Molecular Biomarkers of Oxygen Therapy in Patients with Diabetic Foot Ulcers
Abstract
1. Introduction
2. Cell Metabolism and Energy
3. Molecular Biomarkers in Growth Factor Signaling Transduction
4. Collagen Formation
5. Angiogenesis Biomarkers
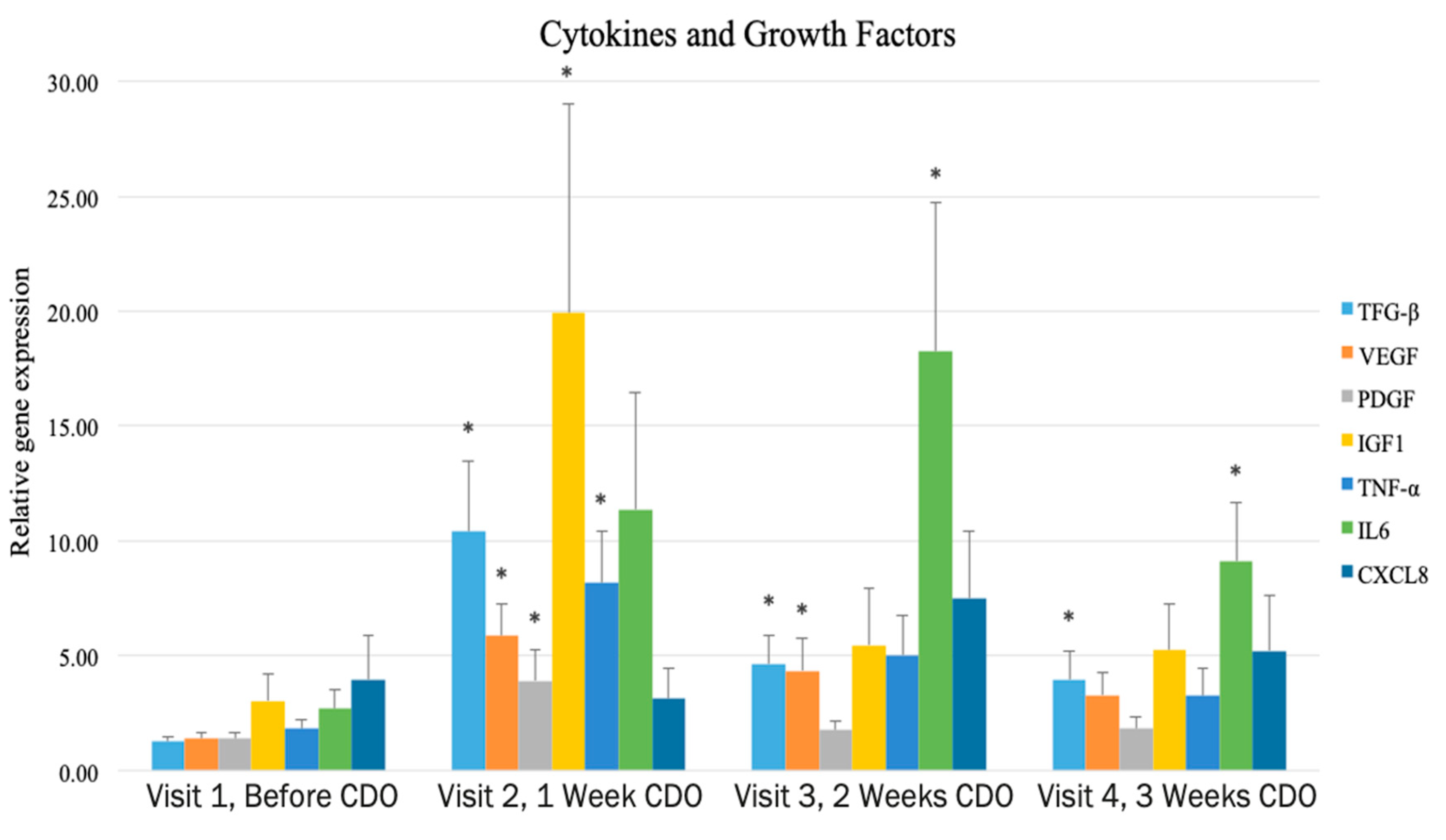
6. Respiratory Burst Process and Cytokine Production
7. Cell Proliferation Molecular Markers
8. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Armstrong, D.G.; Boulton, A.J.M.; Bus, S.A. Diabetic Foot Ulcers and Their Recurrence. N. Engl. J. Med. 2017, 376, 2367–2375. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lazzarini, P.A.; McPhail, S.M.; Van Netten, J.J.; Armstrong, D.G.; Pacella, R.E. Global Disability Burdens of Diabetes-Related Lower-Extremity Complications in 1990 and 2016. Diabetes Care 2020, 43, 964–974. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, B.A.; Senneville, É.; Urbančič-Rovan, V.; Peters, E.J.; on behalf of the International Working Group on the Diabetic Foot(IWGDF); Abbas, Z.G.; Aragón-Sánchez, J.; Diggle, M.; Embil, J.M.; Kono, S.; et al. Guidelines on the diagnosis and treatment of foot infection in persons with diabetes (IWGDF 2019 update). Diabet. Metab. Res. Rev. 2020, 36 (Suppl. 1), e3280. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.G.; A Lavery, L.; Frykberg, R.G.; Wu, S.C.; Boulton, A.J. Validation of a diabetic foot surgery classification. Int. Wound J. 2006, 3, 240–246. [Google Scholar] [CrossRef]
- Skrepnek, G.H.; Armstrong, D.G.; Mills, J.L. 2,500,000 Troubled Soles: Ten-Year Analysis of Diabetic Foot Infections in the United States. J. Vasc. Surg. 2013, 58, 558. [Google Scholar] [CrossRef][Green Version]
- Skrepnek, G.H.; Mills, J.L., Sr.; Armstrong, D.G. A Diabetic Emergency One Million Feet Long: Disparities and Burdens of Illness among Diabetic Foot Ulcer Cases within Emergency Departments in the United States, 2006–2010. PLoS ONE 2015, 10, e0134914. [Google Scholar] [CrossRef]
- Armstrong, D.G.; A Lavery, L.; Harkless, L.B.; Van Houtum, W.H. Amputation and reamputation of the diabetic foot. J. Am. Podiatr. Med. Assoc. 1997, 87, 255–259. [Google Scholar] [CrossRef]
- Lipsky, B.A.; Berendt, A.R.; Cornia, P.B.; Pile, J.C.; Peters, E.J.G.; Armstrong, D.G.; Deery, H.G.; Embil, J.M.; Joseph, W.S.; Karchmer, A.W.; et al. Executive Summary: 2012 Infectious Diseases Society of America Clinical Practice Guideline for the Diagnosis and Treatment of Diabetic Foot Infections. Clin. Infect. Dis. 2012, 54, 1679–1684. [Google Scholar] [CrossRef] [PubMed]
- Boulton, A.J.; Armstrong, D.G.; Kirsner, R.S.; Attinger, C.E.; Lavery, L.A.; Lipsky, B.A.; Mills, J.L.; Steinberg, J.S. Diagnosis and Management of Diabetic Foot Complications; American Diabetes Association: Arlington, VA, USA, 2018. [Google Scholar] [CrossRef]
