Short-Term Temporal Metabolic Behavior in Halophilic Cyanobacterium Synechococcus sp. Strain PCC 7002 after Salt Shock
Abstract
:1. Introduction
2. Results and Discussion
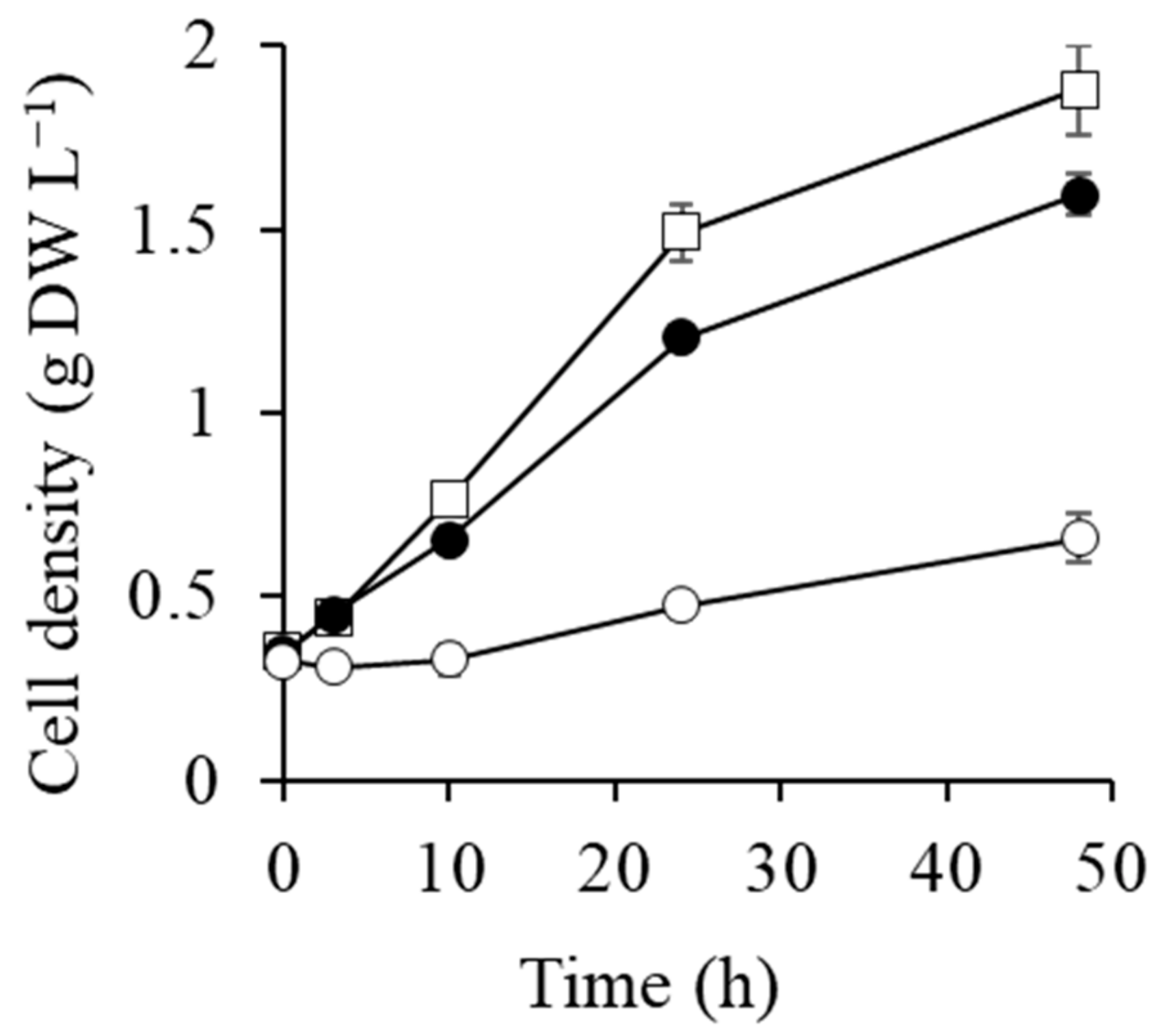
2.1. Cell Growth after Transfer to 0.5 and 1 M NaCl Conditions
2.2. Gene Expression of Na+/H+ Antiporters and K+ Transporters
2.3. Metabolic Analysis under 0.5 M and 1 M NaCl Conditions
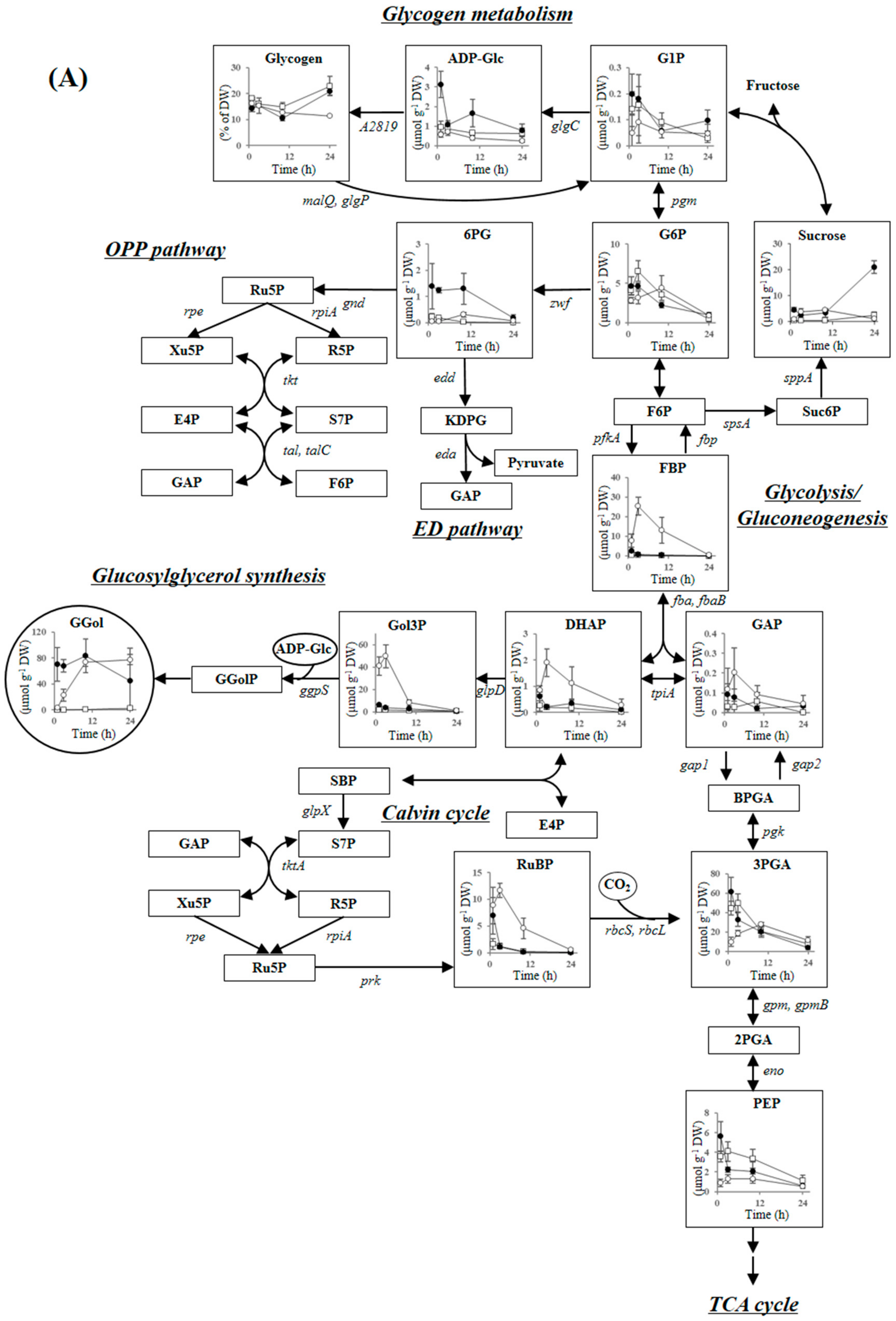
2.3.1. Compatible Solute Synthesis
2.3.2. Glycogen Metabolism and ADP-Glucose Synthesis
2.3.3. Carbon Dioxide Fixation, Glycolysis, the ED Pathway and the OPP Pathway
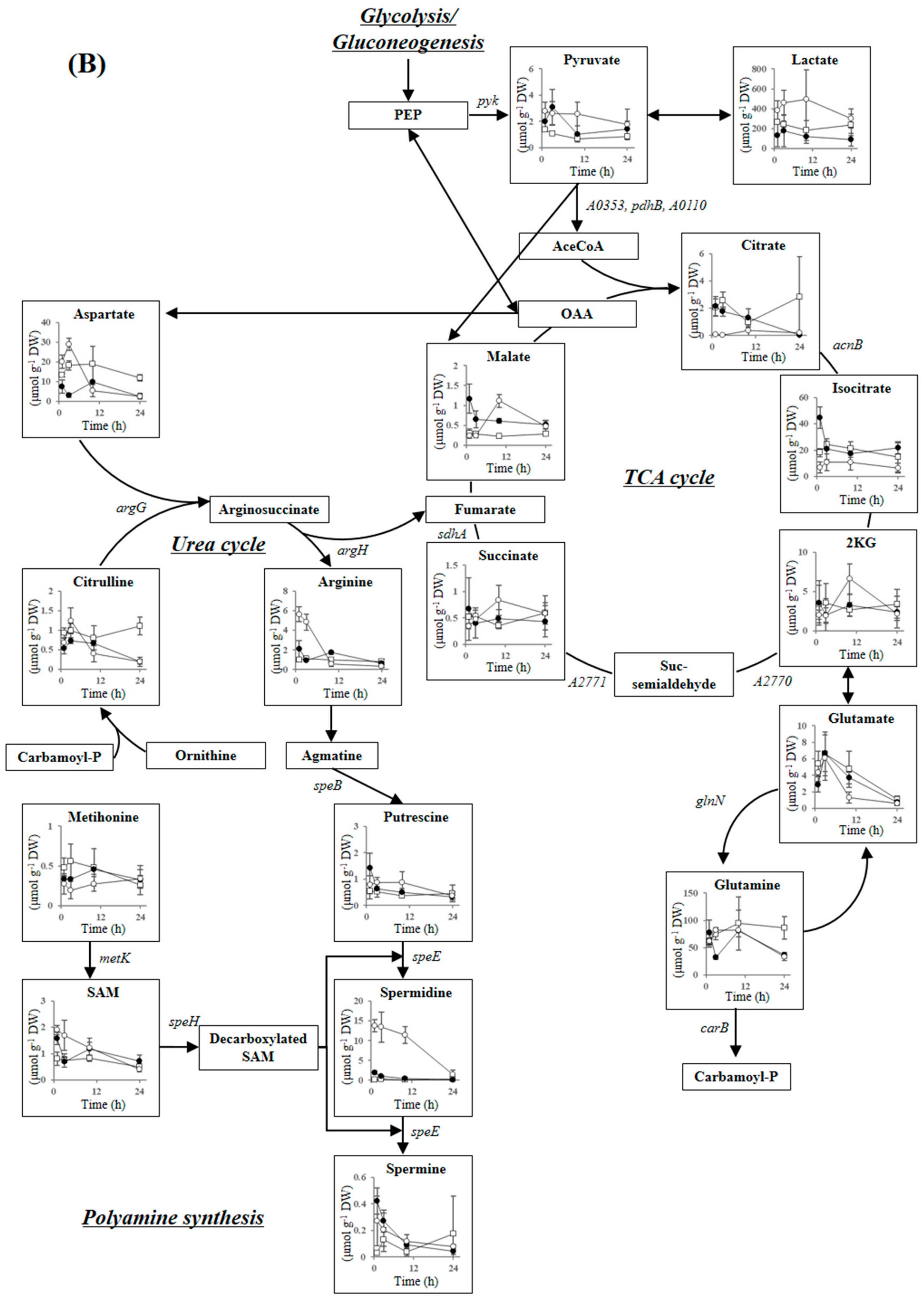
2.3.4. Tricarboxylic Acid Cycle, Urea Cycle, and Polyamine Synthesis
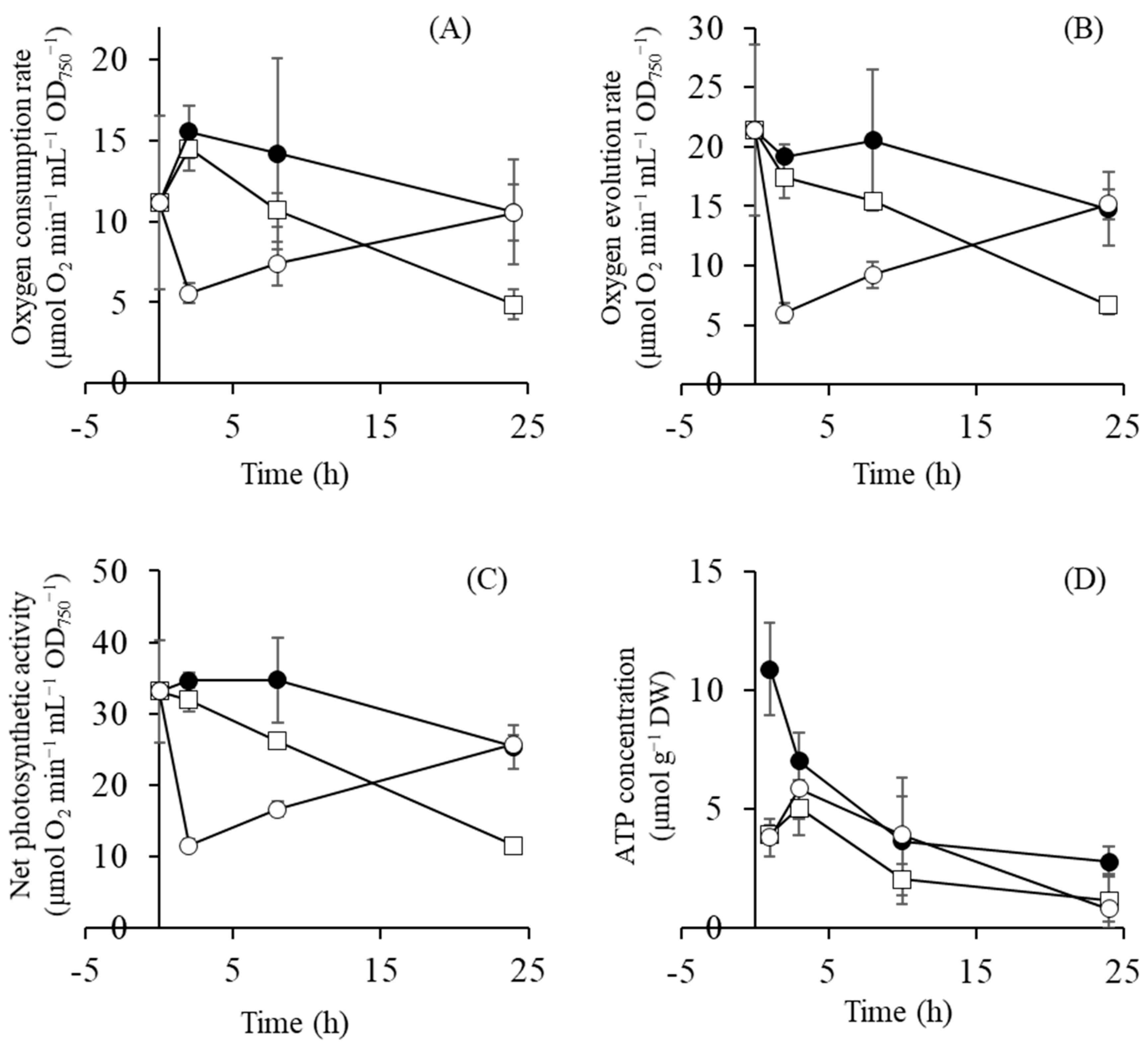
2.4. Gene Expression of Photosynthetic Components
2.5. Concluding Remarks on Salt Stress Response of Synechococcus sp. PCC 7002 at 0.5 M and 1 M NaCl
3. Materials and Methods
3.1. Strain and Culture Medium
3.2. Cell Density
3.3. Extraction of Intercellular Metabolites
3.4. Metabolite Analysis Using CE/MS
3.5. Glucosylglycerol and Sucrose Quantification Using GC/MS
3.6. Glycogen Quantification
3.7. Measurement of Oxygen Consumption Rate and Oxygen Evolution Rate
3.8. Profiles of Transcriptional Activity
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bose, J.; Munns, R.; Shabala, S.; Gilliham, M.; Pogson, B.; Tyerman, S.D. Chloroplast function and ion regulation in plants growing on saline soils: Lessons from halophytes. J. Exp. Bot. 2017, 68, 3129–3143. [Google Scholar] [CrossRef] [PubMed]
- Hagemann, M. Molecular biology of cyanobacterial salt acclimation. FEMS Microbiol. Rev. 2011, 35, 87–123. [Google Scholar] [CrossRef] [PubMed]
- Waditee, R.; Hibino, T.; Nakamura, T.; Incharoensakdi, A.; Takabe, T. Overexpression of a Na+/H+ antiporter confers salt tolerance on a freshwater cyanobacterium, making it capable of growth in sea water. Proc. Natl. Acad. Sci. USA 2002, 99, 4109–4114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waditee, R.; Bhuiyan, M.N.H.; Rai, V.; Aoki, K.; Tanaka, Y.; Hibino, T.; Suzuki, S.; Takano, J.; Jagendorf, A.T.; Takabe, T.; et al. Genes for direct methylation of glycine provide high levels of glycinebetaine and abiotic-stress tolerance in Synechococcus and Arabidopsis. Proc. Natl. Acad. Sci. USA 2005, 102, 1318–1323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pade, N.; Hagemann, M. Salt acclimation of cyanobacteria and their application in biotechnology. Life 2015, 5, 25–49. [Google Scholar] [CrossRef]
- Blumwald, E.; Mehlhorn, R.J.; Packer, L. Ionic osmoregulation during salt adaptation of the cyanobacterium Synechococcus 6311. Plant Physiol. 1983, 73, 377–380. [Google Scholar] [CrossRef] [Green Version]
