Synthesis and Anticancer Activity of Dimeric Polyether Ionophores
Abstract
1. Introduction
2. Materials and Methods
2.1. General Procedures
2.2. Synthesis
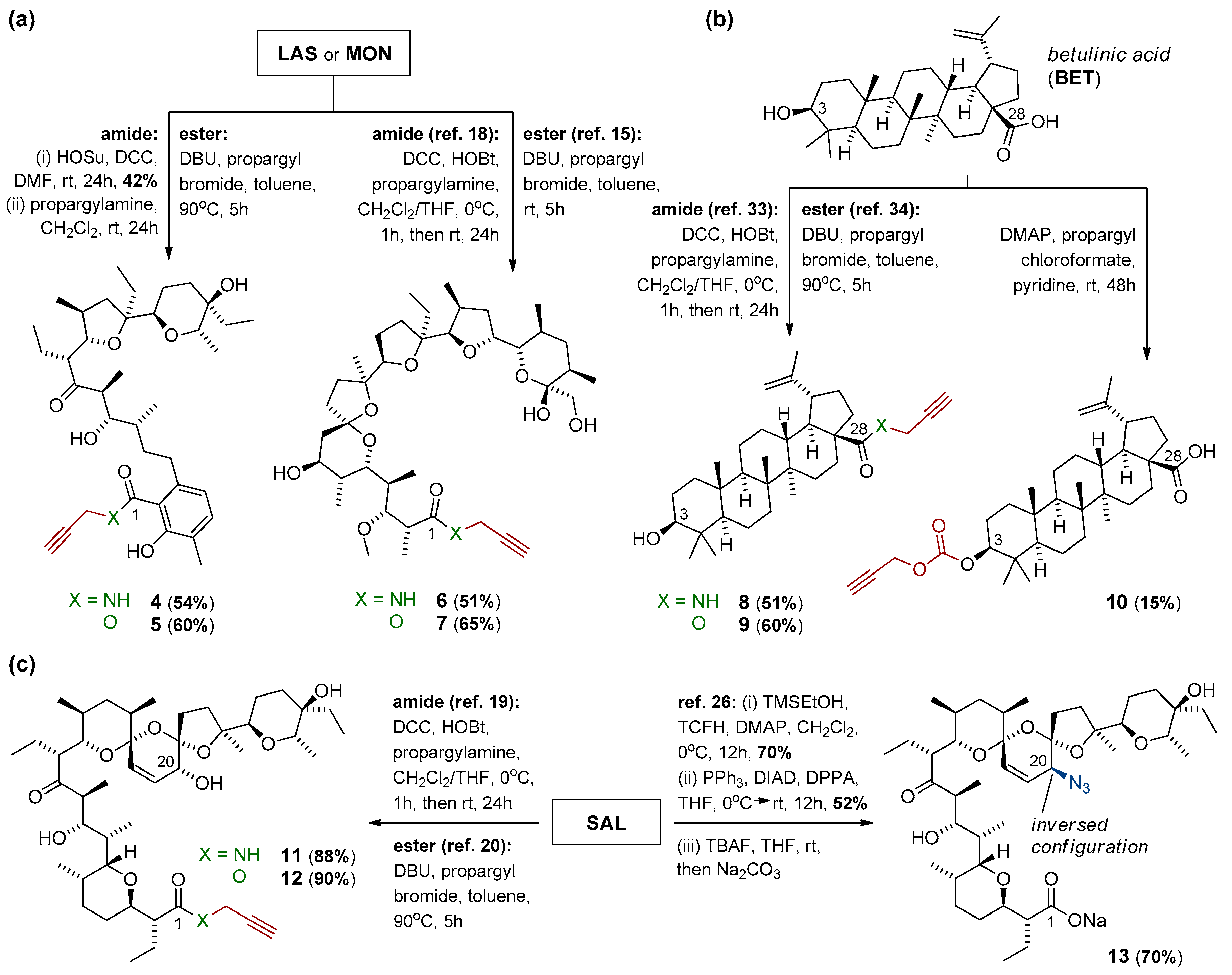
2.2.1. Synthesis of N-Propargyl Amide of Lasalocid Acid (Compound 4)
2.2.2. Synthesis of Propargyl Ester of Lasalocid Acid (Compound 5)
2.2.3. Synthesis of Propargyl Carbonate of Betulinic Acid (Compound 10)
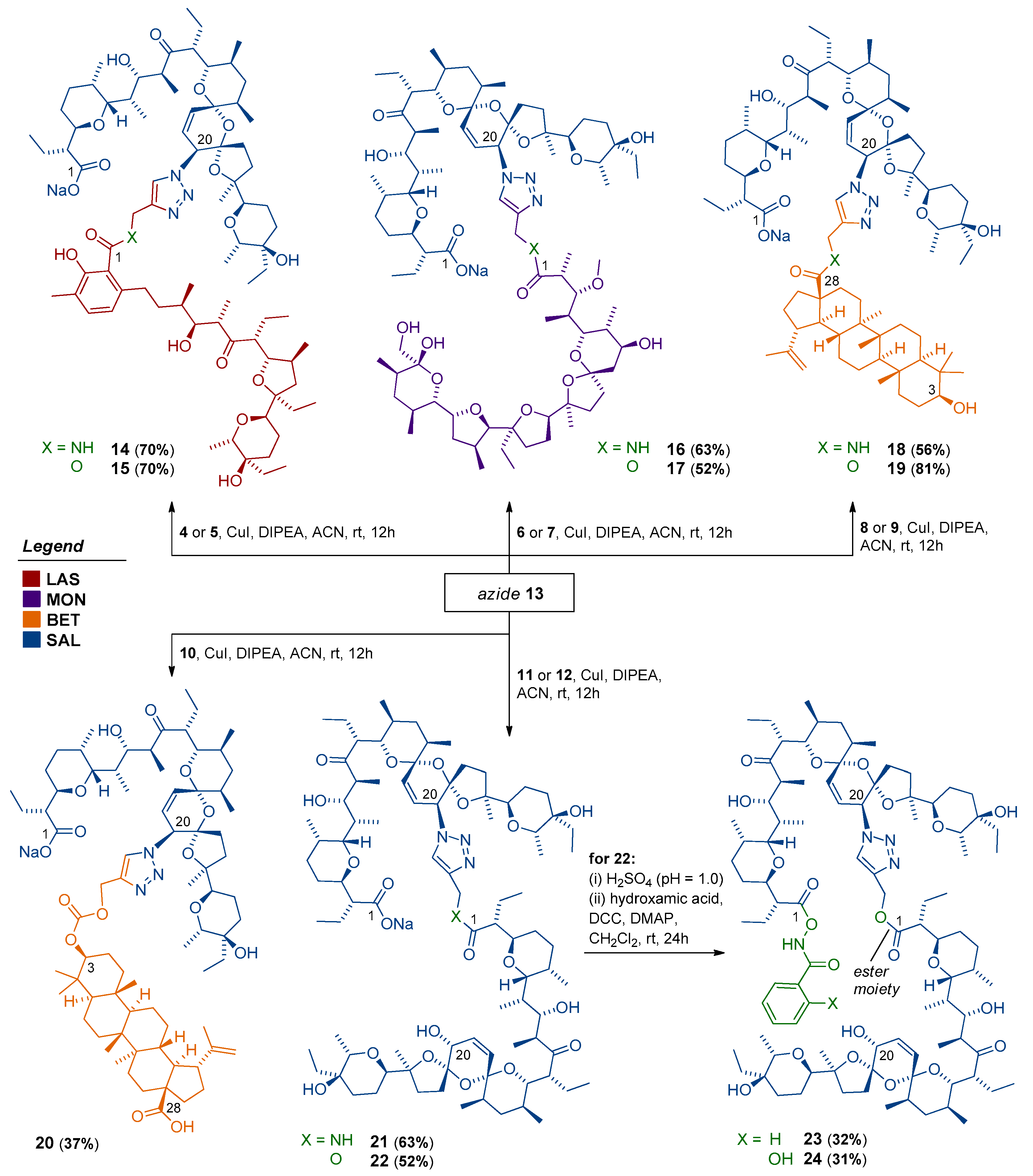
2.2.4. General Procedure for Preparation of Triazole-Linked Dimers (Compounds 14–22)
2.2.5. General Procedure for Preparation of Dimeric Conjugates with Hydroxamic Acids (Compounds 23–24)
2.3. In Vitro Biological Studies
2.3.1. Cell Lines and Culture Conditions
2.3.2. Antiproliferative Activity Assay
3. Results
3.1. Precursor Design and Synthesis
3.2. Dimerization of Polyether Ionophores
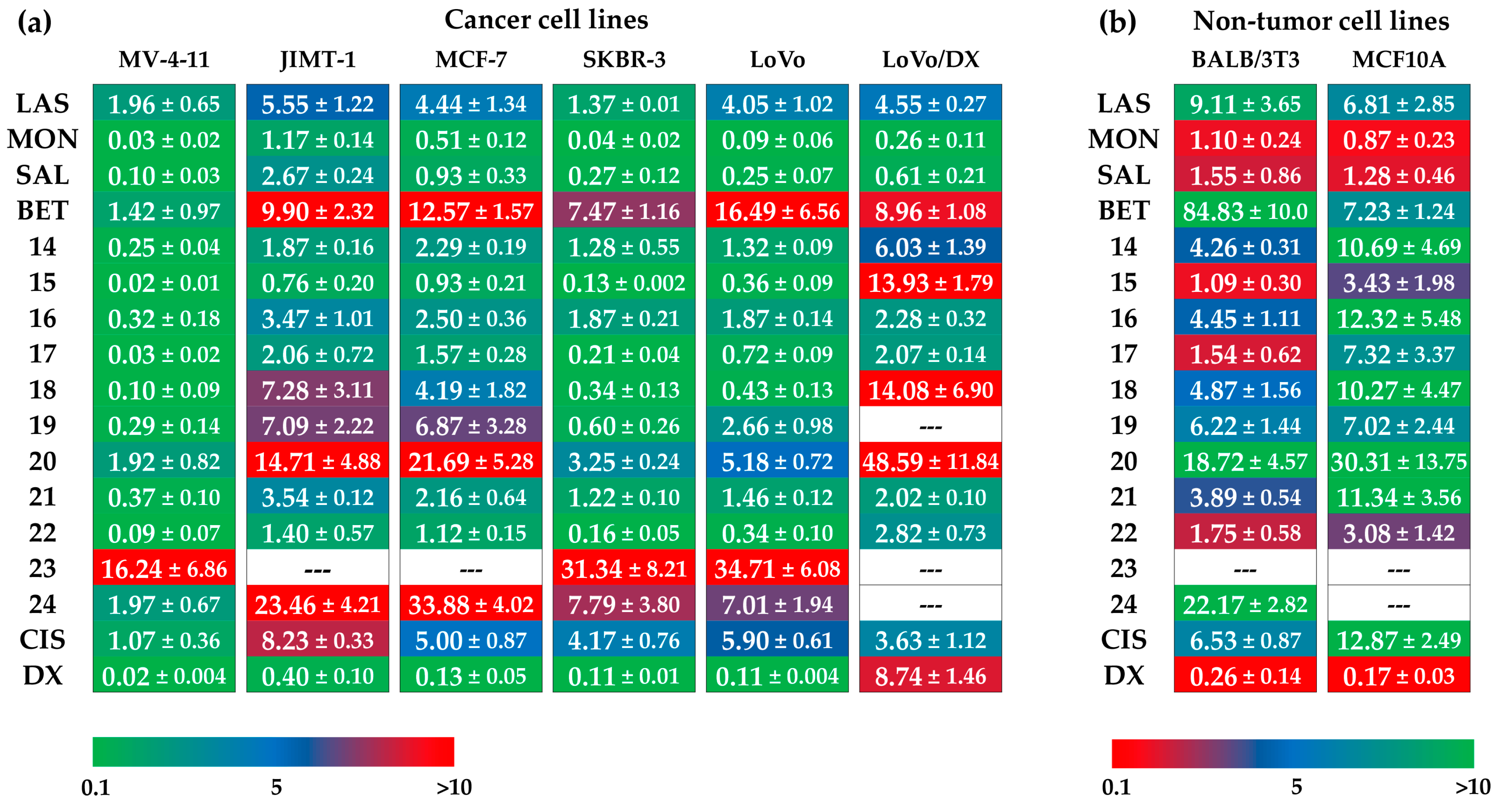
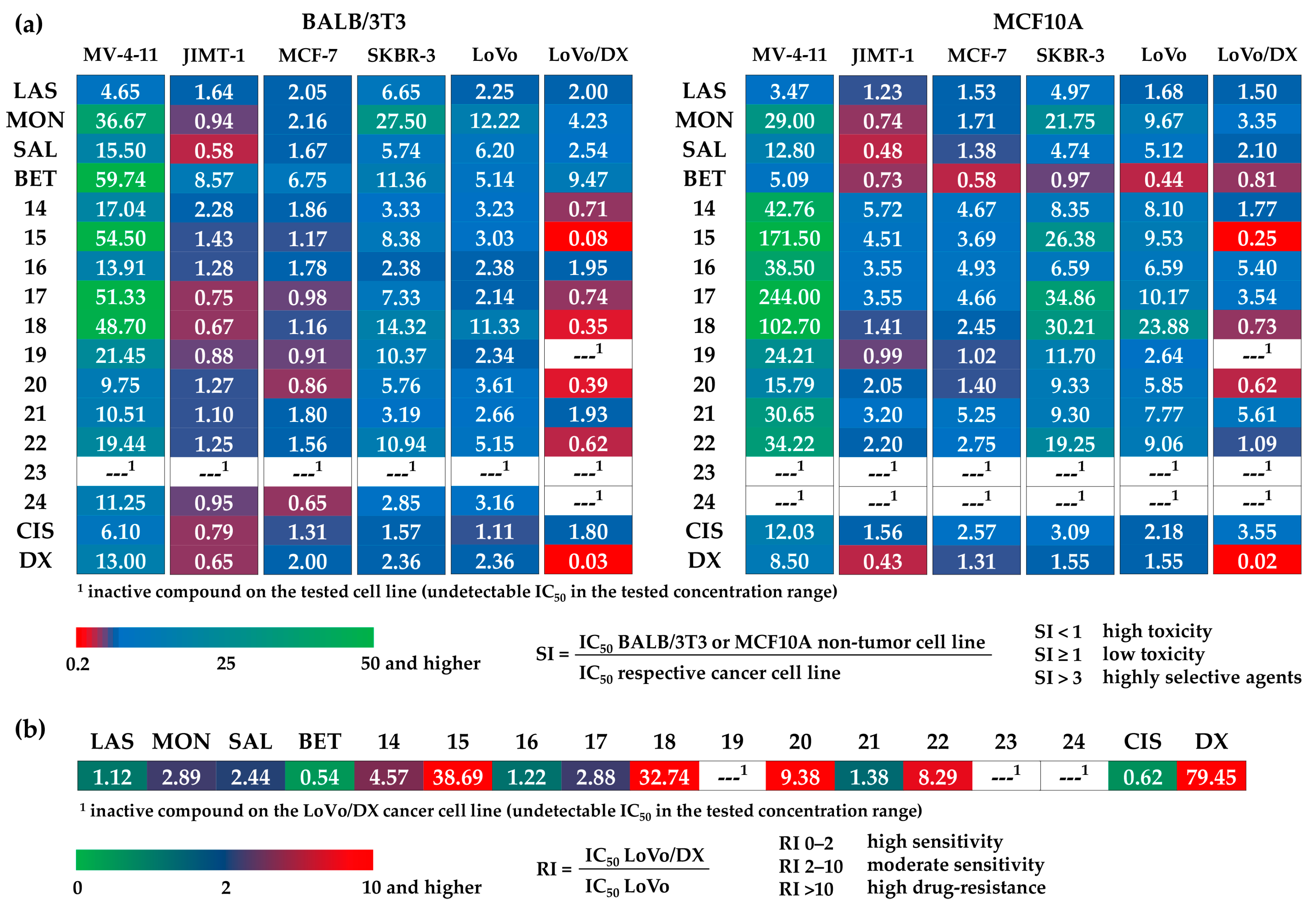
3.3. Antiproliferative Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Dedication
References
- Antoszczak, M.; Huczyński, A. Salinomycin and its derivatives – A new class of multiple-targeted “magic bullets”. Eur. J. Med. Chem. 2019, 176, 208–227. [Google Scholar] [CrossRef]
- Antoszczak, M.; Steverding, D.; Huczyński, A. Anti-parasitic activity of polyether ionophores. Eur. J. Med. Chem. 2019, 166, 32–47. [Google Scholar] [CrossRef]
- Rutkowski, J.; Brzezinski, B. Structures and properties of naturally occurring polyether antibiotics. Biomed. Res. Int. 2013, 2013, 162513. [Google Scholar] [CrossRef]
- Kevin II, D.A.; Meujo, D.A.; Hamann, M.T. Polyether ionophores: Broad-spectrum and promising biologically active molecules for the control of drug-resistant bacteria and parasites. Expert Opin. Drug Discov. 2009, 4, 109–146. [Google Scholar] [CrossRef] [PubMed]
