Effects of Aging and Methionine Restriction on Rat Kidney Metabolome
Abstract
1. Introduction
2. Results
2.1. Effects of Aging and MetR on Markers of Mitochondrial Stress and Tissue Protein Damage
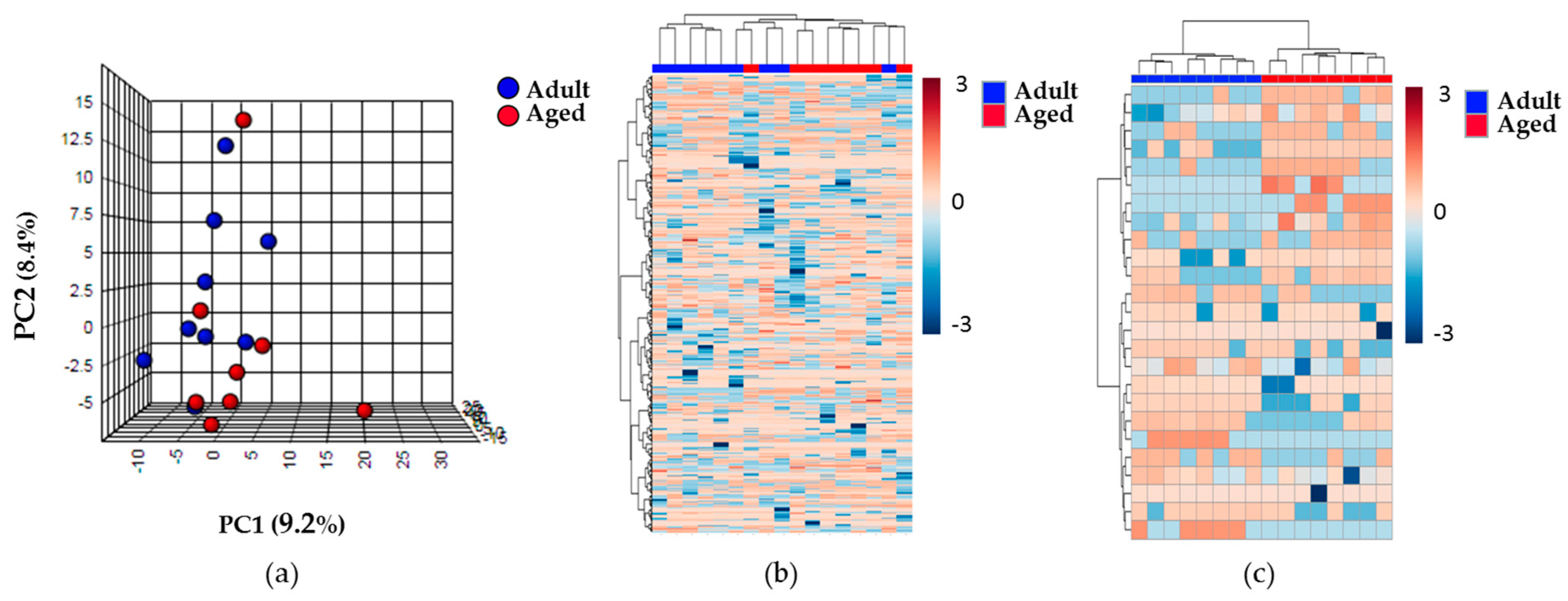
2.2. Effect of Aging on the Renal Cortex Metabolome
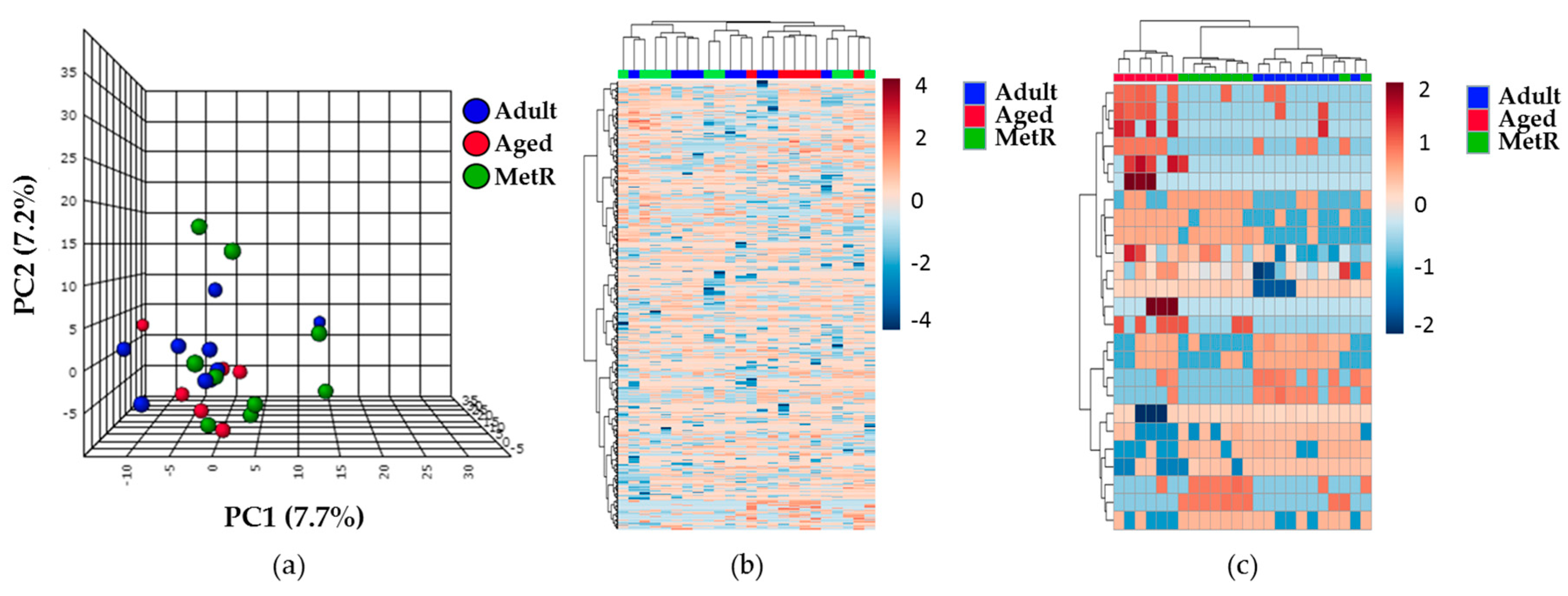
2.3. Effect of MetR on the Renal Cortex Metabolome
3. Discussion
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Sullivan, E.D.; Hughes, J.; Ferenbach, D.A. Renal Aging: Causes and Consequences. J. Am. Soc. Nephrol. 2017, 28, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.J.; Rakheja, D.; Yu, X.; Saxena, R.; Vaziri, N.D.; Silva, F.G. The aging kidney. Kidney Int. 2008, 74, 710–720. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.A.O.; Liebman, S.E.; Scott Lucia, M.; Li, J.; Levi, M. Role of altered renal lipid metabolism and the sterol regulatory element binding proteins in the pathogenesis of age-related renal disease. Kidney Int. 2005, 68, 2608–2620. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Fitzl, G.; Mozet, C.; Martin, H.; Welt, K.; Wieland, E. Effect of age and hypoxia/reoxygenation on mRNA expression of antioxidative enzymes in rat liver and kidneys. Exp. Gerontol. 2002, 37, 1481–1487. [Google Scholar] [CrossRef]
- Csiszar, A.; Toth, J.; Peti-Peterdi, J.; Ungvari, Z. The aging kidney: Role of endothelial oxidative stress and inflammation. Acta Physiol. Hung. 2007, 94, 107–115. [Google Scholar] [CrossRef]
- Uzun, D.; Korkmaz, G.G.; Sitar, M.E.; Cebe, T.; Yanar, K.; Cakatay, U.; Aydın, S. Oxidative damage parameters in renal tissues of aged and young rats based on gender. Clin. Interv. Aging 2013, 8, 809–815. [Google Scholar]
- Ward, W.F.; Qi, W.; Van Remmen, H.; Zackert, W.E.; Roberts, L.J.; Richardson, A. Effects of age and caloric restriction on lipid peroxidation: Measurement of oxidative stress by F2-isoprostane levels. J. Gerontol. A. Biol. Sci. Med. Sci. 2005, 60, 847–851. [Google Scholar] [CrossRef]
- Abdel-Rahman, E.M.; Okusa, M.D. Effects of aging on renal function and regenerative capacity. Nephron. Clin. Pract. 2014, 127, 15–20. [Google Scholar] [CrossRef]
- Dunn, M. The role of arachidonic acid metabolites in renal homeostasis. Non-steroidal anti-inflammatory drugs renal function and biochemical, histological and clinical effects and drug interactions. Drugs 1987, 33, 56–66. [Google Scholar] [CrossRef]
- Pradas, I.; Huynh, K.; Cabré, R.; Ayala, V.; Meikle, P.J.; Jové, M.; Pamplona, R. Lipidomics Reveals a Tissue-Specific Fingerprint. Front. Physiol. 2018, 9, 1165. [Google Scholar] [CrossRef]
- Noh, S.A.; Kim, S.-M.; Park, S.H.; Kim, D.-J.; Lee, J.W.; Kim, Y.G.; Moon, J.-Y.; Lim, S.-J.; Lee, S.-H.; Kim, K.P. Alterations in Lipid Profile of the Aging Kidney Identified by MALDI Imaging Mass Spectrometry. J. Proteom. Res. 2019, 18, 2803–2812. [Google Scholar] [CrossRef] [PubMed]
