Melatonin and Its Protective Role against Biotic Stress Impacts on Plants
Abstract
1. Introduction
2. How Plants Defend Themselves against Biotic Stresses
3. Management Approaches of Biotic Stress
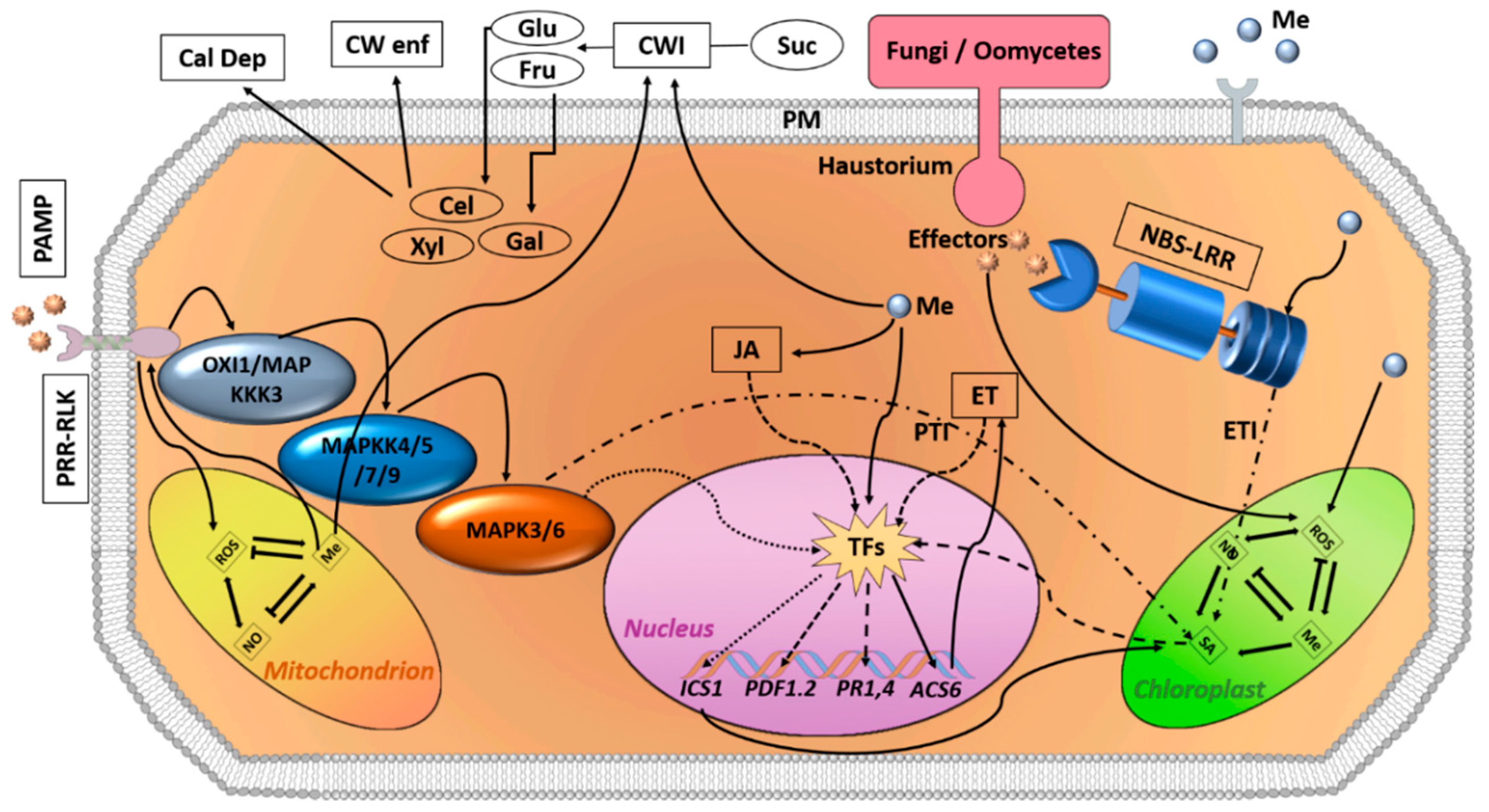
4. Melatonin as an Alleviating Agent against Plant Pathogens
4.1. Melatonin as an Antiviral Agent
4.2. Melatonin Bioactivity against Bacteria
4.3. Antifungal Effect of Melatonin
5. Conclusions and Research Gaps
Funding
Acknowledgments
Conflicts of Interest
References
- Walters, D.R. (Ed.) Why Do Plants Need Defenses? In Plant Defense; Wiley-Blackwell: Edinburgh, UK, 2010; pp. 1–14. [Google Scholar]
- Agrios, G.N. Preface. In Plant Pathology, 5th ed.; Agrios, G.N., Ed.; Academic Press: San Diego, CA, USA, 2005. [Google Scholar]
- Schumann, G.L.; D’Arcy, C.J. Essential Plant Pathology; American Phytopathological Society (APS Press): St. Paul, MI, USA, 2010. [Google Scholar]
- Zaynab, M.; Fatima, M.; Abbas, S.; Sharif, Y.; Umair, M.; Zafar, M.H.; Bahadar, K. Role of secondary metabolites in plant defense against pathogens. Microb. Pathog. 2018, 124, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Monaghan, J.; Zipfel, C. Plant pattern recognition receptor complexes at the plasma membrane. Curr. Opin. Plant Boil. 2012, 15, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Spoel, S.H.; Dong, X. How do plants achieve immunity? Defence without specialized immune cells. Nat. Rev. Immunol. 2012, 12, 89–100. [Google Scholar] [CrossRef] [PubMed]
- De Vleesschauwer, D.; Xu, J.; Hãfte, M.; Höfte, M. Making sense of hormone-mediated defense networking: From rice to Arabidopsis. Front. Plant Sci. 2014, 5, 1–15. [Google Scholar] [CrossRef]
- Grant, M.; Lamb, C. Systemic immunity. Curr. Opin. Plant Boil. 2006, 9, 414–420. [Google Scholar] [CrossRef]
- Loon, L.C.v.; Bakker, P.A.H.M.; Pieterse, C.M.J. Systemic resistance induced by rhizosphere bacteria. Annu. Rev. Phytopathol. 1998, 36, 453–483. [Google Scholar] [CrossRef]
- Santamaria, M.E.; Martínez, M.; Cambra, I.; Grbić, V.; Diaz, I. Understanding plant defence responses against herbivore attacks: An essential first step towards the development of sustainable resistance against pests. Transgenic Res. 2013, 22, 697–708. [Google Scholar] [CrossRef]
- Oerke, E.-C. Estimated crop losses due to pathogens, animal pests and weeds. In Crop Production and Crop Protection; Oerke, E.C., Dehne, H.W., SchÖNbeck, F., Weber, A., Eds.; Elsevier: Amsterdam, The Netherlands, 1999; pp. 72–741. [Google Scholar] [CrossRef]
- Oerke, E.C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Oerke, E.-C.; Dehne, H.-W. Safeguarding production—losses in major crops and the role of crop protection. Crop. Prot. 2004, 23, 275–285. [Google Scholar] [CrossRef]
- Savary, S.; Savary, S.; Zadoks, J.C.; Zadoks, J.C. Analysis of crop loss in the multiple pathosystem groundnut-rust- late leaf spot. I. Six experiments. Crop. Prot. 1992, 11, 99–109. [Google Scholar] [CrossRef]