- Reed, R.H.; Warr, S.R.; Richardson, D.L.; Moore, D.J.; Stewart, W.D. Multiphasic osmotic adjustment in a euryhaline cyanobacterium. FEMS Microbiol. Lett. 1985, 28, 225–229. [Google Scholar] [CrossRef]
- Blumwald, E.; Wolosin, J.M.; Packer, L. Na+ H+ exchange in the cyanobacterium Synechococcus 6311. Biochem. Biophys. Res. Commun. 1984, 122, 452–459. [Google Scholar] [CrossRef]
- Reed, R.H.; Warr, S.R.; Kerby, N.W.; Stewart, W.D. Osmotic shock-induced release of low molecular weight metabolites from free-living and immobilized cyanobacteria. Enzym. Microb. Technol. 1986, 8, 101–104. [Google Scholar] [CrossRef]
- Tsunekawa, K.; Shijuku, T.; Hayashimoto, M.; Kojima, Y.; Onai, K.; Morishita, M.; Ishiura, M.; Kuroda, T.; Nakamura, T.; Kobayashi, H. Identification and characterization of the Na+/H+ antiporter Nhas3 from the thylakoid membrane of Synechocystis sp. PCC 6803. J. Biol. Chem. 2009, 284, 16513–16521. [Google Scholar] [CrossRef] [Green Version]
- Inaba, M.; Sakamoto, A.; Murata, N. Functional expression in Escherichia coli of low-affinity and high-affinity Na+(Li+)/H+ antiporters of Synechocystis. J. Bacteriol. 2001, 183, 1376–1384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mikkat, S.; Milkowski, C.; Hagemann, M. The gene sll0273 of the cyanobacterium Synechocystis sp. strain PCC6803 encodes a protein essential for growth at low Na+/K+ ratios. Plant Cell Environ. 2000, 23, 549–559. [Google Scholar] [CrossRef]
- Berry, S.; Esper, B.; Karandashova, I.; Teuber, M.; Elanskaya, I.; Rögner, M.; Hagemann, M. Potassium uptake in the unicellular cyanobacterium Synechocystis sp. strain PCC 6803 mainly depends on a Ktr-like system encoded by slr1509 (ntpJ). FEBS Lett. 2003, 548, 53–58. [Google Scholar] [CrossRef]
- Marin, K.; Kanesaki, Y.; Los, D.A.; Murata, N.; Suzuki, I.; Hagemann, M. Gene expression profiling reflects physiological processes in salt acclimation of Synechocystis sp. strain PCC 6803. Plant Physiol. 2004, 136, 3290–3300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanesaki, Y.; Suzuki, I.; Allakhverdiev, S.I.; Mikami, K.; Murata, N. Salt stress and hyperosmotic stress regulate the expression of different sets of genes in Synechocystis sp. PCC 6803. Biochem. Biophys. Res. Commun. 2002, 290, 339–348. [Google Scholar] [CrossRef]
- Hagemann, M.; Erdmann, N.; Wittenburg, E. Synthesis of glucosylglycerol in salt-stressed cells of the cyanobacterium Microcystis firma. Arch. Microbiol. 1987, 148, 275–279. [Google Scholar] [CrossRef]
- Erdmann, N. Organic osmoregulatory solutes in blue-green algae. Zeitschrift für Pflanzenphysiologie 1983, 110, 147–155. [Google Scholar] [CrossRef]
