Lipid Metabolism, Carcass Characteristics and Longissimus dorsi Muscle Fatty Acid Composition of Tropical Crossbred Beef Cattle in Response to Desmanthus spp. Forage Backgrounding
Abstract
1. Introduction
2. Results
2.1. IMF, FMP and FA Composition
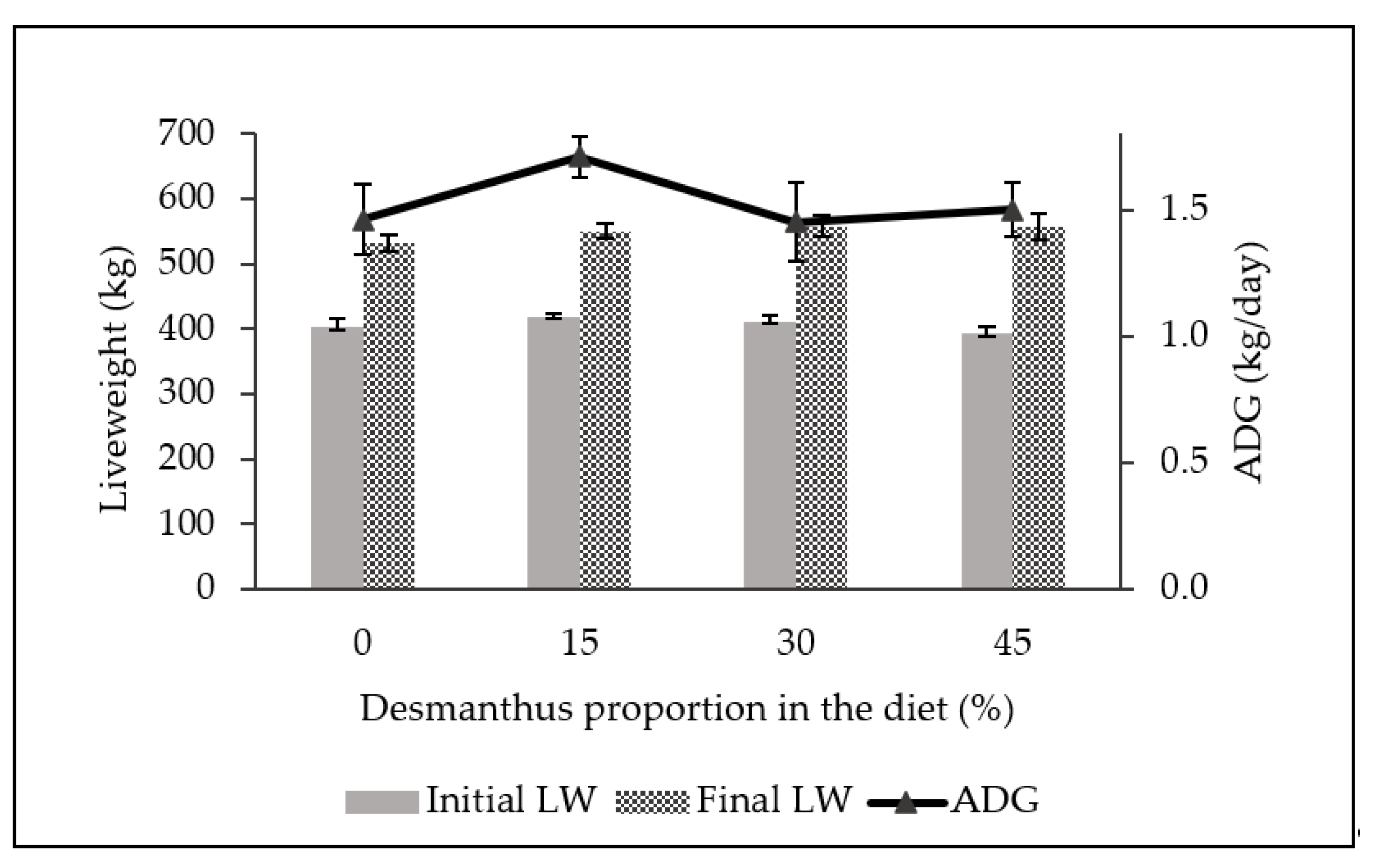
2.2. Feedlot Growth Performance and Carcass Characteristics
3. Discussion
3.1. Intramuscular Fat Content, Fat Melting Point and Fatty Acids Composition
3.2. Feedlot Growth Performance and Carcass Characteristics
4. Materials and Methods
4.1. Animals, Diets and Experimental Design
4.2. Loin Eye Muscle Biopsy and Carcass Sampling
4.3. IMF, FMP and FA Composition Analysis
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Organisation for Economic Co-Operation and Development (OECD). Meat Consumption (indicator). 2021. Available online: https://data.oecd.org/agroutput/meat-consumption.htm (accessed on 1 November 2021).
- FAO. Meat Market Review: Overview of Global Meat Market Developments in 2020; FAO: Rome, Italy, 2021; Available online: https://www.fao.org/3/cb3700en/cb3700en.pdf (accessed on 1 November 2021).
- Cabrera, M.C.; Saadoun, A. An overview of the nutritional value of beef and lamb meat from South America. Meat Sci. 2014, 98, 435–444. [Google Scholar] [CrossRef]
- Troy, D.J.; Tiwari, B.K.; Joo, S. Health implications of beef intramuscular fat consumption. Korean J. Food Sci. Anim. Resour. 2016, 36, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 140, e596–e646. [Google Scholar] [CrossRef] [PubMed]
- Bouvard, V.; Loomis, D.; Guyton, K.Z.; Grosse, Y.; El Ghissassi, F.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; Straif, K.; Stewart, B.W.; et al. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 2015, 16, 1599–1600. [Google Scholar] [CrossRef]
- Johnston, B.C.; Zeraatkar, D.; Han, M.A.; Vernooij, R.W.M.; Valli, C.; El Dib, R.; Marshall, C.; Stover, P.J.; Fairweather-Taitt, S.; Wójcik, G.; et al. Unprocessed red meat and processed meat consumption: Dietary guideline recommendations from the Nutritional Recommendations (NutriRECS) Consortium. Ann. Intern. Med. 2019, 171, 756–764. [Google Scholar] [CrossRef] [PubMed]
- Woollett, L.A.; Spady, D.K.; Dietschy, J.M. Saturated and unsaturated fatty acids independently regulate low density lipoprotein receptor activity and production rate. J. Lipid Res. 1992, 33, 77–88. [Google Scholar] [CrossRef]
- Krauss, R.M.; Kris-Etherton, P.M. Public health guidelines should recommend reducing saturated fat consumption as much as possible: Debate consensus. Am. J. Clin. Nutr. 2020, 112, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Van Vliet, S.; Provenza, F.D.; Kronberg, S.L. Health-promoting phytonutrients are higher in grass-fed meat and milk. Front. Sustain. Food Syst. 2021, 4, 555426. [Google Scholar] [CrossRef]
- Schulze, M.B.; Minihane, A.M.; Saleh, R.N.M.; Risérus, U. Intake and metabolism of omega-3 and omega-6 polyunsaturated fatty acids: Nutritional implications for cardiometabolic diseases. Lancet Diabetes Endocrinol. 2020, 8, 915–930. [Google Scholar] [CrossRef]
- Simopoulos, A.P. An increase in the Omega-6/Omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.D.; Richardson, R.I.; Nute, G.R.; Fisher, A.V.; Campo, M.M.; Kasapidou, E.; Sheard, P.R.; Enser, M. Effects of fatty acids on meat quality: A review. Meat Sci. 2003, 66, 21–32. [Google Scholar] [CrossRef]
- Hwang, Y.H.; Bakhsh, A.; Ismail, I.; Lee, J.G.; Joo, S.T. Effects of intensive alfalfa feeding on meat quality and fatty acid profile of Korean native black goats. Korean J. Food Sci. Anim. Resour. 2018, 38, 1092–1100. [Google Scholar] [CrossRef]
- Flakemore, A.R.; Malau-Aduli, B.S.; Nichols, P.D.; Malau-Aduli, A.E.O. Degummed crude canola oil, sire breed and gender effects on intramuscular long-chain omega-3 fatty acid properties of raw and cooked lamb meat. J. Anim. Sci. Technol. 2017, 59, 17. [Google Scholar] [CrossRef] [PubMed]
- Van Le, H.; Nguyen, D.V.; Vu Nguyen, Q.; Malau-Aduli, B.S.; Nichols, P.D.; Malau-Aduli, A.E.O. Fatty acid profiles of muscle, liver, heart and kidney of Australian prime lambs fed different polyunsaturated fatty acids enriched pellets in a feedlot system. Sci. Rep. 2019, 9, 1238. [Google Scholar] [CrossRef]
- Girard, M.; Dohme-Meier, F.; Silacci, P.; Ampuero Kragten, S.; Kreuzer, M.; Bee, G. Forage legumes rich in condensed tannins may increase n-3 fatty acid levels and sensory quality of lamb meat. J. Sci. Food Agric. 2016, 96, 1923–1933. [Google Scholar] [CrossRef]
- Neves, D.S.B.; Rodrigues Silva, R.; da Silva, F.F.; Santos, L.V.; Filho, G.A.; de Souza, S.O.; da Santos, M.C.; Rocha, W.J.; da Silva, A.P.G.; de Melo Lisboa, M.; et al. Increasing levels of supplementation for crossbred steers on pasture during the dry period of the year. Trop. Anim. Health Prod. 2018, 50, 1411–1416. [Google Scholar] [CrossRef]
- Scollan, N.; Hocquette, J.F.; Nuernberg, K.; Dannenberger, D.; Richardson, I.; Moloney, A. Innovations in beef production systems that enhance the nutritional and health value of beef lipids and their relationship with meat quality. Meat Sci. 2006, 74, 17–33. [Google Scholar] [CrossRef] [PubMed]
