Chemopreventive Property of Sencha Tea Extracts towards Sensitive and Multidrug-Resistant Leukemia and Multiple Myeloma Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Extracts
2.2. Extract Analyses by HPLC-HRMS/MS
2.3. Molecular Network Analyses
2.4. Cell Culture
2.5. Resazurin Assay
2.6. Cell Cycle Analysis and Detection of Apoptotic Cells by Flow Cytometry and Annexin V/PI Staining
2.7. Measurements of ROS
2.8. Assessment of MMP
2.9. COMPARE and Hierarchical Cluster Analyses of Microarray Data
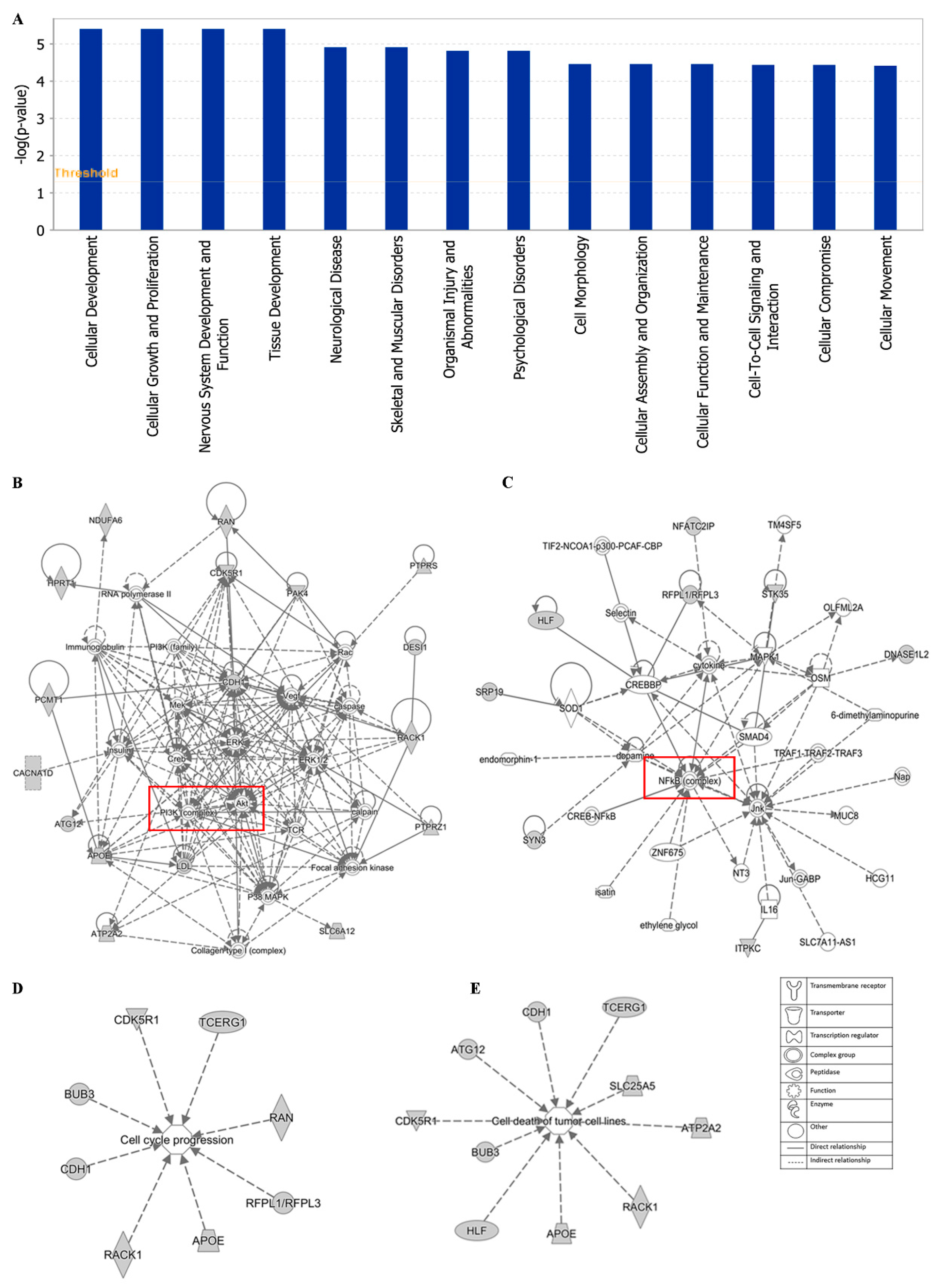
2.10. Ingenuity Pathway Analysis (IPA)
2.11. Statistics
3. Results and Discussion
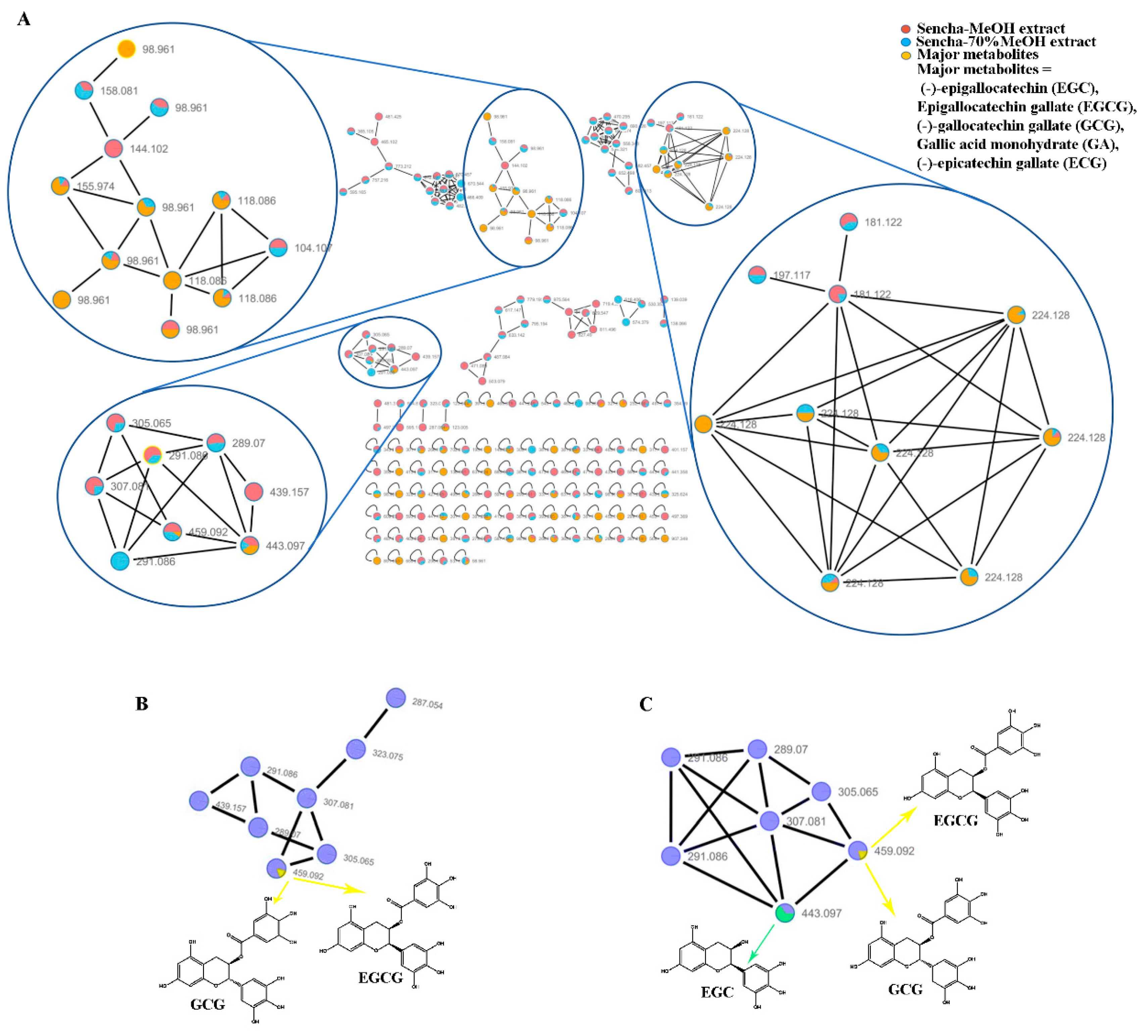
3.1. Molecular Network Analyses for HPLC–HRMS/MS Data
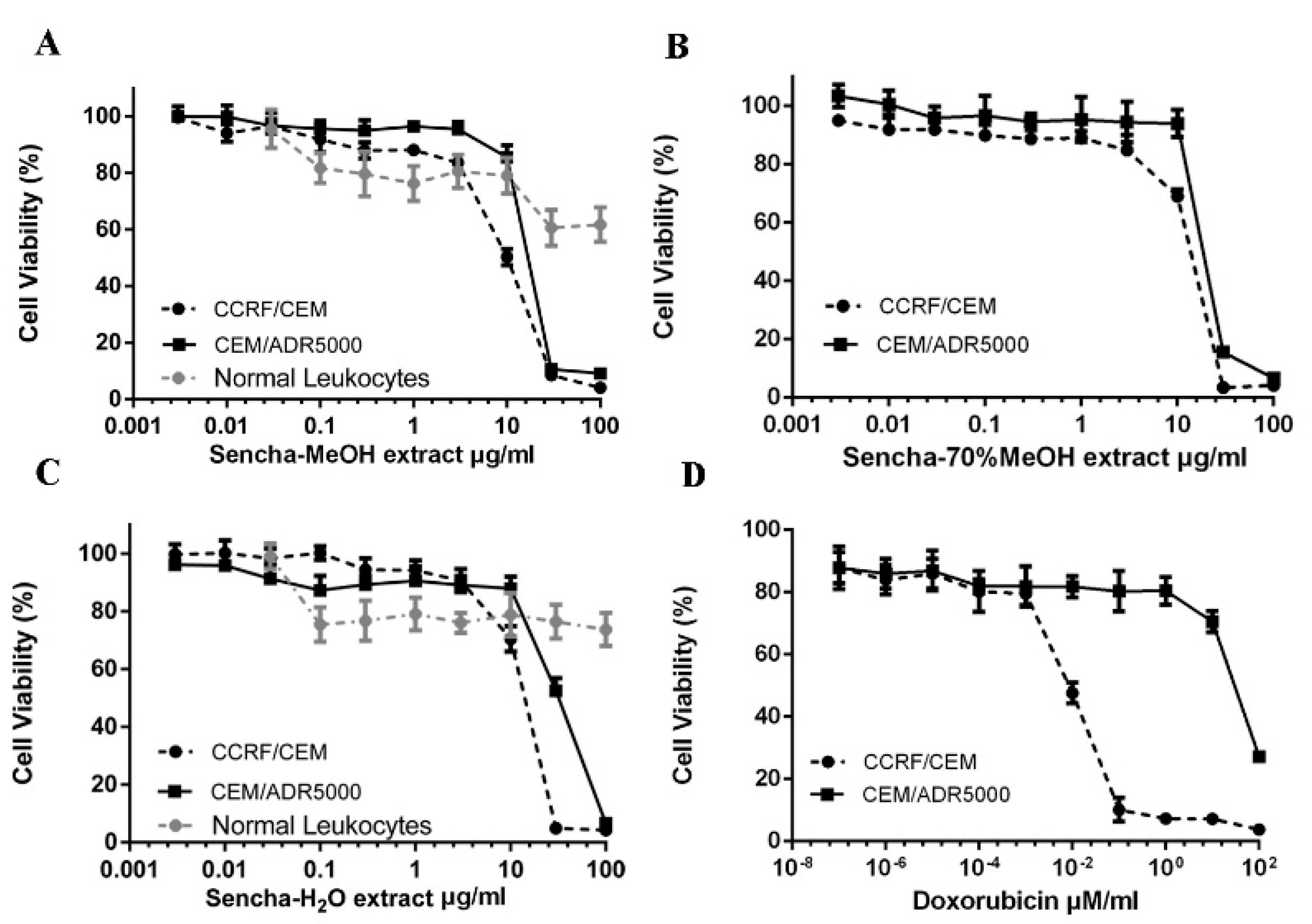
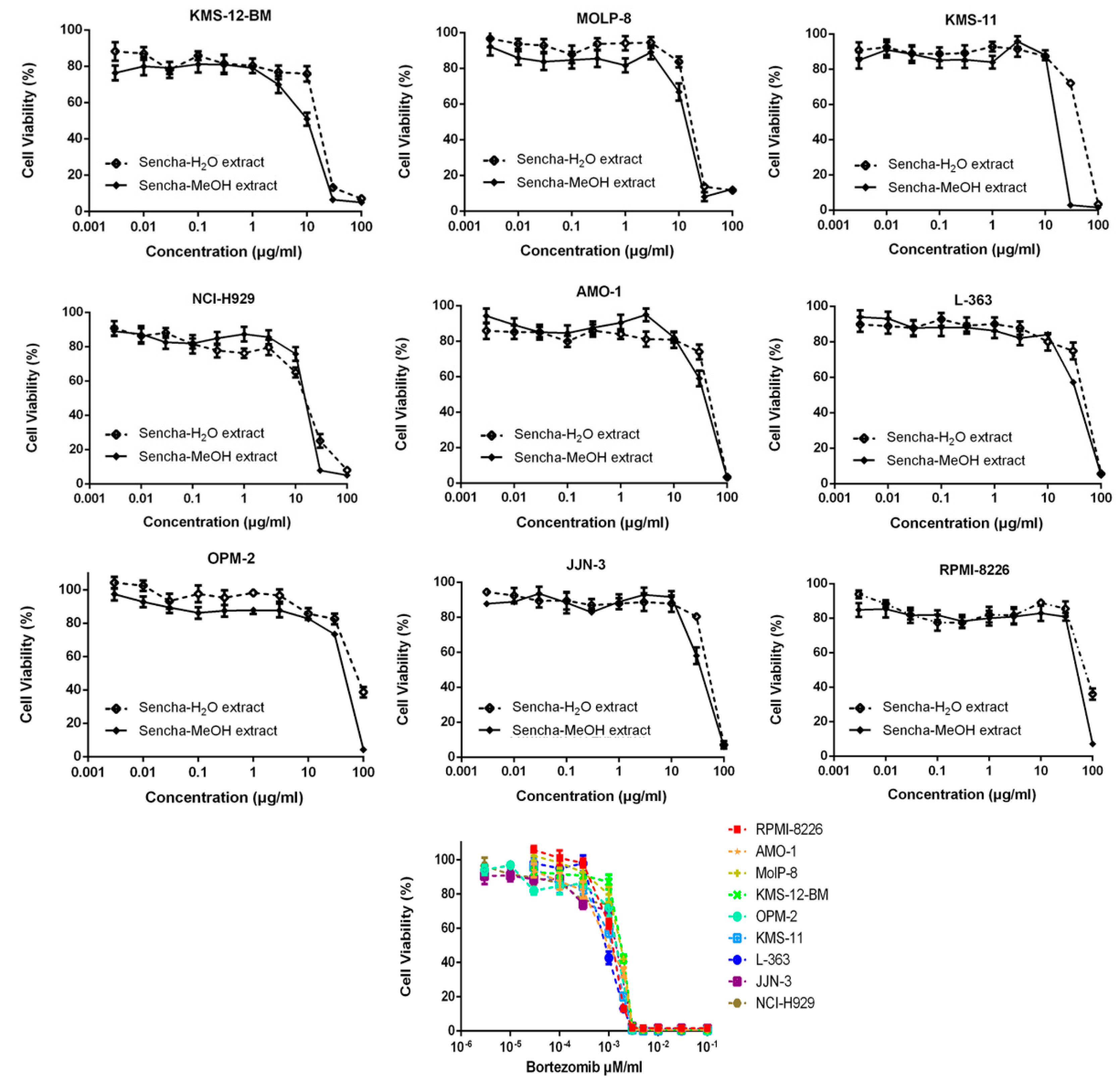
3.2. Resazurin-Based Cytotoxicity of Sencha Extracts
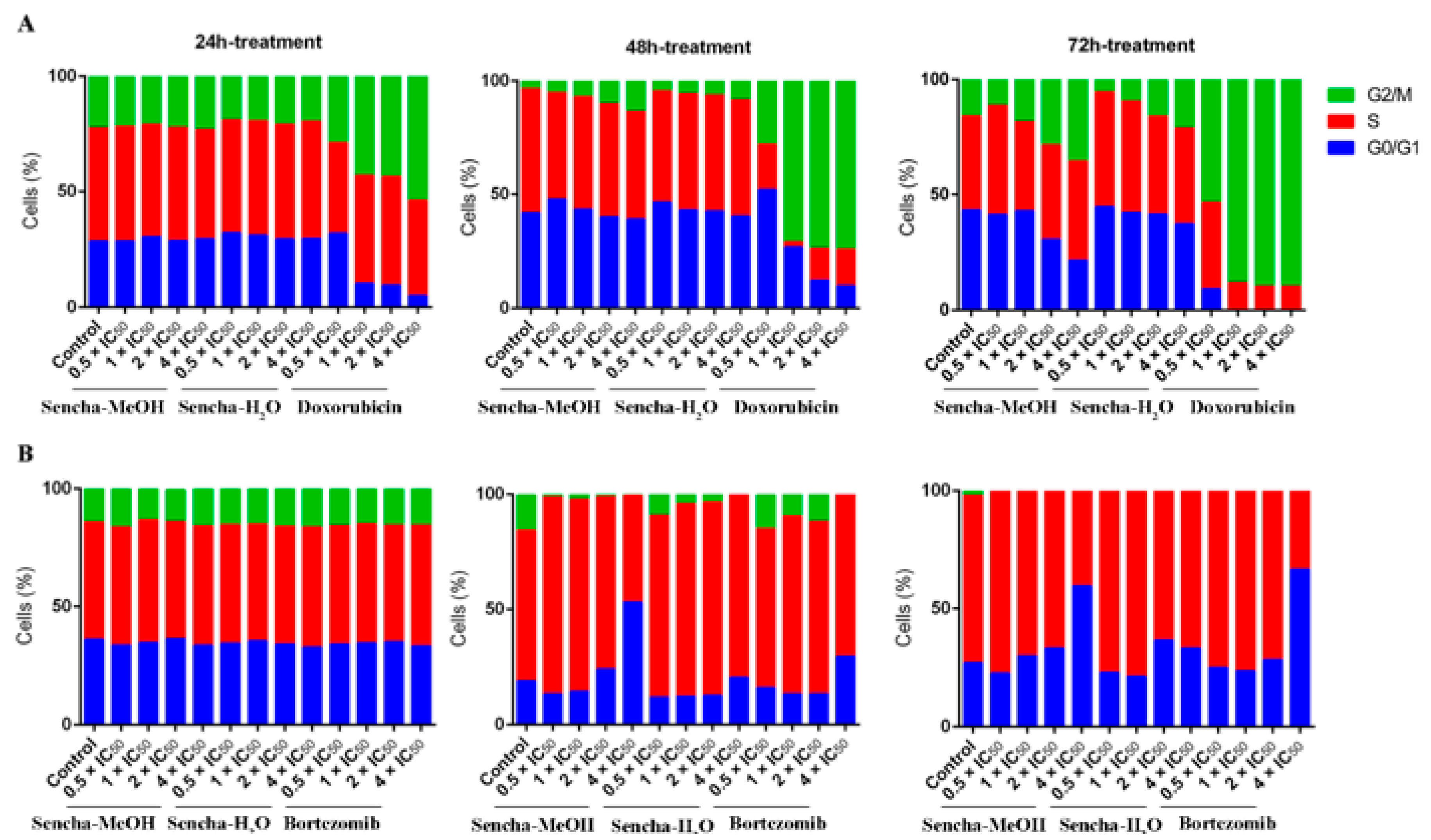
3.3. Cell Cycle Analysis
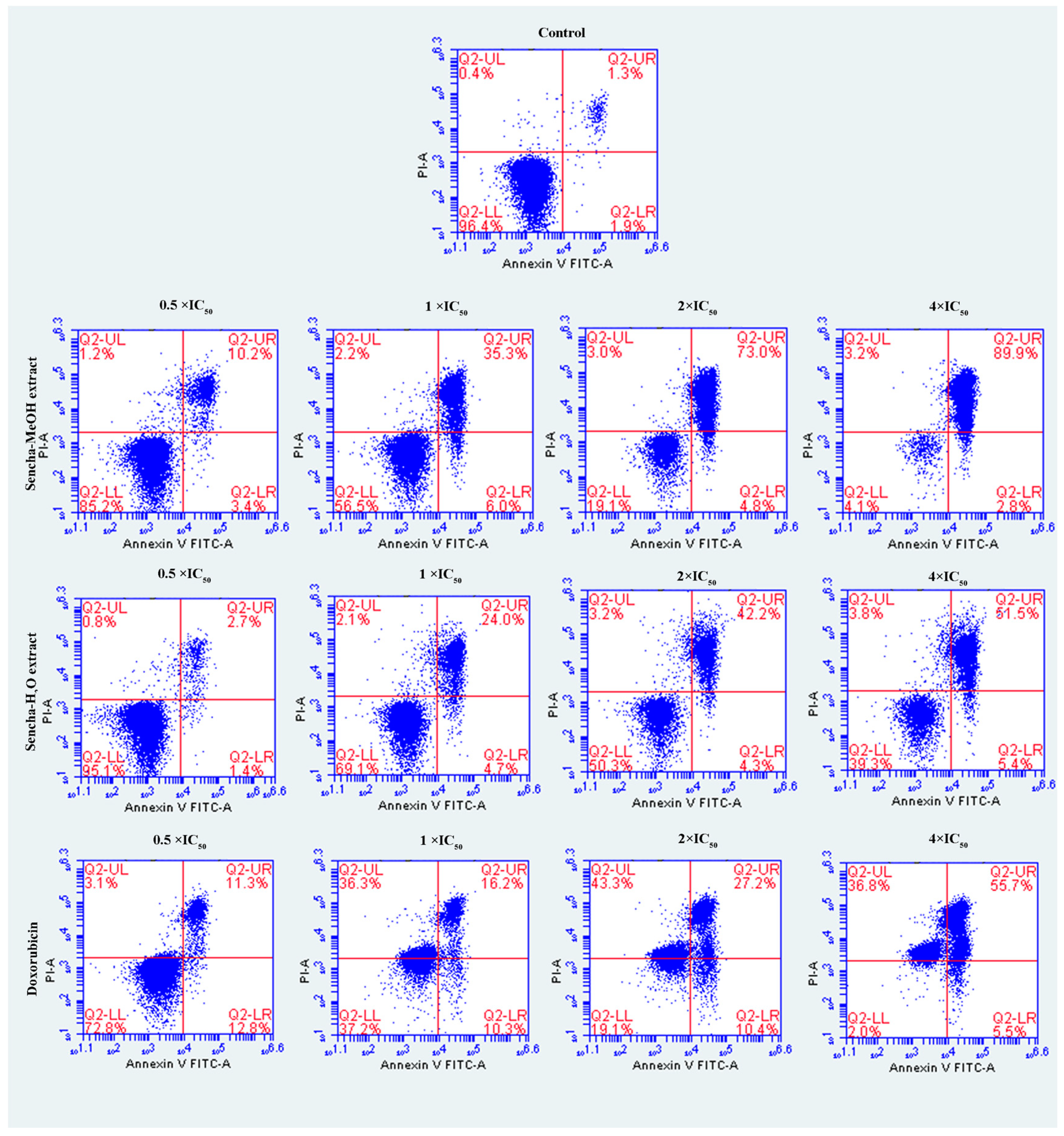
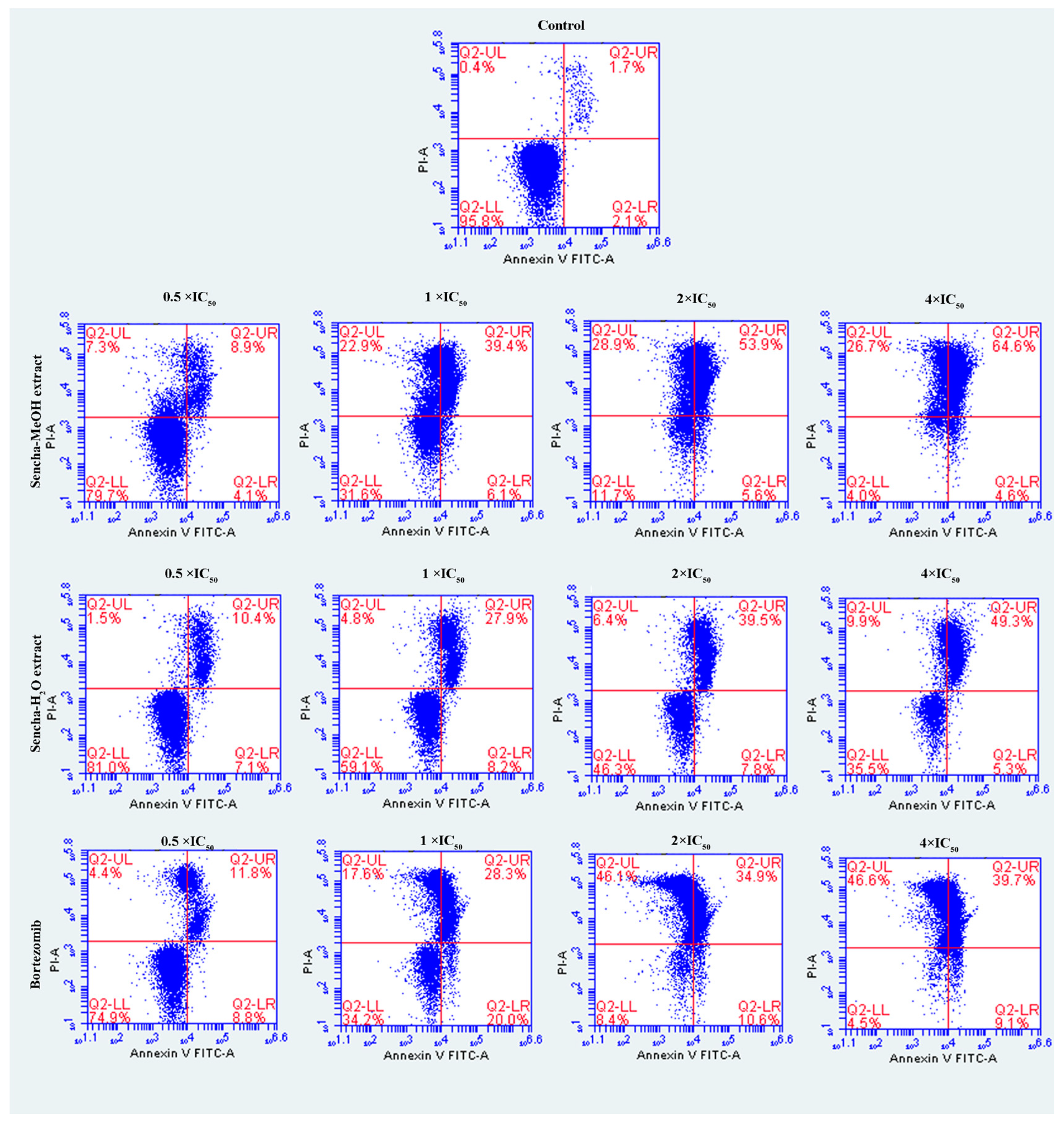
3.4. Induction of Apoptosis by Sencha Extracts
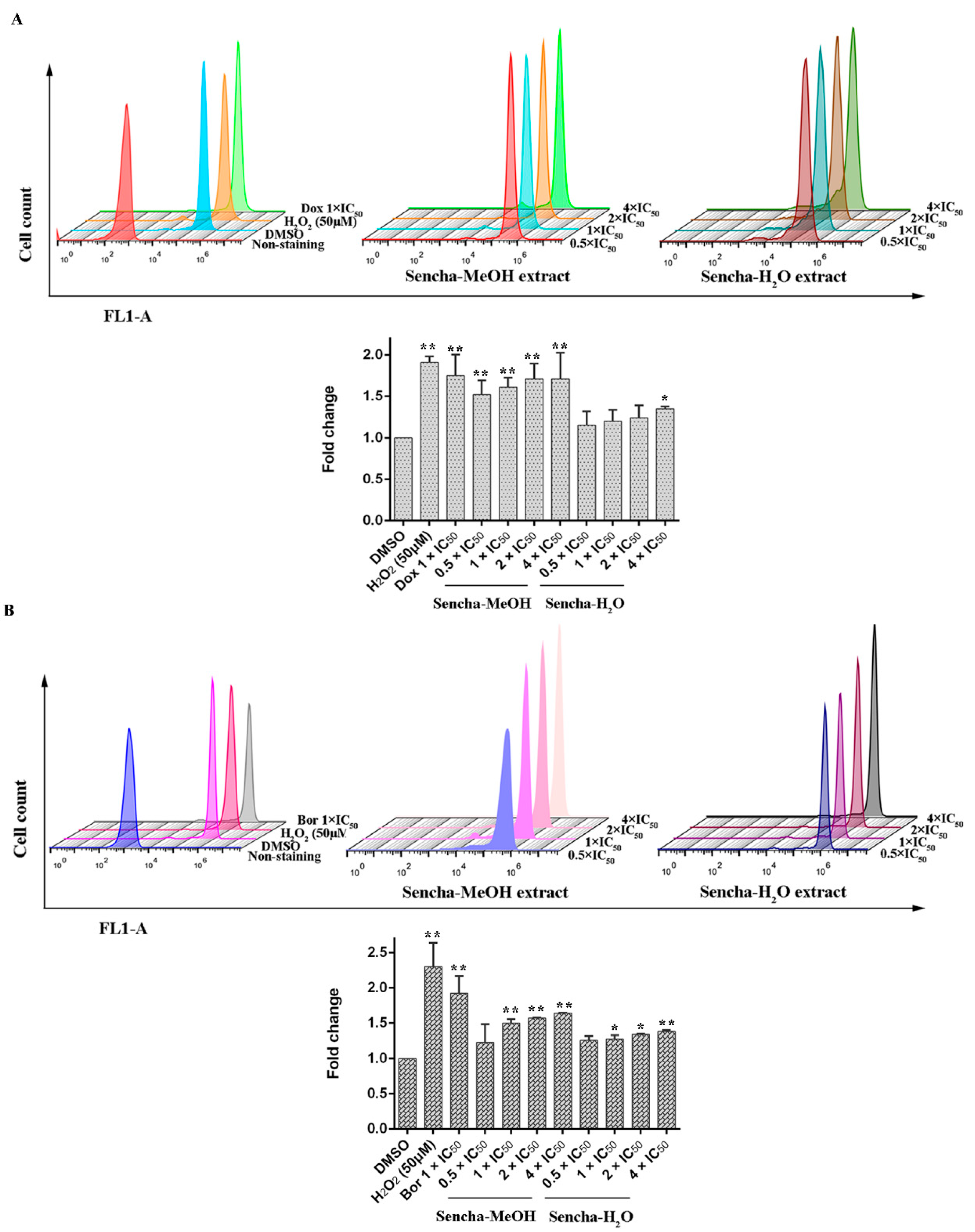
3.5. Measurements of ROS
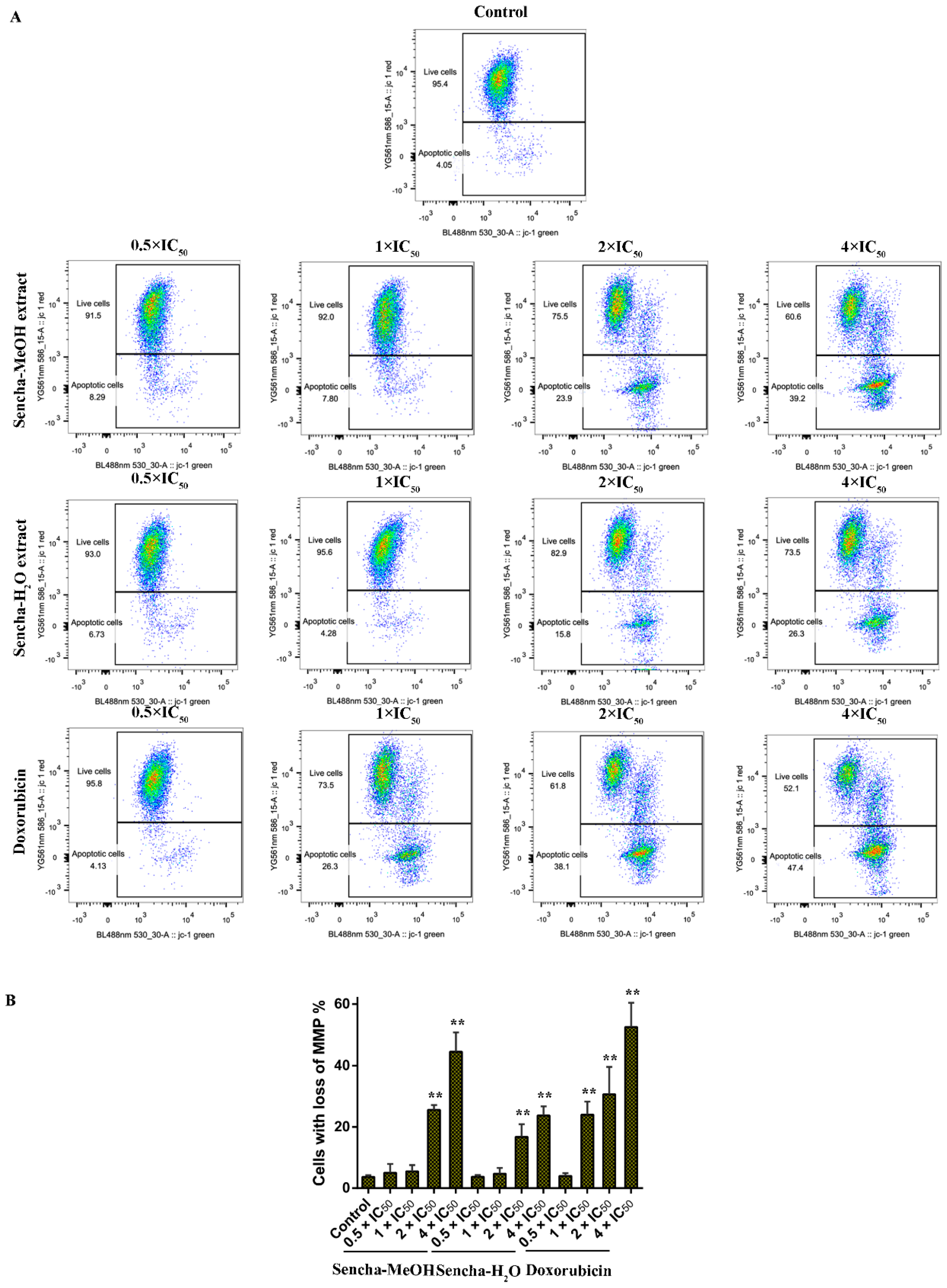
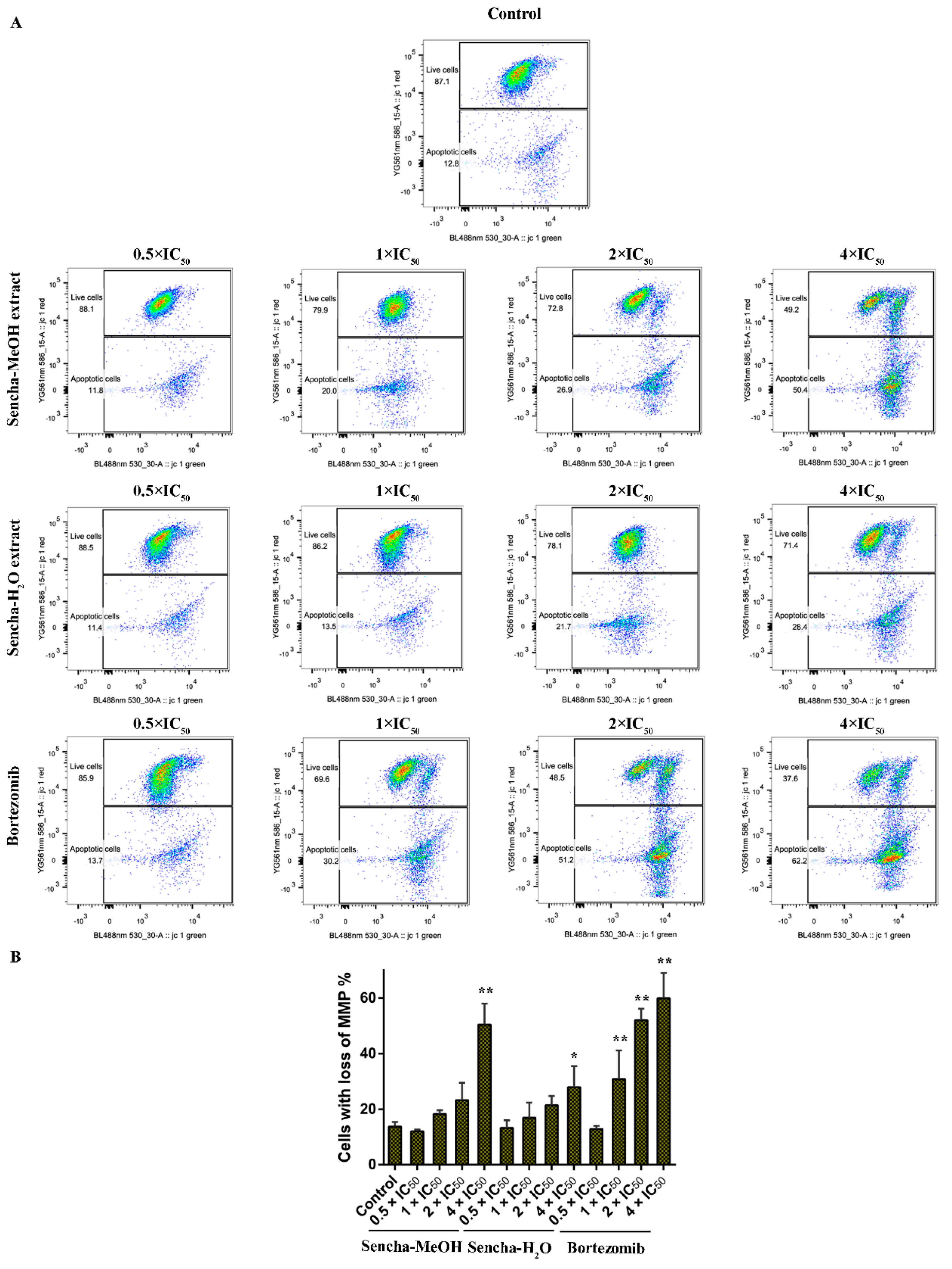
3.6. Assessment of MMP
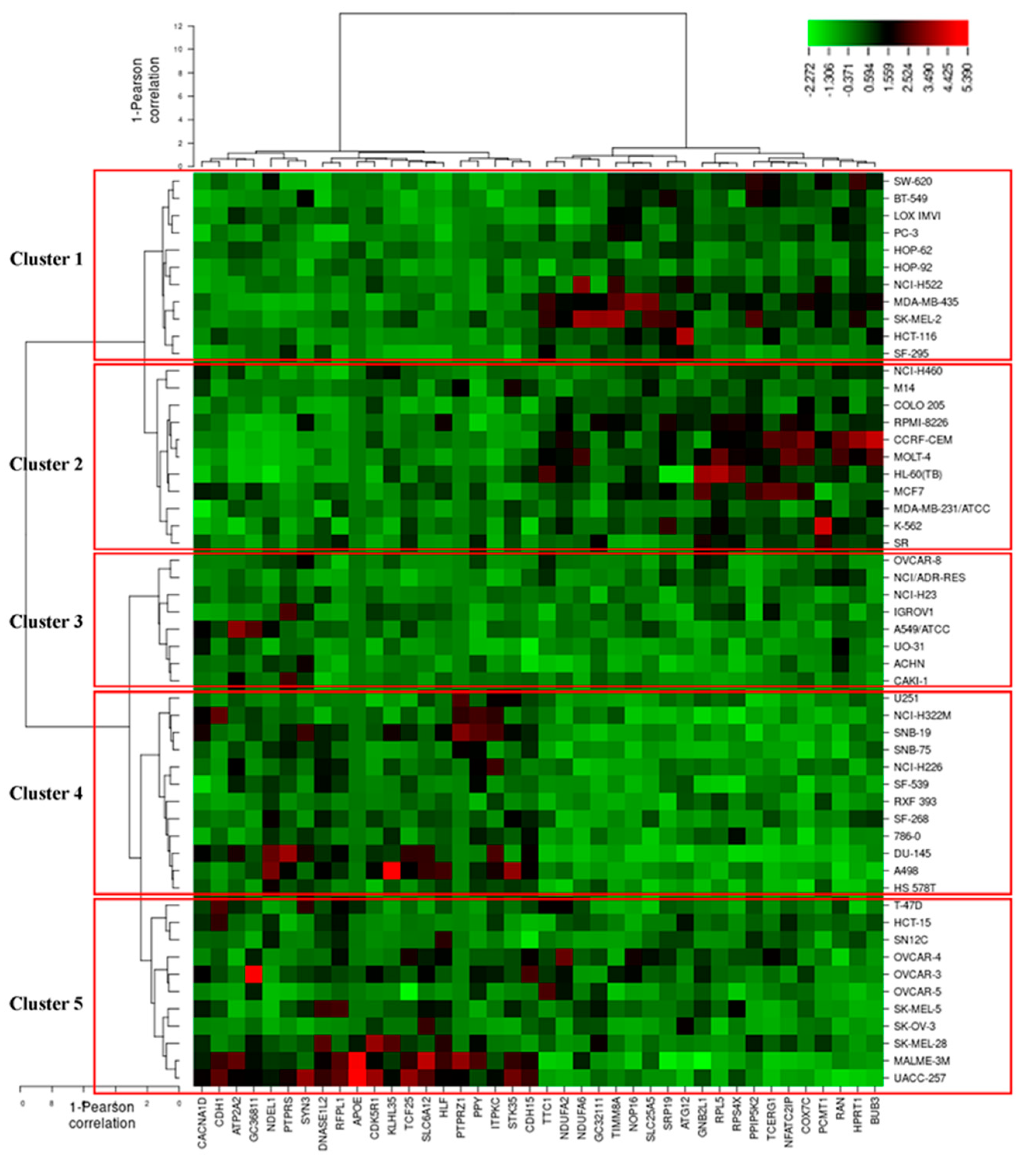
3.7. COMPARE and Hierarchical Cluster Analyses of Microarray Data
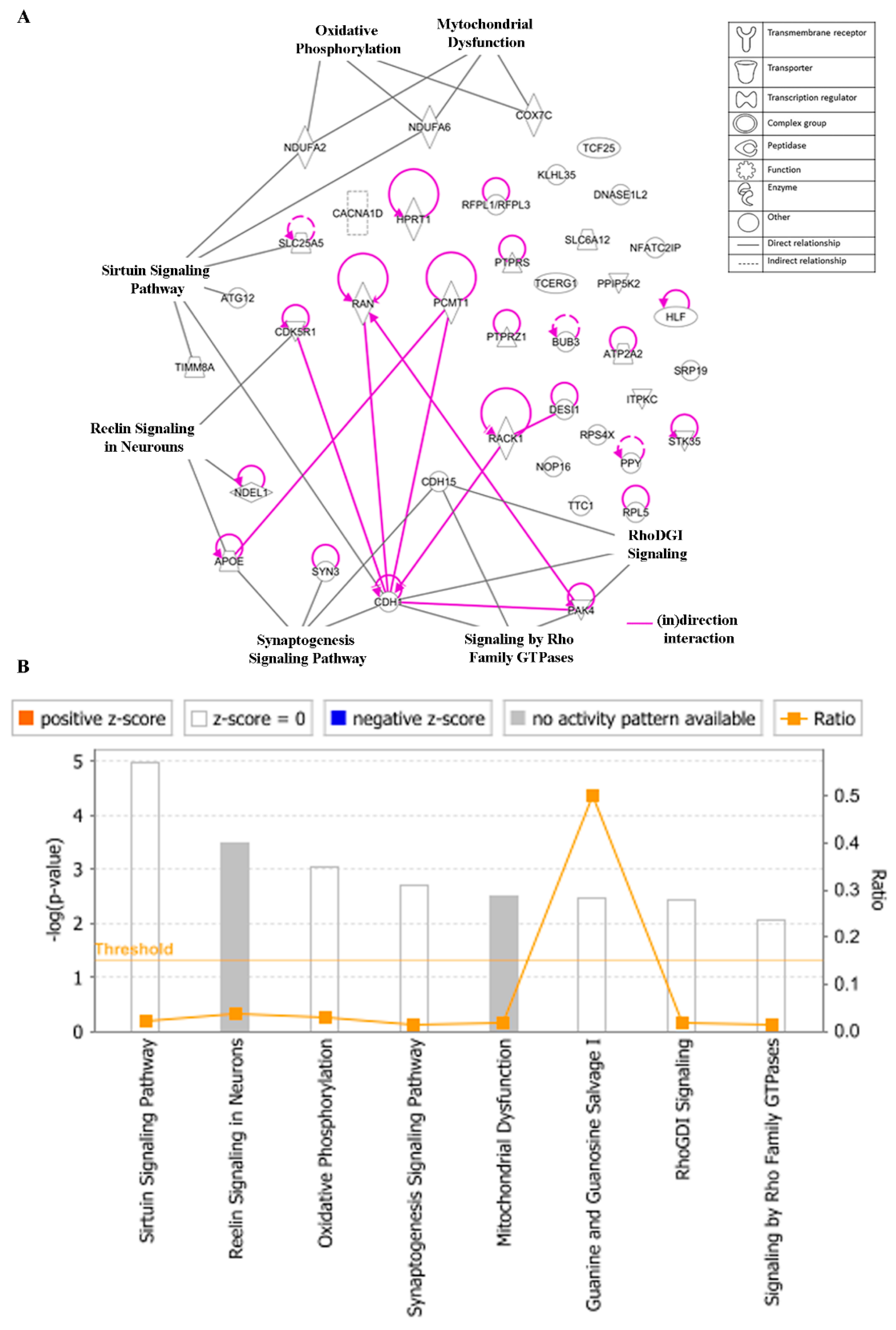
3.8. Ingenuity Pathway Analysis (IPA)
4. Conclusions and Perspectives
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cabrera, C.; Artacho, R.; Giménez, R. Beneficial effects of green tea-a review. J. Am. Coll. Nutr. 2006, 25, 79–99. [Google Scholar] [CrossRef]
- Khan, N.; Mukhtar, H. Tea polyphenols in promotion of human health. Nutrients 2018, 11, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xing, L.; Zhang, H.; Qi, R.; Tsao, R.; Mine, Y. Recent advances in the understanding of the health benefits and molecular mechanisms associated with green tea polyphenols. J. Agric. Food Chem. 2019, 67, 1029–1043. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Chen, Z.M. Green tea, black tea and semi-fermented tea. In Tea: Bioactivity and Therapeutic Potential; Zhen, Y.S., Chen, Z.M., Cheng, S.J., Chen, M.I., Eds.; Taylor and Francis: London, UK; New York, NY, USA, 2002; pp. 35–55. [Google Scholar]
- Tounekti, T.; Joubert, E.; Hernández, I.; Munné-Bosch, S. Improving the polyphenol content of tea. Crit. Rev. Plant Sci. 2013, 32, 192–215. [Google Scholar] [CrossRef]
- Šilarová, P.; Česlová, L.; Meloun, M. Fast gradient HPLC/MS separation of phenolics in green tea to monitor their degradation. Food Chem. 2017, 237, 471–480. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Li, J.; Zhao, W.; Liu, R.; Zhang, L.; Kong, X. Chemical fingerprint analysis for quality control and identification of Ziyang green tea by HPLC. Food Chem. 2015, 171, 405–411. [Google Scholar] [CrossRef]
- Ramdani, D.; Chaudhry, A.S.; Seal, C.J. Alkaloid and polyphenol analysis by HPLC in green and black tea powders and their potential use as additives in ruminant diets. AIP Conf. Proc. 2018, 1927, 030008. [Google Scholar] [CrossRef] [Green Version]
- Nakachi, K.; Matsuyama, S.; Miyake, S.; Suganuma, M.; Imai, K. Preventive effects of drinking green tea on cancer and cardiovascular disease: Epidemiological evidence for multiple targeting prevention. BioFactors 2000, 13, 49–54. [Google Scholar] [CrossRef]
- He, Y.H.; Kies, C. Green and black tea consumption by humans: Impact on polyphenol concentrations in feces, blood and urine. Plant Foods Hum. Nutr. 1994, 46, 221–229. [Google Scholar] [CrossRef]
- Chen, L.; Lee, M.J.; Li, H.; Yang, C.S. Absorption, distribution, and elimination of tea polyphenols in rats. Drug Metab. Dispos. 1997, 25, 1045–1050. [Google Scholar]