- Tel-Or, E.; Spath, S.; Packer, L.; Mehlhorn, R.J. Carbon-13 NMR studies of salt shock-induced carbohydrate turnover in the marine cyanobacterium Agmenellum quadruplicatum. Plant Physiol. 1986, 82, 646–652. [Google Scholar] [CrossRef]
- Mackay, M.A.; Norton, R.S. 13C nuclear magnetic resonance study of biosynthesis of glucosylglycerol by a cyanobacterium under osmotic stress. Microbiology 1987, 133, 1535–1542. [Google Scholar] [CrossRef] [Green Version]
- Warr, S.; Reed, R.; Chudek, J.; Foster, R.; Stewart, W. Osmotic adjustment in Spirulina platensis. Planta 1985, 163, 424–429. [Google Scholar] [CrossRef]
- Soontharapirakkul, K.; Promden, W.; Yamada, N.; Kageyama, H.; Incharoensakdi, A.; Iwamoto-Kihara, A.; Takabe, T. Halotolerant cyanobacterium Aphanothece halophytica contains an Na+-dependent F1F0-ATP synthase with a potential role in salt-stress tolerance. J. Biol. Chem. 2011, 286, 10169–10176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kluge, C.; Dimroth, P. Specific protection by Na+ or Li+ of the F1F0-ATPase of Propionigenium modestum from the reaction with dicyclohexylcarbodiimide. J. Biol. Chem. 1993, 268, 14557–14560. [Google Scholar] [PubMed]
- Kaim, G. The Na+-translocating F1F0 ATP synthase of Propionigenium modestum: Mechanochemical insights into the F0 motor that drives ATP synthesis. Biochim. Biophys. Acta (BBA) Bioenerg. 2001, 1505, 94–107. [Google Scholar] [CrossRef] [Green Version]
- Ludwig, M.; Bryant, D.A. Synechococcus sp. strain PCC 7002 transcriptome: Acclimation to temperature, salinity, oxidative stress, and mixotrophic growth conditions. Front. Microbiol. 2012, 3, 354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dimroth, P.; Wang, H.; Grabe, M.; Oster, G. Energy transduction in the sodium F-ATPase of Propionigenium modestum. Proc. Natl. Acad. Sci. USA 1999, 96, 4924–4929. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engelbrecht, F.; Marin, K.; Hagemann, M. Expression of the ggpS Gene, involved in osmolyte synthesis in the marine cyanobacterium Synechococcus sp. strain PCC 7002, revealed regulatory differences between this strain and the freshwater strain Synechocystis sp. strain PCC 6803. Appl. Environ. Microbiol. 1999, 65, 4822–4829. [Google Scholar]
- Xu, Y.; Tiago Guerra, L.; Li, Z.; Ludwig, M.; Charles Dismukes, G.; Bryant, D.A. Altered carbohydrate metabolism in glycogen synthase mutants of Synechococcus sp. strain PCC 7002: Cell factories for soluble sugars. Metab. Eng. 2013, 16, 56–67. [Google Scholar] [CrossRef]
- Aikawa, S.; Nishida, A.; Ho, S.-H.; Chang, J.-S.; Hasunuma, T.; Kondo, A. Glycogen production for biofuels by the euryhaline cyanobacteria Synechococcus sp. strain PCC 7002 from an oceanic environment. Biotechnol. Biofuels 2014, 7, 88. [Google Scholar] [CrossRef] [Green Version]