- Scollan, N.D.; Dannenberger, D.; Nuernberg, K.; Richardson, I.; MacKintosh, S.; Hocquette, J.F.; Moloney, A.P. Enhancing the nutritional and health value of beef lipids and their relationship with meat quality. Meat Sci. 2014, 97, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Kronberg, S.L.; Scholljegerdes, E.J.; Barceló-Coblijn, G.; Murphy, E.J. Flaxseed treatments to reduce biohydrogenation of α-linolenic acid by rumen microbes in cattle. Lipids 2007, 42, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Alves, S.P.; Francisco, A.; Costa, M.; Santos-Silva, J.; Bessa, R.J.B. Biohydrogenation patterns in digestive contents and plasma of lambs fed increasing levels of a tanniferous bush (Cistus ladanifer L.) and vegetable oils. Anim. Feed Sci. Technol. 2017, 225, 157–172. [Google Scholar] [CrossRef]
- Liu, C.; Xu, C.; Qu, Y.; Guo, P.; Ma, Y.; Wang, B.; Zhang, H.; Luo, H. Effect of alfalfa (Medicago sativa L.) saponins on meat color and myoglobin reduction status in the longissimus thoracis muscle of growing lambs. Anim. Sci. J. 2021, 92, e13556. [Google Scholar] [CrossRef] [PubMed]
- Schlink, A.C.; Burt, R.L. Assessment of the chemical composition of selected tropical legume seeds as animal feed. Trop. Agric. 1993, 70, 169–173. [Google Scholar]
- Suybeng, B.; Charmley, E.; Gardiner, C.P.; Malau-Aduli, B.S.; Malau-Aduli, A.E.O. Methane emissions and the use of desmanthus in beef cattle production in Northern Australia. Animals 2019, 9, 542. [Google Scholar] [CrossRef]
- Suybeng, B.; Charmley, E.; Gardiner, C.P.; Malau-Aduli, B.S.; Malau-Aduli, A.E.O. Supplementing Northern Australian beef cattle with desmanthus tropical legume reduces in-vivo methane emissions. Animals 2020, 10, 2097. [Google Scholar] [CrossRef] [PubMed]
- Department of Agriculture and Fisheries (DAF). The Queensland Beef Supply Chain; DAF: Brisbane, Australia, 2018.
- Blanco, M.; Casasús, I.; Ripoll, G.; Panea, B.; Albertí, P.; Joy, M. Lucerne grazing compared with concentrate-feeding slightly modifies carcase and meat quality of young bulls. Meat Sci. 2010, 84, 545–552. [Google Scholar] [CrossRef]
- Kurve, V.P.; Joseph, P.; Williams, J.B.; Kim, T.J.; Boland, H.; Smith, T.; Schilling, M.W. The effect of feeding native warm season grasses in the stocker phase on the carcass quality, meat quality, and sensory attributes of beef loin steaks from grain-finished steers. Meat Sci. 2016, 112, 31–38. [Google Scholar] [CrossRef]
- Woods, V.B.; Fearon, A.M. Dietary sources of unsaturated fatty acids for animals and their transfer into meat, milk and eggs: A review. Livest. Sci. 2009, 126, 1–20. [Google Scholar] [CrossRef]
- Yang, A.; Larsen, T.W.; Smith, S.B.; Tume, R.K. Δ9 Desaturase activity in bovine subcutaneous adipose tissue of different fatty acid composition. Lipids 1999, 34, 971–978. [Google Scholar] [CrossRef]
- Dierking, R.M.; Kallenbach, R.L.; Grün, I.U. Effect of forage species on fatty acid content and performance of pasture-finished steers. Meat Sci. 2010, 85, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, P.C.; Dawson, L.E.R.; Lively, F.O.; Steen, R.W.J.; Fearon, A.M.; Moss, B.W.; Kilpatrick, D.J. Effects of offering lupins/triticale and vetch/barley silages alone or in combination with grass silage on animal performance, meat quality and the fatty acid composition of lean meat from beef cattle. J. Agric. Sci. 2018, 156, 1005–1016. [Google Scholar] [CrossRef]
- Duckett, S.K.; Neel, J.P.S.; Lewis, R.M.; Fontenot, J.P.; Clapham, W.M. Effects of forage species or concentrate finishing on animal performance, carcass and meat quality. J. Anim. Sci. 2013, 91, 1454–1467. [Google Scholar] [CrossRef] [PubMed]
- Chail, A.; Legako, J.F.; Pitcher, L.R.; Griggs, T.C.; Ward, R.E.; Martini, S.; MacAdam, J.W. Legume finishing provides beef with positive human dietary fatty acid ratios and consumer preference comparable with grain-finished beef. J. Anim. Sci. 2016, 94, 2184–2197. [Google Scholar] [CrossRef] [PubMed]
- Freitas, A.K.d.; Lobato, J.F.P.; Cardoso, L.L.; Tarouco, J.U.; Vieira, R.M.; Dillenburg, D.R.; Castro, I. Nutritional composition of the meat of Hereford and Braford steers finished on pastures or in a feedlot in southern Brazil. Meat Sci. 2014, 96, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.H.; Joo, S.T. Fatty acid profiles, meat quality, and sensory palatability of grain-fed and grass-fed beef from Hanwoo, American, and Australian crossbred cattle. Korean J. Food Sci. Anim. Resour. 2017, 37, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.D.; Enser, M.; Fisher, A.V.; Nute, G.R.; Richardson, R.I.; Sheard, P.R. Manipulating meat quality and composition. Proc. Nutr. Soc. 1999, 58, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.B. Marbling and its nutritional impact on risk factors for cardiovascular disease. Korean J. Food Sci. Anim. Resour. 2016, 36, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Malau-Aduli, A.E.O.; Edriss, M.A.; Siebert, B.D.; Bottema, C.D.K.; Pitchford, W.S. Breed differences and genetic parameters for melting point, marbling score and fatty acid composition of lot-fed cattle. J. Anim. Physiol. Anim. Nutr. 2000, 83, 95–105. [Google Scholar] [CrossRef]
