Pharmacometabolomics of Bronchodilator Response in Asthma and the Role of Age-Metabolite Interactions
Abstract
:1. Introduction
2. Results
2.1. Study Population
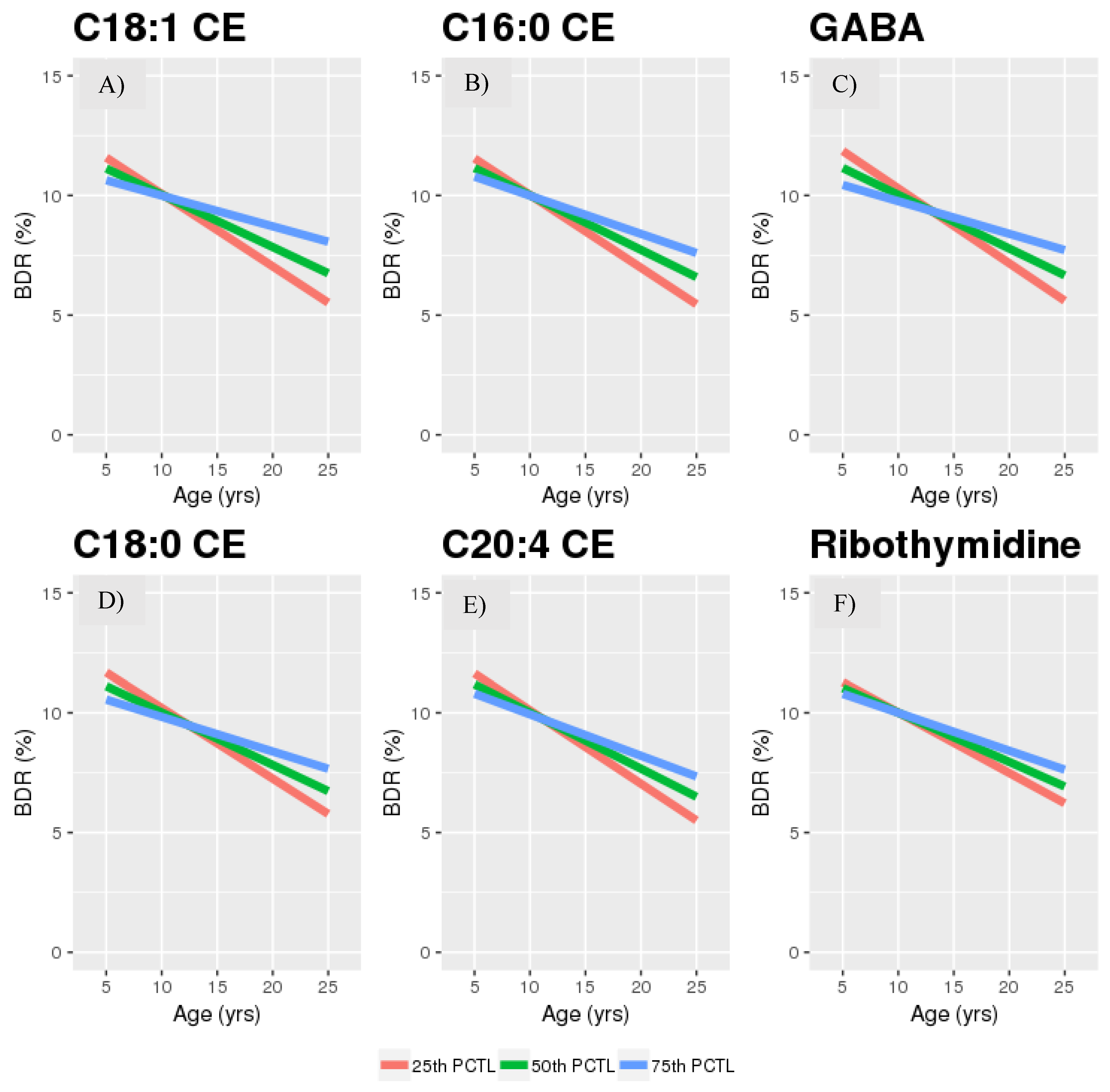
2.2. Age*Metabolite Interactions
2.3. Sensitivity Analyses
3. Discussion
4. Materials and Methods
4.1. Study Population
4.1.1. Discovery Population
4.1.2. Replication Population
4.2. Spirometry
4.3. Metabolomic Profiling
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- The Global Asthma Report 2018; Global Asthma Network: Auckland, New Zealand, 2018; Available online: http://www.globalasthmareport.org/ (accessed on 1 May 2019).
