Diastereomers of Spheroidal Form and Commercially Available Taxifolin Samples
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Moisture Content
2.3. HPLC
2.4. Validation of Quantitative Analysis
2.5. Polarimetry
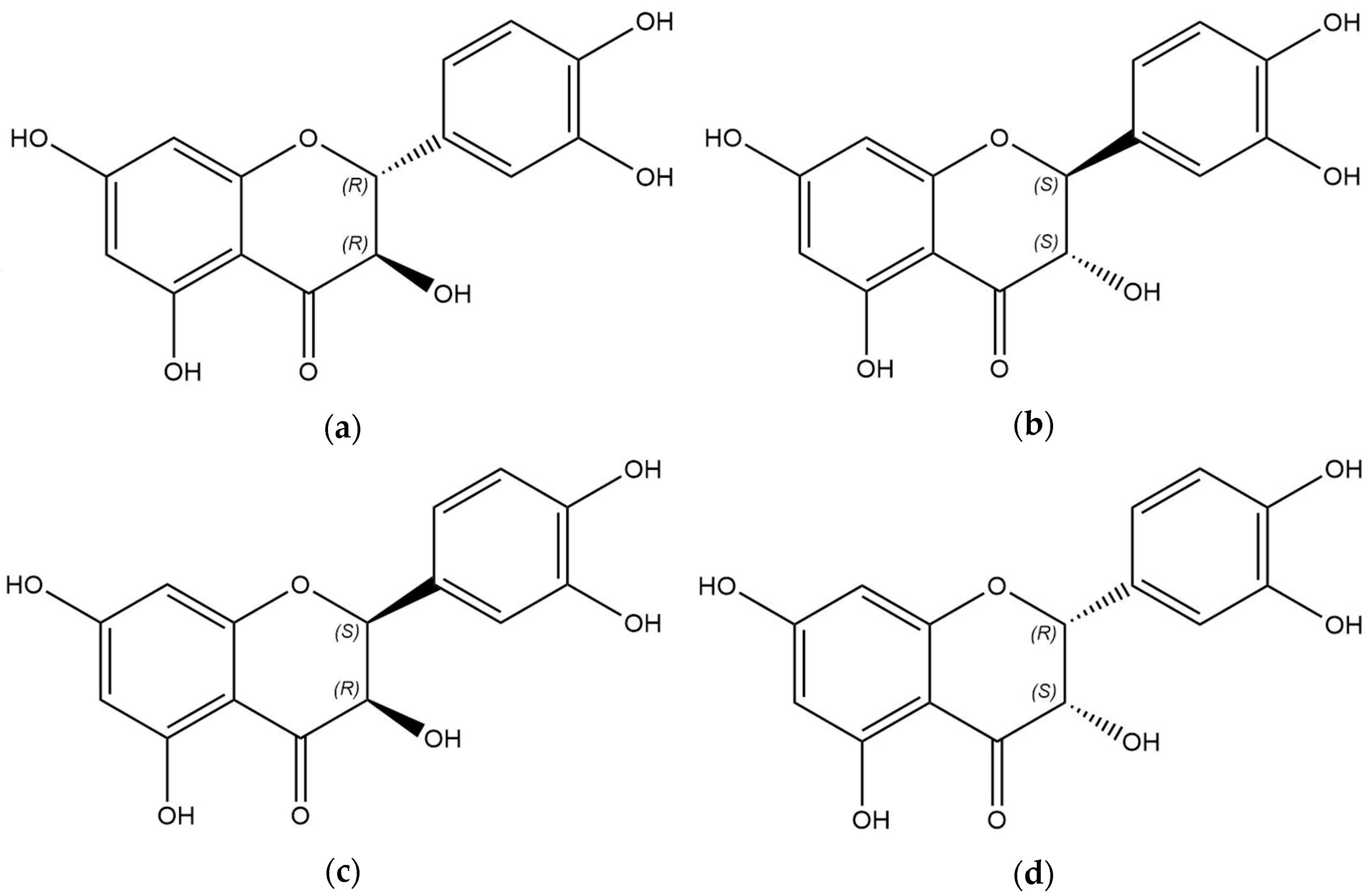
3. Results
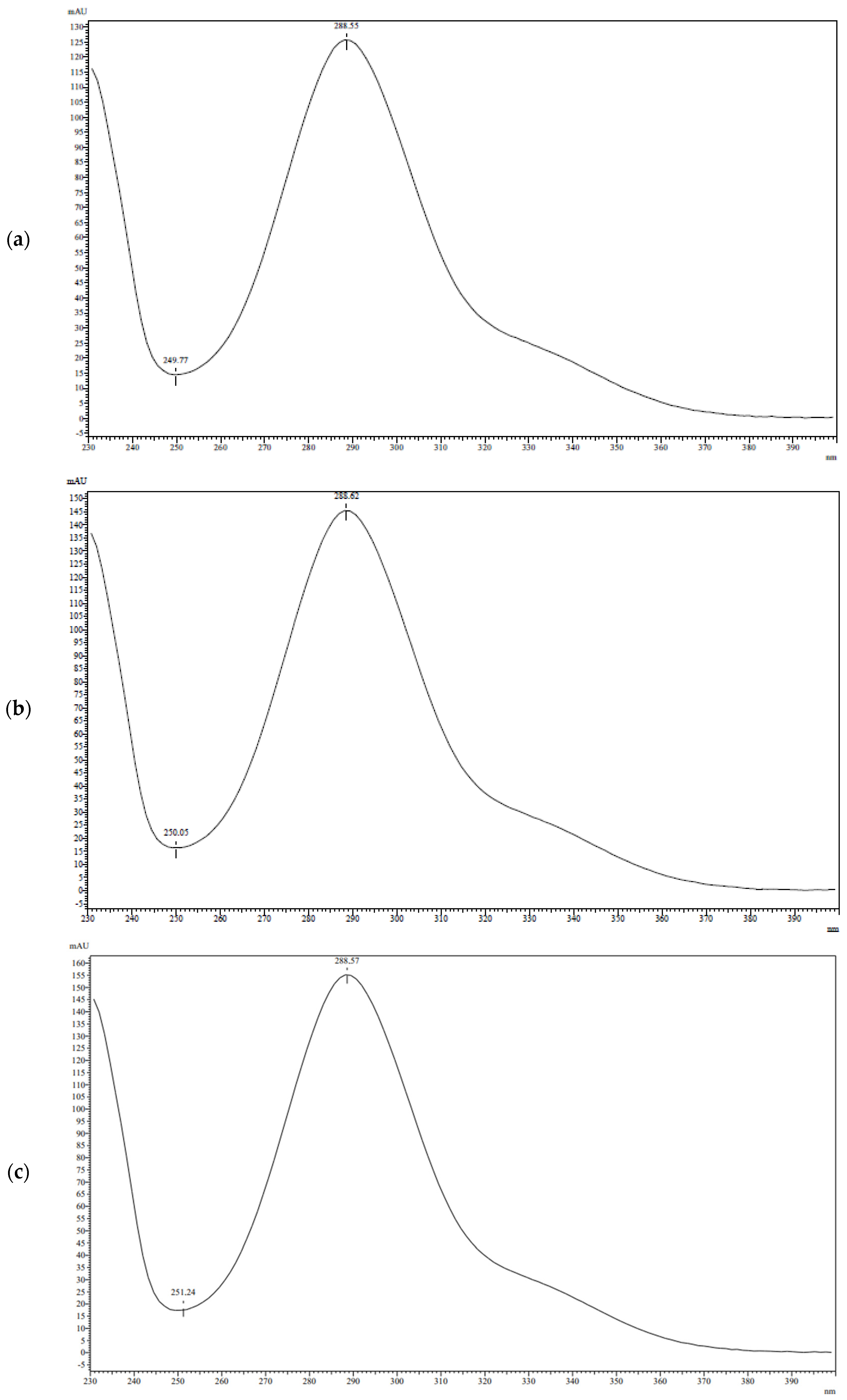
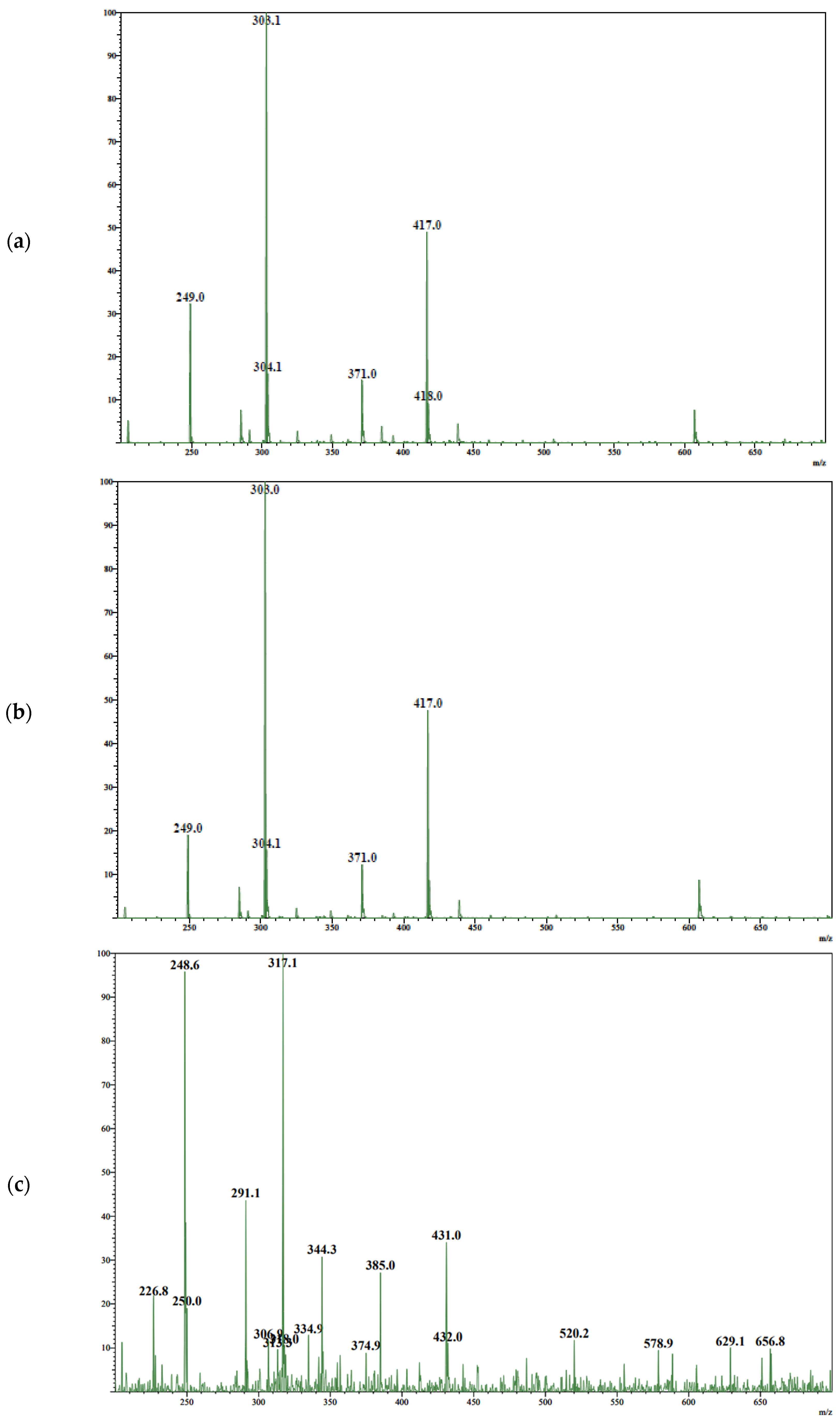
3.1. Analytical Method Development
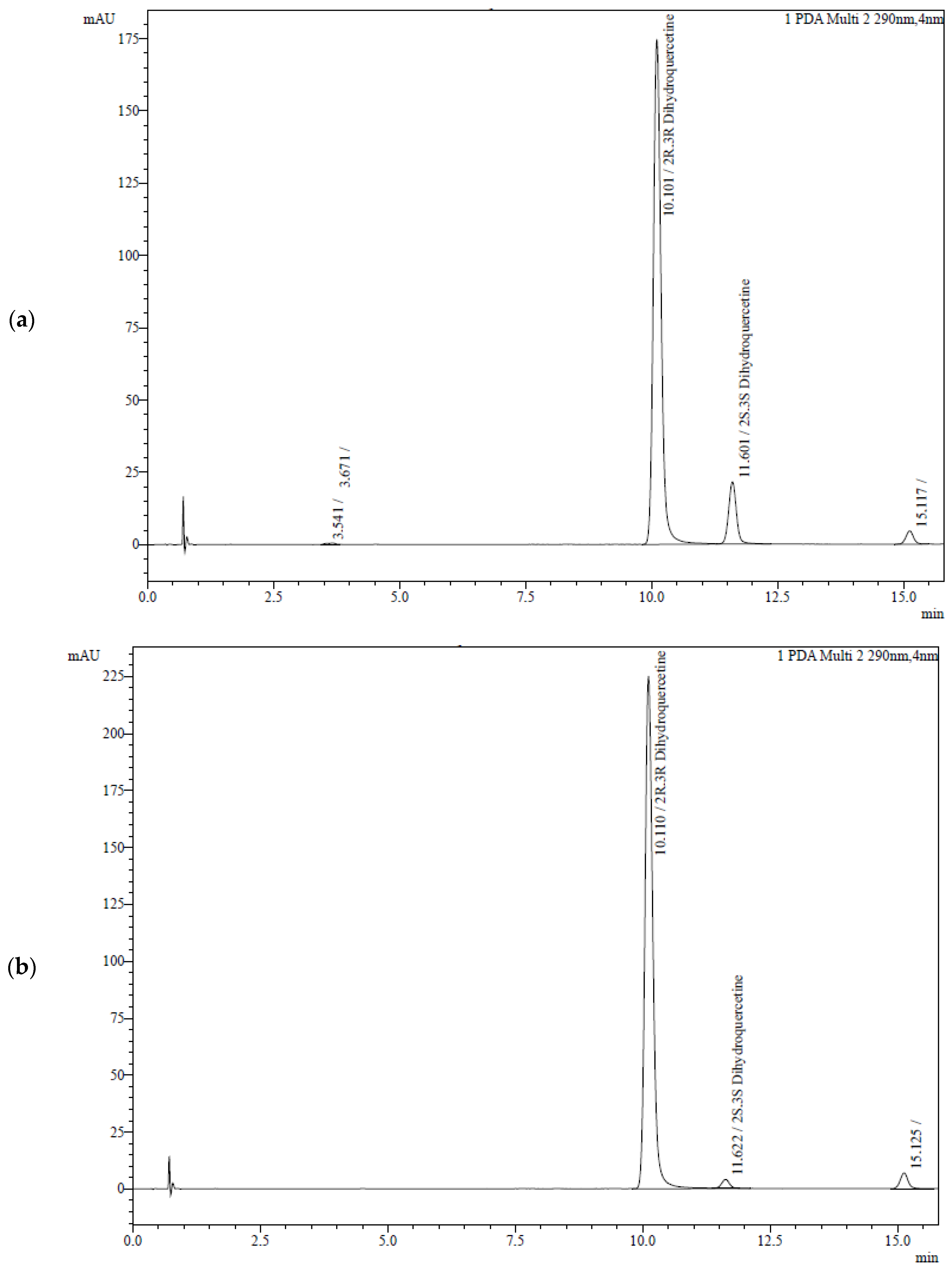
3.2. Analysis of TAXs and TAXr
3.3. Analysis of Commercially Available Samples of Taxifolin
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Das, A.; Baidya, R.; Chakraborty, T.; Samanta, A.K.; Roy, S. Pharmacological basis and new insights of taxifolin: A comprehensive review. Biomed. Pharmacother. 2021, 142, 112004. [Google Scholar] [CrossRef]
- Taldaev, A.; Terekhov, R.; Nikitin, I.; Zhevlakova, A.; Selivanova, I. Insights into the Pharmacological Effects of Flavonoids: The Systematic Review of Computer Modeling. Int. J. Mol. Sci. 2022, 23, 6023. [Google Scholar] [CrossRef]
- Jain, S.; Vaidya, A. Comprehensive review on pharmacological effects and mechanism of actions of taxifolin: A bioactive flavonoid. Pharmacol. Res.-Mod. Chin. Med. 2023, 7, 100240. [Google Scholar] [CrossRef]
- Alay, M.; Sonmez, M.G.; Sakin, A.; Atmaca, M.; Suleyman, H.; Yazici, G.N.; Coban, A.; Suleyman, B.; Bulut, S.; Altuner, D. The effects of taxifolin on neuropathy related with hyperglycemia and neuropathic pain in rats: A biochemical and histopathological evaluation. Adv. Clin. Exp. Med. 2022, 31, 427–435. [Google Scholar] [CrossRef]
- Kondo, S.; Adachi, S.I.; Yoshizawa, F.; Yagasaki, K. Antidiabetic Effect of Taxifolin in Cultured L6 Myotubes and Type 2 Diabetic Model KK-Ay/Ta Mice with Hyperglycemia and Hyperuricemia. Curr. Issues Mol. Biol. 2021, 43, 1293–1306. [Google Scholar] [CrossRef]
