Comparison of Ginsenoside Components of Various Tissues of New Zealand Forest-Grown Asian Ginseng (Panax Ginseng) and American Ginseng (Panax Quinquefolium L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Ginseng Samples
2.2. Chemicals and Reagents
2.3. Preparation of Samples and Reference Standards
2.4. High-Performance Liquid Chromatography Coupled with Quadrupole Time-of-Flight Tandem Mass Spectrometry (HPLC-QTOF-MS)
3. Results and Discussion
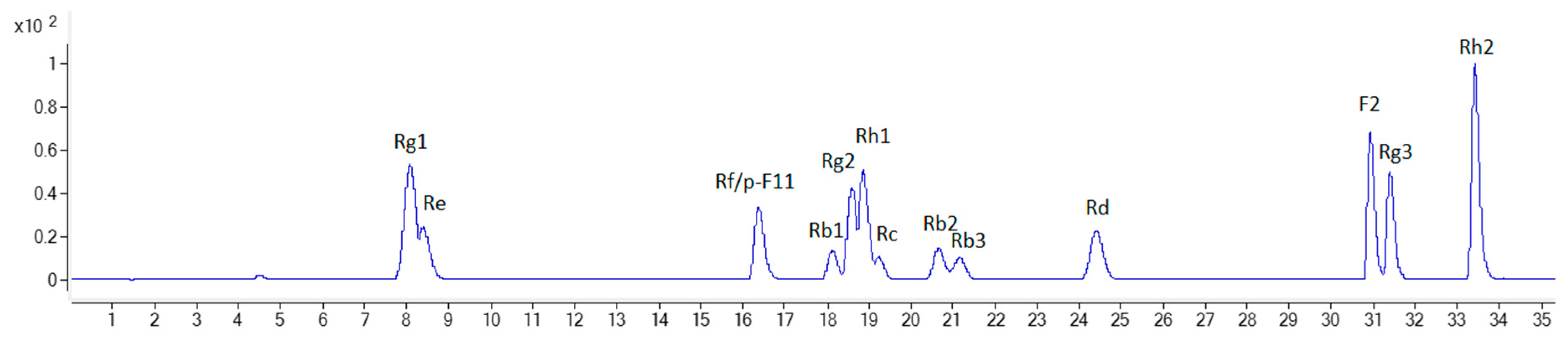
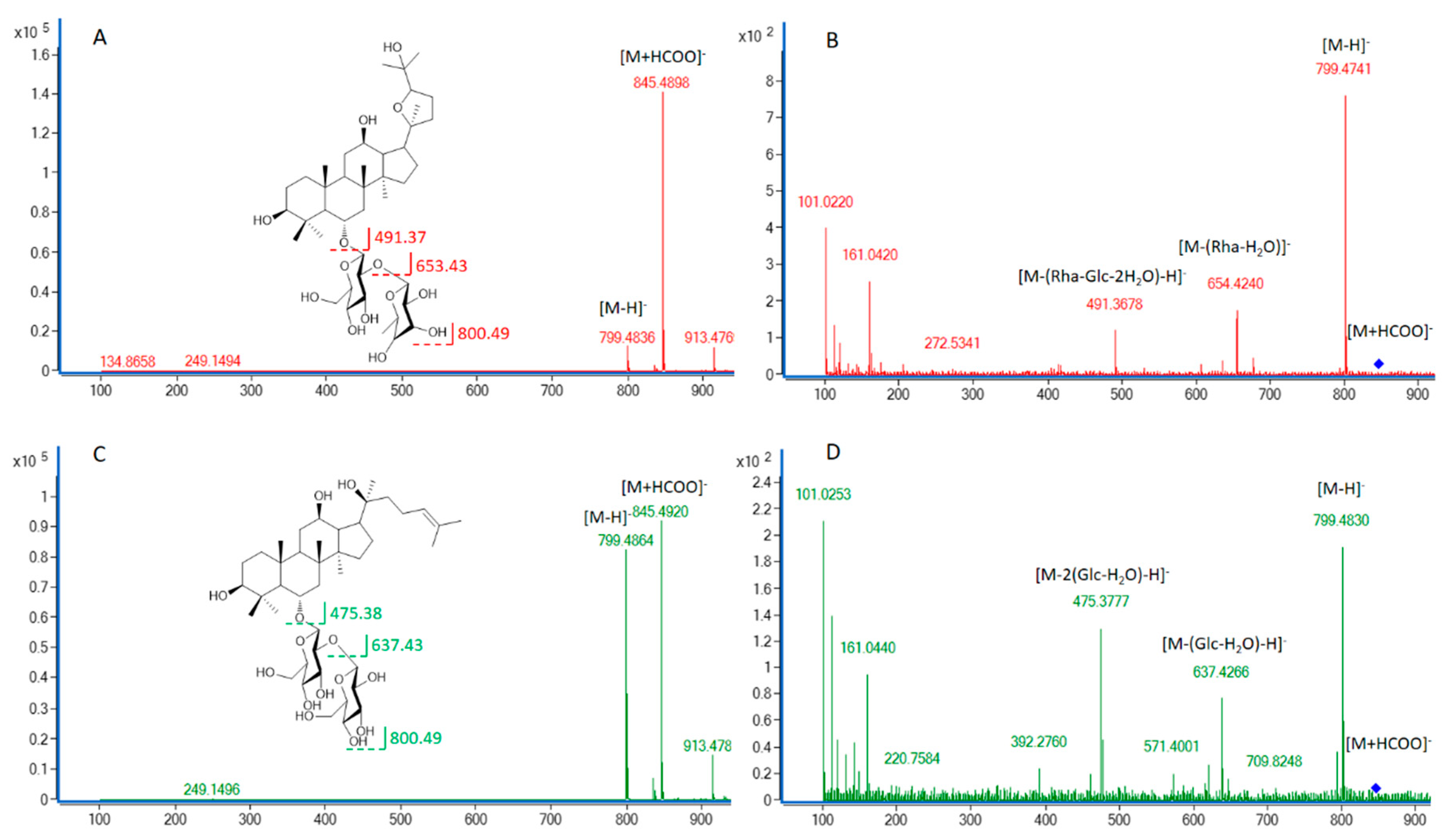
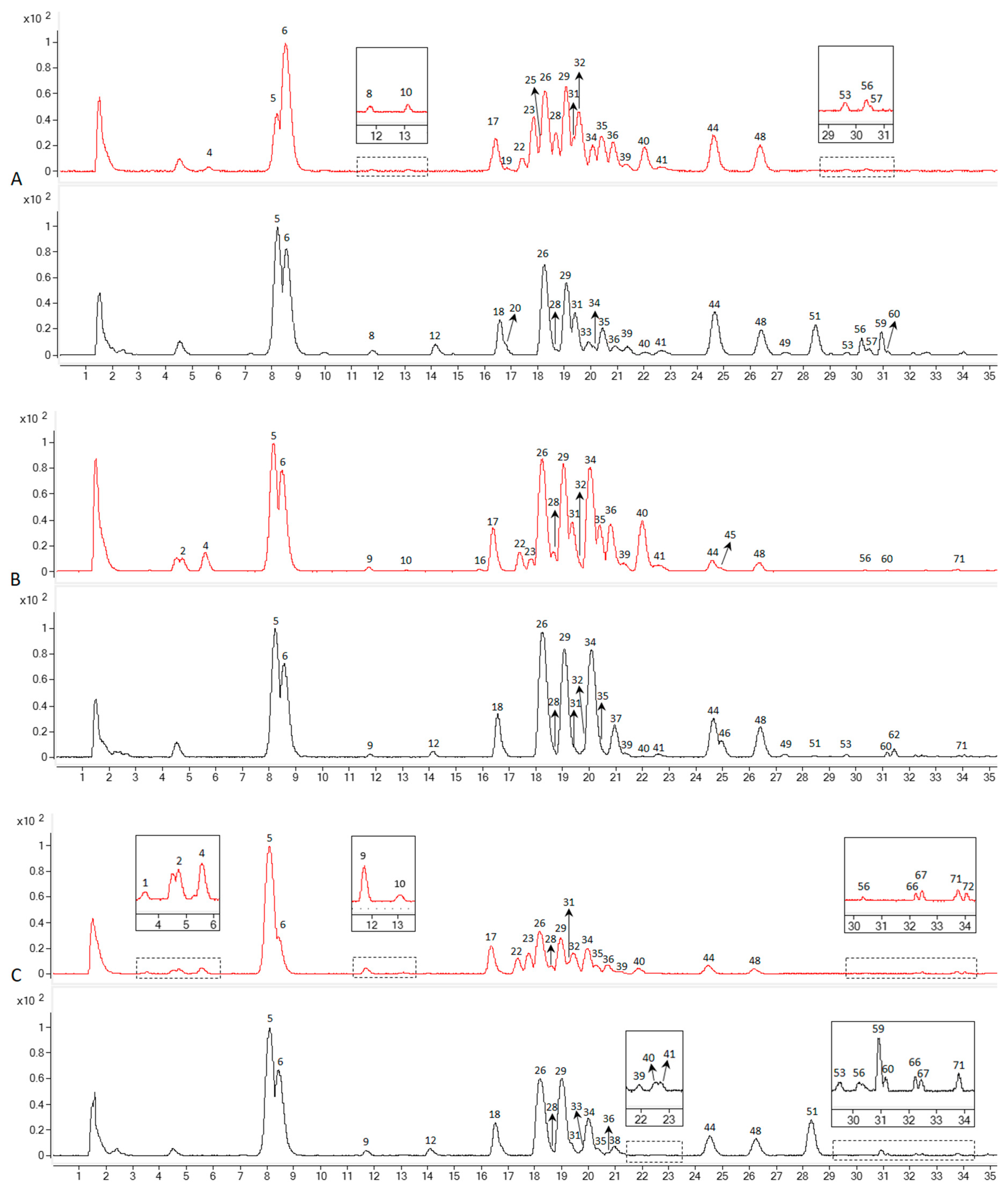
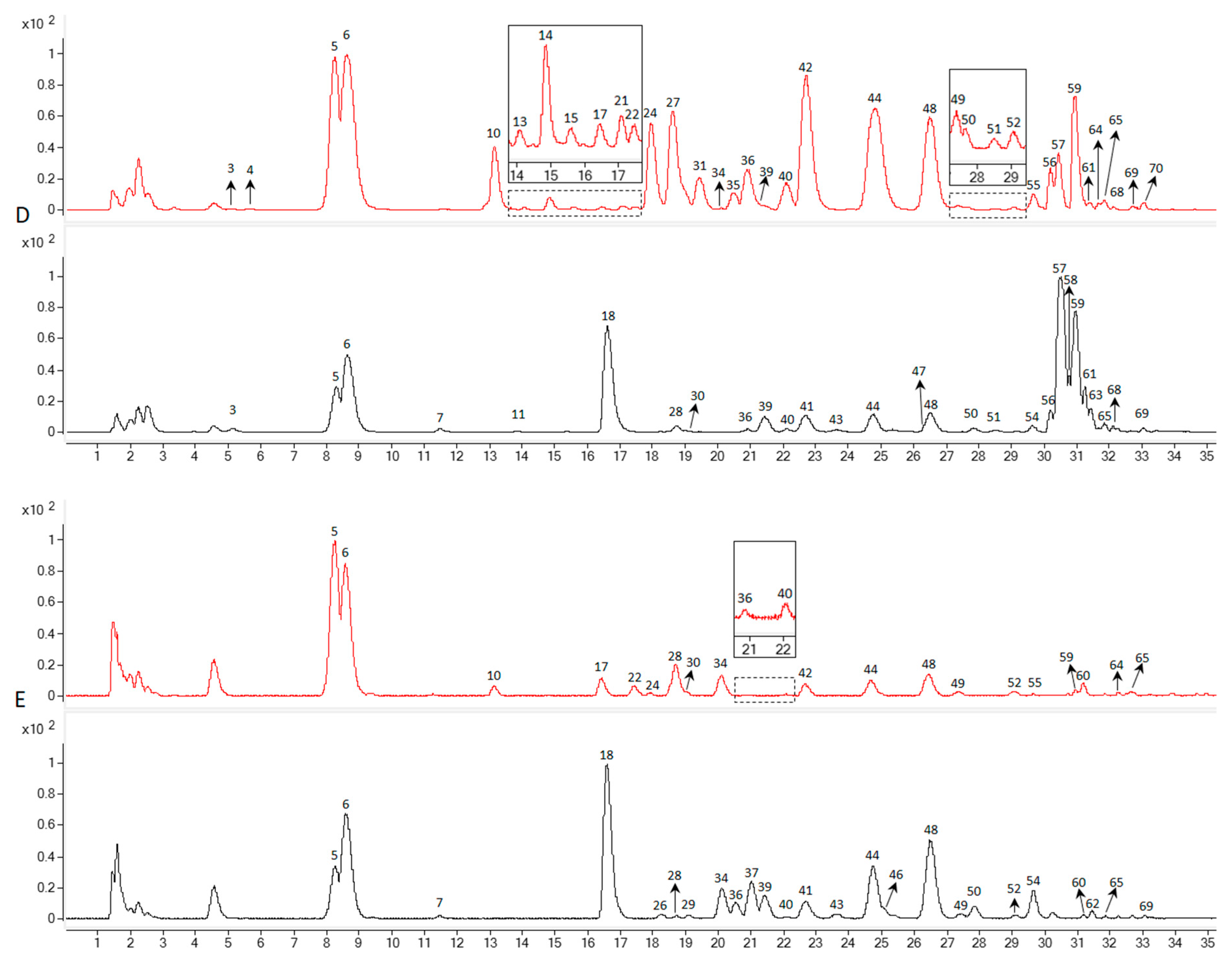
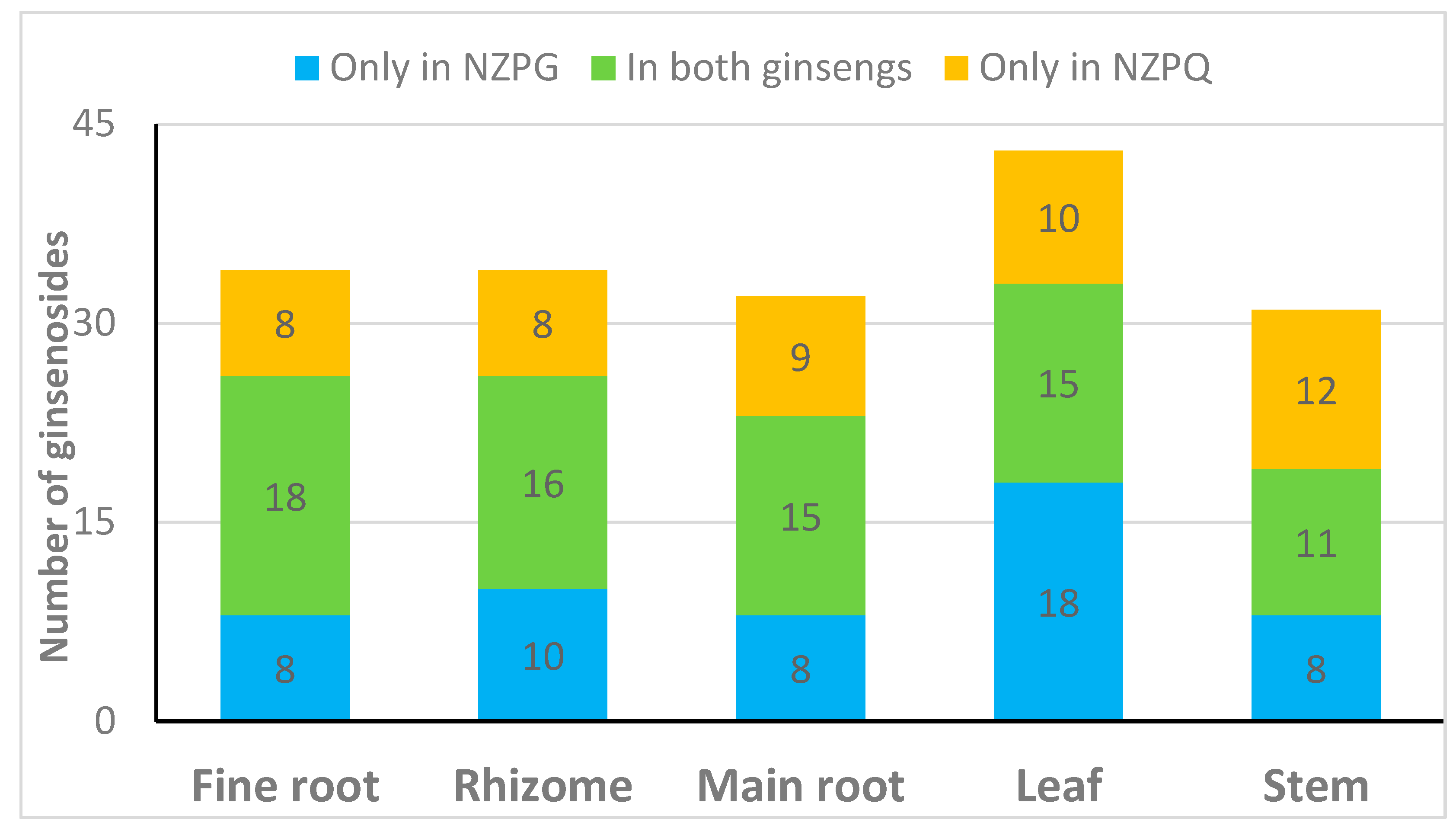
3.1. Identification of the Detected Ginsenosides in Various Parts of NZPG and NZPQ
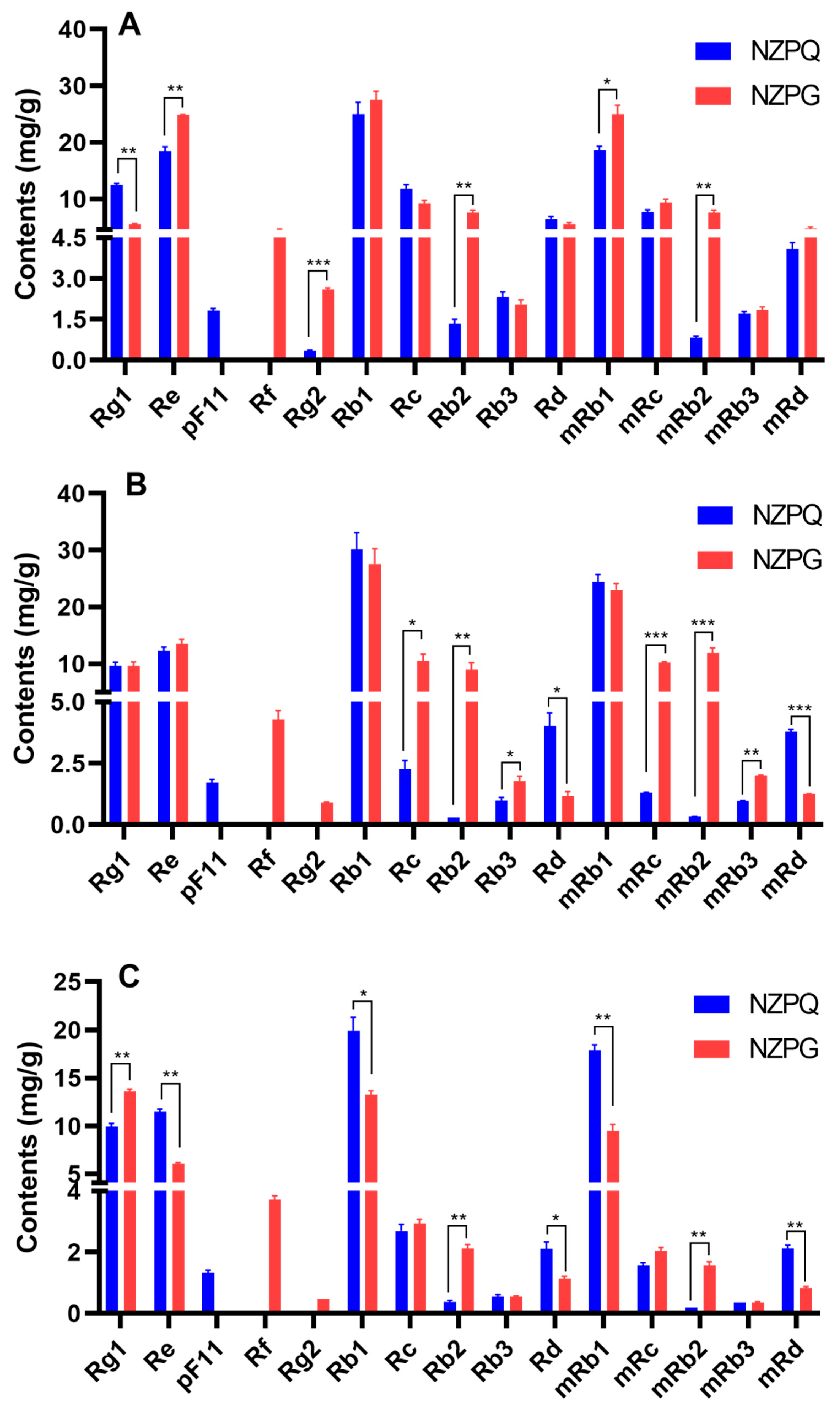
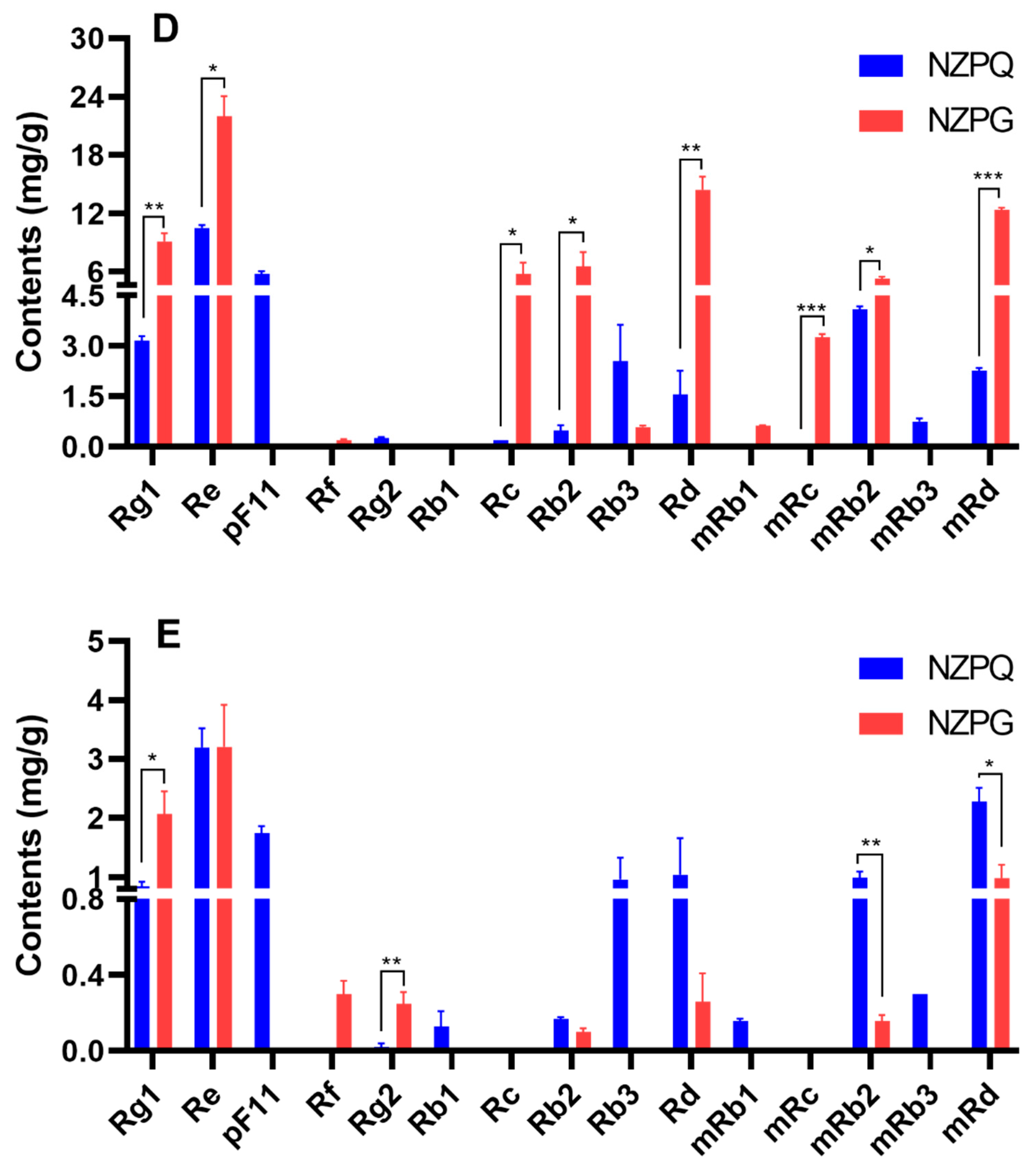
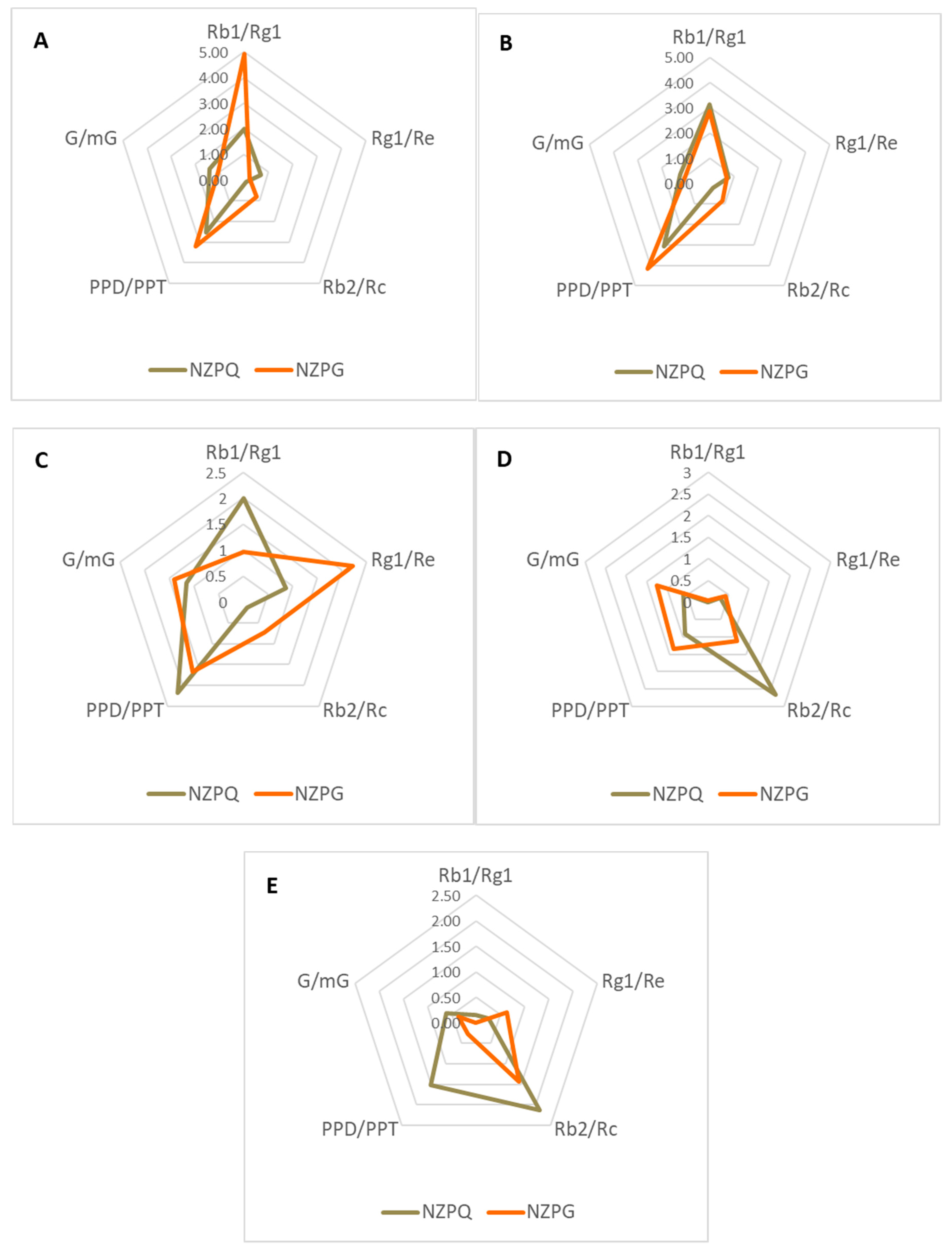
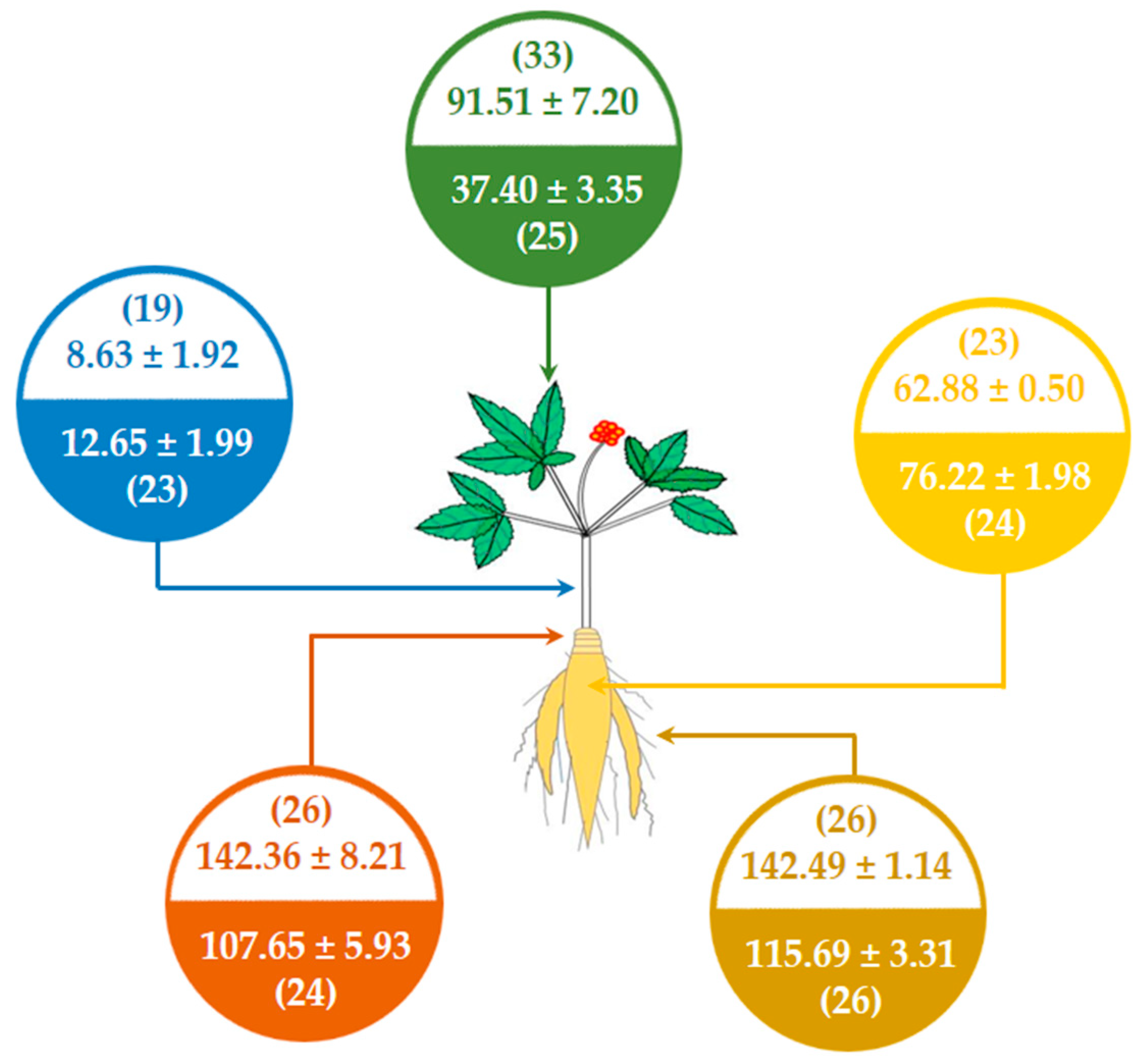
3.2. Quantification of the Main Ginsenosides in Various Parts of NZPG and NZPQ
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Qi, L.W.; Wang, C.Z.; Yuan, C.S. Ginsenosides from American ginseng: Chemical and pharmacological diversity. Phytochemistry 2011, 72, 689–699. [Google Scholar] [CrossRef]
- Angelova, N.; Kong, H.W.; van der Heijden, R.; Yang, S.Y.; Choi, Y.H.; Kim, H.K.; Wang, M.; Hankemeier, T.; van der Greef, J.; Xu, G.; et al. Recent methodology in the phytochemical analysis of ginseng. Phytochem. Anal. 2008, 19, 2–16. [Google Scholar] [CrossRef]
- Han, K.S.; Balan, P.; Hong, H.D.; Choi, W.I.; Cho, C.W.; Lee, Y.C.; Moughan, P.J.; Singh, H. Korean ginseng modulates the ileal microbiota and mucin gene expression in the growing rat. Food Funct. 2014, 5, 1506–1512. [Google Scholar] [CrossRef]
