Relevance of Hydrogen Bonds for the Histamine H2 Receptor-Ligand Interactions: A Lesson from Deuteration †
Abstract
1. Introduction
2. Computational Details
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Deupi, X.; Kobilka, B. Activation of G protein-coupled receptors. Adv. Protein Chem. 2007, 74, 137–166. [Google Scholar] [PubMed]
- Fredriksson, R.; Lagerstrom, M.C.; Lundin, L.G.; Schioth, H.B. The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol. Pharm. 2003, 63, 1256–1272. [Google Scholar] [CrossRef] [PubMed]
- Cong, X.; Topin, J.; Golebiowski, J. Class A GPCRs: Structure, function, modeling and structure-based ligand design. Curr. Pharm. Des. 2017, 23, 4390–4409. [Google Scholar] [CrossRef] [PubMed]
- Congreve, M.; Langmead, C.J.; Mason, J.S.; Marshall, F.H. Progress in structure based drug design for G-protein-coupled receptors. J. Med. Chem. 2011, 54, 4283–4311. [Google Scholar] [CrossRef] [PubMed]
- Keshelava, A.; Solis, G.P.; Hersch, M.; Koval, A.; Kryuchkov, M.; Bergmann, S.; Katanaev, V.L. High capacity in G protein-coupled receptor signaling. Nat. Commun. 2018, 9, 876. [Google Scholar] [CrossRef]
- Franco, M.I.; Turin, L.; Mershin, A.; Skoulakis, E.M.C. Molecular vibration-sensing component in drosophila melanogaster olfaction. Proc. Natl. Acad. Sci. USA 2011, 108, 3797–3802. [Google Scholar] [CrossRef]
- Turin, L. A spectroscopic mechanism for primary olfactory reception. Chem. Senses 1996, 21, 773–791. [Google Scholar] [CrossRef]
- Turin, L. A method for the calculation of odor character from molecular structure. J. Biol. 2002, 216, 367–385. [Google Scholar] [CrossRef]
- Huelga, S.F.; Plenio, M.B. Vibrations, quanta and biology. Contemp. Phys. 2013, 54, 181–207. [Google Scholar] [CrossRef]
- Bittner, E.R.; Madalan, A.; Czader, A.; Roman, G. Quantum origins of molecular recognition and olfaction in Drosophila. J. Chem. Phys. 2012, 137, 22A551. [Google Scholar] [CrossRef]
- Hettinger, T.P. Olfaction is a chemical sense, not a spectral sense. Proc. Natl. Acad. Sci. USA 2011, 108, E349. [Google Scholar] [CrossRef] [PubMed]
- Warshel, A.; Bora, R.P. Defining and quantifying the role of dynamics in enzyme catalysis. J. Chem. Phys. 2016, 144, 180901. [Google Scholar] [CrossRef] [PubMed]
