Sodium Chloride Induced Stress Responses of Antioxidative Activities in Leaves and Roots of Pistachio Rootstock
Abstract
1. Introduction
2. Results
2.1. The Effect of Salinity on Plant Growth and Morphology
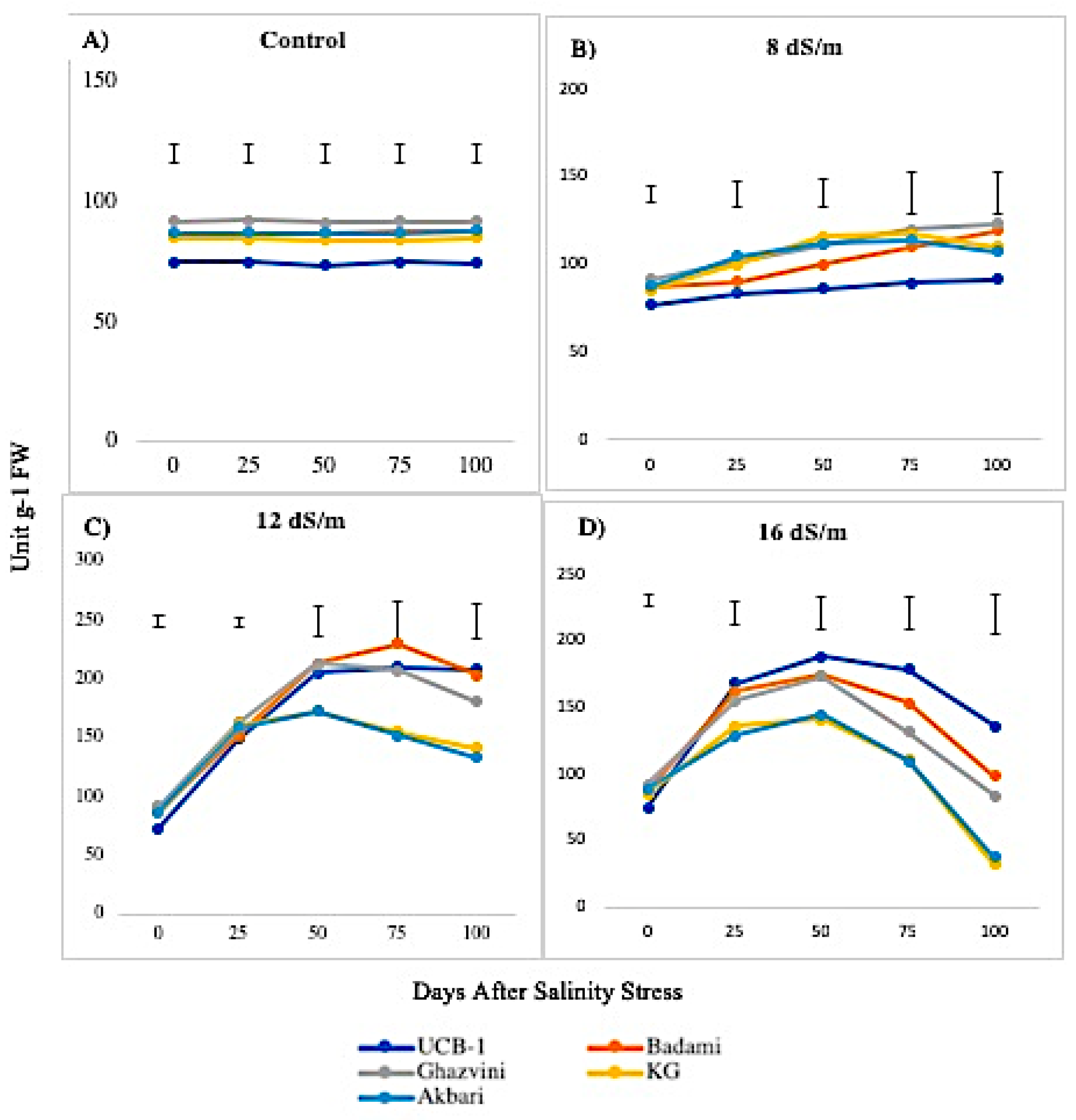
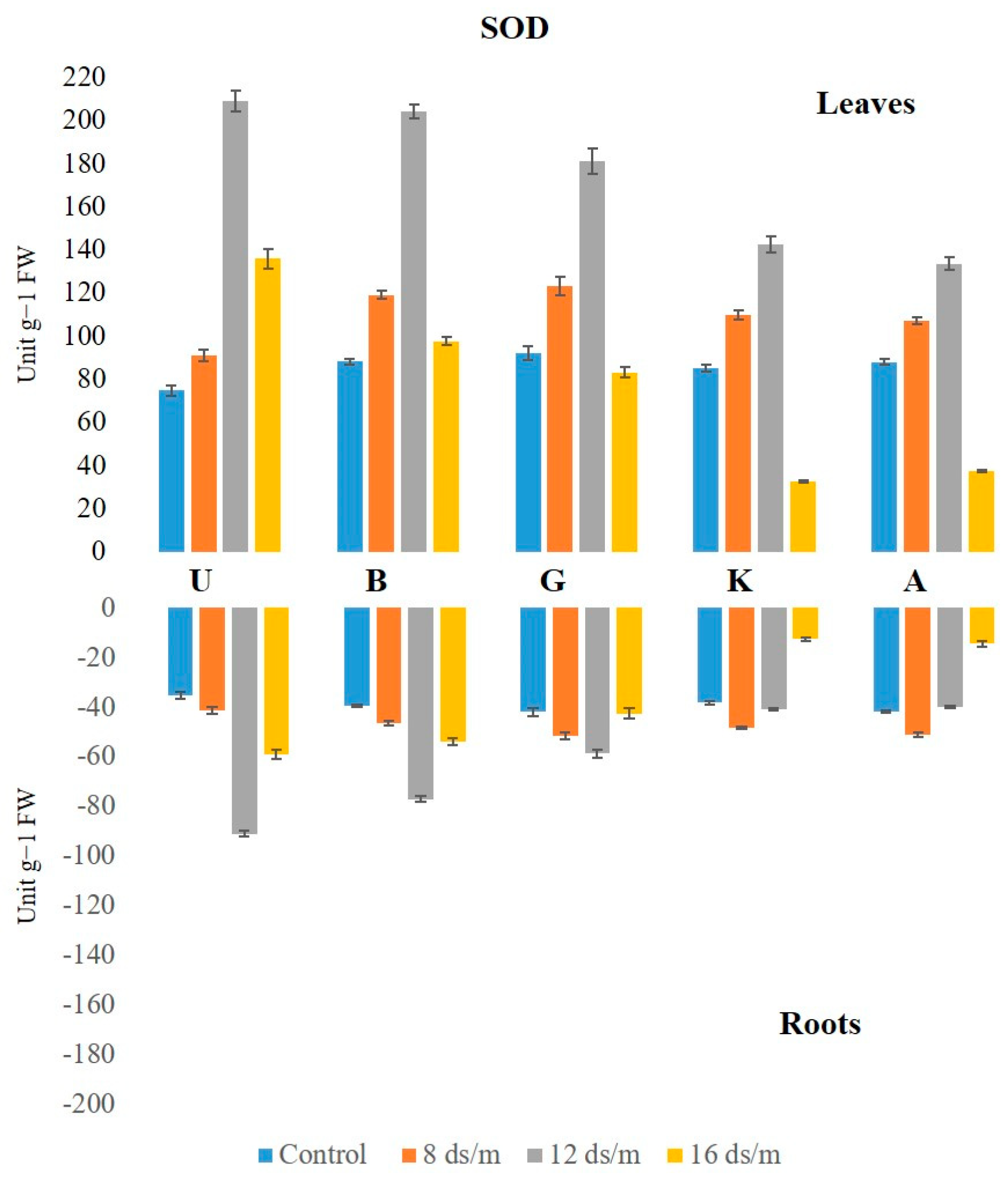
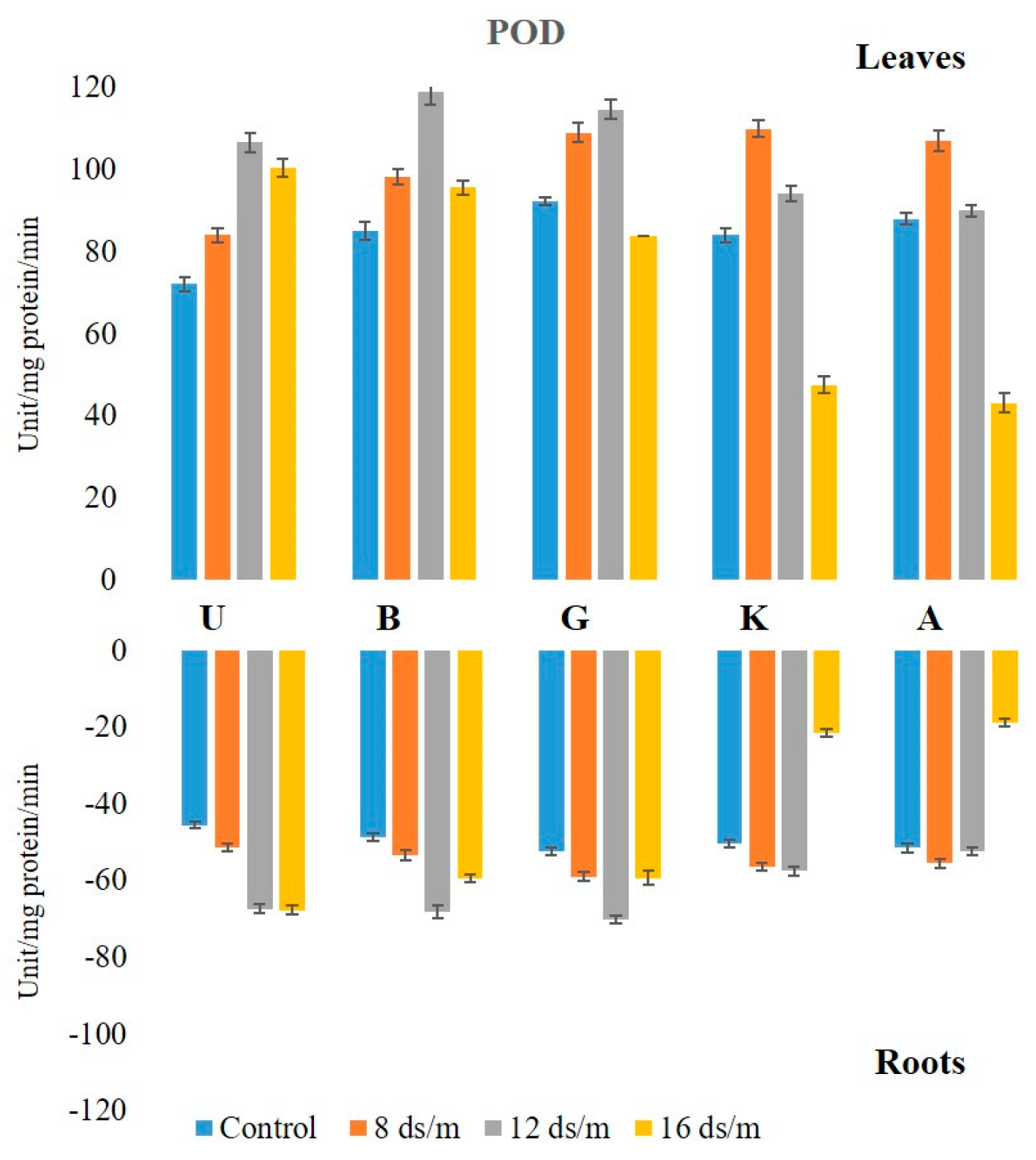
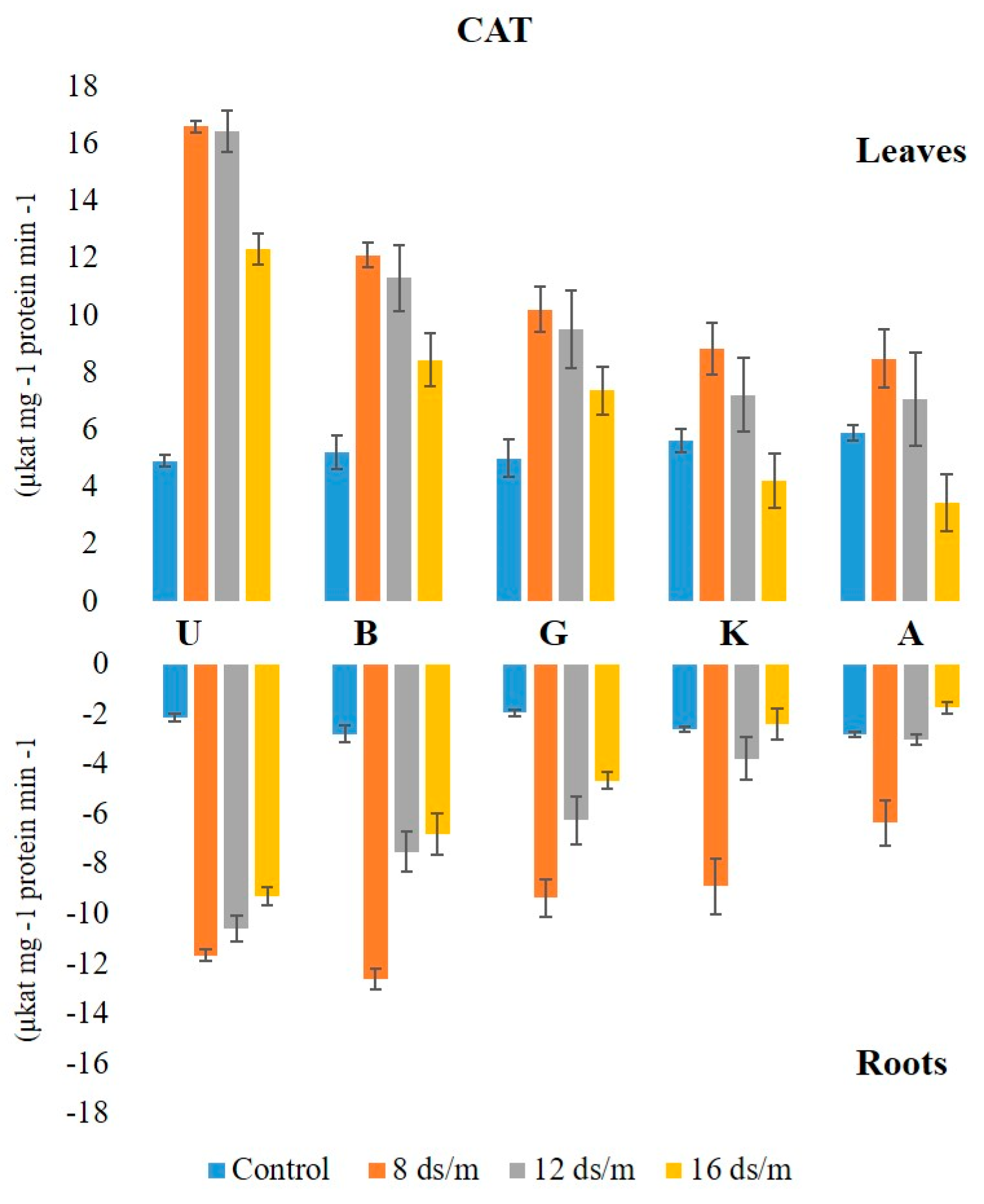
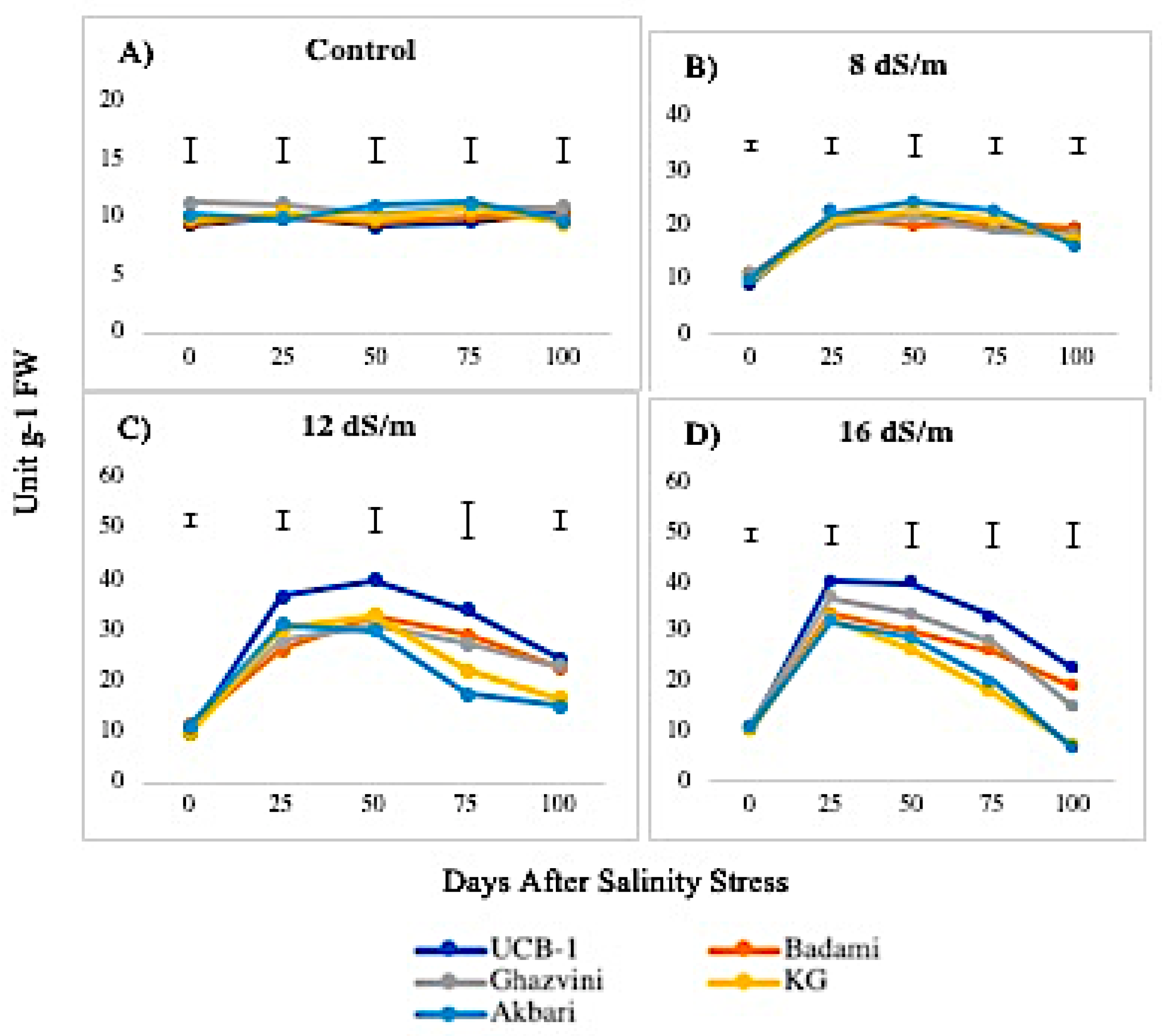
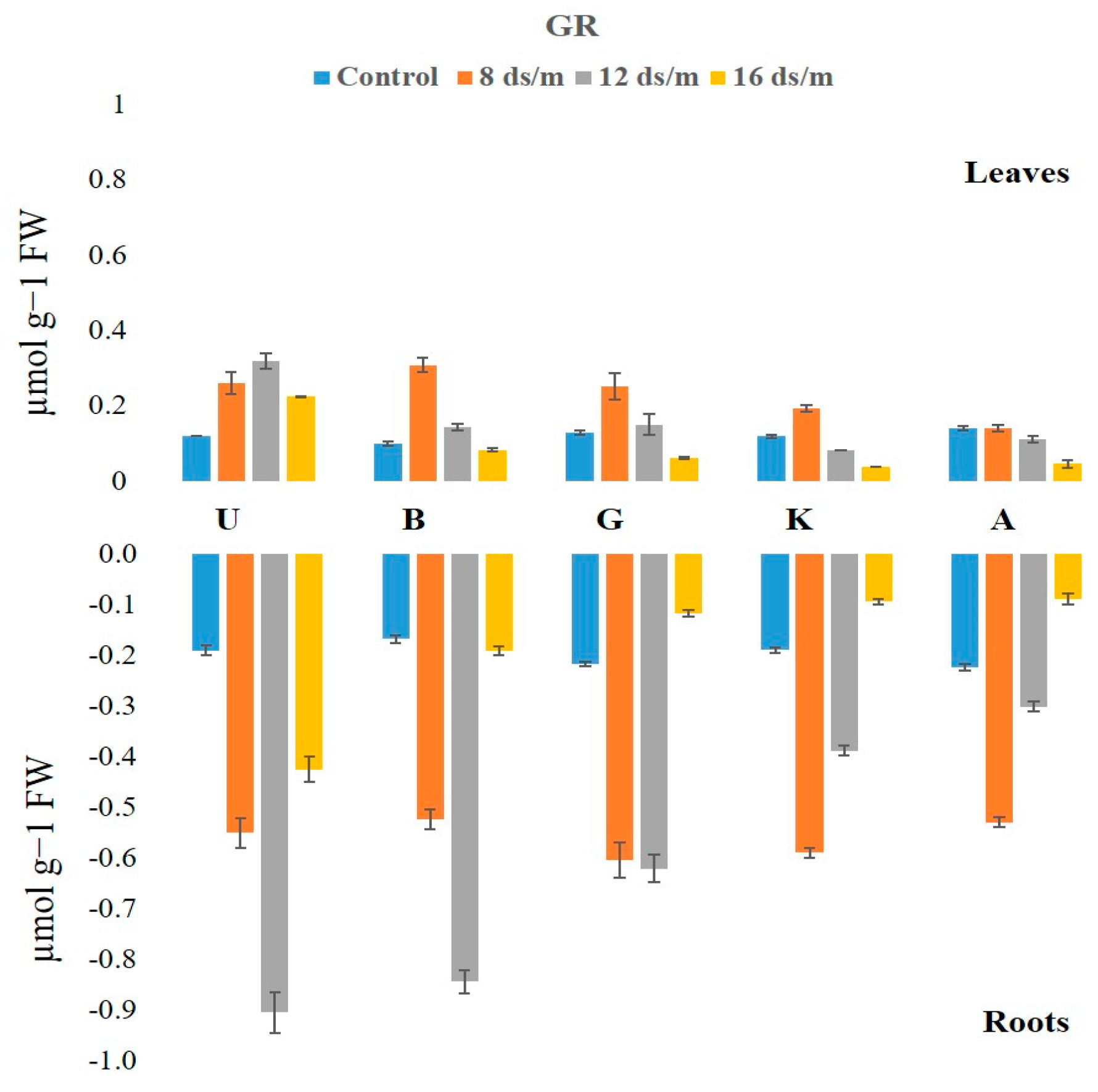
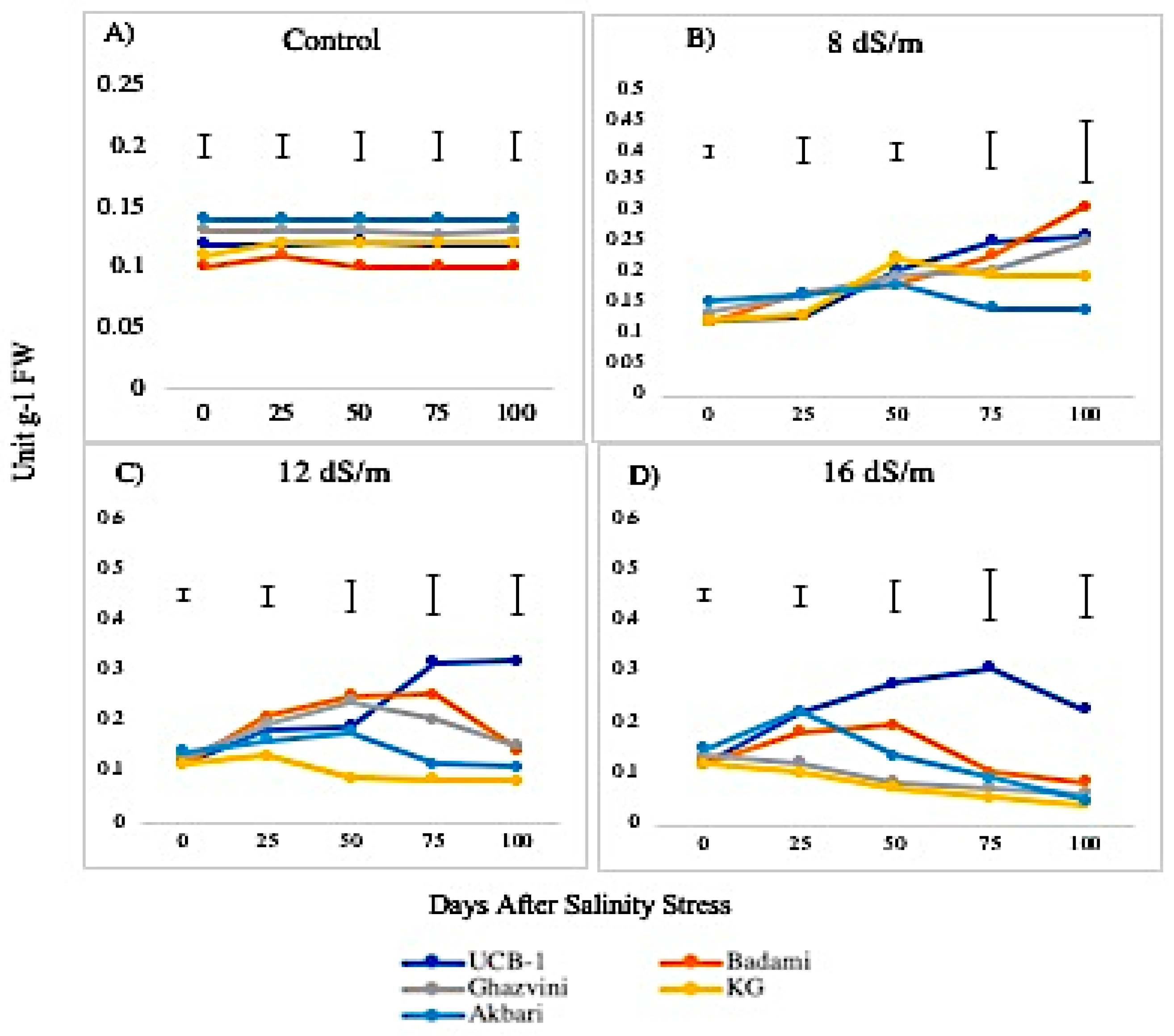
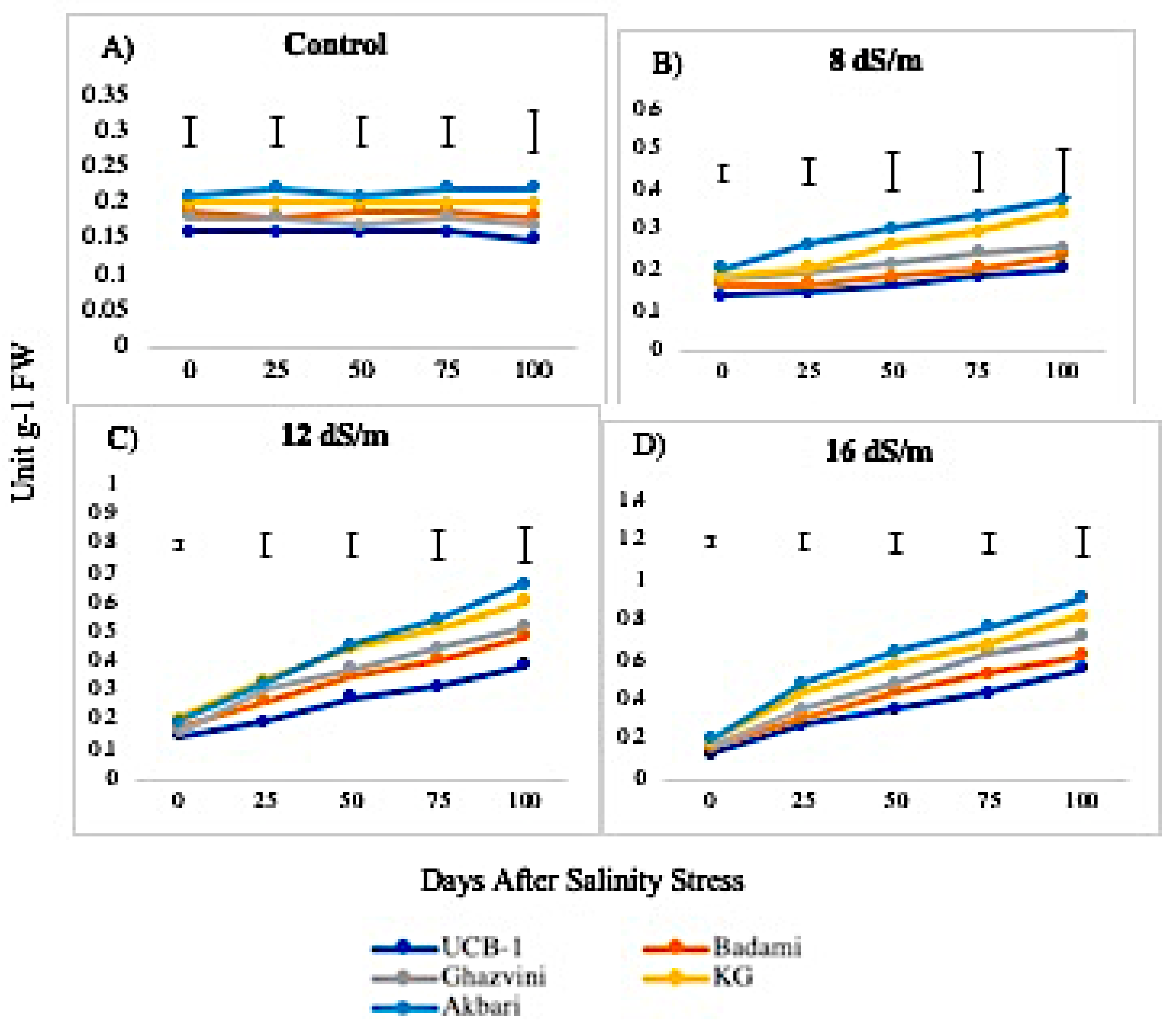
2.2. Antioxidant Enzyme Activity
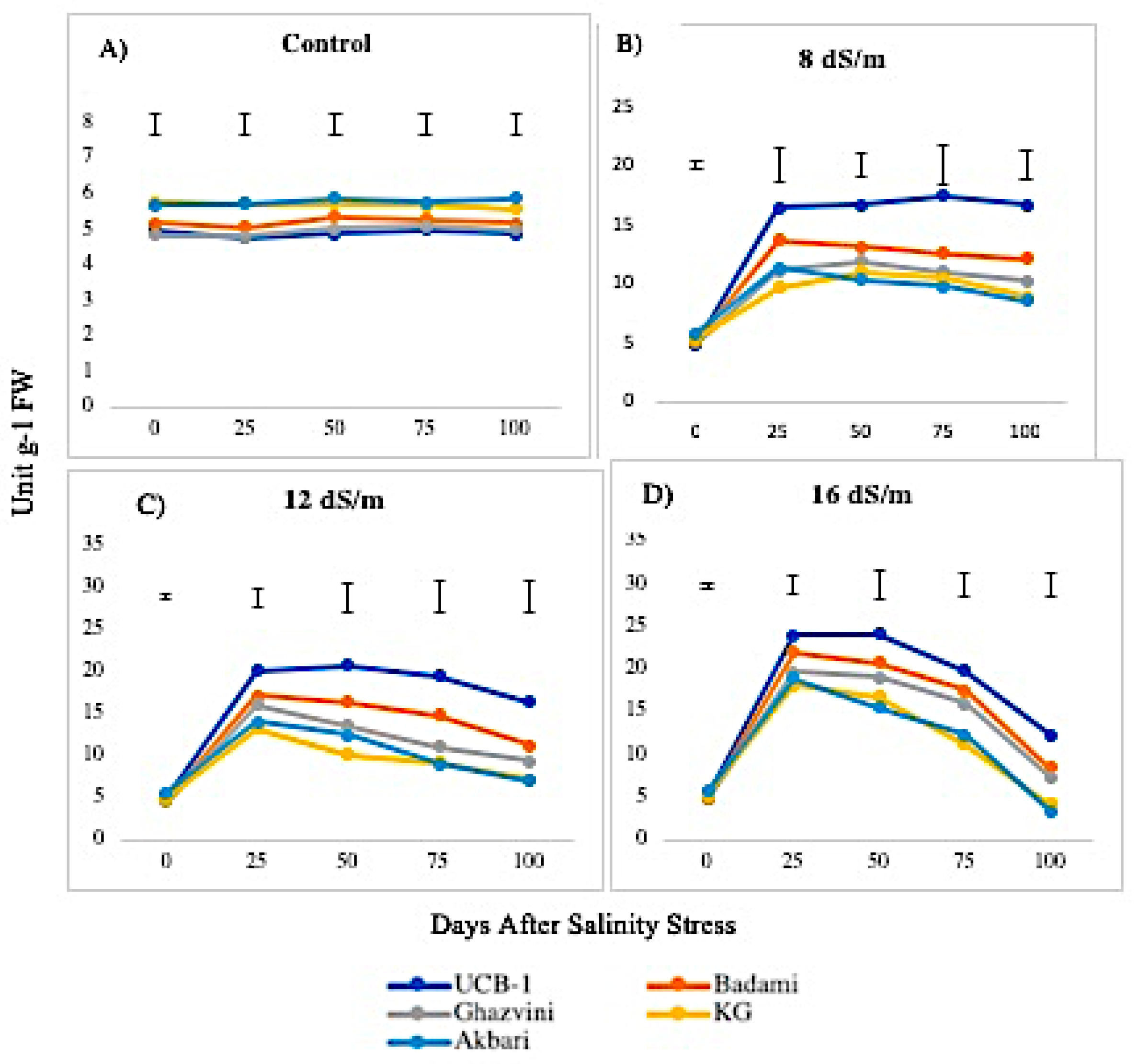
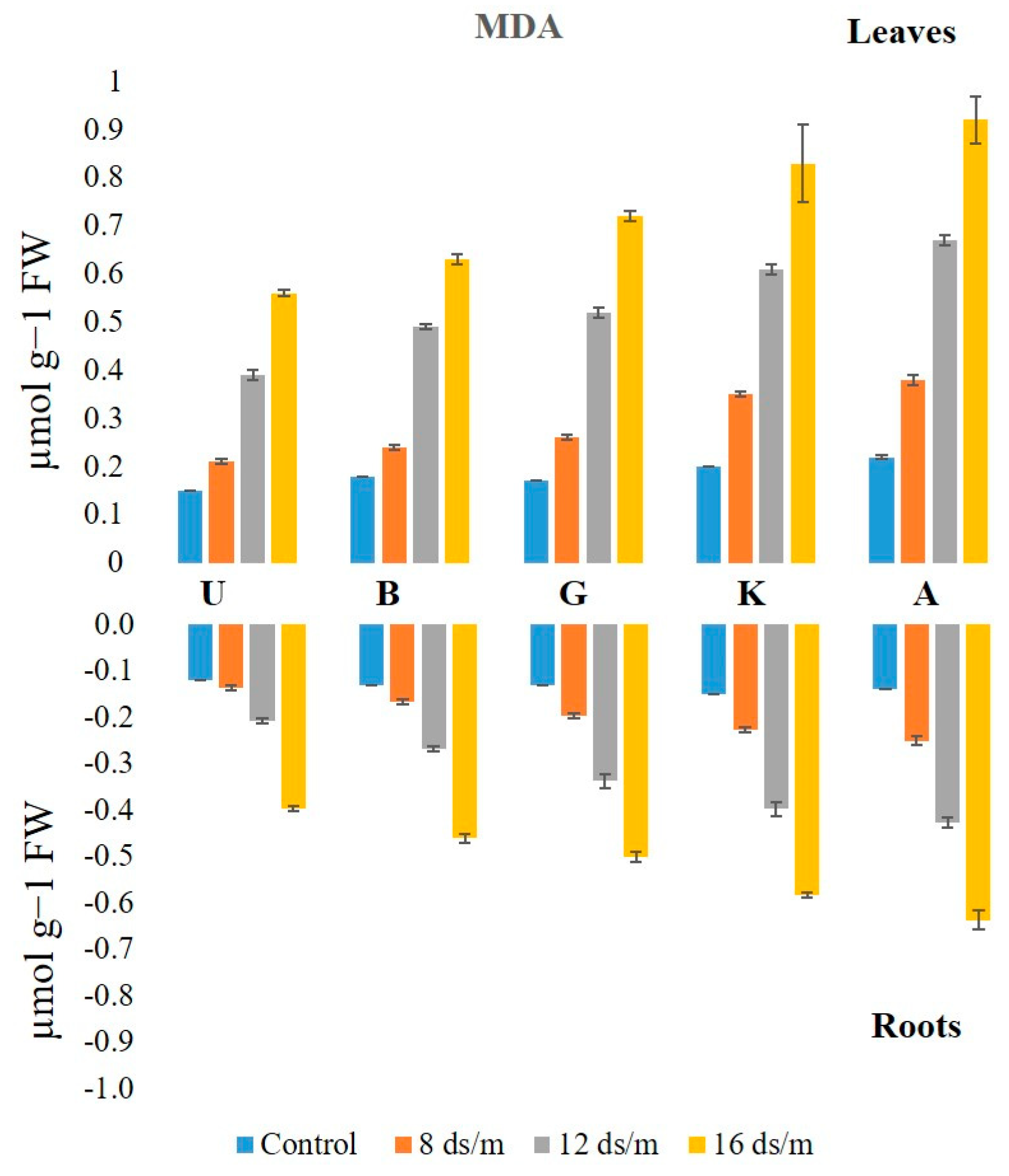
2.3. Lipid Peroxidation
2.4. Correlation Analysis
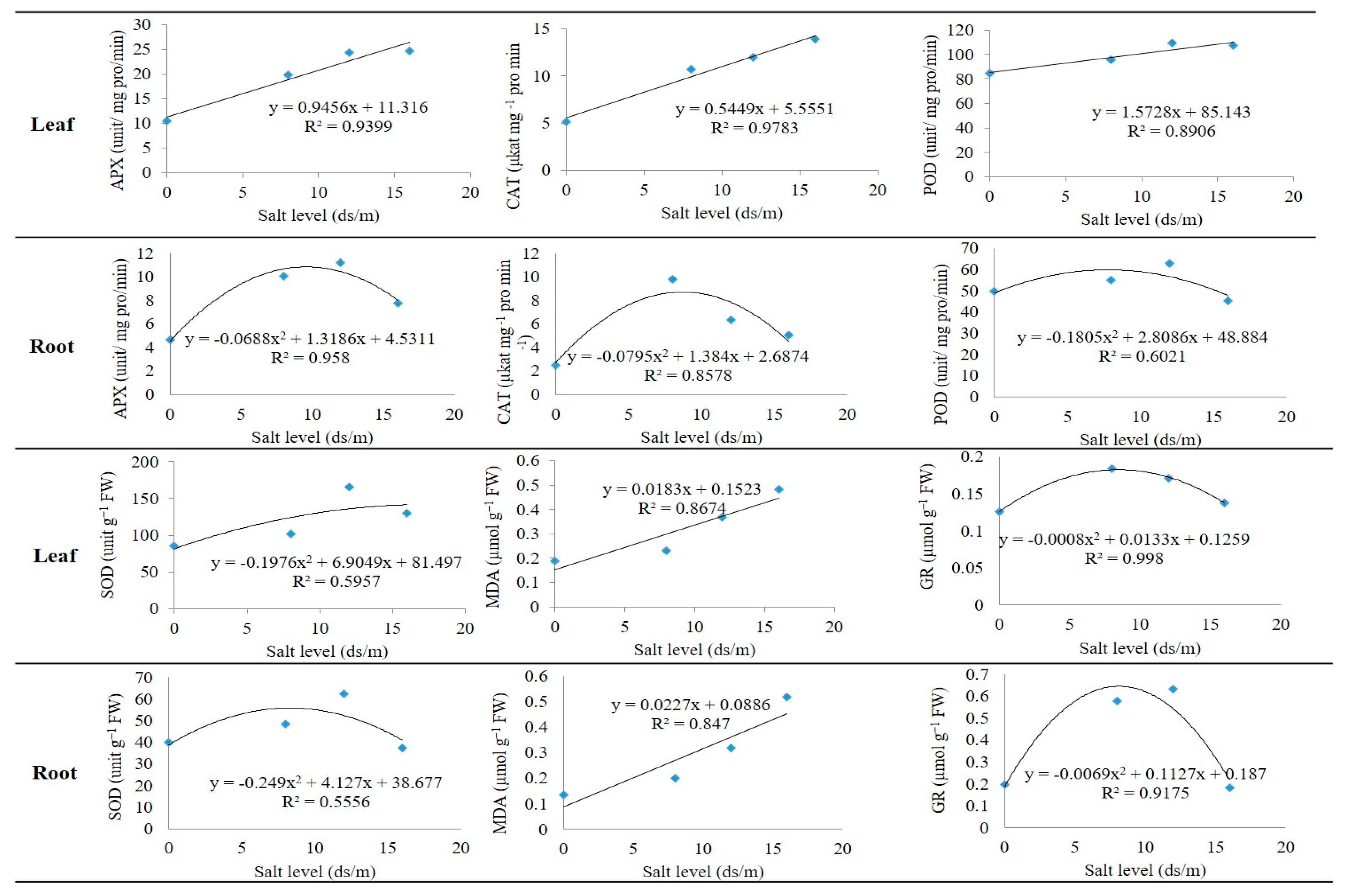
2.5. Model Analysis
3. Discussion
3.1. Quantitative Variation among the Antioxidative Enzymes Is Tissue Specific and Depends on Both Duration and Levels of NaCl-Induced Salt Stress
3.2. Lipid Peroxidation Activity in Relation to Antioxidant Enzymes among Rootstock
3.3. Correlation between H2O2 Activity and Antioxidant Responses
3.4. Genetic Variation in Antioxidant Activity among Rootstock to Salt Stress
4. Materials and Methods
4.1. Plant Material and Growth Conditions
4.2. Salinity Treatment and Sample Collection
4.3. Determination of Enzyme Activity
4.3.1. SOD Activity
4.3.2. POD Activity
4.3.3. CAT Activity
4.3.4. APX Activity
4.3.5. GR Activity
4.4. Lipid Peroxidation
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Aliakbarkhani, S.T.; Farajpour, M.; Asadian, A.H.; Aalifar, M.; Ahmadi, S.; Akbari, M. Variation of nutrients and antioxidant activity in seed and exocarp layer of some Persian pistachio genotypes. Ann. Agric. Sci. 2017, 62, 39–44. [Google Scholar] [CrossRef]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant. Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef]
