Soil Application of Bacillus subtilis Regulates Flavonoid and Alkaloids Biosynthesis in Mulberry Leaves
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Sample Preparation
2.2. Metabolomic Analysis
2.3. Transcriptomic Analysis
2.4. Integrative Analysis of Metabolomics and Transcriptome
2.5. RT-PCR Analysis
3. Results
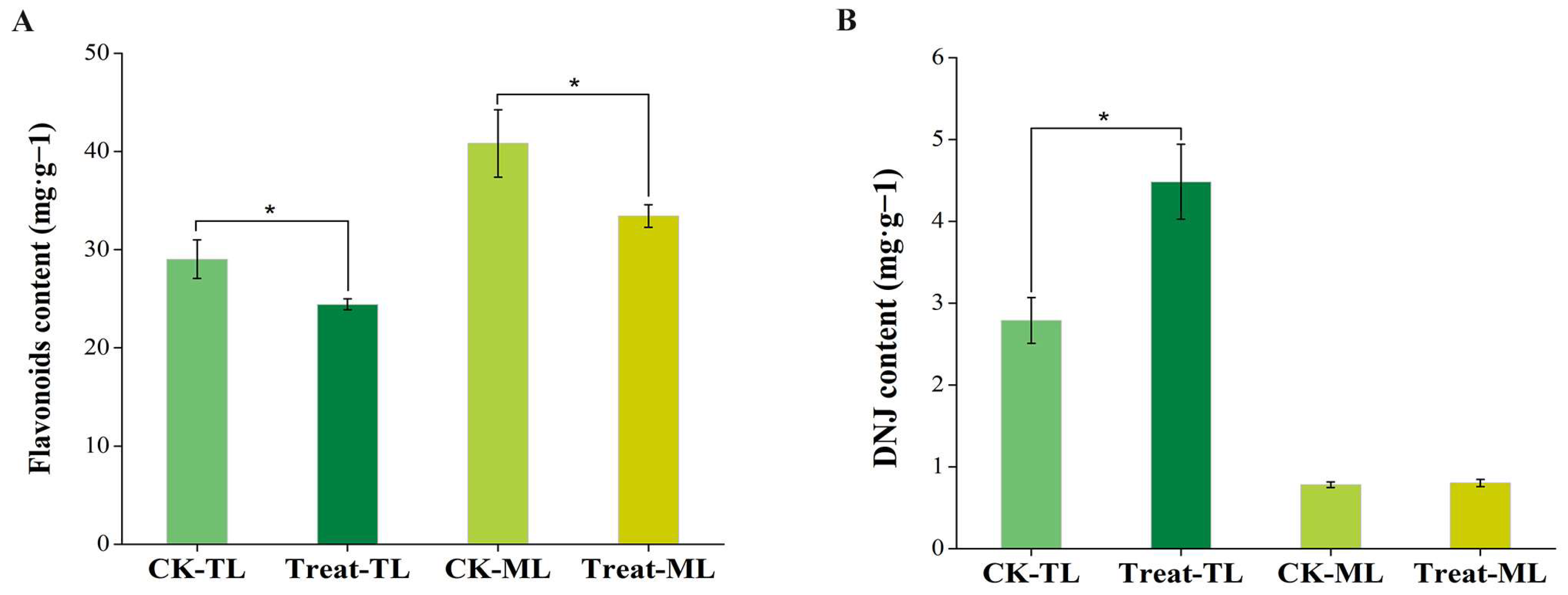
3.1. The Changes in Flavonoid and DNJ Contents in Mulberry Leaves
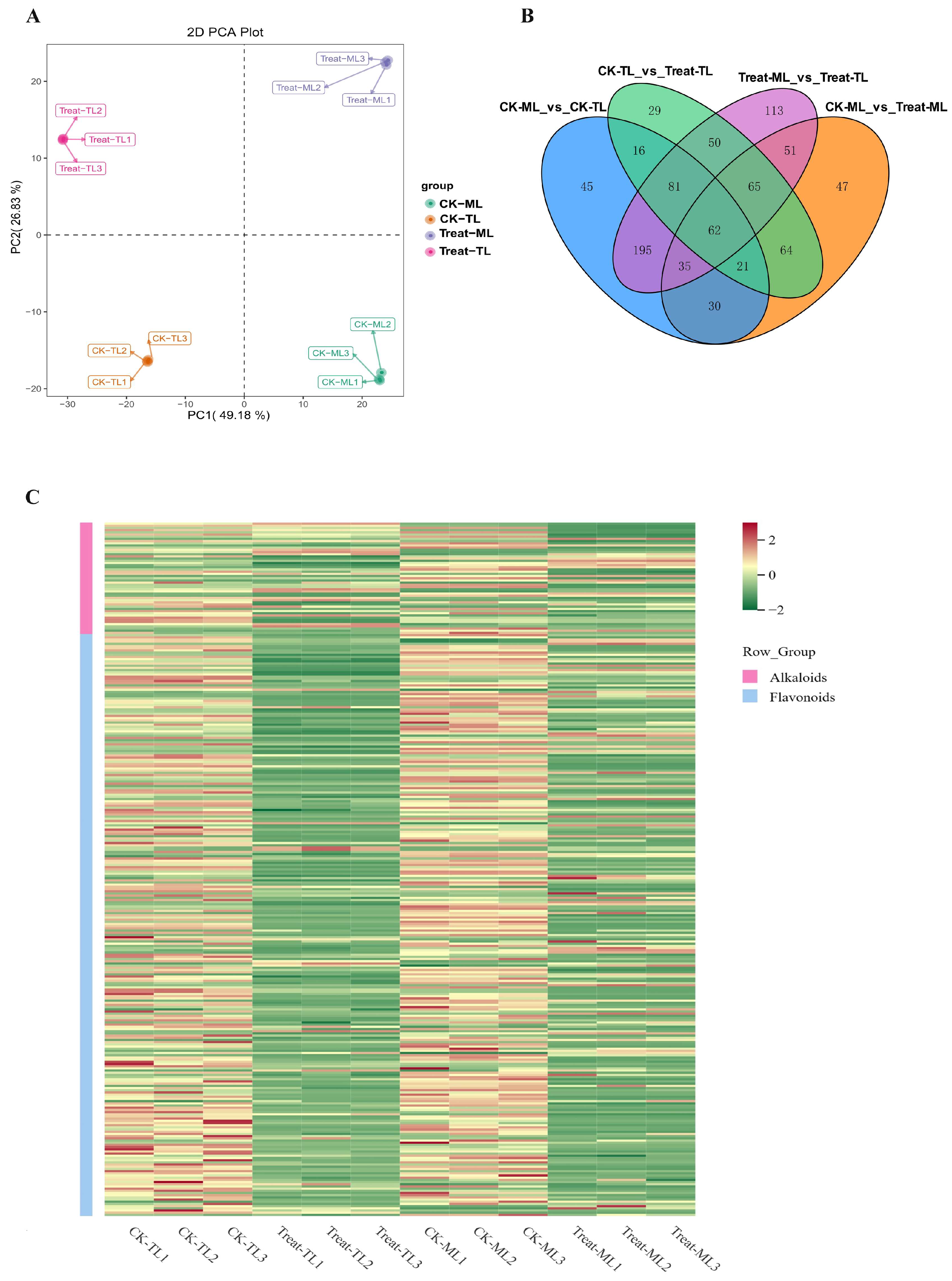
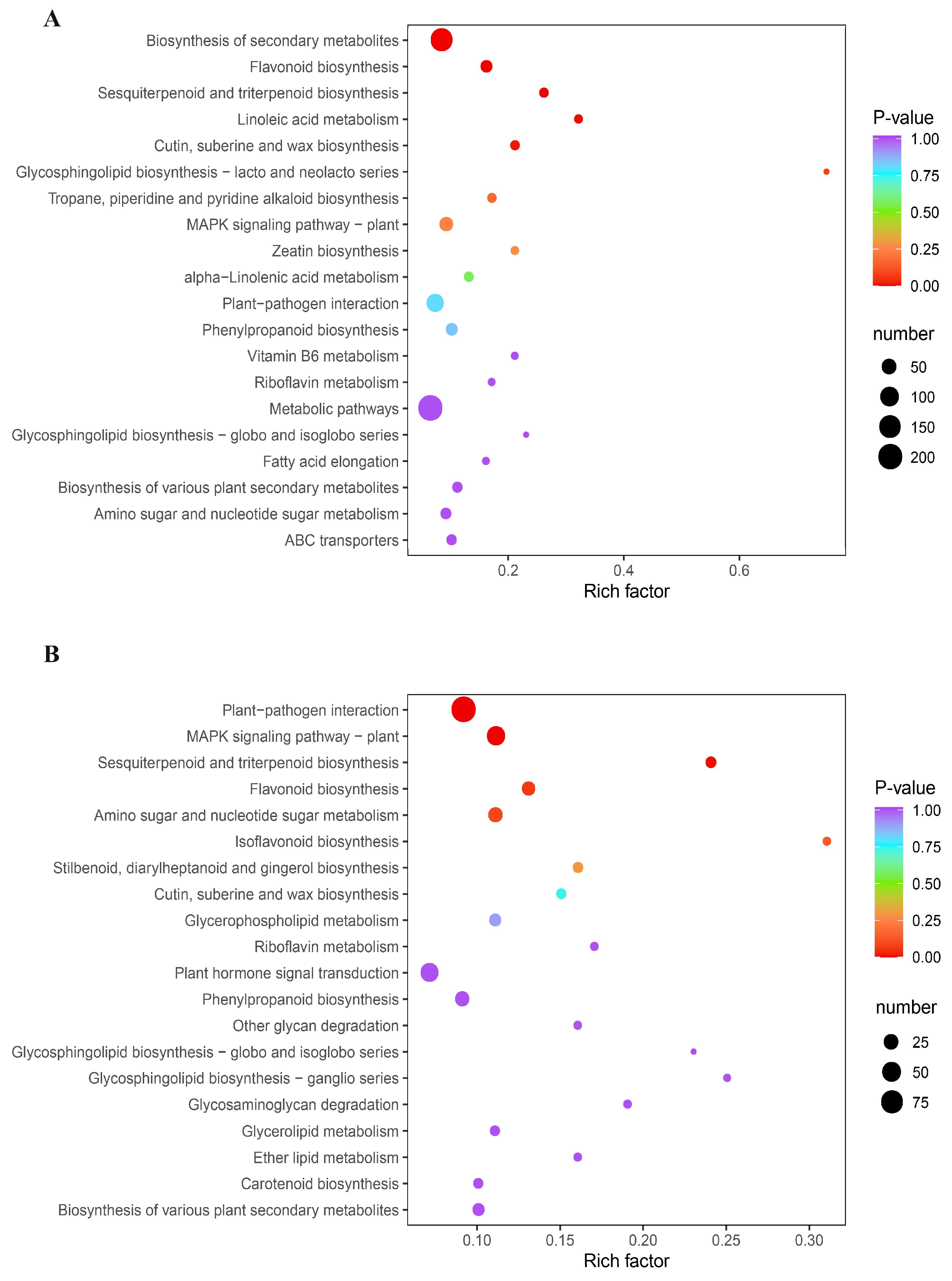
3.2. Diverse Metabolites Enrichment in Mulberry Leaves after B. subtilis Treatment
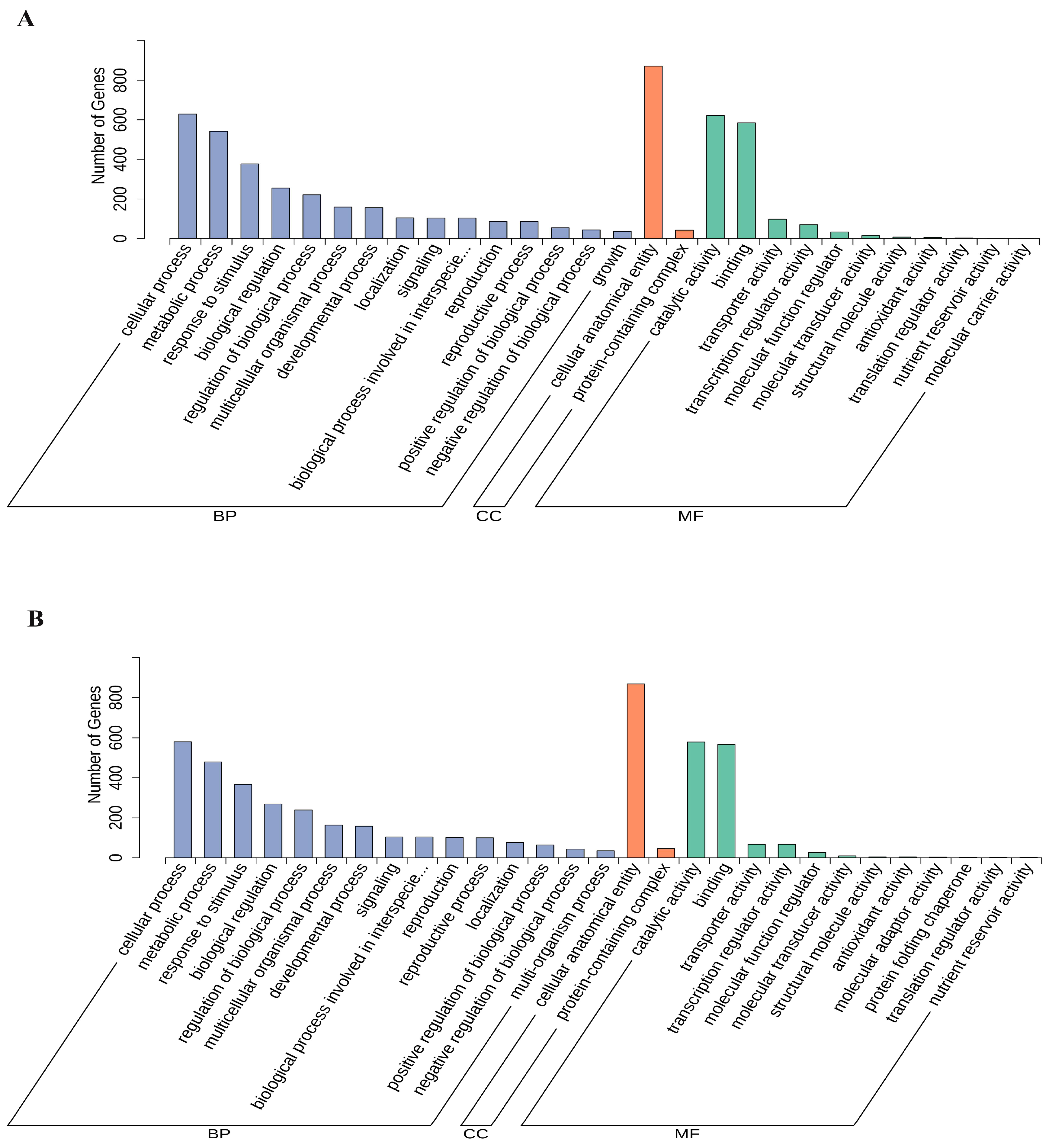
3.3. Transcriptome Changes in Mulberry Leaves after B. subtilis Treatment
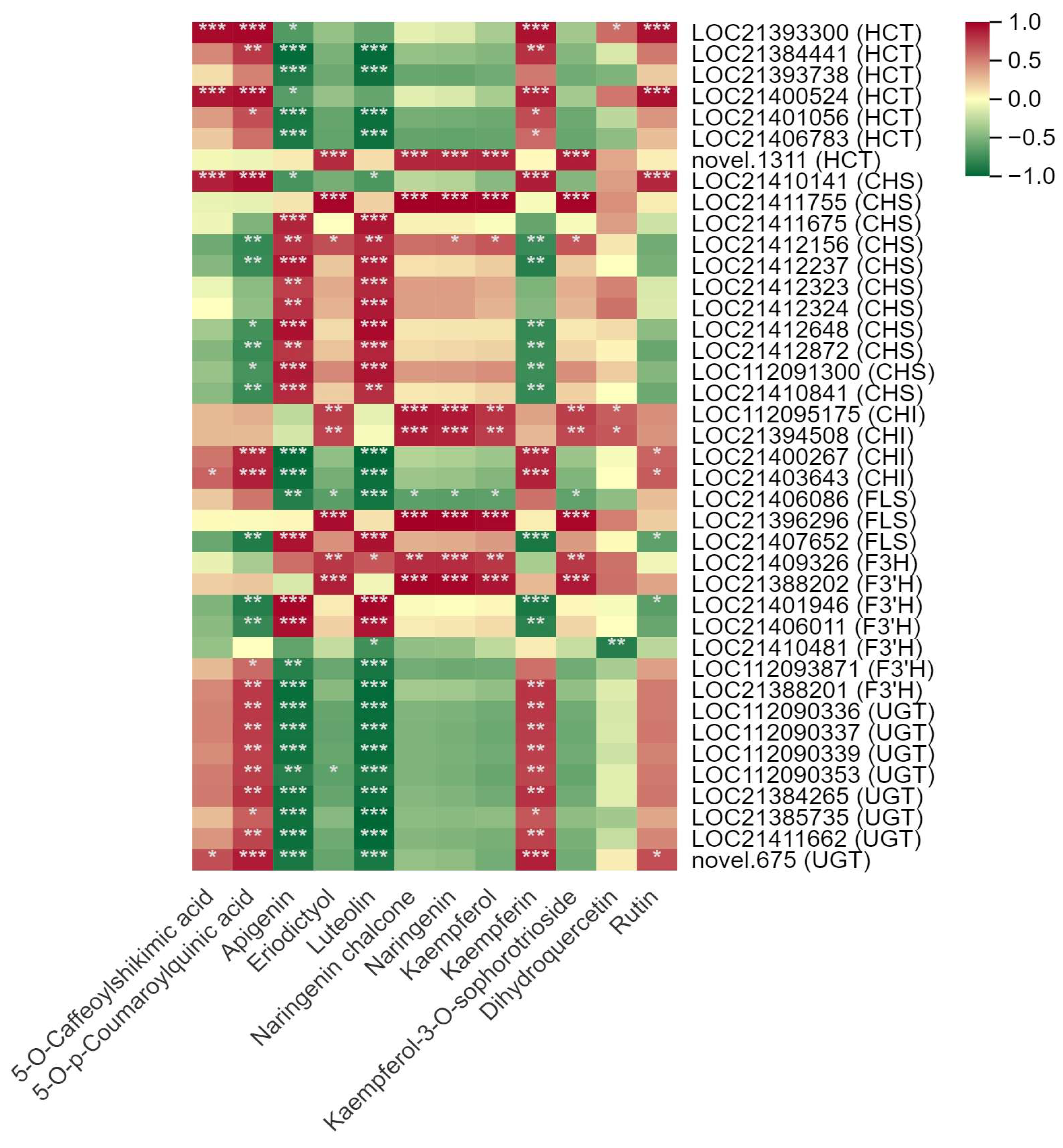
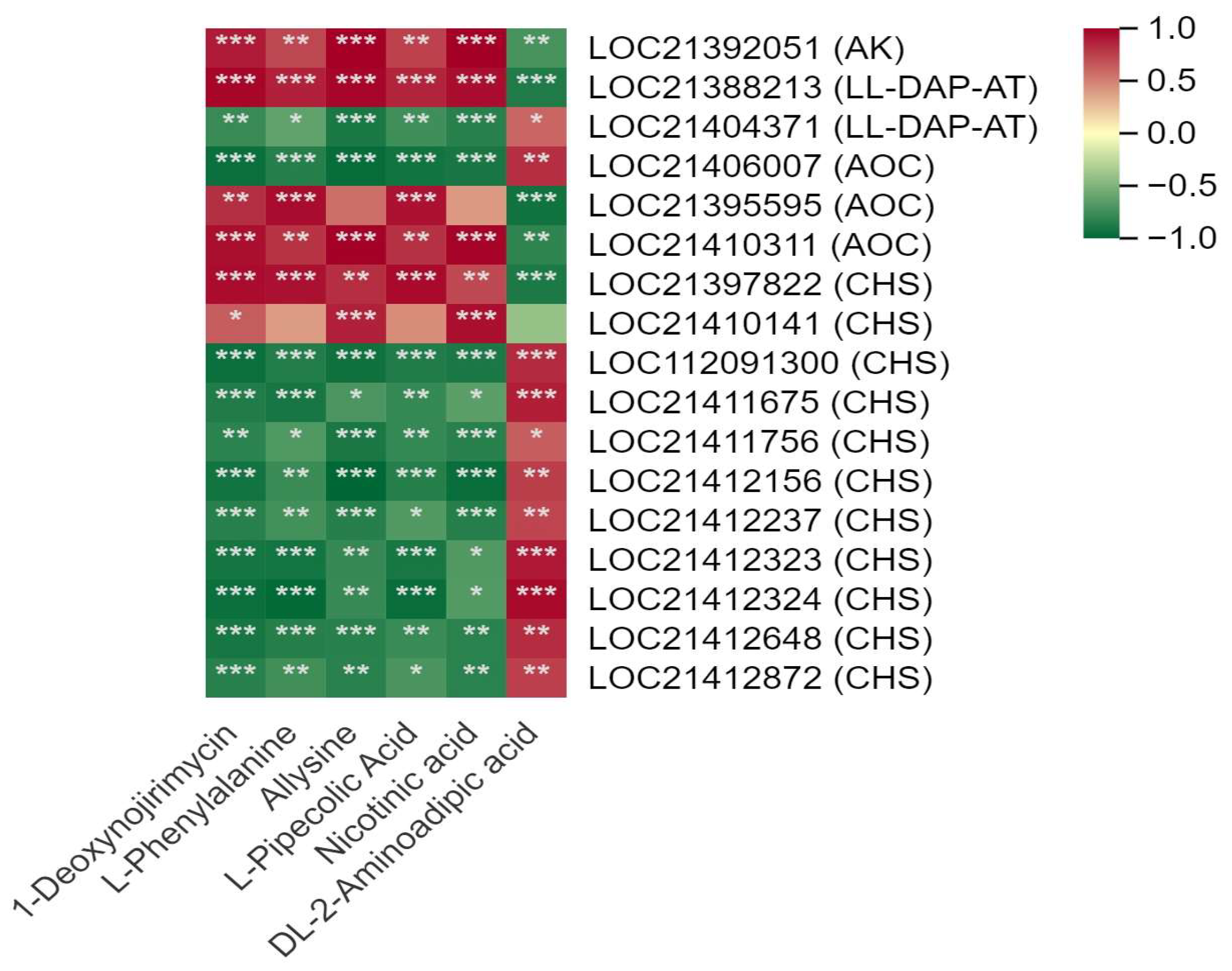
3.4. Correlation between DSMs and DEGs
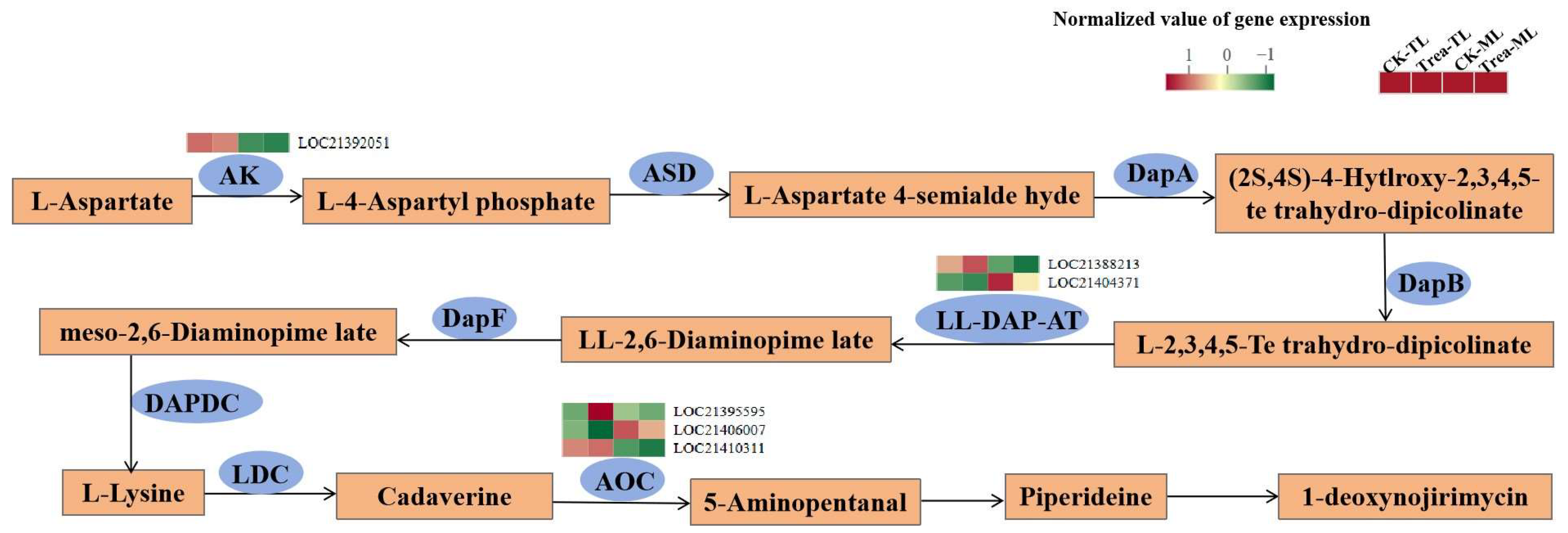
3.5. Analysis of the DEGs in the Flavonoid and Alkaloids Synthesis Pathways
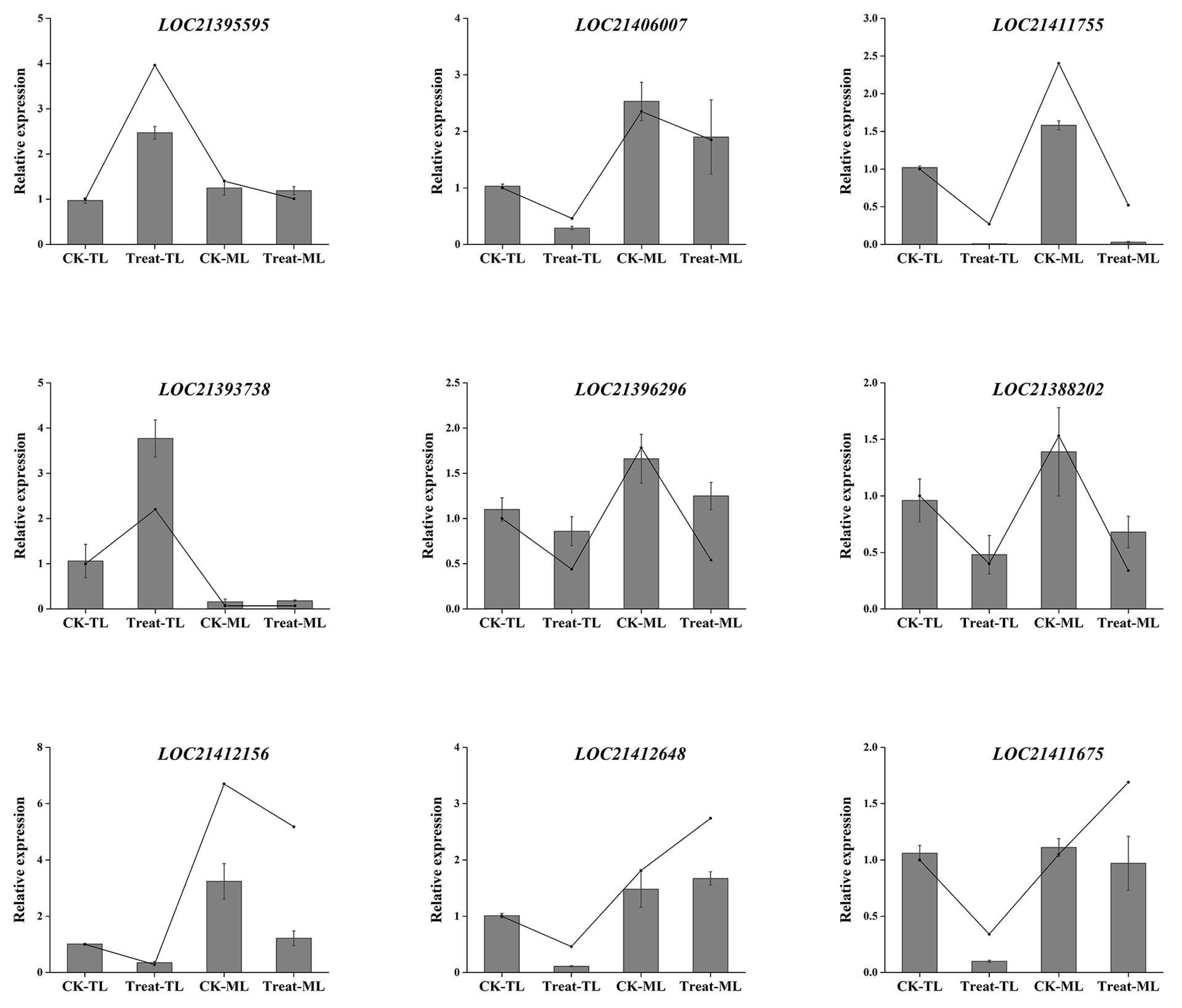
3.6. qRT-PCR Validation of Gene Expression
4. Discussion
4.1. Impact of BS on Flavonoids and Alkaloids Accumulation
4.2. Gene Expression Associated with Flavonoids and Alkaloids Synthesis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rohela, G.K.; Shukla, P.; Kumar, R.; Chowdhury, S.R. Mulberry (Morus spp.): An ideal plant for sustainable development. Trees For. People 2020, 2, 100011. [Google Scholar] [CrossRef]
- Kadam, R.A.; Dhumal, N.D.; Khyade, V.B. The Mulberry, Morus alba (L.): The medicinal herbal source for human health. Int. J. Curr. Microbiol. Appl. Sci 2019, 8, 2941–2964. [Google Scholar] [CrossRef]
- Łochyńska, M. Energy and nutritional properties of the white mulberry (Morus alba L.). J. Agric. Sci. Technol. A 2015, 5, 709–716. [Google Scholar]
- Yadav, S.; Nair, N.; Biharee, A.; Prathap, V.M.; Majeed, J. Updated ethnobotanical notes, phytochemistry and phytopharmacology of plants belonging to the genus Morus (Family: Moraceae). Phytomedicine Plus 2022, 2, 100120. [Google Scholar] [CrossRef]