- Kržan, M.; Vianello, R.; Maršavelski, A.; Repič, M.; Zakšek, M.; Kotnik, K.; Fijan, E.; Mavri, J. The quantum nature of drug-receptor interactions: Deuteration changes binding affinities for histamine receptor ligands. PLoS ONE 2016, 11, e0154002. [Google Scholar] [CrossRef] [PubMed]
- Ubbelohde, A.R.; Gallagher, K.J. Acid-base effects in hydrogen bonds in crystals. Acta Cryst. 1955, 8, 71–83. [Google Scholar] [CrossRef]
- Shao, L.; Hewitt, M.C. The kinetic isotope effect in the search for deuterated drugs. Drug News Perspect. 2010, 23, 398–404. [Google Scholar] [CrossRef]
- Guengerich, F.P. Kinetic deuterium isotope effects in cytochrome P450 oxidation reactions. J. Label. Comp. Radiopharm. 2013, 56, 428–431. [Google Scholar] [CrossRef]
- Shao, L.; Abolin, C.; Hewitt, M.C.; Koch, P.; Varney, M. Derivatives of tramadol for increased duration of effect. Bioorg. Med. Chem. Lett. 2006, 16, 691–694. [Google Scholar] [CrossRef]
- Russak, E.M.; Bednarczyk, E.M. Impact of deuterium substitution on the pharmacokinetics of pharmaceuticals. Ann. Pharm. 2018, 53, 211–216. [Google Scholar] [CrossRef]
- DeWitt, S.H.; Maryanoff, B.E. Deuterated drug molecules: Focus on FDA-approved deutetrabenazine. Biochemistry 2018, 57, 472–473. [Google Scholar] [CrossRef]
- Liu, J.F.; Harbeson, S.L.; Brummel, C.L.; Tung, R.; Silverman, R.; Doller, D. A decade of deuteration in medicinal chemistry. Ann. Rep. Med. Chem. 2017, 50, 519–542. [Google Scholar]
- Schmidt, C. First deuterated drug approved. Nat. Biotechnol. 2017, 35, 493–494. [Google Scholar] [CrossRef] [PubMed]
- Gant, T.G. Using deuterium in drug discovery: Leaving the label in the drug. J. Med. Chem. 2014, 57, 3595–3611. [Google Scholar] [CrossRef] [PubMed]
- Olsson, M.H.M.; Søndergaard, C.R.; Rostkowski, M.; Jensen, J.H. PROPKA3: Consistent treatment of internal and surface residues in empirical pKa predictions. J. Chem. Theory Comput. 2011, 7, 525–537. [Google Scholar] [CrossRef] [PubMed]
- Stark, H. Histamine H4 receptor. A novel drug target for immunoregulation and inflammation; De Gruyter: Berlin, Germany, 2013. [Google Scholar]