- Teng, P.S. Crop Loss Assessment and Pest Management; APS Press: St. Paul, MI, USA, 1987; p. 270. [Google Scholar]
- Wyckhuys, K.A.G.; Hughes, A.C.; Buamas, C.; Johnson, A.C.; Vasseur, L.; Reymondin, L.; Deguine, J.-P.; Sheil, D. Biological control of an agricultural pest protects tropical forests. Commun. Boil. 2019, 2, 10. [Google Scholar] [CrossRef] [PubMed]
- Lamichhane, J.R.; Osdaghi, E.; Behlau, F.; Kohl, J.; Jones, J.B.; Aubertot, J.-N. Thirteen decades of antimicrobial copper compounds applied in agriculture. A review. Agron. Sustain. Dev. 2018, 38, 28. [Google Scholar] [CrossRef]
- Fortunati, E.; Mazzaglia, A.; Balestra, G.M. Sustainable control strategies for plant protection and food packaging sectors by natural substances and novel nanotechnological approaches. J. Sci. Food Agric. 2019, 99, 986–1000. [Google Scholar] [CrossRef] [PubMed]
- Lerner, A.B.; Case, J.D.; Takahashi, Y.; Lee, T.H.; Lee, T.H.; Mori, W. Isolation of melatonin, the pineal gland factor that lightens melanocytes1. J. Am. Chem. Soc. 1958, 80, 2587. [Google Scholar] [CrossRef]
- Hattori, A.; Migitaka, H.; Iigo, M.; Itoh, M.; Yamamoto, K.; Ohtani-Kaneko, R.; Hara, M.; Suzuki, T.; Reiter, R.J. Identification of melatonin in plants and its effects on plasma melatonin levels and binding to melatonin receptors in vertebrates. Biochem. Mol. Boil. Int. 1995, 35, 627–634. [Google Scholar]
- Dubbels, R.; Reiter, R.; Klenke, E.; Goebel, A.; Schnakenberg, E.; Ehlers, C.; Schiwara, H.; Schloot, W. Melatonin in edible plants identified by radioimmunoassay and by high performance liquid chromatography-mass spectrometry. J. Pineal Res. 1995, 18, 28–31. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Galano, A. Melatonin: Exceeding Expectations. Physiology 2014, 29, 325–333. [Google Scholar] [CrossRef]
- Zhao, D.; Yu, Y.; Shen, Y.; Liu, Q.; Zhao, Z.; Sharma, R.; Reiter, R.J. Melatonin Synthesis and Function: Evolutionary History in Animals and Plants. Front. Endocrinol. 2019, 10, 249. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin: A New Plant Hormone and/or a Plant Master Regulator? Trends Plant Sci. 2019, 24, 38–48. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin and reactive oxygen and nitrogen species: A model for the plant redox network. Melatonin Res. 2019, 2, 152–168. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. The multi-regulatory properties of melatonin in plants. In Neurotransmitters in Plants; Ramakrishna, A., Roshchina, V., Eds.; CRC Press: New York, NY, USA; Boca Raton, FL, USA, 2018; pp. 71–101. [Google Scholar] [CrossRef]