- Bianconi, V.; Mannarino, M.R.; Pirro, M. Tea consumption and the risk of atherosclerotic cardiovascular disease and all-cause mortality: The China-PAR project. Eur. J. Prev. Cardiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Mukhtar, H. Dietary agents for prevention and treatment of lung cancer. Cancer Lett. 2015, 359, 155–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wada, K.; Oba, S.; Tsuji, M.; Goto, Y.; Mizuta, F.; Koda, S.; Uji, T.; Hori, A.; Tanabashi, S.; Matsushita, S.; et al. Green tea intake and colorectal cancer risk in Japan: The Takayama study. Jpn. J. Clin. Oncol. 2019, 49, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.Q.; Lu, X.; Min, H.; Wu, Q.Q.; Shi, X.T.; Bian, K.Q.; Zou, X.P. Green tea and liver cancer risk: A meta-analysis of prospective cohort studies in Asian populations. Nutrition 2016, 32, 3–8. [Google Scholar] [CrossRef]
- Kumar, N.B.; Pow-Sang, J.; Egan, K.M.; Spiess, P.E.; Dickinson, S.; Salup, R.; Helal, M.; McLarty, J.; Williams, C.R.; Schreiber, F.; et al. Randomized, placebo-controlled trial of green tea catechins for prostate cancer prevention. Cancer Prev. Res. 2015, 8, 879–887. [Google Scholar] [CrossRef] [Green Version]
- Yang, G.Y.; Liao, J.; Kim, K.; Yurkow, E.J.; Yang, C.S. Inhibition of growth and induction of apoptosis in human cancer cell lines by tea polyphenols. Carcinogenesis 1998, 19, 611–616. [Google Scholar] [CrossRef] [Green Version]
- Seo, E.J.; Wu, C.F.; Ali, Z.; Wang, Y.H.; Khan, S.I.; Walker, L.A.; Khan, I.A.; Efferth, T. Both phenolic and non-phenolic green tea fractions inhibit migration of cancer cells. Front. Pharmacol. 2016, 7, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Srichairatanakool, S.; Ounjaijean, S.; Thephinlap, C.; Khansuwan, U.; Phisalpong, C.; Fucharoen, S. Iron-chelating and free-radical scavenging activities of microwave-processed green tea in iron overload. Hemoglobin 2006, 30, 311–327. [Google Scholar] [CrossRef]
- Jankun, J.; Selman, S.H.; Swiercz, R.; Skrzypczak-Jankun, E. Why drinking green tea could prevent cancer. Nature 1997, 387, 561. [Google Scholar] [CrossRef]
- Ruch, R.J.; Cheng, S.J.; Klaunig, J.E. Prevention of cytotoxicity and inhibition of intercellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis 1989, 10, 1003–1008. [Google Scholar] [CrossRef]
- Rafat Husain, S.; Cillard, J.; Cillard, P. Hydroxyl radical scavenging activity of flavonoids. Phytochemistry 1987, 26, 2489–2491. [Google Scholar] [CrossRef]
- Hou, Z.; Sang, S.; You, H.; Lee, M.J.; Hong, J.; Chin, K.V.; Yang, C.S. Mechanism of Action of (−)-Epigallocatechin-3-Gallate: Auto-oxidation–dependent inactivation of epidermal growth factor receptor and direct effects on growth inhibition in human esophageal cancer KYSE 150 Cells. Cancer Res. 2005, 65, 8049–8056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, L.Y.; Cheung, T.C.; Kong, S.K.; Fung, K.P.; Choy, Y.M.; Chan, Z.Y.; Kwok, T.T. Induction of apoptosis by green tea catechins in human prostate cancer DU145 cells. Life Sci. 2001, 68, 1207–1214. [Google Scholar] [CrossRef]
- Ly, J.D.; Grubb, D.R.; Lawen, A. The mitochondrial membrane potential (Δψm) in apoptosis; an update. Apoptosis 2003, 8, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Tajeddine, N. How do reactive oxygen species and calcium trigger mitochondrial membrane permeabilisation? Biochim. Biophys. Acta 2016, 1860, 1079–1088. [Google Scholar] [CrossRef]
- Thakur, V.S.; Gupta, K.; Gupta, S. Green tea polyphenols causes cell cycle arrest and apoptosis in prostate cancer cells by suppressing class I histone deacetylases. Carcinogenesis 2011, 33, 377–384. [Google Scholar] [CrossRef] [Green Version]
- Qin, J.; Xie, L.P.; Zheng, X.Y.; Wang, Y.B.; Bai, Y.; Shen, H.-F.; Li, L.C.; Dahiya, R. A component of green tea, (−)-epigallocatechin-3-gallate, promotes apoptosis in T24 human bladder cancer cells via modulation of the PI3K/Akt pathway and Bcl-2 family proteins. Biochem. Biophys. Res. Commun. 2007, 354, 852–857. [Google Scholar] [CrossRef]
- Ahmad, N.; Gupta, S.; Mukhtar, H. Green tea polyphenol epigallocatechin-3-gallate differentially modulates nuclear factor κB in cancer cells versus normal cells. Arch. Biochem. Biophys. 2000, 376, 338–346. [Google Scholar] [CrossRef]
- Gu, J.W.; Makey, K.L.; Tucker, K.B.; Chinchar, E.; Mao, X.; Pei, I.; Thomas, E.Y.; Miele, L. EGCG, a major green tea catechin suppresses breast tumor angiogenesis and growth via inhibiting the activation of HIF-1α and NFκB, and VEGF expression. Vasc. Cell 2013, 5, 9. [Google Scholar] [CrossRef] [Green Version]