- Jantratid, E.; Prakongpan, S.; Dressman, J.B.; Amidon, G.L.; Junginger, H.E.; Midha, K.K.; Barends, D.M. Biowaiver monographs for immediate release solid oral dosage forms: Cimetidine. J. Pharm. Sci. 2006, 95, 974–984. [Google Scholar] [CrossRef]
- Islam, M.S.; Narurkar, M.M. Solubility, stability and ionization behaviour of famotidine. J. Pharm. Pharm. 1993, 45, 682–686. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16; Revision, A.03; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Liao, R.Z.; Georgieva, P.; Yu, J.G.; Himo, F. Mechanism of mycolic acid cyclopropane synthase: A theoretical study. Biochemistry 2011, 50, 1505–1513. [Google Scholar] [CrossRef]
- Vianello, R.; Repič, M.; Mavri, J. How are biogenic amines metabolized by monoamine oxidases? Eur. J. Org. Chem. 2012, 36, 7057–7065. [Google Scholar] [CrossRef]
- Maršavelski, A.; Vianello, R. What a difference a methyl group makes: The selectivity of monoamine oxidase B towards histamine and N-methylhistamine. Chem. Eur. J. 2017, 23, 2915–2925. [Google Scholar] [CrossRef]
- Tandarić, T.; Vianello, R. Computational insight into the mechanism of the irreversible inhibition of monoamine oxidase enzymes by the antiparkinsonian propargylamine inhibitors rasagiline and selegiline. ACS Chem. Neurosci. 2019, 10, 3532–3542. [Google Scholar]
- Himo, F. Recent trends in quantum chemical modeling of enzymatic reactions. J. Am. Chem. Soc. 2017, 139, 6780–6786. [Google Scholar] [CrossRef]
- Blomberg, M.R.A.; Borowski, T.; Himo, F.; Liao, R.-Z.; Siegbahn, P.E.M. Quantum chemical studies of mechanisms for metalloenzymes. Chem. Rev. 2014, 114, 3601–3658. [Google Scholar]
- Quesne, M.G.; Borowski, T.; de Visser, S.P. Quantum mechanics/molecular mechanics modeling of enzymatic processes: Caveats and breakthroughs. Chem. Eur. J. 2016, 22, 2562–2581. [Google Scholar] [CrossRef] [PubMed]
- Sousa, S.F.; Ribeiro, A.J.M.; Neves, R.P.P.; Brás, N.F.; Cerqueira, N.M.F.S.A.; Fernandes, P.A.; Ramos, M.J. Application of quantum mechanics/molecular mechanics methods in the study of enzymatic reaction mechanisms. Wires Comput. Mol. Sci. 2017, 7, e1281. [Google Scholar] [CrossRef]
- Quesne, M.G.; Silveri, F.; de Leeuw, N.H.; Catlow, C.R.A. Advances in sustainable catalysis: A computational perspective. Front. Chem. 2019, 7, 182. [Google Scholar] [CrossRef] [PubMed]
