Anti-Apoptotic and Antioxidant Effects of 3-Epi-Iso-Seco-Tanapartholide Isolated from Artemisia argyi against Iodixanol-Induced Kidney Epithelial Cell Death
Abstract
1. Introduction
2. Results
2.1. General Analytical Procedures
2.2. Plant Material and Isolation of Sesquiterpene Compounds
2.3. Cell Culture
2.4. Cellular Viability Test Using the Ez-Cytox Assay
2.5. Quantification of ROS Levels Using DCF Staining
2.6. Evaluation of Apoptosis
2.7. Immunofluorescence Staining
2.8. Western Blotting Analysis
2.9. Statistical Analysis
3. Results
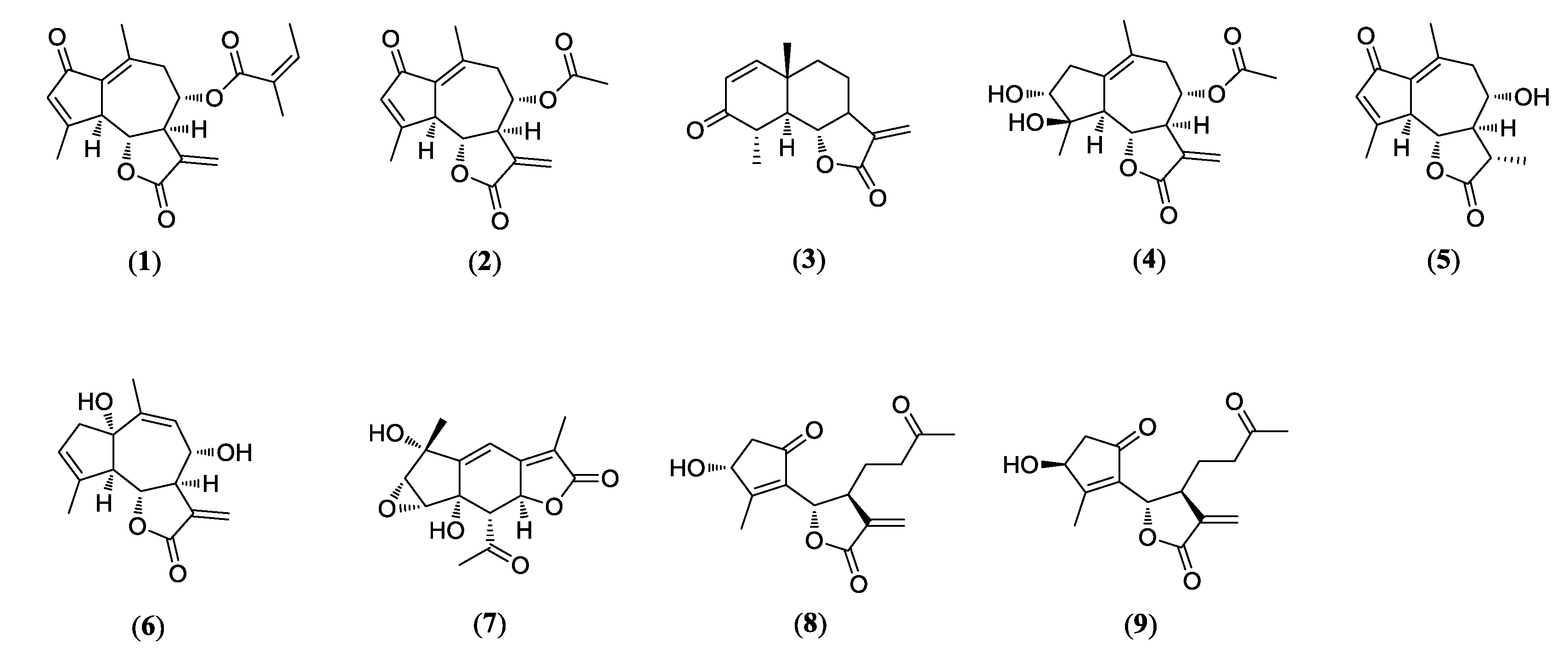
3.1. Identification of Compounds 1–9
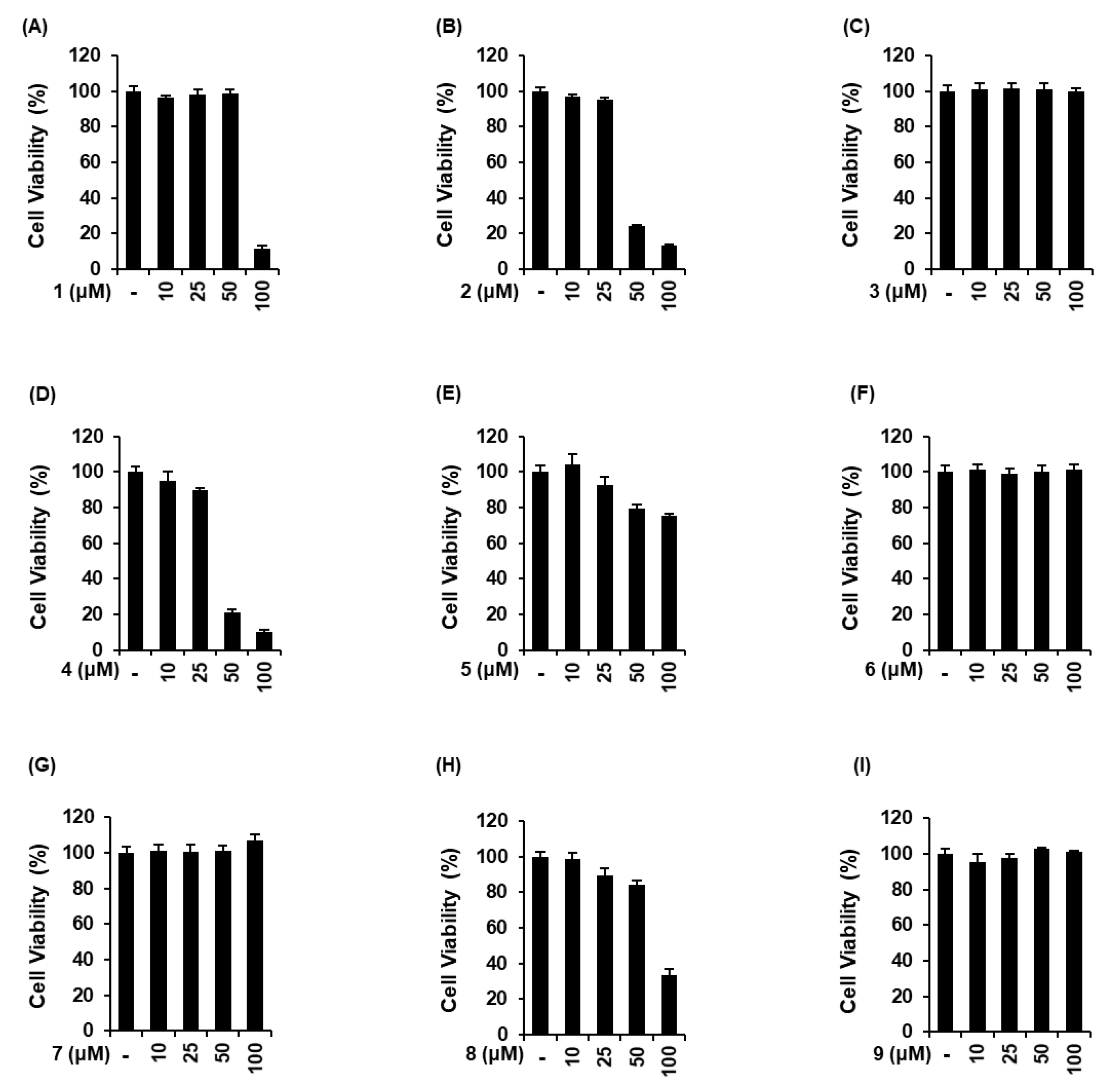
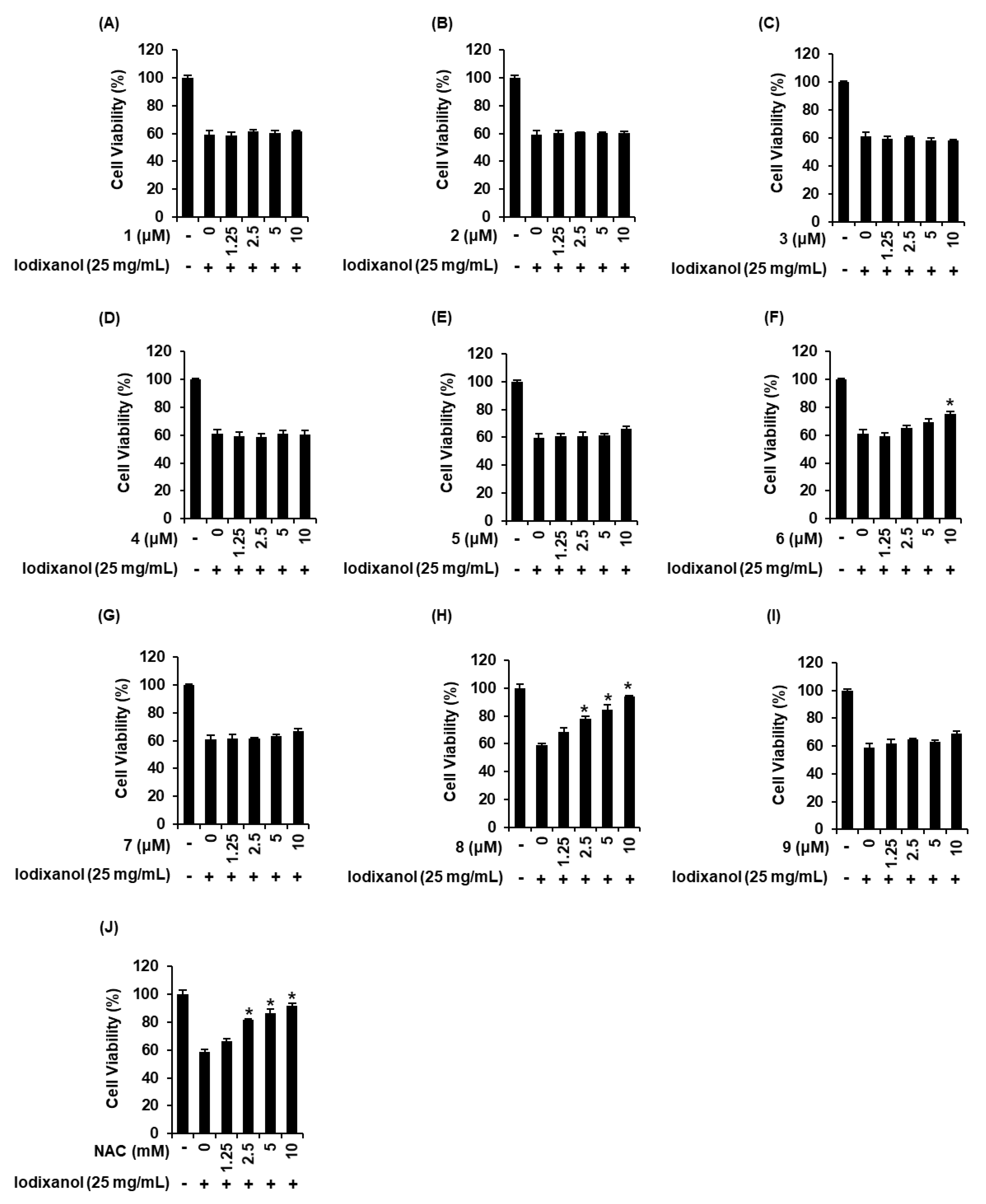
3.2. Comparison of the Protective Effects of 9 Sesquiterpenes Isolated from A. argyi on Contrast Agent-Induced Cytotoxicity in LLC-PK1 Cells
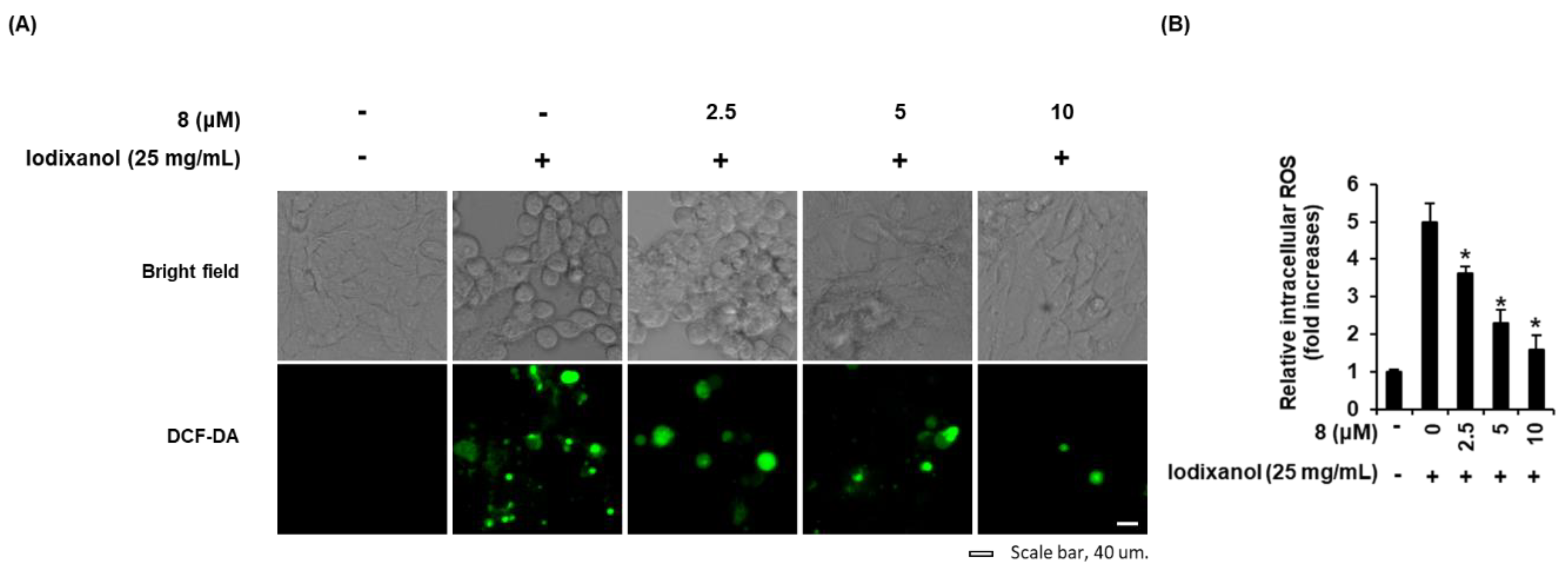
3.3. Effect of 3-Epi-Iso-Seco-Tanapartholide on Contrast Agent-Induced Morphological Changes and ROS Generation in LLC-PK1 Cells
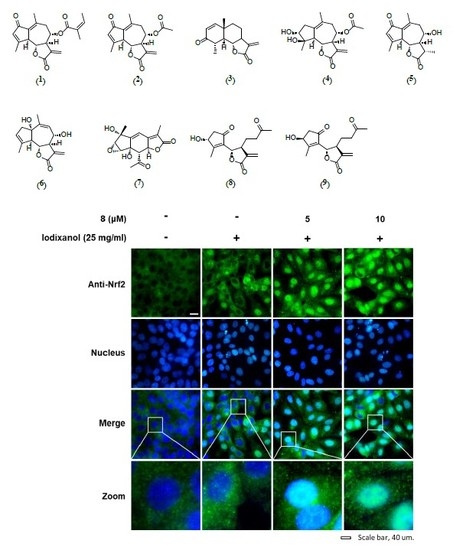
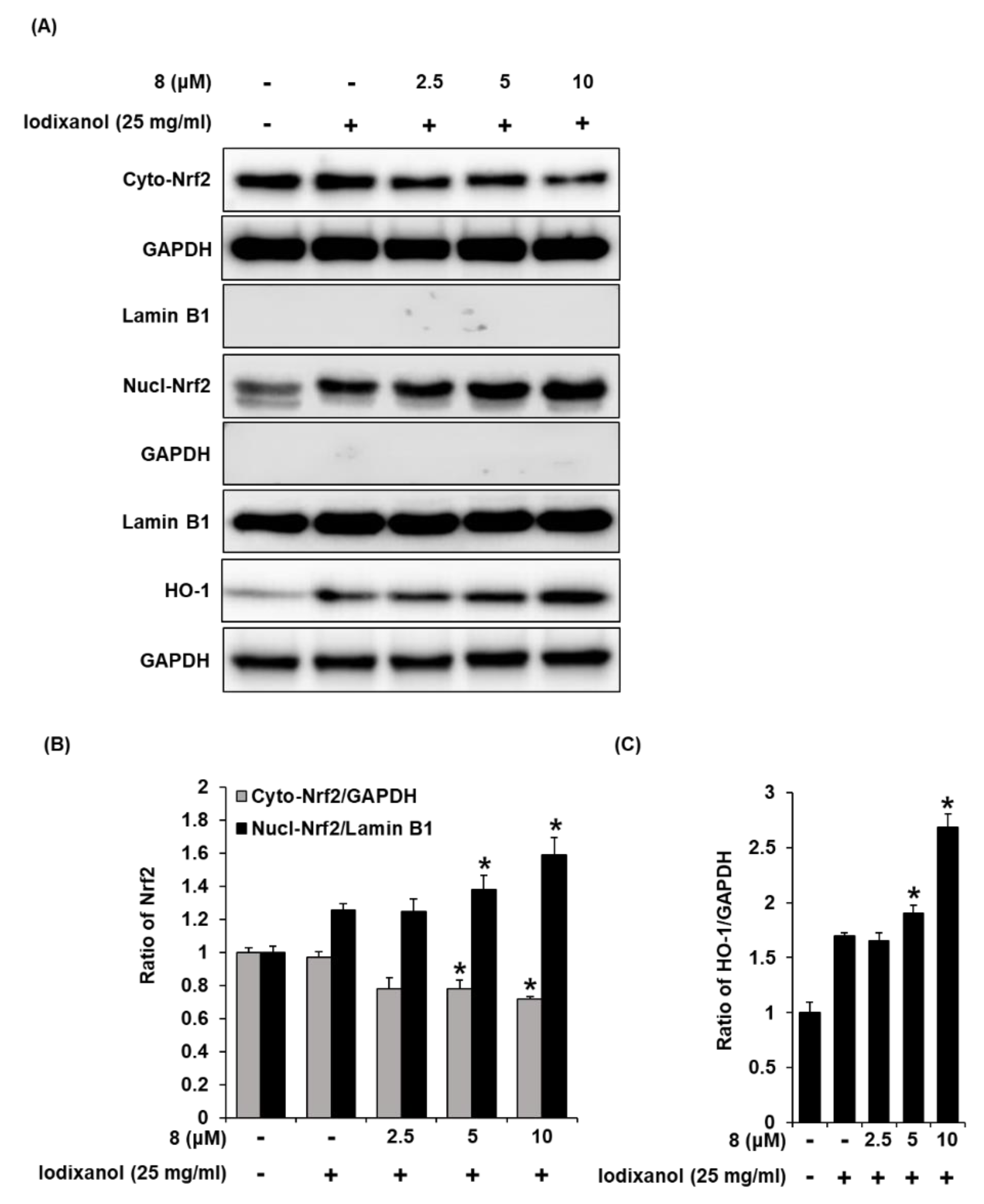
3.4. Effects of 3-Epi-Iso-Seco-Tanapartholide on the Expression Levels of Nrf2/HO-1 Proteins and Nuclear Translocation of Nrf2 Associated with Antioxidant Pathways in Iodixanol-Treated LLC-PK1 Cells
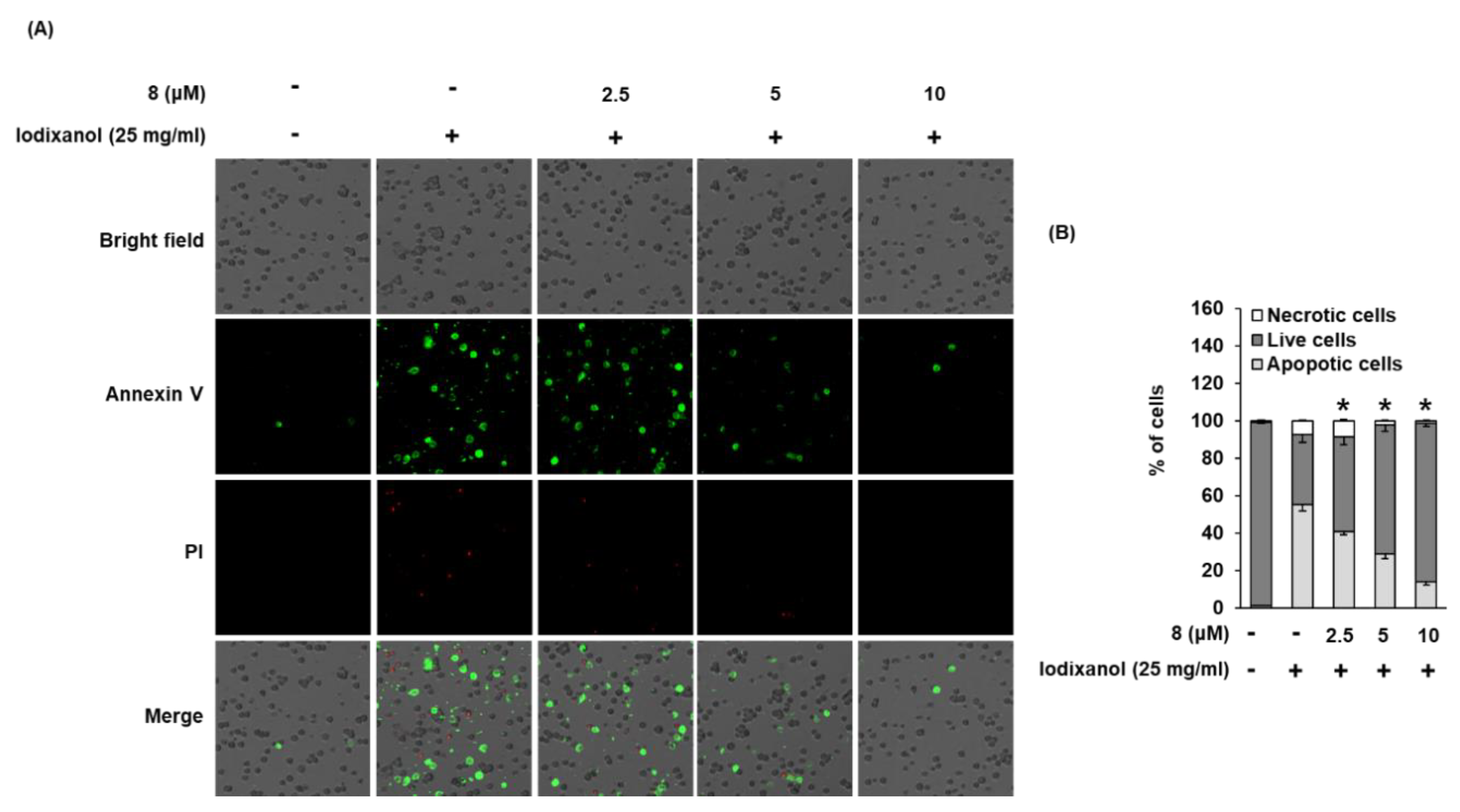
3.5. Effect of 3-Epi-Iso-Seco-Tanapartholide on Iodixanol-Induced Apoptosis in LLC-PK1 Cells
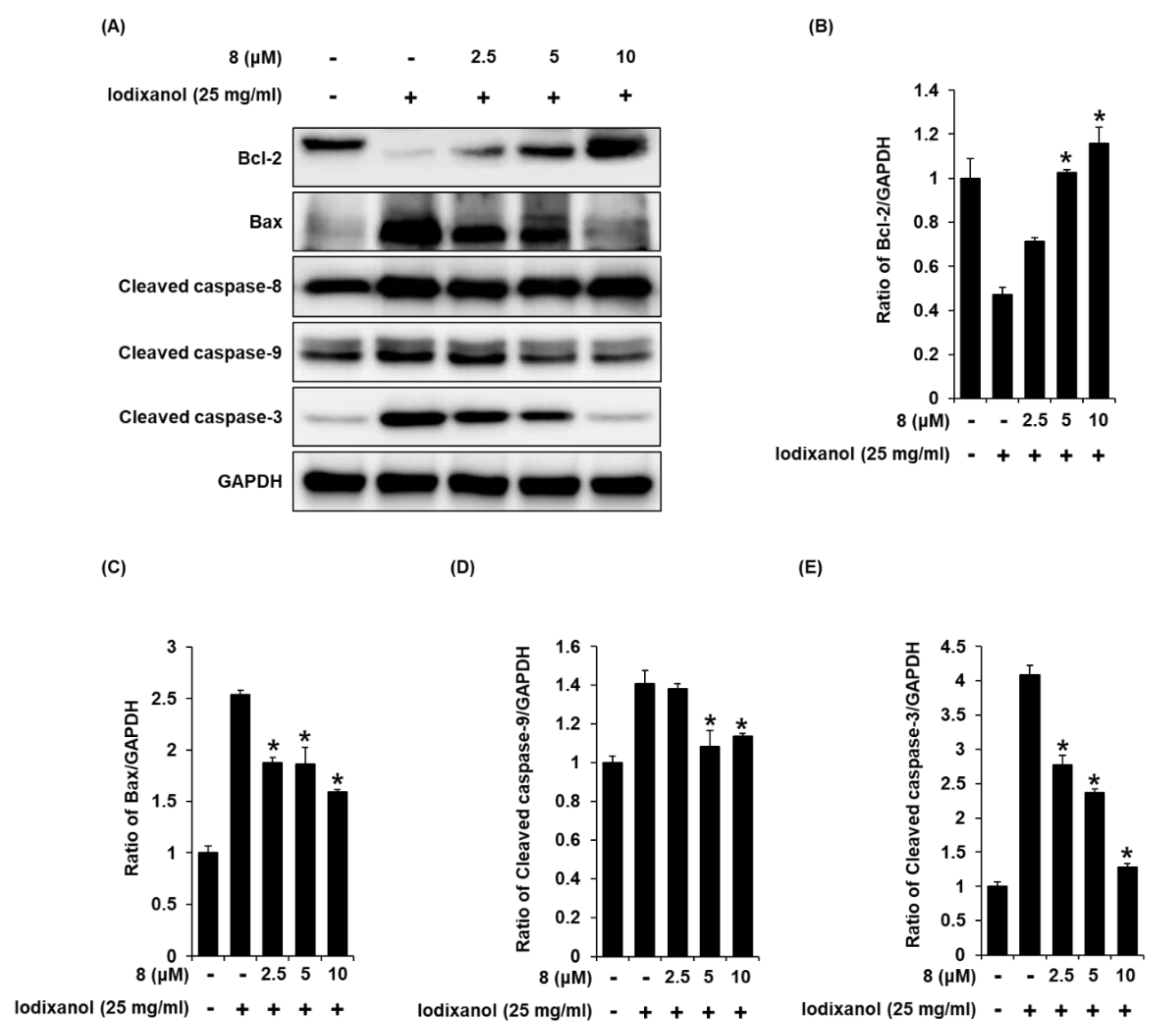
3.6. Effect of 3-Epi-Iso-Seco-Tanapartholide on Expression Levels of Bcl-2, Bax, Cleaved Caspase-8, -9, and -3 Proteins Associated with Apoptosis Pathways in Iodixanol-Treated LLC-PK1 Cells
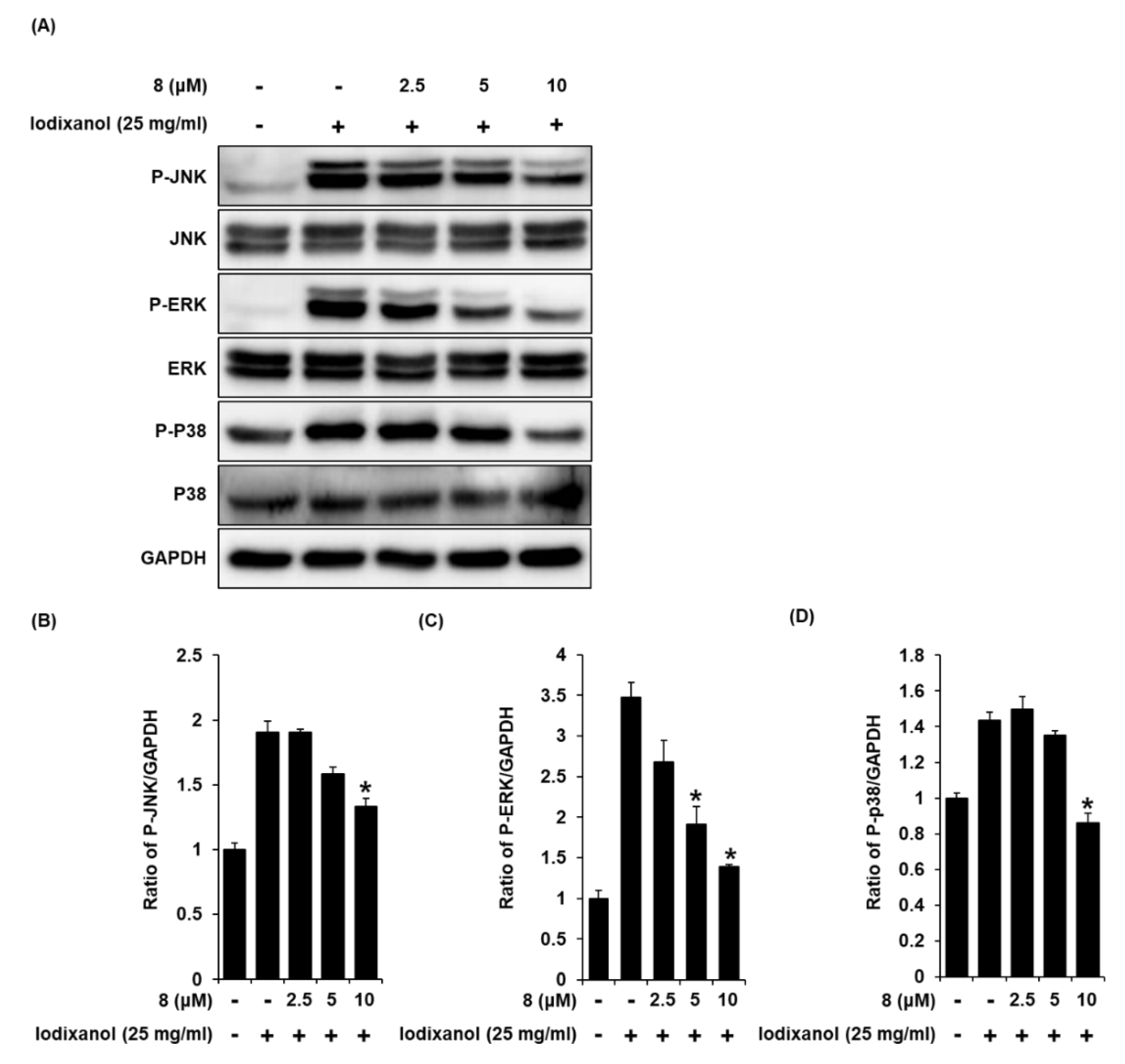
3.7. Effect of 3-Epi-Iso-Seco-Tanapartholide on the Protein Expressions of JNK, ERK, and p38 Proteins Associated with MAPK Pathways in Iodixanol-Treated LLC-PK1 Cells
