Chemical Analysis of Whale Breath Volatiles: A Case Study for Non-Invasive Field Health Diagnostics of Marine Mammals
Abstract
:1. Introduction

2. Methods and Materials
2.1. Fabrication of WBE Sample Collection System
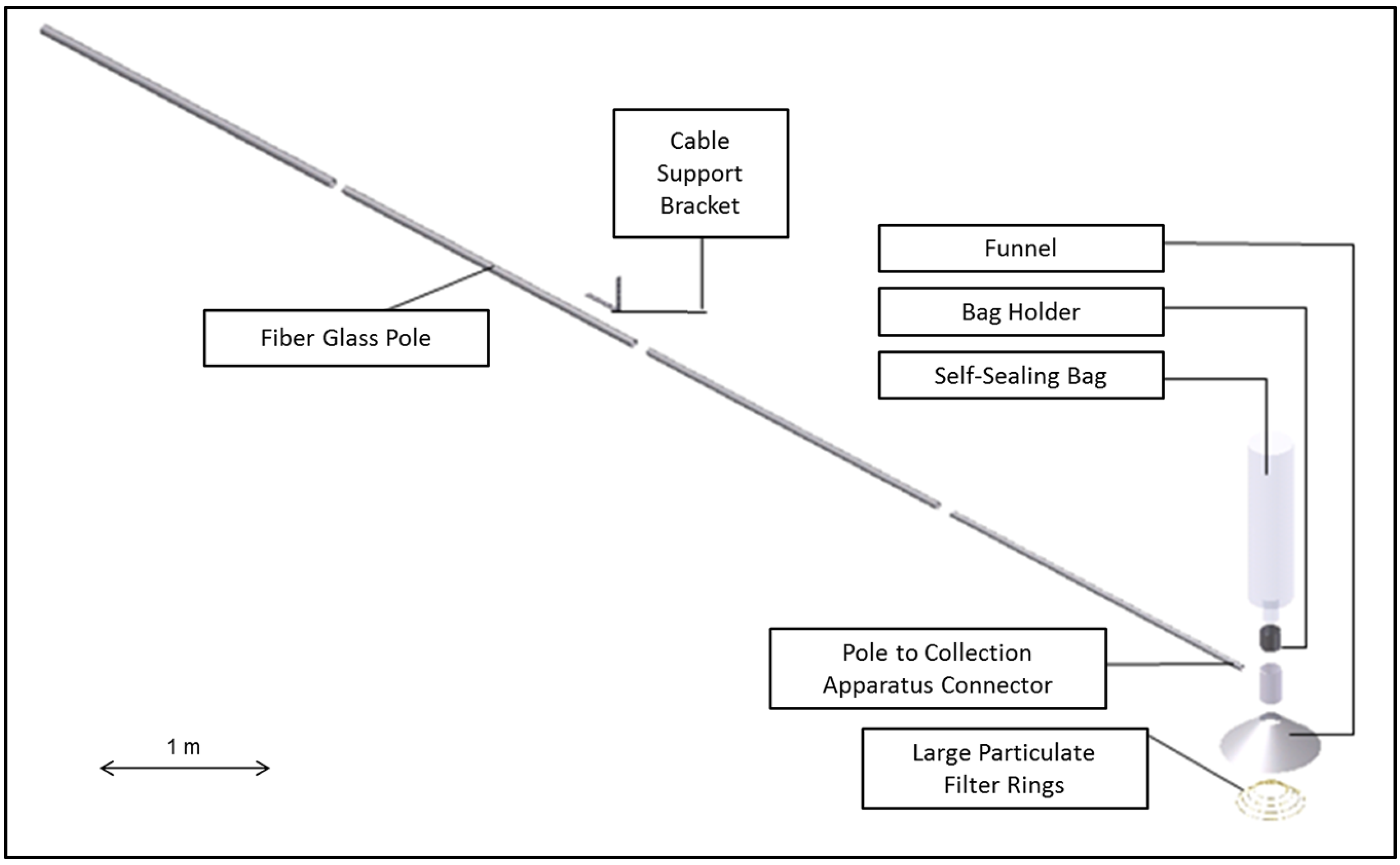
2.2. Field Sampling Methodology

2.3. Stabilization of Breath Gasses for Ground Transport
2.4. Chemical Analysis of the WBE
2.5. Data Analysis and Interpretation
3. Results and Discussion
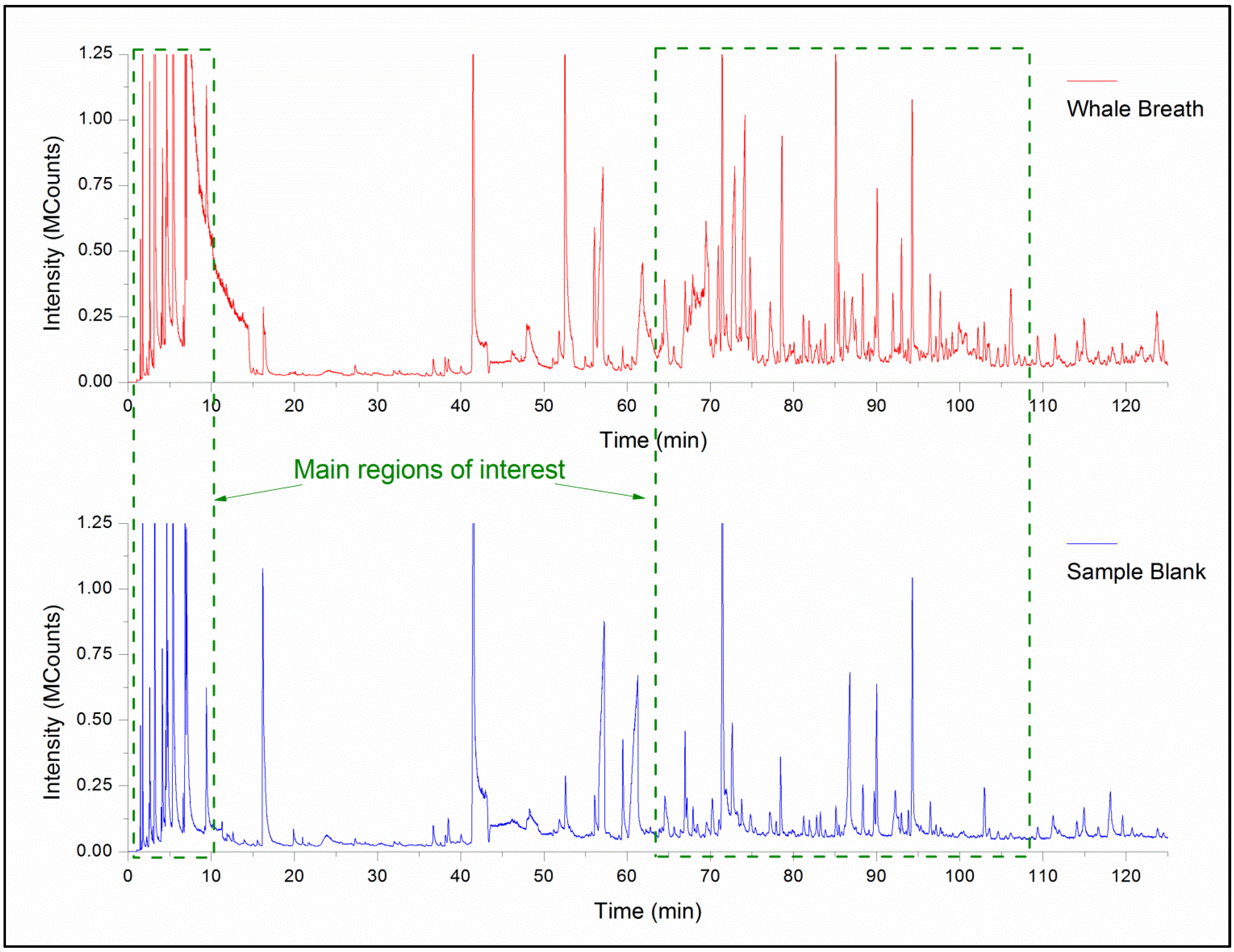
| RT (min) | Chemical Name | CAS | Match | R. Match | RA (%) | Formula | MW | NIST | Human VOC Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 1.477+ | Carbon dioxide | 124-38-9 | 682 | 696 | 2.32 | CO2 | 44 | 18873 | F, Br, Bl |
| 1.549 | Carbonyl sulfide | 463-58-1 | 943 | 952 | 0.06 | COS | 60 | 19006 | Br, M |
| 2.592 | Guanidine | 113-00-8 | 811 | 831 | 4.98 | CH5N3 | 59 | 221220 | |
| 2.668 | Furan | 110-00-9 | 758 | 767 | 0.59 | C4H4O | 68 | 228308 | U, Br, M |
| 2.960+ | Cyclopropane, 1,1-dimethyl- | 1630-94-0 | 899 | 899 | 0.21 | C5H10 | 70 | 34618 | |
| 3.191+ | Carbon disulfide | 75-15-0 | 783 | 785 | 100.00 | CS2 | 76 | 118705 | F, U, Br, Sk, M |
| 3.360 | 2-Propanol, 2-methyl- | 75-65-0 | 796 | 809 | 0.34 | C4H10O | 74 | 19162 | Br, M, Bl |
| 3.999+ | Butane, 2,3-dimethyl- | 79-29-8 | 849 | 855 | 0.44 | C6H14 | 86 | 291518 | Br |
| 4.129+ | Octane | 111-65-9 | 847 | 850 | 0.95 | C8H18 | 114 | 229407 | F, Br, Sk, Sa |
| 4.507 | 2,3-Dihydrofuran | 1191-99-7 | 783 | 849 | 4.37 | C4H6O | 70 | 114595 | F, Br |
| 4.635 | Pentane, 2,3-dimethyl- | 565-59-3 | 756 | 757 | 4.15 | C7H16 | 100 | 61339 | Br |
| 6.643+ | Pentane, 2,2-dimethyl- | 590-35-2 | 852 | 876 | 12.01 | C7H16 | 100 | 114486 | Br |
| 6.867+ | 2,4,6-Trimethyl-3-heptene | 126690-66-2 | 721 | 765 | 1.51 | C10H20 | 140 | 113569 | |
| 9.417 | non-ID | 15.60 | |||||||