- Chen, C.F.; Chiou, W.F.; Zhang, J.T. Comparison of the pharmacological effects of Panax ginseng and Panax quinquefolium. Acta Pharmacol. Sin. 2008, 29, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Qiao, X.; Li, K.; Fan, J.; Bo, T.; Guo, D.A.; Ye, M. Identification and differentiation of Panax ginseng, Panax quinquefolium, and Panax notoginseng by monitoring multiple diagnostic chemical markers. ActaPharmaceutica Sinica. B 2016, 6, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Balan, P.; Popovich, D.G. Chapter 6—Comparison of the ginsenoside composition of Asian ginseng (Panax ginseng) and American ginseng (Panax quinquefolius L.) and their transformation pathways. In Studies in Natural Products Chemistry; Atta ur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 63, pp. 161–195. [Google Scholar]
- Leung, K.S.-Y.; Chan, K.; Bensoussan, A.; Munroe, M.J. Application of atmospheric pressure chemical ionisation mass spectrometry in the identification and differentiation ofPanax Species. Phytochem. Anal. 2007, 18, 146–150. [Google Scholar] [CrossRef]
- Li, L.; Luo, G.A.; Liang, Q.L.; Hu, P.; Wang, Y.M. Rapid qualitative and quantitative analyses of Asian ginseng in adulterated American ginseng preparations by UPLC/Q-TOF-MS. J. Pharm. Biomed. Anal. 2010, 52, 66–72. [Google Scholar] [CrossRef]
- Wu, R.; Chen, X.; Wu, W.J.; Wang, Z.; Wong, Y.E.; Hung, Y.W.; Wong, H.T.; Yang, M.; Zhang, F.; Chan, T.D. Rapid Differentiation of Asian and American Ginseng by Differential Ion Mobility Spectrometry-Tandem Mass Spectrometry Using Stepwise Modulation of Gas Modifier Concentration. J. Am. Soc. Mass. Spectrom. 2019, 31, 2212–2221. [Google Scholar] [CrossRef]
- Cui, S.; Wu, J.; Wang, J.; Wang, X. Discrimination of American ginseng and Asian ginseng using electronic nose and gas chromatography-mass spectrometry coupled with chemometrics. J. Ginseng Res. 2017, 41, 85–95. [Google Scholar] [CrossRef]
