Anti-Obesity Effects of Soybean Embryo Extract and Enzymatically-Modified Isoquercitrin
Abstract
1. Introduction
2. Materials and Methods
2.1. Soy Embryo Extract and Enzymatically-Modified Isoquercitrin
2.2. Cell Cutlure
2.3. Animals
2.4. Analysis of Mitochondrial Function
2.5. Western Blot Analysis
2.6. Statistical Analysis
3. Results
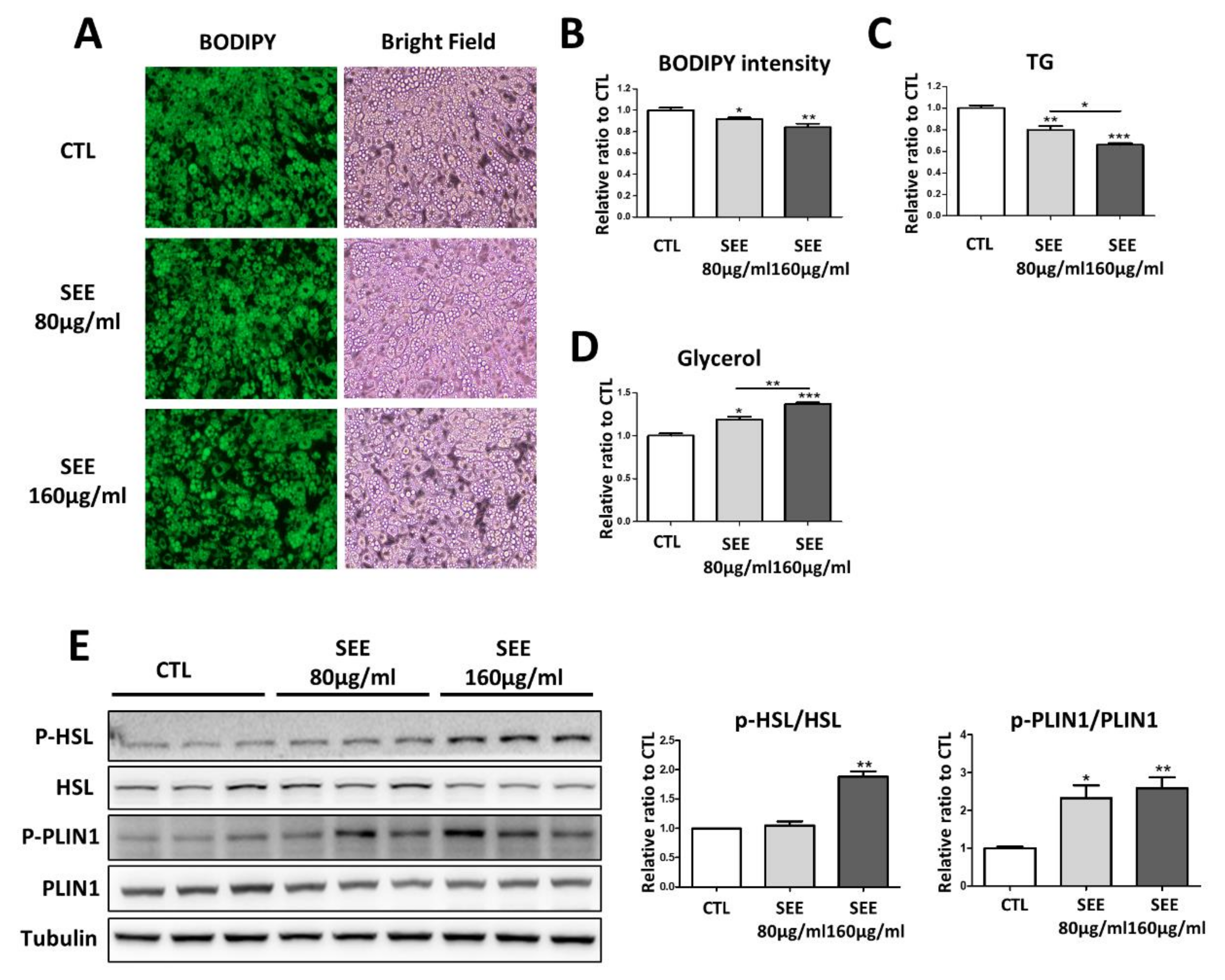
3.1. SEE Reduced Triglyceride Content and Increased Lipolysis in Adipocytes In Vitro
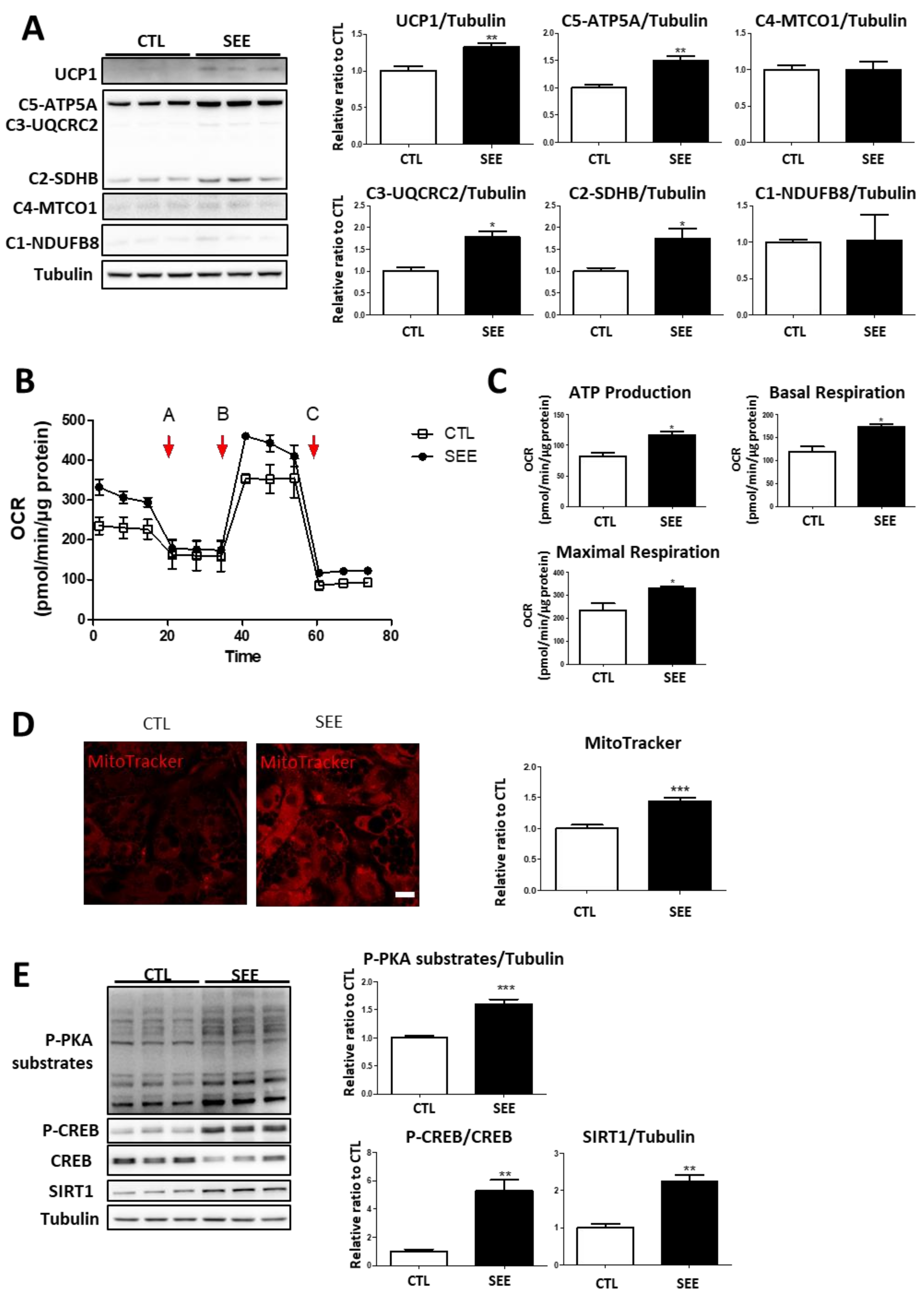
3.2. SEE Upregulated Mitochondrial Content and Respiration via PKA Signaling in Adipocytes In Vitro
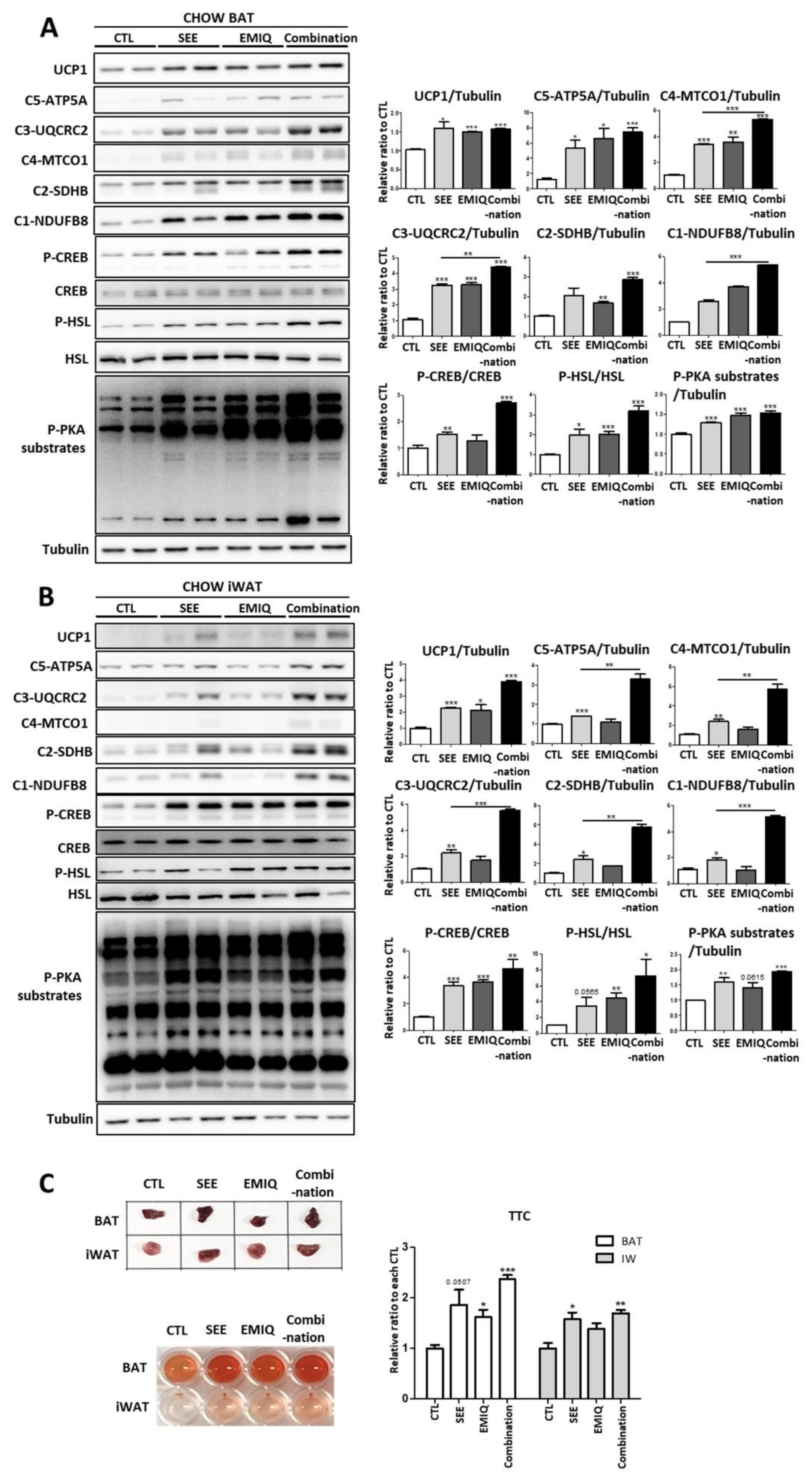
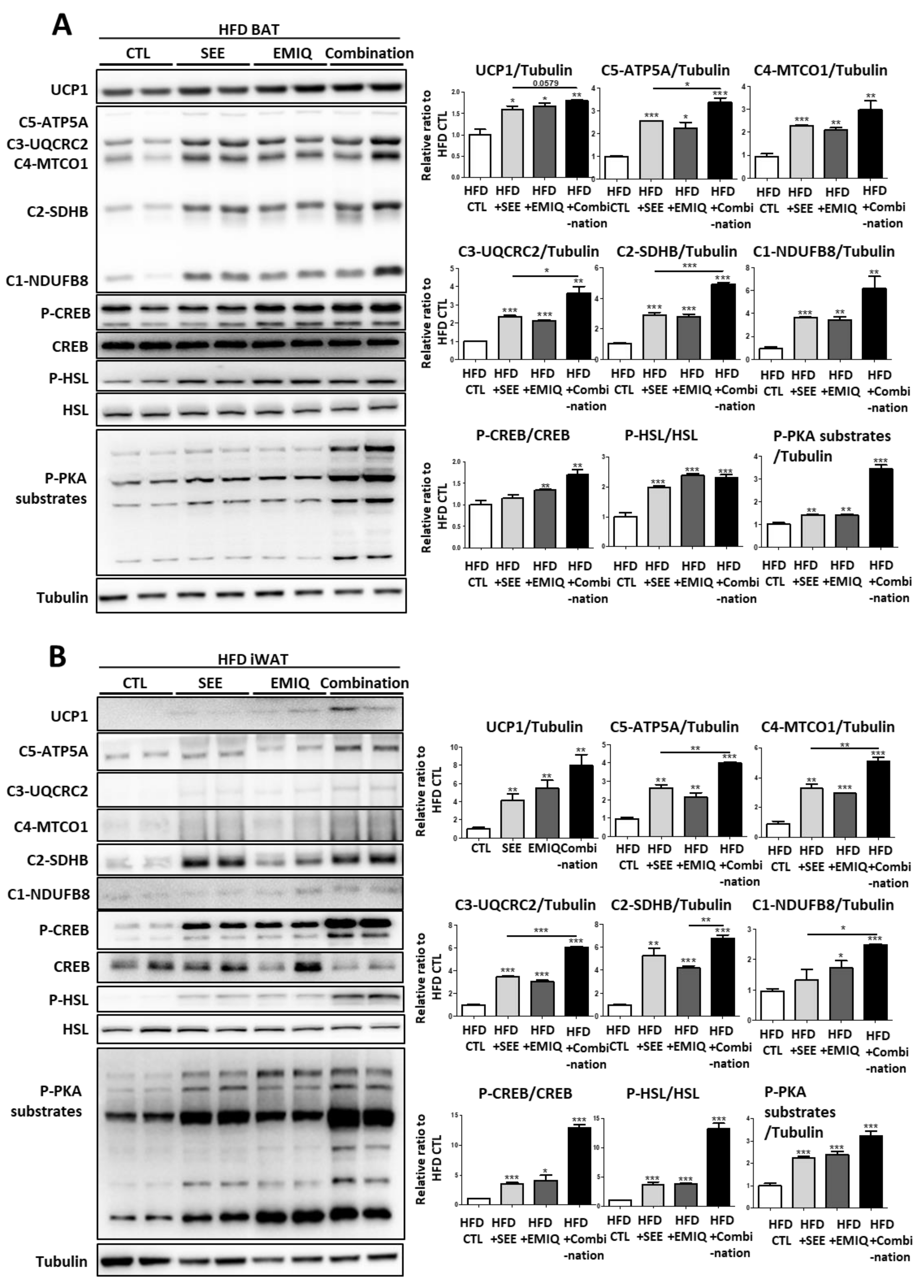
3.3. SEE and EMIQ Had Synergistic Effects on Mitochondrial Content and Activity in Adipose Tissue In Vivo
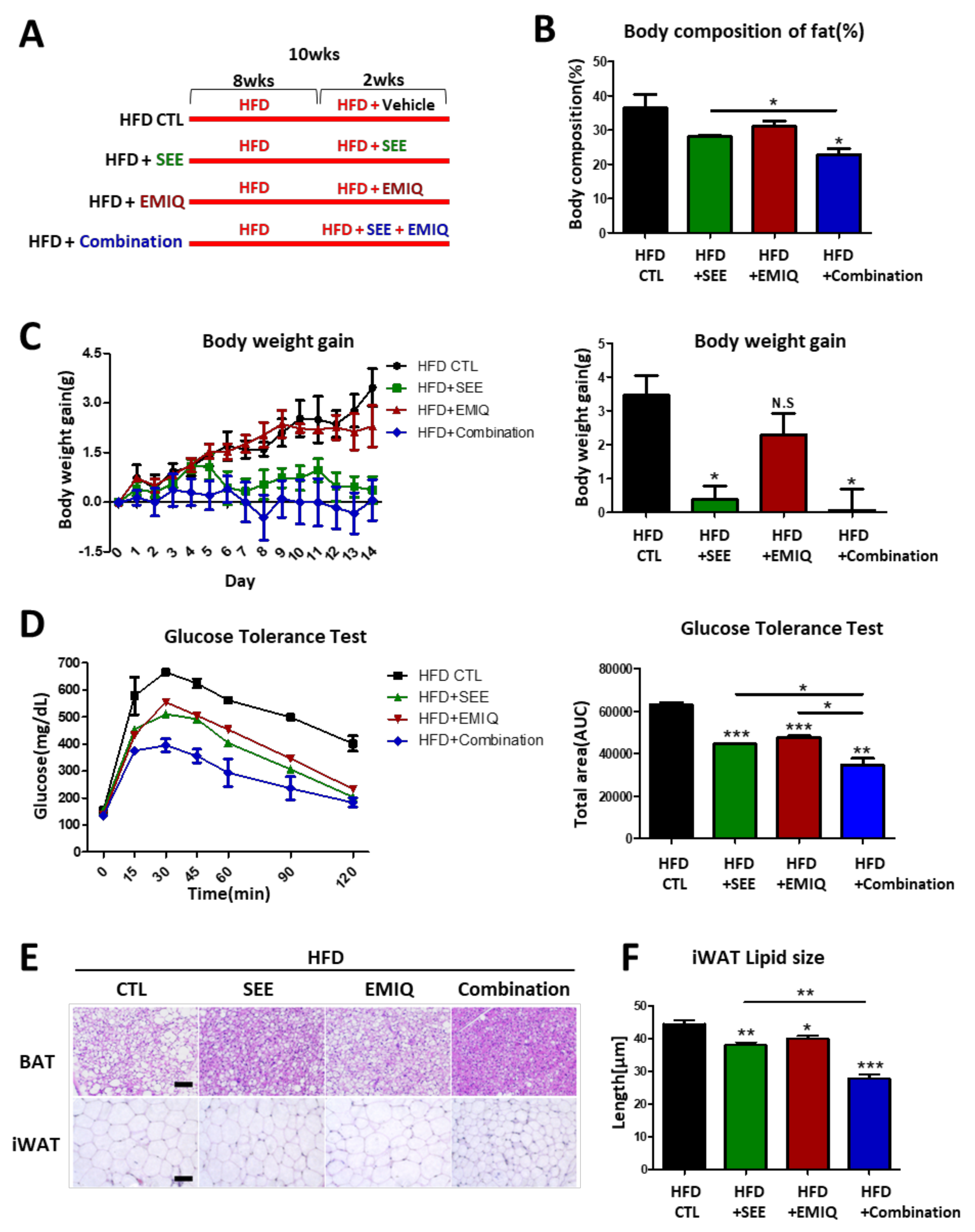
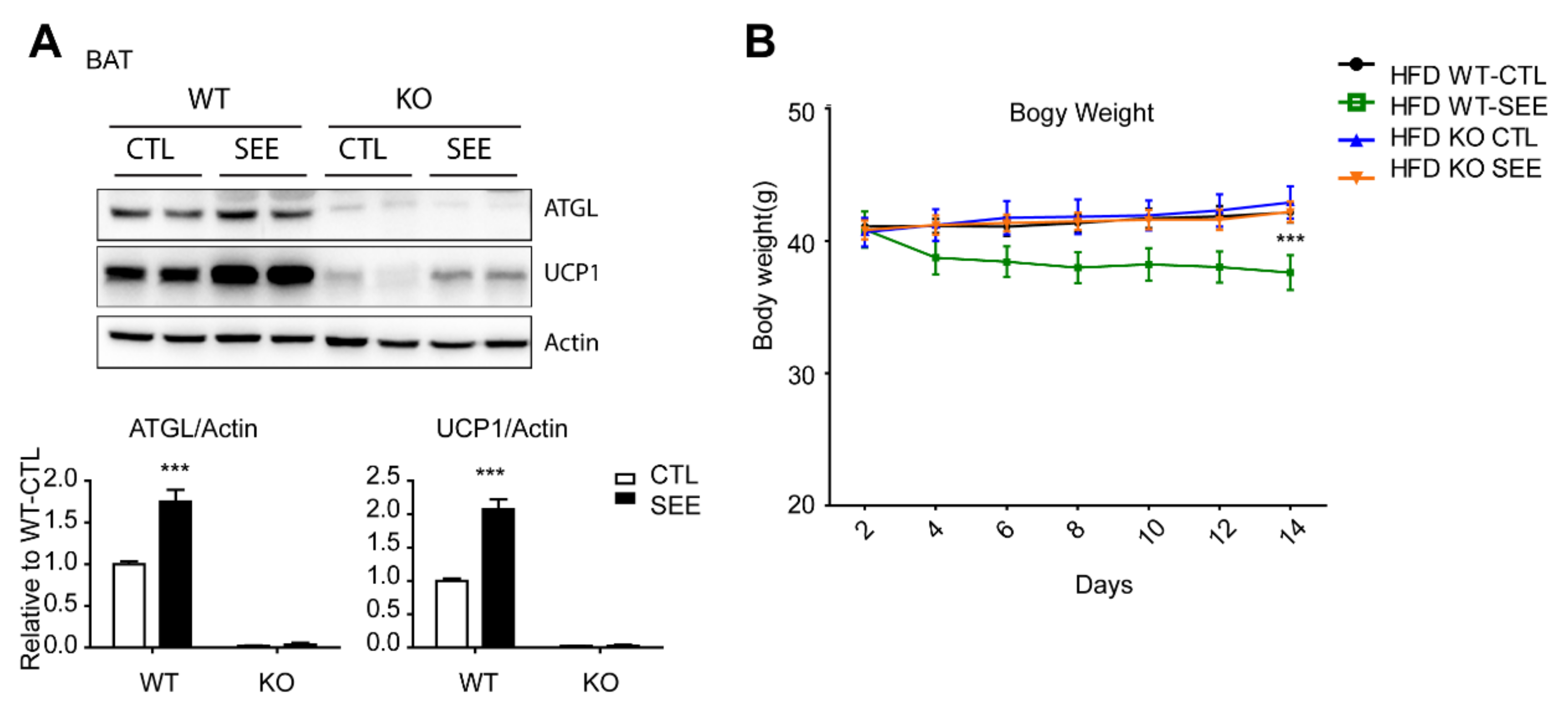
3.4. SEE and EMIQ Had an Anti-Obesity Effect in High-Fat Diet-Induced Obesity Mouse Models