- Batiha, G.E.-S.; Al-Snafi, A.E.; Thuwaini, M.M.; Teibo, J.O.; Shaheen, H.M.; Akomolafe, A.P.; Teibo, T.K.A.; Al-Kuraishy, H.M.; Al-Garbeeb, A.I.; Alexiou, A. Morus alba: A comprehensive phytochemical and pharmacological review. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2023, 396, 1399–1413. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.; Chai, X.; Hou, G.; Zhao, F.; Meng, Q. Phytochemistry, bioactivities and future prospects of mulberry leaves: A review. Food Chem. 2022, 372, 131335. [Google Scholar] [CrossRef] [PubMed]
- Maqsood, M.; Anam Saeed, R.; Sahar, A.; Khan, M.I. Mulberry plant as a source of functional food with therapeutic and nutritional applications: A review. J. Food Biochem. 2022, 46, e14263. [Google Scholar] [CrossRef] [PubMed]
- Mahesh, D.; Vidhathri, B.; Vidyashree, D.; Narayanaswamy, T.; Subbarayappa, C.; Muthuraju, R. Biochemical composition and pharmacological properties of mulberry (Morus spp.)—A review. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 2207–2217. [Google Scholar] [CrossRef]
- Shahana, S.; Nikalje, A.P.G. Phytochemistry and bioactivity of (Mulberry) plant: A comprehensive Morus alba. Asian J. Pharm. Pharmacol. 2019, 5, 207–217. [Google Scholar] [CrossRef]
- Hu, L.; Wang, C.; Guo, X.; Chen, D.; Zhou, W.; Chen, X.; Zhang, Q. Flavonoid levels and antioxidant capacity of mulberry leaves: Effects of growth period and drying methods. Front. Plant Sci. 2021, 12, 684974. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Mu, S.; Li, H.; Li, Y.; Feng, C.; Jin, J.-M.; Tang, S.-Y. Design and application of a novel high-throughput screening technique for 1-deoxynojirimycin. Sci. Rep. 2015, 5, 8563. [Google Scholar] [CrossRef]
- Asano, N. Sugar-mimicking glycosidase inhibitors: Bioactivity and application. Cell. Mol. Life Sci. 2009, 66, 1479–1492. [Google Scholar] [CrossRef]
- Hu, X.-Q.; Jiang, L.; Zhang, J.-G.; Deng, W.; Wang, H.-L.; Wei, Z.-J. Quantitative determination of 1-deoxynojirimycin in mulberry leaves from 132 varieties. Ind. Crop. Prod. 2013, 49, 782–784. [Google Scholar] [CrossRef]