- Saluja, S.; Anderson, S.G.; Hambleton, I.; Shoo, H.; Livingston, M.; Jude, E.B.; Lunt, M.; Dunn, G.; Heald, A.H. Foot ulceration and its association with mortality in diabetes mellitus: A meta-analysis. Diabet. Med. 2020, 37, 211–218. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Swerdlow, M.A.; Armstrong, A.A.; Conte, M.S.; Padula, W.V.; Bus, S.A. Five year mortality and direct costs of care for people with diabetic foot complications are comparable to cancer. J. Foot Ankle Res. 2020, 13, 16. [Google Scholar] [CrossRef]
- Conte, M.S.; Bradbury, A.W.; Kolh, P.; White, J.V.; Dick, F.; Fitridge, R.; Mills, J.L.; Ricco, J.-B.; Suresh, K.R.; Murad, M.H.; et al. Global vascular guidelines on the management of chronic limb-threatening ischemia. J. Vasc. Surg. 2019, 69, 3S–125S.e40. [Google Scholar] [CrossRef]
- Mills, J.L., Sr.; Conte, M.S.; Armstrong, D.G.; Pomposelli, F.B.; Schanzer, A.; Sidawy, A.N.; Andros, G. Society for Vascular Surgery Lower Extremity Guidelines Committee. The Society for Vascular Surgery Lower Extremity Threatened Limb Classification System: Risk stratification based on Wound, Ischemia, and foot Infection (WIfI). J. Vasc. Surg. 2014, 59, 220–234.e2. [Google Scholar] [CrossRef] [PubMed]
- Nussbaum, S.R.; Carter, M.J.; Fife, C.E.; DaVanzo, J.; Haught, R.; Nusgart, M.; Cartwright, D. An Economic Evaluation of the Impact, Cost, and Medicare Policy Implications of Chronic Nonhealing Wounds. Value Health 2018, 21, 27–32. [Google Scholar] [CrossRef]
- Barshes, N.R.; Sigireddi, M.; Wrobel, J.S.; Mahankali, A.; Robbins, J.M.; Kougias, P.; Armstrong, D.G. The system of care for the diabetic foot: Objectives, outcomes, and opportunities. Diabet. Foot Ankle 2013, 4, 21847. [Google Scholar] [CrossRef] [PubMed]
- Howard, M.A.; Asmis, R.; Evans, K.K.; Mustoe, T.A. Oxygen and wound care: A review of current therapeutic modalities and future direction. Wound Repair Regen. 2013, 21, 503–511. [Google Scholar] [CrossRef]
- Hopf, H.W.; Hunt, T.K.; West, J.M.; Blomquist, P.; Goodson, W.; Jensen, J.; Jonsson, K.; Paty, P.B.; Rabkin, J.M.; Upton, R.A.; et al. Wound Tissue Oxygen Tension Predicts the Risk of Wound Infection in Surgical Patients. Arch. Surg. 1997, 132, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- Gottrup, F.; Firmin, R.; Rabkin, J.; Halliday, B.J.; Hunt, T.K. Directly measured tissue oxygen tension and arterial oxygen tension assess tissue perfusion. Crit. Care Med. 1987, 15, 1030–1036. [Google Scholar] [CrossRef]
- Hopf, H.; West, J.; Hunt, T. Clonidine increases tissue oxygen in patients with local tissue hypoxia in non-healing wounds. Wound Repair Regen. 1996, 4, A129. [Google Scholar]
- Edwards, J.; Stapley, S. Debridement of diabetic foot ulcers. Cochrane Database Syst. Rev. 2010, CD003556. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Lavery, L.A.; Nixon, B.P.; Boulton, A.J.M. It’s Not What You Put On, but What You Take Off: Techniques for Debriding and Off-Loading the Diabetic Foot Wound. Clin. Infect. Dis. 2004, 39, S92–S99. [Google Scholar] [CrossRef]
