Macromolecular Crowding Increases the Affinity of the PHD of ING4 for the Histone H3K4me3 Mark
Abstract
1. Introduction
2. Materials and Methods
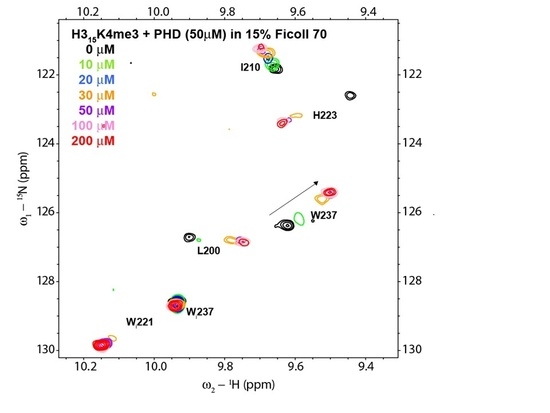
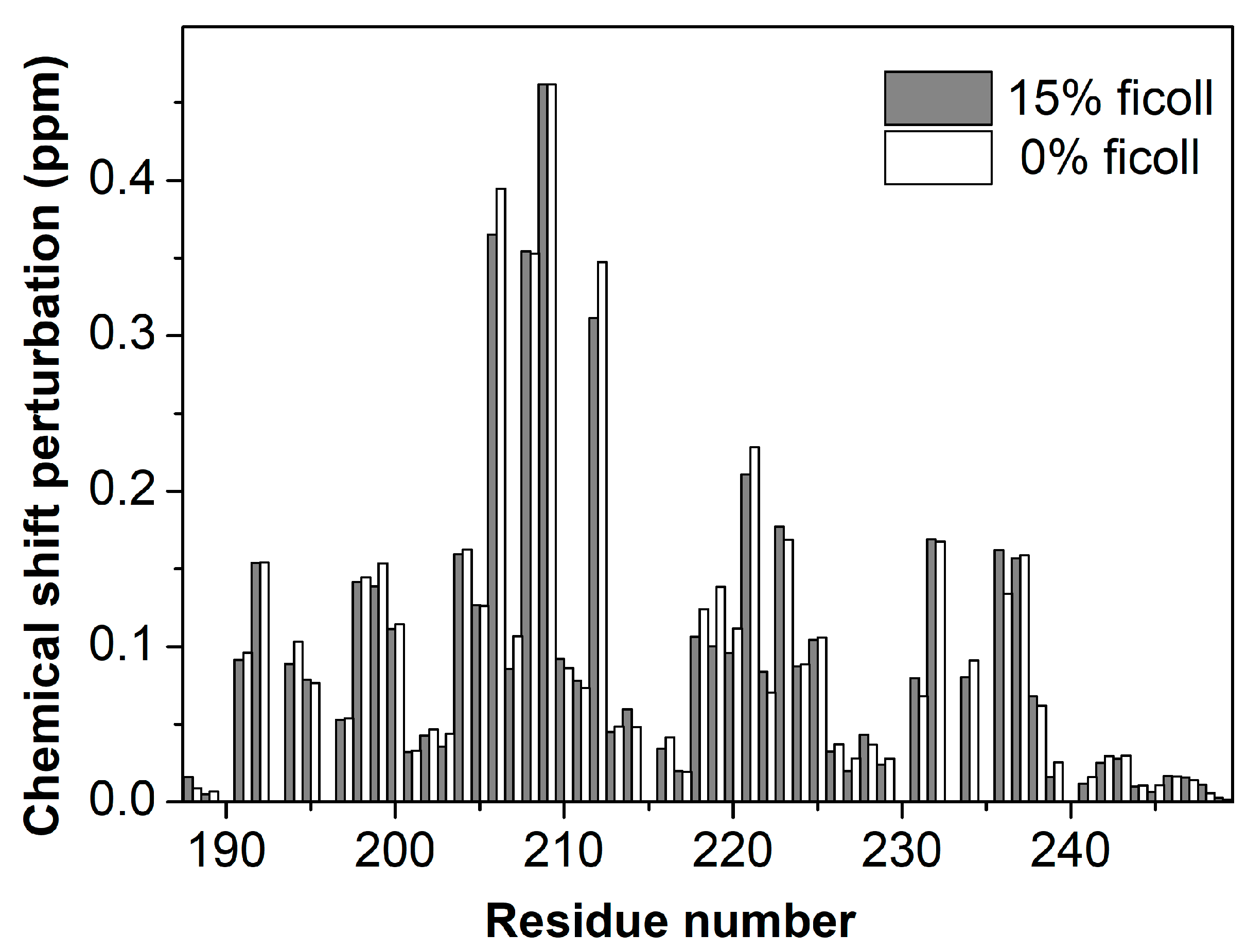
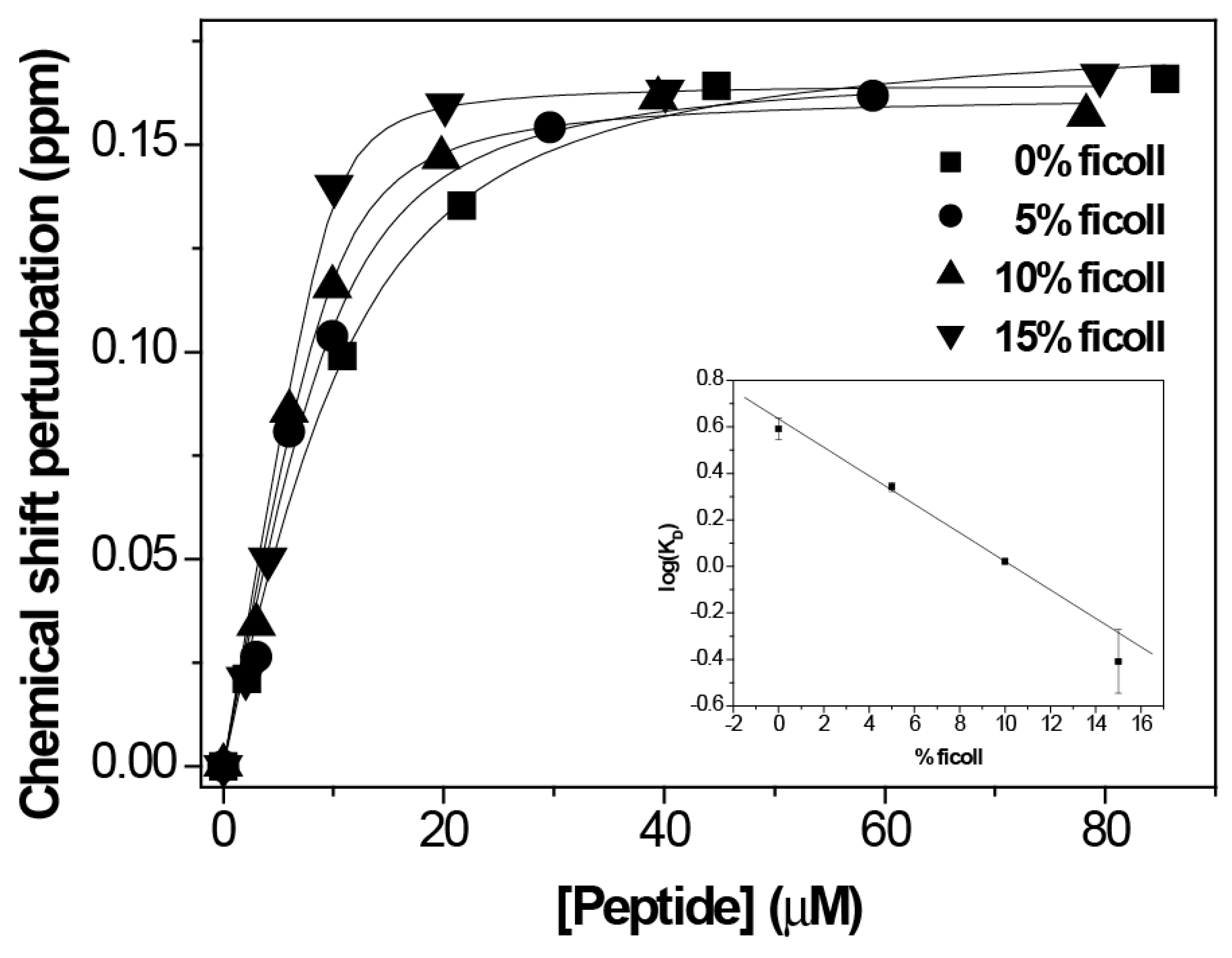
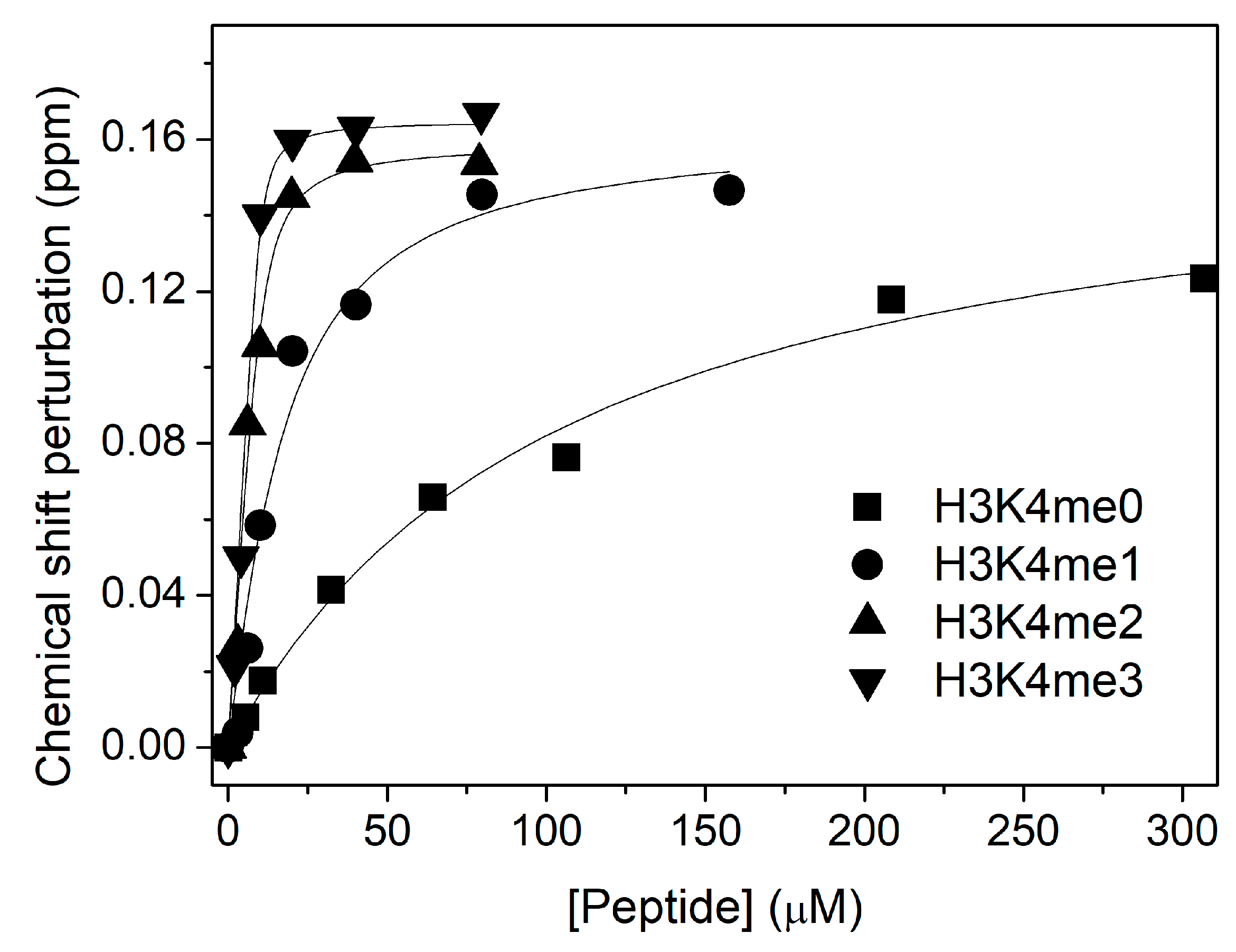
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- de la Cruz, X.; Lois, S.; Sanchez-Molina, S.; Martinez-Balbas, M.A. Do protein motifs read the histone code? Bioessays 2005, 27, 164–175. [Google Scholar] [CrossRef]
- Santos-Rosa, H.; Caldas, C. Chromatin modifier enzymes, the histone code and cancer. Eur. J. Cancer Prev. 2005, 41, 2381–2402. [Google Scholar] [CrossRef] [PubMed]
- Jenuwein, T.; Allis, C.D. Translating the histone code. Science 2001, 293, 1074–1080. [Google Scholar] [CrossRef] [PubMed]
- Nightingale, K.P.; O’Neill, L.P.; Turner, B.M. Histone modifications: Signalling receptors and potential elements of a heritable epigenetic code. Curr. Opin. Genet. Dev. 2006, 16, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Lall, S. Primers on chromatin. Nat. Struct. Mol. Biol. 2007, 14, 1110–1115. [Google Scholar] [CrossRef] [PubMed]
- Taverna, S.D.; Li, H.; Ruthenburg, A.J.; Allis, C.D.; Patel, D.J. How chromatin-binding modules interpret histone modifications: lessons from professional pocket pickers. Nat. Struct. Mol. Biol. 2007, 14, 1025–1040. [Google Scholar] [CrossRef] [PubMed]