- Pitchford, W.S.; Deland, M.P.B.; Siebert, B.D.; Malau-Aduli, A.E.O.; Bottema, C.D.K. Genetic variation in fatness and fatty acid composition of crossbred cattle. J. Anim. Sci. 2002, 80, 2825–2832. [Google Scholar] [CrossRef]
- Turk, S.N.; Smith, S.B. Carcass fatty acid mapping. Meat Sci. 2009, 81, 658–663. [Google Scholar] [CrossRef]
- May, S.G.; Sturdivant, C.A.; Lunt, D.K.; Miller, R.K.; Smith, S.B. Comparison of sensory characteristics and fatty acid composition between Wagyu crossbred and Angus steers. Meat Sci. 1993, 35, 289–298. [Google Scholar] [CrossRef]
- Perry, D.; Nicholls, P.J.; Thompson, J.M. The effect of sirebreed on the melting point and fatty acid composition of subcutaneous fat in steers. J. Anim. Sci. 1998, 76, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Alfaia, C.P.M.; Alves, S.P.; Martins, S.I.V.; Costa, A.S.H.; Fontes, C.M.G.A.; Lemos, J.P.C.; Bessa, R.J.B.; Prates, J.A.M. Effect of the feeding system on intramuscular fatty acids and conjugated linoleic acid isomers of beef cattle, with emphasis on their nutritional value and discriminatory ability. Food Chem. 2009, 114, 939–946. [Google Scholar] [CrossRef]
- Lourenço, M.; Van Ranst, G.; Vlaeminck, B.; De Smet, S.; Fievez, V. Influence of different dietary forages on the fatty acid composition of rumen digesta as well as ruminant meat and milk. Anim. Feed Sci. Technol. 2008, 145, 418–437. [Google Scholar] [CrossRef]
- Toral, P.G.; Monahan, F.J.; Hervas, G.; Frutos, P.; Moloney, A.P. Review: Modulating ruminal lipid metabolism to improve the fatty acid composition of meat and milk. challenges and opportunities. Animal 2018, 12, s272–s281. [Google Scholar] [CrossRef] [PubMed]
- Khiaosa-Ard, R.; Bryner, S.F.; Scheeder, M.R.L.; Wettstein, H.-R.; Leiber, F.; Kreuzer, M.; Soliva, C.R. Evidence for the inhibition of the terminal step of ruminal α-linolenic acid biohydrogenation by condensed tannins. J. Dairy Sci. 2009, 92, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Campidonico, L.; Toral, P.G.; Priolo, A.; Luciano, G.; Valenti, B.; Hervás, G.; Frutos, P.; Copani, G.; Ginane, C.; Niderkorn, V. Fatty acid composition of ruminal digesta and longissimus muscle from lambs fed silage mixtures including red clover, sainfoin, and timothy. J. Anim. Sci. 2016, 94, 1550–1560. [Google Scholar] [CrossRef] [PubMed]
- Tava, A.; Avato, P. Chemical and biological activity of triterpene saponins from medicago species. Nat. Prod. Commun. 2006, 1, 1159–1180. [Google Scholar] [CrossRef]
- Dannenberger, D.; Nuernberg, K.; Nuernberg, G.; Scollan, N.; Steinhart, H.; Ender, K. Effect of pasture vs. concentrate diet on CLA isomer distribution in different tissue lipids of beef cattle. Lipids 2005, 40, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, S.; Buettner, A.; Ender, K.; Nürnberg, G.; Papstein, H.J.; Schieberle, P.; Nürnberg, K. Influence of keeping system on the fatty acid composition in the longissimus muscle of bulls and odorants formed after pressure-cooking. Eur. Food Res. Technol. 2002, 214, 112–118. [Google Scholar] [CrossRef]
- Steen, R.W.J.; Lavery, N.P.; Kilpatrick, D.J.; Porter, M.G. Effects of pasture and high-concentrate diets on the performance of beef cattle, carcass composition at equal growth rates, and the fatty acid composition of beef. N. Z. J. Agric. Res. 2003, 46, 69–81. [Google Scholar] [CrossRef][Green Version]
- Aboujaoude, C.; Pereira, A.S.C.; Feitosa, F.L.B.; Antunes De Lemos, M.V.; Chiaia, H.L.J.; Berton, M.P.; Peripolli, E.; Silva, R.M.D.O.; Ferrinho, A.M.; Mueller, L.F.; et al. Genetic parameters for fatty acids in intramuscular fat from feedlot-finished Nelore carcasses. Anim. Prod. Sci. 2018, 58, 234–243. [Google Scholar] [CrossRef]
- Leal-Gutiérrez, J.D.; Mateescu, R.G. Genetic basis of improving the palatability of beef cattle: Current insights. Food Biotechnol. 2019, 33, 193–216. [Google Scholar] [CrossRef]
- Joseph, S.J.; Robbins, K.R.; Pavan, E.; Pratt, S.L.; Duckett, S.K.; Rekaya, R. Effect of diet supplementation on the expression of bovine genes associated with fatty acid synthesis and metabolism. Bioinform. Biol. Insights 2010, 4, 19–31. [Google Scholar] [CrossRef] [PubMed]
- De Smet, S.; Raes, K.; Demeyer, D. Meat fatty acid composition as affected by fatness and genetic factors: A review. Anim. Res. 2004, 53, 81–98. [Google Scholar] [CrossRef]
- French, P.; Stanton, C.; Lawless, F.; O’Riordan, E.G.; Monahan, F.J.; Caffrey, P.J.; Moloney, A.P. Fatty acid composition, including conjugated linoleic acid, of intramuscular fat from steers offered grazed grass, grass silage, or concentrate-based diets. J. Anim. Sci. 2000, 78, 2849–2855. [Google Scholar] [CrossRef]