- Antoszczak, M. A comprehensive review of salinomycin derivatives as potent anticancer and anti-CSCs agents. Eur. J. Med. Chem. 2019, 166, 48–64. [Google Scholar] [CrossRef] [PubMed]
- Huczyński, A.; Rutkowski, J.; Borowicz, I.; Wietrzyk, J.; Maj, E.; Brzezinski, B. One-pot synthesis and cytotoxicity studies of new Mannich base derivatives of polyether antibiotic – lasalocid acid. Bioorg. Med. Chem. Lett. 2013, 23, 5053. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.Y.; Kim, S.H.; Yu, S.N.; Park, S.G.; Kim, Y.W.; Nam, H.W.; An, H.H.; Yu, H.S.; Kim, Y.W.; Ji, J.H.; et al. Lasalocid induces cytotoxic apoptosis and cytoprotective autophagy through reactive oxygen species in human prostate cancer PC-3 cells. Biomed. Pharmacother. 2017, 88, 1016–1024. [Google Scholar] [CrossRef] [PubMed]
- Ketola, K.; Vainio, P.; Fey, V.; Kallioniemi, O.; Iljin, K. Monensin is a potent inducer of oxidative stress and inhibitor of androgen signaling leading to apoptosis in prostate cancer cells. Mol. Cancer Ther. 2010, 9, 3175. [Google Scholar] [CrossRef]
- Vanneste, M.; Huang, Q.; Li, M.; Moose, D.; Zhao, L.; Stamnes, M.A.; Schultz, M.; Wu, M.; Henry, M.D. High content screening identifies monensin as an EMT-selective cytotoxic compound. Sci. Rep. 2019, 9, 1200. [Google Scholar] [CrossRef]
- Wang, X.; Wu, X.; Zhang, Z.; Ma, C.; Wu, T.; Tang, S.; Zeng, Z.; Huang, S.; Gong, C.; Yuan, C.; et al. Monensin inhibits cell proliferation and tumor growth of chemo-resistant pancreatic cancer cells by targeting the EGFR signaling pathway. Sci. Rep. 2018, 8, 17914. [Google Scholar] [CrossRef]