- Dantas, A.; Al Shueili, B.; Yang, Y.; Nabbi, A.; Fink, D.; Riabowol, K. Biological Functions of the ING Proteins. Cancers 2019, 11, 1817. [Google Scholar] [CrossRef] [PubMed]
- Tallen, G.; Riabowol, K. Keep-ING balance: tumor suppression by epigenetic regulation. FEBS Lett. 2014, 588, 2728–2742. [Google Scholar] [CrossRef]
- Peña, P.V.; Davrazou, F.; Shi, X.; Walter, K.L.; Verkhusha, V.V.; Gozani, O.; Zhao, R.; Kutateladze, T.G. Molecular mechanism of histone H3K4me3 recognition by plant homeodomain of ING2. Nature 2006, 442, 100–103. [Google Scholar] [CrossRef]
- Palacios, A.; Palacios, A.; Moreno, A.; Oliveira, B.L.; Rivera, T.; Prieto, J.; Garcia, P.; Fernandez-Fernandez, M.R.; Bernado, P.; Palmero, I.; et al. The dimeric structure and the bivalent recognition of H3K4me3 by the tumor suppressor ING4 suggests a mechanism for enhanced targeting of the HBO1 complex to chromatin. J. Mol. Biol. 2010, 396, 1117–1127. [Google Scholar] [CrossRef] [PubMed]
- Culurgioni, S.; Munoz, I.G.; Moreno, A.; Palacios, A.; Villate, M.; Palmero, I.; Montoya, G.; Blanco, F.J. The crystal structure of the inhibitor of growth 4 (ING4) dimerization domain reveals the functional organization of the ING family of chromatin binding proteins. J. Biol. Chem. 2012, 287, 10876–10884. [Google Scholar] [CrossRef] [PubMed]