| 15.017+ | Methane, dibromo- | 74-95-3 | 928 | 928 | 3.64 | CH2Br2 | 172 | 118720 | Bl |
| 27.312+ | Toluene | 108-88-3 | 917 | 917 | 0.00 | C7H8 | 92 | 227551 | F, U, Br, Sk, M, Bl, Sa |
| 31.912 | 2-Butene, 2-methyl- | 513-35-9 | 808 | 808 | 0.47 | C5H10 | 70 | 242 | Br |
| 36.689 | non-ID | 0.15 | |||||||
| 47.926+ | o-Xylene | 95-47-6 | 879 | 879 | 0.49 | C8H10 | 106 | 228061 | F, Br, M, Bl, Sa |
| 51.064 | non-ID | 0.84 | |||||||
| 51.844+ | Ethylbenzene | 100-41-4 | 670 | 798 | 0.14 | C8H10 | 106 | 114918 | F, U, Br, Sk, M, Bl |
| 54.970 | Decane | 124-18-5 | 816 | 816 | 1.04 | C10H22 | 142 | 227682 | F, Br, Sk, Sa |
| 56.087+ | Ethanol, 2-butoxy- | 111-76-2 | 834 | 834 | 0.39 | C6H14O2 | 118 | 11812 | U, Br |
| 57.765 | non-ID | 12.19 | |||||||
| 64.212 | Benzene, (1-methylethyl)- | 98-82-8 | 920 | 920 | 0.05 | C9H12 | 120 | 114201 | F, Br |
| 64.494+ | Ethanone, 2,2-dihydroxy-1-phenyl- | 1075-06-5 | 874 | 894 | 0.18 | C8H8O3 | 152 | 289212 | F |
| 65.635 | Benzene, 1,2,3-trimethyl- | 526-73-8 | 719 | 843 | 7.33 | C9H12 | 120 | 125395 | F, Br, M |
| 70.597+ | Indane | 496-11-7 | 618 | 780 | 0.63 | C9H10 | 118 | 20399 | F, Br |
| 70.931 | 3-Aminopyridine | 462-08-8 | 671 | 692 | 0.06 | C5H6N2 | 94 | 288371 | Sk |
| 72.982 | non-ID | 6.15 | |||||||
| 73.547 | Benzene, 1-ethyl-4-methyl- | 622-96-8 | 772 | 827 | 1.19 | C9H12 | 120 | 114034 | F, Br, M |
| 74.159+ | Butane, 2-methoxy-2-methyl- | 994-05-8 | 690 | 700 | 0.87 | C6H14O | 102 | 61443 | |
| 74.776+ | Benzene, 1-methyl-4-(1-methylethyl)- | 99-87-6 | 911 | 920 | 9.71 | C10H14 | 134 | 113988 | U, Br, Sk, Sa |
| 75.411+ | Limonene | 138-86-3 | 764 | 805 | 6.13 | C10H16 | 136 | 57640 | F, U, Br, M, Sa |
| 77.196 | 1-Hexene, 3,5-dimethyl- | 7423-69-0 | 630 | 739 | 2.04 | C8H16 | 112 | 113470 | |
| 77.392+ | Benzyl Alcohol | 100-51-6 | 827 | 891 | 3.18 | C7H8O | 108 | 291348 | F, U, Br, Sk, Sa |
