Effect of Gender, Rearing, and Cooking on the Metabolomic Profile of Porcine Muscles
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Meat Sample
3.2. Sample Preparation
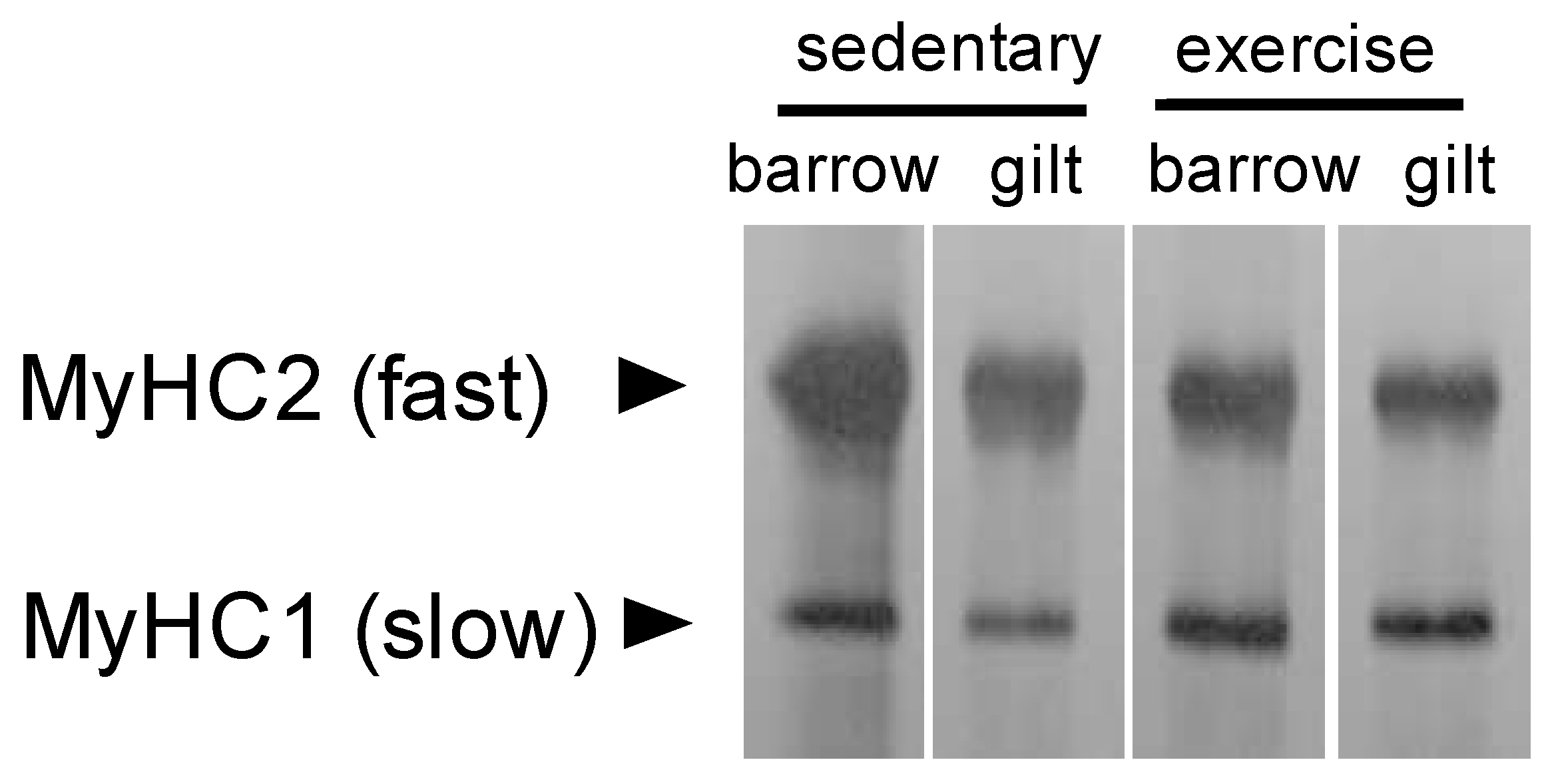
3.3. MyHC Isoform Content Determination
3.4. Sample Pretreatment for Metabolome Analysis
3.5. CE-TOFMS Analysis
3.6. Data Analysis
3.7. Statistics
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schiffman, S.S.; Hornack, K.; Reilly, D. Increased taste thresholds of amino acids with age. Am. J. Clin. Nutr. 1979, 32, 1622–1627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiffman, S.S.; Sennewald, K.; Gagnon, J. Comparison of taste qualities and thresholds of D-and L-amino acids. Physiol. Behav. 1981, 27, 51–59. [Google Scholar] [CrossRef]
- Nishimura, T.; Ra Rhue, M.; Okitani, A.; Kato, H. Components contributing to the improvement of meat taste during storage. Agric. Biol. Chem. 1988, 52, 2323–2330. [Google Scholar]
- Kato, H.; Rhue, M.R.; Nishimura, T. Role of free amino acids and peptides in food taste. In Flavor Chemistry: Trends and Developments, Proceedings of ACS Symposium Series; Teranishi, R., Buttery, R.G., Shalnidi, F., Eds.; American Chemical Society: Washington, DC, USA, 1989; Volume 388, pp. 158–174. [Google Scholar]
- Nelson, G.; Chandrashekar, J.; Hoon, M.A.; Feng, L.; Zhao, G.; Ryba, N.J.; Zuker, C.S. An amino-acid taste receptor. Nature 2002, 416, 199. [Google Scholar] [CrossRef]
- Suzuki, K.; Shioura, H.; Yokota, S.; Katoh, K.; Roh, S.G.; Iida, F.; Komatsu, T.; Syoji, N.; Sakuma, H.; Yamada, S. Search for an index for the taste of Japanese Black cattle beef by panel testing and chemical composition analysis. Anim. Sci. J. 2017, 88, 421–432. [Google Scholar] [CrossRef]
- Schwieterman, M.L.; Colquhoun, T.A.; Jaworski, E.A.; Bartoshuk, L.M.; Gilbert, J.L.; Tieman, D.M.; Odabasi, A.Z.; Moskowitz, H.R.; Folta, K.M.; Klee, H.J. Strawberry flavor: Diverse chemical compositions, a seasonal influence, and effects on sensory perception. PLoS ONE 2014, 9, e88446. [Google Scholar] [CrossRef] [Green Version]