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Azhar, Y.; Parmar, A.; Miller, C.N.; Samuels, J.S.; Rayalam, S. Phytochemicals as novel agents for the induction of browning in white adipose tissue. Nutr. Metab. 2016, 13, 89. [Google Scholar] [CrossRef]
- Srivastava, G.; Apovian, C.M. Current pharmacotherapy for obesity. Nat. Rev. Endocrinol. 2018, 14, 12–24. [Google Scholar] [CrossRef]
- Bartelt, A.; Heeren, J. Adipose tissue browning and metabolic health. Nat. Rev. Endocrinol. 2014, 10, 24–36. [Google Scholar] [CrossRef]
- Lee, Y.-H.; Mottillo, E.P.; Granneman, J.G. Adipose tissue plasticity from wat to bat and in between. Biochim. Biophys. Acta Mol. Basis Dis. 2014, 1842, 358–369. [Google Scholar] [CrossRef]
- Granneman, J.G.; Moore, H.P. Location, location: Protein trafficking and lipolysis in adipocytes. Trends Endocrinol. Metab. 2008, 19, 3–9. [Google Scholar] [CrossRef]
- Carpentier, A.C.; Blondin, D.P.; Virtanen, K.A.; Richard, D.; Haman, F.; Turcotte, E.E. Brown adipose tissue energy metabolism in humans. Front. Endocrinol. 2018, 9, 447. [Google Scholar] [CrossRef]