| 78.631 | 1-Butanol, 4-(1-methylethoxy)- | 31600-69-8 | 768 | 800 | 0.20 | C7H16O2 | 132 | 46521 | |
| 78.888 | Benzene, 1-methyl-3-propyl- | 1074-43-7 | 573 | 781 | 5.70 | C10H14 | 134 | 57774 | |
| 79.357+ | 2(3H)-Furanone, dihydro-4-methyl- | 1679-49-8 | 772 | 833 | 0.60 | C5H8O2 | 100 | 153216 | |
| 80.062 | non-ID | 0.62 | |||||||
| 81.189+ | Benzoyl bromide | 618-32-6 | 916 | 916 | 0.43 | C7H5BrO | 184 | 228984 | |
| 81.875 | 2-Butene-1,4-diol, diformate | 29619-56-5 | 809 | 818 | 3.27 | C6H8O4 | 144 | 4155 | |
| 82.071 | Hexanoic acid, 2-tetrahydrofurylmethyl ester | 2217-34-7 | 684 | 772 | 3.03 | C11H20O3 | 200 | 279279 | |
| 82.594+ | Benzene, 2-ethyl-1,3-dimethyl- | 2870-04-4 | 825 | 830 | 0.73 | C10H14 | 134 | 3144 | |
| 84.652 | non-ID | 0.30 | |||||||
| 85.086+ | Benzene, (2-methyl-1-propenyl)- | 768-49-0 | 891 | 891 | 0.19 | C10H12 | 132 | 113562 | Br |
| 85.722 | 2-Propen-1-one, 1-phenyl- | 768-03-6 | 518 | 618 | 17.49 | C9H8O | 132 | 161642 | |
| 86.131 | 1-Propanol, 3-chloro-, acetate | 628-09-1 | 633 | 699 | 0.27 | C5H9ClO2 | 136 | 3424 | |
| 87.499 | Oxalic acid, isobutyl nonyl ester | - | 723 | 826 | 2.50 | C15H28O4 | 272 | 309374 | |
| 88.330 | 1-Hepten-4-ol | 3521-91-3 | 648 | 704 | 1.43 | C7H14O | 114 | 1768 | |
| 88.601 | non-ID | 4.26 | |||||||
| 88.876 | non-ID | 0.09 | |||||||
| 89.313 | 2-Ethylpiperidine | 1484-80-6 | 770 | 783 | 0.38 | C7H15N | 113 | 231734 | Sk |
| 91.945 | 4-Heptanol | 589-55-9 | 687 | 785 | 1.37 | C7H16O | 116 | 1897 | |
| 93.000 | 1-Butanol, 4-butoxy- | 4161-24-4 | 804 | 810 | 2.19 | C8H18O2 | 146 | 21344 | |
| 96.432 | 2-Allyl-2-methyl-1,3-cyclopentanedione | 26828-48-8 | 632 | 689 | 6.79 | C9H12O2 | 152 | 108207 | |
| 97.644+ | Naphthalene | 91-20-3 | 915 | 919 | 3.98 | C10H8 | 128 | 228342 | F, Br, Sk, M, Sa |
| 97.914 | non-ID | 4.20 | |||||||
| 98.380 | Cyclopentane, 1-isobutylidene-3-methyl- | - | 664 | 717 | 0.20 | C10H18 | 138 | 150621 | |
