Metabolite Profiling of External and Internal Petals in Three Different Colors of Tea Flowers (Camellia sinensis) Using Widely Targeted Metabolomics
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Materials
2.3. Widely Targeted Metabolic Analysis
2.4. Data Processing
3. Results
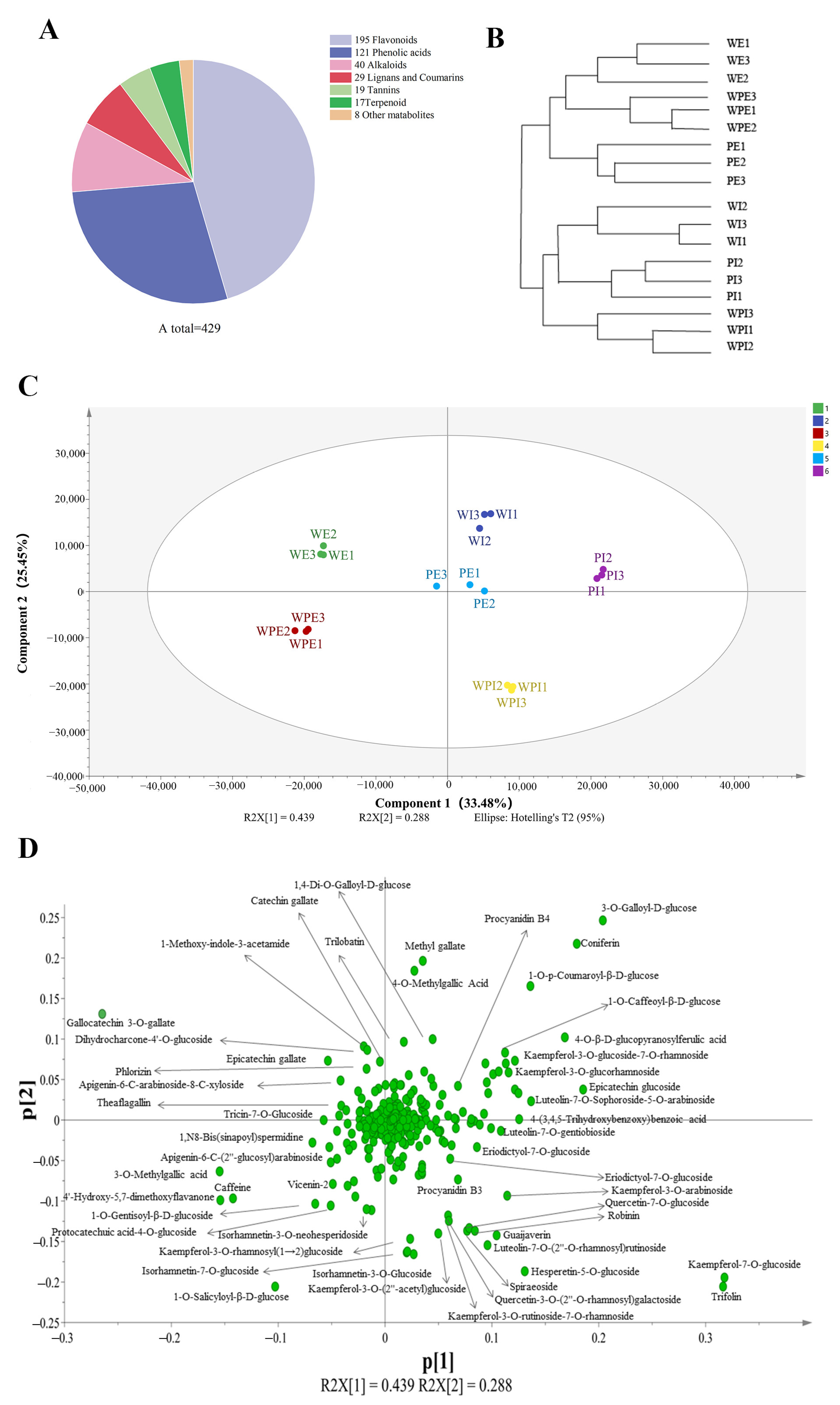
3.1. Overall Characterization of Non-Volatile Metabolites
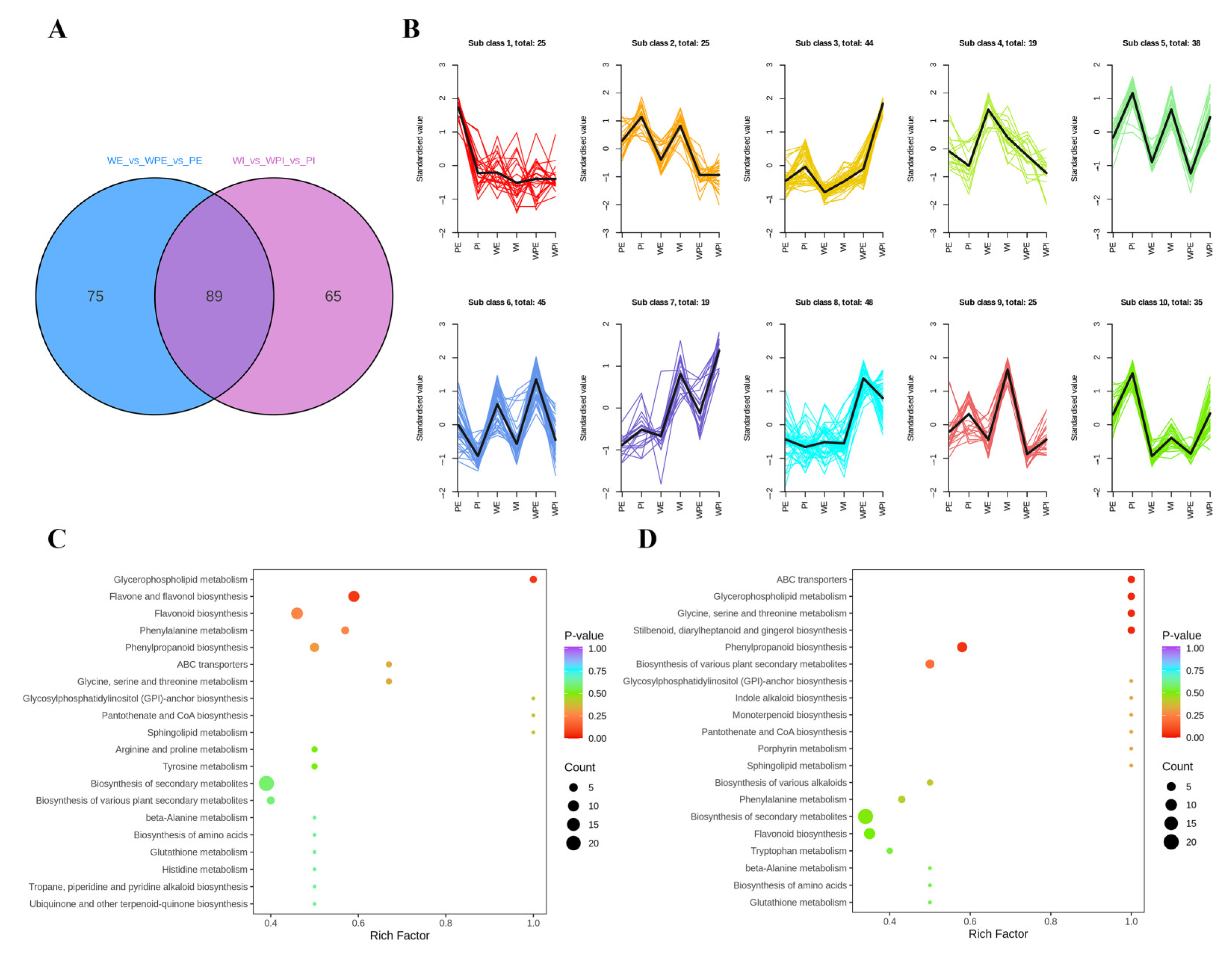
3.2. Comparison of Non-Volatile Secondary Metabolites among the Petals of Three Different Tinctorial Flowers
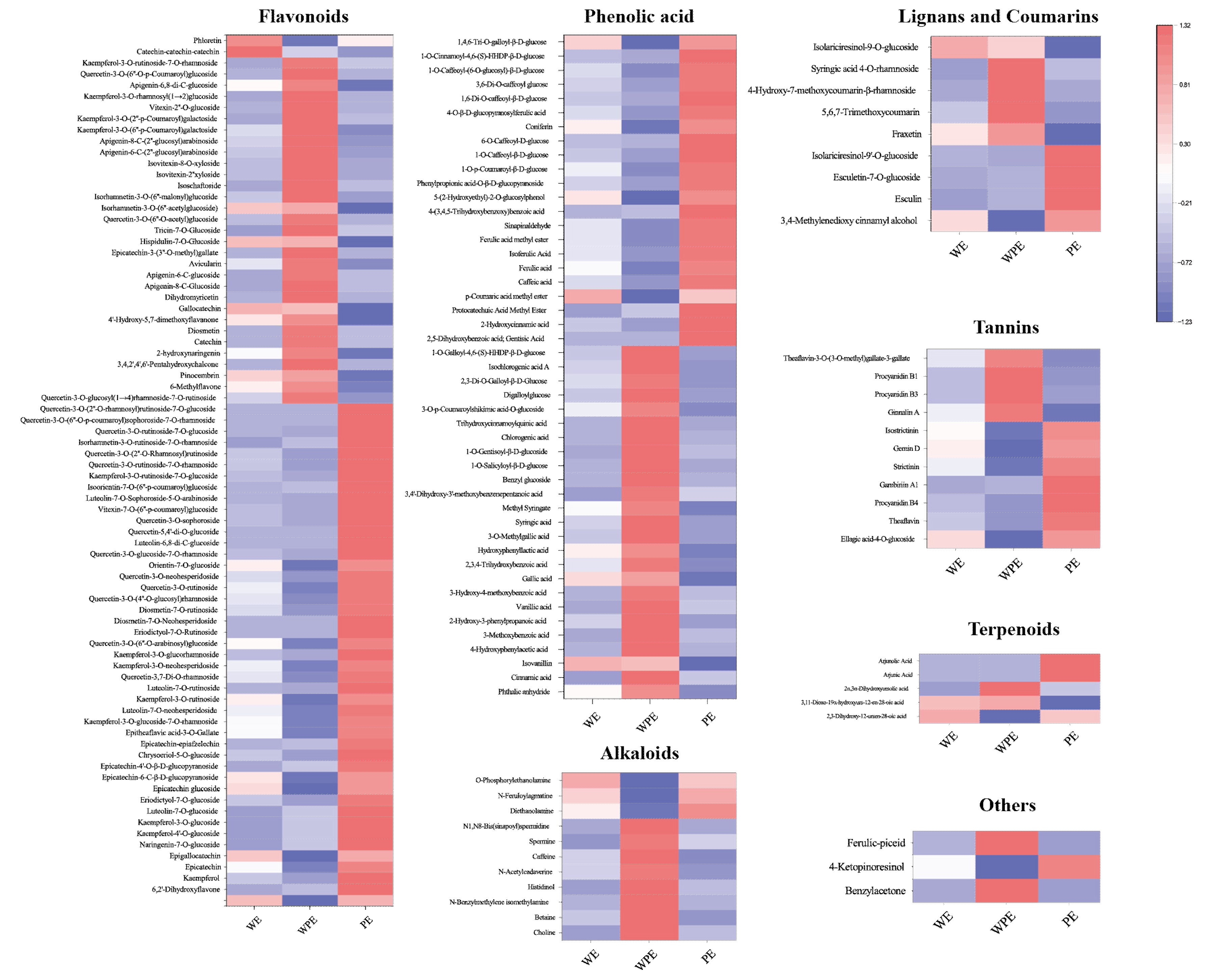
3.2.1. Comparison of Non-Volatile Secondary Metabolites among the External Petals
3.2.2. Comparison of Non-Volatile Secondary Metabolites among the Internal Petals
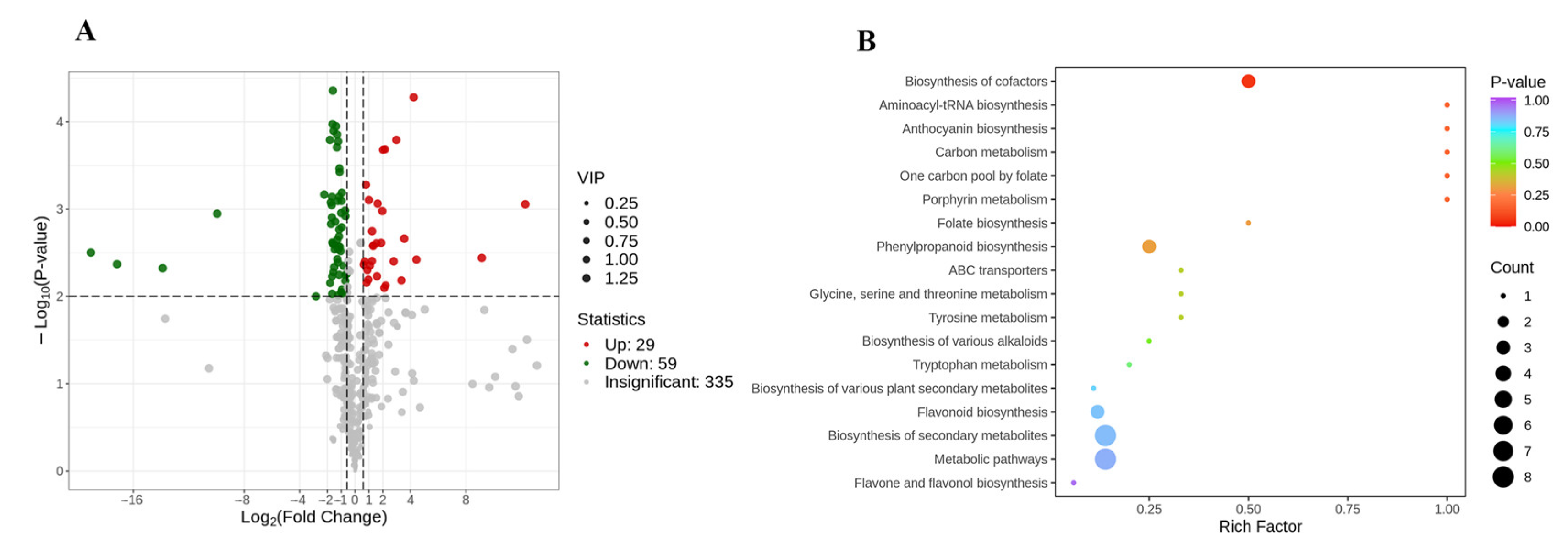
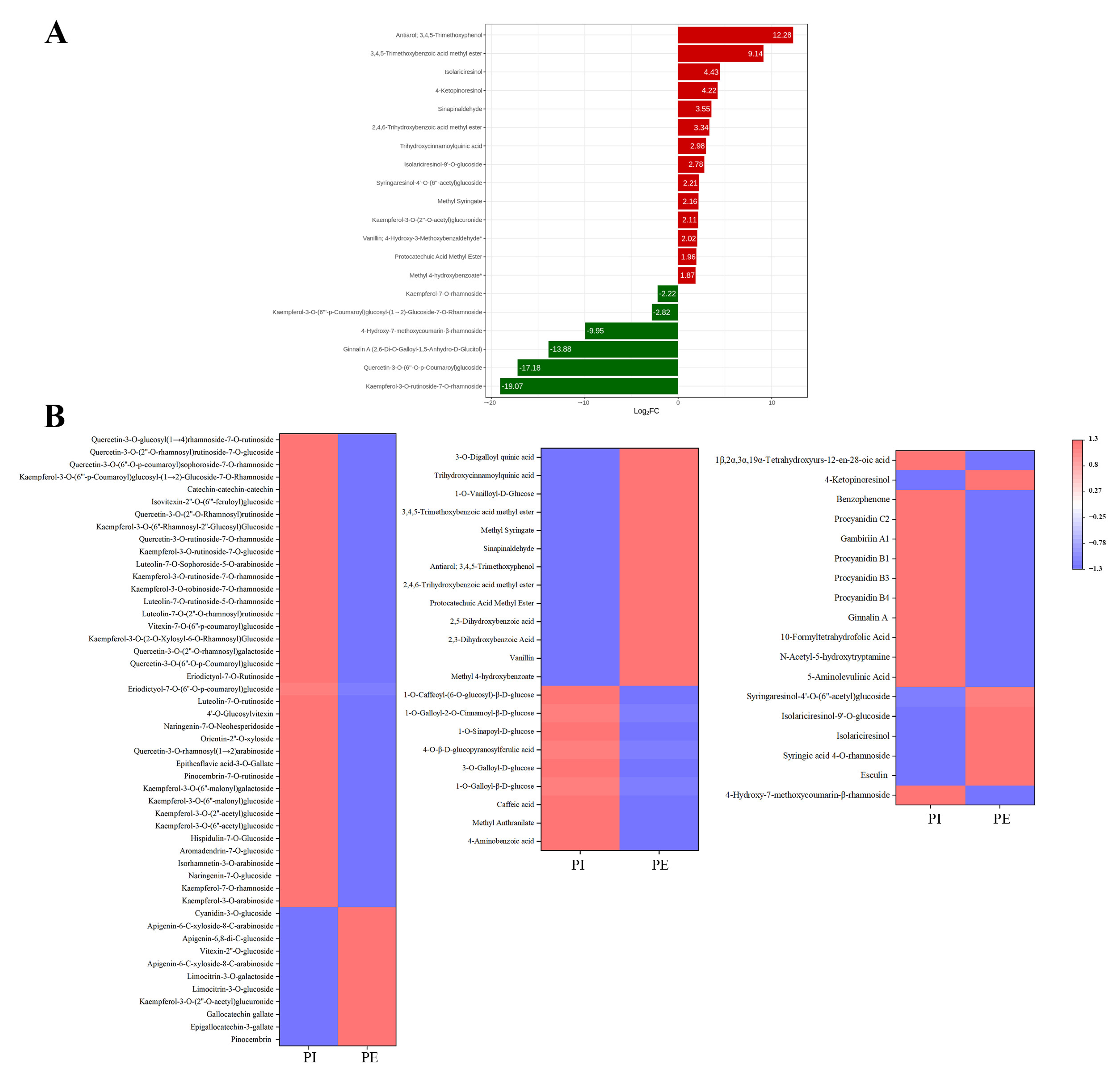
3.3. Comparison of Non-Volatile Secondary Metabolites between the Internal and External Petals of Pink Flowers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, Y.; Fu, X.; Mei, X.; Zhou, Y.; Du, B.; Tu, Y.; Yang, Z. Characterization of functional proteases from flowers of tea (Camellia sinensis) plants. J. Funct. Foods 2016, 25, 149–159. [Google Scholar] [CrossRef]
- Fan, K.; Zhang, Q.; Liu, M.; Ma, L.; Shi, Y.; Ruan, J. Metabolomic and transcriptional analyses reveal the mechanism of C, N allocation from source leaf to flower in tea plant (Camellia sinensis. L). J. Plant Physiol. 2018, 232, 200–208. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, Y.; Zeng, L.; Dong, F.; Tu, Y.; Yang, Z. Occurrence of functional molecules in the flowers of tea (Camellia sinensis) plants: Evidence for a second resource. Molecules 2018, 23, 790. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Tu, Y.; Baldermann, S.; Dong, F.; Xu, Y.; Watanabe, N. Isolation and identification of compounds from the ethanolic extract of flowers of the tea (Camellia sinensis) plant and their contribution to the antioxidant capacity. LWT-Food Sci. Technol. 2009, 42, 1439–1443. [Google Scholar] [CrossRef]