- Kurum, R.; Ulukapi, K.; Aydinskir, K.; Onus, A.N. The influence of salinity on seedling growth of some pumpkin varieties used as rootstock. Notulae Bot. Horti. Agrobot. Cluj. Napoca 2013, 41, 219–225. [Google Scholar] [CrossRef]
- Hima Kumari, P.S.; Anil Kumar, S.; Sivan, P.; Katam, R.; Suravajhala, P.; Rao, K.S.; Varshney, R.K.; Kavi Kishor, P.B. Overexpression of a plasma membrane bound Na+/H+-antiporter-like protein (SbNHXLP) confers salt tolerance and improves fruit yield in tomato by maintaining ion homeostasis. Front. Plant. Sci. 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
- de Azevedo Neto, A.D.; Prisco, J.T.; Enéas-Filho, J.; de Abreu, C.E.B.; Gomes-Filho, E. Effect of salt stress on anti-oxidative enzymes and lipid peroxidation in leaves and roots of salt-tolerant and salt-sensitive maize genotypes. Environ. Exp. Bot. 2006, 56, 87–94. [Google Scholar] [CrossRef]
- Gao, S.; Ouyang, C.; Wang, S.; Xu, Y.; Tang, L.; Chen, F. Effects of salt stress on growth, antioxidant enzyme and phenylalanine ammonia-lyase activities in Jatropa curcas L. seedlings. Plant. Soil Environ. 2008, 54, 374–381. [Google Scholar] [CrossRef]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef]
- Feng, X.; Lai, Z.; Lin, Y.; Lai, G.; Lian, C. Genome-wide identification and characterization of the superoxide dismutase gene family in Musa acuminata cv. Tianbaojiao (AAA group). BMC Genom. 2015, 16, 823. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Noctor, G. Ascorbate and glutathione: The heart of the redox hub. Plant. Physiol. 2011, 155, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wu, C.; Xie, B.; Liu, Y.; Cui, J.; Chen, G.; Zhang, Y. Model analyzing the antioxidant responses of leaves and roots of switch grass to NaCl-salinity stress. Plant. Physiol. Biochem. 2012, 58, 288–296. [Google Scholar] [CrossRef]