- Back, K.; Tan, D.-X.; Reiter, R.J. Melatonin biosynthesis in plants: Multiple pathways catalyze tryptophan to melatonin in the cytoplasm or chloroplasts. J. Pineal Res. 2016, 61, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.-X.; Hardeland, R.; Back, K.; Manchester, L.C.; Alatorre-Jimenez, M.A.; Reiter, R.J. On the significance of an alternate pathway of melatonin synthesis via 5-methoxytryptamine: Comparisons across species. J. Pineal Res. 2016, 61, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Dhole, A.M.; Shelat, H.N. Phytomelatonin: A plant hormone for management of stress. J. Anal. Pharm. Res. 2018, 7, 1. [Google Scholar] [CrossRef]
- Kaur, H.; Mukherjee, S.; Baluska, F.; Bhatla, S.C. Regulatory roles of serotonin and melatonin in abiotic stress tolerance in plants. Plant Signal. Behav. 2015, 10, e1049788. [Google Scholar] [CrossRef] [PubMed]
- Vielma, J.R.; Bonilla, E.; Chacín-Bonilla, L.; Mora, M.; Medina-Leendertz, S.; Bravo, Y. Effects of melatonin on oxidative stress, and resistance to bacterial, parasitic, and viral infections: A review. Acta Trop. 2014, 137, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Vico, A.; Lardone, P.J.; Álvarez-Sánchez, N.; Rodríguez-Rodríguez, A.; Guerrero, J.M. Melatonin: Buffering the Immune System. Int. J. Mol. Sci. 2013, 14, 8638–8683. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.M.; Nabavi, S.F.; Sureda, A.; Xiao, J.; Dehpour, A.R.; Shirooie, S.; Silva, A.S.; Baldi, A.; Khan, H.; Daglia, M. Anti-inflammatory effects of Melatonin: A mechanistic review. Crit. Rev. Food Sci. Nutr. 2019, 59, 4–16. [Google Scholar] [CrossRef]
- Regodon, S.; Martín-Palomino, P.; Fernández-Montesinos, R.; Herrera, J.L.; Carrascosa-Salmoral, M.P.; Píriz, S.; Vadillo, S.; Guerrero, J.M.; Pozo, D.; Perez, P.D.D.P. The use of melatonin as a vaccine agent. Vaccine 2005, 23, 5321–5327. [Google Scholar] [CrossRef]
- Huang, S.-H.; Liao, C.-L.; Chen, S.-J.; Shi, L.-G.; Lin, L.; Chen, Y.-W.; Cheng, C.-P.; Sytwu, H.-K.; Shang, S.-T.; Lin, G.-J. Melatonin possesses an anti-influenza potential through its immune modulatory effect. J. Funct. Foods 2019, 58, 189–198. [Google Scholar] [CrossRef]
- Boga, J.A.; Caballero, B.; Potes, Y.; Perez-Martinez, Z.; Reiter, R.J.; Vega-Naredo, I.; Coto-Montes, A. Therapeutic potential of melatonin related to its role as an autophagy regulator: A review. J. Pineal Res. 2019, 66, 12534. [Google Scholar] [CrossRef]
- Zhao, L.; Chen, L.; Gu, P.; Zhan, X.; Zhang, Y.; Hou, C.; Wu, Z.; Wu, Y.; Wang, Q. Exogenous application of melatonin improves plant resistance to virus infection. Plant Pathol. 2019, 68, 1287–1295. [Google Scholar] [CrossRef]
- Chen, L.; Wang, M.; Li, J.; Feng, C.; Cui, Z.; Zhao, L.; Wang, Q. Exogenous application of melatonin improves eradication of apple stem grooving virus from the infected in vitro shoots by shoot tip culture. Plant Pathol. 2019, 68, 997–1006. [Google Scholar] [CrossRef]