- Vichasilp, C.; Nakagawa, K.; Sookwong, P.; Higuchi, O.; Luemunkong, S.; Miyazawa, T. Development of high 1-deoxynojirimycin (DNJ) content mulberry tea and use of response surface methodology to optimize tea-making conditions for highest DNJ extraction. LWT-Food Sci. Technol. 2012, 45, 226–232. [Google Scholar] [CrossRef]
- Yu, Y.; Li, H.; Zhang, B.; Wang, J.; Shi, X.; Huang, J.; Yang, J.; Zhang, Y.; Deng, Z. Nutritional and functional components of mulberry leaves from different varieties: Evaluation of their potential as food materials. Int. J. Food Prop. 2018, 21, 1495–1507. [Google Scholar] [CrossRef]
- Sugiyama, M.; Takahashi, M.; Katsube, T.; Koyama, A.; Itamura, H. Effects of applied nitrogen amounts on the functional components of mulberry (Morus alba L.) leaves. J. Agric. Food Chem. 2016, 64, 6923–6929. [Google Scholar] [CrossRef]
- Li, Q.; Liao, S.; Zhi, H.; Xing, D.; Xiao, Y.; Yang, Q. Characterization and sequence analysis of potential biofertilizer and biocontrol agent Bacillus subtilis strain SEM-9 from silkworm excrement. Can. J. Microbiol. 2019, 65, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Mahapatra, S.; Yadav, R.; Ramakrishna, W. Bacillus subtilis impact on plant growth, soil health and environment: Dr. Jekyll and Mr. Hyde. J. Appl. Microbiol. 2022, 132, 3543–3562. [Google Scholar] [CrossRef]
- Maksimov, I.; Blagova, D.; Veselova, S.; Sorokan, A.; Burkhanova, G.; Cherepanova, E.; Sarvarova, E.; Rumyantsev, S.; Alekseev, V.Y.; Khayrullin, R. Recombinant Bacillus subtilis 26DCryChS line with gene Btcry1Ia encoding Cry1Ia toxin from Bacillus thuringiensis promotes integrated wheat defense against pathogen Stagonospora nodorum Berk. and greenbug Schizaphis graminum Rond. Biol. Control 2020, 144, 104242. [Google Scholar] [CrossRef]
- Kloepper, J.; Gutierrez-Estrada, A.; McInroy, J. Photoperiod regulates elicitation of growth promotion but not induced resistance by plant growth-promoting rhizobacteria. Can. J. Microbiol. 2007, 53, 159–167. [Google Scholar] [CrossRef]
- Richardson, A.E. Regulating the phosphorus nutrition of plants: Molecular biology meeting agronomic needs. Plant Soil 2009, 322, 17–24. [Google Scholar] [CrossRef]