- Pandey, H.C.; Baig, M.J.; Chandra, A.; Bhatt, R.K. Drought stress induced changes in lipid peroxidation and antioxidant system in genus Avena. J. Environ. Biol. 2012, 31, 435–440. [Google Scholar]
- Pandey, S.; Fartyal, D.; Agarwal, A.; Shukla, T.; James, D.; Kaul, T.; Reddy, M.K. Abiotic stress tolerance in plants: Myriad roles of ascorbate peroxidase. Front. Plant. Sci. 2017, 8, 581. [Google Scholar] [CrossRef] [PubMed]
- Begara-Morales, J.C.; Sánchez-Calvo, B.; Chaki, M.; Mata-Pérez, C.; Valderrama, R.; Padilla, M.N.; Barroso, J.B. Differential molecular response of monodehydroascorbate reductase and glutathione reductase by nitration and S-nitrosylation. J. Exp. Bot. 2015, 66, 5983–5996. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M.H.S.; Etemadi, N.; Arab, M.M.; Aalifar, M.; Arab, M.; Pessarakli, M. Molecular and physiological responses of Iranian Perennial ryegrass as affected by Trinexapac ethyl, Paclobutrazol and Abscisic acid under drought stress. Plant. Physiol. Biochem. 2017, 111, 129–143. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Fang, Y.; Ji, Y.; Jiang, Z.; Wang, L. Effects of salt stress on ion content, antioxidant enzymes and protein profile in different tissues of Broussonetia papyrifera. S. Afr. J. Bot. 2013, 85, 1–9. [Google Scholar] [CrossRef]
- Walker, R.R.; Torokfalvy, E.; Behboudian, M.H. Uptake; distribution of chloride, sodium and potassium ions and growth of salt-treated pistachio plants. Austral. J. Agric. Res. 1987, 38, 383–394. [Google Scholar] [CrossRef]