- Chen, Y.; Zhao, Z.; Chen, H.; Yi, T.; Qin, M.; Liang, Z. Chemical differentiation and quality evaluation of commercial Asian and American ginsengs based on a UHPLC-QTOF/MS/MS metabolomics approach. Phytochem. Anal. 2015, 26, 145–160. [Google Scholar] [CrossRef]
- Smallfield, B.M.; Follett, J.M.; Douglas, M.H.; Douglas, J.A.; Parmenter, G.A. Production of Panax SPP. in New Zealand. Acta Hortic. 1995, 390, 83–91. [Google Scholar] [CrossRef]
- Chen, W.; Balan, P.; Popovich, D.G. Analysis of Ginsenoside Content (Panax ginseng) from Different Regions. Molecules 2019, 24, 3491. [Google Scholar] [CrossRef]
- Chen, W.; Balan, P.; Popovich, D.G. Ginsenosides analysis of New Zealand–grown forest Panax ginseng by LC-QTOF-MS/MS. J. Ginseng Res. 2019, in press. [Google Scholar] [CrossRef]
- Wang, H.P.; Yang, X.B.; Yang, X.W.; Liu, J.X.; Xu, W.; Zhang, Y.B.; Zhang, L.X.; Wang, Y.P. Ginsenjilinol, a new protopanaxatriol-type saponin with inhibitory activity on LPS-activated NO production in macrophage RAW 264.7 cells from the roots and rhizomes of Panax ginseng. J. Asian Nat. Prod. Res. 2013, 15, 579–587. [Google Scholar] [CrossRef]
- Wang, H.P.; Zhang, Y.B.; Yang, X.W.; Zhao, D.Q.; Wang, Y.P. Rapid characterization of ginsenosides in the roots and rhizomes of Panax ginseng by UPLC-DAD-QTOF-MS/MS and simultaneous determination of 19 ginsenosides by HPLC-ESI-MS. J. Ginseng Res. 2016, 40, 382–394. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Choi, B.R.; Kim, Y.C.; Choi, D.J.; Lee, Y.S.; Kim, G.S.; Baek, N.I.; Kim, S.Y.; Lee, D.Y. Comprehensive Profiling and Quantification of Ginsenosides in the Root, Stem, Leaf, and Berry of Panax ginseng by UPLC-QTOF/MS. Molecules 2017, 22, 2147. [Google Scholar] [CrossRef]