- Lee, H.Y.; Byeon, Y.; Tan, D.X.; Reiter, R.J.; Back, K. Arabidopsis serotonin N-acetyltransferase knockout mutant plants exhibit decreased melatonin and salicylic acid levels resulting in susceptibility to an avirulent pathogen. J. Pineal Res. 2015, 58, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Mandal, M.K.; Suren, H.; Ward, B.; Boroujerdi, A.; Kousik, C. Differential roles of melatonin in plant-host resistance and pathogen suppression in cucurbits. J. Pineal Res. 2018, 65, 12505. [Google Scholar] [CrossRef]
- Weeda, S.; Zhang, N.; Zhao, X.; Ndip, G.; Guo, Y.; Buck, G.A.; Fu, C.; Ren, S. Arabidopsis Transcriptome Analysis Reveals Key Roles of Melatonin in Plant Defense Systems. PLoS ONE 2014, 9, e93462. [Google Scholar] [CrossRef]
- Qian, Y.; Tan, D.-X.; Reiter, R.J.; Shi, H. Comparative metabolomic analysis highlights the involvement of sugars and glycerol in melatonin-mediated innate immunity against bacterial pathogen in Arabidopsis. Sci. Rep. 2015, 5, 15815. [Google Scholar] [CrossRef]
- Shi, H.; Chen, Y.; Tan, D.X.; Reiter, R.J.; Chan, Z.; He, C. Melatonin induces nitric oxide and the potential mechanisms relate to innate immunity against bacterial pathogen infection in Arabidopsis. J. Pineal Res. 2015, 59, 102–108. [Google Scholar] [CrossRef]
- Lee, H.Y.; Byeon, Y.; Back, K. Melatonin as a signal molecule triggering defense responses against pathogen attack in Arabidopsisand tobacco. J. Pineal Res. 2014, 57, 262–268. [Google Scholar] [CrossRef]
- Zhao, H.; Xu, L.; Su, T.; Jiang, Y.; Hu, L.; Ma, F. Melatonin regulates carbohydrate metabolism and defenses against Pseudomonas syringae pv. tomato DC3000 infection in Arabidopsis thaliana. J. Pineal Res. 2015, 59, 109–119. [Google Scholar]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin and its relationship to plant hormones. Ann. Bot. 2018, 121, 195–207. [Google Scholar] [CrossRef]
- Lee, H.Y.; Back, K. Mitogen-activated protein kinase pathways are required for melatonin-mediated defense responses in plants. J. Pineal Res. 2016, 60, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Back, K. Melatonin is required for H2O2- and NO-mediated defense signaling through MAPKKK3 and OXI1 in Arabidopsis thaliana. J. Pineal Res. 2017, 62, 12379. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin as a Chemical Substance or as Phytomelatonin Rich-Extracts for Use as Plant Protector and/or Biostimulant in Accordance with EC Legislation. Agronomy 2019, 9, 570. [Google Scholar] [CrossRef]
- Aghdam, M.S.; Fard, J.R. Melatonin treatment attenuates postharvest decay and maintains nutritional quality of strawberry fruits (Fragaria×anannasa cv. Selva) by enhancing GABA shunt activity. Food Chem. 2017, 221, 1650–1657. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Wang, P.; Li, M.; Ke, X.; Li, C.; Liang, N.; Wu, S.; Ma, X.; Li, C.; Zou, Y.; et al. Exogenous melatonin improves Malus resistance to Marssonina apple blotch. J. Pineal Res. 2013, 54, 426–434. [Google Scholar] [CrossRef]