- Aliakbarkhani, S.T.; Akbari, M.; Hassankhah, A.; Talaie, A.; Moghadam, M.F. Phenotypic and genotypic variation in Iranian pistachios. J. Genet. Eng. Biotechnol. 2015, 13, 235–241. [Google Scholar] [CrossRef]
- Van Breusegem, F.; Vranová, E.; Dat, J.F.; Inzé, D. The role of active oxygen species in plant signal transduction. Plant. Sci. 2001, 161, 405–414. [Google Scholar] [CrossRef]
- Chawla, S.; Jain, S.; Jain, V. Salinity induced oxidative stress and antioxidant system in salt-tolerant and salt-sensitive cultivars of rice (Oryza sativa L.). J. Plant. Biochem. Biotechnol. 2013, 22, 27–34. [Google Scholar] [CrossRef]
- Prashanth, S.R.; Sadhasivam, V.; Parida, A. Over expression of cytosolic copper/zinc superoxide dismutase from a mangrove plant Avicennia marina in indica rice var Pusa Basmati-1 confers abiotic stress tolerance. Transgenic Res. 2008, 17, 281–291. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trend Plant. Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant. Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Yıldıztugay, E.; Sekmen, A.H.; Turkan, I.; Kucukoduk, M. Elucidation of physiological and biochemical mechanisms of an endemic halophyte Centaurea tuzgoluensis under salt stress. Plant. Physiol. Biochem. 2011, 49, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Akbari, M.; Mahna, N.; Ramesh, K.; Bandehagh, A.; Mazzuca, S. Ion homeostasis, osmoregulation, and physiological changes in the roots and leaves of pistachio rootstocks in response to salinity. Protoplasma 2018, 255, 1349–1362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ma, H.; Chen, T.; Pen, J.; Yu, S.; Zhao, X. Morphological and physiological responses of cotton (Gossypium hirsutum L.) plants to salinity. PLoS ONE 2014, 9, e112807. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, J.Z.; Chow, W.S.; Sun, L.L.; Chen, J.W.; Chen, Y.J.; Peng, C.L. The influence of low temperature on photosynthesis and antioxidant enzymes in sensitive banana and tolerant plantan (Musa sp.) cultivars. Photosynthetica 2011, 49, 201–208. [Google Scholar] [CrossRef]