- Matsuda, H.; Hamao, M.; Nakamura, S.; Kon’I, H.; Murata, M.; Yoshikawa, M. Medicinal flowers. XXXIII.1 anti-hyperlipidemic and anti-hyperglycemic effects of chakasaponins I-III and structure of chakasaponin IV from flower buds of Chinese tea plant (Camellia sinensis). Chem. Pharm. Bull. 2012, 60, 674–689. [Google Scholar] [CrossRef] [PubMed]
- Dan, C.; Guijie, C.; Yi, S.; Xiaoxiong, Z.; Hong, Y. Physiological genetics, chemical composition, health benefits and toxicology of tea (Camellia sinensis L.) flower: A review. Food Res. Int. 2020, 137, 109584. [Google Scholar]
- Dissanayake, C.-Y.; Moon, H.-H.; Yang, K.-M.; Lee, Y.; Han, C.-H. The effects of green tea (Camellia sinensis) flower extract on melanin synthesis in B16-F10 melanoma cells. Korean J. Vet. Res. 2018, 58, 65–72. [Google Scholar] [CrossRef]
- Yaomin, W.; Ning, R.; Gary, O.R.; Bo, L.; Yon, R.; Youying, T.; Yi, C.C. Anti-proliferative effect and cell cycle arrest induced by saponins extracted from tea (Camellia sinensis) flower in human ovarian cancer cells. J. Funct. Foods 2017, 37, 310–321. [Google Scholar]
- Niichiro, K.; Toshio, M.; Chiaki, M.; Kiyofumi, N.; Shuhei, O.; Ayaka, N.; Masayuki, Y.; Osamu, M. The antiproliferative effect of chakasaponins I and II, floratheasaponin A, and epigallocatechin 3-O-gallate isolated from Camellia sinensis on human digestive tract carcinoma cell lines. Int. J. Mol. Sci. 2016, 17, 1979. [Google Scholar]
- Wang, Y.; Xia, C.; Chen, L.; Chen, Y.C.; Tu, Y. Saponins extracted from tea (Camellia sinensis) flowers induces autophagy in ovarian cancer cells. Molecules 2020, 25, 5245. [Google Scholar] [CrossRef]