- Terwilliger, T.; Abdul-Hay, M. Acute lymphoblastic leukemia: A comprehensive review and 2017 update. Blood Cancer J. 2017, 7, e577. [Google Scholar] [CrossRef] [Green Version]
- Turesson, I.; Bjorkholm, M.; Blimark, C.H.; Kristinsson, S.; Velez, R.; Landgren, O. Rapidly changing myeloma epidemiology in the general population: Increased incidence, older patients, and longer survival. Eur. J. Haematol. 2018, 101, 237–244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexander, D.D.; Mink, P.J.; Adami, H.O.; Cole, P.; Mandel, J.S.; Oken, M.M.; Trichopoulos, D. Multiple myeloma: A review of the epidemiologic literature. Int. J. Cancer 2007, 120 (Suppl. 12), 40–61. [Google Scholar] [CrossRef] [PubMed]
- Andres, M.; Feller, A.; Arndt, V. Trends of incidence, mortality, and survival of multiple myeloma in Switzerland between 1994 and 2013. Cancer Epidemiol. 2018, 53, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Katz, A.J.; Chia, V.M.; Schoonen, W.M.; Kelsh, M.A. Acute lymphoblastic leukemia: An assessment of international incidence, survival, and disease burden. Cancer Causes Control 2015, 26, 1627–1642. [Google Scholar] [CrossRef]
- Maiso, P.; Huynh, D.; Moschetta, M.; Sacco, A.; Aljawai, Y.; Mishima, Y.; Asara, J.M.; Roccaro, A.M.; Kimmelman, A.C.; Ghobrial, I.M. Metabolic signature identifies novel targets for drug resistance in multiple myeloma. Cancer Res. 2015, 75, 2071–2082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mansoori, B.; Mohammadi, A.; Davudian, S.; Shirjang, S.; Baradaran, B. The different mechanisms of cancer drug resistance: A brief review. Adv. Pharm Bull. 2017, 7, 339–348. [Google Scholar] [CrossRef]
- Fletcher, J.I.; Williams, R.T.; Henderson, M.J.; Norris, M.D.; Haber, M. ABC transporters as mediators of drug resistance and contributors to cancer cell biology. Drug Resis. Updat. 2016, 26, 1–9. [Google Scholar] [CrossRef]
- Niruri, R.; Narayani, I.; Ariawati, K.; Herawati, S. P-glycoprotein expression on patients with acute lymphoblastic leukemia. J. Health Sci. Med. 2017, 1, 39–41. [Google Scholar] [CrossRef] [Green Version]
- Miyakoshi, S.; Kami, M.; Yuji, K.; Matsumura, T.; Takatoku, M.; Sasaki, M.; Narimatsu, H.; Fujii, T.; Kawabata, M.; Taniguchi, S.; et al. Severe pulmonary complications in Japanese patients after bortezomib treatment for refractory multiple myeloma. Blood 2006, 107, 3492–3494. [Google Scholar] [CrossRef] [Green Version]
- Bockorny, M.; Bockorny, M.; Chakravarty, S.; Schulman, P.; Bockorny, B.; Bona, R. Severe heart failure after bortezomib treatment in a patient with multiple myeloma: A case report and review of the literature. Acta Haematol. 2012, 128, 244–247. [Google Scholar] [CrossRef]
- Stammler, G.; Volm, M. Green tea catechins (EGCG and EGC) have modulating effects on the activity of doxorubicin in drug-resistant cell lines. Anticancer Drugs 1997, 8, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.; Kuete, V.; Kadioglu, O.; Börtzler, J.; Khalid, H.; Greten, H.J.; Efferth, T. Cytotoxicity of the bisphenolic honokiol from Magnolia officinalis against multiple drug-resistant tumor cells as determined by pharmacogenomics and molecular docking. Phytomedicine 2014, 21, 1525–1533. [Google Scholar] [CrossRef] [PubMed]
- Kimmig, A.; Gekeler, V.; Neumann, M.; Frese, G.; Handgretinger, R.; Kardos, G.; Diddens, H.; Niethammer, D. Susceptibility of multidrug-resistant human leukemia cell lines to human interleukin 2-activated killer cells. Cancer Res. 1990, 50, 6793–6799. [Google Scholar] [PubMed]
- O’Brien, J.; Wilson, I.; Orton, T.; Pognan, F. Investigation of the Alamar Blue (resazurin) fluorescent dye for the assessment of mammalian cell cytotoxicity. Eur. J. Biochem. 2000, 267, 5421–5426. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Sauerbrey, A.; Olbrich, A.; Gebhart, E.; Rauch, P.; Weber, H.O.; Hengstler, J.G.; Halatsch, M.-E.; Volm, M.; Tew, K.D.; et al. Molecular modes of action of artesunate in tumor cell lines. Mol. Pharmacol. 2003, 64, 382–394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mbaveng, A.T.; Damen, F.; Çelik, İ.; Tane, P.; Kuete, V.; Efferth, T. Cytotoxicity of the crude extract and constituents of the bark of Fagara tessmannii towards multi-factorial drug resistant cancer cells. J. Ethnopharmacol. 2019, 235, 28–37. [Google Scholar] [CrossRef]