- Lebrun, E.; Tomic-Canic, M.; Kirsner, R.S. The role of surgical debridement in healing of diabetic foot ulcers. Wound Repair Regen. 2010, 18, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Lavery, L.A.; Niederauer, M.Q.; Papas, K.K.; Armstrong, D.G. Does Debridement Improve Clinical Outcomes in People with DFU Ulcers Treated with CDO? Wounds 2019, 31, 246–251. [Google Scholar] [PubMed]
- Allen, D.B.; Maguire, J.J.; Mahdavian, M.; Wicke, C.; Marcocci, L.; Scheuenstuhl, H.; Chang, M.; Le, A.X.; Hopf, H.W.; Hunt, T.K. Wound Hypoxia and Acidosis Limit Neutrophil Bacterial Killing Mechanisms. Arch. Surg. 1997, 132, 991–996. [Google Scholar] [CrossRef] [PubMed]
- Hopf, H.W.; Humphrey, L.M.; Puzziferri, N.; West, J.M.; Attinger, C.E.; Hunt, T.K. Adjuncts to preparing wounds for closure: Hyperbaric oxygen, growth factors, skin substitutes, negative pressure wound therapy (vacuum-assisted closure). Foot Ankle Clin. 2001, 6, 661–682. [Google Scholar] [CrossRef]
- Sen, C.K. Wound healing essentials: Let there be oxygen. Wound Repair Regen. 2009, 17, 1–18. [Google Scholar] [CrossRef]
- Gupta, A.; Raghubir, R. Energy metabolism in the granulation tissue of diabetic rats during cutaneous wound healing. Mol. Cell. Biochem. 2005, 270, 71–77. [Google Scholar] [CrossRef]
- Hohn, D.C.; Ponce, B.; Burton, R.W.; Hunt, T.K. Antimicrobial systems of the surgical wound. I. A comparison of oxidative metabolism and microbicidal capacity of phagocytes from wounds and from peripheral blood. Am. J. Surg. 1977, 133, 597–600. [Google Scholar] [CrossRef]
- Matsuda, T.; Clark, N.; Hariyani, G.D.; Bryant, R.S.; Hanumadass, M.L.; Kagan, R.J. The Effect of Burn Wound Size on Resting Energy Expenditure. J. Trauma Inj. Infect. Crit. Care 1987, 27, 115–118. [Google Scholar] [CrossRef]
- Im, M.J.; Hoopes, J.E. Energy metabolism in healing skin wounds. J. Surg. Res. 1970, 10, 459–464. [Google Scholar] [CrossRef]
- Tandara, A.A.; Mustoe, T.A. Oxygen in Wound Healing? More than a Nutrient. World J. Surg. 2004, 28, 294–300. [Google Scholar] [CrossRef]
- LaVan, F.B.; Hunt, T.K. Oxygen and Wound Healing. Clin. Plast. Surg. 1990, 17, 463–472. [Google Scholar] [CrossRef]
- Hess, C.L.; Howard, M.A.; Attinger, C.E. A Review of Mechanical Adjuncts in Wound Healing: Hydrotherapy, Ultrasound, Negative Pressure Therapy, Hyperbaric Oxygen, and Electrostimulation. Ann. Plast. Surg. 2003, 51, 210–218. [Google Scholar] [CrossRef]
- Hunt, T.K. Basic Principles of Wound Healing. J. Trauma Inj. Infect. Crit. Care 1990, 30, 122–128. [Google Scholar] [CrossRef]