- Park, W.H.; Seol, J.G.; Kim, E.S.; Kang, W.K.; Im, Y.H.; Jung, C.W.; Kim, B.K.; Lee, Y.Y. Monensin-mediated growth inhibition in human lymphoma cells through cell cycle arrest and apoptosis. Br. J. Haematol. 2002, 119, 400. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.B.; Onder, T.T.; Jiang, G.; Tao, K.; Kuperwasser, C.; Weinberg, R.A.; Lander, E.S. Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 2009, 138, 645–659. [Google Scholar] [CrossRef] [PubMed]
- Naujokat, C.; Steinhart, R. Salinomycin as a drug for targeting human cancer stem cells. J. Biomed. Biotechnol. 2012, 2012, 950658. [Google Scholar] [CrossRef] [PubMed]
- Antoszczak, M. A medicinal chemistry perspective on salinomycin as a potent anticancer and anti-CSCs agent. Eur. J. Med. Chem. 2019, 164, 366–377. [Google Scholar] [CrossRef]
- Klejborowska, G.; Jędrzejczyk, M.; Stępczyńska, N.; Maj, E.; Wietrzyk, J.; Huczyński, A. Antiproliferative activity of ester derivatives of monensin A at the C-1 and C-26 positions. Chem. Biol. Drug Des. 2019, 94, 1859–1864. [Google Scholar] [CrossRef]
- Li, B.; Wu, J.; Zhang, W.; Li, Z.; Chen, G.; Zhou, Q.; Wu, S. Synthesis and biological activity of salinomycin-hydroxamic acid conjugates. Bioorg. Med. Chem. Lett. 2017, 27, 1624–1626. [Google Scholar] [CrossRef]
- Huczyński, A.; Klejborowska, G.; Antoszczak, M.; Maj, E.; Wietrzyk, J. Anti-proliferative activity of monensin and its tertiary amide derivatives. Bioorg. Med. Chem. Lett. 2015, 25, 4539–4543. [Google Scholar] [CrossRef]