- Ormaza, G.; Medagli, B.; Ibanez de Opakua, A.; Rodriguez, J.A.; Merino, N.; Villate, M.; Onesti, S.; Blanco, F.J. The tumor suppressor inhibitor of growth 4 binds double-stranded DNA through its disordered central region. FEBS Lett. 2017, 591, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Palacios, A.; Garcia, P.; Padro, D.; Lopez-Hernandez, E.; Martin, I.; Blanco, F.J. Solution structure and NMR characterization of the binding to methylated histone tails of the plant homeodomain finger of the tumour suppressor ING4. FEBS Lett. 2006, 205 580, 6903–6908. [Google Scholar] [CrossRef]
- Palacios, A.; Munoz, I.G.; Pantoja-Uceda, D.; Marcaida, M.J.; Torres, D.; Martin-Garcia, J.M.; Luque, I.; Montoya, G.; Blanco, F.J. Molecular basis of histone H3K4me3 recognition by ING4. J. Biol. Chem. 2008, 283, 15956–15964. [Google Scholar] [CrossRef] [PubMed]
- Minton, A.P. Implications of macromolecular crowding for protein assembly. Curr. Opin. Struct. Biol. 2000, 10, 34–39. [Google Scholar] [CrossRef]
- Rivas, G.; Minton, A.P. Macromolecular Crowding In Vitro, In Vivo, and In Between. Trends Biochem. Sci. 2016, 41, 970–981. [Google Scholar] [CrossRef]
