Gas Chromatography Mass Spectrometry (GC-MS) Quantification of Metabolites in Stool Using 13C Labelled Compounds
Abstract
1. Introduction
2. Results
2.1. Qualitative Data from Unmodified Stools
2.2. Quantitative Data from Unaltered Stools
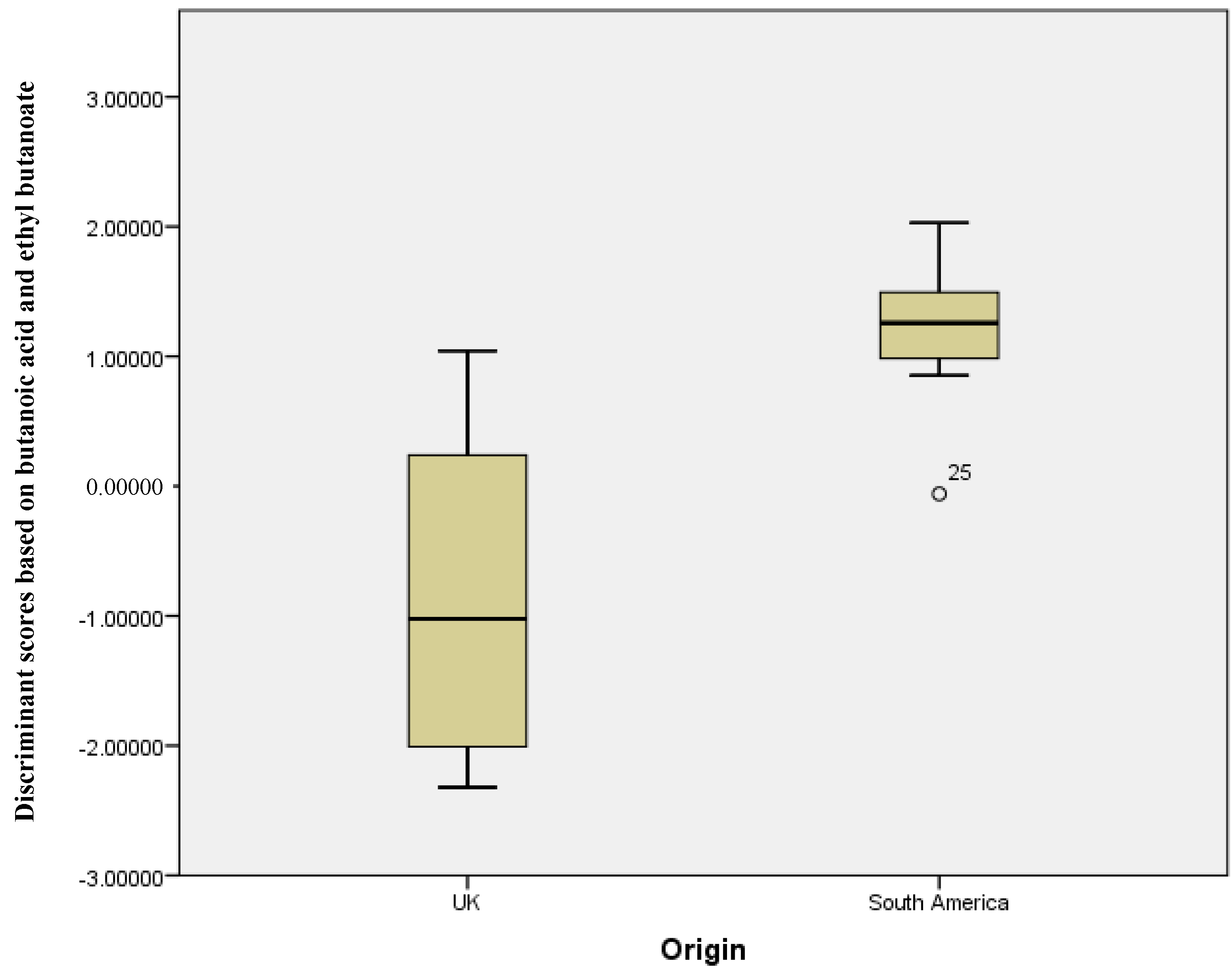
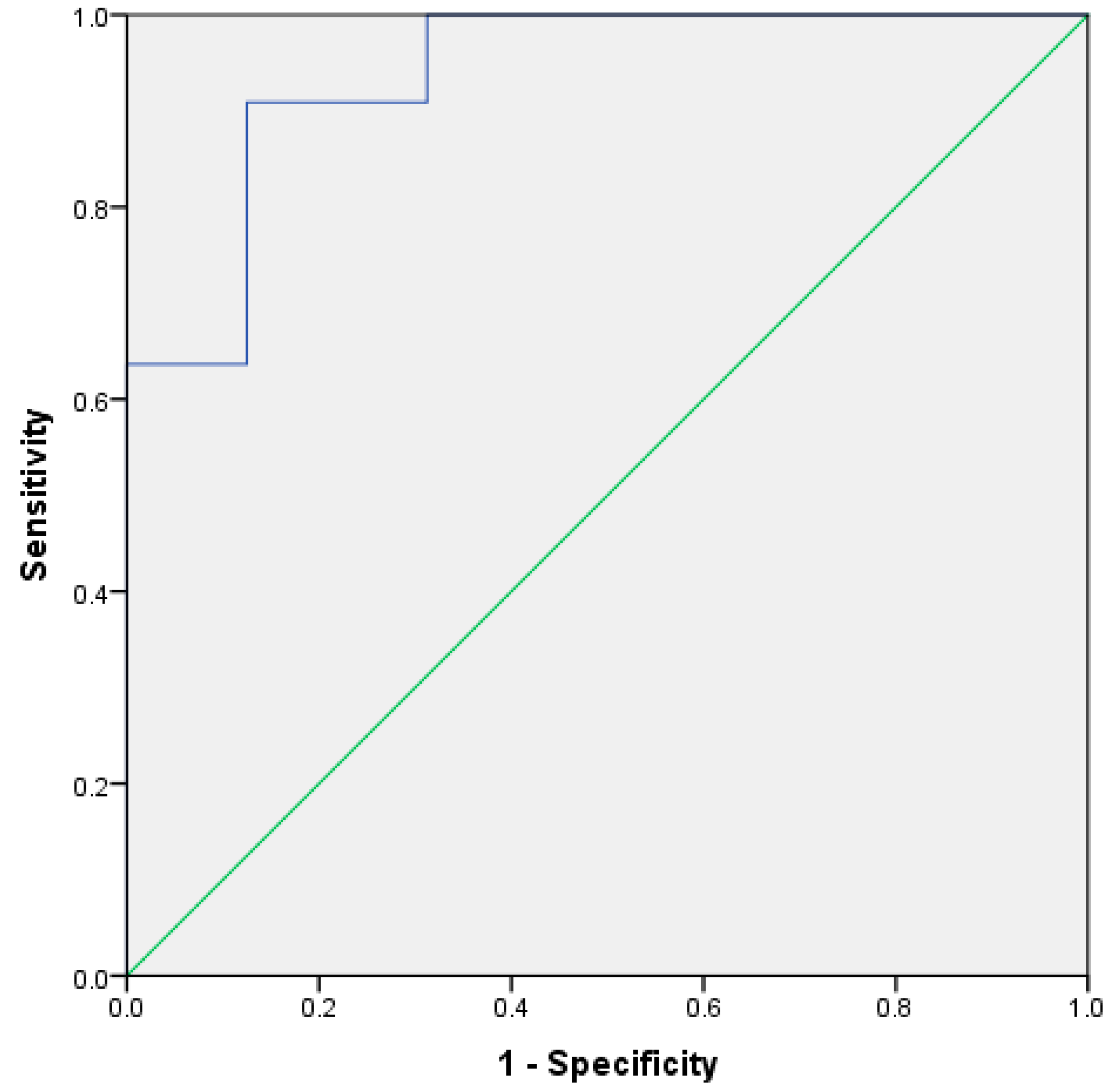
2.3. UK Samples versus South America Samples
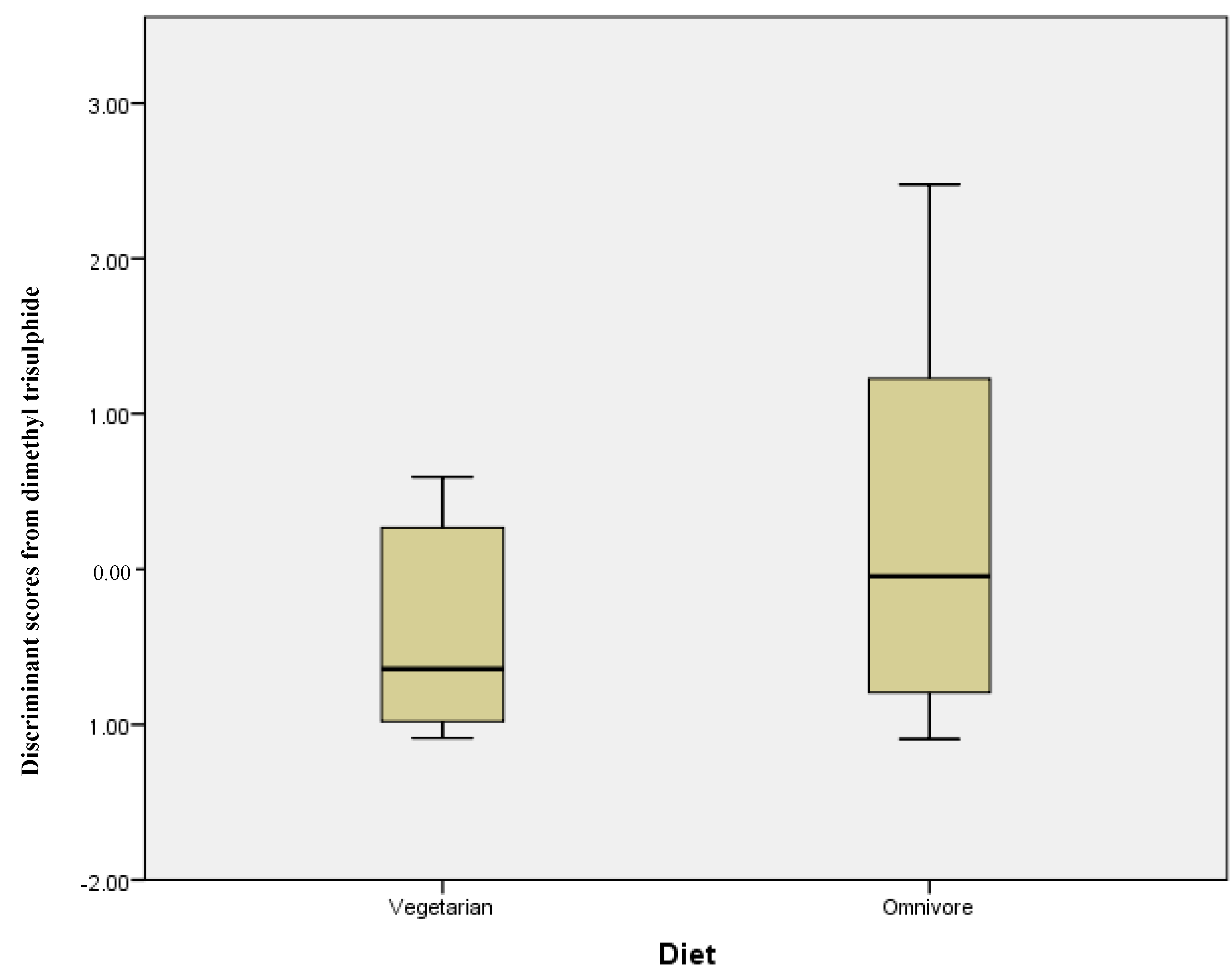
2.4. Omnivore versus Vegetarian
2.4.1. Qualitative Data from Alkaline (pH 13) Stools
2.4.2. Quantitative Data from Alkaline (pH 13) Stools
3. Discussion
4. Materials and Methods
4.1. Instrumentation and Separation Methodology
4.2. Loading the Thermal Desorption (TD) Tubes
4.3. Standard Solutions
4.4. Calibration Curves
4.5. Sample Preparation
4.6. Trimethylamine Quantification in Ph13 Stool Samples
4.7. Analysis
4.7.1. Qualitative Analysis
4.7.2. Mass Calculations Using 13C Labelled Compounds
4.7.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
References
- Garner, C.E.; Smith, S.; Bardhan, P.K.; Ratcliffe, N.M.; Probert, C.S.J. A pilot study of faecal volatile organic compounds in faeces from cholera patients in Bangladesh to determine their utility in disease diagnosis. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 1171–1173. [Google Scholar] [CrossRef] [PubMed]
- Garner, C.E.; Smith, S.; de Lacy Costello, B.; White, P.; Spencer, R.; Probert, C.S.J.; Ratcliffe, N.M. Volatile organic compounds from feces and their potential for diagnosis of gastrointestinal disease. FASEB J. 2007, 21, 1675–1688. [Google Scholar] [CrossRef] [PubMed]
- Probert, C.S.; Reade, S.; Ahmed, I. Fecal volatile organic compounds: A novel, cheaper method of diagnosing inflammatory bowel disease? Expert Rev. Clin. Immunol. 2014, 10, 1129–1131. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, I.; Greenwood, R.; de Lacy Costello, B.; Ratcliffe, N.M.; Probert, C.S. An Investigation of Fecal Volatile Organic Metabolites in Irritable Bowel Syndrome. PLoS ONE 2013, 8, e58204. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, S.F.; McGuire, N.D.; de Lacy Costello, B.P.J.; Ewen, R.J.; Jayasena, D.H.; Vaughan, K.; Ahmed, I.; Probert, C.S.; Ratcliffe, N.M. The use of a gas chromatograph coupled to a metal oxide sensor for rapid assessment of stool samples from irritable bowel syndrome and inflammatory bowel disease patients. J. Breath Res. 2014, 8, 026001. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-J.; Li, S.; Gan, R.-Y.; Zhou, T.; Xu, D.-P.; Li, H.-B. Impacts of Gut Bacteria on Human Health and Diseases. Int. J. Mol. Sci. 2015, 16, 7493–7519. [Google Scholar] [CrossRef] [PubMed]
- Amann, A.; Costello Bde, L.; Miekisch, W.; Schubert, J.; Buszewski, B.; Pleil, J.; Ratcliffe, N.; Risby, T. The human volatilome: Volatile organic compounds (VOCs) in exhaled breath, skin emanations, urine, feces and saliva. J. Breath Res. 2014, 8, 034001. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Links between diet, gut microbiota composition and gut metabolism. Proc. Nutr. Soc. 2015, 74, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Silvester, K.R.; Bingham, S.A.; Pollock, J.R.A.; Cummings, J.H.; Neill, I.K.O. Effect of meat and resistant starch on fecal excretion of apparent N-nitroso compounds and ammonia from the human large bowel Effect of Meat and Resistant Starch on Fecal Excretion of Apparent N-Nitroso Compounds and Ammonia From the Human Large Bowel. Nutr. Cancer 2009, 29, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Sethi, S.; Nanda, R.; Chakraborty, T. Clinical application of volatile organic compound analysis for detecting infectious diseases. Clin. Microbiol. Rev. 2013, 26, 462–475. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Aoll, J.; Niclass, Y.; Velazco, M.I.; Wünsche, L.; Pika, J.; Starkenmann, C. Qualitative and Quantitative Analysis of Volatile Constituents from Latrines. Environ. Sci. Technol. 2013, 47, 7876–7882. [Google Scholar] [CrossRef] [PubMed]