- Omata, K.; Abraham, N.G.; Escalante, B.; Schwartzman, M.L. Age-related changes in renal cytochrome P-450 arachidonic acid metabolism in spontaneously hypertensive rats. Am. J. Physiol. 1992, 262, F8–F16. [Google Scholar] [CrossRef] [PubMed]
- Hornych, A. Ageing and Prostaglandins. In The Eicosanoids; Curtis-Prior, P., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2004; pp. 299–318. ISBN 9780470020623. [Google Scholar]
- Drenjančević, I.; Jukić, I.; Mihaljević, Z.; Ćosić, A.; Kibel, A. The Metabolites of Arachidonic Acid in Microvascular Function. In Microcirculation Revisited—From Molecules to Clinical Practice; InTech: London, UK, 2016. [Google Scholar]
- Pamplona, R.; Barja, G. Mitochondrial oxidative stress, aging and caloric restriction: The protein and methionine connection. Biochim. Biophys. Acta Bioenergy 2006, 1757, 496–508. [Google Scholar] [CrossRef] [PubMed]
- Richie, J.P.; Leutzinger, Y.; Parthasarathy, S.; Malloy, V.; Orentreich, N.; Zimmerman, J.A. Methionine restriction increases blood glutathione and longevity in F344 rats. FASEB J. 1994, 8, 1302–1307. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.A.; Buehner, G.; Chang, Y.; Harper, J.M.; Sigler, R.; Smith-Wheelock, M. Methionine-deficient diet extends mouse lifespan, slows immune and lens aging, alters glucose, T4, IGF-I and insulin levels, and increases hepatocyte MIF levels and stress resistance. Aging Cell 2005, 4, 119–125. [Google Scholar] [CrossRef]
- Yoshida, S.; Yamahara, K.; Kume, S.; Koya, D.; Yasuda-Yamahara, M.; Takeda, N.; Osawa, N.; Chin-Kanasaki, M.; Adachi, Y.; Nagao, K.; et al. Role of dietary amino acid balance in diet restriction-mediated lifespan extension, renoprotection, and muscle weakness in aged mice. Aging Cell 2018, 17, e12796. [Google Scholar] [CrossRef]
- Sanchez-Roman, I.; Gomez, A.; Gomez, J.; Suarez, H.; Sanchez, C.; Naudi, A.; Ayala, V.; Portero-Otin, M.; Lopez-Torres, M.; Pamplona, R.; et al. Forty percent methionine restriction lowers DNA methylation, complex I ROS generation, and oxidative damage to mtDNA and mitochondrial proteins in rat heart. J. Bioenergy Biomembr 2011, 43, 699–708. [Google Scholar] [CrossRef]
- Caro, P.; Gomez, J.; Lopez-Torres, M.; Sanchez, I.; Naudi, A.; Jove, M.; Pamplona, R.; Barja, G. Forty percent and eighty percent methionine restriction decrease mitochondrial ROS generation and oxidative stress in rat liver. Biogerontology 2008, 9, 183–196. [Google Scholar] [CrossRef]
- Caro, P.; Gomez, J.; Sanchez, I.; Naudi, A.; Ayala, V.; López-Torres, M.; Pamplona, R.; Barja, G. Forty percent methionine restriction decreases mitochondrial oxygen radical production and leak at complex I during forward electron flow and lowers oxidative damage to proteins and mitochondrial DNA in rat kidney and brain mitochondria. Rejuvenation Res. 2009, 12, 421–434. [Google Scholar] [CrossRef]