- Kim, S.N.; Ahn, S.Y.; Song, H.D.; Kwon, H.J.; Saha, A.; Son, Y.; Cho, Y.K.; Jung, Y.S.; Jeong, H.W.; Lee, Y.H. Antiobesity effects of coumestrol through expansion and activation of brown adipose tissue metabolism. J. Nutr. Biochem. 2020, 76, 108300. [Google Scholar] [CrossRef]
- Murosaki, S.; Lee, T.R.; Muroyama, K.; Shin, E.S.; Cho, S.Y.; Yamamoto, Y.; Lee, S.J. A combination of caffeine, arginine, soy isoflavones, and l-carnitine enhances both lipolysis and fatty acid oxidation in 3t3-l1 and hepg2 cells in vitro and in kk mice in vivo. J. Nutr. 2007, 137, 2252–2257. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bhathena, S.J.; Velasquez, M.T. Beneficial role of dietary phytoestrogens in obesity and diabetes. Am. J. Clin. Nutr. 2002, 76, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.R.; Shim, J.; Kim, M.J. Genistin: A novel potent anti-adipogenic and anti-lipogenic agent. Molecules 2020, 25, 2042. [Google Scholar] [CrossRef]
- Dang, Z.-C.; Audinot, V.; Papapoulos, S.E.; Boutin, J.A.; Löwik, C.W. Peroxisome proliferator-activated receptor γ (pparγ) as a molecular target for the soy phytoestrogen genistein. J. Biol. Chem. 2003, 278, 962–967. [Google Scholar] [CrossRef]