- Aldai, N.; Dugan, M.E.R.; Kramer, J.K.G.; Martínez, A.; López-Campos, O.; Mantecón, A.R.; Osoro, K. Length of concentrate finishing affects the fatty acid composition of grass-fed and genetically lean beef: An emphasis on trans-18:1 and conjugated linoleic acid profiles. Animal 2011, 5, 1643–1652. [Google Scholar] [CrossRef]
- McAfee, A.J.; McSorley, E.M.; Cuskelly, G.J.; Moss, B.W.; Wallace, J.M.W.; Bonham, M.P.; Fearon, A.M. Red meat consumption: An overview of the risks and benefits. Meat Sci. 2010, 84, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nuernberg, K.; Dannenberger, D.; Nuernberg, G.; Ender, K.; Voigt, J.; Scollan, N.D.; Wood, J.D.; Nute, G.R.; Richardson, R.I. Effect of a grass-based and a concentrate feeding system on meat quality characteristics and fatty acid composition of longissimus muscle in different cattle breeds. Livest. Prod. Sci. 2005, 94, 137–147. [Google Scholar] [CrossRef]
- Tansawat, R.; Maughan, C.A.J.; Ward, R.E.; Martini, S.; Cornforth, D.P. Chemical characterisation of pasture- and grain-fed beef related to meat quality and flavour attributes. Int. J. Food Sci. Technol. 2013, 48, 484–495. [Google Scholar] [CrossRef]
- Raes, K.; De Smet, S.; Demeyer, D. Effect of dietary fatty acids on incorporation of long chain polyunsaturated fatty acids and conjugated linoleic acid in lamb, beef and pork meat: A review. Anim. Feed Sci. Technol. 2004, 113, 199–221. [Google Scholar] [CrossRef]
- Enser, M.; Hallett, K.G.; Hewett, B.; Fursey, G.A.J.; Wood, J.D.; Harrington, G. Fatty acid content and composition of UK beef and lamb muscle in relation to production system and implications for human nutrition. Meat Sci. 1998, 49, 329–341. [Google Scholar] [CrossRef]
- Nuernberg, K.; Nuernberg, G.; Ender, K.; Lorenz, S.; Winkler, K.; Rickert, R.; Steinhart, H. N-3 fatty acids and conjugated linoleic acids of longissimus muscle in beef cattle. Eur. J. Lipid Sci. Technol. 2002, 104, 463–471. [Google Scholar] [CrossRef]
- Lee, J.H.; Min, B.R. Carcass characteristics and meat quality of Kiko crossbred male goats as influenced by feeding phytochemical tannin containing supplementations. Agric. Sci. 2021, 12, 445–463. [Google Scholar]
- Zheng, Y.; Wang, S.; Yan, P. The meat quality, muscle fiber characteristics and fatty acid profile in Jinjiang and F1 Simmental × Jinjiang yellow cattle. Asian-Australas. J. Anim. Sci. 2018, 31, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Cater, N.B.; Denke, M.A. Behenic acid is a cholesterol-raising saturated fatty acid in humans. Am. J. Clin. Nutr. 2001, 73, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.M.; Jenkins, T.C. Docosahexaenoic acid elevates trans-18:1 isomers but is not directly converted into trans-18:1 isomers in ruminal batch cultures. J. Dairy Sci. 2011, 94, 4676–4683. [Google Scholar] [CrossRef] [PubMed]
- Sampath, H.; Ntambi, J.M. The fate and intermediary metabolism of stearic acid. Lipids 2005, 40, 1187–1191. [Google Scholar] [CrossRef] [PubMed]
- Pewan, S.B.; Otto, J.R.; Kinobe, R.T.; Adegboye, O.A.; Malau-Aduli, A.E.O. Nutritional enhancement of health beneficial omega-3 long-chain polyunsaturated fatty acids in the muscle, liver, kidney, and heart of Tattykeel Australian white MARGRA lambs fed pellets fortified with omega-3 oil in a feedlot system. Biology (Basel) 2021, 10, 912. [Google Scholar] [CrossRef] [PubMed]
- Moreira, D.K.T.; Santos, P.S.; Gambero, A.; Macedo, G.A. Evaluation of structured lipids with behenic acid in the prevention of obesity. Food Res. Int. 2017, 95, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Maughan, B.; Provenza, F.D.; Tansawat, R.; Maughan, C.; Martini, S.; Ward, R.; Clemensen, A.; Song, X.; Cornforth, D.; Villalba, J.J. Importance of grass-legume choices on cattle grazing behavior, performance, and meat characteristics. J. Anim. Sci. 2014, 92, 2309–2324. [Google Scholar] [CrossRef] [PubMed]
- Blanco, M.; Joy, M.; Panea, B.; Albert, P.; Ripoll, G.; Carrasco, S.; Revilla, R.; Casas, I. Effects of the forage content of the winter diet on the growth performance and carcass quality of steers finished on mountain pasture with a barley supplement. Anim. Prod. Sci. 2012, 52, 823–831. [Google Scholar] [CrossRef]
- Monteiro, A.C.G.; Navas, D.R.; Lemos, J.P.C. Effects of castration and time-on-feed on Mertolenga breed beef quality. Animal 2014, 8, 675–682. [Google Scholar] [CrossRef] [PubMed]
- De Brito, G.F.; McGrath, S.R.; Holman, B.W.B.; Friend, M.A.; Fowler, S.M.; van de Ven, R.J.; Hopkins, D.L. The effect of forage type on lamb carcass traits, meat quality and sensory traits. Meat Sci. 2016, 119, 95–101. [Google Scholar] [CrossRef]
- Ladeira, M.M.; Schoonmaker, J.P.; Swanson, K.C.; Duckett, S.K.; Gionbelli, M.P.; Rodrigues, L.M.; Teixeira, P.D. Review: Nutrigenomics of marbling and fatty acid profile in ruminant meat. Animal 2018, 12, s282–s294. [Google Scholar] [CrossRef] [PubMed]
- National Health and Medical Research Council. Australian Code of Practice for the Care and Use of Animals for Scientific Purposes, 8th ed.; National Health and Medical Research Council: Canberra, Australia, 2013; ISBN 186-496-5-975.