- Isom, D.G.; Castaneda, C.A.; Cannon, B.R.; Garcia-Moreno, E.B. Large shifts in pKa values of lysine residues buried inside a protein. Proc. Natl. Acad. Sci. USA 2011, 108, 5260–5265. [Google Scholar] [CrossRef] [PubMed]
- Harms, M.J.; Castaneda, C.A.; Schlessman, J.L.; Sue, G.R.; Garcia-Moreno, E.B. The pKa values of acidic and basic residues buried at the same internal location in a protein are governed by different factors. J. Mol. Biol. 2009, 389, 34–47. [Google Scholar] [CrossRef]
- Hodošček, M.; Hadži, D. Proton transfer in the HCOOH·CH3NH2 complex. Ab initio study with various basis sets and solvent reaction field. J. Mol. Struct. 1989, 198, 461–473. [Google Scholar] [CrossRef]
- Scheiner, S.; Duan, X. Effect of intermolecular orientation upon proton transfer within a polarizable medium. Biophys. J. 1991, 60, 874–883. [Google Scholar] [CrossRef][Green Version]
- De Souza, J.M.; Freire, P.T.C.; Bordallo, H.N.; Argyriou, D.N. Structural isotopic effects in the smallest chiral amino acid: Observation of a structural phase transition in fully deuterated alanine. J. Phys. Chem. B 2007, 111, 5034–5039. [Google Scholar] [CrossRef]
- Shi, C.; Zhang, X.; Yu, C.-H.; Yao, Y.-F.; Zhang, W. Geometric isotope effect of deuteration in a hydrogen-bonded host-guest crystal. Nat. Commun. 2018, 9, 481. [Google Scholar] [CrossRef]
- Rivera-Rivera, L.A.; Wang, Z.; McElmurry, B.A.; Willaert, F.F.; Lucchese, R.R.; Bevan, J.W.; Suenram, R.D.; Lovas, F.J. A ground state morphed intermolecular potential for the hydrogen bonded and van der Waals isomers in OC:HI and a prediction of an anomalous deuterium isotope effect. J. Chem. Phys. 2010, 133, 184305. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.K.; Murli, C.; Sharma, S.M. High pressure Raman spectroscopic study of deuterated γ-glycine. J. Phys. Chem. B 2008, 112, 15867–15874. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, R.O.; Freire, P.T.C.; Bordallo, H.N.; Lima, J.A., Jr.; Melo, F.E.A.; Mendes Filho, J.; Argyriou, D.N.; Lima, R.J.C. High-pressure Raman spectra of deuterated L-alanine crystal. J. Raman Spectrosc. 2009, 40, 958–963. [Google Scholar] [CrossRef]