- Ren, N.; Chen, L.; Li, B.; Rankin, G.O.; Chen, Y.C.; Tu, Y. Purified tea (Camellia sinensis (L.) Kuntze) flower saponins induce the p53-dependent intrinsic apoptosis of cisplatin-resistant ovarian cancer cells. Int. J. Mol. Sci. 2020, 21, 4324. [Google Scholar] [CrossRef]
- Hisakazu, Y.; Yoshio, S.; Nobutaka, T.; Kiyotoshi, T.; Masaki, F. Endogenous inhibitors for spore germination in Lygodium japonicum and their inhibitory effects on pollen germinations in Camellia japonica and Camellia sinensis. Agric. Biol. Chem. 2014, 44, 1697–1699. [Google Scholar]
- Yan, H.; Hu, S.; Jiawei, Z.; Peng, L.; Weijiang, S. Effects of processing treatments on quality and antioxidant activity of tea plant flower. Food Sci. 2020, 41, 165–170. [Google Scholar]
- Tang, D.; Shen, Y.; Li, F.; Yue, R.; Duan, J.; Ye, Z.; Lin, Y.; Zhou, W.; Yang, Y.; Chen, L.; et al. Integrating metabolite and transcriptome analysis revealed the different mechanisms of characteristic compound biosynthesis and transcriptional regulation in tea flowers. Front. Plant Sci. 2022, 13, 1016692. [Google Scholar] [CrossRef]
- Xu, R.J.; Wang, L.; Wang, M.C.; Ye, H.; Tu, Y.Y.; Zeng, X.X. Determination of sugers, catechins and free amino acids in tea flowers by high performance liquid chromatography. Food Sci. 2012, 33, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Kim, E.; Yang, L.; Huang, Y.; Ren, N.; Li, B.; He, P.; Tu, Y.; Wu, Y. Effect of a combined microwave-assisted drying and air drying on improving active nutraceutical compounds, flavor quality, and antioxidant properties of (Camellia sinensis L. cv. Longjing 43) flowers. Food Qual. Saf. 2021, 5, 40. [Google Scholar] [CrossRef]
- Wang, L.; Xu, R.; Hu, B.; Li, W.; Sun, Y.; Tu, Y.; Zeng, X. Analysis of free amino acids in Chinese teas and flower of tea plant by high performance liquid chromatography combined with solid-phase extraction. Food Chem. 2010, 123, 1259–1266. [Google Scholar] [CrossRef]