| 99.099 | o-Toluic acid, 4-nitrophenyl ester | - | 751 | 895 | 0.50 | C14H11NO4 | 257 | 307460 | |
| 99.913 | non-ID | 1.28 | |||||||
| 100.077 | Benzoic acid, 3-amino-, methyl ester | 4518-10-9 | 644 | 654 | 0.48 | C8H9NO2 | 151 | 73503 | |
| 102.217 | 2-Butene, 2-nitro- | 4812-23-1 | 596 | 690 | 0.06 | C4H7NO2 | 101 | 75242 | |
| 103.393 | Oxalic acid, isobutyl octyl ester | - | 788 | 788 | 2.98 | C14H26O4 | 258 | 309373 | |
| 103.528 | 2-Propenoic acid, 2-hydroxyethyl ester | 818-61-1 | 483 | 733 | 0.59 | C5H8O3 | 116 | 71287 | |
| 104.608+ | 1,9-Nonanediol | 3937-56-2 | 769 | 769 | 0.99 | C9H20O2 | 160 | 114694 | |
| 105.468+ | Benzothiazole | 95-16-9 | 827 | 889 | 0.09 | C7H5NS | 135 | 228674 | Br, Sk |
| 106.168 | Ethanol, 2-phenoxy- | 122-99-6 | 820 | 861 | 1.68 | C8H10O2 | 138 | 250161 | F, Br, Sk, Sa |
| 107.094 | 2-Naphthalenol | 135-19-3 | 784 | 867 | 3.69 | C10H8O | 144 | 92317 | |
| 107.739 | non-ID | 0.78 |

4. Conclusions
- (1)
- Use a thermal desorption (TD) tube to directly introduce the breath sample into the mass spectrometer instrument with minimal sample loss. The TD tube is considered a gold standard in active VOC sampling practice, especially in outdoor environments. However, this is an expensive option as significant equipment investment in consumables and infrastructure is required. In addition to the thermal desorption unit, reusable silicone coated stainless steel tubes are also available, which include Tenax®-TA packing. These devices must also be conditioned. TD requires large quantity of ultra-high purity purging gas for conditioning and cleaning cycles of sorbent packing between uses. Lastly, the sorbent needs to be replaced periodically to ensure maximum absorption capacity as it is only stable for a limited period of time after cleaning, even when properly stored in optimal conditions.