- Jiang, T.; Bratcher, C.L. Differentiation of commercial ground beef products and correlation between metabolites and sensory attributes: A metabolomic approach. Food Res. Int. 2016, 90, 298–306. [Google Scholar] [CrossRef]
- Lee, S.M.; Kwon, G.Y.; Kim, K.-O.; Kim, Y.-S. Metabolomic approach for determination of key volatile compounds related to beef flavor in glutathione-Maillard reaction products. Anal. Chim. Acta 2011, 703, 204–211. [Google Scholar] [CrossRef]
- Carrillo, J.A.; He, Y.; Li, Y.; Liu, J.; Erdman, R.A.; Sonstegard, T.S.; Song, J. Integrated metabolomic and transcriptome analyses reveal finishing forage affects metabolic pathways related to beef quality and animal welfare. Sci. Rep. 2016, 6, 25948. [Google Scholar] [CrossRef] [PubMed]
- Argyri, A.A.; Mallouchos, A.; Panagou, E.Z.; Nychas, G.-J.E. The dynamics of the HS/SPME–GC/MS as a tool to assess the spoilage of minced beef stored under different packaging and temperature conditions. Int. J. Food Microbiol. 2015, 193, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Muroya, S.; Oe, M.; Ojima, K.; Watanabe, A. Metabolomic approach to key metabolites characterizing postmortem aged loin muscle of Japanese Black (Wagyu) cattle. Asian Australas. J. Anim. Sci. 2019, 32, 1172–1185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muroya, S.; Oe, M.; Nakajima, I.; Ojima, K.; Chikuni, K. CE-TOF MS-based metabolomic profiling revealed characteristic metabolic pathways in postmortem porcine fast and slow type muscles. Meat Sci. 2014, 98, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Welzenbach, J.; Neuhoff, C.; Looft, C.; Schellander, K.; Tholen, E.; Große-Brinkhaus, C. Different statistical approaches to investigate porcine muscle metabolome profiles to highlight new biomarkers for pork quality assessment. PLoS ONE 2016, 11, e0149758. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.M.; Kim, B.C. Muscle fiber characteristics, myofibrillar protein isoforms, and meat quality. Livest. Sci. 2009, 122, 105–118. [Google Scholar] [CrossRef]