- Korenblum, E.; Dong, Y.; Szymanski, J.; Panda, S.; Jozwiak, A.; Massalha, H.; Meir, S.; Rogachev, I.; Aharoni, A. Rhizosphere microbiome mediates systemic root metabolite exudation by root-to-root signaling. Proc. Natl. Acad. Sci. USA 2020, 117, 3874–3883. [Google Scholar] [CrossRef] [PubMed]
- Mhlongo, M.I.; Piater, L.A.; Steenkamp, P.A.; Labuschagne, N.; Dubery, I.A. Metabolic Profiling of PGPR-Treated Tomato Plants Reveal Priming-Related Adaptations of Secondary Metabolites and Aromatic Amino Acids. Metabolites 2020, 10, 210. [Google Scholar] [CrossRef]
- Mashabela, M.D.; Piater, L.A.; Dubery, I.A.; Tugizimana, F.; Mhlongo, M.I. Rhizosphere Tripartite Interactions and PGPR-Mediated Metabolic Reprogramming towards ISR and Plant Priming: A Metabolomics Review. Biology 2022, 11, 346. [Google Scholar] [CrossRef]
- Korenblum, E.; Aharoni, A. Phytobiome metabolism: Beneficial soil microbes steer crop plants’ secondary metabolism. Pest Manag. Sci. 2019, 75, 2378–2384. [Google Scholar] [CrossRef]
- Nazari, F.; Safaie, N.; Soltani, B.M.; Shams-Bakhsh, M.; Sharifi, M. Bacillus subtilis affects miRNAs and flavanoids production in Agrobacterium-Tobacco interaction. Plant Physiol. Biochem. 2017, 118, 98–106. [Google Scholar] [CrossRef]
- Bashir, S.; Javed, S.; Al-Anazi, K.M.; Farah, M.A.; Ali, S. Bioremediation of Cadmium Toxicity in Wheat (Triticum aestivum L.) Plants Primed with L-Proline, Bacillus subtilis and Aspergillus niger. Int. J. Environ. Res. Public Health 2022, 19, 12683. [Google Scholar] [CrossRef]
- Mashabela, M.D.; Tugizimana, F.; Steenkamp, P.A.; Piater, L.A.; Dubery, I.A.; Mhlongo, M.I. Untargeted metabolite profiling to elucidate rhizosphere and leaf metabolome changes of wheat cultivars (Triticum aestivum L.) treated with the plant growth-promoting rhizobacteria Paenibacillus alvei (T22) and Bacillus subtilis. Front. Microbiol. 2022, 13, 971836. [Google Scholar] [CrossRef]