- Shevelev, A.B.; La Porta, N.; Isakova, E.P.; Martens, S.; Biryukova, Y.K.; Belous, A.S.; Sivokhin, D.A.; Trubnikova, E.V.; Zylkova, M.V.; Belyakova, A.V.; et al. In Vivo Antimicrobial and Wound-Healing Activity of Resveratrol, Dihydroquercetin, and Dihydromyricetin against Staphylococcus aureus, Pseudomonas aeruginosa, and Candida albicans. Pathogens 2020, 9, 296. [Google Scholar] [CrossRef]
- Terekhov, R.P.; Selivanova, I.A.; Anurova, M.N.; Zhevlakova, A.K.; Nikitin, I.D.; Cong, Z.; Ma, S.; Yang, F.; Dong, Z.; Liao, Y. Comparative Study of Wound-Healing Activity of Dihydroquercetin Pseudopolymorphic Modifications. Bull. Exp. Biol. Med. 2021, 170, 444–447. [Google Scholar] [CrossRef]
- Wang, Y.; Ding, C.; Zhao, Y.; Zhang, J.; Ding, Q.; Zhang, S.; Wang, N.; Yang, J.; Xi, S.; Zhao, T.; et al. Sodium alginate/poly(vinyl alcohol)/taxifolin nanofiber mat promoting diabetic wound healing by modulating the inflammatory response, angiogenesis, and skin flora. Int. J. Biol. Macromol. 2023, 252, 126530. [Google Scholar] [CrossRef]
- Butt, S.S.; Khan, K.; Badshah, Y.; Rafiq, M.; Shabbir, M. Evaluation of pro-apoptotic potential of taxifolin against liver cancer. PeerJ 2021, 9, e11276. [Google Scholar] [CrossRef]
- Obeidat, H.M.; Althunibat, O.Y.; Alfwuaires, M.A.; Aladaileh, S.H.; Algefare, A.I.; Almuqati, A.F.; Alasmari, F.; Aldal’in, H.K.; Alanezi, A.A.; Alsuwayt, B.; et al. Cardioprotective Effect of Taxifolin against Isoproterenol-Induced Cardiac Injury through Decreasing Oxidative Stress, Inflammation, and Cell Death, and Activating Nrf2/HO-1 in Mice. Biomolecules 2022, 12, 1546. [Google Scholar] [CrossRef]