- Mbaveng, A.T.; Damen, F.; Simo Mpetga, J.D.; Awouafack, M.D.; Tane, P.; Kuete, V.; Efferth, T. Cytotoxicity of crude extract and isolated constituents of the dichrostachys cinerea bark towards multifactorial drug-resistant cancer cells. Evid. Based Complement. Alternat. Med. 2019, 2019, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Abdelfatah, S.; Fleischer, E.; Klinger, A.; Wong, V.K.W.; Efferth, T. Identification of inhibitors of the polo-box domain of polo-like kinase 1 from natural and semisynthetic compounds. Invest. New Drugs 2019, 2019, 1–9. [Google Scholar] [CrossRef]
- Krishan, A. Rapid flow cytofluorometric analysis of mammalian cell cycle by propidium iodide staining. J. Cell Biol. 1975, 66, 188–193. [Google Scholar] [CrossRef] [Green Version]
- Vermes, I.; Haanen, C.; Steffens-Nakken, H.; Reutellingsperger, C. A novel assay for apoptosis Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. J. Immunol. Methods 1995, 184, 39–51. [Google Scholar] [CrossRef] [Green Version]
- Rescher, U.; Gerke, V. Annexins—Unique membrane binding proteins with diverse functions. J. Cell Sci. 2004, 117, 2631–2639. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cossarizza, A.; Ferraresi, R.; Troiano, L.; Roat, E.; Gibellini, L.; Bertoncelli, L.; Nasi, M.; Pinti, M. Simultaneous analysis of reactive oxygen species and reduced glutathione content in living cells by polychromatic flow cytometry. Nat. Protoc. 2009, 4, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Özenver, N.; Saeed, M.; Demirezer, L.Ö.; Efferth, T. Aloe-emodin as drug candidate for cancer therapy. Oncotarget 2018, 9, 17770–17796. [Google Scholar] [CrossRef] [Green Version]
- Amundson, S.A.; Do, K.T.; Vinikoor, L.C.; Lee, R.A.; Koch-Paiz, C.A.; Ahn, J.; Reimers, M.; Chen, Y.; Scudiero, D.A.; Weinstein, J.N.; et al. Integrating global gene expression and radiation survival parameters across the 60 cell Lines of the national cancer institute anticancer drug screen. Cancer Res. 2008, 68, 415–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paull, K.D.; Shoemaker, R.H.; Hodes, L.; Monks, A.; Scudiero, D.A.; Rubinstein, L.; Plowman, J.; Boyd, M.R. Display and analysis of patterns of differential activity of drugs against human tumor cell lines: Development of mean graph and COMPARE algorithm. J. Natl. Cancer Inst. 1989, 81, 1088–1092. [Google Scholar] [CrossRef] [PubMed]
- Fagan, V.; Bonham, S.; Carty, M.P.; Saenz-Mendez, P.; Eriksson, L.A.; Aldabbagh, F. COMPARE analysis of the toxicity of an iminoquinone derivative of the imidazo[5,4-f]benzimidazoles with NAD(P)H:quinone oxidoreductase 1 (NQO1) activity and computational docking of quinones as NQO1 substrates. Bioorg. Med. Chem. 2012, 20, 3323–3332. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.E.M.; Mahmoud, N.; Sugimoto, Y.; Efferth, T.; Abdel-Aziz, H. Molecular determinants of sensitivity or resistance of cancer cells toward sanguinarine. Front. Pharmacol. 2018, 9, 1–15. [Google Scholar] [CrossRef]
- Dawood, M.; Hamdoun, S.; Efferth, T. Multifactorial modes of action of arsenic trioxide in cancer cells as analyzed by classical and network pharmacology. Front. Pharmacol. 2018, 9, 1–19. [Google Scholar] [CrossRef]
- Kadioglu, O.; Efferth, T. Pharmacogenomic characterization of cytotoxic compounds from salvia officinalis in cancer cells. J. Nat. Prod. 2015, 78, 762–775. [Google Scholar] [CrossRef]
- Jiménez-Marín, Á.; Collado-Romero, M.; Ramirez-Boo, M.; Arce, C.; Garrido, J.J. Biological pathway analysis by ArrayUnlock and Ingenuity Pathway Analysis. BMC Proc. 2009, 3, S6. [Google Scholar] [CrossRef] [Green Version]