- Han, Z.; Ahsan, M.; Adil, M.F.; Chen, X.; Nazir, M.M.; Shamsi, I.H.; Zeng, F.; Zhang, G. Identification of the gene network modules highly associated with the synthesis of phenolics compounds in barley by transcriptome and metabolome analysis. Food Chem. 2020, 323, 126862. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Liu, A.; Li, L. Metabolomics and transcriptome analysis of the biosynthesis mechanism of flavonoids in the seeds of Euryale ferox Salisb at different developmental stages. Mol. Genet. Genom. 2021, 296, 953–970. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Shakeel, M.; Wang, D.; Qu, C.; Yang, S.; Ahmad, S.; Song, Z. Metabolome and transcriptome profiling unveil the mechanisms of light-induced anthocyanin synthesis in rabbiteye blueberry (vaccinium ashei: Reade). BMC Plant Biol. 2022, 22, 223. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, L.; Yang, J.; Hu, H.; Wei, G.; Cui, J.; Xu, J. Transcriptome and Metabolome Analyses Reveal Differences in Terpenoid and Flavonoid Biosynthesis in Cryptomeria fortunei Needles across Different Seasons. Front. Plant Sci. 2022, 13, 862746. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Chen, G.; Ma, B.; Zhong, C.; He, N. Metabolic profiling and transcriptome analysis of mulberry leaves provide insights into flavonoid biosynthesis. J. Agric. Food Chem. 2020, 68, 1494–1504. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.-Q.; Cheng, S.-Y.; Zhang, J.-Q.; Lin, H.-F.; Chen, Y.-Y.; Yue, S.-J.; Tian, M.; Tang, Y.-P.; Zhao, Y.-C. Morus alba L. Leaves–Integration of Their Transcriptome and Metabolomics Dataset: Investigating Potential Genes Involved in Flavonoid Biosynthesis at Different Harvest Times. Front. Plant Sci. 2021, 12, 736332. [Google Scholar] [CrossRef] [PubMed]
- Farouk, S.; AL-Huqail, A.A.; El-Gamal, S.M. Improvement of phytopharmaceutical and alkaloid production in periwinkle plants by endophyte and abiotic elicitors. Horticulturae 2022, 8, 237. [Google Scholar] [CrossRef]