- Naudí, A.; Caro, P.; Jové, M.; Gómez, J.; Boada, J.; Ayala, V.; Portero-Otín, M.; Barja, G.; Pamplona, R. Methionine Restriction Decreases Endogenous Oxidative Molecular Damage and Increases Mitochondrial Biogenesis and Uncoupling Protein 4 in Rat Brain. Rejuvenation Res. 2007, 10, 473–484. [Google Scholar] [CrossRef]
- Sanz, A.; Caro, P.; Ayala, V.; Portero-Otin, M.; Pamplona, R.; Barja, G. Methionine restriction decreases mitochondrial oxygen radical generation and leak as well as oxidative damage to mitochondrial DNA and proteins. FASEB J. 2006, 20, 1064–1073. [Google Scholar] [CrossRef] [PubMed]
- Perrone, C.E.; Malloy, V.L.; Orentreich, D.S.; Orentreich, N. Metabolic adaptations to methionine restriction that benefit health and lifespan in rodents. Exp. Gerontol. 2013, 48, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Orentreich, N.; Matias, J.; DeFelice, A.; Zimmerman, J. Low methionine ingestion by rats extends life span. J. Nutr. 1993, 123, 269–274. [Google Scholar] [PubMed]
- Zimmerman, J.; Malloy, V.; Krajcik, R.; Orentreich, N. Nutritional control of aging. Exp. Gerontol. 2003, 38, 47–52. [Google Scholar] [CrossRef]
- Malloy, V.L.; Krajcik, R.A.; Bailey, S.J.; Hristopoulos, G.; Plummer, J.D.; Orentreich, N. Methionine restriction decreases visceral fat mass and preserves insulin action in aging male Fischer 344 rats independent of energy restriction. Aging Cell 2006, 5, 305–314. [Google Scholar] [CrossRef]
- Perrone, C.E.; Mattocks, D.A.L.; Jarvis-Morar, M.; Plummer, J.D.; Orentreich, N. Methionine restriction effects on mitochondrial biogenesis and aerobic capacity in white adipose tissue, liver, and skeletal muscle of F344 rats. Metabolism 2010, 59, 1000–1011. [Google Scholar] [CrossRef]
- Perrone, C.E.; Mattocks, D.A.L.; Plummer, J.D.; Chittur, S.V.; Mohney, R.; Vignola, K.; Orentreich, D.S.; Orentreich, N. Genomic and metabolic responses to methionine-restricted and methionine-restricted, cysteine-supplemented diets in Fischer 344 rat inguinal adipose tissue, liver and quadriceps muscle. J. Nutrigenet. Nutrigenom. 2012, 5, 132–157. [Google Scholar] [CrossRef]
- Hasek, B.E.; Stewart, L.K.; Henagan, T.M.; Boudreau, A.; Lenard, N.R.; Black, C.; Shin, J.; Huypens, P.; Malloy, V.L.; Plaisance, E.P.; et al. Dietary methionine restriction enhances metabolic flexibility and increases uncoupled respiration in both fed and fasted states. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 299, R728–R739. [Google Scholar] [CrossRef]
- Hasek, B.E.; Boudreau, A.; Shin, J.; Feng, D.; Hulver, M.; Van, N.T.; Laque, A.; Stewart, L.K.; Stone, K.P.; Wanders, D.; et al. Remodeling the integration of lipid metabolism between liver and adipose tissue by dietary methionine restriction in rats. Diabetes 2013, 62, 3362–3372. [Google Scholar] [CrossRef]