- Guidelines for the Diagnosis and Managment of Asthma. In National Asthma Education and Prevention Program, Third Expert Panel on the Diagnosis and Management of Asthma; National Heart Lung and Blood Institute: Bethesda, MD, USA, 2007; Report No: 07-4051. Available online: https://www.ncbi.nlm.nih.gov/books/NBK7232/ (accessed on 1 May 2019).
- Kaddurah-Daouk, R.; Weinshilboum, R.M. Pharmacometabolomics Research N: Pharmacometabolomics: Implications for clinical pharmacology and systems pharmacology. Clin. Pharmacol. Ther. 2014, 95, 154–167. [Google Scholar] [CrossRef] [PubMed]
- Sordillo, J.E.; McGeachie, M.; Lutz, S.M.; Lasky-Su, J.; Tantisira, K.; Tsai, C.H.; Dahlin, A.; Kelly, R.; Wu, A.C. Longitudinal analysis of bronchodilator response in asthmatics and effect modification of age-related trends by genotype. Pediatr. Pulmonol. 2019, 54, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Galant, S.P.; Morphew, T.; Newcomb, R.L.; Hioe, K.; Guijon, O.; Liao, O. The relationship of the bronchodilator response phenotype to poor asthma control in children with normal spirometry. J. Pediatr. 2011, 158, 953–959 e951. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.L.; Wu, C.L.; Lu, Y.T. Effects of Age on 1-Second Forced Expiratory Volume Response to Bronchodilation. Int. J. Gerontol. 2009, 3, 149–155. [Google Scholar] [CrossRef] [Green Version]
- Anthonisen, N.R.; Lindgren, P.G.; Tashkin, D.P.; Kanner, R.E.; Scanlon, P.D.; Connett, J.E. Lung Health Study Research G: Bronchodilator response in the lung health study over 11 yrs. Eur. Respir. J. 2005, 26, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Arshad, S.H.; Raza, A.; Lau, L.; Bawakid, K.; Karmaus, W.; Zhang, H.; Ewart, S.; Patil, V.; Roberts, G.; Kurukulaaratchy, R. Pathophysiological characterization of asthma transitions across adolescence. Respir. Res. 2014, 15, 153. [Google Scholar] [CrossRef] [PubMed]
- Oldham, W.M.; Clish, C.B.; Yang, Y.; Loscalzo, J. Hypoxia-mediated increases in L-2-hydroxyglutarate coordinate the metabolic response to reductive stress. Cell Metab. 2015, 22, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Miric, M.; Ristic, S.; Joksimovic, B.N.; Medenica, S.; Racic, M.; Ristic, S.; Joksimovic, V.R.; Skipina, M. Reversion of methacholine induced bronchoconstriction with inhaled diazepam in patients with asthma. Rev. Med. Chil. 2016, 144, 434–441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forkuo, G.S.; Guthrie, M.L.; Yuan, N.Y.; Nieman, A.N.; Kodali, R.; Jahan, R.; Stephen, M.R.; Yocum, G.T.; Treven, M.; Poe, M.M.; et al. Development of GABAA receptor subtype-selective imidazobenzodiazepines as novel asthma treatments. Mol. Pharm. 2016, 13, 2026–2038. [Google Scholar] [CrossRef] [PubMed]