- Poppi, D.P.; Quigley, S.P.; Silva, T.A.C.C.; McLennan, S.R. Challenges of beef cattle production from tropical pastures. Rev. Bras. Zootec. 2018, 47, e20160419. [Google Scholar] [CrossRef]
- Agriculture and Resource Management Council of Australia and New Zealand. Model Code of Practice for the Welfare of Animals: Livestock at Slaughtering Establishments; CSIRO Publishing: Collingwood, Australia, 2001; ISBN 0-643-06911-9. [Google Scholar]
- Commonwealth Scientific and Industrial Research Organisation. Nutrient Requirements of Domesticated Ruminants; CSIRO Publishing: Collingwood, Australia, 2007; ISBN 978-064-309-2-624. [Google Scholar]
- Malau-Aduli, A.E.O.; Siebert, B.D.; Bottema, C.D.K.; Pitchford, W.S. Breed comparison of the fatty acid composition of muscle phospholipids in Jersey and Limousin cattle. J. Anim. Sci. 1998, 76, 766–773. [Google Scholar] [CrossRef][Green Version]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Flakemore, R.A.; Balogun, R.O.; McEvoy, P.D.; Malau-Aduli, B.S.; Nichols, P.D.; Malau-Aduli, A.E.O. Genetic variation in intramuscular fat of prime lambs supplemented with varying concentrations of degummed crude canola oil. Int. J. Nutr. Food Sci. 2014, 3, 203–209. [Google Scholar] [CrossRef]
- AOCS—American Oil Chemists’ Society. Slip Melting Point ISO Standard; AOCS Official Method Cc 3b-92; AOCS: Urbana, IL, USA, 2017. [Google Scholar]
- Pewan, S.B.; Otto, J.R.; Kinobe, R.T.; Adegboye, O.A.; Malau-Aduli, A.E.O. MARGRA lamb eating quality and human health-promoting omega-3 long-chain polyunsaturated fatty acid profiles of Tattykeel Australian white sheep: Linebreeding and gender effects. Antioxidants 2020, 9, 1118. [Google Scholar] [CrossRef] [PubMed]
- Malau-Aduli, A.E.O.; Holman, B.W.B.; Kashani, A.; Nichols, P.D. Sire breed and sex effects on the fatty acid composition and content of heart, kidney, liver, adipose and muscle tissues of purebred and first-cross prime lambs. Anim. Prod. Sci. 2016, 56, 2122. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.R.; Nichols, P.D.; Barnes, J.; Davies, N.W.; Peacock, E.J.; Carter, C.G. Regiospecificity profiles of storage and membrane lipids from the gill and muscle tissue of Atlantic salmon (Salmo salar L.) grown at elevated temperature. Lipids 2006, 41, 865–876. [Google Scholar] [CrossRef]
- Flakemore, A.R.; Malau-Aduli, B.S.; Nichols, P.D.; Malau-Aduli, A.E.O. Omega-3 fatty acids, nutrient retention values, and sensory meat eating quality in cooked and raw Australian lamb. Meat Sci. 2017, 123, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Clayton, E.H. Graham Centre Monograph No. 4: Long-Chain Omega-3 Polyunsaturated Fatty Acids in Ruminant Nutrition: Benefits to Animals and Humans; Nicholls, T.N.C., Ed.; NSW Department of Primary Industries: Wagga Wagga, Australia, 2014; ISBN 978-1-74256-678-8.
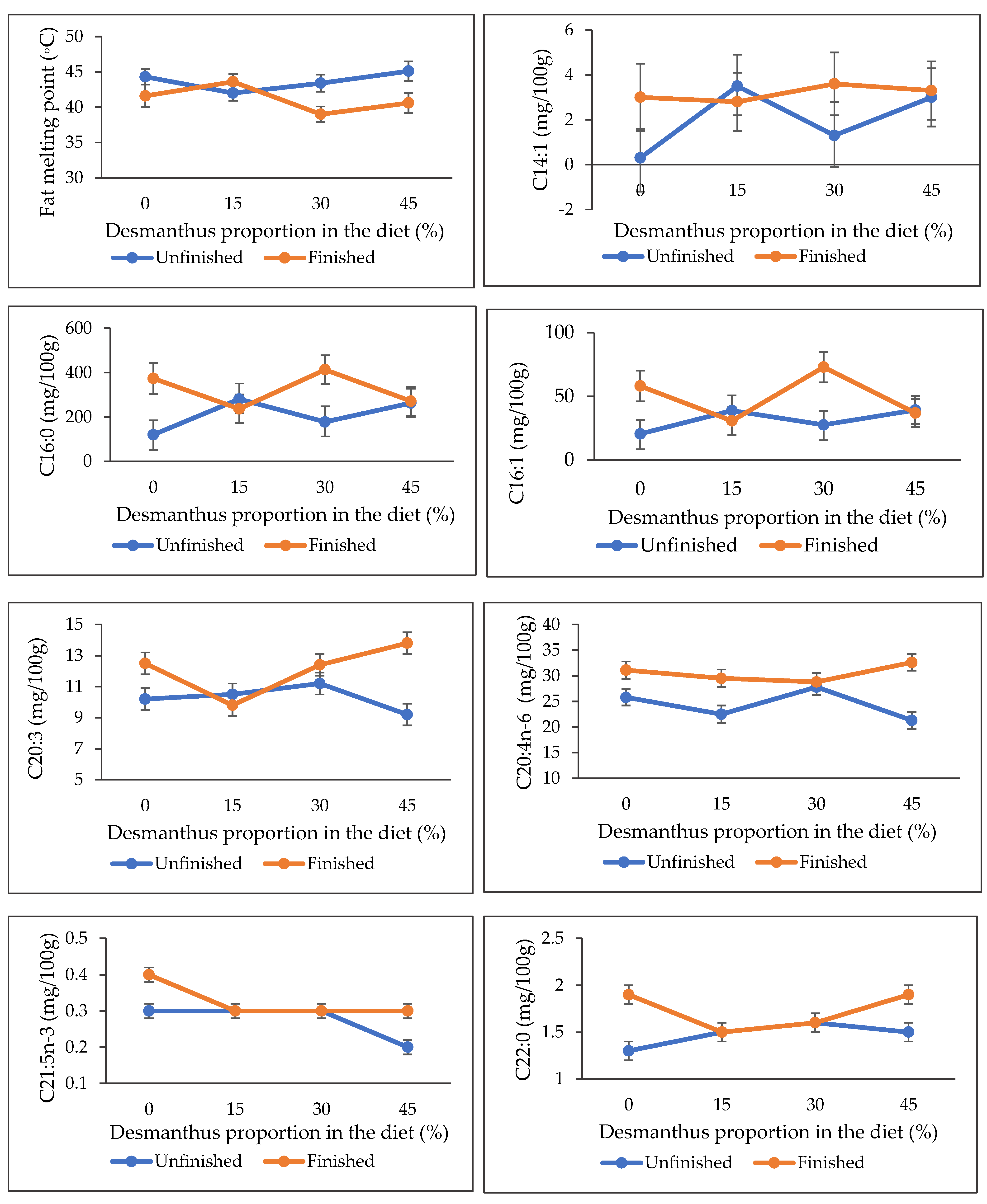
| Variable 1 | Desmanthus Proportion in the Diet (%) | SEM 2 | p-Value | |||
|---|---|---|---|---|---|---|
| 0 | 15 | 30 | 45 | |||
| IMF (%) | 2.6 | 2.3 | 2.1 | 2.3 | 0.11 | 0.50 |
| FMP (°C) | 43.1 | 43.5 | 43.1 | 44.5 | 0.71 | 0.94 |
| Fatty Acids (mg/100g) | ||||||
| C13:0 | 0.2 | 0.1 | 0.3 | 0.1 | 0.03 | 0.51 |
| C14:0 | 9.7 | 12.8 | 14.3 | 15.6 | 2.11 | 0.77 |
| C14:1 | 1.4 | 2.1 | 2.1 | 2.8 | 0.40 | 0.69 |
| C15:0 | 3.5 | 4.9 | 4.0 | 6.0 | 0.64 | 0.54 |
| C16:0 | 156.9 | 209.9 | 203.5 | 215.7 | 21.83 | 0.67 |
| C16:1 | 23.8 | 28.2 | 33.5 | 30.9 | 3.16 | 0.70 |
| C17:0 | 9.4 | 11.0 | 12.4 | 11.7 | 1.06 | 0.78 |
| C17:1 | 9.2 | 10.3 | 11.3 | 10.7 | 0.96 | 0.88 |
| C18:0 | 102.7 | 135.2 | 137.1 | 139.6 | 11.40 | 0.60 |
| C18:1 (Oleic) | 190.0 | 250.3 | 288.6 | 258.8 | 28.48 | 0.58 |