- (2)
- Use a mobile gas detection system to detect specific compounds directly from the whale blow in the field. For example, differential mobility spectrometry (DMS) is a portable sensor platform currently applied in our group to directly analyze human exhaled breath, and it represents an alternative to traditional GC/MS lab based instrument testing. However the spectral resolution of DMS is lower compared to MS, but it should still able to generate information rich spectral profiles in near real time (5–15 min) for the different chemical species present in the WBE.
- (3)
- Once further testing is performed on exhaled cetacean breath, it should be possible to develop a mass spectral library and database for biogenic breath biomarkers from these marine mammals. This is not limited to MS alone, and other VOC sampling and analysis techniques such as DMS should be considered. It would also be useful to perform tandem MS/MS for chemical identification of the profiled breath metabolites. All of these approaches will help us to characterize unidentified biomarkers that may be associated with heath status in these important animals.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Amann, A.; Poupart, G.; Telser, S.; Ledochowski, M.; Schmid, A.; Mechtcheriakov, S. Applications of breath gas analysis in medicine. Int. J. Mass Spectrom. 2004, 239, 227–233. [Google Scholar] [CrossRef]
- Amann, A.; Schmid, A.; Scholl-Buergi, S.; Telser, S.; Hinterhuber, H. Breath analysis for medical diagnosis and therapeutic monitoring. Spectrosc. Eur. 2005, 17, 18–20. [Google Scholar]
- Hornuss, C.; Praun, S.; Villinger, J.; Dornauer, A.; Moehnle, P.; Dolch, M.; Weninger, E.; Chouker, A.; Feil, C.; Briegel, J.; et al. Real-time monitoring of propofol in expired air in humans undergoing total intravenous anesthesia. Anesthesiology 2007, 106, 665–674. [Google Scholar] [CrossRef]
- Phillips, M.; Cataneo, R.N.; Cummin, A.R.C.; Gagliardi, A.J.; Gleeson, K.; Greenberg, J.; Maxfield, R.A.; Rom, W.N. Detection of lung cancer with volatile markers in the breath. Chest 2003, 123, 2115–2123. [Google Scholar] [CrossRef]
- Phillips, M.; Gleeson, K.; Hughes, J.M.B.; Greenberg, J.; Cataneo, R.N.; Baker, L.; McVay, W.P. Volatile organic compounds in breath as markers of lung cancer: A cross-sectional study. Lancet 1999, 353, 1930–1933. [Google Scholar] [CrossRef]
- Phillips, M.E.A. Volatile markers of breast cancer in the breath. Breast J. 2003, 9, 184–191. [Google Scholar] [CrossRef]
- Risby, T.H.; Solga, S.F. Current status of clinical breath analysis. Appl. Phys. B: Lasers Opt. 2006, 85, 421–426. [Google Scholar]
- Takita, A.; Masui, K.; Kazama, T. On-line monitoring of end-tidal propofol concentration in anesthetized patients. Anesthesiology 2007, 106, 659–664. [Google Scholar] [CrossRef]
- Wehinger, A.; Schmid, A.; Mechtcheriakov, S.; Ledochowski, M.; Grabmer, C.; Gastl, G.A.; Amann, A. Lung cancer detection by proton transfer reaction mass-spectrometric analysis of human breath gas. Int. J. Mass Spectrom. 2007, 265, 49–59. [Google Scholar] [CrossRef]
- Zolotov, Y.A. Breath Analysis. J. Anal. Chem. 2005, 60, Article 497. [Google Scholar]
- Vautz, W.; Nolte, J.; Bufe, A.; Baumbach, J.I.; Peters, M. Analyses of mouse breath with ion mobility spectrometry: A feasibility study. J. Appl. Physiol. 2010, 108, 697–704. [Google Scholar] [CrossRef]