- Milton, S.L.; Prentice, H.M. Beyond anoxia: The physiology of metabolic downregulation and recovery in the anoxia-tolerant turtle. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2007, 147, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Stys, P.K.; Ransom, B.R.; Waxman, S.G.; Davis, P.K. Role of extracellular calcium in anoxic injury of mammalian central white matter. Proc. Natl. Acad. Sci. USA 1990, 87, 4212–4216. [Google Scholar] [CrossRef]
- Sen, C.K.; Roy, S. Redox signals in wound healing. Biochim. Biophys. Acta (BBA) Gen. Subj. 2008, 1780, 1348–1361. [Google Scholar] [CrossRef] [PubMed]
- Niederauer, M.Q.; Michalek, J.E.; Liu, Q.; Papas, K.K.; Lavery, L.A.; Armstrong, D.G. Continuous diffusion of oxygen improves diabetic foot ulcer healing when compared with a placebo control: A randomised, double-blind, multicentre study. J. Wound Care 2018, 27, S30–S45. [Google Scholar] [CrossRef]
- Frykberg, R.G.; Franks, P.; Edmonds, M.; Brantley, J.N.; Téot, L.; Wild, T.; Garoufalis, M.G.; Lee, A.M.; Thompson, J.A.; Reach, G.; et al. A Multinational, Multicenter, Randomized, Double-Blinded, Placebo-Controlled Trial to Evaluate the Efficacy of Cyclical Topical Wound Oxygen (TWO2) Therapy in the Treatment of Chronic Diabetic Foot Ulcers: The TWO2 Study. Diabetes Care 2019, 43, 616–624. [Google Scholar] [CrossRef]
- Serena, T.E.; Bullock, N.M.; Cole, W.; Lantis, J.; Li, L.; Moore, S.; Patel, K.; Sabo, M.; Wahab, N.; Price, P. Topical oxygen therapy in the treatment of diabetic foot ulcers: A multicentre, open, randomised controlled clinical trial. J. Wound Care 2021, 30, S7–S14. [Google Scholar] [CrossRef]
- Yu, J.; Lu, S.; McLaren, A.-M.; Perry, J.A.; Cross, K.M. Topical oxygen therapy results in complete wound healing in diabetic foot ulcers. Wound Repair Regen. 2016, 24, 1066–1072. [Google Scholar] [CrossRef]
- Sundaresan, M.; Yu, Z.-X.; Ferrans, V.J.; Sulciner, D.J.; Gutkind, J.S.; Irani, K.; Goldschmidt-Clermont, P.J.; Finkel, T. Regulation of reactive-oxygen-species generation in fibroblasts by Rac 1. Biochem. J. 1996, 318, 379–382. [Google Scholar] [CrossRef]
- Sen, C.K.; Khanna, S.; Babior, B.M.; Hunt, T.K.; Ellison, E.C.; Roy, S. Oxidant-induced Vascular Endothelial Growth Factor Expression in Human Keratinocytes and Cutaneous Wound Healing. J. Biol. Chem. 2002, 277, 33284–33290. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.; Hunt, T.K.; Hussain, M.Z. Hydrogen peroxide stimulates macrophage vascular endothelial growth factor release. Am. J. Physiol. Circ. Physiol. 2001, 280, H2357–H2363. [Google Scholar] [CrossRef]
- Sundaresan, M.; Yu, Z.-X.; Ferrans, V.J.; Irani, K.; Finkel, T. Requirement for generation of H2O2 for platelet-derived growth factor signal transduction. Science 1995, 270, 296–299. [Google Scholar] [CrossRef] [PubMed]
- Gordillo, G.M.; Sen, C.K. Revisiting the essential role of oxygen in wound healing. Am. J. Surg. 2003, 186, 259–263. [Google Scholar] [CrossRef]