- Cheng, X.; Li, D.; Jiang, Z.; Qu, C.; Yan, H.; Wu, Q. Metabolite profiling and transcriptomic analyses demonstrate the effects of biocontrol agents on alkaloid accumulation in Fritillaria thunbergii. BMC Plant Biol. 2023, 23, 435. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, M.; Chun, S.C.; Oh, J.W.; Paramasivan, M.; Saini, R.K.; Sahayarayan, J.J. Bacillus subtilis CBR05 for tomato (Solanum lycopersicum) fruits in South Korea as a novel plant probiotic bacterium (PPB): Implications from total phenolics, flavonoids, and carotenoids content for fruit quality. Agronomy 2019, 9, 838. [Google Scholar] [CrossRef]
- Akram, W.; Ahmad, A.; Yasin, N.A.; Anjum, T.; Ali, B.; Fatima, S.; Ahmed, S.; Simirgiotis, M.J.; Li, G. Mechanical strengthening and metabolic re-modulations are involved in protection against Fusarium wilt of tomato by B. subtilis IAGS174. J. Plant Interact. 2021, 16, 411–421. [Google Scholar] [CrossRef]
- Dimkić, I.; Janakiev, T.; Petrović, M.; Degrassi, G.; Fira, D. Plant-associated Bacillus and Pseudomonas antimicrobial activities in plant disease suppression via biological control mechanisms—A review. Physiol. Mol. Plant Pathol. 2022, 117, 101754. [Google Scholar] [CrossRef]
- Hashem, A.; Tabassum, B.; Abd_Allah, E.F. Bacillus subtilis: A plant-growth promoting rhizobacterium that also impacts biotic stress. Saudi J. Biol. Sci. 2019, 26, 1291–1297. [Google Scholar] [CrossRef]
- Bhambhani, S.; Kondhare, K.R.; Giri, A.P. Diversity in chemical structures and biological properties of plant alkaloids. Molecules 2021, 26, 3374. [Google Scholar] [CrossRef]
- Blake, C.; Christensen, M.N.; Kovács, Á.T. Molecular aspects of plant growth promotion and protection by Bacillus subtilis. Mol. Plant-Microbe Interact. 2021, 34, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Berens, M.L.; Wolinska, K.W.; Spaepen, S.; Ziegler, J.; Nobori, T.; Nair, A.; Krüler, V.; Winkelmüller, T.M.; Wang, Y.; Mine, A. Balancing trade-offs between biotic and abiotic stress responses through leaf age-dependent variation in stress hormone cross-talk. Proc. Natl. Acad. Sci. USA 2019, 116, 2364–2373. [Google Scholar] [CrossRef] [PubMed]