- Teselkin, Y.O.; Babenkova, I.V.; Tjukavkina, N.A.; Rulenko, I.A.; Kolesnik, Y.A.; Kolhir, V.K.; Eichholz, A.A. Influence of dihydroquercetin on the lipid peroxidation of mice during post-radiation period. Phytother. Res. 1998, 12, 517–519. [Google Scholar] [CrossRef]
- Iwasa, M.; Kato, H.; Iwashita, K.; Yamakage, H.; Kato, S.; Saito, S.; Ihara, M.; Nishimura, H.; Kawamoto, A.; Suganami, T.; et al. Taxifolin Suppresses Inflammatory Responses of High-Glucose-Stimulated Mouse Microglia by Attenuating the TXNIP–NLRP3 Axis. Nutrients 2023, 15, 2738. [Google Scholar] [CrossRef]
- Hu, Z.; Xuan, L.; Wu, T.; Jiang, N.; Liu, X.; Chang, J.; Wang, T.; Han, N.; Tian, X. Taxifolin attenuates neuroinflammation and microglial pyroptosis via the PI3K/Akt signaling pathway after spinal cord injury. Int. Immunopharmacol. 2023, 114, 109616. [Google Scholar] [CrossRef]
- Vrhovsek, U.; Masuero, D.; Gasperotti, M.; Franceschi, P.; Caputi, L.; Viola, R.; Mattivi, F. A versatile targeted metabolomics method for the rapid quantification of multiple classes of phenolics in fruits and beverages. J. Agric. Food Chem. 2012, 60, 8831–8840. [Google Scholar] [CrossRef]
- Slimestad, R.; Fossen, T.; Vågen, I.M. Onions: A Source of Unique Dietary Flavonoids. J. Agric. Food Chem. 2007, 55, 10067–10080. [Google Scholar] [CrossRef]
- Schauss, A.G.; Tselyico, S.S.; Kuznetsova, V.A.; Yegorova, I. Toxicological and Genotoxicity Assessment of a Dihydroquercetin-Rich Dahurian Larch Tree (Larix gmelinii Rupr) Extract (Lavitol). Int. J. Toxicol. 2015, 34, 162–181. [Google Scholar] [CrossRef]
- Thuan, N.H.; Shrestha, A.; Trung, N.T.; Tatipamula, V.B.; Van Cuong, D.; Canh, N.X.; Van Giang, N.; Kim, T.S.; Sohng, J.K.; Dhakal, D. Advances in biochemistry and the biotechnological production of taxifolin and its derivatives. Biotechnol. Appl. Biochem. 2022, 69, 848–861. [Google Scholar] [CrossRef]