- Sen, C.K. The general case for redox control of wound repair. Wound Repair Regen. 2003, 11, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Khanna, S.; Sen, C.K. Redox regulation of the VEGF signaling path and tissue vascularization: Hydrogen peroxide, the common link between physical exercise and cutaneous wound healing. Free Radic. Biol. Med. 2008, 44, 180–192. [Google Scholar] [CrossRef]
- Roy, S.; Khanna, S.; Rink, C.; Biswas, S.; Sen, C.K. Characterization of the acute temporal changes in excisional murine cutaneous wound inflammation by screening of the wound-edge transcriptome. Physiol. Genom. 2008, 34, 162–184. [Google Scholar] [CrossRef] [PubMed]
- Lavery, L.A.; Killeen, A.L.; Farrar, D.; Akgul, Y.; Crisologo, P.A.; Malone, M.; Davis, K.E. The effect of continuous diffusion of oxygen treatment on cytokines, perfusion, bacterial load, and healing in patients with diabetic foot ulcers. Int. Wound J. 2020, 17, 1986–1995. [Google Scholar] [CrossRef]
- Hunt, T.K.; Zederfeldt, B.; Goldstick, T.K. Oxygen and healing. Am. J. Surg. 1969, 118, 521–525. [Google Scholar] [CrossRef]
- Prockop, D.; Kivirikko, K.; Tuderman, L.; Guzman, N. The biosynthesis of collagen and its disorders (part 1). N. Engl. J. Med. 1979, 301, 13–23. [Google Scholar] [CrossRef]
- Prockop, D.; Kivirikko, K.; Tuderman, L.; Guzman, N. The biosynthesis of collagen and its disorder (part 2). N. Engl. J. Med. 1979, 301, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, K.; Jensen, J.; Goodson, W.; Scheuenstuhl, H.; West, J.; Hopf, H.; Hunt, T. Tissue oxygenation, anemia, and perfusion in relation to wound healing in surgical patients. Ann. Surg. 1991, 214, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Niinikoski, J. Effect of oxygen supply on wound healing and formation of experimental granulation tissue. Acta Physiol. Scand. Suppl. 1969, 334, 1–72. [Google Scholar]
- Hunt, T.K.; Pai, M.P. The effect of varying ambient oxygen tensions on wound metabolism and collagen synthesis. Surg. Gynecol. Obstet. 1972, 135, 561–567. [Google Scholar] [PubMed]
- Stephens, F.O.; Hunt, T.K. Effect of Changes in Inspired Oxygen and Carbon Dioxide Tensions on Wound Tensile Strength. Ann. Surg. 1971, 173, 515–519. [Google Scholar] [CrossRef]
- Hutton, J.J.; Tappel, A.; Udenfriend, S. Cofactor and substrate requirements of collagen proline hydroxylase. Arch. Biochem. Biophys. 1967, 118, 231–240. [Google Scholar] [CrossRef]
- Myllyla, R.; Tuderman, L.; Kivirikko, K. Mechanism of the prolyl hydroxlase reaction. 2. Kinetic analysis of the reaction se-quence. Eur. J. Biochem. 1977, 80, 349–357. [Google Scholar] [CrossRef]
- Asmis, R.; Qiao, M.; Zhao, Q. Low-Flow Oxygenation of Full-Excisional Skin Wounds on Diabetic Mice Improves Wound Heal-ing by Accelerating Wound Closure and Reepithelialization. Int. Wound J. 2010, 7, 349–357. [Google Scholar] [CrossRef]