- Jia, P.; Kao, C.F.; Kuo, P.H.; Zhao, Z. A comprehensive network and pathway analysis of candidate genes in major depressive disorder. BMC Syst. Biol. 2011, 5, S12. [Google Scholar] [CrossRef] [Green Version]
- Natrajan, R.; Weigelt, B.; Mackay, A.; Geyer, F.C.; Grigoriadis, A.; Tan, D.S.P.; Jones, C.; Lord, C.J.; Vatcheva, R.; Rodriguez-Pinilla, S.M.; et al. An integrative genomic and transcriptomic analysis reveals molecular pathways and networks regulated by copy number aberrations in basal-like, HER2 and luminal cancers. Breast Cancer Res. Treat. 2010, 121, 575–589. [Google Scholar] [CrossRef]
- Dawood, M.; Ooko, E.; Efferth, T. Collateral sensitivity of parthenolide via NF-kappaB and HIF-alpha inhibition and epigenetic changes in drug-resistant cancer cell lines. Front. Pharmacol. 2019, 10, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allard, P.M.; Péresse, T.; Bisson, J.; Gindro, K.; Marcourt, L.; Pham, V.C.; Roussi, F.; Litaudon, M.; Wolfender, J.L. Integration of molecular networking and in-silico MS/MS fragmentation for natural products dereplication. Anal. Chem. 2016, 88, 3317–3323. [Google Scholar] [CrossRef]
- Otto, T.; Sicinski, P. Cell cycle proteins as promising targets in cancer therapy. Nat. Rev. Cancer 2017, 17, 93–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, G.; Wargovich, M.J.; Dixon, D.A. Anti-proliferative effects of green tea polyphenol EGCG on Ha-Ras-induced transformation of intestinal epithelial cells. Cancer Lett. 2006, 238, 260–270. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Feyes, D.K.; Agarwal, R.; Mukhtar, H.; Nieminen, A.L. Green tea constituent epigallocatechin-3-gallate and induction of apoptosis and cell cycle arrest in human carcinoma cells. J. Natl. Cancer Inst. 1997, 89, 1881–1886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forester, S.C.; Lambert, J.D. The role of antioxidant versus pro-oxidant effects of green tea polyphenols in cancer prevention. Mol. Nutr. Food Res. 2011, 55, 844–854. [Google Scholar] [CrossRef] [Green Version]
- Ott, M.; Gogvadze, V.; Orrenius, S.; Zhivotovsky, B. Mitochondria, oxidative stress and cell death. Apoptosis 2007, 12, 913–922. [Google Scholar] [CrossRef]
- Indo, H.P.; Davidson, M.; Yen, H.C.; Suenaga, S.; Tomita, K.; Nishii, T.; Higuchi, M.; Koga, Y.; Ozawa, T.; Majima, H.J. Evidence of ROS generation by mitochondria in cells with impaired electron transport chain and mitochondrial DNA damage. Mitochondrion 2007, 7, 106–118. [Google Scholar] [CrossRef]
- Trachootham, D.; Alexandre, J.; Huang, P. Targeting cancer cells by ROS-mediated mechanisms: A radical therapeutic approach? Nat. Rev. Drug Discov. 2009, 8, 579–591. [Google Scholar] [CrossRef]
- Ran, Z.H.; Xu, Q.; Tong, J.L.; Xiao, S.D. Apoptotic effect of Epigallocatechin-3-gallate on the human gastric cancer cell line MKN45 via activation of the mitochondrial pathway. World J. Gastroenterol. 2007, 13, 4255–4259. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Koch, E. Complex interactions between phytochemicals. The multi-target therapeutic concept of phytotherapy. Curr. Drug Targets 2011, 12, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Mancini, M.; Yarden, Y. Mutational and network level mechanisms underlying resistance to anti-cancer kinase inhibitors. Semin. Cell Dev. Biol. 2016, 50, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Konkimalla, V.B.; Wang, Y.F.; Sauerbrey, A.; Meinhardt, S.; Zintl, F.; Mattern, J.; Volm, M. Prediction of broad spectrum resistance of tumors towards anticancer drugs. Clin. Cancer Res. 2008, 14, 2405–2412. [Google Scholar] [CrossRef] [Green Version]
- Davis, T.B.; Yang, M.; Wang, H.; Lee, C.; Yeatman, T.J.; Pledger, W.J. PTPRS drives adaptive resistance to MEK/ERK inhibitors through SRC. Oncotarget 2019, 10, 6768–6780. [Google Scholar] [CrossRef] [PubMed]
- Byrgazov, K.; Kastner, R.; Gorna, M.; Hoermann, G.; Koenig, M.; Lucini, C.B.; Ulreich, R.; Benesch, M.; Strenger, V.; Lackner, H.; et al. NDEL1-PDGFRB fusion gene in a myeloid malignancy with eosinophilia associated with resistance to tyrosine kinase inhibitors. Leukemia 2017, 31, 237–240. [Google Scholar] [CrossRef] [Green Version]