- Lees, E.K.; Król, E.; Grant, L.; Shearer, K.; Wyse, C.; Moncur, E.; Bykowska, A.S.; Mody, N.; Gettys, T.W.; Delibegovic, M. Methionine restriction restores a younger metabolic phenotype in adult mice with alterations in fibroblast growth factor 21. Aging Cell 2014, 13, 817–827. [Google Scholar] [CrossRef]
- Ghosh, S.; Forney, L.A.; Wanders, D.; Stone, K.P.; Gettys, T.W. An integrative analysis of tissue-specific transcriptomic and metabolomic responses to short-term dietary methionine restriction in mice. PLoS ONE 2017, 12, e0177513. [Google Scholar] [CrossRef] [PubMed]
- Lees, E.K.; Banks, R.; Cook, C.; Hill, S.; Morrice, N.; Grant, L.; Mody, N.; Delibegovic, M. Direct comparison of methionine restriction with leucine restriction on the metabolic health of C57BL/6J mice. Sci. Rep. 2017, 7, 9977. [Google Scholar] [CrossRef] [PubMed]
- Jové, M.; Ayala, V.; Ramírez-Núñez, O.; Naudí, A.; Cabré, R.; Spickett, C.M.; Portero-Otín, M.; Pamplona, R. Specific lipidome signatures in central nervous system from methionine-restricted mice. J. Proteom. Res. 2013, 12, 2679–2689. [Google Scholar] [CrossRef] [PubMed]
- Hulbert, A.J.; Pamplona, R.; Buffenstein, R.; Buttemer, W.A. Life and Death: Metabolic Rate, Membrane Composition, and Life Span of Animals. Physiol. Rev. 2007, 87, 1175–1213. [Google Scholar] [CrossRef]
- Houtkooper, R.H.; Argmann, C.; Houten, S.M.; Cantó, C.; Jeninga, E.H.; Andreux, P.A.; Thomas, C.; Doenlen, R.; Schoonjans, K.; Auwerx, J. The metabolic footprint of aging in mice. Sci. Rep. 2011, 1, 134. [Google Scholar] [CrossRef]
- Yu, Z.; Zhai, G.; Singmann, P.; He, Y.; Xu, T.; Prehn, C.; Römisch-Margl, W.; Lattka, E.; Gieger, C.; Soranzo, N.; et al. Human serum metabolic profiles are age dependent. Aging Cell 2012, 11, 960–967. [Google Scholar] [CrossRef]
- Gonzalez-Covarrubias, V. Lipidomics in longevity and healthy aging. Biogerontology 2013, 14, 663–672. [Google Scholar] [CrossRef]
- Tomás-Loba, A.; Bernardes de Jesus, B.; Mato, J.M.; Blasco, M.A. A metabolic signature predicts biological age in mice. Aging Cell 2013, 12, 93–101. [Google Scholar] [CrossRef]
- De Guzman, J.M.; Ku, G.; Fahey, R.; Youm, Y.-H.; Kass, I.; Ingram, D.K.; Dixit, V.D.; Kheterpal, I. Chronic caloric restriction partially protects against age-related alteration in serum metabolome. Age 2013, 35, 1091–1104. [Google Scholar] [CrossRef]
- Lee, S.H.; Park, S.; Kim, H.-S.; Jung, B.H. Metabolomic approaches to the normal aging process. Metabolomics 2014, 10, 1268–1292. [Google Scholar] [CrossRef]
- Cheng, S.; Larson, M.G.; McCabe, E.L.; Murabito, J.M.; Rhee, E.P.; Ho, J.E.; Jacques, P.F.; Ghorbani, A.; Magnusson, M.; Souza, A.L.; et al. Distinct metabolomic signatures are associated with longevity in humans. Nat. Commun. 2015, 6, 6791. [Google Scholar] [CrossRef] [PubMed]