- Aikawa, S.; Izumi, Y.; Matsuda, F.; Hasunuma, T.; Chang, J.-S.; Kondo, A. Synergistic enhancement of glycogen production in Arthrospira platensis by optimization of light intensity and nitrate supply. Bioresour. Technol. 2012, 108, 211–215. [Google Scholar] [CrossRef]
- Ballicora, M.A.; Iglesias, A.A.; Preiss, J. ADP-glucose pyrophosphorylase: A regulatory enzyme for plant starch synthesis. Photosynth. Res. 2004, 79, 1–24. [Google Scholar] [CrossRef]
- Solomon, A.; Beer, S.; Waisel, Y.; Jones, G.; Paleg, L. Effects of NaCl on the carboxylating activity of Rubisco from Tamarix jordanis in the presence and absence of proline-related compatible solutes. Physiol. Plant. 1994, 90, 198–204. [Google Scholar] [CrossRef]
- Wedel, N.; Soll, J.; Paap, B.K. CP12 provides a new mode of light regulation of Calvin cycle activity in higher plants. Proc. Natl. Acad. Sci. USA 1997, 94, 10479–10484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wedel, N.; Soll, J. Evolutionary conserved light regulation of Calvin cycle activity by NADPH-mediated reversible phosphoribulokinase/CP12/glyceraldehyde-3-phosphate dehydrogenase complex dissociation. Proc. Natl. Acad. Sci. USA 1998, 95, 9699–9704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rae, B.D.; Long, B.M.; Whitehead, L.F.; Förster, B.; Badger, M.R.; Price, G.D. Cyanobacterial carboxysomes: Microcompartments that facilitate CO2 fixation. J. Mol. Microbiol. Biotechnol. 2013, 23, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Danshina, P.; Schmalhausen, E.; Avetisyan, A.; Muronetz, V. Mildly oxidized glyceraldehyde-3-phosphate dehydrogenase as a possible regulator of glycolysis. IUBMB Life 2001, 51, 309–314. [Google Scholar]
- Chen, X.; Schreiber, K.; Appel, J.; Makowka, A.; Fähnrich, B.; Roettger, M.; Hajirezaei, M.R.; Sönnichsen, F.D.; Schönheit, P.; Martin, W.F.; et al. The Entner–Doudoroff pathway is an overlooked glycolytic route in cyanobacteria and plants. Proc. Natl. Acad. Sci. USA 2016, 113, 5441–5446. [Google Scholar] [CrossRef] [Green Version]
- Srivastava, A.K. Assessment of salinity-induced antioxidative defense system of diazotrophic cyanobacterium Nostoc muscorum. J. Microbiol. Biotechnol. 2010, 20, 1506–1512. [Google Scholar] [CrossRef] [Green Version]
- Cooley, J.W.; Vermaas, W.F.J. Succinate dehydrogenase and other respiratory pathways in thylakoid membranes of Synechocystis sp. strain PCC 6803: Capacity comparisons and physiological function. J. Bacteriol. 2001, 183, 4251–4258. [Google Scholar] [CrossRef] [Green Version]
- Lea-Smith, D.J.; Ross, N.; Zori, M.; Bendall, D.S.; Dennis, J.S.; Scott, S.A.; Smith, A.G.; Howe, C.J. Thylakoid terminal oxidases are essential for the cyanobacterium Synechocystis sp. PCC 6803 to survive rapidly changing light intensities. Plant Physiol. 2013, 162, 484–495. [Google Scholar] [CrossRef] [Green Version]