- Mashima, D.; Oka, Y.; Gotoh, T.; Tomonaga, S.; Sawano, S.; Nakamura, M.; Tatsumi, R.; Mizunoya, W. Correlation between skeletal muscle fiber type and free amino acid levels in Japanese Black steers. Anim. Sci. J. 2019, 90, 604–609. [Google Scholar] [CrossRef] [Green Version]
- Valin, C.; Touraille, C.; Vigneron, P.; Ashmore, C. Prediction of lamb meat quality traits based on muscle biopsy fibre typing. Meat Sci. 1982, 6, 257–263. [Google Scholar] [CrossRef]
- Brooke, M.H.; Engel, W.K. The histographic analysis of human muscle biopsies with regard to fiber types: I. Adult male and female. Neurology 1969, 19, 221–233. [Google Scholar] [CrossRef]
- Simoneau, J.A.; Bouchard, C. Human variation in skeletal muscle fiber-type proportion and enzyme activities. Am. J. Physiol. 1989, 257, E567–E572. [Google Scholar] [CrossRef]
- Staron, R.S.; Hagerman, F.C.; Hikida, R.S.; Murray, T.F.; Hostler, D.P.; Crill, M.T.; Ragg, K.E.; Toma, K. Fiber type composition of the vastus lateralis muscle of young men and women. J. Histochem. Cytochem. 2000, 48, 623–629. [Google Scholar] [CrossRef] [Green Version]
- Holloszy, J.O.; Booth, F.W. Biochemical adaptations to endurance exercise in muscle. Annu. Rev. Physiol. 1976, 38, 273–291. [Google Scholar] [CrossRef]
- Allen, D.L.; Harrison, B.C.; Maass, A.; Bell, M.L.; Byrnes, W.C.; Leinwand, L.A. Cardiac and skeletal muscle adaptations to voluntary wheel running in the mouse. J. Appl. Physiol. 2001, 90, 1900–1908. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kriketos, A.; Pan, D.; Sutton, J.; Hoh, J.; Baur, L.; Cooney, G.; Jenkins, A.; Storlien, L. Relationships between muscle membrane lipids, fiber type, and enzyme activities in sedentary and exercised rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1995, 269, R1154–R1162. [Google Scholar] [CrossRef] [PubMed]
- Bigard, A.X.; Sanchez, H.; Birot, O.; Serrurier, B. Myosin heavy chain composition of skeletal muscles in young rats growing under hypobaric hypoxia conditions. J. Appl. Physiol. 2000, 88, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Mottram, D.S. Flavour formation in meat and meat products: A review. Food Chem. 1998, 62, 415–424. [Google Scholar] [CrossRef]