- Wendell, S.G.; Baffi, C.; Holguin, F. Fatty acids, inflammation, and asthma. J. Allergy Clin. Immunol. 2014, 133, 1255–1264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tall, A.R.; Yvan-Charvet, L. Cholesterol, inflammation and innate immunity. Nat. Rev. Immunol. 2015, 15, 104–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montecino-Rodriguez, E.; Berent-Maoz, B.; Dorshkind, K. Causes, consequences, and reversal of immune system aging. J. Clin. Investig. 2013, 123, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Lopez, A.; Krehl, W.A.; Hodges, R.E. Relationship between total cholesterol and cholesteryl esters with age in human blood plasma. Am. J. Clin. Nutr. 1967, 20, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Yde, C.C.; Arnberg, K.; Molgaard, C.; Michaelsen, K.F.; Larnkjaer, A.; Bertram, H.C. NMR-based metabolomic profiling of overweight adolescents: An elucidation of the effects of inter-/intraindividual differences, gender, and pubertal development. Biomed. Res. Int. 2014, 2014, 537157. [Google Scholar] [CrossRef] [PubMed]
- Weir, J.M.; Wong, G.; Barlow, C.K.; Greeve, M.A.; Kowalczyk, A.; Almasy, L.; Comuzzie, A.G.; Mahaney, M.C.; Jowett, J.B.; Shaw, J.; et al. Plasma lipid profiling in a large population-based cohort. J. Lipid Res. 2013, 54, 2898–2908. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelly, R.S.; Dahlin, A.; McGeachie, M.J.; Qiu, W.; Sordillo, J.; Wan, E.S.; Wu, A.C.; Lasky-Su, J. Asthma metabolomics and the potential for integrative omics in research and the clinic. Chest 2017, 151, 262–277. [Google Scholar] [CrossRef] [PubMed]
- Haid, M.; Muschet, C.; Wahl, S.; Romisch-Margl, W.; Prehn, C.; Moller, G.; Adamski, J. Long-term stability of human plasma metabolites during storage at −80 degrees C. J. Proteome Res. 2018, 17, 203–211. [Google Scholar] [CrossRef]
- Breier, M.; Wahl, S.; Prehn, C.; Fugmann, M.; Ferrari, U.; Weise, M.; Banning, F.; Seissler, J.; Grallert, H.; Adamski, J.; et al. Targeted metabolomics identifies reliable and stable metabolites in human serum and plasma samples. PLoS ONE 2014, 9, e89728. [Google Scholar] [CrossRef]
- Forno, E.; Lasky-Su, J.; Himes, B.; Howrylak, J.; Ramsey, C.; Brehm, J.; Klanderman, B.; Ziniti, J.; Melen, E.; Pershagen, G.; et al. Genome-wide association study of the age of onset of childhood asthma. J. Allergy Clin. Immunol. 2012, 130, 83–90 e84. [Google Scholar] [CrossRef]
- Himes, B.E.; Lasky-Su, J.; Wu, A.C.; Wilk, J.B.; Hunninghake, G.M.; Klanderman, B.; Murphy, A.J.; Lazarus, R.; Soto-Quiros, M.E.; Avila, L.; et al. Asthma-susceptibility variants identified using probands in case-control and family-based analyses. BMC Med. Genet. 2010, 11, 122. [Google Scholar] [CrossRef]