- Aerts, N.; Pereira Mendes, M.; Van Wees, S.C. Multiple levels of crosstalk in hormone networks regulating plant defense. Plant J. 2021, 105, 489–504. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Salcedo, E.M.; Tassotti, M.; Del Rio, D.; Hernández, F.; Martínez, J.J.; Mena, P. (Poly) phenolic fingerprint and chemometric analysis of white (Morus alba L.) and black (Morus nigra L.) mulberry leaves by using a non-targeted UHPLC–MS approach. Food Chem. 2016, 212, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, D.; Yang, Z.; Zeng, Q.; Luo, Y.; He, N. Flavones produced by mulberry flavone synthase type I constitute a defense line against the ultraviolet-B stress. Plants 2020, 9, 215. [Google Scholar] [CrossRef]
- Pandith, S.A.; Ramazan, S.; Khan, M.I.; Reshi, Z.A.; Shah, M.A. Chalcone synthases (CHSs): The symbolic type III polyketide synthases. Planta 2020, 251, 15. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Park, C.H.; Li, X.; Kim, Y.B.; Yang, J.; Sung, G.B.; Park, N.I.; Kim, S.; Park, S.U. Accumulation of rutin and betulinic acid and expression of phenylpropanoid and triterpenoid biosynthetic genes in mulberry (Morus alba L.). J. Agric. Food Chem. 2015, 63, 8622–8630. [Google Scholar] [CrossRef] [PubMed]
- Gharibi, S.; Tabatabaei, B.E.S.; Saeidi, G.; Talebi, M.; Matkowski, A. The effect of drought stress on polyphenolic compounds and expression of flavonoid biosynthesis related genes in Achillea pachycephala Rech. f. Phytochemistry 2019, 162, 90–98. [Google Scholar] [CrossRef]
- Hua, Y.; Dai, B.; Luo, Y.; Ding, Y. Integrated analysis of multiple metabolome and transcriptome revealed the accumulation of flavonoids and associated molecular regulation mechanisms in Rubus chingii Hu at different developmental stages. Plant Physiol. Biochem. 2023, 204, 108085. [Google Scholar] [CrossRef]
- Hoffmann, L.; Besseau, S.; Geoffroy, P.; Ritzenthaler, C.; Meyer, D.; Lapierre, C.; Pollet, B.; Legrand, M. Silencing of hydroxycinnamoyl-coenzyme A shikimate/quinate hydroxycinnamoyltransferase affects phenylpropanoid biosynthesis. Plant Cell 2004, 16, 1446–1465. [Google Scholar] [CrossRef]