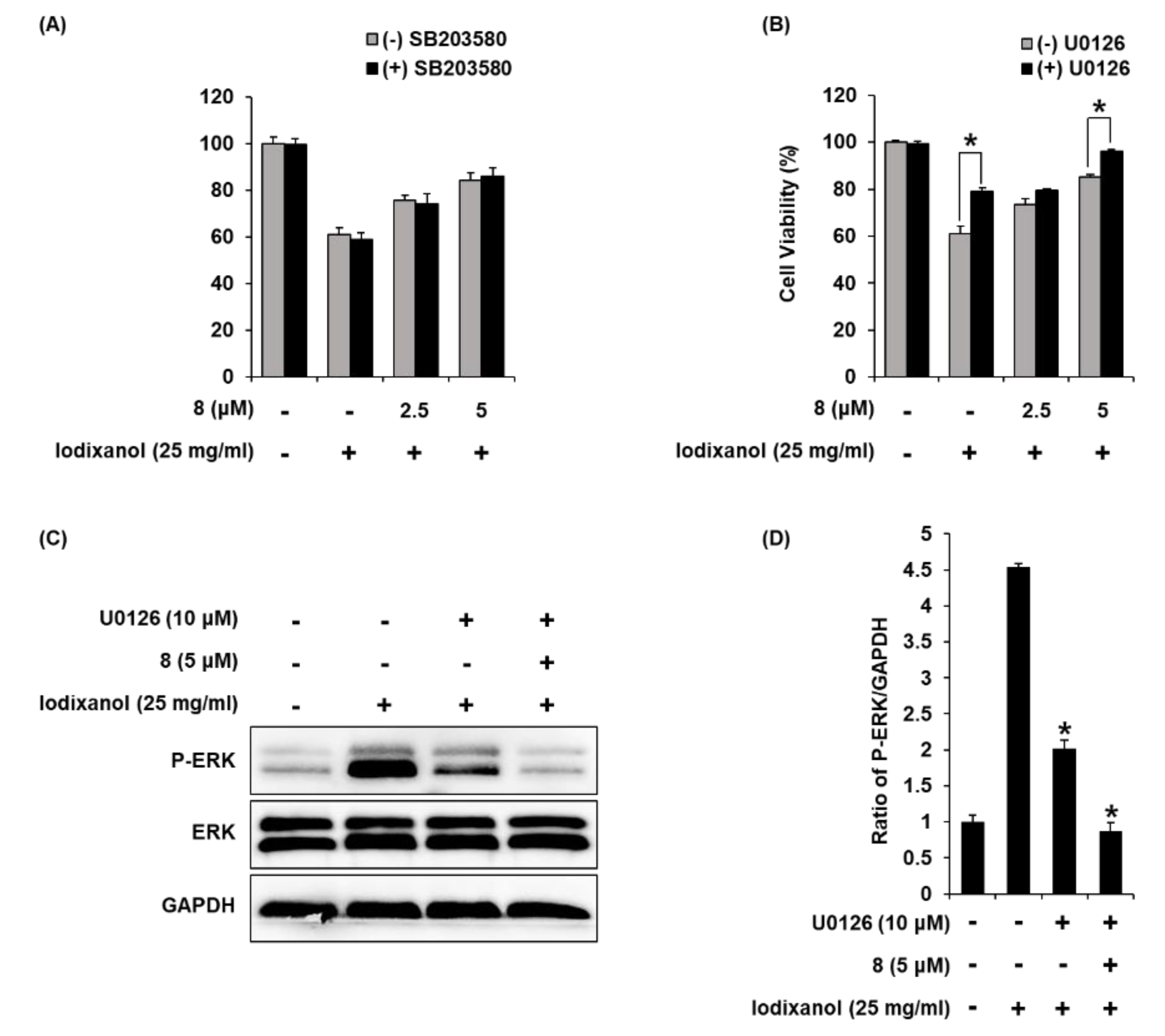
3.8. Effects of Combined Treatment with Inhibitors of MAPK Pathways (SB203580 and U0126) and 3-Epi-Iso-Seco-Tanapartholide on Iodixanol-Treated LLC-PK1 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Andreucci, M.; Solomon, R.; Tasanarong, A. Side effects of radiographic contrast media: Pathogenesis, risk factors, and prevention. Biomed. Res. Int. 2014, 2014, 20. [Google Scholar] [CrossRef]
- Sadat, U. Radiographic contrast-media-induced acute kidney injury: Pathophysiology and prophylactic strategies. ISRN Radiol. 2013, 2013, 496438. [Google Scholar] [CrossRef]
- Mohammed, N.M.; Mahfouz, A.; Achkar, K.; Rafie, I.M.; Hajar, R. Contrast-induced nephropathy. Heart Views 2013, 14, 106. [Google Scholar] [CrossRef]
- Bucher, A.M.; De Cecco, C.N.; Schoepf, U.J.; Meinel, F.G.; Krazinski, A.W.; Spearman, J.V.; McQuiston, A.D.; Wang, R.; Bucher, J.; Vogl, T.J.; et al. Is contrast medium osmolality a causal factor for contrast-induced nephropathy? Biomed. Res. Int. 2014, 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- From, A.M.; Al Badarin, F.J.; McDonald, F.S.; Bartholmai, B.J.; Cha, S.S.; Rihal, C.S. Iodixanol versus low-osmolar contrast media for prevention of contrast induced nephropathy: Meta-analysis of randomized, controlled trials. Circ. Cardiovasc. Interv. 2010, 3, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Mruk, B. Renal safety of iodinated contrast media depending on their osmolarity–current outlooks. Pol. J. Radiol. 2016, 81, 157. [Google Scholar] [CrossRef] [PubMed]
- Andreucci, M.; Faga, T.; Serra, R.; De Sarro, G.; Michael, A. Update on the renal toxicity of iodinated contrast drugs used in clinical medicine. Drug Healthc. Patient Saf. 2017, 9, 25. [Google Scholar] [CrossRef]