- Hunninghake, G.M.; Lasky-Su, J.; Soto-Quiros, M.E.; Avila, L.; Liang, C.; Lake, S.L.; Hudson, T.J.; Spesny, M.; Fournier, E.; Sylvia, J.S.; et al. Sex-stratified linkage analysis identifies a female-specific locus for IgE to cockroach in Costa Ricans. Am. J. Respir. Crit. Care. Med. 2008, 177, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Lasky-Su, J.; Lange, N.; Brehm, J.M.; Damask, A.; Soto-Quiros, M.; Avila, L.; Celedon, J.C.; Canino, G.; Cloutier, M.M.; Hollis, B.W.; et al. Genome-wide association analysis of circulating vitamin D levels in children with asthma. Hum. Genet. 2012, 131, 1495–1505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelly, R.S.; Chawes, B.L.; Blighe, K.; Virkud, Y.V.; Croteau-Chonka, D.C.; McGeachie, M.J.; Clish, C.B.; Bullock, K.; Celedon, J.C.; Weiss, S.T.; et al. An integrative transcriptomic and metabolomic study of lung function in children with asthma. Chest 2018, 154, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Kastenmüller, G.; He, Y.; Belcredi, P.; Möller, G.; Prehn, C.; Mendes, J.; Wahl, S.; Roemisch-Margl, W.; Ceglarek, U.; et al. Differences between Human Plasma and Serum Metabolite Profiles. PLoS ONE 2011, 6, e21230. [Google Scholar] [CrossRef] [PubMed]
- Bowler, R.P.; Wendt, C.H.; Fessler, M.B.; Foster, M.W.; Kelly, R.S.; Lasky-Su, J.; Rogers, A.J.; Stringer, K.A.; Winston, B.W.; American Thoracic Society Workgroup on M; et al. New Strategies and Challenges in Lung Proteomics and Metabolomics. An Official American Thoracic Society Workshop Report. Ann. Am. Thorac. Soc. 2017, 14, 1721–1743. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.S.; Lasky-Su, J. Response. Chest 2018, 153, 1283–1284. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.S.; McGeachie, M.J.; Lee-Sarwar, K.A.; Kachroo, P.; Chu, S.H.; Virkud, Y.V.; Huang, M.; Litonjua, A.A.; Weiss, S.T.; Lasky-Su, J. Partial least squares discriminant analysis and bayesian networks for metabolomic prediction of childhood asthma. Metabolites 2018, 8, 68. [Google Scholar] [CrossRef]
- Kelly, R.S.; Sordillo, J.E.; Lasky-Su, J.; Dahlin, A.; Perng, W.; Rifas-Shiman, S.L.; Weiss, S.T.; Gold, D.R.; Litonjua, A.A.; Hivert, M.F.; et al. Plasma metabolite profiles in children with current asthma. Clin. Exp. Allergy 2018, 48, 1297–1304. [Google Scholar] [CrossRef]
- Pite, H.; Morais-Almeida, M.; Rocha, S.M. Metabolomics in asthma: Where do we stand? Curr. Opin. Pulm. Med. 2018, 24, 94–103. [Google Scholar] [CrossRef]
- Nambiar, S.; Bong How, S.; Gummer, J.; Trengove, R.; Moodley, Y. Metabolomics in chronic lung diseases. Respirology 2019. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]
- The Childhood Asthma Management Program Research Group. The childhood asthma management program (CAMP): Design, rationale, and methods. Control. Clin. Trials 1999, 20, 91–120. [Google Scholar] [CrossRef]
- Kelly, R.S.; Virkud, Y.; Giorgio, R.; Celedon, J.C.; Weiss, S.T.; Lasky-Su, J. Metabolomic profiling of lung function in Costa-Rican children with asthma. Biochim. Biophys Acta Mol. Basis Dis. 2017, 1863, 1590–1595. [Google Scholar] [CrossRef] [PubMed]
- Townsend, M.K.; Clish, C.B.; Kraft, P.; Wu, C.; Souza, A.L.; Deik, A.A.; Tworoger, S.S.; Wolpin, B.M. Reproducibility of metabolomic profiles among men and women in 2 large cohort studies. Clin. Chem. 2013, 59, 1657–1667. [Google Scholar] [CrossRef] [PubMed]