- Luby-Phelps, K.; Castle, P.E.; Taylor, D.L.; Lanni, F. Hindered diffusion of inert tracer particles in the cytoplasm of mouse 3T3 cells. Proc. Natl. Acad. Sci. USA 1987, 84, 4910–4913. [Google Scholar] [CrossRef]
- Monterroso, B.; Minton, A.P. Effect of high concentration of inert cosolutes on the refolding of an enzyme: carbonic anhydrase B in sucrose and ficoll 70. J. Biol. Chem. 2007, 282, 33452–33458. [Google Scholar] [CrossRef]
- Venturoli, D.; Rippe, B. Ficoll and dextran vs. globular proteins as probes for testing glomerular permselectivity: effects of molecular size, shape, charge, and deformability. Am. J. Physiol. Renal Physiol. 2005, 288, F605–F613. [Google Scholar] [CrossRef]
- Ormaza, G.; Rodriguez, J.A.; Ibanez de Opakua, A.; Merino, N.; Villate, M.; Gorrono, I.; Rabano, M.; Palmero, I.; Vilaseca, M.; Kypta, R.; et al. The Tumor Suppressor ING5 Is a Dimeric, Bivalent Recognition Molecule of the Histone H3K4me3 Mark. J. Mol. Biol. 2019, 431, 2298–2319. [Google Scholar] [CrossRef]
- Rivas, G.; Minton, A.P. Toward an understanding of biochemical equilibria within living cells. Biophys. Rev. 2018, 10, 241–253. [Google Scholar] [CrossRef] [PubMed]
- You, X.; Nguyen, A.W.; Jabaiah, A.; Sheff, M.A.; Thorn, K.S.; Daugherty, P.S. Intracellular protein interaction mapping with FRET hybrids. Proc. Natl. Acad. Sci. USA 2006, 103, 18458–18463. [Google Scholar] [CrossRef] [PubMed]