- Che-Othman, M.H.; Jacoby, R.P.; Millar, A.H.; Taylor, N.L. Wheat mitochondrial respiration shifts from the tricarboxylic acid cycle to the GABA shunt under salt stress. New Phytol. 2019. [Google Scholar] [CrossRef]
- Hamdani, S.; Gauthier, A.; Msilini, N.; Carpentier, R. Positive charges of polyamines protect PSII in isolated thylakoid membranes during photoinhibitory conditions. Plant Cell Physiol. 2011, 52, 866–873. [Google Scholar] [CrossRef] [Green Version]
- Bueno, M.; Cordovilla, M.-P. Polyamines in halophytes. Front. Plant Sci. 2019, 10, 439. [Google Scholar] [CrossRef] [Green Version]
- Chen, D.; Shao, Q.; Yin, L.; Younis, A.; Zheng, B. Polyamine function in plants: Metabolism, regulation on development, and roles in abiotic stress responses. Front. Plant Sci. 2019, 9, 1945. [Google Scholar] [CrossRef] [PubMed]
- Raksajit, W.; Yodsang, P.; Maenpaa, P.; Incharoensakdi, A. Characterization of spermidine transport system in a cyanobacterium, Synechocystis sp. PCC 6803. J. Microbiol. Biotechnol. 2009, 19, 447–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tonon, G.; Kevers, C.; Faivre-Rampant, O.; Graziani, M.; Gaspar, T. Effect of NaCl and mannitol iso-osmotic stresses on proline and free polyamine levels in embryogenic Fraxinus angustifolia callus. J. Plant Physiol. 2004, 161, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Zapata, P.J.; Serrano, M.; García-Legaz, M.F.; Pretel, M.T.; Botella, M.A. Short term effect of salt shock on ethylene and polyamines depends on plant salt sensitivity. Front. Plant Sci. 2017, 8, 855. [Google Scholar] [CrossRef] [Green Version]
- Mulo, P.; Sicora, C.; Aro, E.-M. Cyanobacterial psbA gene family: Optimization of oxygenic photosynthesis. Cell. Mol. Life Sci. 2009, 66, 3697–3710. [Google Scholar] [CrossRef] [Green Version]
- Bölling, C.; Fiehn, O. Metabolite profiling of Chlamydomonas reinhardtii under nutrient deprivation. Plant Physiol. 2005, 139, 1995–2005. [Google Scholar] [CrossRef] [Green Version]
- Hasunuma, T.; Kikuyama, F.; Matsuda, M.; Aikawa, S.; Izumi, Y.; Kondo, A. Dynamic metabolic profiling of cyanobacterial glycogen biosynthesis under conditions of nitrate depletion. J. Exp. Bot. 2013, 64, 2943–2954. [Google Scholar] [CrossRef] [Green Version]
- Fiehn, O.; Kopka, J.; Dörmann, P.; Altmann, T.; Trethewey, R.N.; Willmitzer, L. Metabolite profiling for plant functional genomics. Nat. Biotechnol. 2000, 18, 1157–1161. [Google Scholar] [CrossRef]
- Izumi, Y.; Aikawa, S.; Matsuda, F.; Hasunuma, T.; Kondo, A. Aqueous size-exclusion chromatographic method for the quantification of cyanobacterial native glycogen. J. Chromatogr. B 2013, 930, 90–97. [Google Scholar] [CrossRef] [PubMed]