- Meinert, L.; Tikk, K.; Tikk, M.; Brockhoff, P.B.; Bredie, W.L.; Bjergegaard, C.; Aaslyng, M.D. Flavour development in pork. Influence of flavour precursor concentrations in longissimus dorsi from pigs with different raw meat qualities. Meat Sci. 2009, 81, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Tikk, K.; Haugen, J.-E.; Andersen, H.J.; Aaslyng, M.D. Monitoring of warmed-over flavour in pork using the electronic nose–correlation to sensory attributes and secondary lipid oxidation products. Meat Sci. 2008, 80, 1254–1263. [Google Scholar] [CrossRef]
- Van der Zwaard, S.; Brocherie, F.; Kom, B.L.; Millet, G.P.; Deldicque, L.; van der Laarse, W.J.; Girard, O.; Jaspers, R.T. Adaptations in muscle oxidative capacity, fiber size, and oxygen supply capacity after repeated-sprint training in hypoxia combined with chronic hypoxic exposure. J. Appl. Physiol. 2018, 124, 1403–1412. [Google Scholar] [CrossRef]
- Mizunoya, W.; Wakamatsu, J.; Tatsumi, R.; Ikeuchi, Y. Protocol for high-resolution separation of rodent myosin heavy chain isoforms in a mini-gel electrophoresis system. Anal. Biochem. 2008, 377, 111–113. [Google Scholar] [CrossRef]
- Soga, T.; Ohashi, Y.; Ueno, Y.; Naraoka, H.; Tomita, M.; Nishioka, T. Quantitative metabolome analysis using capillary electrophoresis mass spectrometry. J. Proteome Res. 2003, 2, 488–494. [Google Scholar] [CrossRef]
- Sugimoto, M.; Wong, D.T.; Hirayama, A.; Soga, T.; Tomita, M. Capillary electrophoresis mass spectrometry-based saliva metabolomics identified oral, breast and pancreatic cancer-specific profiles. Metabolomics 2010, 6, 78–95. [Google Scholar] [CrossRef] [Green Version]
- Junker, B.H.; Klukas, C.; Schreiber, F. VANTED: A system for advanced data analysis and visualization in the context of biological networks. BMC Bioinform. 2006, 7, 109. [Google Scholar]

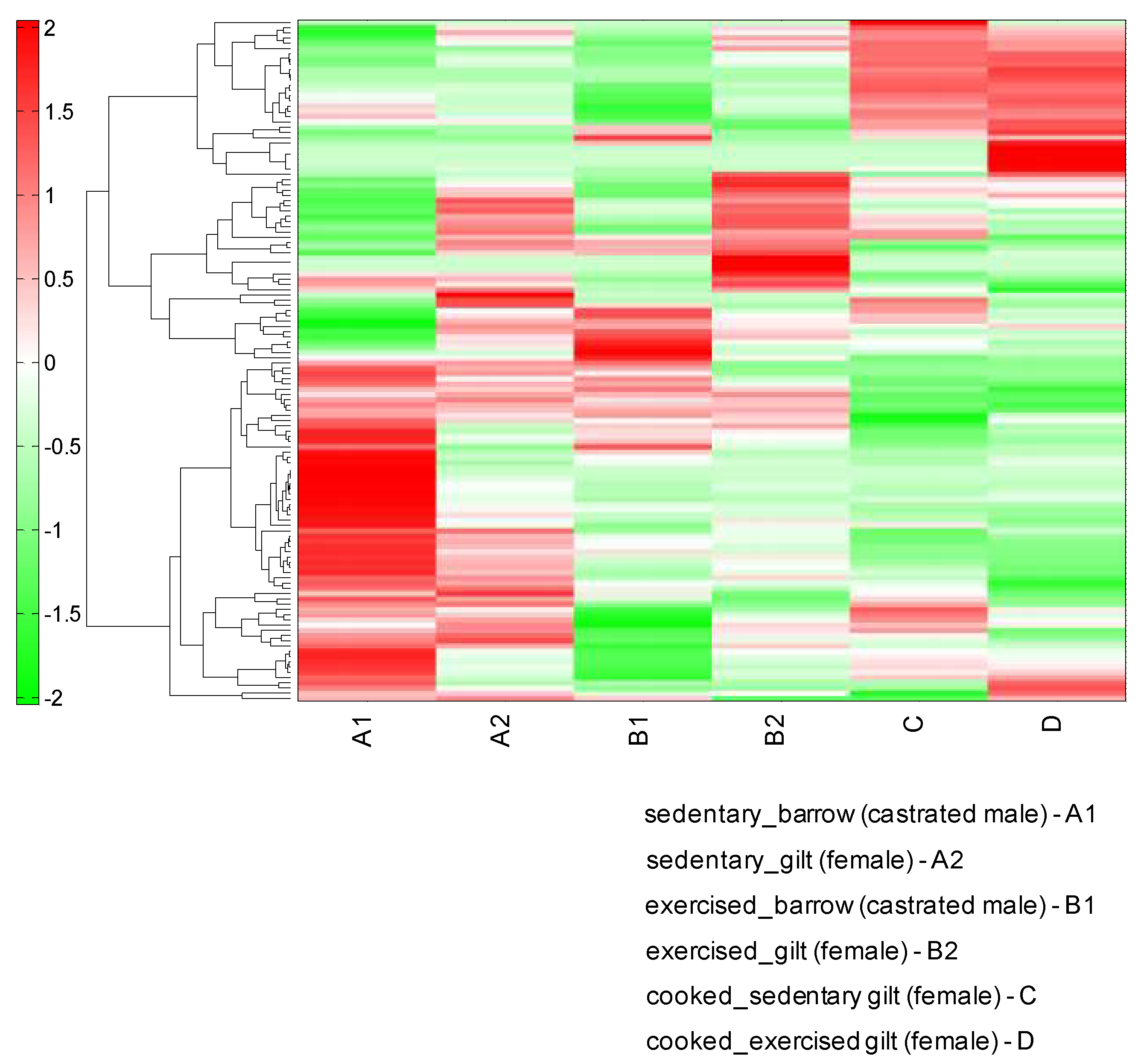
| Compound Name | Gilt/Barrow | Exercise/Sedentary | Cooked/Uncooked | |||
|---|---|---|---|---|---|---|
| Sedentary | Exercise | Barrow | Gilt | Sedentary | Exercise | |