- Wang, L.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Blunder, M.; Liu, X.; Malainer, C.; Blazevic, T.; Schwaiger, S.; Rollinger, J.M.; Heiss, E.H.; et al. Natural product agonists of peroxisome proliferator-activated receptor gamma (pparγ): A review. Biochem. Pharmacol. 2014, 92, 73–89. [Google Scholar] [CrossRef]
- Valentová, K.; Vrba, J.; Bancířová, M.; Ulrichová, J.; Křen, V. Isoquercitrin: Pharmacology, toxicology, and metabolism. Food Chem. Toxicol. 2014, 68, 267–282. [Google Scholar] [CrossRef]
- Salim, E.I.; Kaneko, M.; Wanibuchi, H.; Morimura, K.; Fukushima, S. Lack of carcinogenicity of enzymatically modified isoquercitrin in f344/ducrj rats. Food Chem. Toxicol. 2004, 42, 1949–1969. [Google Scholar] [CrossRef]
- Hasumura, M.; Yasuhara, K.; Tamura, T.; Imai, T.; Mitsumori, K.; Hirose, M. Evaluation of the toxicity of enzymatically decomposed rutin with 13-weeks dietary administration to wistar rats. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2004, 42, 439–444. [Google Scholar] [CrossRef]
- Jiang, H.; Yoshioka, Y.; Yuan, S.; Horiuchi, Y.; Yamashita, Y.; Croft, K.D.; Ashida, H. Enzymatically modified isoquercitrin promotes energy metabolism through activating ampkα in male c57bl/6 mice. Food Funct. 2019, 10, 5188–5202. [Google Scholar] [CrossRef]
- Kim, S.N.; Kwon, H.J.; Akindehin, S.; Jeong, H.W.; Lee, Y.H. Effects of epigallocatechin-3-gallate on autophagic lipolysis in adipocytes. Nutrients 2017, 9, 680. [Google Scholar] [CrossRef]
- Lee, Y.H.; Petkova, A.P.; Mottillo, E.P.; Granneman, J.G. In vivo identification of bipotential adipocyte progenitors recruited by beta3-adrenoceptor activation and high-fat feeding. Cell Metab. 2012, 15, 480–491. [Google Scholar] [CrossRef]
- Li, P.; Zhu, Z.; Lu, Y.; Granneman, J.G. Metabolic and cellular plasticity in white adipose tissue ii: Role of peroxisome proliferator-activated receptor-alpha. Am. J. Physiol. Endocrinol. Metab. 2005, 289, E617–E626. [Google Scholar] [CrossRef]
- Son, Y.; Cho, Y.K.; Saha, A.; Kwon, H.J.; Park, J.H.; Kim, M.; Jung, Y.S.; Kim, S.N.; Choi, C.; Seong, J.K.; et al. Adipocyte-specific beclin1 deletion impairs lipolysis and mitochondrial integrity in adipose tissue. Mol. Metab. 2020, 39, 101005. [Google Scholar] [CrossRef]