- Walton, C.; Fowler, D.P.; Turner, C.; Jia, W.; Whitehead, R.N.; Griffiths, L.; Dawson, C.; Waring, R.H.; Ramsden, D.B.; Cole, J.A.; et al. Analysis of Volatile Organic Compounds of Bacterial Origin in Chronic Gastrointestinal Diseases. Inflamm. Bowel Dis. 2013, 19, 2069–2078. [Google Scholar] [CrossRef] [PubMed]
- Baranska, A.; Mujagic, Z.; Smolinska, A.; Dallinga, J.W.; Jonkers, D.M.A.E.; Tigchelaar, E.F.; Dekens, J.; Zhernakova, A.; Ludwig, T.; Masclee, A.A.M.; et al. Volatile organic compounds in breath as markers for irritable bowel syndrome: A metabolomic approach. Aliment. Pharmacol. Ther. 2016, 44, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Christophersen, C.T.; Sorich, M.J.; Gerber, J.P.; Angley, M.T.; Conlon, M.A. Elevated Fecal Short Chain Fatty Acid and Ammonia Concentrations in Children with Autism Spectrum Disorder. Dig. Dis. Sci. 2012, 57, 2096–2102. [Google Scholar] [CrossRef] [PubMed]
- De Preter, V.; Van Staeyen, G.; Esser, D.; Rutgeerts, P.; Verbeke, K. Development of a screening method to determine the pattern of fermentation metabolites in faecal samples using on-line purge-and-trap gas chromatographic-mass spectrometric analysis. J. Chromatogr. A 2009, 1216, 1476–1483. [Google Scholar] [CrossRef] [PubMed]
- Shreiner, A.B.; Kao, J.Y.; Young, V.B. The gut microbiome in health and in disease. Curr. Opin. Gastroenterol. 2015, 31, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Probert, C.S.J.; Jones, P.R.H.; Probert, S.J. A novel method for rapidly diagnosing the causes of diarrhoea. Gut 2004, 53, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Shoji, H.; Sato, H.; Nagata, S.; Ohtsuka, Y.; Shimizu, T.; Yamashiro, Y. Effects of Oral Administration of Bifidobacterium breve on Fecal Lactic Acid and Short-chain Fatty Acids in Low Birth Weight Infants. J. Pediatr. Gastroenterol. Nutr. 2007, 44, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Primec, M.; Mičetić-Turk, D.; Langerholc, T. Analysis of short-chain fatty acids in human feces: A scoping review. Anal. Biochem. 