| 1-Methylhistidine, 3-Methylhistidine | 0.59 | 1.09 | 0.80 | 1.46 | 0.64 | 0.64 |
| 2-(Creatinine-3-yl) propionic acid | N.D. | N.D. | N.D. | N.D. | N.D. | N/A |
| 2-Aminoisobutyric acid, 2-Aminobutyric acid | 0.78 | 1.10 | 0.58 | 0.81 | 0.65 | 0.73 |
| 2-Hydroxybutyric acid | 1.10 | zero | zero | zero | 0.79 | N.D. |
| 2-Hydroxyvaleric acid | 2.38 | 0.67 | 1.76 | 0.49 | 0.87 | 0.96 |
| 3-Hydroxybutyric acid | 1.28 | 1.90 | 0.92 | 1.37 | 1.00 | 0.73 |
| 3-Methyladenine | N/A | 1.25 | N/A | 1.05 | zero | zero |
| 4-Methylpyrazole | 1.18 | zero | 0.89 | zero | zero | N/A |
| 5’-Deoxy-5’-methylthioadenosine | N.D. | N.D. | N.D. | N.D. | N.D. | N/A |
| 5-Oxoproline | 0.71 | 1.10 | 0.42 | 0.65 | 0.67 | 2.39 |
| Adenosine | N.D. | N.D. | N/A | N.D. | N.D. | N/A |
| ADMA | 0.96 | 1.22 | 0.63 | 0.81 | zero | 1.67 |
| ADP | 0.93 | zero | 3.11 | zero | 3.96 | N/A |
| ADP-ribose | N/A | 0.66 | N/A | 0.84 | 0.40 | 0.53 |
| Ala | 0.73 | 0.84 | 0.73 | 0.84 | 0.70 | 0.82 |
| AMP | 0.93 | 0.10 | 14.49 | 1.63 | 7.84 | 5.90 |
| Anserine_divalent | 1.03 | 1.60 | 0.83 | 1.29 | 0.72 | 0.63 |
| Arg | 0.84 | 1.42 | 0.60 | 1.01 | 1.28 | 1.38 |
| Argininosuccinic acid | zero | N.D. | zero | N.D. | N.D. | N.D. |
| Asn | 0.72 | 1.39 | 0.51 | 0.99 | 1.09 | 1.15 |
| Asp | 0.45 | 0.93 | 0.58 | 1.20 | 1.05 | 1.07 |
| ATP | N.D. | N.D. | N.D. | N.D. | N/A | N/A |
| Betaine | 1.33 | 1.44 | 0.96 | 1.04 | 0.90 | 0.76 |
| Butyrylcarnitine | 0.36 | 1.49 | 0.19 | 0.79 | 0.67 | 0.74 |
| Carnitine | 0.94 | 1.24 | 0.67 | 0.88 | 0.72 | 0.63 |
| Carnosine | 1.33 | 1.26 | 1.01 | 0.96 | 0.96 | 0.84 |
| Choline | 0.43 | 0.87 | 0.35 | 0.70 | 0.40 | 0.48 |
| Citrulline | 0.71 | 1.26 | 0.43 | 0.76 | 0.74 | 0.65 |
| Creatine | 1.06 | 1.13 | 0.91 | 0.98 | 0.91 | 0.78 |
| Creatinine | 1.33 | 1.34 | 0.82 | 0.82 | 1.25 | 3.68 |
| Cys | 4.65 | 1.53 | 3.22 | 1.06 | 1.47 | 1.02 |
| Cysteine glutathione disulfide | 0.21 | N/A | zero | 0.56 | zero | 0.55 |
| Cystine | 0.06 | N.D. | zero | zero | zero | N.D. |
| Cytidine | 1.05 | 1.12 | 0.78 | 0.84 | 0.83 | 0.96 |
| Daminozide Ala-Ala | N/A | N/A | N.D. | 1.14 | 1.90 | 1.42 |