- Miekisch, W; Schubert, J.K.; Noeldge-Schomburg, G.F.E. Diagnostic potential of breath analysis—focus on volatile organic compounds. Clin. Chim. Acta 2004, 347, 25–39. [Google Scholar]
- Kharitonov, S.A.; Barnes, P.J. Biomarkers of some pulmonary diseases in exhaled breath. Biomarkers 2002, 7, 1–32. [Google Scholar] [CrossRef]
- Grob, N.M.; Aytekin, M.; Dweik, R.A. Biomarkers in exhaled breath condensate: A review of collection, processing and analysis. J. Breath Res. 2008, 2, Article 037004. [Google Scholar]
- Costello, B.D.L.; Amann, A.; Al-Kateb, H.; Flynn, C.; Filipiak, W.; Khalid, T.; Osborne, D.; Ratcliffe, N.M. A review of the volatiles from the healthy human body. J. Breath Res. 2014, 8, Article 014001. [Google Scholar]
- Phillips, M.; Basa-Dalay, V.; Bothamley, G.; Cataneo, R.N.; Lam, P.K.; Natividad, M.P.R.; Schmitt, P.; Wai, J. Breath biomarkers of active pulmonary tuberculosis. Tuberculosis 2010, 90, 145–151. [Google Scholar] [CrossRef]
- Michael, P; Renee, N.C.; Anirudh, C.; Patrick, J.D.; Anantrai, D.; David, A.L.; Kim, L.N.; Peter, S.; James, W. Effect of influenza vaccination on oxidative stress products in breath. J. Breath Res. 2010, 4, Article 026001. [Google Scholar]
- Phillips, M.; Basa-Dalay, V.; Blais, J.; Bothamley, G.; Chaturvedi, A.; Modi, K.D.; Pandya, M.; Natividad, M.P.R.; Patel, U.; Ramraje, N.N.; et al. Point-of-care breath test for biomarkers of active pulmonary tuberculosis. Tuberculosis 2012, 92, 314–320. [Google Scholar] [CrossRef]
- Chambers, S.T.; Scott-Thomas, A.; Epton, M. Developments in novel breath tests for bacterial and fungal pulmonary infection. Curr. Opin. Pulm. Med. 2012, 18, 228–232. [Google Scholar] [CrossRef]
- Machado, R.F.; Laskowski, D.; Deffenderfer, O.; Burch, T.; Zheng, S.; Mazzone, P.J.; Mekhail, T.; Jennings, C.; Stoller, J.K.; Pyle, J.; et al. Detection of lung cancer by sensor array analyses of exhaled breath. Am. J. Resp. Crit. Care 2005, 171, 1286–1291. [Google Scholar] [CrossRef]
- Mazzone, P.J. Analysis of Volatile Organic Compounds in the Exhaled Breath for the Diagnosis of Lung Cancer. J. Thorac. Oncol. 2008, 3, 774–780. [Google Scholar] [CrossRef]
- Horváth, I.; Lázár, Z.; Gyulai, N.; Kollai, M.; Losonczy, G. Exhaled biomarkers in lung cancer. Eur. Respir. J. 2009, 34, 261–275. [Google Scholar]
- Fuchs, P; Loeseken, C.; Schubert, J.K.; Miekisch, W. Breath gas aldehydes as biomarkers of lung cancer. Int. J. Cancer 2010, 126, 2663–2670. [Google Scholar]
- Amann, A.; Corradi, M.; Mazzone, P.; Mutti, A. Lung cancer biomarkers in exhaled breath. Expert Rev. Mol. Diagn. 2011, 11, 207–217. [Google Scholar] [CrossRef]
- Bajtarevic, A.; Ager, C.; Pienz, M.; Klieber, M.; Schwarz, K.; Ligor, M.; Ligor, T.; Filipiak, W.; Denz, H.; Fiegl, M.; et al. Noninvasive detection of lung cancer by analysis of exhaled breath. BMC Cancer 2009, 9, Article 348. [Google Scholar]
- Haick, H.; Broza, Y.Y.; Mochalski, P.; Ruzsanyi, V.; Amann, A. Assessment, origin, and implementation of breath volatile cancer markers. Chem. Soc. Rev. 2014, 43, 1423–1449. [Google Scholar] [CrossRef]
- Beck, O.; Stephanson, N.; Sandqvist, S.; Franck, J. Detection of Drugs of Abuse in Exhaled Breath from Users Following Recovery from Intoxication. J. Anal. Toxicol. 2012, 36, 638–646. [Google Scholar] [CrossRef]
- Maniscalco, M.; de Laurentiis, G.; Pentella, C.; Mormile, M.; Sanduzzi, A.; Carratù, P.; Sofia, M. Exhaled breath condensate as matrix for toluene detection: A preliminary study. Biomarkers 2006, 11, 233–240. [Google Scholar] [CrossRef]