- Tang, B.L. Sirt1 and the mitochondria. Mol. Cells 2016, 39, 87–95. [Google Scholar] [PubMed]
- Mottillo, E.P.; Balasubramanian, P.; Lee, Y.H.; Weng, C.; Kershaw, E.E.; Granneman, J.G. Coupling of lipolysis and de novo lipogenesis in brown, beige, and white adipose tissues during chronic β3-adrenergic receptor activation. J. Lipid Res. 2014, 55, 2276–2286. [Google Scholar] [CrossRef]
- Li, H.; Qi, J.; Li, L. Phytochemicals as potential candidates to combat obesity via adipose non-shivering thermogenesis. Pharmacol. Res. 2019, 104393. [Google Scholar] [CrossRef] [PubMed]
- Zang, Y.; Igarashi, K.; Yu, C. Anti-obese and anti-diabetic effects of a mixture of daidzin and glycitin on c57bl/6j mice fed with a high-fat diet. Biosci. Biotechnol. Biochem. 2015, 79, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Crespillo, A.; Alonso, M.; Vida, M.; Pavón, F.J.; Serrano, A.; Rivera, P.; Romero-Zerbo, Y.; Fernández-Llebrez, P.; Martínez, A.; Pérez-Valero, V.; et al. Reduction of body weight, liver steatosis and expression of stearoyl-coa desaturase 1 by the isoflavone daidzein in diet-induced obesity. Br. J. Pharmacol. 2011, 164, 1899–1915. [Google Scholar] [CrossRef]