- Tekbaş, Ö.F.; Ogur, R.; Korkmaz, A.; Kilic, A.; Reiter, R.J. Melatonin as an antibiotic: New insights into the actions of this ubiquitous molecule. J. Pineal Res. 2008, 44, 222–226. [Google Scholar]
- Wiid, I.; Helden, E.H.-V.; Hon, D.; Lombard, C.; Van Helden, P. Potentiation of Isoniazid Activity against Mycobacterium tuberculosis by Melatonin. Antimicrob. Agents Chemother. 1999, 43, 975–977. [Google Scholar] [CrossRef]
- Ma, N.; Zhang, J.; Reiter, R.J.; Ma, X. Melatonin mediates mucosal immune cells, microbial metabolism, and rhythm crosstalk: A therapeutic target to reduce intestinal inflammation. Med. Res. Rev. 2019. [Google Scholar] [CrossRef]
- Chen, X.; Sun, C.; Laborda, P.; He, Y.; Zhao, Y.; Li, C.; Liu, F. Melatonin treatments reduce the pathogenicity and inhibit the growth of Xanthomonas oryzae pv. oryzicola. Plant Patho. 2019, 68, 288–296. [Google Scholar] [CrossRef]
- Chen, X.; Sun, C.; Laborda, P.; Zhao, Y.; Palmer, I.; Fu, Z.Q.; Qiu, J.; Liu, F. Melatonin Treatment Inhibits the Growth of Xanthomonas oryzae pv. oryzae. Front. Microbiol. 2018, 9, 2280. [Google Scholar] [CrossRef]
- Nehela, Y.; Killiny, N. Infection with phytopathogenic bacterium inhibits melatonin biosynthesis, decreases longevity of its vector, and suppresses the free radical-defense. J. Pineal Res. 2018, 65, 12511. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Chang, Y.; Zeng, H.; Liu, G.; He, C.; Shi, H. RAV transcription factors are essential for disease resistance against cassava bacterial blight via activation of melatonin biosynthesis genes. J. Pineal Res. 2018, 64, 12454. [Google Scholar] [CrossRef] [PubMed]
- Yavuz, T.; Kaya, D.; Behçet, M.; Ozturk, E.; Yavuz, O. Effects of melatonin on Candida sepsis in an experimental rat model. Adv. Ther. 2007, 24, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Terron, M.P.; Cubero, J.; Barriga, C.; Ortega, E.; Rodriguez, A.B. Phagocytosis of Candida albicans and superoxide anion Levels in ring dove (Streptopelia risoria) heterophils: Effect of melatonin. J. Neuroendocr. 2003, 15, 1111–1115. [Google Scholar] [CrossRef]
- Paredes, S.; Barriga, C.; Ortega, E. Comparative study of the heterophil phagocytic function in young and old ring doves (Streptopelia risoria) and its relationship with melatonin levels. J. Comp. Physiol. B 2004, 174, 421–427. [Google Scholar]
- Cengiz, M.I.; Cengiz, S.; Wang, H.-L. Melatonin and Oral Cavity. Int. J. Dent. 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Liu, C.; Chen, L.; Zhao, R.; Li, R.; Zhang, S.; Yu, W.; Sheng, J.; Shen, L. Melatonin Induces Disease Resistance to Botrytis cinerea in Tomato Fruit by Activating Jasmonic Acid Signaling Pathway. J. Agric. Food Chem. 2019, 67, 6116–6124. [Google Scholar] [CrossRef]