2017, 526, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Fordtran, J. Organic anions in fecal contents. N. Engl. J. Med. 1971, 284, 329–330. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Carrio, J.; Salazar, N.; Margolles, A.; González, S.; Gueimonde, M.; de los Reyes-Gavilán, C.G.; Suárez, A. Free Fatty Acids Profiles Are Related to Gut Microbiota Signatures and Short-Chain Fatty Acids. Front. Immunol. 2017, 8, 823. [Google Scholar] [CrossRef] [PubMed]
- Simenhoff, M.L.; Burke, J.F.; Saukkonen, J.J.; Ordinario, A.T.; Doty, R.; Dunn, S. Biochemical Profile of Uremic Breath. N. Engl. J. Med. 1977, 297, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Davies, S.; Spanel, P.; Smith, D. Quantitative analysis of ammonia on the breath of patients in end-stage renal failure. Kidney Int. 1997, 52, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Ong, J.P.; Aggarwal, A.; Krieger, D.; Easley, K.A.; Karafa, M.T.; van Lente, F.; Arroliga, A.C.; Mullen, K.D. Correlation between Ammonia Levels and the Severity of Hepatic Encephalopathy. Am. J. Med. 2003, 114, 188–193. [Google Scholar] [CrossRef]
- de Lacy Costello, B.; Amann, A.; Al-Kateb, H.; Flynn, C.; Filipiak, W.; Khalid, T.; Osborne, D.; Ratcliffe, N.M. A review of the volatiles from the healthy human body. J. Breath Res. 2014, 8, 14001–14029. [Google Scholar] [CrossRef] [PubMed]
- de Filippo, C.; Cavalieri, D.; di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef] [PubMed]
- Kwok, L.; Zhang, J.; Guo, Z.; Gesudu, Q.; Zheng, Y.; Qiao, J.; Huo, D.; Zhang, H. Characterization of Fecal Microbiota across Seven Chinese Ethnic Groups by Quantitative Polymerase Chain Reaction. PLoS ONE 2014, 9, e93631. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.; Saunier, K.; Hanisch, C.; Norin, E.; Alm, L.; Midtvedt, T.; Cresci, A.; Silvi, S.; Orpianesi, C.; Verdenelli, M.C.; et al. Differences in fecal microbiota in different European study populations in relation to age, gender, and country: A cross-sectional study. Appl. Environ. Microbiol. 2006, 72, 1027–1033. [Google Scholar] [CrossRef] [PubMed]