- Pleil, J.D. Role of Exhaled Breath Biomarkers in Environmental Health Science. J. Toxicol. Env. Heal. B 2008, 11, 613–629. [Google Scholar] [CrossRef]
- Turner, M.A.; Bandelow, S.; Edwards, L.; Patel, P.; Martin, H.J.; Wilson, I.D.; Thomas, C.L.P. The effect of a paced auditory serial addition test (PASAT) intervention on the profile of volatile organic compounds in human breath: a pilot study. J. Breath Res. 2013, 7, Article 017102. [Google Scholar]
- Hunt, K.E.; Moore, M.J.; Rolland, R.M.; Kellar, N.M.; Hall, A.J.; Kershaw, J.; Raverty, S.A.; Davis, C.E.; Yeates, L.C.; Fauquier, D.A.; et al. Overcoming the challenges of studying conservation physiology in large whales: A review of available methods. Conserv. Physiol. 2013, 1, Article cot006. [Google Scholar]
- Dweik, R.A. The great challenge for exhaled breath analysis: Embracing complexity, delivering simplicity. J. Breath Res. 2011, 5, Article 030201. [Google Scholar]
- Dummer, J.; Storer, M.; Swanney, M.; McEwan, M.; Scott-Thomas, A.; Bhandari, S.; Chambers, S.; Dweik, R.; Epton, M. Analysis of biogenic volatile organic compounds in human health and disease. Trac-Trend. Anal. Chem. 2011, 30, 960–967. [Google Scholar]
- Schubert, J.K.; Miekisch, W.; Geiger, K.; Nöldge-Schomburg, G.F. Breath analysis in critically ill patients: Potential and limitations. Expert Rev. Mol. Diagn. 2004, 4, 619–629. [Google Scholar] [CrossRef]
- Bojko, B.; Cudjoe, E.; Pawliszyn, J.; Wasowicz, M. Solid-phase microextraction. How far are we from clinical practice? Trac-Trend. Anal. Chem. 2011, 30, 1505–1512. [Google Scholar]
- Amann, A.; Miekisch, W.; Pleil, J.; Risby, T.; Schubert, J. Methodological issues of sample collection and analysis of exhaled breath. Eur. Respir. Mon. 2010, 49, 96–114. [Google Scholar]
- Groves, W.A.; Zellers, E.T. Investigation of Organic Vapor Losses to Condensed Water Vapor in Tedlar® Bags Used for Exhaled-Breath Sampling. Am. Ind. Hyg. Assoc. J. 1996, 57, 257–263. [Google Scholar] [CrossRef]
- Marco, M.L.S.; Simona, M.C.; Frans, J.M.H. The suitability of Tedlar bags for breath sampling in medical diagnostic research. Physiol. Meas. 2007, 28, 73–84. [Google Scholar] [CrossRef]
- Jonathan, B.; Jens, H.; Rene, G.; Armin, H. On the use of Tedlar® bags for breath-gas sampling and analysis. J. Breath Res. 2008, 2, Article 046001. [Google Scholar]
- Soyer, O.U.; Dizdar, E.A.; Keskin, O.; Lilly, C.; Kalayci, O. Comparison of two methods for exhaled breath condensate collection. Allergy 2006, 61, 1016–1018. [Google Scholar] [CrossRef]
- Davidsson, A.; Schmekel, B. Efficacy of two breath condensers. J. Clin. Lab. Anal. 2010, 24, 219–223. [Google Scholar] [CrossRef]
- Lindstrom, A.B.; Pleil, J.D. A review of the USEPA’s single breath canister (SBC) method for exhaled volatile organic biomarkers. Biomarkers 2002, 7, 189–208. [Google Scholar] [CrossRef]
- Ligor, T.; Ligor, M.; Amann, A.; Ager, C.; Bachler, M.; Dzien, A.; Buszewski, B. The analysis of healthy volunteers’ exhaled breath by the use of solid-phase microextraction and GC-MS. J. Breath Res. 2008, 2, Article 046006. [Google Scholar]
- Phillips, M. Method for the Collection and Assay of Volatile Organic Compounds in Breath. Anal. Biochem. 1997, 247, 272–278. [Google Scholar] [CrossRef]
- Frère, C.H.; Krzyszczyk, E.; Patterson, E.M.; Hunter, S.; Ginsburg, A.; Mann, J. Thar She Blows! A Novel Method for DNA Collection from Cetacean Blow. PLoS One 2010, 5, Article e12299. [Google Scholar]