- Skiera, I.; Antoszczak, M.; Trynda, J.; Wietrzyk, J.; Boratyński, P.; Kacprzak, K.; Huczyński, A. Antiproliferative activity of polyether antibiotic-Cinchona alkaloid conjugates obtained via click chemistry. Chem. Biol. Drug Des. 2015, 86, 911–917. [Google Scholar] [CrossRef]
- Antoszczak, M.; Maj, E.; Stefańska, J.; Wietrzyk, J.; Janczak, J.; Brzezinski, B.; Huczyński, A. Synthesis, antiproliferative and antibacterial activity of new amides of salinomycin. Bioorg. Med. Chem. Lett. 2014, 24, 1724–1729. [Google Scholar] [CrossRef]
- Antoszczak, M.; Popiel, K.; Stefańska, J.; Wietrzyk, J.; Maj, E.; Janczak, J.; Michalska, G.; Brzezinski, B.; Huczyński, A. Synthesis, cytotoxicity and antibacterial activity of new esters of polyether antibiotic – salinomycin. Eur. J. Med. Chem. 2014, 76, 435–444. [Google Scholar] [CrossRef]
- Versini, A.; Colombeau, L.; Hienzsch, A.; Gaillet, C.; Retailleau, P.; Debieu, S.; Müller, S.; Cañeque, T.; Rodriguez, R. Salinomycin derivatives kill breast cancer stem cells by lysosomal iron targeting. Chem. Eur. J. 2020, 26, 7416. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Shi, Q.; Shao, J.; Yuan, Y.; Yang, Z.; Chen, S.; Zhou, X.; Wen, S.; Jiang, Z.X. Synthesis and biological evaluation of 20-epi-amino-20-deoxysalinomycin derivatives. Eur. J. Med. Chem. 2018, 148, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Mai, T.T.; Hamaï, A.; Hienzsch, A.; Cañeque, T.; Müller, S.; Wicinski, J.; Cabaud, O.; Leroy, C.; David, A.; Acevedo, V.; et al. Salinomycin kills cancer stem cells by sequestering iron in lysosomes. Nat. Chem. 2017, 9, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Borgström, B.; Huang, X.; Hegardt, C.; Oredsson, S.; Strand, D. Structure-activity relationships in salinomycin: Cytotoxicity and phenotype selectivity of semi-synthetic derivatives. Chem. Eur. J. 2017, 23, 2077–2083. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wu, J.; Li, B.; Xia, J.; Wu, H.; Wang, L.; Hao, J.; Zhou, Q.; Wu, S. Synthesis and biological activity evaluation of 20-epi-salinomycin and its 20-O-acyl derivatives. RSC Adv. 2016, 6, 41885. [Google Scholar] [CrossRef]
- Shi, Q.; Li, Y.; Bo, S.; Li, X.; Zhao, P.; Liu, Q.; Yang, Z.; Cong, H.; Deng, H.; Chen, M.; et al. Discovery of a 19F MRI sensitive salinomycin derivative with high cytotoxicity towards cancer cells. Chem. Commun. 2016, 52, 5136–5139. [Google Scholar] [CrossRef]
- Borgström, B.; Huang, X.; Pošta, M.; Hegardt, C.; Oredsson, S.; Strand, D. Synthetic modification of salinomycin: Selective O-acylation and biological evaluation. Chem. Commun. 2013, 49, 9944–9946. [Google Scholar] [CrossRef]
- Huczyński, A.; Domańska, A.; Paluch, I.; Stefańska, J.; Brzezinski, B.; Bartl, F. Synthesis of new semi-synthetic dipodands and tripodands from naturally occurring polyether ionophores. Tetrahedron Lett. 2008, 49, 5572–5575. [Google Scholar] [CrossRef]
- Huang, M.; Deng, Z.; Tian, J.; Liu, T. Synthesis and biological evaluation of salinomycin triazole analogues as anticancer agents. Eur. J. Med. Chem. 2017, 127, 900–908. [Google Scholar] [CrossRef]
- Antoszczak, M.; Maj, E.; Borgström, B.; Oredsson, S.; Huczyński, A.; Wietrzyk, J.; Strand, D. Bivalent polyether ionophores: Synthesis and biological evaluation of C2-symmetric salinomycin dimers. Tetrahedron Lett. 2017, 58, 2396–2399. [Google Scholar] [CrossRef]