- Etemadi, N.; Sheikh-Mohammadi, M.H.; Nikbakht, A.; Sabzalian, M.R.; Pessarakli, M. Influence of trinexapac-ethyl in improving drought resistance of wheatgrass and tall fescue. Acta Physiol. Plant. 2015, 37, 53. [Google Scholar] [CrossRef]
- Meloni, D.A.; Oliva, M.A.; Martinez, C.A.; Cambraia, J. Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environ. Exp. Bot. 2003, 49, 69–76. [Google Scholar] [CrossRef]
- Amor, N.B.; Jiménez, A.; Megdiche, W.; Lundqvist, M.; Sevilla, F.; Abdelly, C. Kinetics of the anti-oxidant response to salinity in the halophyte Cakile maritima. J. Integr. Plant. Biol. 2007, 49, 982–992. [Google Scholar] [CrossRef]
- Al-Taweel, K.; Iwaki, T.; Yabuta, Y.; Shigeoka, S.; Murata, N.; Wadano, A. A bacterial transgene for catalase protects translation of D1 protein during exposure of salt-stressed tobacco leaves to strong light. Plant. Physiol. 2007, 145, 258–265. [Google Scholar] [CrossRef]
- Azooz, M.M.; Ismail, A.M.; Elhamd, M.A. Growth, lipid peroxidation and antioxidant enzyme activities as a selection criterion for the salt tolerance of maize cultivars grown under salinity stress. Int. J. Agric. Biol. 2009, 11, 21–26. [Google Scholar]
- Lin, K.H.; Pu, S.F. Tissue-and genotype-specific ascorbate peroxidase expression in sweet potato in response to salt stress. Biol. Plant. 2010, 54, 664–670. [Google Scholar] [CrossRef]