| Gene | Accession No. | Function | Induction Factor | |
|---|---|---|---|---|
| 0.5 M NaCl | 1 M NaCl | |||
| nhaS3 | A0577 | Na+/H+ antiporter localized in thylakoid membrane | ‒–*1 | 2.4(± 0.1) |
| atpA-II | G0151 | F1F0-type Na+-ATPase, subunit alpha | 2.0 (± 0.3) | 2.4(± 0.2) |
| atpB-II | G0148 | F1F0-type Na+-ATPase, subunit A | 2.5 (± 0.2) | 1.3 (± 0.3) |
| atpC-II | G0145 | F1F0-type Na+-ATPase, subunit epsilon | 2.4 (± 0.3) | 4.0 (± 0.2) |
| atpD | A0749 | F1F0-type Na+-ATPase, subunit beta | 2.5 (± 0.3) | 2.9 (± 0.7) |
| atpF-II | G0150 | F1F0-type Na+-ATPase, subunit B | 2.0 (± 0.4) | 2.1 (± 0.5) |
| atpG-II | G0152 | F1F0-type Na+-ATPase, gamma subunit | 2.3 (± 0.2) | ‒–*1 |
| atpH-II | G0149 | F1F0-type Na+-ATPase, subunit C | 1.5 (± 0.1) | 2.1(± 0.0) |
| kdpA | G0060 | K+-transporting ATPase, A subunit | 32.0 (± 3.1) | 2.0 (± 0.3) |
| kdpB | G0059 | K+-transporting ATPase, B subunit | 33.0 (± 4.7) | 2.2 (± 0.3) |
| kdpC | G0055 | K+-transporting ATPase, C subunit | 13.9 (± 2.6) | 1.3 (± 0.2) |
| kdpD | G0054 | K+-transporting ATPase, D subunit | 20.2 (± 6.5) | 1.3 (± 0.1) |
| kdpF | G0057 | K+-transporting ATPase, F subunit | 20.6 (± 0.3) | 1.5 (± 0.1) |
| Gene | Accession No. | Function | Induction Factor | |
|---|---|---|---|---|
| 0.5 M NaCl | 1 M NaCl | |||
| glpD | A2852 | Glycerol-3-phosphate dehydrogenase | 9.9 (± 1.1) | 7.9 (± 1.0) |
| ggpS | A2851 | Glucosylglycerol-3-phosphate synthase | 9.3 (± 2.7) | 6.9 (± 1.7) |
| stpA | A2841 | Glucosylglycerol-3-phosphatase | 2.6 (± 0.3) | 1.5 (± 0.2) |
| spsA | A0888 | Sucrose-phosphate synthase | 2.5 (± 0.1) | 2.6 (± 0.4) |
| sppA | A0887 | Sucrose-phosphate phosphatase | 2.1 (± 0.2) | 2.0 (± 0.5) |
| glgC | A0095 | Glycerol-3-phosphate dehydrogenase | −2.1 (± 0.2) | −2.4 (± 0.1) |
| A2819 | A2819 | 1,4-Alpha-glucan branching enzyme | −2.1 (± 0.1) | −1.8 (± 0.0) |
| glgP | A2139 | Alpha-glucan phosphorylase | 2.1 (± 0.3) | 2.2 (± 0.1) |
| malQ | A0330 | 4-Alpha-glucanotransferase | 2.5 (± 0.4) | 2.0 (± 0.4) |
| Gene | Accession No. | Function | Induction Factor | |
|---|---|---|---|---|
| 0.5 M NaCl | 1 M NaCl | |||
| rbcL | A1798 | Rubisco large subunit gene | −6.2 (± 1.9) | −5.9 (± 0.4) |
| rbcS | A1796 | Rubisco small subunit | −5.5 (± 0.1) | −6.8 (± 0.3) |
| rbcX | A1797 | Rubisco chaperone | −5.0 (± 0.3) | −6.3 (± 0.3) |
| prk | A2665 | Phosphoribulokinase | −1.5 (± 0.2) | −2.5 (± 0.5) |
| prk | A2857 | Phosphoribulokinase | −2.6 (± 0.4) | −3.5 (± 0.1) |
| gap2 | A0106 | Glyceraldehyde-3-phosphate dehydrogenase (Gapdh2) | −4.1 (± 0.6) | −2.8 (± 0.5) |
| glpX | A1301 | Fructose-1,6-bisphosphatase/sedoheptulose 1,7-bisphosphatase | −4.0 (± 0.5) | −4.5 (± 1.6) |