- Mullauer, F.B.; Kessler, J.H.; Medema, J.P. Betulinic acid, a natural compounds with potent anticancer effects. Anticancer Drugs 2010, 21, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S. Betulinic acid: A natural product with anticancer activity. Mol. Nutr. Food Res. 2009, 53, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Dang Thi, T.A.; Kim Tuyet, N.T.; Pham The, C.; Thanh Nguyen, H.; Ba Thi, C.; Thi Phuong, H.; Van Boi, L.; Van Nguyen, T.; D’hooghe, M. Synthesis and cytotoxic evaluation of novel amide-triazole-linked triterpenoid-AZT conjugates. Tetrahedron Lett. 2015, 56, 218–224. [Google Scholar] [CrossRef]
- Dang Thi, T.A.; Kim Tuyet, N.T.; Pham The, C.; Thanh Nguyen, H.; Ba Thi, C.; Doan Duy, T.; D’hooghe, M.; Van Nguyen, T. Synthesis and cytotoxic evaluation of novel ester-triazole-linked triterpenoid-AZT conjugates. Bioorg. Med. Chem. Lett. 2014, 24, 5190–5194. [Google Scholar] [CrossRef] [PubMed]
- Nevozhay, D. Cheburator software for automatically calculating drug inhibitory concentrations from in vitro screening assays. PLoS One 2014, 9, e106186. [Google Scholar] [CrossRef] [PubMed]
- Sulik, M.; Stępień, K.; Stefańska, J.; Huczyński, A.; Antoszczak, M. Antibacterial activity of singly and doubly modified salinomycin derivatives. Bioorg. Med. Chem. Lett. 2020, 30, 127062. [Google Scholar] [CrossRef]
- Antoszczak, M.; Steverding, D.; Sulik, M.; Janczak, J.; Huczyński, A. Anti-trypanosomal activity of doubly modified salinomycin derivatives. Eur. J. Med. Chem. 2019, 173, 90–98. [Google Scholar] [CrossRef]
- Antoszczak, M.; Klejborowska, G.; Kruszyk, M.; Maj, E.; Wietrzyk, J.; Huczyński, A. Synthesis and antiproliferative activity of silybin conjugates with salinomycin and monensin. Chem. Biol. Drug Des. 2015, 86, 1378–1386. [Google Scholar] [CrossRef]