- Cesarino, I.; Vanholme, R.; Goeminne, G.; Vanholme, B.; Boerjan, W. Shikimate hydroxycinnamoyl transferase (HCT) activity assays in Populus nigra. Bio-Protocol 2013, 3, e978. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, D.; Liu, J.; Yu, X.; Wang, R.; Wei, Y.; Wen, C.; Ouyang, Z. Transcriptomic analysis of key genes involved in chlorogenic acid biosynthetic pathway and characterization of MaHCT from Morus alba L. Protein Expr. Purif. 2019, 156, 25–35. [Google Scholar] [CrossRef]
- Su, Z.; Sun, M.; Cai, Z.; Guo, S.; Li, J.; Zhao, B.; Shen, Z.; Ma, R.; Yan, J.; Yu, M. Identification and expression analysis of chlorogenic acid biosynthesis key gene PpHCT in peach. Hortic. Plant J. 2023, 9, 670–680. [Google Scholar] [CrossRef]
- Wang, D.; Zhao, L.; Wang, D.; Liu, J.; Yu, X.; Wei, Y.; Ouyang, Z. Transcriptome analysis and identification of key genes involved in 1-deoxynojirimycin biosynthesis of mulberry (Morus alba L.). PeerJ 2018, 6, e5443. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Luo, Y.; Xia, X.; He, J.; Zhang, J.; Zeng, Q.; Li, D.; Ma, B.; Zhang, S.; Zhai, C. Dehydrogenase MnGutB1 catalyzes 1-deoxynojirimycin biosynthesis in mulberry. Plant Physiol. 2023, 192, 1307–1320. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Y.; Huang, J.; Deng, Z.; Wang, Y.; Jiang, X.; Wang, J. Soil Application of Bacillus subtilis Regulates Flavonoid and Alkaloids Biosynthesis in Mulberry Leaves. Metabolites 2024, 14, 180. https://doi.org/10.3390/metabo14040180
Yu Y, Huang J, Deng Z, Wang Y, Jiang X, Wang J. Soil Application of Bacillus subtilis Regulates Flavonoid and Alkaloids Biosynthesis in Mulberry Leaves. Metabolites. 2024; 14(4):180. https://doi.org/10.3390/metabo14040180
Chicago/Turabian StyleYu, Yanfang, Jinzhi Huang, Zhenhua Deng, Yawei Wang, Xinfeng Jiang, and Junwen Wang. 2024. "Soil Application of Bacillus subtilis Regulates Flavonoid and Alkaloids Biosynthesis in Mulberry Leaves" Metabolites 14, no. 4: 180. https://doi.org/10.3390/metabo14040180
APA StyleYu, Y., Huang, J., Deng, Z., Wang, Y., Jiang, X., & Wang, J. (2024). Soil Application of Bacillus subtilis Regulates Flavonoid and Alkaloids Biosynthesis in Mulberry Leaves. Metabolites, 14(4), 180. https://doi.org/10.3390/metabo14040180