- Eun, C.S.; Min, P.S.; Hee, K.T.; Eun, K.Y.; Hwan, J.Y.; Seok, K.M. A Mathod for Extracting Taxifolin Glycoside with High Content and Taxifolin Non-Saccharide. KR 102380457 B1, 30 March 2022. [Google Scholar]
- Lee, S.B.; Cha, K.H.; Selenge, D.; Solongo, A.; Nho, C.W. The Chemopreventive Effect of Taxifolin Is Exerted through ARE-Dependent Gene Regulation. Biol. Pharm. Bull. 2007, 30, 1074–1079. [Google Scholar] [CrossRef]
- Lashin, S.A.; Ostronkov, V.S. Method of Dihydroquercetin Production. RU 2330677 C1, 8 October 2008. [Google Scholar]
- Levdanskij, V.A.; Levdanskij, A.V.; Kuznecov, B.N. Method for Producing Dihydroquercetin. RU 2454410 C1, 27 June 2011. [Google Scholar]
- Wiesbeck, F. Process for the Production of Taxifolin from Wood. EP 2809310 B1, 20 January 2016. [Google Scholar]
- Wiesbeck, F. Process for the Manufacture of Taxifolin from Wood. US 9073889 B2, 7 July 2015. [Google Scholar]
- Babkin, V.A.; Ostrouhova, L.A.; Babkin, D.V.; Yu, A.; Malkov. Method of Preparing Dihydroquercetin. RU 2158598 C2, 27 January 2000. [Google Scholar]
- Kislicyn, A.N.; Mal’chikov, E.L. Method of Isolation of Dihydroquercetin from Larchwood and Installation for Its Implementation. RU 2346941 C2, 20 February 2009. [Google Scholar]
- Hutoryanskij, V.A.; Bazhenov, B.N.; Sajbotalov, M.Y.U.; Tyukavkina, N.A. Method of Preparation of Dihydroquercetin. RU 96111031 A, 27 September 1997. [Google Scholar]
- Terekhov, R.P.; Selivanova, I.A.; Tyukavkina, N.A.; Shylov, G.V.; Utenishev, A.N.; Porozov, Y.B. Taxifolin tubes: Crystal engineering and characteristics. Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 2019, 75, 175–182. [Google Scholar] [CrossRef]