- Hartmann, M.; Jönsson, K.; Zederfeldt, B. Effect of tissue perfusion and oxygenation on accumulation of collagen in healing wounds. Randomized study in patients after major abdominal operations. Eur. J. Surg. 1992, 158, 521–526. [Google Scholar]
- Hopf, H.W.; Gibson, J.J.; Angeles, A.P.; Constant, J.S.; Feng, J.J.; Rollins, M.D.; Zamirul Hussain, M.; Hunt, T.K. Hyperoxia and angiogenesis. Wound Repair Regen. 2005, 13, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Mussini, E.; Hutton, J.J., Jr.; Udenfriend, S. Collagen proline hydroxylase in wound healing, granuloma formation, scurvy, and growth. Science 1967, 157, 927–929. [Google Scholar] [CrossRef]
- Berthod, F.; Germain, L.; Tremblay, N.; Auger, F.A. Extracellular matrix deposition by fibroblasts is necessary to promote capil-lary-like tube formation in vitro. J. Cell. Physiol. 2006, 207, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Hunt, T.K.; Aslam, R.S.; Beckert, S.; Wagner, S.; Ghani, Q.P.; Hussain, M.Z.; Roy, S.; Sen, C.K. Aerobically derived lactate stimulates re-vascularization and tissue repair via redox mechanisms. Antioxid. Redox Signal. 2007, 9, 1115–1124. [Google Scholar] [CrossRef]
- Knighton, D.R.; Silver, I.A.; Hunt, T.K. Regulation of wound-healing angiogenesis-effect of oxygen gradients and inspired oxygen concentration. Surgery 1981, 90, 262–270. [Google Scholar] [PubMed]
- Sheikh, A.Y.; Rollins, M.D.; Hopf, H.W.; Hunt, T.K. Hyperoxia improves microvascular perfusion in a murine wound model. Wound Repair Regen. 2005, 13, 303–308. [Google Scholar] [CrossRef]
- Maniscalco, W.M.; Watkins, R.H.; Finkelstein, J.N.; Campbell, M.H. Vascular endothelial growth factor mRNA increases in alveolar epithelial cells during recovery from oxygen injury. Am. J. Respir. Cell Mol. Biol. 1995, 13, 377–386. [Google Scholar] [CrossRef]
- Deaton, P.R.; McKellar, C.T.; Culbreth, R.; Veal, C.F.; Cooper, J.A. Hyperoxia stimulates interleukin-8 release from alveolar macrophages and U937 cells: Attenuation by dexamethasone. Am. J. Physiol. Lung Cell. Mol. Physiol. 1994, 267, 187–192. [Google Scholar] [CrossRef]
- Darrington, R.S.; Godden, D.J.; Park, M.S.; Ralston, S.H.; Wallace, H.M. The effect of hyperoxia on the expression of cytokine mRNA in endothelial cells. Biochem. Soc. Trans. 1997, 25, 292S. [Google Scholar] [CrossRef]
- Sheikh, A.Y.; Gibson, J.J.; Rollins, M.D.; Hopf, H.W.; Hussain, Z.; Hunt, T.K. Effect of Hyperoxia on Vascular Endothelial Growth Factor Levels in a Wound Model. Arch. Surg. 2000, 135, 1293–1297. [Google Scholar] [CrossRef]
- Shenberger, J.S.; Zhang, L.; Powell, R.J.; Barchowsky, A. Hyperoxia enhances VEGF release from A549 cells via post-transcriptional processes. Free Radic. Biol. Med. 2007, 43, 844–852. [Google Scholar] [CrossRef]
- Gordillo, G.M.; Roy, S.; Khanna, S.; Schlanger, R.; Khandelwal, S.; Phillips, G.; Sen, C.K. Topical oxygen therapy induces vascular endothelial growth factor expression and im-proves closure of clinically presented chronic wounds. Clin. Exp. Pharmacol. Physiol. 2008, 35, 957–964. [Google Scholar] [CrossRef]