- Tallen, G.; Riabowol, K. DisorderING promotes epigenetic order. FEBS Lett. 2017, 591, 257–259. [Google Scholar] [CrossRef]
- Avvakumov, N.; Lalonde, M.E.; Saksouk, N.; Paquet, E.; Glass, K.C.; Landry, A.J.; Doyon, Y.; Cayrou, C.; Robitaille, G.A.; Richard, D.E.; et al. Conserved molecular interactions within the HBO1 acetyltransferase complexes regulate cell proliferation. Mol. Cell. Biol. 2011, 32, 689–703. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palacios, A.; Blanco, F.J. Macromolecular Crowding Increases the Affinity of the PHD of ING4 for the Histone H3K4me3 Mark. Biomolecules 2020, 10, 234. https://doi.org/10.3390/biom10020234
Palacios A, Blanco FJ. Macromolecular Crowding Increases the Affinity of the PHD of ING4 for the Histone H3K4me3 Mark. Biomolecules. 2020; 10(2):234. https://doi.org/10.3390/biom10020234
Chicago/Turabian StylePalacios, Alicia, and Francisco J Blanco. 2020. "Macromolecular Crowding Increases the Affinity of the PHD of ING4 for the Histone H3K4me3 Mark" Biomolecules 10, no. 2: 234. https://doi.org/10.3390/biom10020234
APA StylePalacios, A., & Blanco, F. J. (2020). Macromolecular Crowding Increases the Affinity of the PHD of ING4 for the Histone H3K4me3 Mark. Biomolecules, 10(2), 234. https://doi.org/10.3390/biom10020234