| ccmK | A2613 | Carboxysome shell protein (hexamer) | −3.4 (± 0.4) | −5.1 (± 2.3) |
| ccmK | A2612 | Carboxysome shell protein (hexamer) | −1.9 (± 0.3) | −3.1 (± 0.4) |
| ccmO | A2389 | Carboxysome shell protein (pseudohexamer) | 2.4 (± 0.3) | 1.1 (± 0.0) |
| ccmL | A1801 | Carboxysome shell protein | −4.2 (± 0.3) | −4.8 (± 1.1) |
| ccmM | A1800 | Carboxysome assembly protein | −2.7 (± 0.2) | −2.4 (± 0.2) |
| pgm | A0165 | Phosphoglucomutase | −2.6 (± 0.4) | −2.4 (± 0.2) |
| pgm | A1492 | Phosphoglucomutase | −1.1 (± 0.2) | 2.3 (± 0.2) |
| pfkA | A0162 | 6-Phosphofructokinase | 1.9 (± 0.0) | 2.6 (± 0.0) |
| fbp | A0329 | Fructose-1,6-bisphosphatase | 2.4 (± 0.3) | ‒–*1 |
| fba | A1352 | Fructose-1,6-bisphosphate aldolase class II | −2.3 (± 0.5) | −2.5 (± 0.2) |
| fbaB | A0010 | Fructose-1,6-bisphosphate aldolase class I | 3.4 (± 0.3) | 1.7 (± 0.2) |
| tpiA | A0595 | Triosephosphate isomerase | −1.2 (± 0.1) | −2.3 (± 0.1) |
| gap1 | A2697 | Glyceraldehyde-3-phosphate dehydrogenase (Gapdh1) | 3.4 (± 0.7) | 2.2 (± 0.4) |
| pgk | A1585 | Phosphoglycerate kinase | −3.0 (± 0.3) | −2.4 (± 0.1) |
| gpmB | A2560 | Phosphoglycerate mutase | −2.9 (± 0.1) | −2.4 (± 0.1) |
| eno | A0073 | 2-Phosphopyruvate hydratase | −2.8 (± 0.2) | −4.0 (± 0.6) |
| zwf | A1459 | Glucose-6-phosphate 1-dehydrogenase | 4.4 (± 0.4) | 2.5 (± 0.2) |
| edd | A0652 | 6-Phosphogluconate dehydratase | −2.5 (± 0.3) | −2.4 (± 0.3) |
| eda | A0130 | 2-Keto-3-deoxygluconate-6-phosphate aldolase | 3.6 (± 0.6) | 3.7 (± 0.7) |
| Gene | Accession No. | Function | Induction Factor | |
|---|---|---|---|---|
| 0.5 M NaCl | 1 M NaCl | |||
| pyk | A1658 | Pyruvate kinase | −3.1 (± 0.4) | −2.6 (± 0.2) |
| A0353 | A0353 | Pyruvate dehydrogenase E1 alpha subunit | −3.4 (± 0.4) | −4.0 (± 0.2) |
| pdhB | A0655 | Pyruvate dehydrogenase E1 beta subunit | −3.8 (± 0.5) | −3.7 (± 0.3) |
| A0110 | A0110 | Pyruvate dehydrogenase E2 subunit | −2.5 (± 0.2) | −3.5 (± 0.5) |
| acnB | A1683 | Methylisocitrate dehydratase | −1.5 (± 0.1) | −3.0 (± 0.5) |
| glnN | A0246 | Glutamine synthetase | −2.4 (± 0.2) | −3.0 (± 0.2) |
| carB | A2488 | Carbamoyl-phosphate synthase large subunit | −2.4 (± 0.6) | −1.7 (± 0.2) |
| A2770 | A2770 | 2-Ketoglutarate decarboxylase | 2.2 (± 0.3) | 2.7 (± 0.7) |
| A2771 | A2771 | Succinate-semialdehyde dehydrogenase | 3.3 (± 0.3) | 1.9 (± 0.1) |
| sdhA | A2569 | Succinate dehydrogenase flavoprotein subunit | 2.2 (± 0.2) | ‒–*1 |
| argG | A2806 | Argininosuccinate synthase | −2.6 (± 0.3) | −2.3 (± 0.2) |
| argH | A2487 | Argininosuccinate lyase | −1.6 (± 0.2) | −2.3 (± 0.1) |
| speB | A1109 | Agmatinase | −3.4 (± 0.6) | −1.8 (± 0.1) |
| speE | A2283 | Spermidine synthase | −5.5 (± 0.6) | −3.2 (± 0.3) |