- Zhang, S.; Zheng, X.; Reiter, R.J.; Feng, S.; Wang, Y.; Liu, S.; Jin, L.; Li, Z.; Datla, R.; Ren, M. Melatonin Attenuates Potato Late Blight by Disrupting Cell Growth, Stress Tolerance, Fungicide Susceptibility and Homeostasis of Gene Expression in Phytophthora infestans. Front. Plant Sci. 2017, 8, 1993. [Google Scholar] [CrossRef]
- Chang, F.; Cao, X.; Peng, J.; Tan, X.; Chen, T.; Zeng, J.; Cao, Y.; Qiao, D. Induced resistance to Plasmodiophora brassicae in Arabidopsis by melatonin. Chin. J. Appl. Environ. Bio. 2018, 24, 75–80. [Google Scholar]
- Sun, Y.; Liu, Z.; Lan, G.; Jiao, C.; Sun, Y. Effect ofexogenous melatonin on resistance of cucumber to downy mildew. Sci. Hortic. 2019, 255, 231–241. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, S.; Zhang, J.; Reiter, R.J.; Wang, Y.; Qiu, D.; Luo, X.; Khalid, A.R.; Wang, H.; Feng, L.; et al. Synergistic anti-oomycete effect of melatonin with a biofungicide against oomycetic black shank disease. J. Pineal Res. 2018, 65, 12492. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhao, Q.; Gao, T.; Wang, H.; Zhang, Z.; Liang, B.; Wei, Z.; Liu, C.; Ma, F. The mitigation effects of exogenous melatonin on replant disease in apple. J. Pineal Res. 2018, 65, 12523. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B.; Hernández-Ruiz, J. Functions of melatonin in plants: A review. J. Pineal Res. 2015, 59, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Jiao, J.; Ma, Y.; Chen, S.; Liu, C.; Song, Y.; Qin, Y.; Yuan, C.; Liu, Y. Melatonin-Producing Endophytic Bacteria from Grapevine Roots Promote the Abiotic Stress-Induced Production of Endogenous Melatonin in Their Hosts. Front. Plant Sci. 2016, 7, 542. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Jiao, J.; Fan, X.; Sun, H.; Zhang, Y.; Jiang, J.; Liu, C. Endophytic Bacterium Pseudomonas fluorescens RG11 May Transform Tryptophan to Melatonin and Promote Endogenous Melatonin Levels in the Roots of Four Grape Cultivars. Front. Plant Sci. 2017, 7, 305. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, X.; Chen, T.; Ji, Y.; Shi, K.; Wang, L.; Zheng, X.; Kong, J. Melatonin in Apples and Juice: Inhibition of Browning and Microorganism Growth in Apple Juice. Molecules 2018, 23, 521. [Google Scholar] [CrossRef]
- Lin, Y.; Fan, L.; Xia, X.; Wang, Z.; Yin, Y.; Cheng, Y.; Li, Z. Melatonin decreases resistance to postharvest green mold on citrus fruit by scavenging defense-related reactive oxygen species. Postharvest Boil. Technol. 2019, 153, 21–30. [Google Scholar] [CrossRef]
- Wei, Y.; Hu, W.; Wang, Q.; Zeng, H.; Li, X.; Yan, Y.; Reiter, R.J.; He, C.; Shi, H. Identification, transcriptional and functional analysis of heat-shock protein 90s in banana (Musa acuminata L.) highlight their novel role in melatonin-mediated plant response to Fusarium wilt. J. Pineal Res. 2017, 62, 12367. [Google Scholar] [CrossRef]