- Diaz-Vivancos, P.; Faize, M.; Barba-Espin, G.; Faize, L.; Petri, C.; Hernández, J.A.; Burgos, L. Ectopic expression of cytosolic superoxide dismutase and ascorbate peroxidase leads to salt stress tolerance in transgenic plums. Plant. Biotechnol. J. 2013, 11, 976–985. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wisniewski, M.; Meilan, R.; Cui, M.; Webb, R.; Fuchigami, L. Overexpression of cytosolic ascorbate peroxidase in tomato confers tolerance to chilling and salt stress. J. Am. Soc. Hortic. Sci. 2005, 130, 167–173. [Google Scholar] [CrossRef]
- Demiral, T.; Türkan, I. Comparative lipid peroxidation, antioxidant defense systems and proline content in roots of two rice cultivars differing in salt tolerance. Environ. Exp. Bot. 2005, 53, 247–257. [Google Scholar] [CrossRef]
- Bandeoğlu, E.; Eyidoğan, F.; Yücel, M.; Öktem, H.A. Antioxidant responses of shoots and roots of lentil to NaCl-salinity stress. Plant. Growth Regul. 2004, 42, 69–77. [Google Scholar] [CrossRef]
- Hamed, K.B.; Castagna, A.; Salem, E.; Ranieri, A.; Abdelly, C. Sea fennel (Crithmum maritimum L.) under salinity conditions: A comparison of leaf and root antioxidant responses. Plant. Growth Regul. 2007, 53, 185–194. [Google Scholar] [CrossRef]
- Hediye Sekmen, A.; Türkan, İ.; Takio, S. Differential responses of anti oxidative enzymes and lipid peroxidation to salt stress in salt-tolerant Plantago maritima and salt-sensitive Plantago Media. Physiol. Plant. 2007, 131, 399–411. [Google Scholar]
- Gharsallah, C.; Fakhfakh, H.; Grubb, D.; Gorsane, F. Effect of salt stress on ion concentration, proline content, antioxidant enzyme activities and gene expression in tomato cultivars. AoB Plants 2016, 8, plw055. [Google Scholar] [CrossRef]