| metK | A1714 | S-adenosylmethionine (SAM) synthetase | −2.5 (± 0.3) | −2.9 (± 0.2) |
| speH | A0430 | SAM decarboxylase | −10.2 (± 1.8) | −4.0 (± 0.1) |
| Gene | Accession No. | Function | Induction Factor | |
|---|---|---|---|---|
| 0.5 M NaCl | 1 M NaCl | |||
| psaA | A1961 | PSI P700 apoprotein A1 | −2.2 (± 0.1) | −1.6 (± 0.0) |
| psaB | A1962 | PSI P700 apoprotein A2 | −2.3 (± 0.2) | −1.4 (± 0.0) |
| psbA-II | A0157 | PSII D1 subunit | −3.3 (± 0.3) | −1.6 (± 0.3) |
| psbA | A1418 | PSII D1 subunit | −3.5 (± 0.4) | −1.7 (± 0.3) |
| psbA | A2164 | PSII D1 subunit | 1.7 (± 0.2) | 3.5 (± 1.7) |
| psbD | A1560 | PSII D2 subunit | −2.1 (± 0.1) | −1.8 (± 0.0) |
| apcA | A1930 | Allophycocyanin (APC) subunit alpha | −4.8 (± 0.2) | −3.3 (± 0.6) |
| apcB | A1929 | APC subunit beta | −4.0 (± 0.6) | −3.6 (± 0.6) |
| apcE | A2009 | PBS linker protein | −2.5 (± 0.2) | −1.5 (± 0.1) |
| cpcA | A2210 | Phycocyanin (PC) subunit alpha | −4.6 (± 0.2) | −2.6 (± 0.2) |
| cpcB | A2209 | PC subunit beta | −4.4 (± 0.3) | −2.6 (± 0.6) |
| cpcE | A2213 | PC alpha subunit phycocyanobilin lyase | ‒–*1 | 3.0 (± 0.4) |
| cpcF | A2214 | PC subunit alpha phycocyanobilin lyase | 1.2 (± 0.1) | 3.8 (± 0.2) |
| petA | A1910 | Cytochrome f | −3.5 (± 0.4) | −5.8 (± 1.5) |
| petB | A0842 | Cytochrome b6 | −2.4 (± 0.1) | −3.1 (± 0.1) |
| petF | A2325 | Ferredoxin | −3.4 (± 0.6) | −3.7 (± 0.4) |
| petH | A0853 | Ferredoxin-NADP reductase | −2.9 (± 0.3) | −4.5 (± 0.4) |
| atpA | A0734 | F1F0 ATP synthase subunit alpha | −3.5 (± 0.8) | −3.8 (± 0.5) |
| atpB | A0739 | F1F0 ATP synthase subunit A | −3.3 (± 0.3) | −11.6 (± 3.1) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aikawa, S.; Nishida, A.; Hasunuma, T.; Chang, J.-S.; Kondo, A. Short-Term Temporal Metabolic Behavior in Halophilic Cyanobacterium Synechococcus sp. Strain PCC 7002 after Salt Shock. Metabolites 2019, 9, 297. https://doi.org/10.3390/metabo9120297
Aikawa S, Nishida A, Hasunuma T, Chang J-S, Kondo A. Short-Term Temporal Metabolic Behavior in Halophilic Cyanobacterium Synechococcus sp. Strain PCC 7002 after Salt Shock. Metabolites. 2019; 9(12):297. https://doi.org/10.3390/metabo9120297
Chicago/Turabian StyleAikawa, Shimpei, Atsumi Nishida, Tomohisa Hasunuma, Jo-Shu Chang, and Akihiko Kondo. 2019. "Short-Term Temporal Metabolic Behavior in Halophilic Cyanobacterium Synechococcus sp. Strain PCC 7002 after Salt Shock" Metabolites 9, no. 12: 297. https://doi.org/10.3390/metabo9120297
APA StyleAikawa, S., Nishida, A., Hasunuma, T., Chang, J.-S., & Kondo, A. (2019). Short-Term Temporal Metabolic Behavior in Halophilic Cyanobacterium Synechococcus sp. Strain PCC 7002 after Salt Shock. Metabolites, 9(12), 297. https://doi.org/10.3390/metabo9120297