| Diethanolamine | 1.06 | zero | 1.24 | zero | zero | N.D. |
| Dyphylline | 2.69 | 0.83 | 2.88 | 0.89 | 0.99 | 0.90 |
| Ethanolamine | 0.91 | 1.01 | 0.87 | 0.97 | 0.56 | 0.50 |
| Ethanolamine phosphate | 0.80 | 0.84 | 1.04 | 1.09 | zero | 0.68 |
| Fructose 6-phosphate | 36.20 | 0.42 | 69.38 | 0.80 | 1.55 | 0.98 |
| GABA | 1.13 | 1.16 | 0.76 | 0.78 | zero | zero |
| Gln | 0.87 | 0.77 | 0.88 | 0.78 | 0.50 | 0.66 |
| Glu | 0.62 | 1.79 | 0.36 | 1.04 | 1.06 | 0.94 |
| Glu-Glu | 2.20 | 1.90 | 1.38 | 1.19 | 1.27 | 1.00 |
| Gluconic acid | 2.48 | 2.10 | 1.35 | 1.14 | 0.88 | 0.49 |
| Gluconolactone | N/A | 1.50 | N/A | 1.15 | 1.14 | zero |
| Glucose 1-phosphate | N/A | 0.51 | N/A | 0.85 | 1.15 | 0.72 |
| Glucose 6-phosphate | 43.39 | 0.39 | 83.94 | 0.76 | 0.81 | 0.54 |
| Glutathione (GSH) | 0.97 | 0.89 | 1.09 | 1.00 | 0.78 | 0.65 |
| Glutathione (GSSG)_divalent | 0.25 | 0.81 | 0.21 | 0.68 | 0.17 | 0.26 |
| Gly | 0.83 | 1.14 | 0.63 | 0.87 | 0.75 | 0.75 |
| Gly-Asp | zero | N.D. | zero | N.D. | N.D. | N/A |
| Gly-Gly | N.D. | N/A | N.D. | N/A | N.D. | zero |
| Gly-Leu | N.D. | N.D. | N.D. | N.D. | N/A | N.D. |
| Glyceric acid | N/A | 2.59 | N/A | 0.81 | 0.40 | 0.65 |
| Glycerol | 1.34 | 0.63 | 1.87 | 0.88 | 0.94 | 0.95 |
| Glycerol 3-phosphate | 0.97 | 1.24 | 0.57 | 0.73 | 0.51 | 0.93 |
| Glycerophosphocholine | 1.00 | 0.64 | 1.58 | 1.01 | 0.61 | 0.71 |
| GMP | 1.12 | 1.30 | 1.05 | 1.22 | 1.06 | 0.89 |
| Guanine | 0.45 | 1.03 | 0.35 | 0.80 | 0.74 | 1.13 |
| Guanosine | 0.91 | 0.84 | 0.76 | 0.70 | 0.93 | 1.04 |
| His | 0.73 | 1.15 | 0.62 | 0.99 | 1.06 | 0.89 |
| His-Glu | N.D. | N.D. | N.D. | N.D. | N.D. | N/A |
| Homocarnosine | 1.41 | 1.28 | 1.18 | 1.07 | 0.94 | 0.81 |
| Hydroxyproline | 0.79 | 0.96 | 0.62 | 0.76 | 0.48 | 0.70 |
| Hypotaurine | 0.43 | 0.70 | 0.66 | 1.06 | 0.46 | 0.60 |
| Hypoxanthine | 0.46 | 1.15 | 0.33 | 0.83 | 1.00 | 1.23 |
| Ile | 0.84 | 1.24 | 0.60 | 0.88 | 1.31 | 1.47 |
| IMP | 1.50 | 1.63 | 1.03 | 1.12 | 0.94 | 0.93 |
| Inosine | 1.24 | 1.02 | 1.03 | 0.85 | 0.97 | 0.98 |
| Isobutyric acid Butyric acid | 0.84 | N/A | zero | 1.46 | zero | zero |