- Li, J.; Chen, L.; Kim, E.; Li, B.; Tu, Y. Research on flavonoid glycosides of tea flower in different tea plant cultivars. J. Zhejiang Univ. (Agric. Life Sci.) 2019, 45, 707–714. [Google Scholar]
- Fu, M.; Yang, X.; Zheng, J.; Wang, L.; Yang, X.; Tu, Y.; Ye, J.; Zhang, W.; Liao, Y.; Cheng, S.; et al. Unraveling the regulatory mechanism of color diversity in Camellia japonica petals by integrative transcriptome and metabolome analysis. Front. Plant Sci. 2021, 12, 685136. [Google Scholar] [CrossRef]
- Chen, M.B.; Rothenberg, D.O.N.; Zhang, L.Y. Analysis of aroma components of pink and white tea flowers with the purple bud in SPME/GC-MS. Guangdong Agric. Sci. 2018, 45, 88–94. [Google Scholar]
- Mei, X.; Hu, L.; Deng, C.; Li, Y.; Long, L.; Tian, W.; Song, Y.; Zhou, C. Analysis of volatile metabolites in white and purple flowers of tea (Camellia sinensis). J. Food Saf. Qual. 2022, 13, 4447–4453. [Google Scholar]
- Shen, X.; Shi, L.; Pan, H.; Li, B.; Wu, Y.; Tu, Y. Identification of triterpenoid saponins in flowers of four Camellia Sinensis cultivars from Zhejiang province: Differences between cultivars, developmental stages, and tissues. Ind. Crop. Prod. 2017, 95, 140–147. [Google Scholar] [CrossRef]
- Chen, Q.; Li, Q.; Liang, Y.; Zu, M.; Chen, N.; Canup, B.S.; Luo, L.; Wang, C.; Zeng, L.; Xiao, B. Natural exosome-like nanovesicles from edible tea flowers suppress metastatic breast cancer via ROS generation and microbiota modulation. Acta Pharm. Sin. B 2022, 12, 907–923. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Li, W.; He, R.; Li, Y.; Tsoi, B.; Zhai, Y.; Kurihara, H. Anti-inflammatory effects of a polyphenols-rich extract from tea (Camellia sinensis) flowers in acute and chronic mice models. Oxidative Med. Cell. Longev. 2012, 2012, 537923. [Google Scholar] [CrossRef]