- Smirnov, S.N.; Golubev, N.S.; Denisov, G.S.; Benedict, H.; Schah-Mohammedi, P.; Limbach, H.-H. Hydrogen/deuterium isotope effects on the NMR chemical shifts and geometries of intermolecular low-barrier hydrogen-bonded complexes. J. Am. Chem. Soc. 1996, 118, 4094–4101. [Google Scholar] [CrossRef]
- Vianello, R.; Mavri, J. Microsolvation of the histamine monocation in aqueous solution: The effect on structure, hydrogen bonding ability and vibrational spectrum. New J. Chem. 2012, 36, 954–962. [Google Scholar] [CrossRef]
- Wheeler, S.E. Homodesmotic reactions for thermochemistry. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2012, 2, 204–220. [Google Scholar] [CrossRef]
- Perdan-Pirkmajer, K.; Mavri, J.; Kržan, M. Histamine (re)uptake by astrocytes: An experimental and computational study. J. Mol. Model. 2010, 16, 1151–1158. [Google Scholar] [CrossRef]
- Llinàs, A.; Glen, R.C.; Goodman, J.M. Solubility challenge: Can you predict solubilities of 32 molecules using a database of 100 reliable measurements? J. Chem. Inf. Model. 2008, 48, 1289–1303. [Google Scholar] [CrossRef]
- Birdsall, N.J. Cloning and structure-function of the H2 histamine receptor. Trends Pharm. Sci. 1991, 12, 9–10. [Google Scholar] [CrossRef]
- Gantz, I.; DelValle, J.; Wang, L.D.; Tashiro, T.; Munzert, G.; Guo, Y.J.; Konda, Y.; Yamada, T. Molecular basis for the interaction of histamine with the histamine H2 receptor. J. Biol. Chem. 1992, 267, 20840–20843. [Google Scholar]
- Black, J.W.; Duncan, W.A.; Durant, C.J.; Ganelin, C.R.; Parsons, E.M. Definition and antagonism of histamine H2-receptors. Nature 1972, 236, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Tehan, B.G.; Bortolato, A.; Blaney, F.E.; Weir, M.P.; Mason, J.S. Unifying family A GPCR theories of activation. Pharm. 2014, 143, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Heo, Y.A.; Scott, L.J. Deutetrabenazine; a review in chorea associated with Huntington’s disease. Drugs 2017, 17, 1857–1864. [Google Scholar] [CrossRef] [PubMed]
- Claassen, D.O.; Carroll, B.; De Boer, L.M.; Wu, E.; Ayyagari, R.; Gandhi, S.; Stamler, D. Indirect tolerability comparison of Deutetrabenazine and Tetrabenazine for Huntington disease. J. Clin. Mov. Disord. 2017, 4, 3. [Google Scholar] [CrossRef]