- Selivanova, I.A.; Terekhov, R.P. Crystal engineering as a scientific basis for modification of physicochemical properties of bioflavonoids. Russ. Chem. Bull. 2019, 68, 2155–2162. [Google Scholar] [CrossRef]
- Terekhov, R.P.; Selivanova, I.A.; Tyukavkina, N.A.; Ilyasov, I.R.; Zhevlakova, A.K.; Dzuban, A.V.; Bogdanov, A.G.; Davidovich, G.N.; Shylov, G.V.; Utenishev, A.N.; et al. Assembling the Puzzle of Taxifolin Polymorphism. Molecules 2020, 25, 5437. [Google Scholar] [CrossRef]
- Taldaev, A.; Terekhov, R.P.; Selivanova, I.A.; Pankov, D.I.; Anurova, M.N.; Markovina, I.Y.; Cong, Z.; Ma, S.; Dong, Z.; Yang, F.; et al. Modification of Taxifolin Properties by Spray Drying. Sci. Pharm. 2022, 90, 67. [Google Scholar] [CrossRef]
- Taldaev, A.; Savina, A.D.; Olicheva, V.V.; Ivanov, S.V.; Terekhov, R.P.; Ilyasov, I.R.; Zhevlakova, A.K.; Selivanova, I.A. Protective Properties of Spheroidal Taxifolin Form in Streptozotocin-Induced Diabetic Rats. Int. J. Mol. Sci. 2023, 24, 11962. [Google Scholar] [CrossRef]
- Kolhir, V.K.; Bykov, V.A.; Baginskaja, A.I.; Sokolov, S.Y.; Glazova, N.G.; Leskova, T.E.; Sakovich, G.S.; Tjukavkina, N.A.; Kolesnik, Y.A.; Rulenko, I.A. Antioxidant activity of a dihydroquercetin isolated from Larix gmelinii (Rupr.) Rupr. wood. Phytother. Res. 1996, 10, 478–482. [Google Scholar] [CrossRef]
- Selivanova, I.A.; Tyukavkina, N.A.; Kolesnik, Y.A.; Nesterov, V.N.; Kuleshova, L.N.; Khutoryanskii, V.A.; Bazhenov, B.N.; Saibotalov, M.Y. Study of the crystalline structure of dihydroquercetin. Pharm. Chem. J. 1999, 33, 222–224. [Google Scholar] [CrossRef]
- The State Pharmacopoeia of the Russian Federation, XV Edition. Available online: https://pharmacopoeia.regmed.ru/pharmacopoeia/izdanie-15/?PAGEN_1=5 (accessed on 2 November 2023).