| CLA | 3.1 | 4.1 | 5.4 | 4.9 | 0.49 | 0.48 |
| C18:2n-6 (LA) | 44.7 | 52.8 | 48.1 | 54.8 | 1.47 | 0.25 |
| C18:3n-3 (ALA) | 15.3 | 17.1 | 15.8 | 16.9 | 0.45 | 0.47 |
| C18:3n-6 | 0.4 | 0.6 | 0.6 | 0.6 | 0.04 | 0.35 |
| C18:4n-3 | 2.4 | 2.6 | 1.9 | 2.3 | 0.11 | 0.25 |
| C19:1 | 1.4 | 1.6 | 2.1 | 1.9 | 0.21 | 0.64 |
| C20:0 | 0.8 | 1.2 | 1.3 | 1.3 | 0.13 | 0.48 |
| C20:1 | 1.6 | 2.2 | 2.3 | 2.5 | 0.22 | 0.38 |
| C20:2n-6 | 0.7 | 1.1 | 0.9 | 0.9 | 0.06 | 0.30 |
| C20:3 | 9.9 | 10.8 | 10.2 | 9.6 | 0.26 | 0.45 |
| C20:4n-3 | 3.1 | 3.1 | 2.9 | 2.9 | 0.10 | 0.78 |
| C20:4n-6 (ARA) | 26.3 | 26.7 | 26.6 | 25.0 | 0.85 | 0.90 |
| C20:5n-3 (EPA) | 9.7 | 9.9 | 9.5 | 8.6 | 0.31 | 0.43 |
| C21:0 | 0.2 | 0.3 | 0.3 | 0.3 | 0.01 | 0.98 |
| C21:5n-3 | 0.2 | 0.3 | 0.3 | 0.2 | 0.02 | 0.56 |
| C22:0 | 1.4 | 1.5 | 2.0 | 1.6 | 0.06 | 0.04 3 |
| C22:1 | 0.8 | 0.8 | 0.9 | 0.7 | 0.04 | 0.74 |
| C22:4n-6 | 1.8 | 2.0 | 2.0 | 1.9 | 0.05 | 0.58 |
| C22:5n-6 | 0.5 | 0.5 | 0.4 | 0.5 | 0.01 | 0.72 |
| C22:5n-3 (DPA) | 13.9 | 15.0 | 14.0 | 13.4 | 0.48 | 0.76 |
| C22:6n-3 (DHA) | 2.8 | 2.3 | 2.2 | 2.1 | 0.11 | 0.20 |
| C23:0 | 1.6 | 1.6 | 1.8 | 1.6 | 0.04 | 0.60 |
| C24:0 | 2.4 | 2.3 | 3.0 | 2.7 | 0.17 | 0.38 |
| C24:1 | 1.1 | 1.0 | 1.2 | 1.1 | 0.04 | 0.60 |
| EPA + DHA | 12.5 | 12.3 | 11.8 | 10.7 | 0.40 | 0.41 |
| EPA + DPA + DHA | 26.4 | 27.2 | 25.8 | 24.1 | 0.83 | 0.62 |
| Total FA | 644.5 | 816.5 | 859.3 | 852.5 | 71.66 | 0.59 |
| ∑SFA | 285.7 | 367.8 | 381.8 | 392.6 | 37.07 | 0.65 |
| ∑MUFA | 229.7 | 296.1 | 342.1 | 310.7 | 33.35 | 0.60 |
| ∑PUFA | 134.3 | 150.6 | 141.8 | 144.6 | 3.82 | 0.59 |
| ∑n-3 PUFA | 47.4 | 50.3 | 49.2 | 46.2 | 1.28 | 0.70 |
| ∑n-6 PUFA | 74.3 | 83.9 | 76.7 | 83.8 | 2.33 | 0.58 |
| PUFA/SFA | 0.6 | 0.5 | 0.4 | 0.5 | 0.03 | 0.59 |
| n-6/n-3 PUFA | 1.5 | 1.6 | 1.7 | 1.8 | 0.02 | 0.01 3 |
| Variable 1 | Unfinished | Feedlot Finished | p-Value 2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 15 | 30 | 45 | 0 | 15 | 30 | 45 | SEM | D | F | D*F | |
| IMF (%) | 3.5 | 2.6 | 2.4 | 2.2 | 4.5 | 4.5 | 4.5 | 4.5 | 0.18 | 0.33 | 0.01 | 0.29 |
| FMP (°C) | 44.3 | 42.0 | 43.4 | 45.1 | 41.6 | 43.6 | 39.0 | 40.6 | 0.48 | 0.49 | 0.01 | 0.04 |
| Fatty Acids (mg/100g) | ||||||||||||
| C13:0 | 0.3 | 0.2 | 0.5 | 0.3 | 0.0 | 0.0 | 0.0 | 0.0 | 0.04 | 0.27 | 0.01 | 0.27 |
| C14:0 | 3.9 | 18.9 | 8.9 | 18.5 | 16.6 | 13.4 | 36.3 | 17.5 | 2.63 | 0.42 | 0.07 | 0.06 |
| C14:1 | 0.3 | 3.5 | 1.3 | 3.0 | 3.0 | 2.8 | 3.6 | 3.3 | 0.55 | 0.74 | 0.03 | 0.04 |
| C15:0 | 2.2 | 5.2 | 4.3 | 3.9 | 3.2 | 3.6 | 7.7 | 3.8 | 0.55 | 0.30 | 0.46 | 0.25 |
| C16:0 | 119.8 | 280.6 | 177.5 | 262.9 | 373.9 | 236.5 | 413.1 | 271.3 | 26.22 | 0.92 | 0.01 | 0.03 |
| C16:1 | 20.5 | 38.8 | 27.6 | 39.2 | 58.1 | 30.7 | 72.9 | 36.9 | 4.34 | 0.62 | 0.02 | 0.04 |
| C17:0 | 7.2 | 13.2 | 11.3 | 14.2 | 12.2 | 10.4 | 21.5 | 12.2 | 1.17 | 0.32 | 0.20 | 0.09 |
| C17:1 | 7.2 | 11.9 | 9.9 | 12.7 | 20.4 | 11.4 | 23.4 | 13.9 | 1.35 | 0.59 | 0.01 | 0.09 |
| C18:0 | 86.8 | 167.7 | 140.4 | 165.6 | 169.1 | 155.1 | 214.9 | 170.5 | 11.38 | 0.54 | 0.06 | 0.22 |
| C18:1 (Oleic) | 151.4 | 331.0 | 244.5 | 316.9 | 581.6 | 356.4 | 649.0 | 407.1 | 39.88 | 0.77 | 0.01 | 0.08 |
| CLA | 2.5 | 4.4 | 5.5 | 5.5 | 6.4 | 5.5 | 8.9 | 6.4 | 0.60 | 0.35 | 0.04 | 0.68 |
| C18:2n-6 (LA) | 43.0 | 48.6 | 53.1 | 48.9 | 85.1 | 74.2 | 81.6 | 80.8 | 2.73 | 0.55 | 0.01 | 0.22 |
| C18:3n-3 (ALA) | 14.8 | 15.1 | 16.5 | 15.4 | 6.8 | 7.4 | 6.5 | 7.4 | 0.73 | 0.93 | 0.01 | 0.76 |
| C18:3n-6 | 0.2 | 0.7 | 0.4 | 0.6 | 1.0 | 0.9 | 1.1 | 0.9 | 0.06 | 0.64 | 0.01 | 0.06 |
| C18:4n-3 | 2.3 | 2.9 | 2.7 | 2.3 | 3.4 | 2.7 | 3.0 | 2.8 | 0.10 | 0.68 | 0.07 | 0.12 |
| C19:1 | 1.0 | 2.0 | 1.8 | 2.4 | 2.3 | 1.3 | 2.8 | 1.6 | 0.22 | 0.61 | 0.64 | 0.15 |