- Zeng, A.L.; Yan, W.; Liu, Y.W.; Wang, Z.; Hu, Q.; Nie, E.; Zhou, X.; Li, R.; Wang, X.F.; Jiang, T.; et al. Tumour exosomes from cells harbouring PTPRZ1–MET fusion contribute to a malignant phenotype and temozolomide chemoresistance in glioblastoma. Oncogene 2017, 36, 5369–5381. [Google Scholar] [CrossRef] [Green Version]
- Jiang, L.; Chan, J.Y.W.; Fung, K.P. Epigenetic loss of CDH1 correlates with multidrug resistance in human hepatocellular carcinoma cells. Biochem. Biophys. Res. Commun. 2012, 422, 739–744. [Google Scholar] [CrossRef]
- Fu, X.; Feng, J.; Zeng, D.; Ding, Y.; Yu, C.; Yang, B. PAK4 confers cisplatin resistance in gastric cancer cells via PI3K/Akt- and MEK/ERK-dependent pathways. Biosci. Rep. 2014, 34. [Google Scholar] [CrossRef]
- Yang, F.; Fang, H.; Wang, D.; Chen, Y.; Zhai, Y.; Zhou, B.-B.S.; Li, H. HPRT1 activity loss is associated with resistance to thiopurine in ALL. Oncotarget 2017, 9, 2268–2278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsofack, S.P.; Meunier, L.; Sanchez, L.; Madore, J.; Provencher, D.; Mes-Masson, A.M.; Lebel, M. Low expression of the X-linked ribosomal protein S4 in human serous epithelial ovarian cancer is associated with a poor prognosis. BMC Cancer 2013, 13, 303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, F.; Lee, J.T.; Navolanic, P.M.; Steelman, L.S.; Shelton, J.G.; Blalock, W.L.; Franklin, R.A.; McCubrey, J.A. Involvement of PI3K/Akt pathway in cell cycle progression, apoptosis, and neoplastic transformation: A target for cancer chemotherapy. Leukemia 2003, 17, 590–603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shankar, S.; Chen, Q.; Srivastava, R.K. Inhibition of PI3K/AKT and MEK/ERK pathways act synergistically to enhance antiangiogenic effects of EGCG through activation of FOXO transcription factor. J. Mol. Signal. 2008, 3, 7. [Google Scholar] [CrossRef] [Green Version]
- Hoesel, B.; Schmid, J.A. The complexity of NF-κB signaling in inflammation and cancer. Mol. Cancer 2013, 12, 86. [Google Scholar] [CrossRef] [Green Version]
- Xia, Y.; Shen, S.; Verma, I.M. NF-κB, an active player in human cancers. Cancer Immunol. Res. 2014, 2, 823–830. [Google Scholar] [CrossRef] [Green Version]
- Kordes, U.; Krappmann, D.; Heissmeyer, V.; Ludwig, W.D.; Scheidereit, C. Transcription factor NF-κB is constitutively activated in acute lymphoblastic leukemia cells. Leukemia 2000, 14, 399–402. [Google Scholar] [CrossRef] [Green Version]
- Tang, X.; Liu, D.; Shishodia, S.; Ozburn, N.; Behrens, C.; Lee, J.J.; Hong, W.K.; Aggarwal, B.B.; Wistuba, I.I. Nuclear factor-κB (NF-κB) is frequently expressed in lung cancer and preneoplastic lesions. Cancer 2006, 107, 2637–2646. [Google Scholar] [CrossRef]
- Suhail, M.; Perveen, A.; Husain, A.; Rehan, M. Exploring inhibitory mechanisms of green tea catechins as inhibitors of a cancer therapeutic target, nuclear factor-κB (NF-κB). Biosci. Biotech. Res Asia 2019, 16. [Google Scholar] [CrossRef]
- Luo, K.W.; Wei, C.; Lung, W.Y.; Wei, X.Y.; Cheng, B.H.; Cai, Z.M.; Huang, W.R. EGCG inhibited bladder cancer SW780 cell proliferation and migration both in vitro and in vivo via down-regulation of NF-κB and MMP-9. J. Nutr. Biochem. 2017, 41, 56–64. [Google Scholar] [CrossRef]
- Lagha, A.B.; Grenier, D. Tea polyphenols inhibit the activation of NF-κB and the secretion of cytokines and matrix metalloproteinases by macrophages stimulated with Fusobacterium nucleatum. Sci. Rep. 2016, 6, 34520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Cell Lines | Sencha-MeOH | Sencha-70% MeOH | Sencha-H2O | Doxorubicin | ||||
|---|---|---|---|---|---|---|---|---|
| IC50(μg) | Resistance Degree | IC50(μg) | Resistance Degree | IC50(μg) | Resistance Degree | IC50(μM) | Resistance Degree | |
| CCRF/CEM | 8.38 ± 0.72 | 2.21 | 11.34 ± 1.86 | 1.90 | 11.50 ± 1.30 | 2.94 | 0.0093 ± 0.00 | 7758.78 |
| CEM/ADR5000 | 18.52 ± 1.98 | 21.57 ± 2.69 | 33.8 ± 3.55 | 72.43 ± 2.61 | ||||
| Cell Line | KMS-12-BM | MolP-8 | KMS-11 | NCI-H929 | AMO-1 | L-363 | OPM-2 | JJN-3 | RPMI-8226 |
|---|---|---|---|---|---|---|---|---|---|