- Heng, M.C.; Harker, J.; Csathy, G.; Marshall, C.; Brazier, J.; Sumampong, S.; Paterno Gomez, E. Angiogenesis in necrotic ulcers treated with hyperbaric oxygen. Ostomy Wound Manag. 2000, 46, 18–28. [Google Scholar]
- Brown, J.R.; Goldblatt, D.; Buddle, J.; Morton, L.; Thrasher, A.J. Diminished production of anti-inflammatory mediators during neutrophil apoptosis and macrophage phagocytosis in chronic granulomatous disease (CGD). J. Leukoc. Biol. 2003, 73, 591–599. [Google Scholar] [CrossRef]
- Babior, B.M. Oxygen-dependent microbial killing by phagocytes (first of two parts). N. Engl. J. Med. 1978, 298, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Rabkin, J.M.; Hunt, T.K. Infection and oxygen. In Problem Wounds: The Role of Oxygen; Davis, J.C., Hunt, T.K., Eds.; Elsevier: New York, NY, USA, 1988; pp. 1–16. [Google Scholar]
- Cianci, P. Advances in the treatment of the diabetic foot: Is there a role for adjunctive hyperbaric oxygen therapy? Wound Repair Regen. 2004, 12, 2–10. [Google Scholar] [CrossRef]
- Wattel, F.; Mathieu, D. Oxygen and wound healing. Bull. Acad. Natl. Med. 2005, 189, 853–864. [Google Scholar]
- Knighton, D.; Halliday, B.; Hunt, T. Oxygen as an antibiotic: The effect of inspired oxygen on infection. Arch. Surg. 1984, 119, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Knighton, D.R.; Halliday, B.; Hunt, T.K. Oxygen as an Antibiotic. Arch. Surg. 1986, 121, 191–195. [Google Scholar] [CrossRef]
- Driver, V.R.; Yao, M.; Kantarci, A.; Gu, G.; Park, N.; Hasturk, H. A prospective, randomized clinical study evaluating the effect of transdermal continuous oxygen therapy on biological processes and foot ulcer healing in persons with diabetes mellitus. Ostomy Wound Manag. 2013, 59, 19–26. [Google Scholar]
- Weissenbach, M.; Clahsen, T.; Weber, C.; Spitzer, D.; Wirth, D.; Vestweber, D.; Heinrich, P.C.; Schaper, F. Interleukin-6 is a direct mediator of T cell migration. Eur. J. Immunol. 2004, 34, 2895–2906. [Google Scholar] [CrossRef]
- Wright, H.; Cross, A.L.; Edwards, S.W.; Moots, R.J. Effects of IL-6 and IL-6 blockade on neutrophil function in vitro and in vivo. Rheumatology 2014, 53, 1321–1331. [Google Scholar] [CrossRef]
- Pandit, A.S.; Faldman, D.S. Effect of oxygen treatment and dressing oxygen permeability on wound healing. Wound Repair Re-gen. 1994, 2, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, K.A.; Liu, Z.-J.; Xiao, M.; Chen, H.; Goldstein, L.J.; Buerk, D.G.; Nedeau, A.; Thom, S.R.; Velazquez, O.C. Diabetic impairments in NO-mediated endothelial progenitor cell mobilization and homing are reversed by hyperoxia and SDF-1α. J. Clin. Investig. 2007, 117, 1249–1259. [Google Scholar] [CrossRef]