| Isoglutamic acid | 0.71 | zero | 0.41 | zero | zero | N.D. |
| Lactic acid | 1.43 | 1.23 | 1.14 | 0.98 | 0.82 | 0.77 |
| Leu | 0.86 | 1.43 | 0.59 | 0.99 | 1.38 | 1.39 |
| Lys | 0.74 | 1.38 | 0.53 | 0.99 | 1.18 | 1.26 |
| Malic acid | 0.46 | 1.63 | 0.34 | 1.20 | 0.76 | 0.41 |
| Malonylcarnitine | N/A | N.D. | N.D. | zero | zero | N.D. |
| Met | 1.07 | 1.62 | 0.63 | 0.96 | 1.81 | 1.83 |
| Methionine sulfoxide | N.D. | N.D. | N.D. | N.D. | N/A | N/A |
| myo-Inositol 1-phosphate myo-Inositol 3-phosphate | N.D. | N/A | N.D. | N.D. | N/A | 1.00 |
| N-Acetyllysine | 1.81 | 1.57 | 1.20 | 1.04 | 1.95 | 1.88 |
| N-Acetylneuraminic acid | N.D. | N.D. | N.D. | N.D. | N/A | N/A |
| N-Acetylornithine | 1.43 | 2.00 | 0.86 | 1.20 | 2.11 | 1.82 |
| N-Methylalanine | 0.76 | 1.22 | 0.56 | 0.89 | 0.57 | 0.60 |
| N5-Ethylglutamine | 0.44 | 0.94 | 0.65 | 1.39 | 0.72 | 0.75 |
| N6,N6,N6-Trimethyllysine | 1.80 | 1.21 | 1.92 | 1.29 | 0.74 | 0.65 |
| N6-Methyllysine | 0.66 | 0.73 | 1.04 | 1.15 | 1.00 | 0.95 |
| NADH | 0.89 | 2.43 | 0.28 | 0.76 | 1.21 | 1.02 |
| Nicotinamide | 2.27 | 0.89 | 2.16 | 0.85 | 0.79 | 1.05 |
| O-Acetylcarnitine | 0.83 | 1.72 | 0.32 | 0.67 | 1.43 | 1.51 |
| O-Acetylhomoserine 2-Aminoadipic acid | 0.73 | 0.88 | 0.57 | 0.69 | 0.50 | 0.69 |
| Ornithine | 0.89 | 2.10 | 0.40 | 0.94 | 0.76 | 0.79 |
| Pantothenic acid | 0.71 | 1.20 | 0.57 | 0.96 | zero | 0.81 |
| Phe | 1.01 | 1.27 | 0.71 | 0.90 | 1.54 | 1.62 |
| Phosphorylcholine | 0.36 | 0.77 | 0.45 | 0.98 | 0.81 | 1.09 |
| Pro | 0.68 | 1.05 | 0.53 | 0.82 | 0.75 | 0.84 |
| Putrescine | 0.91 | 0.99 | 1.00 | 1.09 | zero | zero |
| Ribulose 5-phosphate | 1.40 | 1.34 | 1.07 | 1.03 | 1.43 | 1.45 |
| S-Adenosylhomocysteine | 1.07 | 0.84 | 1.26 | 1.00 | 1.16 | 1.35 |
| S-Adenosylmethionine | 1.45 | 1.12 | 1.38 | 1.06 | 0.59 | 0.67 |
| S-Methylcysteine | 0.90 | 1.43 | 0.79 | 1.25 | 0.87 | 0.57 |
| Saccharopine | N/A | 0.70 | N/A | 1.17 | 0.75 | 0.60 |
| Sedoheptulose 7-phosphate | zero | zero | 0.40 | N.D. | N.D. | N.D. |
| Ser | 0.68 | 1.34 | 0.51 | 1.01 | 1.14 | 1.09 |