- European Pharmacopoeia (Ph. Eur.) 11th Edition—European Directorate for the Quality of Medicines & HealthCare. Available online: https://www.edqm.eu/en/european-pharmacopoeia-ph.-eur.-11th-edition (accessed on 2 November 2023).
- Lundgren, L.N.; Theander, O. Cis- and trans-dihydroquercetin glucosides from needles of Pinus sylvestris. Phytochemistry 1988, 27, 829–832. [Google Scholar] [CrossRef]
- Kasai, R.; Hirono, S.; Tanaka, O.; Chou, W.H.; Chen, F.H. Sweet Dihydroflavonol Rhamnoside from Leaves of Engelhardtia chrysolepis, a Chinese Folk Medicine, Hung-qi. Chem. Pharm. Bull. 1988, 36, 4167–4170. [Google Scholar] [CrossRef]
- Inada, A.; Murata, H.; Somekawa, M.; Nakanishi, T. Phytochemical Studies of Seeds of Medicinal Plants. II. A New Dihydroflavonol Glycoside and a New 3-Methyl-1-butanol Glycoside from Seeds of Platycodon grandiflorum A. DE CANDOLLE. Chem. Pharm. Bull. 1992, 40, 3081–3083. [Google Scholar] [CrossRef]
- Sakurai, A.; Okumura, Y. Chemical Studies on the Mistletoe. V. The Structure of Taxillusin, a New Flavonoid Glycoside Isolated from Taxillus kaempferi. Bull. Chem. Soc. Jpn. 2006, 56, 542–544. [Google Scholar] [CrossRef]
- Dzhavakhyan, M.A.; Prozhogina, Y.E.; Pavelieva, O.K.; Kalenikova, E.I. Natural Deep Eutectic Solvents as Alternative Flavonoid Extractants from the Sedative Plant Composition. Drug Dev. Regist. 2022, 11, 75–83. [Google Scholar] [CrossRef]
- Marakhova, A.I.; Sorokina, A.A.; Stanishevskiy, Y.M. Application of through standardization principle in the analysis of flavonoids motherwort (leonurus, l.) herb and it’s preparation. Drug Dev. Regist. 2016, 1, 150–154. [Google Scholar]
- Kalashnikova, O.A.; Ryzhov, V.M.; Kurkin, V.A. Method of Quantitative Determination of the Total Flavonoid Content in Leaves of Giant Cephalaria. Pharm. Chem. J. 2023, 57, 382–387. [Google Scholar] [CrossRef]
- Dimova, G.; Sabotinova, D.; Rosenova, Y.; Zhelev, I. Changes in the content of phenolic compounds in cotinus coggygria scop. leaves collected in different months and Antimicrobial Activity of Extracts Thereof. Certif. J. Rosenovaal. World J. Pharm. Res. 2023, 12, 1125. [Google Scholar] [CrossRef]
- Andonova, T.; Muhovski, Y.; Slavov, I.; Vrancheva, R.; Georgiev, V.; Apostolova, E.; Naimov, S.; Mladenov, R.; Pavlov, A.; Dimitrova-Dyulgerova, I. Phenolic Profile, Antioxidant and DNA-Protective Capacity, and Microscopic Characters of Ailanthus altissima Aerial Substances. Plants 2023, 12, 920. [Google Scholar] [CrossRef] [PubMed]
- Poluyanov, A.M.; Sokolova, A.Y.; Koynova, A.D.; Kulikova, S.D.; Malashenko, E.A.; Bobkova, N.V. Identification and Quantitative Determination of Flavonoids by HPLC-UV Method in the Raw Materials of Some Representatives of the Genus Rumex of Three Vegetation Time. Drug Dev. Regist. 2023, 12, 134–142. [Google Scholar] [CrossRef]
- Gerasimov, M.A.; Perova, I.B.; Eller, K.I.; Akimov, M.Y.; Sukhanova, A.M.; Rodionova, G.M.; Ramenskaya, G.V. Investigation of Polyphenolic Compounds in Different Varieties of Black Chokeberry Aronia melanocarpa. Molecules 2023, 28, 4101. [Google Scholar] [CrossRef] [PubMed]
- Kurkin, V.A.; Zimenkina, N.I. HPLC Determination of Myricitrin in Juglans nigra L. Bark. Pharm. Chem. J. 2021, 55, 881–885. [Google Scholar] [CrossRef]
- Epshtein, N.A. Validation of Chromatographic Methods: Checking the Peak Purity and the Specificity of Methods with Diode Array Detectors (Review). Drug Dev. Regist. 2020, 9, 129–136. [Google Scholar] [CrossRef]