- Kabeerdoss, J.; Shobana Devi, R.; Regina Mary, R.; Ramakrishna, B.S. Faecal microbiota composition in vegetarians: Comparison with omnivores in a cohort of young women in southern India. Br. J. Nutr. 2012, 108, 953–957. [Google Scholar] [CrossRef] [PubMed]
- Van Faassen, A.; Hazen, M.J.; van den Brandt, P.A.; van den Bogaard, A.E.; Hermus, R.J.; Janknegt, R.A. Bile acids and pH values in total feces and in fecal water from habitually omnivorous and vegetarian subjects. Am. J. Clin. Nutr. 1993, 58, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Ferrocino, I.; di Cagno, R.; de Angelis, M.; Turroni, S.; Vannini, L.; Bancalari, E.; Rantsiou, K.; Cardinali, G.; Neviani, E.; Cocolin, L. Fecal Microbiota in Healthy Subjects Following Omnivore, Vegetarian and Vegan Diets: Culturable Populations and rRNA DGGE Profiling. PLoS ONE 2015, 10, e0128669. [Google Scholar] [CrossRef] [PubMed]
- Falasconi, M.; Pardo, M.; Sberveglieri, G.; Battistutta, F.; Piloni, M.; Zironi, R. Study of white truffle aging with SPME-GC-MS and the Pico2-electronic nose. Sens. Actuators B Chem. 2005, 106, 88–94. [Google Scholar] [CrossRef]
- Wernig, F.; Buegger, F.; Pritsch, K.; Splivallo, R. Composition and authentication of commercial and home-made white truffle-flavored oils. Food Control 2018, 87, 9–16. [Google Scholar] [CrossRef]
- Cyclohexanol, 5-methyl-2-(1-methylethyl)-, (1α,2β,5α)-. NIST Chemistry Webbook, SRD 69. Available online: https://webbook.nist.gov/cgi/cbook.cgi?ID=C15356704 (accessed on 24 October 2018).
- Masood, N.; Chaudhry, A.; Tariq, P. In vitro antibacterial activities of kalonji, cumin and poppy seed. Pak. J. Bot. 2008, 40, 461–467. [Google Scholar]
- Methyl Isopropyl Ketone | C5H10O—PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/11251 (accessed on 24 October 2018).