- Mascanfroni, I.D.; Takenaka, M.C.; Yeste, A.; Patel, B.; Wu, Y.; Kenison, J.E.; Siddiqui, S.; Basso, A.S.; Otterbein, L.E.; Pardoll, D.M.; et al. Metabolic control of type 1 regulatory T cell differentiation by AHR and HIF1-alpha. Nat. Med. 2015, 21, 638–646. [Google Scholar] [CrossRef]
- O’Sullivan, J.F.; Morningstar, J.E.; Yang, Q.; Zheng, B.; Gao, Y.; Jeanfavre, S.; Scott, J.; Fernandez, C.; Zheng, H.; O’Connor, S.; et al. Dimethylguanidino valeric acid is a marker of liver fat and predicts diabetes. J. Clin. Investig. 2017, 127, 4394–4402. [Google Scholar]
- Rowan, S.; Jiang, S.; Korem, T.; Szymanski, J.; Chang, M.L.; Szelog, J.; Cassalman, C.; Dasuri, K.; McGuire, C.; Nagai, R.; et al. Involvement of a gut-retina axis in protection against dietary glycemia-induced age-related macular degeneration. Proc. Natl. Acad. Sci. USA 2017, 114, E4472–E4481. [Google Scholar] [CrossRef]
- Paynter, N.P.; Balasubramanian, R.; Giulianini, F.; Wang, D.D.; Tinker, L.F.; Gopal, S.; Deik, A.A.; Bullock, K.; Pierce, K.A.; Scott, J.; et al. Metabolic predictors of incident coronary heart disease in women. Circulation 2018, 137, 841–853. [Google Scholar] [CrossRef]

| Baseline (n = 560) | Study End (n = 563) | Follow-up (n = 295) | |||||
|---|---|---|---|---|---|---|---|
| Characteristic | n | % | n | % | n | % | |
| Sex | Male | 359 | 64.1% | 357 | 63.4% | 189 | 64.1% |
| Female | 201 | 35.9% | 206 | 36.6% | 106 | 35.9% | |
| Race | White | 395 | 70.5% | 399 | 70.9% | 213 | 72.2% |
| Black | 82 | 14.6% | 81 | 14.4% | 35 | 11.9% | |
| Hispanic | 56 | 10.0% | 56 | 9.9% | 21 | 7.1% | |
| Other | 27 | 4.8% | 27 | 4.8% | 26 | 8.8% | |
| Treatment Group | Budesonide | 151 | 27.0% | 156 | 27.7% | 78 | 26.4% |
| Nedocromil | 171 | 30.5% | 169 | 30.0% | 83 | 28.1% | |
| Placebo | 238 | 42.5% | 238 | 42.3% | 134 | 45.4% | |
| Age at blood sample | mean (SD) [range] | 8.8 (2.1) | [5.1, 13.2] | 12.8 [2.2] | [9.1, 17.2] | 16.8 (2.9) | [12.2, 25.9] |
| BDR at blood sample | mean (SD) [range] | 0.11 (0.10) | [−0.17,0.82] | 0.09 [0.08] | [−0.08, 0.59] | 0.08 (0.07) | [−0.14,0.49] |
| Other available time-points | Baseline | - | - | 558 | 99.1% | 294 | 99.7% |
| End | 558 | 99.6% | - | - | 295 | 100.0% | |
| Follow-up | 294 | 52.5% | 295 | 52.4% | - | - | |
| Metabolite | Beta | Interaction p-Value | Interaction q-Value a |
|---|---|---|---|
| 2-hydroxyglutarate | −0.004 | 1.77 × 10−4 | 0.089 |
| adipate | −0.004 | 0.001 | 0.136 |
| GABA | 0.004 | 0.004 | 0.468 |
| 2-O-methyladenosine | 0.002 | 0.005 | 0.468 |
| 3-methyladipate/pimelate | −0.002 | 0.005 | 0.468 |
| C18:1 CE | 0.005 | 0.006 | 0.468 |
| ectoine | −0.002 | 0.007 | 0.468 |
| saccharin | 0.001 | 0.008 | 0.468 |
| C18:3 CE | 0.004 | 0.010 | 0.468 |
| sebacate | −0.002 | 0.011 | 0.468 |
| suberate | −0.002 | 0.011 | 0.468 |
| C36:1 DAG | −0.002 | 0.011 | 0.468 |
| linoleoyl ethanolamide | 0.002 | 0.012 | 0.477 |