- Voronin, K.S.; Fenin, A.A.; Zhevlakova, A.K.; Zavadskii, S.P.; Selivanova, I.A. Polyphenolic Profile of Larch Knotwood. Pharm. Chem. J. 2021, 55, 781–786. [Google Scholar] [CrossRef]
- Voronin, K.S.; Fenin, A.A.; Zhevlakova, A.K.; Pyzhov, V.S.; Selivanova, I.A. Development and Validation of a Method for Simultaneous Quantification of Dihydroquercetin and Secoisolariciresinol. Pharm. Chem. J. 2023, 57, 740–744. [Google Scholar] [CrossRef]
- Xu, Y.; Li, Z.; Wang, Y.; Li, C.; Zhang, M.; Chen, H.; Chen, W.; Zhong, Q.; Pei, J.; Chen, W.; et al. Unraveling the Antioxidant Activity of 2R,3R-dihydroquercetin. Int. J. Mol. Sci. 2023, 24, 14220. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, A.G.M.; Branco, A.; Câmara, C.A.; Silva, T.M.S.; Silva, T.M.G.; de Oliveira, A.P.; Santos, A.D.d.C.; Dutra, L.M.; Rolim, L.A.; de Oliveira, G.G.; et al. Identification of flavonoids in Hymenaea martiana Hayne (Fabaceae) by HPLC-DAD-MSn analysis. Nat. Prod. Res. 2021, 35, 2414–2419. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Yang, D.; Ha, S.H.; Lee, S.Y. Biosynthesis of dihydroquercetin in Escherichia coli from glycerol. bioRxiv 2020, 11, 401000. [Google Scholar] [CrossRef]
- Lee, H.; Jeong, W.T.; So, Y.S.; Lim, H.; Bin; Lee, J. Taxifolin and sorghum ethanol extract protect against hepatic insulin resistance via the mir-195/irs1/pi3k/akt and ampk signalling pathways. Antioxidants 2021, 10, 1331. [Google Scholar] [CrossRef] [PubMed]
- Abad-García, B.; Garmón-Lobato, S.; Berrueta, L.A.; Gallo, B.; Vicente, F. A fragmentation study of dihydroquercetin using triple quadrupole mass spectrometry and its application for identification of dihydroflavonols in Citrus juices. Rapid Commun. Mass Spectrom. 2009, 23, 2785–2792. [Google Scholar] [CrossRef] [PubMed]
- Kiehlmann, E.; Li, E.P.M. Isomerization of dihydroquercetin. J. Nat. Prod. 1995, 58, 450–455. [Google Scholar] [CrossRef]
- Choonara, I.; Haynes, B.; Cholerton, S.; Breckenridge, A.; Park, B. Enantiomers of warfarin and vitamin K1 metabolism. Br. J. Clin. Pharmacol. 1986, 22, 729–732. [Google Scholar] [CrossRef] [PubMed]
- Boulton, D.W.; Fawcett, J.P. Enantioselective Disposition of Albuterol in Humans. Clin. Rev. Allergy Immunol. 1996, 14, 115–138. [Google Scholar] [CrossRef]
- FDA’s policy statement for the development of new stereoisomeric drugs. Chirality 1992, 4, 338–340. [CrossRef]
- Gaggeri, R.; Rossi, D.; Christodoulou, M.S.; Passarella, D.; Leoni, F.; Azzolina, O.; Collina, S. Chiral Flavanones from Amygdalus lycioides Spach: Structural Elucidation and Identification of TNFalpha Inhibitors by Bioactivity-guided Fractionation. Molecules 2012, 17, 1665–1674. [Google Scholar] [CrossRef]
- Towatari, K.; Yoshida, K.; Mori, N.; Shimizu, K.; Kondo, R.; Sakai, K. Polyphenols from the Heartwood of Cercidiphyllum japonicum and their Effects on Proliferation of Mouse Hair Epithelial Cells. Planta Med. 2002, 68, 995–998. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, G.I.; Nishioka, I.; Goto, Y.; Kinjo, J.E.; Nohara, T. Tannins and Related Compounds. LII. Studies on the Constituents of the Leaves of Thujopsis dolabrata SIEB. et ZUCC. Chem. Pharm. Bull. 1987, 35, 1105–1108. [Google Scholar] [CrossRef]