| C20:0 | 0.7 | 1.4 | 1.2 | 1.7 | 1.8 | 1.3 | 1.6 | 1.2 | 0.14 | 0.93 | 0.38 | 0.20 |
| C20:1 | 1.2 | 2.4 | 2.3 | 3.4 | 4.3 | 3.2 | 3.9 | 3.5 | 0.30 | 0.87 | 0.01 | 0.19 |
| C20:2n-6 | 0.6 | 1.2 | 1.0 | 1.1 | 1.5 | 1.0 | 1.4 | 1.1 | 0.08 | 0.94 | 0.08 | 0.05 |
| C20:3 | 10.2 | 10.5 | 11.2 | 9.2 | 12.5 | 9.8 | 12.4 | 13.8 | 0.32 | 0.14 | 0.01 | 0.01 |
| C20:4n-3 | 3.4 | 2.9 | 3.1 | 2.7 | 1.8 | 1.9 | 1.6 | 2.0 | 0.12 | 0.73 | 0.01 | 0.13 |
| C20:4n-6 (ARA) | 25.8 | 22.5 | 27.8 | 21.3 | 31.1 | 29.5 | 28.8 | 32.6 | 0.76 | 0.44 | 0.01 | 0.03 |
| C20:5n-3 (EPA) | 9.2 | 8.7 | 9.6 | 7.4 | 8.8 | 8.0 | 7.1 | 8.3 | 0.25 | 0.47 | 0.16 | 0.11 |
| C21:0 | 0.2 | 0.3 | 0.3 | 0.3 | 0.1 | 0.1 | 0.2 | 0.1 | 0.02 | 0.93 | 0.01 | 0.48 |
| C21:5n-3 | 0.3 | 0.3 | 0.3 | 0.2 | 0.4 | 0.3 | 0.3 | 0.3 | 0.01 | 0.17 | 0.06 | 0.01 |
| C22:0 | 1.3 | 1.5 | 1.6 | 1.5 | 1.9 | 1.5 | 1.6 | 1.9 | 0.05 | 0.55 | 0.01 | 0.02 |
| C22:1 | 0.7 | 0.8 | 0.9 | 0.7 | 0.9 | 0.9 | 0.7 | 0.7 | 0.04 | 0.59 | 0.90 | 0.11 |
| C22:4n-6 | 1.8 | 2.0 | 2.2 | 2.0 | 3.6 | 3.5 | 3.4 | 3.7 | 0.01 | 0.89 | 0.01 | 0.37 |
| C22:5n-6 | 0.5 | 0.5 | 0.5 | 0.4 | 0.7 | 0.6 | 0.5 | 0.6 | 0.13 | 0.50 | 0.01 | 0.17 |
| C22:5n-3 (DPA) | 13.6 | 13.7 | 15.4 | 12.8 | 16.6 | 15.9 | 14.2 | 14.9 | 0.37 | 0.60 | 0.04 | 0.20 |
| C22:6n-3 (DHA) | 2.5 | 2.0 | 2.2 | 1.8 | 2.7 | 2.1 | 2.2 | 2.0 | 0.09 | 0.01 3 | 0.38 | 0.97 |
| C23:0 | 1.6 | 1.7 | 2.0 | 1.8 | 1.4 | 1.3 | 1.2 | 1.4 | 0.05 | 0.81 | 0.01 | 0.06 |
| C24:0 | 1.6 | 1.7 | 1.9 | 1.8 | 2.0 | 1.6 | 1.7 | 1.8 | 0.05 | 0.68 | 0.89 | 0.24 |
| C24:1 | 1.0 | 1.1 | 1.2 | 1.2 | 1.1 | 0.9 | 0.9 | 0.9 | 0.04 | 0.92 | 0.07 | 0.41 |
| EPA+DHA | 11.7 | 10.7 | 11.8 | 9.1 | 11.3 | 10.1 | 9.5 | 10.3 | 0.30 | 0.19 | 0.34 | 0.18 |
| EPA+DPA+DHA | 25.2 | 24.4 | 27.2 | 21.9 | 27.9 | 26.0 | 23.6 | 25.1 | 0.61 | 0.37 | 0.41 | 0.18 |
| Total FA | 523.7 | 1005.1 | 787.5 | 997.7 | 1513.8 | 1006.5 | 1635.5 | 1128.8 | 93.38 | 0.84 | 0.01 | 0.07 |
| ∑SFA | 223.9 | 472.2 | 349.0 | 473.4 | 668.5 | 422.6 | 707.0 | 479.9 | 44.25 | 0.90 | 0.02 | 0.06 |
| ∑MUFA | 184.7 | 391.5 | 289.6 | 381.7 | 675.7 | 407.2 | 766.0 | 468.1 | 46.70 | 0.75 | 0.01 | 0.07 |
| ∑PUFA | 129.7 | 139.8 | 151.7 | 132.5 | 186.7 | 164.3 | 175.3 | 177.8 | 4.20 | 0.67 | 0.01 | 0.19 |
| ∑n-3 PUFA | 45.9 | 45.7 | 49.9 | 42.7 | 40.3 | 38.2 | 34.9 | 37.7 | 1.14 | 0.77 | 0.01 | 0.27 |
| ∑n-6 PUFA | 72.0 | 75.7 | 85.1 | 73.4 | 123.0 | 109.6 | 116.8 | 119.7 | 3.49 | 0.52 | 0.01 | 0.23 |
| PUFA/SFA | 0.6 | 0.4 | 0.5 | 0.4 | 0.4 | 0.4 | 0.3 | 0.4 | 0.03 | 0.59 | 0.04 | 0.06 |
| n-6/n-3 PUFA | 1.5 | 1.7 | 1.7 | 1.8 | 3.1 | 2.9 | 3.5 | 3.1 | 0.12 | 0.26 | 0.01 | 0.28 |
| Variable 1 | Unfinished | Finished | p-Value 2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 15 | 30 | 45 | 0 | 15 | 30 | 45 | SEM | D | F | D*F | |
| Slaughter weight (kg) | 463.8 | 457.7 | 443.5 | 447.5 | 532.8 | 550.5 | 558.0 | 557.3 | 8.11 | 0.96 | 0.01 | 0.21 |
| HCW (kg) | 225.3 | 219.0 | 216.7 | 220.8 | 267.2 | 275.0 | 274.2 | 274.9 | 4.42 | 0.98 | 0.01 | 0.63 |
| Dressing percentage (%) | 52.8 | 52.0 | 53.1 | 53.6 | 54.5 | 54.3 | 53.4 | 53.6 | 0.26 | 0.86 | 0.04 | 0.30 |
| P8 fat (mm) | 4.3 | 4.8 | 3.3 | 3.0 | 9.7 | 9.2 | 7.5 | 8.7 | 0.50 | 0.28 | 0.01 | 0.84 |
| Variable 1 | Desmanthus Proportion in the Diet (%) | |||
|---|---|---|---|---|
| 0 | 15 | 30 | 45 | |
| DM (%) | 87.3 | 68.4 | 56.9 | 48.6 |
| CP | 11.6 | 11.6 | 11.5 | 11.4 |
| NDF | 65.2 | 64.9 | 64.6 | 64.4 |
| ADF | 40.1 | 40.4 | 40.8 | 41.1 |
| Hemicellulose | 25.1 | 24.4 | 23.8 | 23.3 |
| ME (MJ/kg DM) 2 | 8.1 | 7.9 | 7.6 | 7.4 |
| DMI (kg/head) | 8.8 | 8.5 | 8.2 | 7.6 |
| Initial LW (kg) | 332.0 | 330.2 | 332.7 | 332.8 |
| Final LW (kg) | 434.1 | 437.8 | 427.5 | 420.1 |
| ADG (kg/day) | 0.63 | 0.66 | 0.57 | 0.53 |