- Ge, Y.-B.; Wang, Z.-G.; Xiong, Y.; Huang, X.-J.; Mei, Z.-N.; Hong, Z.-G. Anti-inflammatory and blood stasis activities of essential oil extracted from Artemisia argyi leaf in animals. J. Nat. Med. 2016, 70, 531–538. [Google Scholar] [CrossRef]
- Adams, J.D.; Garcia, C.; Garg, G. Mugwort (Artemisia vulgaris, Artemisia douglasiana, Artemisia argyi) in the treatment of menopause, premenstrual syndrome, dysmenorrhea and attention deficit hyperactivity disorder. Chin. Med. 2012, 3, 116–123. [Google Scholar] [CrossRef]
- Shin, N.-R.; Ryu, H.-W.; Ko, J.-W.; Park, S.-H.; Yuk, H.-J.; Kim, H.-J.; Kim, J.-C.; Jeong, S.-H.; Shin, I.-S. Artemisia argyi attenuates airway inflammation in ovalbumin-induced asthmatic animals. J. Ethnopharmacol. 2017, 209, 108–115. [Google Scholar] [CrossRef]
- Song, X.; Wen, X.; He, J.; Zhao, H.; Li, S.; Wang, M. Phytochemical components and biological activities of Artemisia argyi. J. Funct. Foods 2019, 52, 648–662. [Google Scholar] [CrossRef]
- Lim, J.C.; Park, S.Y.; Nam, Y.; Nguyen, T.T.; Sohn, U.D. The protective effect of eupatilin against hydrogen peroxide-induced injury involving 5-lipoxygenase in feline esophageal epithelial cells. Korean J. Physiol. Pharmacol. 2012, 16, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Zeng, K.-W.; Wang, S.; Dong, X.; Jiang, Y.; Tu, P.-F. Sesquiterpene dimer (DSF-52) from Artemisia argyi inhibits microglia-mediated neuroinflammation via suppression of NF-κB, JNK/p38 MAPKs and Jak2/Stat3 signaling pathways. Phytomedicine 2014, 21, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.Y.; Park, S.K.; Guo, T.J.; Ha, J.S.; Lee, D.S.; Kim, J.M.; Lee, U.; Kim, D.O.; Heo, H.J. Reversal of trimethyltin-induced learning and memory deficits by 3, 5-dicaffeoylquinic acid. Oxid. Med. Cell. Longev. 2016, 2016, 13. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Kim, C.