- Wei, C.; Liang, G.; Zilong, G.; Wensheng, W.; Hongyan, Z.; Xianqing, L.; Sibin, Y.; Lizhong, X.; Jie, L. A Novel integrated method for large-scale detection, identification, and quantification of widely targeted metabolites: Application in the study of rice metabolomics. Mol. Plant 2013, 6, 1769–1780. [Google Scholar]
- Liu, H.; Liu, Q.; Chen, Y.; Zhu, Y.; Zhou, X.; Li, B. Full-length transcriptome sequencing provides insights into flavonoid biosynthesis in Camellia nitidissima petals. Gene 2023, 850, 146924. [Google Scholar] [CrossRef]
- Liu, C.; Yu, Q.; Li, Z.; Jin, X.; Xing, W. Metabolic and transcriptomic analysis related to flavonoid biosynthesis during the color formation of Michelia crassipes tepal. Plant Physiol. Biochem. 2020, 155, 938–951. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Nie, J.; Shi, L.; Xie, Y.; Tan, D.; Yang, X.; Zhang, C.; Zheng, J. Transcriptomic and metabolomic profiling reveals the mechanisms of color and taste development in cherry tomato cultivars. LWT-Food Sci. Technol. 2022, 167, 113810. [Google Scholar] [CrossRef]
- Liao, Y.; Zhou, X.; Zeng, L. How does tea (Camellia sinensis) produce specialized metabolites which determine its unique quality and function: A review. Crit. Rev. Food Sci. Nutr. 2021, 62, 113810. [Google Scholar] [CrossRef]
- Norihiko, T.; Yoshiyuki, T.; Atsushi, N.; Toshio, H. Anthocyanins from red flower tea (Benibana-cha), Camellia sinensis. Phytochemistry 2001, 56, 11–17. [Google Scholar]
- Liu, F.; Wang, Y.; Ding, Z.; Zhao, L.; Xiao, J.; Wang, L.; Ding, S. Transcriptomic analysis of flower development in tea (Camellia sinensis (L.)). Gene 2017, 631, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wang, B.W.; Feng, G.; Wang, H.F.; Zhang, Y.H.; Wang, J.H.; Liu, Z.Q.; Deng, W.W. Effect of different drying methods on quality of tea plant flowers. Food Sci. Technol. 2017, 42, 82–87. [Google Scholar]
- Lin, Y.; Wu, S.; Lin, J. Determination of teapolyphenols and caffeine in tea flowers (Camellia sinensis) and their hydroxyl radical scavenging and nitric oxide suppressing effects. J. Agric. Food Chem. 2003, 51, 975–980. [Google Scholar] [CrossRef]