- Jové, M.; Naudí, A.; Gambini, J.; Borras, C.; Cabré, R.; Portero-Otín, M.; Viña, J.; Pamplona, R. A Stress-Resistant Lipidomic Signature Confers Extreme Longevity to Humans. J. Gerontol. A. Biol. Sci. Med. Sci. 2017, 72, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Cabré, R. Vulnerabilitat Neuronal Selectiva associada a L’envelliment Fisiològic del Sistema Nervioso Central. Ph.D. Thesis, University of Lleida, Lleida, Spain, 2015. [Google Scholar]
- Dean, J.M.; Lodhi, I.J. Structural and functional roles of ether lipids. Protein Cell 2018, 9, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Lancaster, G.I.; Meikle, P.J. Plasmalogens: A potential therapeutic target for neurodegenerative and cardiometabolic disease. Prog. Lipid Res. 2019, 100993. [Google Scholar] [CrossRef] [PubMed]
- Milhas, D.; Clarke, C.J.; Hannun, Y.A. Sphingomyelin metabolism at the plasma membrane: Implications for bioactive sphingolipids. FEBS Lett. 2010, 584, 1887–1894. [Google Scholar] [CrossRef]
- D’Angelo, G.; Capasso, S.; Sticco, L.; Russo, D. Glycosphingolipids: Synthesis and functions. FEBS J. 2013, 280, 6338–6353. [Google Scholar] [CrossRef]
- Russo, D.; Parashuraman, S.; D’Angelo, G. Glycosphingolipid–Protein Interaction in Signal Transduction. Int. J. Mol. Sci. 2016, 17, 1732. [Google Scholar] [CrossRef]
- Leng, S.; Winter, T.; Aukema, H.M. Dietary LA and sex effects on oxylipin profiles in rat kidney, liver, and serum differ from their effects on PUFAs. J. Lipid Res. 2017, 58, 1702–1712. [Google Scholar] [CrossRef]
- Anthony, T.G.; Morrison, C.D.; Gettys, T.W. Remodeling of Lipid Metabolism by Dietary Restriction of Essential Amino Acids. Diabetes 2013, 62, 2635–2644. [Google Scholar] [CrossRef]
- Gómez, J.; Caro, P.; Naudí, A.; Portero-Otin, M.; Pamplona, R.; Barja, G. Effect of 8.5% and 25% caloric restriction on mitochondrial free radical production and oxidative stress in rat liver. Biogerontology 2007, 8, 555–566. [Google Scholar] [CrossRef]
- Plaisance, E.P.; Greenway, F.L.; Boudreau, A.; Hill, K.L.; Johnson, W.D.; Krajcik, R.A.; Perrone, C.E.; Orentreich, N.; Cefalu, W.T.; Gettys, T.W. Dietary methionine restriction increases fat oxidation in obese adults with metabolic syndrome. J. Clin. Endocrinol. Metab. 2011, 96, E836–E840. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; He, L.; Wan, D.; Yang, H.; Yao, K.; Wu, G.; Wu, X.; Yin, Y. Methionine restriction on lipid metabolism and its possible mechanisms. Amino Acids 2016, 48, 1533–1540. [Google Scholar] [CrossRef] [PubMed]
- Cefalu, W.T.; Wang, Z.Q.; Bell–Farrow, A.D.; Terry, J.G.; Sonntag, W.; Waite, M.; Parks, J. Chronic caloric restriction alters muscle membrane fatty acid content. Exp. Gerontol. 2000, 35, 331–341. [Google Scholar] [CrossRef]
- Tacconi, M.T.; Lligoña, L.; Salmona, M.; Pitsikas, N.; Algeri, S. Aging and food restriction: Effect on lipids of cerebral cortex. Neurobiol. Aging 1991, 12, 55–59. [Google Scholar] [CrossRef]