-E.; Park, S.-Y.; Kim, K.O.; Hiep, N.T.; Lee, D.; Jang, H.-J.; Lee, J.W.; Kang, K.S. Protective effect of Artemisia argyi and its flavonoid constituents against contrast-induced cytotoxicity by iodixanol in LLC-PK1 cells. Int. J. Mol. Sci. 2018, 19, 1387. [Google Scholar] [CrossRef]
- Park, J.Y.; Lee, D.; Jang, H.-J.; Jang, D.S.; Kwon, H.C.; Kim, K.H.; Kim, S.-N.; Hwang, G.S.; Kang, K.S.; Eom, D.-W. Protective effect of Artemisia asiatica extract and its active compound eupatilin against cisplatin-induced renal damage. Evid. Based Complement. Alternat. Med. 2015, 2015, 6. [Google Scholar] [CrossRef]
- Kim, K.O.; Lee, D.; Hiep, N.T.; Song, J.H.; Lee, H.J.; Lee, D.; Kang, K.S. Protective effect of phenolic compounds isolated from Mugwort (Artemisia argyi) against contrast-induced apoptosis in kidney epithelium cell line LLC-PK1. Molecules 2019, 24, 195. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Shimada, H.; Matsuda, H.; Yamahara, J.; Murakami, N. Bioactive constituents of Chinese natural medicines. I. New sesquiterpene ketones with vasorelaxant effect from Chinese moxa, the processed leaves of Artemisia argyi Levl. et Vant.: Moxartenone and moxartenolide. Chem. Pharm. Bull. (Tokyo) 1996, 44, 1656–1662. [Google Scholar] [CrossRef]
- Lee, S.H.; Kang, H.M.; Song, H.C.; Lee, H.; Lee, U.C.; Son, K.H.; Kim, S.H.; Kwon, B.M. Sesquiterpene lactones, inhibitors of farnesyl protein transferase, isolated from the flower of Artemisia sylvatica. Tetrahedron 2000, 56, 4711–4715. [Google Scholar] [CrossRef]
- Ando, M.; Wada, T.; Kusaka, H.; Takase, K.; Hirata, N.; Yanagi, Y. Studies on the syntheses of sesquiterpene lactones. 10. Improved syntheses of (+)-tuberiferin and the related. alpha.-methylene-. gamma.-lactones and their biological activities. J. Org. Chem. 1987, 52, 4792–4796. [Google Scholar] [CrossRef]