- de Villiers, A.; Venter, P.; Pasch, H. Recent advances and trends in the liquid-chromatography–mass spectrometry analysis of flavonoids. J. Chromatogr. A 2016, 1430, 16–78. [Google Scholar] [CrossRef]
- Liu, F.; Wang, Y.; Corke, H.; Zhu, H. Dynamic changes in flavonoids content during congou black tea processing. LWT-Food Sci. Technol. 2022, 170, 114073. [Google Scholar] [CrossRef]
- Zou, L.; Shen, S.; Wei, Y.; Jia, H.; Li, T.; Yin, X.; Lu, C.; Cui, Q.; He, F.; Deng, W.; et al. Evaluation of the effects of solar withering on nonvolatile compounds in white tea through metabolomics and transcriptomics. Food Res. Int. 2022, 162, 112088. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Yu, X.; Zhang, Y.; Lu, X.; Liu, Q.; Jia, Y.; He, C. Sensory descriptive analysis of green tea: Correlation with chemical components. IOP Conf. Ser. Earth Environ. Sci. 2020, 461, 012013. [Google Scholar] [CrossRef]
- Sheng, J.; Su, W.; Shi, J.; Shi, B. Research progress of flavonoids in tea flower. J. Food Saf. Qual. 2022, 13, 1019–1026. [Google Scholar]
- Rui, M.; Lin, Y.; Teng, C.; Yang, Q. Antioxidant activity in vitro of tea flowers. Food Ind. 2019, 40, 218–221. [Google Scholar]
- Menezes, J.C.J.M.; Campos, V.R. Natural biflavonoids as potential therapeutic agents against microbial diseases. Sci. Total Environ. 2021, 769, 145168. [Google Scholar] [CrossRef]
- Sartinah, A.; Ihsan, S.; Kasmawati, H.; Andriyani, R.; Adjeng, A.N.T.; Arba, M. Radical scavenging assay and determination flavonoid and phenolic total of extract and fractions of raghu bark (Dracontomelon dao (Blanco) Merr). Res. J. Pharm. Technol. 2020, 13, 2335–2339. [Google Scholar]
- Voltaire, S.; Poliana, D.G.; Ligia, D.F.M.; Isabel, C.T. Tracking bioactive compounds with colour changes in foods—A review. Dyes Pigment 2013, 98, 601–608. [Google Scholar]