- Goldstein, L.J.; Gallagher, K.A.; Bauer, S.M.; Bauer, R.J.; Baireddy, V.; Liu, Z.J.; Buerk, D.G.; Thom, S.R.; Velazquez, O.C. Endothelial pro-genitor cell release into circulation is triggered by hyperoxia-induced increases in bone marrow nitric oxide. Stem Cells 2006, 24, 2309–2318. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, K.A.; Goldstein, L.J.; Thom, S.R.; Velazquez, O.C. Hyperbaric Oxygen and Bone Marrow–Derived Endothelial Progenitor Cells in Diabetic Wound Healing. Vascular 2006, 14, 328–337. [Google Scholar] [CrossRef]
- Thom, S.R.; Bhopale, V.M.; Velazquez, O.C.; Goldstein, L.J.; Thom, L.H.; Buerk, D.G. Stem cell mobilization by hyperbaric oxygen. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H1378–H1386. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Jude, E.B. The Role of Matrix Metalloproteinases in Wound Healing. J. Am. Podiatr. Med. Assoc. 2002, 92, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.G.; Gurtner, G.C. A histologically hostile environment made more hospitable? Nat. Rev. Endocrinol. 2018, 14, 511–512. [Google Scholar] [CrossRef] [PubMed]
| Growth Factors | |
|---|---|
| IGF-1 | protein production and cell proliferation and migration |
| PDGF | cell growth and division and chemotaxis |
| TGF-β | angiogenesis, fibroblast proliferation, collagen synthesis and deposition, extracellular matrix (ECM) remodeling, tissue remodeling, granulation tissue stimulant and anti-inflammatory mediator |
| VEGF | angiogenesis and collagen deposition and epithelialization |
| IGF-1 | protein production and cell proliferation and migration |
| cytokines | |
| CXCL8 | angiogenesis, epithelialization, fibroblast migration and inflammatory mediator |
| IL-6 | leukocyte infiltration, angiogenesis, collagen accumulation, anti-inflammatory, granulation tissue stimulant and mitogenic |
| TNF-α | leukocyte recruitment, cell regulator, ECM synthesis and inflammatory mediator |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oropallo, A.R.; Serena, T.E.; Armstrong, D.G.; Niederauer, M.Q. Molecular Biomarkers of Oxygen Therapy in Patients with Diabetic Foot Ulcers. Biomolecules 2021, 11, 925. https://doi.org/10.3390/biom11070925
Oropallo AR, Serena TE, Armstrong DG, Niederauer MQ. Molecular Biomarkers of Oxygen Therapy in Patients with Diabetic Foot Ulcers. Biomolecules. 2021; 11(7):925. https://doi.org/10.3390/biom11070925
Chicago/Turabian StyleOropallo, Alisha R., Thomas E. Serena, David G. Armstrong, and Mark Q. Niederauer. 2021. "Molecular Biomarkers of Oxygen Therapy in Patients with Diabetic Foot Ulcers" Biomolecules 11, no. 7: 925. https://doi.org/10.3390/biom11070925
APA StyleOropallo, A. R., Serena, T. E., Armstrong, D. G., & Niederauer, M. Q. (2021). Molecular Biomarkers of Oxygen Therapy in Patients with Diabetic Foot Ulcers. Biomolecules, 11(7), 925. https://doi.org/10.3390/biom11070925








