size (Cohen’s D) |
|---|---|---|---|---|---|
| Digit span (forward: highest correct) | 7.78 (1.5) | 7.46 (0.85) | 0.37 | 0.55 | 0.26 |
| Digit span (forward: overall correct) | 41.3 (11.98) | 38.27 (6.55) | 0.25 | 0.75 | 0.31 |
| Digit span (backward: highest correct) | 7.34 (1.52) | 6.85 (1.81) | 0.28 | 0.67 | 0.29 |
| Digit span (backward: overall correct) | 40.85 (10.62) | 42 (9.04) | 0.66 | 0.79 | 0.12 |
| Stroop (% correct) | 59.03 (15.39) | 62.96 (15.99) | 0.35 | 0.6 | 0.25 |
| Multi-tasking (% total accuracy) | 66.6 (7.97) | 64.24 (8.47) | 0.29 | 0.58 | 0.29 |
| Symbol search (no. correct) | 31.66 (6.3) | 33.03 (8.47) | 0.49 | 0.65 | 0.18 |
| N-back (% correct) | 91.19 (9.57) | 90.42 (10.05) | 0.76 | 0.83 | 0.08 |
| Series simple (no. correct) | 15.3 (5.9) | 15.27 (5.48) | 0.98 | 0.98 | 0.00 |
| Series complex (no. correct) | 12 (4.51) | 13.41 (4.35) | 0.23 | 0.92 | 0.32 |
| CVLT (total correct) | 22.4 (2.96) | 20.51 (3.75) | 0.04 | 0.24 | 0.54 |
| CVLT (no. of words—post interference) | 11.33 (2.3) | 9.03 (2.93) | 0.001 | 0.012 * | 0.85 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzin, G.; Halpert Frolinger, T.; Yogev, D.; Hadanny, A.; Catalogna, M.; Rassovsky, Y.; Efrati, S. Oxygen: The Rate-Limiting Factor for Episodic Memory Performance, Even in Healthy Young Individuals. Biomolecules 2020, 10, 1328. https://doi.org/10.3390/biom10091328
Suzin G, Halpert Frolinger T, Yogev D, Hadanny A, Catalogna M, Rassovsky Y, Efrati S. Oxygen: The Rate-Limiting Factor for Episodic Memory Performance, Even in Healthy Young Individuals. Biomolecules. 2020; 10(9):1328. https://doi.org/10.3390/biom10091328
Chicago/Turabian StyleSuzin, Gil, Tom Halpert Frolinger, Dror Yogev, Amir Hadanny, Merav Catalogna, Yuri Rassovsky, and Shai Efrati. 2020. "Oxygen: The Rate-Limiting Factor for Episodic Memory Performance, Even in Healthy Young Individuals" Biomolecules 10, no. 9: 1328. https://doi.org/10.3390/biom10091328
APA StyleSuzin, G., Halpert Frolinger, T., Yogev, D., Hadanny, A., Catalogna, M., Rassovsky, Y., & Efrati, S. (2020). Oxygen: The Rate-Limiting Factor for Episodic Memory Performance, Even in Healthy Young Individuals. Biomolecules, 10(9), 1328. https://doi.org/10.3390/biom10091328