- Kuryłowicz, A.; Cąkała-Jakimowicz, M.; Puzianowska-Kuźnicka, M. Targeting abdominal obesity and its complications with dietary phytoestrogens. Nutrients 2020, 12, 582. [Google Scholar] [CrossRef]
- Chen, M.N.; Lin, C.C.; Liu, C.F. Efficacy of phytoestrogens for menopausal symptoms: A meta-analysis and systematic review. Climacteric 2015, 18, 260–269. [Google Scholar] [CrossRef]
- Jiang, H.; Horiuchi, Y.; Hironao, K.Y.; Kitakaze, T.; Yamashita, Y.; Ashida, H. Prevention effect of quercetin and its glycosides on obesity and hyperglycemia through activating ampkα in high-fat diet-fed icr mice. J. Clin. Biochem. Nutr. 2020, 67, 74–83. [Google Scholar] [CrossRef]
- Paulo, A.; Martins, S.; Branco, P.; Dias, T.; Borges, C.; Rodrigues, A.I.; Costa Mdo, C.; Teixeira, A.; Mota-Filipe, H. The opposing effects of the flavonoids isoquercitrin and sissotrin, isolated from pterospartum tridentatum, on oral glucose tolerance in rats. Phytother. Res. 2008, 22, 539–543. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.; Im, S.; Cho, Y.k.; Choi, C.; Son, Y.; Kwon, D.; Jung, Y.-S.; Lee, Y.-H. Anti-Obesity Effects of Soybean Embryo Extract and Enzymatically-Modified Isoquercitrin. Biomolecules 2020, 10, 1394. https://doi.org/10.3390/biom10101394
Kim M, Im S, Cho Yk, Choi C, Son Y, Kwon D, Jung Y-S, Lee Y-H. Anti-Obesity Effects of Soybean Embryo Extract and Enzymatically-Modified Isoquercitrin. Biomolecules. 2020; 10(10):1394. https://doi.org/10.3390/biom10101394
Chicago/Turabian StyleKim, Minsu, Seowoo Im, Yoon keun Cho, Cheoljun Choi, Yeonho Son, Doyoung Kwon, Young-Suk Jung, and Yun-Hee Lee. 2020. "Anti-Obesity Effects of Soybean Embryo Extract and Enzymatically-Modified Isoquercitrin" Biomolecules 10, no. 10: 1394. https://doi.org/10.3390/biom10101394
APA StyleKim, M., Im, S., Cho, Y. k., Choi, C., Son, Y., Kwon, D., Jung, Y.-S., & Lee, Y.-H. (2020). Anti-Obesity Effects of Soybean Embryo Extract and Enzymatically-Modified Isoquercitrin. Biomolecules, 10(10), 1394. https://doi.org/10.3390/biom10101394