- Bianchi, F.; Careri, M.; Mangia, A.; Musci, M. Retention indices in the analysis of food aroma volatile compounds in temperature-programmed gas chromatography: Database creation and evaluation of precision and robustness. J. Sep. Sci. 2007, 30, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.T.; Yang, Z.C.; Ding, S.F. Prediction of retention indexes: II. Structure-retention index relationship on polar columns. J. Chromatogr. A 1991, 586, 85–112. [Google Scholar] [CrossRef]
- Schluter, S.; Steinhart, H.; Schwarz, F.J.; Kirchgessner, M. Changes in the Odorants of Boiled Carp Fillet (Cyprinus carpio L.) as Affected by Increasing Methionine Levels in Feed. J. Agric. Food Chem. 1999, 40, 461–467. [Google Scholar] [CrossRef]
- Babushok, V.I.; Linstrom, P.J.; Zenkevich, I.G. Retention Indices for Frequently Reported Compounds of Plant Essential Oils. J. Phys. Chem. Ref. Data 2011, 40, 043101. [Google Scholar] [CrossRef]
- Liu, J.-L.; Yan, C.-W.; Li, Y.-T.; Wu, Z.-Y.; Zhang, W.-J. Diacetonitrile-1κN,3κN-bis{μ-trans-N-[3-(dimethylamino)propyl]-N′-(2-hydroxyethyl)oxamidato(2–)}-1:2κ5N,N′,O:O′,N′′;2:3κ5O′,N′′:N,N′,O-dithiocyanato-1κN,3κN-tricopper(II). Acta Crystallogr. Sect. C Cryst. Struct. Commun. 2008, 64, m149–m152. [Google Scholar] [CrossRef] [PubMed]
| Origin | Number of Samples | Number of Times Detected | Mean (ng/g) | Std. Deviation (ng/g) | Range (ng/g) | |
|---|---|---|---|---|---|---|
| Dimethyl sulphide CAS 75-18-3 | Total | 38 | 37 | 3058 | 4531 | 25–25,626 |
| UK | 16 | 16 | 2474 | 2913 | 26–8637 | |
| South America | 11 | 10 | 2890 | 2780 | 621–8079 | |
| Acetone CAS 67-64-1 | Total | 38 | 38 | 1481 | 626 | 442–3005 |
| UK | 16 | 16 | 1335 | 614 | 442–2521 | |
| South America | 11 | 11 | 1652 | 487 | 819–2377 | |
| Ethyl butanoate CAS 105-54-4 | Total | 38 | 21 | 352 | 659 | 39–2468 |
| UK | 16 | 14 | 424 | 714 | 39–2468 | |
| South America | 11 | 3 | 121 | 263 | 118–828 | |
| Ethyl 2-methylbutanoate CAS 7452-79-1 | Total | 38 | 12 | 8 | 29 | 0.34–180 |
| UK | 16 | 10 | 16 | 44 | 0.3–180 | |
| South America | 11 | 2 | 4 | 12.8 | 9–42 | |
| Dimethyl disulphide CAS 624-92-0 | Total | 38 | 38 | 479 | 406 | 35–1302 |
| UK | 16 | 16 | 301 | 275 | 48–864 | |
| South America | 11 | 11 | 583 | 445 | 36–1313 | |
| 1-Octen-3-one CAS 4312-99-6 | Total | 38 | 1 | 0.31 | 2 | 12 |
| UK | 16 | 1 | 0.74 | 3 | 12 | |
| South America | 11 | 0 | 0.00 | 0 | Not detected | |
| Dimethyl trisulphide CAS 3658-80-8 | Total | 38 | 38 | 132 | 116 | 10–409 |
| UK | 16 | 16 | 81 | 69 | 10–253 | |
| South America | 11 | 11 | 156 | 119 | 13–364 | |
| 1-Octen-3-ol CAS 3391-86-4 | Total | 38 | 13 | 4 | 13 | 0.4–58 |
| UK | 16 | 10 | 11 | 18 | 1–58 | |
| South America | 11 | 3 | 0.35 | 0.72 | 0.4–2 | |
| Ethanoic acid CAS 64-19-7 | Total | 38 | 38 | 8756 | 2246 | 672–12,963 |
| UK | 16 | 16 | 7400 | 2607 | 672–11,343 | |
| South America | 11 | 11 | 9583 | 1411 | 7968–12,963 | |
| Butanoic acid CAS 107-92-6 | Total | 38 | 38 | 7556 | 2310 | 2493–11,553 |