- Li, X.; Yao, F.; Fan, H.; Li, K.; Sun, L.; Liu, Y. Intraconversion of Polar Ginsenosides, Their Transformation into Less-Polar Ginsenosides, and Ginsenoside Acetylation in Ginseng Flowers upon Baking and Steaming. Molecules 2018, 23, 759. [Google Scholar] [CrossRef]
- Zhang, Y.; Pi, Z.; Liu, C.; Song, F.; Liu, Z.; Liu, S. Analysis of Low-polar Ginsenosides in Steamed Panax Ginseng at High-temperature by HPLC-ESI-MS/MS. Chem. Res. Chin. Univ. 2012, 28, 31–36. [Google Scholar]
- Cho, E.J.; Lee, D.J.; Wee, C.D.; Kim, H.L.; Cheong, Y.H.; Cho, J.S.; Sohn, B.K. Effects of AMF inoculation on growth of Panax ginseng C.A. Meyer seedlings and on soil structures in mycorrhizosphere. Sci. Hortic. 2009, 122, 633–637. [Google Scholar] [CrossRef]
- Khan Chowdhury, E.; Jeon, J.; Ok Rim, S.; Park, Y.H.; Kyu Lee, S.; Bae, H. Composition, diversity and bioactivity of culturable bacterial endophytes in mountain-cultivated ginseng in Korea. Sci. Rep. 2017, 7, 10098. [Google Scholar] [CrossRef]
- Szakiel, A.; Pączkowski, C.; Henry, M. Influence of environmental abiotic factors on the content of saponins in plants. Phytochem. Rev. 2011, 10, 471–491. [Google Scholar] [CrossRef]

| No. | R.t | Measured Value [ion form] | Formula | Source | Identification | Ref. |
|---|---|---|---|---|---|---|
| 1 | 3.50 | 815.4775 [M - H]; 861.4822 [M + HCOO] | C42H72O15 | PG (m) | Ginsenjilinol | [15] |
| 2 | 4.71 | 961.5291 [M - H]; 1007.5387 [M + HCOO] | C48H82O19 | PG (m, r) | 20glc-Rf | [11] |
| 3 | 5.09 | 931.5215 [M - H]; 977.5257 [M + HCOO] | C47H80O18 | PG (l); PQ (l) | NG-R1 | [11] |
| 4 | 5.62 | 931.5162 [M - H]; 977.5212 [M + HCOO] | C47H80O18 | PG (f, r, m, l) | Re4 | [16] |
| 5 | 8.19 | 799.4857 [M - H]; 845.4898 [M + HCOO] | C42H72O14 | PG (f, r, m, s, l); PQ (f, r, m, s, l) | Rg1 | std |