- Wei, J.; Li, D.-X.; Zhang, J.-R.; Shan, C.; Rengel, Z.; Song, Z.-B.; Chen, Q. Phytomelatonin receptor PMTR1-mediated signaling regulates stomatal closure in Arabidopsis thaliana. J. Pineal Res. 2018, 65, 12500. [Google Scholar] [CrossRef]
- Zhan, H.; Nie, X.; Zhang, T.; Li, S.; Wang, X.; Du, X.; Tong, W.; Song, W. Melatonin: A Small Molecule but Important for Salt Stress Tolerance in Plants. Int. J. Mol. Sci. 2019, 20, 709. [Google Scholar] [CrossRef]
- Almoneafy, A.A.; Kakar, K.U.; Nawaz, Z.; Li, B.; Saand, M.A.; Chun-Lan, Y.; Xie, G.-L. Tomato plant growth promotion and antibacterial related-mechanisms of four rhizobacterial Bacillus strains against Ralstonia solanacearum. Symbiosis 2014, 63, 59–70. [Google Scholar] [CrossRef]
- Almoneafy, A.A.; Ojaghian, M.R.; Seng-Fu, X.; Ibrahim, M.; Guan-Lin, X.; Yu, S.; Wen-Xiao, T.; Bin, L. Synergistic effect of acetyl salicylic acid and DL-Beta-aminobutyric acid on biocontrol efficacy of Bacillus strains against tomato bacterial wilt. Trop. Plant Pathol. 2013, 38, 102–113. [Google Scholar] [CrossRef]
- Kakar, K.U.; Duan, Y.P.; Nawaz, Z.; Sun, G.; Almoneafy, A.A.; Hassan, M.A.; Elshakh, A.; Li, B.; Xie, G.-L. A novel rhizobacterium Bk7 for biological control of brown sheath rot of rice caused by Pseudomonas fuscovaginae and its mode of action. J. Plant Pathol. 2014, 138, 819–834. [Google Scholar] [CrossRef]
- Liu, T.; Zhao, F.; Liu, Z.; Zuo, Y.; Hou, J.; Wang, Y. Identification of melatonin in Trichoderma spp. and detection of melatonin content under controlled-stress growth conditions from T. asperellum. J. Basic Micro. 2016, 56, 838–843. [Google Scholar] [CrossRef]
| Plant Name | Pathogen Name | Melatonin Dosage (µM) | Delivery Method | Involved Mechanism | Resulting Effect | Ref. |
|---|---|---|---|---|---|---|
| Nicotiana glutinosa and Solanum lycopersicum | Tobacco mosaic virus (TMV) | 100 (twice) | Root irrigation | Salicylic acid (SA) concentrations lead to increased expression of PR1 and PR5 genes | Reduction of virus concentration in infected plants | [37] |
| Malus domestica | Apple stem grooving virus (ASGV) | 15 | To the shoot proliferation medium | Not reconnoitered | Eradication of virus from previously infected shoot tips | [38] |
| Plant | Pathogen | Melatonin Dosage (µM) | Delivery Method | Mechanism | Effect | Ref. |
|---|---|---|---|---|---|---|
| Arabidopsis thaliana, Nicotiana benthamiana | Pseudomonas syringae | 1 or 10 | Leaf treatment | ▲ Expression of defense genes PR1, PR5, and PDF1.2 | Inhibition of pathogen propagation | [44] |
| A. thaliana | P. syringae | 10 | Leaf treatment | ▲ Pathogenesis-related (PR) genes by the harmonizing signaling between SA and ET | Increase resistance against pathogen | [39] |
| A. thaliana, N. benthamiana | P. syringae | 1 | Leaf treatment | ▲ Induction of PR genes through MAPK signaling cascades | Disease resistance | [47] |