| Spermidine | 0.82 | 1.01 | 0.69 | 0.85 | 0.66 | 0.86 |
| Spermine | 2.92 | 1.35 | 1.82 | 0.84 | 1.08 | 1.14 |
| Stachydrine | 1.52 | N/A | zero | 0.78 | 0.83 | 0.91 |
| Succinic acid | 0.67 | 0.62 | 0.89 | 0.82 | 0.54 | 0.76 |
| Taurine | 0.50 | 0.89 | 0.61 | 1.08 | 0.51 | 0.67 |
| Terephthalic acid | N.D. | N.D. | N.D. | N.D. | N.D. | N/A |
| Thiamine | 1.08 | 1.72 | 0.52 | 0.84 | 1.04 | 1.00 |
| Thiamine phosphate | 1.31 | 1.17 | 1.21 | 1.09 | zero | zero |
| Thr | 0.69 | 1.35 | 0.50 | 0.98 | 1.07 | 1.03 |
| Thr-Asp Ser-Glu | 1.10 | N.D. | zero | zero | 1.52 | N/A |
| Trigonelline | 1.08 | 1.58 | 0.59 | 0.86 | 0.98 | 0.70 |
| Trp | 0.92 | 1.24 | 0.74 | 0.99 | 1.32 | 1.36 |
| Tyr | 0.92 | 1.60 | 0.60 | 1.05 | 1.50 | 1.55 |
| UDP-glucose UDP-galactose | N.D. | zero | N.D. | N.D. | N.D. | N.D. |
| UDP-N-acetylgalactosamine UDP-N-acetylglucosamine | N.D. | N/A | N.D. | N/A | N.D. | zero |
| UMP | 1.77 | 1.97 | 1.11 | 1.24 | 0.99 | 0.74 |
| Urea | 0.99 | 0.91 | 0.73 | 0.67 | 0.76 | 0.76 |
| Uridine | 1.14 | 0.85 | 0.97 | 0.73 | 0.83 | 0.99 |
| Val | 0.74 | 1.24 | 0.55 | 0.92 | 1.09 | 1.14 |
| β-Ala | 0.89 | 0.96 | 0.76 | 0.82 | 0.82 | 0.64 |
| β-Ala-Lys | 1.98 | 1.44 | 1.41 | 1.02 | 0.66 | 0.74 |
| γ-Butyrobetaine | 1.06 | 1.44 | 0.61 | 0.83 | 0.81 | 0.76 |
| γ-Glu-Cys | N.D. | N/A | N.D. | N/A | N.D. | zero |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sawano, S.; Oza, K.; Murakami, T.; Nakamura, M.; Tatsumi, R.; Mizunoya, W. Effect of Gender, Rearing, and Cooking on the Metabolomic Profile of Porcine Muscles. Metabolites 2020, 10, 10. https://doi.org/10.3390/metabo10010010
Sawano S, Oza K, Murakami T, Nakamura M, Tatsumi R, Mizunoya W. Effect of Gender, Rearing, and Cooking on the Metabolomic Profile of Porcine Muscles. Metabolites. 2020; 10(1):10. https://doi.org/10.3390/metabo10010010
Chicago/Turabian StyleSawano, Shoko, Keishi Oza, Tetsuya Murakami, Mako Nakamura, Ryuichi Tatsumi, and Wataru Mizunoya. 2020. "Effect of Gender, Rearing, and Cooking on the Metabolomic Profile of Porcine Muscles" Metabolites 10, no. 1: 10. https://doi.org/10.3390/metabo10010010