- Wang, S.; Sun, J.; Zeng, K.W.; Chen, X.G.; Zhou, W.Q.; Zhang, C.; Jin, H.W.; Jiang, Y.; Tu, P.F. Sesquiterpenes from Artemisia argyi: Absolute configurations and biological activities. Eur J. Org. Chem. 2014, 2014, 973–983. [Google Scholar] [CrossRef]
- Ahmed, A.A.; Gati, T.; Hussein, T.A.; Ali, A.T.; Tzakou, O.A.; Couladis, M.A.; Mabry, T.J.; Toth, G. Ligustolide A and B, two novel sesquiterpenes with rare skeletons and three 1,10-seco-guaianolide derivatives from Achillea ligustica. Tetrahedron 2003, 59, 3729–3735. [Google Scholar] [CrossRef]
- Kwon, H.C.; Choi, S.Z.; Lee, W.B.; Min, Y.D.; Yang, M.C.; Chung, A.K.; Lee, K.H.; Lee, K.R. Sesquiterpene lactones of Artemisia sylvatica. J. Pharm. Soc. Korea 2001, 45, 147–152. [Google Scholar]
- Todorova, M.N.; Vogler, B.; Tsankova, E.T. Acrifolide, a novel sesquiterpene lactone from Achillea crithmifolia. Nat. Prod. Lett. 2000, 14, 463–468. [Google Scholar] [CrossRef]
- Makiyi, E.F.; Frade, R.F.; Lebl, T.; Jaffray, E.G.; Cobb, S.E.; Harvey, A.L.; Slawin, A.M.; Hay, R.T.; Westwood, N.J. Iso-seco-tanapartholides: Isolation, synthesis and biological evaluation. Eur. J. Org. Chem. 2009, 2009, 5711–5715. [Google Scholar] [CrossRef] [PubMed]
- Humes, H.D.; Hunt, D.A.; White, M.D. Direct toxic effect of the radiocontrast agent diatrizoate on renal proximal tubule cells. Am. J. Physiol. Renal Physiol. 1987, 252, F246–F255. [Google Scholar] [CrossRef]
- Ward, D.B.; Valentovic, M.A. Contrast induced acute kidney injury and direct cytotoxicity of iodinated radiocontrast media on renal proximal tubule cells. J. Pharmacol. Exp. Ther. 2019, 370, 160–171. [Google Scholar] [CrossRef]
- Xu, R.; Tao, A.; Bai, Y.; Deng, Y.; Chen, G. Effectiveness of N-acetylcysteine for the prevention of contrast-induced nephropathy: A systematic review and meta-analysis of randomized controlled trials. J. Am. Heart Assoc. 2016, 5, e003968. [Google Scholar] [CrossRef]