| 6 | 8.55 | 945.5370 [M - H]; 991.5439 [M + HCOO] | C48H82O18 | PG (f, r, m, s, l); PQ (f, r, m, s, l) | Re | std |
| 7 | 11.49 | 799.4835 [M - H]; 845.4920 [M + HCOO] | C42H72O14 | PQ (s, l) | Rg1 isomer | [16] |
| 8 | 11.53 | 841.4871 [M - H] | C44H74O15 | PG (f); PQ (f) | Ac-Rg1 | [16] |
| 9 | 11.70 | 885.4780 [M - H] | C45H74O17 | PG (m, r); PQ (m, r) | m-Rg1 | [11] |
| 10 | 13.17 | 1031.5386 [M - H] | C51H84O21 | PG (f, r, m, s, l) | m-Re | [11] |
| 11 | 13.83 | 799.4821 [M - H]; 845.4770 [M + HCOO] | C42H72O14 | PQ (l) | Rg1 isomer | [16] |
| 12 | 14.14 | 887.4954 [M + HCOO] | C44H74O15 | PQ (f, m, r) | Yesanchinoside D | [16] |
| 13 | 14.12 | 815.4697 [M - H] | C42H72O15 | PG (l) | Re5 | [15] |
| 14 | 14.81 | 961.5322 [M - H]; 1007.5338 [M + HCOO] | C48H82O19 | PG (l) | VG-R4 | [17] |
| 15 | 15.55 | 829.4839 [M + HCOO] | C42H72O13 | PG (l) | G-La | [18] |
| 16 | 15.88 | 1117.5382 [M - H] | C54H86O24 | PG (r) | mf-Rd6/isomer | [6] |
| 17 | 16.40 | 799.4807 [M - H]; 845.4855 [M + HCOO] | C42H72O14 | PG (f, r, m, s, l) | Rf | std |
| 18 | 16.55 | 799.4772 [M - H]; 845.4862 [M + HCOO] | C42H72O14 | PQ (f, r, m, s, l) | p-F11 | std |
| 19 | 16.84 | 1325.6255 [M - H] | C62H102O30 | PG (f) | m-Ra3/m-NG-R4 | [6] |
| 20 | 16.84 | 653.3698 [M - H]; 699.4271 [M + HCOO] | C36H62O10 | PQ (f) | G-Ki/G-Km/G-ST2 | [6] |
| 21 | 17.09 | 799.4497 [M - H]; 845.4817 [M + HCOO] | C42H72O14 | PG (l) | Rf isomer | [16] |
| 22 | 17.40 | 769.4681 [M - H]; 815.4726 [M + HCOO] | C41H70O13 | PG (f, r, m, s, l) | NG-R2 | [11] |
| 23 | 17.82 | 1209.6224 [M - H] | C58H98O26 | PG (f, r, m) | Ra1 | [11] |
| 24 | 17.90 | 769.4739 [M - H]; 815.4754 [M + HCOO] | C41H70O13 | PG (s, l) | G-F5 | [17] |
| 25 | 18.02 | 1239.6273 [M - H] | C59H100O27 | PG (f) | Ra3/NG-R4 | [17] |
| 26 | 18.22 | 1107.5929 [M - H] | C54H92O23 | PG (f, r, m); PQ (f, r, m, s) | Rb1 | std |