| Sencha-MeOH extract (μg) | 11.37 ± 1.03 | 16.51 ± 2.32 | 17.13 ± 2.53 | 18.23 ± 0.59 | 39.26 ± 2.72 | 61.65 ± 9.37 | 42.20 ± 4.80 | 51.74 ± 3.74 | 68.90 ± 6.25 |
| Sencha-H2O extract (μg) | 14.85 ± 1.44 | 24.38 ± 3.58 | 52.66 ± 4.22 | 25.52 ± 5.56 | 42.58 ± 2.81 | 71.19 ± 1.88 | 76.63 ± 1.36 | >80 | >80 |
| Bortezomib (μM) | 0.0019 ± 0.00013 | 0.0018 ± 0.000078 | 0.0016 ±0.00038 | 0.0014 ± 0.00032 | 0.0020 ± 0.000024 | 0.0018± 0.00016 | 0.0018 ± 0.00020 | 0.0015 ± 0.00044 | 0.0016 ± 0.00035 |
| COMPARE | Experimental | GenBank | Gene | ||
|---|---|---|---|---|---|
| Coefficient | ID | Accession | Symbol | Name | Function |
| −0.605 | GC39119 | Y11999 | ITPKC | Inositol 1,4,5-trisphosphate 3-kinase C RNA | Phosphorylation of inositol 2,4,5-triphosphate to inositol 2,4,5,6-tetraphosphate |
| −0.596 | GC35270 | U40317 | PTPRS | Protein tyrosine phosphatase, receptor type, S RNA | Inhibition of neurite and axonal outgrowth |
| −0.527 | GC37416 | AF038203 | NDEL1 | Nude nuclear distribution gene E homolog (A. nidulans)-like 1 RNA | Organization of the cellular microtubule array and microtubule anchoring at the centrosome |
| −0.526 | GC32502 | M12529 | APOE | Apolipoprotein E RNA | Function in lipoprotein-mediated lipid transport |
| −0.523 | GC31111 | U27699 | SLC6A12 | Solute carrier family 6 (neurotransmitter transporter, betaine/GABA), member 12 RNA | Regulation of gabaergic transmission in the brain |
| −0.516 | GC34680 | U62647 | DNASE1L2 | Deoxyribonuclease I-like 2 RNA | Breakdown of the nucleus during corneocyte formation of epidermal keratinocytes |
| −0.512 | GC32691 | X80343 | CDK5R1 | Cyclin-dependent kinase 5, regulatory subunit 1 (p35) RNA | Required for neurite outgrowth and cortical lamination |
| −0.511 | GC29695 | M76558 | CACNA1D | Calcium channel, voltage-dependent, L type, alpha 1D subunit RNA | Mediate the entry of calcium ions into excitable cells |
| −0.511 | GC34733 | W27128 | STK35 | Unknown RNA | Association with PDLIM1 is controversial |
| −0.509 | GC30627 | X68985 | HLF | Hepatic leukemia factor RNA | Accumulate according to a robust circadian rhythm |
| −0.503 | GC30214 | M23115 | ATP2A2 | Atpase, Ca++ transporting, cardiac muscle, slow twitch 2 RNA | Catalyze the hydrolysis of ATP coupled with the translocation of calcium |
| −0.495 | GC30715 | AA471042 | KLHL35 | Kelch-like 35 (Drosophila) RNA | Interactions with 2 proteins |
| −0.494 | GC37908 | AF046873 | SYN3 | Synapsin III RNA | Involved in the regulation of neurotransmitter release and synaptogenesis |
| −0.494 | GC33357 | M93426 | PTPRZ1 | Protein tyrosine phosphatase, receptor-type, Z polypeptide 1 RNA | Negatively regulate oligodendrocyte precursor proliferation in the embryonic spinal cord |
| −0.489 | GC35321 | AA844998 | PPY | Pancreatic polypeptide RNA | A regulator of pancreatic and gastrointestinal functions |
| −0.488 | GC34918 | AJ010228 | RFPL1 | Ret finger protein-like 1 RNA | Negatively regulate the G2-M phase transition by promoting cyclin B1/CCNB1 and CDK1 proteasomal degradation |
| −0.475 | GC34162 | L08599 | CDH1 | Cadherin 1, type 1, E-cadherin (epithelial) RNA | Contribute to the sorting of heterogeneous cell types |
| −0.473 | GC36409 | AI332820 | TCF25 | Transcription factor 25 (basic helix-loop-helix) RNA | Control of cell death and repress transcription of SRF |
| −0.473 | GC33285 | D83542 | CDH15 | Cadherin 15, type 1, M-cadherin (myotubule) RNA | Contribute to the sorting of heterogeneous cell types |
| −0.47 | GC36811 | AF005046 | PAK4 | P21 protein (Cdc42/Rac)-activated kinase 4 RNA | Serve as targets for the small GTP binding proteins Cdc42 and Rac |
| 0.501 | GC28045 | M31642 | HPRT1 | Hypoxanthine phosphoribosyltransferase 1 RNA | Play a central role in the generation of purine nucleotides |
| 0.474 | GC29847 | AF017789 | TCERG1 | Transcription elongation regulator 1 RNA | Regulate transcription elongation in a TATA box-dependent manner |
| 0.456 | GC35203 | AA152202 | NFATC2IP | Nuclear factor of activated T-cells, cytoplasmic, calcineurin-dependent 2 interacting protein RNA | Regulate the magnitude of NFAT-driven transcription of a specific subset of cytokine genes |