- Hogg, C.J.; Rogers, T.L.; Shorter, A.; Barton, K.; Miller, P.J.O.; Nowacek, D. Determination of steroid hormones in whale blow: It is possible. Mar. Mammal Sci. 2009, 25, 605–618. [Google Scholar] [CrossRef]
- Schroeder, P.; Raverty, S.; Cameron, C.; Zabek, E.; Eshghi, A.; Bain, D.; Wood, B.; Rhodes, L.; Hanson, B. Investigation into the microbial culture and molecular screening of exhaled breaths of endangered southern resident killer whales (SRKW) and pathogen screening of the seasurface microlayer (SML) in Puget Sound. Available online: http://depts.washington.edu/uwconf/psgb/proceedings/table_of_contents.html (accessed on 3 September 2014).
- Acevedo-Whitehouse, K.; Rocha-Gosselin, A.; Gendron, D. A novel non-invasive tool for disease surveillance of free-ranging whales and its relevance to conservation programs. Anim. Conserv. 2010, 13, 217–225. [Google Scholar]
- Times-Standard Local News. Klamath whale likely died from a fungal skin infection; dead gray whale in Bandon, Ore,. not calf who left river. Available online: http://www.times-standard.com/localnews/ci_19637852 (accessed on 5 May 2014).
- Tylor, A. Mama, the Klamath River Whale, Swimming in River 48 Days. Available online: http://ashalatylorimages.wordpress.com/2011/08/11/mama-the-klamath-river-whale-swimming-in-river-48-days-81011/ (accessed on 3 September 2014).
- Tylor, A. How to Capture Whale Breath from Mama, The Klamath River Whale. Available online: http://ashalatylorimages.wordpress.com/2011/08/07/how-to-capture-whale-breath-from-mama-the-klamath-river-whale-update/ (accessed on 3 September 2014).
- Saturn Varian Chemstation, version 6.6; MS Data Review; Saturn Varian Inc.: Palo Alto, CA, USA, 2005.
- Rowles, T.; Ilyashenko, V. Summary of Finding on Investigation of the Stinky Whale Condition in Eastern North Pacific Gray Whales. In Proceedings of International Whale Conference (IWC) 59th annual meeting, Anchorage, AK, USA, 28–31 May 2007. Paper IWC/59/CC15.
- Ilyashenko, V. Consideration of Management Implications “Stinky” Gray Whales for the Eastern North Pacific stock. In Proceedings of International Whale Conference (IWC) 59th annual meeting, Anchorage, AK, USA, 28–31 May 2007. Paper IWC/59/ASW7.
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cumeras, R.; Cheung, W.H.K.; Gulland, F.; Goley, D.; Davis, C.E. Chemical Analysis of Whale Breath Volatiles: A Case Study for Non-Invasive Field Health Diagnostics of Marine Mammals. Metabolites 2014, 4, 790-806. https://doi.org/10.3390/metabo4030790
Cumeras R, Cheung WHK, Gulland F, Goley D, Davis CE. Chemical Analysis of Whale Breath Volatiles: A Case Study for Non-Invasive Field Health Diagnostics of Marine Mammals. Metabolites. 2014; 4(3):790-806. https://doi.org/10.3390/metabo4030790
Chicago/Turabian StyleCumeras, Raquel, William H.K. Cheung, Frances Gulland, Dawn Goley, and Cristina E. Davis. 2014. "Chemical Analysis of Whale Breath Volatiles: A Case Study for Non-Invasive Field Health Diagnostics of Marine Mammals" Metabolites 4, no. 3: 790-806. https://doi.org/10.3390/metabo4030790
APA StyleCumeras, R., Cheung, W. H. K., Gulland, F., Goley, D., & Davis, C. E. (2014). Chemical Analysis of Whale Breath Volatiles: A Case Study for Non-Invasive Field Health Diagnostics of Marine Mammals. Metabolites, 4(3), 790-806. https://doi.org/10.3390/metabo4030790