- Jeon, T.I.; Lim, B.O.; Yu, B.P.; Lim, Y.; Jeon, E.J.; Park, D.K. Effect of dietary restriction on age-related increase of liver susceptibility to peroxidation in rats. Lipids 2001, 36, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Pamplona, R.; Barja, G.; Portero-Otín, M. Membrane fatty acid unsaturation, protection against oxidative stress, and maximum life span: A homeoviscous-longevity adaptation? Ann. N. Y. Acad. Sci. 2002, 959, 475–490. [Google Scholar] [CrossRef]
- Faulks, S.C.; Turner, N.; Else, P.L.; Hulbert, A.J. Calorie restriction in mice: Effects on body composition, daily activity, metabolic rate, mitochondrial reactive oxygen species production, and membrane fatty acid composition. J. Gerontol. A. Biol. Sci. Med. Sci. 2006, 61, 781–794. [Google Scholar] [CrossRef]
- Selman, C.; Kerrison, N.D.; Cooray, A.; Piper, M.D.W.; Lingard, S.J.; Barton, R.H.; Schuster, E.F.; Blanc, E.; Gems, D.; Nicholson, J.K.; et al. Coordinated multitissue transcriptional and plasma metabonomic profiles following acute caloric restriction in mice. Physiol. Genom. 2006, 27, 187–200. [Google Scholar] [CrossRef]
- Jové, M.; Naudí, A.; Ramírez-Núñez, O.; Portero-Otín, M.; Selman, C.; Withers, D.J.; Pamplona, R. Caloric restriction reveals a metabolomic and lipidomic signature in liver of male mice. Aging Cell 2014, 13, 828–837. [Google Scholar] [CrossRef]
- Miller, K.N.; Burhans, M.S.; Clark, J.P.; Howell, P.R.; Polewski, M.A.; DeMuth, T.M.; Eliceiri, K.W.; Lindstrom, M.J.; Ntambi, J.M.; Anderson, R.M. Aging and caloric restriction impact adipose tissue, adiponectin, and circulating lipids. Aging Cell 2017, 16, 497–507. [Google Scholar] [CrossRef]
- Cabré, R.; Naudí, A.; Dominguez-Gonzalez, M.; Ayala, V.; Jové, M.; Mota-Martorell, N.; Piñol-Ripoll, G.; Gil-Villar, M.P.; Rué, M.; Portero-Otín, M.; et al. Sixty years old is the breakpoint of human frontal cortex aging. Free Radic. Biol. Med. 2017, 103, 14–22. [Google Scholar] [CrossRef]
- Pizarro, C.; Arenzana-Rámila, I.; Pérez-del-Notario, N.; Pérez-Matute, P.; González-Sáiz, J.-M. Plasma lipidomic profiling method based on ultrasound extraction and liquid chromatography mass spectrometry. Anal. Chem. 2013, 85, 12085–12092. [Google Scholar] [CrossRef]
- Castro-Perez, J.M.; Kamphorst, J.; DeGroot, J.; Lafeber, F.; Goshawk, J.; Yu, K.; Shockcor, J.P.; Vreeken, R.J.; Hankemeier, T. Comprehensive LC−MS E Lipidomic Analysis using a Shotgun Approach and Its Application to Biomarker Detection and Identification in Osteoarthritis Patients. J. Proteom. Res. 2010, 9, 2377–2389. [Google Scholar] [CrossRef]
- Jové, M.; Serrano, J.C.E.; Ortega, N.; Ayala, V.; Anglès, N.; Reguant, J.; Morelló, J.R.; Romero, M.P.; Motilva, M.J.; Prat, J.; et al. Multicompartmental LC-Q-TOF-Based Metabonomics as an Exploratory Tool to Identify Novel Pathways Affected by Polyphenol-Rich Diets in Mice. J. Proteom. Res. 2011, 10, 3501–3512. [Google Scholar] [CrossRef]