| A. thaliana | P. syringae | 20 | Added to nutrient solution | Involvement of sugars and glycerol in melatonin-mediated innate immunity in SA- and NO-dependent pathways | Disease resistance | [42] |
| A. thaliana | P. syringae | 20 | Added to nutrient solution | ▲ NO and melatonin levels in leaves ▲ Defense-related genes | Improvement of disease resistance | [43] |
| A. thaliana | P. syringae | 50 | Added to plant culture medium | ▲ Activities of CWI and vacuolar invertase (VI) | Cell-wall reinforcement and callose deposition during infection | [45] |
| A. thaliana | P. syringae | 1 | Leaf treatment | Induction of PR1 and ICS1 expression genes through MAPK cascades in coexistence H2O2 and NO | Disease resistance | [48] |
| Plant Name | Pathogen Name | Melatonin Dosage (µM) | Delivery Method | Involved Mechanism | Resulting Effect | Ref. |
|---|---|---|---|---|---|---|
| Malus prunifolia | Diplocarpon mali | 50–500 | Root irrigation | Maintain intracellular H2O2 concentrations ▲ Activities of plant defense-related enzymes | Alleviating disease damage Fungal infection resistance Lesion reduction | [51] |
| Musa acuminata | Fusarium oxysporum | 100 | Leaf and root treatment | ▲ Resistance induced via regulating the expression of MaHSP90s gene | Improvement of disease resistance | [74] |
| Fragaria ananassa | Botrytis cinerea and Rhizopus stolonifer | 100 | Fruit dipping | ▲ H2O2 levels ▲ Antioxidant enzyme activities | Reduction of postharvest decay in stored strawberry fruits | [50] |
| Citrullus lanatus | Podosphaera xanthii and Phythophthora capsici | 100 | Leaf treatment | ▲ Upregulation of PTI- and ETI-associated genes | Disease resistance | [40] |
| A. thaliana | -------- | 1 × 10−4 or 100 | Seedling rinsing | ▲ Upregulation of genes involved in abscisic acid (ABA), ET, SA, and JA pathways | Increase plant resilience to biotic and abiotic stress. | [41] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moustafa-Farag, M.; Almoneafy, A.; Mahmoud, A.; Elkelish, A.; Arnao, M.B.; Li, L.; Ai, S. Melatonin and Its Protective Role against Biotic Stress Impacts on Plants. Biomolecules 2020, 10, 54. https://doi.org/10.3390/biom10010054
Moustafa-Farag M, Almoneafy A, Mahmoud A, Elkelish A, Arnao MB, Li L, Ai S. Melatonin and Its Protective Role against Biotic Stress Impacts on Plants. Biomolecules. 2020; 10(1):54. https://doi.org/10.3390/biom10010054
Chicago/Turabian StyleMoustafa-Farag, Mohamed, Abdulwareth Almoneafy, Ahmed Mahmoud, Amr Elkelish, Marino B. Arnao, Linfeng Li, and Shaoying Ai. 2020. "Melatonin and Its Protective Role against Biotic Stress Impacts on Plants" Biomolecules 10, no. 1: 54. https://doi.org/10.3390/biom10010054
APA StyleMoustafa-Farag, M., Almoneafy, A., Mahmoud, A., Elkelish, A., Arnao, M. B., Li, L., & Ai, S. (2020). Melatonin and Its Protective Role against Biotic Stress Impacts on Plants. Biomolecules, 10(1), 54. https://doi.org/10.3390/biom10010054