| C18:0 CE | 0.004 | 0.014 | 0.489 |
| C22:5 CE | 0.003 | 0.015 | 0.492 |
| C16:0 CE | 0.005 | 0.021 | 0.576 |
| cortisone | 0.002 | 0.022 | 0.576 |
| C54:1 TAG | −0.002 | 0.022 | 0.576 |
| C10:2 carnitine | −0.001 | 0.024 | 0.576 |
| arginine | 0.004 | 0.024 | 0.576 |
| C6 carnitine | 0.002 | 0.025 | 0.576 |
| taurodeoxycholate/taurochenodeoxycholate | −0.002 | 0.026 | 0.576 |
| C56:2 TAG | −0.003 | 0.027 | 0.576 |
| C36:0 DAG | −0.004 | 0.028 | 0.576 |
| C30:0 DAG | −0.001 | 0.029 | 0.589 |
| C36:2 DAG or TAG fragment | −0.002 | 0.032 | 0.614 |
| C58:10 TAG | 0.001 | 0.038 | 0.622 |
| sphingosine | 0.002 | 0.039 | 0.622 |
| C36:2 DAG | −0.002 | 0.041 | 0.622 |
| C20:3 CE | 0.003 | 0.042 | 0.622 |
| phenyllactate | −0.003 | 0.042 | 0.622 |
| C20:4 CE | 0.003 | 0.043 | 0.622 |
| C32:1 DAG | −0.002 | 0.043 | 0.622 |
| C5 carnitine | 0.002 | 0.043 | 0.622 |
| C54:2 TAG | −0.002 | 0.044 | 0.622 |
| C16:1 CE | 0.003 | 0.045 | 0.622 |
| ribothymidine | 0.002 | 0.046 | 0.622 |
| taurocholate | −0.002 | 0.047 | 0.622 |
| C3 carnitine | 0.002 | 0.050 | 0.641 |
| Metabolite | CAMP | GACRS | ||||
|---|---|---|---|---|---|---|
| Beta | Interaction p-Value | Interaction q-Value a | Beta | Interaction p-Value | Interaction q-Value a | |
| 2-hydroxyglutarate * | −0.004 | 1.80 × 10−4 | 0.089 | −0.015 | 0.018 | 0.997 |
| GABA. | 0.004 | 0.004 | 0.468 | 0.01 | 0.085 | 0.997 |
| 3-methyladipate/pimelate | −0.002 | 0.005 | 0.468 | −0.01 | 0.133 | 0.997 |
| C18:1 CE * | 0.005 | 0.006 | 0.468 | 0.023 | 0.041 | 0.997 |
| C18:3 CE | 0.004 | 0.01 | 0.468 | 0.009 | 0.203 | 0.997 |
| C36:1 DAG | −0.002 | 0.011 | 0.468 | 0.001 | 0.823 | 0.997 |
| linoleoyl ethanolamide | 0.002 | 0.012 | 0.477 | 0.001 | 0.807 | 0.997 |
| C18:0 CE. | 0.004 | 0.014 | 0.489 | 0.012 | 0.101 | 0.997 |
| C22:5 CE | 0.003 | 0.015 | 0.492 | 0.011 | 0.125 | 0.997 |
| C16:0 CE. | 0.005 | 0.021 | 0.576 | 0.023 | 0.056 | 0.997 |
| Cortisone | 0.002 | 0.022 | 0.576 | 0.001 | 0.812 | 0.997 |
| C54:1 TAG | −0.002 | 0.022 | 0.576 | −0.001 | 0.760 | 0.997 |
| C10:2 carnitine | −0.001 | 0.024 | 0.576 | −0.002 | 0.683 | 0.997 |
| Arginine | 0.004 | 0.024 | 0.576 | −0.002 | 0.747 | 0.997 |
| C6 carnitine | 0.002 | 0.025 | 0.576 | −0.001 | 0.841 | 0.997 |
| taurodeoxycholate/taurochenodeoxycholate | −0.002 | 0.026 | 0.576 | −0.004 | 0.321 | 0.997 |
| C56:2 TAG | −0.003 | 0.027 | 0.576 | −0.003 | 0.572 | 0.997 |
| C30:0 DAG | −0.001 | 0.029 | 0.589 | 0.001 | 0.799 | 0.997 |
| C58:10 TAG | 0.001 | 0.038 | 0.622 | −0.003 | 0.572 | 0.997 |
| C36:2 DAG | −0.002 | 0.04 | 0.622 | 0.001 | 0.949 | 0.997 |
| C20:3 CE | 0.003 | 0.042 | 0.622 | 0.01 | 0.189 | 0.997 |
| C20:4 CE. | 0.003 | 0.043 | 0.622 | 0.017 | 0.076 | 0.997 |
| C32:1 DAG | −0.002 | 0.043 | 0.622 | 0.001 | 0.832 | 0.997 |
| C5 carnitine | 0.002 | 0.043 | 0.622 | −0.001 | 0.861 | 0.997 |
| C54:2 TAG | −0.002 | 0.044 | 0.622 | −0.002 | 0.760 | 0.997 |