- Sana, T.R.; Roark, J.C.; Li, X.; Waddell, K.; Fischer, S.M. Molecular formula and METLIN Personal Metabolite Database matching applied to the identification of compounds generated by LC/TOF-MS. J. Biomol. Tech. 2008, 19, 258–266. [Google Scholar]
- Xia, J.; Psychogios, N.; Young, N.; Wishart, D.S. MetaboAnalyst: A web server for metabolomic data analysis and interpretation. Nucleic Acids Res. 2009, 37, W652–W660. [Google Scholar] [CrossRef]
- Xia, J.; Mandal, R.; Sinelnikov, I.V.; Broadhurst, D.; Wishart, D.S. MetaboAnalyst 2.0—A comprehensive server for metabolomic data analysis. Nucleic Acids Res. 2012, 40, W127–W133. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Marcu, A.; Guo, A.C.; Liang, K.; Vázquez-Fresno, R.; Sajed, T.; Johnson, D.; Li, C.; Karu, N.; et al. HMDB 4.0: The human metabolome database for 2018. Nucleic Acids Res. 2018, 46, D608–D617. [Google Scholar] [CrossRef]
- Kind, T.; Liu, K.-H.; Lee, D.Y.; DeFelice, B.; Meissen, J.K.; Fiehn, O. LipidBlast in silico tandem mass spectrometry database for lipid identification. Nat. Methods 2013, 10, 755–758. [Google Scholar] [CrossRef]
| Compound | Adult | Aged | MetR |
|---|---|---|---|
| 2-SC | 69.55 ± 7.41 | 68.82 ± 7.80 | 72.58 ± 3.01 |
| GSA | 5232.67 ± 458.74 | 5216.56 ± 178.61 | 5729.25 ± 376.60 |
| AASA | 284.64 ± 37.76 | 276.84 ± 21.80 | 291.35 ± 21.36 |
| CEL | 384.75 ± 42.28 | 322.70 ± 38.12 | 329.99 ± 22.78 |
| MDAL | 228.53 ± 25.27 | 203.44 ± 25.44 | 204.02 ± 43.33 |
| CMC | 103.78 ± 15.42 | 99.27 ± 9.04 | 103.43 ± 7.04 |
| CML | 1595.83 ± 75.82 | 1410.95 ± 123.92 | 1494.69 ± 110.89 |
| Compound | Adduct | m/z | RT | Log FC | p | Aged vs Adult |
|---|---|---|---|---|---|---|
| PC(P-34:2) a | M+H+ | 742.5863 | 7.64 | 7.83 | 0.0253 | up |
| MAG(18:0) a* | M+Na+ | 381.2909 | 4.47 | 13.00 | 0.00159 | up |
| DAG(32:2) a* | M+H+-H2O | 529.4520 | 8.41 | 7.02 | 0.0389 | up |
| TAG(50:2) a | M+NH4+ | 848.7654 | 9.55 | 9.02 | 0.0134 | up |
| TAG(56:8) a | M+H+ | 903.7554 | 9.78 | -8.74 | 0.0372 | down |
| TAG(56:5) a | M+NH4+ | 926.8102 | 10.01 | 9.15 | 0.0242 | up |
| Palmitaldehyde b | M+NH4+ | 258.2738 | 9.11 | 7.20 | 0.00955 | up |
| EET methyl ester b | M+H+-H2O | 317.2477 | 11.83 | -6.73 | 0.0473 | down |
| AA methyl ester b | M+NH4+ | 336.2908 | 11.80 | -6.85 | 0.0275 | down |
| Dihomo-PGI2 b | M+H+-H2O | 363.2525 | 11.47 | -9.34 | 0.00401 | down |
| ADP-ribose b | M+H+-H2O | 542.0597 | 0.58 | -9.47 | 0.0423 | down |
| NADH b | M+Na+ | 688.1027 | 5.90 | -8.42 | 0.0418 | down |
| Compound | m/z | RT | p value | LogFC MetR vs Aged | MetR vs Aged | LogFC MetR vs Adult | MetR vs Adult |
|---|---|---|---|---|---|---|---|
| LysoPE(18:3) * | 493.303 | 0.83 | 0.0197 | 10.67 | up | 5.68 | up |
| LysoPI(15:0) | 559.2942 | 2.97 | 0.0234 | -9.06 | down | -0.03 | down |
| PA(P-30:1) | 585.4237 | 6.41 | 0.00000662 | 14.26 | up | 12.35 | up |
| PC(20:0) | 604.3236 | 2.13 | 0.0128 | 10.73 | up | 7.02 | up |
| PC(38:6) * | 823.5946 | 5.58 | 0.00246 | 8.39 | up | 0.07 | up |