| 27 | 18.61 | 769.4658 [M - H]; 815.4750 [M + HCOO] | C41H70O13 | PG (l) | G-F3 | [17] |
| 28 | 18.66 | 783.4858 [M - H]; 829.4930 [M + HCOO] | C42H72O13 | PG (f, r, m, s); PQ (f, m, s, l) | Rg2 | std |
| 29 | 19.04 | 1193.5932 [M - H] | C57H94O26 | PG (f, r, m); PQ (f, r, m, s) | m-Rb1 | [11] |
| 30 | 19.05 | 683.4329 [M + HCOO] | C36H62O9 | PG (s); PQ (l) | Rh1 | std |
| 31 | 19.31 | 1077.5818 [M - H] | C53H90O22 | PG (f, r, m, l); PQ (f, r, m) | Rc | std |
| 32 | 19.49 | 1209.6254 [M - H] | C58H98O26 | PG (f, r, m) | Ra2 | [11] |
| 33 | 19.75 | 1193.5849 [M - H] | C57H94O26 | PQ (f, m) | m-Rb1 isomer | [16] |
| 34 | 20.01 | 955.4865 [M - H] | C48H76O19 | PG (f, r, m, s, l); PQ (f, r, m, s) | Ro | [11] |
| 35 | 20.38 | 1163.5798 [M - H] | C56H92O25 | PG (f, r, m, l); PQ (f, r, m) | m-Rc | [16] |
| 36 | 20.80 | 1077.5813 [M - H] | C53H90O22 | PG (f, r, m, s, l); PQ (f, m, s, l) | Rb2 | std |
| 37 | 20.98 | 955.4853 [M - H] | C48H76O19 | PQ (r, s) | Ro isomer | [16] |
| 38 | 20.95 | 1193.5892 [M - H] | C57H94O26 | PQ (m) | m-Rb1 isomer | [16] |
| 39 | 21.35 | 1077.5782 [M - H] | C53H90O22 | PG (f, r, m, l); PQ (f, r, m, s, l) | Rb3 | std |
| 40 | 21.95 | 1163.5767 [M - H] | C56H92O25 | PG (f, r, m, s, l); PQ (f, r, m, s, l) | m-Rb2 | [11] |
| 41 | 22.59 | 1163.5782 [M - H] | C56H92O25 | PG (f, r, m); PQ (f, r, m, s, l) | m-Rb3 | [11] |
| 42 | 22.69 | 637.4292 [M - H]; 683.4310 [M + HCOO] | C36H62O9 | PG (s, l) | F1 | [17] |
| 43 | 23.56 | 1163.5822 [M - H] | C56H92O25 | PQ (s, l) | m-Rb3 isomer | [16] |
| 44 | 24.65 | 945.5364 [M - H]; 991.5469 [M + HCOO] | C48H82O18 | PG (f, r, m, s, l); PQ (f, r, m, s, l) | Rd | std |
| 45 | 24.59 | 1119.5871 [M - H] | C55H92O23 | PG (r) | Rs1 | [19] |
| 46 | 24.99 | 793.4306 [M - H] | C42H66O14 | PQ (r, s) | Zingibroside R1 | [11] |