| UK | 16 | 16 | 5783 | 2160 | 2493–8376 | |
| South America | 11 | 11 | 8740 | 800 | 7043–9953 | |
| 3-Methylbutanoic acid CAS 503-74-2 | Total | 38 | 36 | 2726 | 2210 | 63–8262 |
| UK | 16 | 16 | 1363 | 1029 | 64–3602 | |
| South America | 11 | 10 | 3391 | 2555 | 220–6885 | |
| Pentanoic acid CAS 109-52-4 | Total | 38 | 37 | 4443 | 4374 | 88–21,886 |
| UK | 16 | 16 | 2078 | 1720 | 88–5970 | |
| South America | 11 | 11 | 7494 | 5879 | 946–21,886 | |
| Indole CAS 120-72-9 | Total | 38 | 38 | 3717 | 1119 | 290–5477 |
| UK | 16 | 16 | 3002 | 797 | 1508–4309 | |
| South America | 11 | 11 | 4174 | 649 | 3286–5477 | |
| 3-Methyl-1H-indole CAS 83-34-1 | Total | 38 | 38 | 1323 | 776 | 37–3483 |
| UK | 16 | 16 | 1340 | 799 | 425–3054 | |
| South America | 11 | 11 | 1703 | 777 | 636–3483 | |
| Trimethylamine CAS 75-50-3 | Total | 15 | 15 | 241 | 157 | 40–920.5 |
| Compound | Sig. |
|---|---|
| Ethyl butanoate | 0.023 |
| 1-Octen-3-ol | 0.034 |
| Ethanoic acid | 0.011 |
| Butanoic acid | 0.000 |
| Pentanoic acid | 0.001 |
| Indole | 0.000 |
| Diet | Number of Samples | Number of Times Detected | Mean (ng/g) | Std. Deviation (ng/g) | Range (ng/g) | |
|---|---|---|---|---|---|---|
| Dimethyl sulphide 75-18-3 | Vegetarian | 14 | 14 | 4182 | 6818 | 91–25,626 |
| Omnivore | 24 | 23 | 2403 | 2354 | 26–8089 | |
| Acetone CAS 67-64-1 | Vegetarian | 14 | 14 | 1285 | 522 | 442–2287 |
| Omnivore | 24 | 24 | 1595 | 663 | 453–3006 | |
| Ethyl butanoate CAS 105-54-4 | Vegetarian | 14 | 7 | 243 | 511 | 39–1659 |
| Omnivore | 24 | 14 | 415 | 735 | 40–2468 | |
| Ethyl 2-methylbutanoate CAS 7452-79-1 | Vegetarian | 14 | 4 | 1 | 4 | 0.3–13 |
| Omnivore | 24 | 8 | 12 | 37 | 8–180 | |
| Dimethyl disulphide CAS 624-92-0 | Vegetarian | 14 | 14 | 355 | 335 | 37–1058 |
| Omnivore | 24 | 24 | 553 | 432 | 35–1303 | |
| 1-Octen-3-one CAS 4312-99-6 | Vegetarian | 14 | 1 | 0.85 | 0 | 12 |
| Omnivore | 24 | 0 | 0.00 | 0 | Not detected | |
| Dimethyl trisulphide CAS 3658-80-8 | Vegetarian | 14 | 14 | 85 | 69 | 11–199 |
| Omnivore | 24 | 24 | 160 | 130 | 10–410 | |
| 1-Octen-3-ol CAS 3391-86-4 | Vegetarian | 14 | 6 | 8 | 14 | 2–38 |
| Omnivore | 24 | 7 | 3 | 12 | 0.4–58 | |
| Ethanoic acid CAS 64-19-7 | Vegetarian | 14 | 14 | 8799 | 2155 | 4298–12,233 |
| Omnivore | 24 | 24 | 8731 | 2343 | 672–12,963 | |
| Butanoic acid CAS 107-92-6 | Vegetarian | 14 | 14 | 7452 | 2938 | 2493–11,553 |
| Omnivore | 24 | 24 | 7618 | 1922 | 2681–9633 | |
| 3-Methylbutanoic acid CAS 503-74-2 | Vegetarian | 14 | 14 | 2319 | 1974 | 64–5821 |
| Omnivore | 24 | 22 | 2965 | 2344 | 220–8262 | |
| Pentanoic acid CAS 109-52-4 | Vegetarian | 14 | 14 | 3123 | 2841 | 88–10,216 |
| Omnivore | 24 | 23 | 5213 | 4953 | 730–21,886 | |
| Indole CAS 120-72-9 | Vegetarian | 14 | 14 | 3340 | 1505 | 290–5477 |
| Omnivore | 24 | 24 | 3938 | 772 | 2342–5070 | |
| 3-Methyl-1H-indole CAS 83-34-1 | Vegetarian | 14 | 14 | 1160 | 633 | 37–2267 |
| Omnivore | 24 | 24 | 1419 | 846 | 404–3483 |
| Chemical Family | Unmodified | pH Modified | Percentage Decrease (Unmodified to pH13 Modified) |