- Liu, R.; Nair, D.; Ix, J.; Moore, D.H.; Bent, S. N-acetylcysteine for the prevention of contrast-induced nephropathy. A systematic review and meta-analysis. J. Gen. Intern. Med. 2005, 20, 193–200. [Google Scholar] [CrossRef]
- Kang, X.; Hu, D.Y.; Li, C.B.; Ai, Z.S.; Peng, A. N-acetylcysteine for the prevention of contrast-induced nephropathy in patients with pre-existing renal insufficiency or diabetes: A systematic review and meta-analysis. Ren. Fail. 2015, 37, 297–303. [Google Scholar] [CrossRef]
- Nazıroğlu, M.; Yoldaş, N.; Uzgur, E.N.; Kayan, M. Role of contrast media on oxidative stress, Ca 2+ signaling and apoptosis in kidney. J. Membr. Biol. 2013, 246, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Duan, Y.; Zheng, J.; Wang, Y.; Wang, G.; Norgren, S.; Hei, T.K. Nephroprotective effects of N-acetylcysteine amide against contrast-induced nephropathy through upregulating thioredoxin-1, inhibiting ASK1/p38MAPK pathway, and suppressing oxidative stress and apoptosis in rats. Oxid. Med. Cell. Longev. 2016, 2016, 8715185. [Google Scholar] [CrossRef] [PubMed]
- Fishbane, S. N-acetylcysteine in the prevention of contrast-induced nephropathy. Clin J. Am. Soc. Nephrol. 2008, 3, 281–287. [Google Scholar] [CrossRef]
- Dhanapal, A.; Ming, T.W.; Aung, H.P.; Hao, S.J. Preliminary screening of Artemisia argyi for antioxidant potentials. Int. J. Pharmacog. Phytochem. Res. 2016, 8, 347–355. [Google Scholar]
- Bartikova, H.; Hanusova, V.; Skalova, L.; Ambroz, M.; Bousova, I. Antioxidant, pro-oxidant and other biological activities of sesquiterpenes. Curr. Top. Med. Chem. 2014, 14, 2478–2494. [Google Scholar] [CrossRef]