- Dean, M.; Sung, V.W. Review of deutetrabenazine: A novel treatment for chorea associated with Huntington s disease. Drug Des. Devel. Ther. 2018, 12, 313–319. [Google Scholar] [CrossRef]
- Kaur, S.; Gupta, M. Deuteration as a tool for optimization of metabolic stability and toxicity of drugs. Glob. J. Pharmaceu. Sci. 2017, 1, 555566. [Google Scholar]
- Tung, R.D. Deuterium medicinal chemistry comes of age. Future Med. Chem. 2016, 8, 491–494. [Google Scholar] [CrossRef]
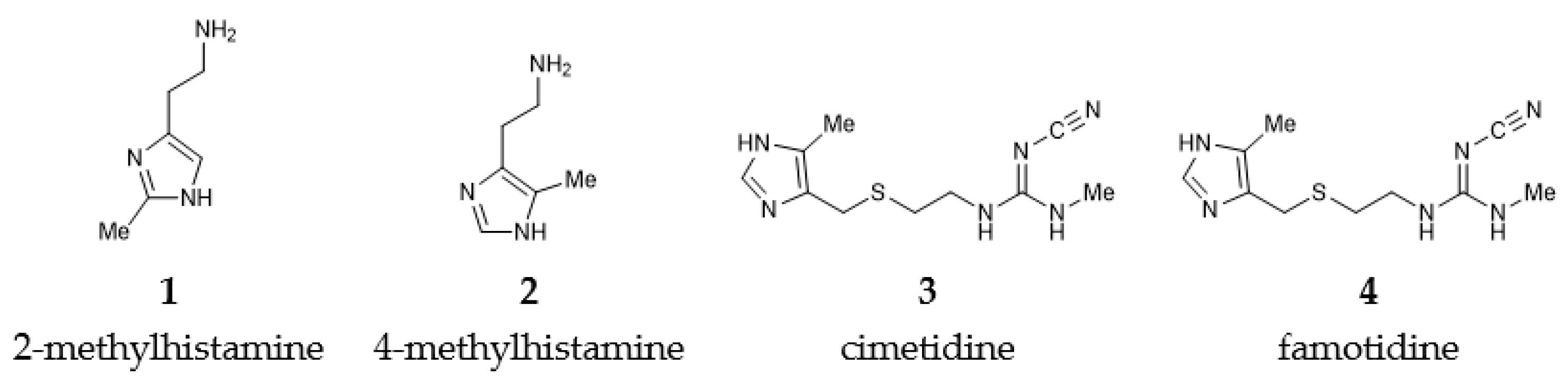
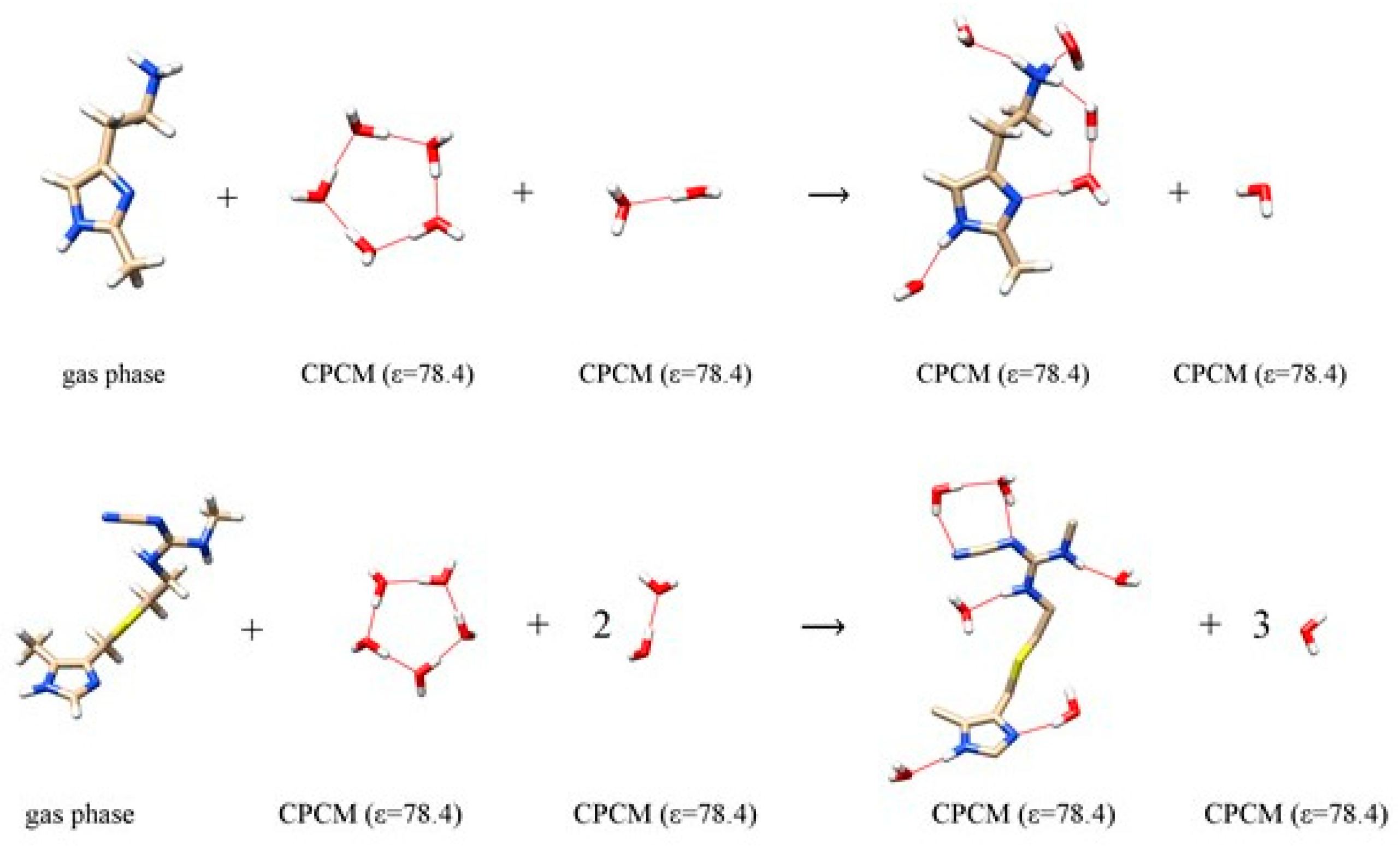
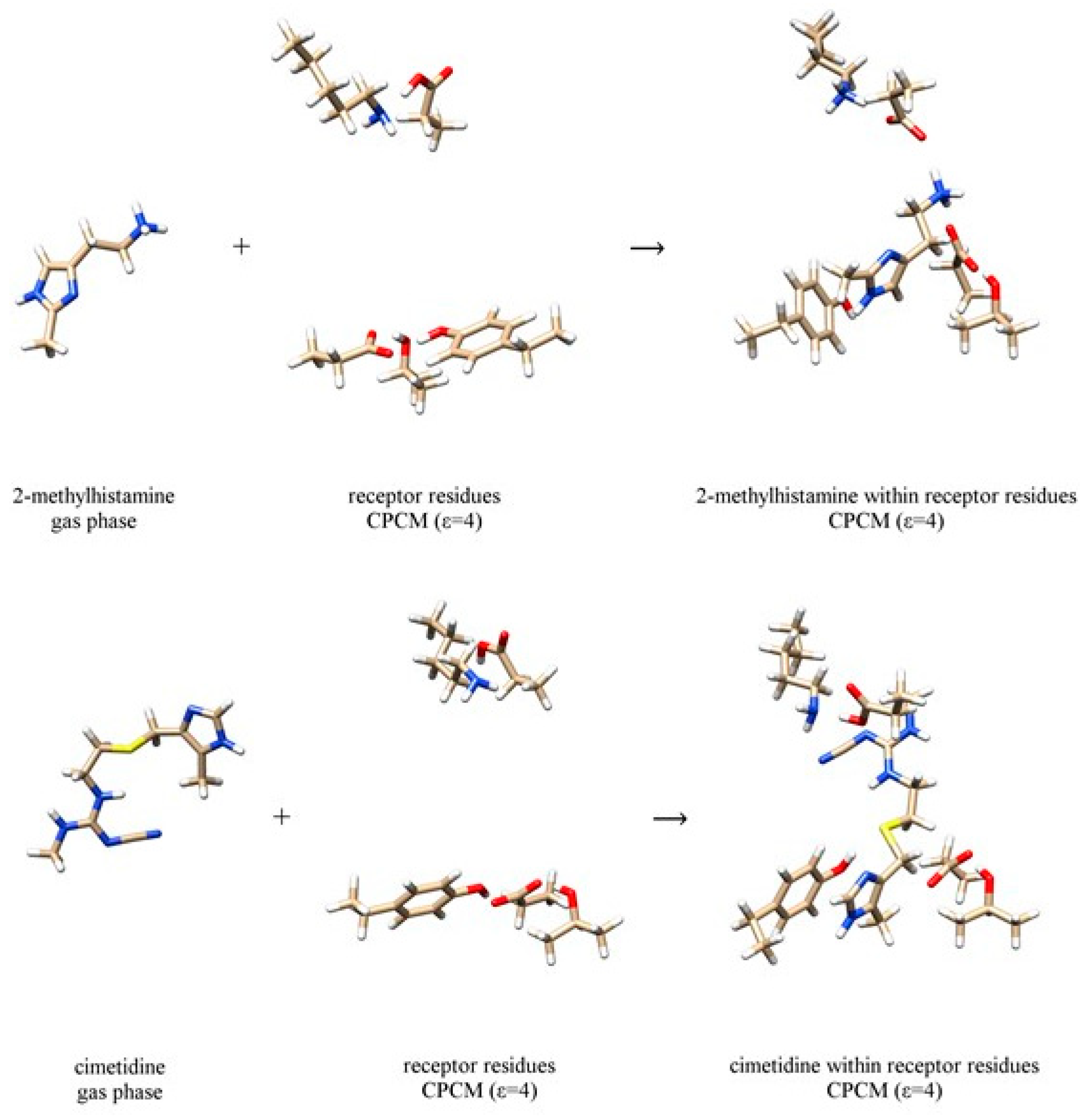
| Ligand | In H2O | In D2O | ΔΔEBIND,CALC | ΔΔEBIND,EXP | ||||
|---|---|---|---|---|---|---|---|---|
| ΔEHYDR | ΔEINTER | ΔEBIND | ΔEHYDR | ΔEINTER | ΔEBIND | |||
| 2-methylhistamine (1) | –66.92 | –71.99 | –5.07 | –66.88 | –69.57 | –2.69 | 2.38 | 2.08 |
| 4-methylhistamine (2) | –67.38 | –86.13 | –18.75 | –67.55 | –87.40 | –19.85 | –1.10 | –0.49 |
| cimetidine (3) | –19.69 | –27.27 | –7.58 | –19.54 | –27.21 | –7.67 | –0.09 | 0.00 |
| famotidine (4) | –14.29 | –31.17 | –17.88 | –13.97 | –31.90 | –17.93 | –0.05 | 0.00 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kržan, M.; Keuschler, J.; Mavri, J.; Vianello, R. Relevance of Hydrogen Bonds for the Histamine H2 Receptor-Ligand Interactions: A Lesson from Deuteration. Biomolecules 2020, 10, 196. https://doi.org/10.3390/biom10020196
Kržan M, Keuschler J, Mavri J, Vianello R. Relevance of Hydrogen Bonds for the Histamine H2 Receptor-Ligand Interactions: A Lesson from Deuteration. Biomolecules. 2020; 10(2):196. https://doi.org/10.3390/biom10020196
Chicago/Turabian StyleKržan, Mojca, Jan Keuschler, Janez Mavri, and Robert Vianello. 2020. "Relevance of Hydrogen Bonds for the Histamine H2 Receptor-Ligand Interactions: A Lesson from Deuteration" Biomolecules 10, no. 2: 196. https://doi.org/10.3390/biom10020196
APA StyleKržan, M., Keuschler, J., Mavri, J., & Vianello, R. (2020). Relevance of Hydrogen Bonds for the Histamine H2 Receptor-Ligand Interactions: A Lesson from Deuteration. Biomolecules, 10(2), 196. https://doi.org/10.3390/biom10020196