| 47 | 25.84 | 1119.5844 [M - H] | C55H92O23 | PQ (l) | Rs2 | [19] |
| 48 | 26.35 | 1031.5348 [M - H] | C51H84O21 | PG (f, r, m, s, l); PQ (f, r, m, s, l) | m-Rd | [11] |
| 49 | 27.36 | 987.5413 [M - H] | C50H84O19 | PG (s, l); PQ (f, r, s) | Ac-Rd | [18] |
| 50 | 27.76 | 1031.5348 [M - H] | C51H84O21 | PG (l); PQ (s, l) | m-Rd isomer | [16] |
| 51 | 28.41 | 945.5354 [M - H]; 991.5435 [M + HCOO] | C48H82O18 | PG (l); PQ (f, r, m, l) | GyXVII | [17] |
| 52 | 29.12 | 1117.5369 [M - H] | C54H86O24 | PG (s, l); PQ (s) | mf-Rd6 isomer | [6] |
| 53 | 29.56 | 987.5449 [M - H] | C50H84O19 | PG (f); PQ (f, r, m) | Ac-Rd isomer | [16] |
| 54 | 29.63 | 1117.5314 [M - H] | C54H86O24 | PQ (s, l) | mf-Rd6 isomer | [6] |
| 55 | 29.67 | 987.5436 [M - H] | C50H84O19 | PG (s, l) | Ac-Rd isomer | [16] |
| 56 | 30.10 | 915.5285 [M - H]; 961.5338 [M + HCOO] | C47H80O17 | PG (f, r, m, l); PQ (f, m, l) | NG-Fe | [17] |
| 57 | 30.42 | 915.5226 [M - H]; 961.5279 [M + HCOO] | C47H80O17 | PG (f, l); PQ (f, l) | VG-R16 | [16] |
| 58 | 30.75 | 1001.5258 [M + HCOO] | C48H76O19 | PQ (l) | Ro isomer | [16] |
| 59 | 30.91 | 829.4906 [M + HCOO] | C42H72O13 | PG (s, l); PQ (f, m, l) | F2 | std |
| 60 | 31.15 | 793.4334 [M - H] | C42H66O14 | PG (r, s); PQ (f, m, r, s) | Chikusetsusaponin IVa | [11] |
| 61 | 31.4 | 783.4806 [M - H]; 829.4874 [M + HCOO] | C42H72O13 | PG (l); PQ (l) | Rg3 | std |
| 62 | 31.43 | 793.4301 [M - H] | C42H66O14 | PQ (r, s) | Chikusetsusaponin IVa isomer | [16] |
| 63 | 31.43 | 825.4879 [M - H]; 871.4974 [M + HCOO] | C52H72O11 | PQ (l) | Unknown | - |
| 64 | 31.65 | 675.3529 [M - H]; 721.3579 [M + HCOO] | C40H54O9 | PG (s, l) | Unknown | - |
| 65 | 31.83 | 675.3505 [M - H]; 721.3589 [M + HCOO] | C40H54O9 | PG (s, l); PQ (s, l) | Unknown | - |