- Chen, X.; Zhong, Z.; Xu, Z.; Chen, L.; Wang, Y. 2′, 7′-Dichlorodihydrofluorescein as a fluorescent probe for reactive oxygen species measurement: Forty years of application and controversy. Free Radic. Res. 2010, 44, 587–604. [Google Scholar] [CrossRef]
- Tebay, L.E.; Robertson, H.; Durant, S.T.; Vitale, S.R.; Penning, T.M.; Dinkova-Kostova, A.T.; Hayes, J.D. Mechanisms of activation of the transcription factor Nrf2 by redox stressors, nutrient cues, and energy status and the pathways through which it attenuates degenerative disease. Free Radic. Biol. Med. 2015, 88, 108–146. [Google Scholar] [CrossRef]
- Naidu, S.; Vijayan, V.; Santoso, S.; Kietzmann, T.; Immenschuh, S. Inhibition and genetic deficiency of p38 MAPK up-regulates heme oxygenase-1 gene expression via Nrf2. J. Immunol. 2009, 182, 7048–7057. [Google Scholar] [CrossRef]
- Tongqiang, L.; Shaopeng, L.; Xiaofang, Y.; Nana, S.; Xialian, X.; Jiachang, H.; Ting, Z.; Xiaoqiang, D. Salvianolic acid B prevents iodinated contrast media-induced acute renal injury in rats via the PI3K/Akt/Nrf2 pathway. Oxid. Med. Cell. Longev. 2016, 2016, 7079487. [Google Scholar] [CrossRef]
- Yano, T.; Itoh, Y.; Sendo, T.; Kubota, T.; Oishi, R. Cyclic AMP reverses radiocontrast media-induced apoptosis in LLC-PK1 cells by activating A kinase/PI3 kinase. Kidney Int. 2003, 64, 2052–2063. [Google Scholar] [CrossRef][Green Version]
- Itoh, Y.; Yano, T.; Sendo, T.; Oishi, R. Clinical and experimental evidence for prevention of acute renal failure induced by radiographic contrast media. J. Pharmacol. Sci. 2005, 7, 473–488. [Google Scholar] [CrossRef] [PubMed]
- Itoh, Y.; Yano, T.; Sendo, T.; Sueyasu, M.; Hirano, K.; Kanaide, H.; Oishi, R. Involvement of de novo ceramide synthesis in radiocontrast-induced renal tubular cell injury. Kidney Int. 2006, 69, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Kolyada, A.Y.; Liangos, O.; Madias, N.E.; Jaber, B.L. Protective effect of erythropoietin against radiocontrast-induced renal tubular epithelial cell injury. Am. J. Nephrol. 