| Conditions | Characteristics | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No | Stationary Phase | Eluents | Elution Time, min | Elution Mode (B eluent, %) | T, °C | Flow Rate, mL/min | trans-Isomer | cis-Isomer | Rs | |||||
| A | B | RT, min | TF | NTP | RT, min | TF | NTP | |||||||
| 1 | Luna 5 µ C18(2) 100 Å, 250 × 4.6 mm | 0.01% H3PO4 in water | MeCN | 20 | isocratic (30) | 25 | 1.00 | 5.4 | 1.7 | 1668 | 6.1 | 2.1 | 1796 | - |
2 | Luna 5µ CN 100 Å, 250 × 4.6 mm | 0.01% H3PO4 in water | MeCN | 30 | gradient (10–90) | 25 | 1.00 | 8.5 | 0.9 | 4551 | 9.2 | 1.2 | 17,767 | 1.7 |
| 3 | (10–55) | 9.3 | 1.8 | 3150 | 9.9 | 2.1 | 18,263 | 1.2 | ||||||
| 4 | (20–50) | 6.2 | 0.8 | 4914 | - | - | - | - | ||||||
| 5 | 15 | (30–50) | 4.9 | 1.0 | 4403 | - | - | - | - | |||||
| 6 | Kinetex® 2.6 μm Biphenyl 100 Å, 100 × 3.0 mm | 0.1% formic acid in water | 0.2% formic acid in MeOH | 15 | gradient (15–40) | 60 | 0.65 | 4.9 | 1.3 | 15,721 | 5.6 | 1.2 | 18,708 | 4.0 |
| 7 | (7–21) | 10.1 | 1.1 | 20,531 | 11.6 | 1.0 | 26,285 | 5.3 | ||||||
| Sample Name | Moist, % | Quantity of Taxifolin Diastereomers | trans/cis-Isomers Ration | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| trans-Isomer | cis-Isomer | Sum of Isomers | |||||||||||||
| mg/mL | % | mg/mL | % | mg/mL | % | ||||||||||
| M | ±SD | M | ±SD | M | ±SD | M | ±SD | M | ±SD | M | ±SD | M | ±SD | ||
| TAXs | 5.0 | 0.2 | 0.072 | 0.001 | 75.8 | 1.1 | 0.009 | n/a | 9.5 | n/a | 0.081 | 0.001 | 85.3 | 1.3 | 8:1 |
| TAXr | 7.3 | 0.1 | 0.091 | 0.001 | 98.2 | 1.5 | 0.003 | n/a | 3.7 | n/a | 0.094 | 0.001 | 101.9 | 1.5 | 27:1 |
| Taxifolia | 5.2 | 0.5 | 0.089 | 0.001 | 93.9 | 1.4 | 0.001 | n/a | 1.5 | n/a | 0.090 | 0.001 | 95.1 | 1.4 | 63:1 |
| Robios | 8.5 | 0.3 | 0.082 | 0.001 | 89.6 | 1.3 | 0.001 | n/a | 0.8 | n/a | 0.083 | 0.001 | 90.4 | 1.3 | 112:1 |
| Sample Name | [α]D, ° | |||
|---|---|---|---|---|
| In Ethanol | In Acetone | |||
| M | ±SD | M | ±SD | |
| TAXs | 22.1 | 0.5 | 32.0 | 1.7 |
| TAXr | 21.9 | 0.5 | 30.4 | 1.6 |
| Taxifolia | 18.8 | 0.7 | 20.5 | 1.2 |
| Robios | 18.9 | 0.6 | 24.4 | 1.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terekhov, R.P.; Melnikov, E.S.; Nikitin, I.D.; Tokareva, M.A.; Rodina, T.A.; Savina, A.D.; Pankov, D.I.; Zhevlakova, A.K.; Beloborodov, V.L.; Selivanova, I.A. Diastereomers of Spheroidal Form and Commercially Available Taxifolin Samples. Sci. Pharm. 2024, 92, 5. https://doi.org/10.3390/scipharm92010005
Terekhov RP, Melnikov ES, Nikitin ID, Tokareva MA, Rodina TA, Savina AD, Pankov DI, Zhevlakova AK, Beloborodov VL, Selivanova IA. Diastereomers of Spheroidal Form and Commercially Available Taxifolin Samples. Scientia Pharmaceutica. 2024; 92(1):5. https://doi.org/10.3390/scipharm92010005
Chicago/Turabian StyleTerekhov, Roman P., Evgeny S. Melnikov, Ilya D. Nikitin, Margarita A. Tokareva, Tatyana A. Rodina, Anastasiya D. Savina, Denis I. Pankov, Anastasiya K. Zhevlakova, Vladimir L. Beloborodov, and Irina A. Selivanova. 2024. "Diastereomers of Spheroidal Form and Commercially Available Taxifolin Samples" Scientia Pharmaceutica 92, no. 1: 5. https://doi.org/10.3390/scipharm92010005
APA StyleTerekhov, R. P., Melnikov, E. S., Nikitin, I. D., Tokareva, M. A., Rodina, T. A., Savina, A. D., Pankov, D. I., Zhevlakova, A. K., Beloborodov, V. L., & Selivanova, I. A. (2024). Diastereomers of Spheroidal Form and Commercially Available Taxifolin Samples. Scientia Pharmaceutica, 92(1), 5. https://doi.org/10.3390/scipharm92010005