- Zhang, S.; Guo, H.; Pei, X.; Li, C.; Cheng, H. Expression of flavonoid3′-hydrolyase from eupatorium adenophorum in Tobacca. Sci. Agric. Sin. 2009, 42, 4182–4186. [Google Scholar]
- Nijveldt, R.J.; van Nood, E.; van Hoorn, D.E.; Boelens, P.G.; van Norren, K.; van Leeuwen, P.A. Flavonoids: A review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Pu, Z.-J.; Yue, S.-J.; Zhou, G.-S.; Yan, H.; Shi, X.-Q.; Zhu, Z.-H.; Huang, S.-L.; Peng, G.-P.; Chen, Y.-Y.; Bai, J.-Q.; et al. The comprehensive evaluation of safflowers in different producing areas by combined analysis of color, chemical compounds, and biological activity. Molecules 2019, 24, 3381. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, L.; Wang, S.; Niu, X. Analysis of anthocyanins and flavonols in petals of 10 Rhododendron species from the Sygera Mountains in southeast Tibet. Plant Physiol. Biochem. 2016, 104, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Huang, S.; Yang, J.; Li, Z.; Zhang, M.; Fang, Y.; Yang, Q.; Jin, W. Metabolite profiling of violet, white and pink flowers revealing flavonoids composition patterns in Rhododendron pulchrum sweet. J. Biosci. 2021, 46, 3. [Google Scholar] [CrossRef]
- Du, H.; Lai, L.; Wang, F.; Sun, W.; Zhang, L.; Li, X.; Wang, L.; Jiang, L.; Zheng, Y. Characterisation of flower colouration in 30 Rhododendron species via anthocyanin and flavonol identification and quantitative traits. Plant Biol. 2018, 20, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Su, M.; Damaris, R.N.; Hu, Z.; Yang, P.; Deng, J. Metabolomic analysis on the petal of ‘Chen Xi’ rose with light-induced color changes. Plants 2021, 10, 2065. [Google Scholar] [CrossRef]
- Li, X.; Wang, J.; Sun, Z.; Wang, J.; Yin, H.; Fan, Z.; Li, J. Flavonoid components and their effect on flower colors in Camellia nitidissima, white C. japonica and their three hybrid cultivars. Acta Hortic. Sin. 2019, 46, 1145–1154. [Google Scholar]