| Variable 1 | Forage 2 | |||||
|---|---|---|---|---|---|---|
| Rhodes Grass | Lucerne | JCU2 | JCU4 | JCU7 | Desmanthus | |
| C14:0 | 0.6 | 0.6 | 0.3 | 0.3 | 0.3 | 0.3 |
| C15:0 | 0.9 | 1.6 | 0.4 | 0.3 | 0.5 | 0.4 |
| C16:0 | 31.3 | 26.4 | 20.5 | 20.2 | 20.1 | 20.3 |
| C16:1 | 2.8 | 3.9 | 3.2 | 2.7 | 2.7 | 2.9 |
| C17:0 | 2.5 | 1.7 | 1.1 | 1.1 | 1.2 | 1.1 |
| C17:1 | 0.3 | 0.2 | 0.2 | 0.1 | 0.2 | 0.2 |
| C18:0 | 4.0 | 5.5 | 5.1 | 5.1 | 4.9 | 5.0 |
| C18:1 | 3.3 | 4.0 | 5.3 | 5.4 | 6.4 | 5.7 |
| C18:2n-6 (LA) | 14.5 | 16.4 | 18.8 | 20.8 | 21.5 | 20.4 |
| C18:3n-3 (ALA) | 25.6 | 26.5 | 36.9 | 34.4 | 33.4 | 34.9 |
| C18:3n-6 | 1.9 | 1.8 | 0.9 | 0.7 | 0.8 | 0.8 |
| CLA | 0.3 | 0.6 | 0.1 | 0.4 | 0.2 | 0.2 |
| C19:1 | 0.0 | 0.2 | 0.4 | 0.2 | 0.1 | 0.2 |
| C20:0 | 1.5 | 1.4 | 1.1 | 1.1 | 0.8 | 1.0 |
| C20:1 | 0.6 | 0.7 | 0.4 | 0.6 | 0.4 | 0.5 |
| C20:2n-6 | 0.0 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| C20:3 | 0.6 | 0.6 | 0.2 | 0.2 | 0.3 | 0.2 |
| C20:4n-3 | 0.5 | 0.5 | 0.2 | 0.2 | 0.2 | 0.2 |
| C20:5n-3 (EPA) | 0.1 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 |
| C21:0 | 0.4 | 0.5 | 0.4 | 0.4 | 0.4 | 0.4 |
| C21:5n-3 | 0.2 | 0.3 | 0.1 | 0.1 | 0.1 | 0.1 |
| C22:0 | 2.4 | 1.8 | 1.5 | 1.8 | 1.9 | 1.7 |
| C22:1 | 0.3 | 0.9 | 0.1 | 0.4 | 0.2 | 0.2 |
| C22:4n-6 | 0.0 | 0.1 | 0.1 | 0.2 | 0.1 | 0.1 |
| C22:5n-3 (DPA) | 0.9 | 0.2 | 0.0 | 0.0 | 0.0 | 0.0 |
| C22:5n-6 | 0.0 | 0.2 | 0.0 | 0.1 | 0.0 | 0.0 |
| C22:6n-3 (DHA) | 0.5 | 0.3 | 0.2 | 0.3 | 0.1 | 0.2 |
| C23:0 | 0.7 | 0.7 | 0.4 | 0.4 | 0.6 | 0.5 |
| C24:0 | 2.6 | 1.8 | 1.9 | 2.0 | 2.6 | 2.2 |
| C24:1 | 0.3 | 0.2 | 0.2 | 0.1 | 0.1 | 0.1 |
| EPA+DHA | 0.6 | 0.4 | 0.2 | 0.3 | 0.1 | 0.2 |
| EPA+DPA+DHA | 1.5 | 0.6 | 0.2 | 0.3 | 0.1 | 0.2 |
| ∑SFA | 47.1 | 42.1 | 32.7 | 32.8 | 33.2 | 32.9 |
| ∑MUFA | 7.7 | 10.1 | 9.7 | 9.6 | 10.1 | 9.8 |
| ∑PUFA | 45.2 | 47.8 | 57.6 | 57.6 | 56.7 | 57.3 |
| ∑n-3 PUFA | 27.9 | 27.9 | 37.4 | 35.1 | 33.8 | 35.4 |
| ∑n-6 PUFA | 16.4 | 18.7 | 19.9 | 21.9 | 22.5 | 21.4 |
| PUFA/SFA | 1.0 | 1.1 | 1.8 | 1.8 | 1.7 | 1.8 |
| n-6/n-3 PUFA | 0.6 | 0.7 | 0.5 | 0.6 | 0.7 | 0.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mwangi, F.W.; Blignaut, D.J.C.; Charmley, E.; Gardiner, C.P.; Malau-Aduli, B.S.; Kinobe, R.T.; Malau-Aduli, A.E.O. Lipid Metabolism, Carcass Characteristics and Longissimus dorsi Muscle Fatty Acid Composition of Tropical Crossbred Beef Cattle in Response to Desmanthus spp. Forage Backgrounding. Metabolites 2021, 11, 804. https://doi.org/10.3390/metabo11120804
Mwangi FW, Blignaut DJC, Charmley E, Gardiner CP, Malau-Aduli BS, Kinobe RT, Malau-Aduli AEO. Lipid Metabolism, Carcass Characteristics and Longissimus dorsi Muscle Fatty Acid Composition of Tropical Crossbred Beef Cattle in Response to Desmanthus spp. Forage Backgrounding. Metabolites. 2021; 11(12):804. https://doi.org/10.3390/metabo11120804
Chicago/Turabian StyleMwangi, Felista W., David J. C. Blignaut, Edward Charmley, Christopher P. Gardiner, Bunmi S. Malau-Aduli, Robert T. Kinobe, and Aduli E. O. Malau-Aduli. 2021. "Lipid Metabolism, Carcass Characteristics and Longissimus dorsi Muscle Fatty Acid Composition of Tropical Crossbred Beef Cattle in Response to Desmanthus spp. Forage Backgrounding" Metabolites 11, no. 12: 804. https://doi.org/10.3390/metabo11120804
APA StyleMwangi, F. W., Blignaut, D. J. C., Charmley, E., Gardiner, C. P., Malau-Aduli, B. S., Kinobe, R. T., & Malau-Aduli, A. E. O. (2021). Lipid Metabolism, Carcass Characteristics and Longissimus dorsi Muscle Fatty Acid Composition of Tropical Crossbred Beef Cattle in Response to Desmanthus spp. Forage Backgrounding. Metabolites, 11(12), 804. https://doi.org/10.3390/metabo11120804