| C16:1 CE | 0.003 | 0.045 | 0.622 | 0.01 | 0.195 | 0.997 |
| Ribothymidine. | 0.002 | 0.046 | 0.622 | 0.01 | 0.088 | 0.997 |
| Taurocholate | −0.002 | 0.047 | 0.622 | −0.005 | 0.166 | 0.997 |
| C3 carnitine | 0.002 | 0.05 | 0.641 | 0.003 | 0.629 | 0.997 |
| Metabolite | CAMP | Costa Rica | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | Study End | Follow-up | ||||||||||||||
| Beta | 95% CI | p-Value | q-Value a | Beta | 95% CI | p-Value | q-Value a | Beta | 95% CI | p-Value | q-Value a | Beta | 95% CI | p-Value | q-Value a | |
| 2-hydroxy glutarate | −0.015 | (−0.034, 0.004) | 0.114 | 0.314 | −0.013 | (−0.029, 0.002) | 0.082 | 0.082 | 0.032 | (0.014, 0.05) | 0.001 * | 0.001 | −0.008 | (−0.038, 0.021) | 0.581 | 0.931 |
| C18:1 CE | −0.008 | (−0.02, 0.003) | 0.159 | 0.314 | −0.017 | (−0.028, −0.007) | 0.001 * | 0.009 | −0.02 | (−0.032, −0.008) | 0.002 * | 0.002 | −0.005 | (−0.022, 0.011) | 0.52 | 0.931 |
| C16:0 CE | 9.5 × 10−5 | (−0.01, 0.011) | 0.986 | 0.986 | −0.012 | (−0.022, −0.003) | 0.014 * | 0.016 | −0.009 | (−0.02, 0.002) | 0.108 * | 0.126 | −0.008 | (−0.024, 0.008) | 0.317 | 0.921 |
| GABA | −0.012 | (−0.028, 0.004) | 0.132 | 0.314 | −0.019 | (−0.034, −0.004) | 0.012 * | 0.016 | −0.029 | (−0.046, −0.013) | 0.001 * | 0.001 | −0.008 | (−0.038, 0.021) | 0.581 | 0.931 |
| C18:0 CE | 3.7 × 10−4 | (−0.014, 0.014) | 0.959 | 0.986 | −0.018 | (−0.031, −0.005) | 0.005 * | 0.012 | −0.036 | (−0.051, −0.02) | 7.7 × 10−6 * | 2.7 × 10−5 | 0.006 | (−0.017, 0.029) | 0.625 | 0.931 |
| C20:4 CE | −0.009 | (−0.021, 0.004) | 0.18 | 0.314 | −0.018 | (−0.031, −0.006) | 0.005 * | 0.012 | −0.006 | (−0.021, 0.008) | 0.404 | 0.404 | −0.012 | (−0.032, 0.009) | 0.265 | 0.921 |
| Ribo thymidine | −0.009 | (−0.025, 0.006) | 0.236 | 0.330 | −0.018 | (−0.031, −0.005) | 0.007 * | 0.012 | −0.062 | (−0.077, −0.047) | 6.2 × 10−15 * | 4.3 × 10−14 | 4.7 × 10−4 | (−0.031, 0.032) | 0.977 | 0.972 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kelly, R.S.; Sordillo, J.E.; Lutz, S.M.; Avila, L.; Soto-Quiros, M.; Celedón, J.C.; McGeachie, M.J.; Dahlin, A.; Tantisira, K.; Huang, M.; et al. Pharmacometabolomics of Bronchodilator Response in Asthma and the Role of Age-Metabolite Interactions. Metabolites 2019, 9, 179. https://doi.org/10.3390/metabo9090179
Kelly RS, Sordillo JE, Lutz SM, Avila L, Soto-Quiros M, Celedón JC, McGeachie MJ, Dahlin A, Tantisira K, Huang M, et al. Pharmacometabolomics of Bronchodilator Response in Asthma and the Role of Age-Metabolite Interactions. Metabolites. 2019; 9(9):179. https://doi.org/10.3390/metabo9090179
Chicago/Turabian StyleKelly, Rachel S., Joanne E. Sordillo, Sharon M. Lutz, Lydiana Avila, Manuel Soto-Quiros, Juan C. Celedón, Michael J. McGeachie, Amber Dahlin, Kelan Tantisira, Mengna Huang, and et al. 2019. "Pharmacometabolomics of Bronchodilator Response in Asthma and the Role of Age-Metabolite Interactions" Metabolites 9, no. 9: 179. https://doi.org/10.3390/metabo9090179