| 66 | 32.23 | 677.3731 [M - H]; 723.3730 [M + HCOO] | C40H54O9 | PG (m); PQ (m) | Unknown | - |
| 67 | 32.46 | 677.3743 [M - H]; 723.3746 [M + HCOO] | C40H54O9 | PG (m); PQ (m) | Unknown | - |
| 68 | 32.69 | 653.3673 [M - H]; 699.3744 [M + HCOO] | C36H62O10 | PG (l); PQ (l) | G-Ki/G-Km/G-ST2 | [6] |
| 69 | 33.05 | 513.2942 [M - H]; 559.3044 [M + HCOO] | C27H46O9 | PG (l); PQ (s, l) | Unknown | - |
| 70 | 33.42 | 667.4373 [M + HCOO] | C36H62O8 | PG (l) | Rh2 | std |
| 71 | 33.81 | 595.2873 [M + HCOO] | C33H42O7 | PG (r, m, s); PQ (r, m) | Unknown | - |
| 72 | 34.05 | 515.9695 [M - H]; 561.3247 [M + HCOO] | C27H48O9 | PG (m) | Unknown | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.; Balan, P.; Popovich, D.G. Comparison of Ginsenoside Components of Various Tissues of New Zealand Forest-Grown Asian Ginseng (Panax Ginseng) and American Ginseng (Panax Quinquefolium L.). Biomolecules 2020, 10, 372. https://doi.org/10.3390/biom10030372
Chen W, Balan P, Popovich DG. Comparison of Ginsenoside Components of Various Tissues of New Zealand Forest-Grown Asian Ginseng (Panax Ginseng) and American Ginseng (Panax Quinquefolium L.). Biomolecules. 2020; 10(3):372. https://doi.org/10.3390/biom10030372
Chicago/Turabian StyleChen, Wei, Prabhu Balan, and David G Popovich. 2020. "Comparison of Ginsenoside Components of Various Tissues of New Zealand Forest-Grown Asian Ginseng (Panax Ginseng) and American Ginseng (Panax Quinquefolium L.)" Biomolecules 10, no. 3: 372. https://doi.org/10.3390/biom10030372
APA StyleChen, W., Balan, P., & Popovich, D. G. (2020). Comparison of Ginsenoside Components of Various Tissues of New Zealand Forest-Grown Asian Ginseng (Panax Ginseng) and American Ginseng (Panax Quinquefolium L.). Biomolecules, 10(3), 372. https://doi.org/10.3390/biom10030372