2008, 28, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Quintavalle, C.; Brenca, M.; De Micco, F.; Fiore, D.; Romano, S.; Romano, M.; Apone, F.; Bianco, A.; Zabatta, M.; Troncone, G. In vivo and in vitro assessment of pathways involved in contrast media-induced renal cells apoptosis. Cell Death Dis. 2011, 2, e155. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Z.; Chen, J.; Liu, Y.; Liu, T.; Chen, K.; Li, G. Role of ERK1/2 and JNK phosphorylation in iodine contrast agent-induced apoptosis in diabetic rat kidneys. Ren. Fail. 2015, 37, 1349–1355. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, D.; Kim, K.O.; Lee, D.; Kang, K.S. Anti-Apoptotic and Antioxidant Effects of 3-Epi-Iso-Seco-Tanapartholide Isolated from Artemisia argyi against Iodixanol-Induced Kidney Epithelial Cell Death. Biomolecules 2020, 10, 867. https://doi.org/10.3390/biom10060867
Lee D, Kim KO, Lee D, Kang KS. Anti-Apoptotic and Antioxidant Effects of 3-Epi-Iso-Seco-Tanapartholide Isolated from Artemisia argyi against Iodixanol-Induced Kidney Epithelial Cell Death. Biomolecules. 2020; 10(6):867. https://doi.org/10.3390/biom10060867
Chicago/Turabian StyleLee, Dahae, Kem Ok Kim, Dongho Lee, and Ki Sung Kang. 2020. "Anti-Apoptotic and Antioxidant Effects of 3-Epi-Iso-Seco-Tanapartholide Isolated from Artemisia argyi against Iodixanol-Induced Kidney Epithelial Cell Death" Biomolecules 10, no. 6: 867. https://doi.org/10.3390/biom10060867
APA StyleLee, D., Kim, K. O., Lee, D., & Kang, K. S. (2020). Anti-Apoptotic and Antioxidant Effects of 3-Epi-Iso-Seco-Tanapartholide Isolated from Artemisia argyi against Iodixanol-Induced Kidney Epithelial Cell Death. Biomolecules, 10(6), 867. https://doi.org/10.3390/biom10060867