- Cavalcanti, F.R.; Oliveira, J.T.A.; Martins-Miranda, A.S.; Viégas, R.A.; Silveira, J.A.G. Superoxide dismutase, catalase and peroxidase activities do not confer protection against oxidative damage in salt-stressed cowpea leaves. New Phytol. 2004, 163, 563–571. [Google Scholar] [CrossRef]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutases: I. Occurrence in higher plants. Plant. Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Kar, M.; Mishra, D. Catalase, peroxidase, and polyphenoloxidase activities during rice leaf senescence. Plant. Physiol. 1976, 57, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Cakmak, I.; Marschner, H. Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves. Plant. Physiol. 1992, 98, 1222–1227. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant. Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Sgherri, C.L.M.; Navari-Izzo, F. Sunflower seedlings subjected to increasing water deficit stress: Oxidative stress and defense mechanisms. Physiol. Plant. 1995, 93, 25–30. [Google Scholar] [CrossRef]
- Hodges, D.M.; DeLong, J.M.; Forney, C.F.; Prange, R.K. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 1999, 207, 604–611. [Google Scholar] [CrossRef]
- Duncan, D.B. Multiple range and multiple F tests. Biometrics 1955, 11, 1–42. [Google Scholar] [CrossRef]


| Leaf | Leaf | Root | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| APX | CAT | POD | SOD | MDA | GR | APX | CAT | POD | SOD | MDA | GR | |
| APX | 1.00 | 0.99 | 0.98 | 0.83 | 0.97 | −0.99 | 0.99 | |||||
| CAT | 0.56 | 1.00 | 0.51 | 0.55 | 0.35 | 0.45 | −0.56 | 0.58 | ||||
| POD | 0.96 | 0.34 | 1.00 | 0.97 | 0.94 | 0.86 | 0.98 | −0.95 | 0.94 | |||
| SOD | 0.85 | 0.28 | 0.91 | 1.00 | 0.88 | 0.81 | 0.96 | 0.93 | −0.84 | 0.81 | ||
| MDA | −0.99 | −0.55 | −0.95 | −0.85 | 1.00 | −0.96 | −0.96 | −0.86 | −0.95 | 1 | −0.99 | |
| GR | 0.95 | 0.65 | 0.88 | 0.70 | −0.94 | 1.00 | 0.91 | 0.90 | 0.72 | 0.89 | −0.94 | 0.96 |
| Root | ||||||||||||
| APX | 1.00 | |||||||||||