|---|---|---|---|
| Aldehydes | 6 | 3 | 50% |
| Esters and thioesters | 36 | 30 | 17% |
| Ketones | 12 | 7 | 42% |
| Alcohols | 20 | 11 | 45% |
| Acids | 10 | 3 | 70% |
| Nitrogen containing | 4 | 6 | 50% |
| Sulphides | 8 | 3 | 63% |
| Aromatic compounds | 6 | 5 | 29% |
| Miscellaneous | 2 | 0 | 100% |
| Terpenes | 30 | 22 | 40% |
| Unidentified | 32 | 16 | 50% |
| CAS | Compound | Retention Time (min) | Retention Indices Sample | Retention Indices Literature |
|---|---|---|---|---|
| 563-80-4 | 3-Methyl-2-butanone | 4.56 | 972 | 970 [36] |
| 556-24-1 | 3-Methylbutanoic acid methyl ester | 5.49 | 1017 | 1024 [37] |
| 97-87-0 | Propanoic acid 2-methylbutyl ester | 8.18 | 1152 | 1154 [37] |
| 539-90-2 | Butanoic acid 2-methylpropyl ester | 8.45 | 1158 | 11,152 [37] |
| 141-06-0 | Pentanoic acid propyl ester | 9.73 | 1215 | 1200–1233 [38] |
| 1618-26-4 | 2,4-Dithiapentane | 10.6 | 1260 | 1260 [39] |
| 2313-61-3 | 2-heptanol | 11.66 | 1308 | 1315.3 [40] |
| 15356-70-4 | 5methyl-2-(1-methylethyl)-(1a,2b,5a) (Menthol) | 16.9 | 1631 | 1630.4 [40] |
| 55012-32-3 | 4-Isopropyl benzaldehyde (cuminaldehyde) | 18.91 | 1783 | 1781.4 [40] |
| CAS | Compound | Retention Time (min) | Retention Indices Sample | Retention Indices Literature |
|---|---|---|---|---|
| 75-50-3 | Trimethylamine | 1.95 | - | - |
| 108-64-5 | 3-Methyl butanoic acid ethyl ester | 6.48 | 1074 | 1068 [38] |
| 926-64-7 | Acetonitrile (dimethylamino) | 10.02 | 1230 | 1243 [41] |
| Number of Participants | Ethnic Origin | Omnivore | Vegetarian |
|---|---|---|---|
| 16 | UK | 8 | 8 |
| 11 | South America (Brazil, Mexico) | 10 | 1 |
| 3 | Mainland Europe (Czech Republic, Latvia, and Spain) | 1 | 2 |
| 5 | Asia (China, Vietnam, Iran) | 4 | 1 |
| 3 | Africa (Maldives, Nigeria) | 1 | 2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gould, O.; De Lacy Costello, B.; Smart, A.; Jones, P.; Macmaster, A.; Ransley, K.; Ratcliffe, N. Gas Chromatography Mass Spectrometry (GC-MS) Quantification of Metabolites in Stool Using 13C Labelled Compounds. Metabolites 2018, 8, 75. https://doi.org/10.3390/metabo8040075
Gould O, De Lacy Costello B, Smart A, Jones P, Macmaster A, Ransley K, Ratcliffe N. Gas Chromatography Mass Spectrometry (GC-MS) Quantification of Metabolites in Stool Using 13C Labelled Compounds. Metabolites. 2018; 8(4):75. https://doi.org/10.3390/metabo8040075
Chicago/Turabian StyleGould, Oliver, Ben De Lacy Costello, Amy Smart, Peter Jones, Angus Macmaster, Karen Ransley, and Norman Ratcliffe. 2018. "Gas Chromatography Mass Spectrometry (GC-MS) Quantification of Metabolites in Stool Using 13C Labelled Compounds" Metabolites 8, no. 4: 75. https://doi.org/10.3390/metabo8040075
APA StyleGould, O., De Lacy Costello, B., Smart, A., Jones, P., Macmaster, A., Ransley, K., & Ratcliffe, N. (2018). Gas Chromatography Mass Spectrometry (GC-MS) Quantification of Metabolites in Stool Using 13C Labelled Compounds. Metabolites, 8(4), 75. https://doi.org/10.3390/metabo8040075