- Yaoa, L.; Jiang, Y.; Singanusong, R.; Datta, N.; Raymont, K. Phenolic acids in Australian Melaleuca, Guioa, Lophostemon, Banksia and Helianthus honeys and their potential for floral authentication. Food Res. Int. 2005, 38, 651–658. [Google Scholar] [CrossRef]
- Wang, P.; Liu, Y.; Zhang, L.; Wang, W.; Hou, H.; Zhao, Y.; Jiang, X.; Yu, J.; Tan, H.; Wang, Y.; et al. Functional demonstration of plant flavonoid carbocations proposed to be involved in the biosynthesis of proanthocyanidins. Plant J. Cell Mol. Biol. 2020, 101, 18–36. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, T.; Ma, X.; Zhou, Y.; Yang, H.; Wang, Y.; Chen, T.; Chen, Q.; Deng, Y. Metabolite Profiling of External and Internal Petals in Three Different Colors of Tea Flowers (Camellia sinensis) Using Widely Targeted Metabolomics. Metabolites 2023, 13, 784. https://doi.org/10.3390/metabo13070784
Zhang T, Ma X, Zhou Y, Yang H, Wang Y, Chen T, Chen Q, Deng Y. Metabolite Profiling of External and Internal Petals in Three Different Colors of Tea Flowers (Camellia sinensis) Using Widely Targeted Metabolomics. Metabolites. 2023; 13(7):784. https://doi.org/10.3390/metabo13070784
Chicago/Turabian StyleZhang, Tao, Xue Ma, Yuanyuan Zhou, Hui Yang, Yuxin Wang, Taolin Chen, Qincao Chen, and Yanli Deng. 2023. "Metabolite Profiling of External and Internal Petals in Three Different Colors of Tea Flowers (Camellia sinensis) Using Widely Targeted Metabolomics" Metabolites 13, no. 7: 784. https://doi.org/10.3390/metabo13070784
APA StyleZhang, T., Ma, X., Zhou, Y., Yang, H., Wang, Y., Chen, T., Chen, Q., & Deng, Y. (2023). Metabolite Profiling of External and Internal Petals in Three Different Colors of Tea Flowers (Camellia sinensis) Using Widely Targeted Metabolomics. Metabolites, 13(7), 784. https://doi.org/10.3390/metabo13070784