| 0.452 | GC27723 | U46570 | TTC1 | Tetratricopeptide repeat domain 1 RNA | Unfolded protein binding |
| 0.45 | GC37383 | AI708889 | COX7C | Cytochrome c oxidase subunit viic RNA | Cytochrome-c oxidase activity |
| 0.445 | GC36655 | U14966 | RPL5 | Ribosomal protein L5 RNA | Responsible for the synthesis of proteins |
| 0.438 | GC38242 | X12791 | SRP19 | Signal recognition particle 19kda RNA | Bind directly to 7S RNA and mediates binding of the 54 kda subunit of the SRP |
| 0.436 | GC31697 | AI553745 | NOP16 | NOP16 nucleolar protein homolog (yeast) RNA | RNA binding |
| 0.436 | GC28146 | J02683 | SLC25A5 | Solute carrier family 25 (mitochondrial carrier; adenine nucleotide translocator), member 5 RNA | Catalyze the exchange of cytoplasmic ADP with mitochondrial ATP across the mitochondrial inner membrane |
| 0.434 | GC29122 | AF054183 | RAN | RAN, member RAS oncogene family RNA | Participate both to the import and the export from the nucleus of proteins and RNAs |
| 0.421 | GC37648 | M58458 | RPS4X | Ribosomal protein S4, X-linked RNA | Positive regulation of cell population proliferation |
| 0.421 | GC28143 | D25547 | PCMT1 | Protein-L-isoaspartate (D-aspartate) O-methyltransferase RNA | Repair and/or degradation of damaged proteins |
| 0.418 | GC30306 | U66035 | TIMM8A | Translocase of inner mitochondrial membrane 8 homolog A (yeast) RNA | A chaperone-like protein that protects the hydrophobic precursors from aggregation |
| 0.414 | GC30977 | AF047185 | NDUFA2 | Unknown RNA | Complex I functions in the transfer of electrons from NADH to the respiratory chain |
| 0.414 | GC35707 | AA151922 | ATG12 | ATG12 autophagy related 12 homolog (S. Cerevisiae) RNA | Involved in autophagy vesicles formation |
| 0.411 | GC29399 | AI223047 | NDUFA6 | NADH dehydrogenase (ubiquinone) 1 alpha subcomplex, 6, 14kDa RNA | Complex I functions in the transfer of electrons from NADH to the respiratory chain |
| 0.408 | GC38177 | AB007893 | PPIP5K2 | Diphosphoinositol pentakisphosphate kinase 2 RNA | Act in concert with the IP6K kinases IP6K1, IP6K2, and IP6K3 to synthesize the diphosphate group-containing inositol pyrophosphates diphosphoinositol pentakisphosphate |
| 0.401 | GC37613 | M24194 | GNB2L1 | Guanine nucleotide binding protein (G protein), beta polypeptide 2-like 1 RNA | Unknown |
| 0.399 | GC37789 | AF047473 | BUB3 | Budding uninhibited by benzimidazoles 3 homolog (yeast) RNA | Regulate chromosome segregation during oocyte meiosis. |
| 0.398 | GC32111 | R38263 | DESL1 | PPPDE peptidase domain containing 2 RNA | Deconjugate SUMO1, SUMO2, and SUMO3 from some substrate proteins |
| Sensitive | Resistant | |
|---|---|---|
| Partition (log10 IC50) | <−4.7 M | ≥−4.7 M |
| Cluster 1 | 8 | 3 |
| Cluster 2 | 8 | 3 |
| Cluster 3 | 2 | 6 |
| Cluster 4 | 3 | 9 |
| Cluster 5 | 4 | 7 |
| Chi-square test | p = 0.037 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, X.; Saeed, M.E.M.; Hegazy, M.-E.F.; Kampf, C.J.; Efferth, T. Chemopreventive Property of Sencha Tea Extracts towards Sensitive and Multidrug-Resistant Leukemia and Multiple Myeloma Cells. Biomolecules 2020, 10, 1000. https://doi.org/10.3390/biom10071000
Lu X, Saeed MEM, Hegazy M-EF, Kampf CJ, Efferth T. Chemopreventive Property of Sencha Tea Extracts towards Sensitive and Multidrug-Resistant Leukemia and Multiple Myeloma Cells. Biomolecules. 2020; 10(7):1000. https://doi.org/10.3390/biom10071000
Chicago/Turabian StyleLu, Xiaohua, Mohamed E. M. Saeed, Mohamed-Elamir F. Hegazy, Christopher J. Kampf, and Thomas Efferth. 2020. "Chemopreventive Property of Sencha Tea Extracts towards Sensitive and Multidrug-Resistant Leukemia and Multiple Myeloma Cells" Biomolecules 10, no. 7: 1000. https://doi.org/10.3390/biom10071000
APA StyleLu, X., Saeed, M. E. M., Hegazy, M.-E. F., Kampf, C. J., & Efferth, T. (2020). Chemopreventive Property of Sencha Tea Extracts towards Sensitive and Multidrug-Resistant Leukemia and Multiple Myeloma Cells. Biomolecules, 10(7), 1000. https://doi.org/10.3390/biom10071000