| CAT | 0.97 | 1.00 | ||||||||||
| POD | 0.83 | 0.79 | 1.00 | |||||||||
| SOD | 0.98 | 0.93 | 0.88 | 1.00 | ||||||||
| MDA | −0.98 | −0.97 | −0.83 | −0.95 | 1.00 | |||||||
| GR | 0.96 | 0.96 | 0.80 | 0.94 | −0.99 | 1.00 | ||||||
| Source of Variation | df | Mean Square (Leaf) | Mean Square (Root) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| APX | CAT | POD | SOD | MDA | GR | APX | CAT | POD | SOD | MDA | GR | ||
| Group | 1 | 64.7 * | 70 | 823.6 * | 1127 ** | 0.02 * | 0.007 | 11.71 * | 12.19 | 205 ** | 358.4 * | 0.01 * | 0.025 |
| Error | 3 | 5.49 | 60 | 36.8 | 9.69 | 0.001 | 0.002 | 0.84 | 1.54 | 2.08 | 11.69 | 0.001 | 0.003 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akbari, M.; Katam, R.; Husain, R.; Farajpour, M.; Mazzuca, S.; Mahna, N. Sodium Chloride Induced Stress Responses of Antioxidative Activities in Leaves and Roots of Pistachio Rootstock. Biomolecules 2020, 10, 189. https://doi.org/10.3390/biom10020189
Akbari M, Katam R, Husain R, Farajpour M, Mazzuca S, Mahna N. Sodium Chloride Induced Stress Responses of Antioxidative Activities in Leaves and Roots of Pistachio Rootstock. Biomolecules. 2020; 10(2):189. https://doi.org/10.3390/biom10020189
Chicago/Turabian StyleAkbari, Mohammad, Ramesh Katam, Rabab Husain, Mostafa Farajpour, Silvia Mazzuca, and Nasser Mahna. 2020. "Sodium Chloride Induced Stress Responses of Antioxidative Activities in Leaves and Roots of Pistachio Rootstock" Biomolecules 10, no. 2: 189. https://doi.org/10.3390/biom10020189
APA StyleAkbari, M., Katam, R., Husain, R., Farajpour, M., Mazzuca, S., & Mahna, N. (2020). Sodium Chloride Induced Stress Responses of Antioxidative Activities in Leaves and Roots of Pistachio Rootstock. Biomolecules, 10(2), 189. https://doi.org/10.3390/biom10020189