| PC(42:2) | 870.688 | 7.40 | 0.0478 | -7.80 | down | -7.05 | down |
| PC(P-38:2) | 820.6198 | 8.70 | 0.00248 | -7.57 | down | -0.07 | down |
| PG(32:1) | 721.5002 | 7.72 | 0.0397 | 0.05 | up | -0.12 | down |
| PS(P-29:0) | 642.453 | 5.67 | 0.03 | -5.80 | down | 1.62 | up |
| GlcAβ-Cer(d36:1) | 742.5863 | 7.64 | 0.002 | -10.15 | down | -1.76 | down |
| SM(d30:0) * | 666.5661 | 8.41 | 0.02 | 9.08 | up | 7.20 | up |
| MAG(20:3) | 381.2906 | 4.48 | 0.00482 | -6.16 | down | 8.82 | up |
| DAG(32:2) * | 529.4533 | 8.41 | 0.0173 | 0.61 | up | 7.37 | up |
| TAG(40:2) | 848.7654 | 9.56 | 0.00188 | -10.11 | down | 1.85 | up |
| TAG(61:6) | 941.8264 | 9.94 | 0.0235 | 11.11 | up | 1.74 | up |
| Compound | m/z | RT | p value | LogFC MetR vs Aged | MetR vs Aged | LogFC MetR vs Adult | MetR vs Adult |
|---|---|---|---|---|---|---|---|
| Methylcytidine | 258.0986 | 0.48 | 0.0224 | -2.27 | down | 0.69 | up |
| ADP-ribose | 542.0597 | 0.58 | 0.0036 | -0.02 | down | -2.36 | down |
| Inosine | 269.0814 | 0.67 | 0.0291 | 0.01 | up | -0.12 | down |
| Glutathione disulfide | 613.1499 | 0.46 | 0.0169 | -2.46 | down | -0.51 | down |
| Angiotensin (1-7) | 937.4213 | 11.50 | 0.0375 | 2.29 | up | 2.58 | up |
| epoxyHDHA | 399.2466 | 10.04 | 0.0183 | 2.62 | up | 0.48 | up |
| 10,11-DiHDPE | 363.2511 | 11.47 | 0.0236 | 2.35 | up | 0.05 | up |
| 20-carboxy-LTB4 | 367.2129 | 7.33 | 0.0143 | -0.11 | down | 0.00 | down |
| 6-keto PGE1 | 369.2334 | 6.89 | 0.0192 | -2.37 | down | -0.51 | down |
| 5β-Cholestane-3α,7α-diol | 427.3541 | 13.89 | 0.0242 | -0.07 | down | 1.98 | up |
| Glycocholic Acid | 466.3105 | 8.93 | 0.000176 | 2.89 | up | 2.85 | up |
| Dodecenoyl-CoA | 912.2679 | 13.69 | 0.0435 | -1.18 | down | 1.03 | up |
| Formyl-CoA | 778.0916 | 10.56 | 0.0215 | 0.55 | up | -1.75 | down |
| Palmitaldehyde | 258.2745 | 9.11 | 0.00461 | 0.01 | up | 2.69 | up |
| Propionylcarnitine | 235.1639 | 9.19 | 0.0278 | 0.00 | up | 2.36 | up |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pradas, I.; Jové, M.; Cabré, R.; Ayala, V.; Mota-Martorell, N.; Pamplona, R. Effects of Aging and Methionine Restriction on Rat Kidney Metabolome. Metabolites 2019, 9, 280. https://doi.org/10.3390/metabo9110280
Pradas I, Jové M, Cabré R, Ayala V, Mota-Martorell N, Pamplona R. Effects of Aging and Methionine Restriction on Rat Kidney Metabolome. Metabolites. 2019; 9(11):280. https://doi.org/10.3390/metabo9110280
Chicago/Turabian StylePradas, Irene, Mariona Jové, Rosanna Cabré, Victoria Ayala, Natalia Mota-Martorell, and Reinald Pamplona. 2019. "Effects of Aging and Methionine Restriction on Rat Kidney Metabolome" Metabolites 9, no. 11: 280. https://doi.org/10.3390/metabo9110280
APA StylePradas, I., Jové, M., Cabré, R., Ayala, V., Mota-Martorell, N., & Pamplona, R. (2019). Effects of Aging and Methionine Restriction on Rat Kidney Metabolome. Metabolites, 9(11), 280. https://doi.org/10.3390/metabo9110280