- Rooseboom, M.; Commandeur, J.N.; Vermeulen, N.P. Enzyme catalyzed activation of anticancer prodrugs. Pharmacol. Rev. 2004, 56, 53–102. [Google Scholar] [CrossRef]
- Huczyński, A.; Antoszczak, M.; Kleczewska, N.; Lewandowska, M.; Maj, E.; Stefańska, J.; Wietrzyk, J.; Janczak, J.; Celewicz, L. Synthesis and biological activity of salinomycin conjugates with floxuridine. Eur. J. Med. Chem. 2015, 93, 33–41. [Google Scholar] [CrossRef]
- Antoszczak, M.; Urbaniak, A.; Delgado, M.; Maj, E.; Borgström, B.; Wietrzyk, J.; Huczyński, A.; Yuan, Y.; Chambers, T.C.; Strand, D. Biological activity of doubly modified salinomycin analogs – Evaluation in vitro and ex vivo. Eur. J. Med. Chem. 2018, 156, 510–523. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sulik, M.; Maj, E.; Wietrzyk, J.; Huczyński, A.; Antoszczak, M. Synthesis and Anticancer Activity of Dimeric Polyether Ionophores. Biomolecules 2020, 10, 1039. https://doi.org/10.3390/biom10071039
Sulik M, Maj E, Wietrzyk J, Huczyński A, Antoszczak M. Synthesis and Anticancer Activity of Dimeric Polyether Ionophores. Biomolecules. 2020; 10(7):1039. https://doi.org/10.3390/biom10071039
Chicago/Turabian StyleSulik, Michał, Ewa Maj, Joanna Wietrzyk, Adam Huczyński, and Michał Antoszczak. 2020. "Synthesis and Anticancer Activity of Dimeric Polyether Ionophores" Biomolecules 10, no. 7: 1039. https://doi.org/10.3390/biom10071039
APA StyleSulik, M., Maj, E., Wietrzyk, J., Huczyński, A., & Antoszczak, M. (2020). Synthesis and Anticancer Activity of Dimeric Polyether Ionophores. Biomolecules, 10(7), 1039. https://doi.org/10.3390/biom10071039






