Abstract
Foods contain dietary fibers which can be classified into soluble and insoluble forms. The nutritional composition of fast foods is considered unhealthy because it negatively affects the production of short-chain fatty acids (SCFAs). Dietary fiber is resistant to digestive enzymes in the gut, which modulates the anaerobic intestinal microbiota (AIM) and fabricates SCFAs. Acetate, butyrate, and propionate are dominant in the gut and are generated via Wood–Ljungdahl and acrylate pathways. In pancreatic dysfunction, the release of insulin/glucagon is impaired, leading to hyperglycemia. SCFAs enhance insulin sensitivity or secretion, beta-cell function, leptin release, mitochondrial function, and intestinal gluconeogenesis in human organs, which positively affects type 2 diabetes (T2D). Research models have shown that SCFAs either enhance the release of peptide YY (PYY) and glucagon-like peptide-1 (GLP-1) from L-cells (entero-endocrine), or promotes the release of leptin hormone in adipose tissues through G-protein receptors GPR-41 and GPR-43. Dietary fiber is a component that influences the production of SCFAs by AIM, which may have beneficial effects on T2D. This review focuses on the effectiveness of dietary fiber in producing SCFAs in the colon by the AIM as well as the health-promoting effects on T2D.
1. Introduction
The gut microbiota (GM) is a complicated and dynamic ecosystem that interacts with the host, maintaining a mutualistic relationship. The microbes can influence numerous physiological mechanisms, including those involved in glucose regulation, lipid metabolism, pathogen resistance, and micronutrient production [1]. Thus, modulating GM might be a reasonable approach to preventing inflammatory and metabolic diseases. For example, previous studies employing animal models showed that modulating GM had salutary effects on obesity, insulin sensitivity, and type 2 diabetes (T2D) [2,3]. However, no evidence was found that specific microbial communities were directly linked to these diseases, while some evidence suggested that gut microbial activity is beneficially associated with T2D [4]. In addition, the composition of GM is influenced by internal and external factors. Genetics plays a major role in elucidating the gut microbial composition, and several potential strategies have been employed to induce positive changes in gut microbial communities via fermentative activity [5]. The GM is an essential storehouse for human health; 60 bacterial phyla have been identified in the human gastrointestinal tract (GIT) (including Firmicutes, Bacteroides, Actinobacteria, Fusobacteria, Proteobacteria, Verrucomicrobia, Cyanobacteria, and Spirochaetes) [6].
Dietary fiber is an essential component of food composed of a complex polymer of phenylpropanoid units [7,8]. It is mainly classified into four subgroups: resistant starches, lignins, resistant oligosaccharides (galacto-oligosaccharides, fructo-oligosaccharides, etc.), and non-starchy polysaccharides (cellulose, hemicellulose, and pectin) [9]. Soluble fiber is resistant to gastrointestinal digestive enzymes and is utilized by the anaerobic intestinal microbiota (AIM) to produce short-chain fatty acids (SCFAs), whereas insoluble fiber is not degraded or utilized by the human GIT [9,10]. Regular consumption of soluble dietary fiber may modulate the intestinal microbiota, positively affecting T2D [11,12,13,14]. Legumes are considered to have an excess amount of fibers including resistant starch (RS) [15,16]. The recommended amount of dietary fiber by the World Health Organization, Food and Agriculture Organization (WHO/FAO), and European Food Safety Authority (EFSA) is 25 g/day, depending on the laxation of healthy individuals [17,18]. Intervention with dietary fiber in the AIM has numerous benefits for human health, such as energy consumption, AIM integrity, and regulation of immune functions; arabinoxylan has shown beneficial effects in T2D, linked to AIM amendment and metabolites produced during fermentation [19]. Types of water-soluble/insoluble dietary fiber are shown in Figure 1.
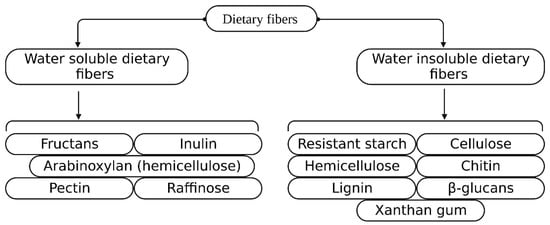
Figure 1.
Dietary fiber classification based on water solubility/insolubility.
SCFAs are a sub-class of fatty acids composed of carbon atoms (six or fewer), acetate (C2), propionate (C3), butyrate (C4), pentanoic acid (C5), and hexanoic acid (C6) [20]. Among them, the focus is primarily on acetate, propionate, and butyrate due to their excess production in GIT [21]. These fatty acids are generated from dietary fiber through fermentation via the AIM in the mammalian colon and have shown beneficial effects on metabolic activity [22]. The consumption of excess amounts of starchy foods and avoidance of physical exercise leads to the disruption of energy balance and generates intricate symptoms, collectively called metabolic syndrome; hypertension, obesity, glycemic imbalance, and T2D are typical manifestations of metabolic syndrome [23].
The higher intake of dietary fibers affects T2D, non-digestible oligosaccharides are fermented by gut microbial communities, producing SCFAs, which may positively contribute to different organs of the body [24]. Several systematic reviews have previously focused on fibers, lifestyle interventions, probiotics, and fecal microbial transplantation, and its effects on T2D [25,26]. This review focuses on the effectiveness of fiber in producing SCFAs in the colon by the AIM as well as the health-promoting effects in T2D, and the negative effects of fast-food consumption on T2D have also been addressed. This review provides a reference for subsequent research.
2. Fibers
2.1. Dietary Fiber
Dietary fiber, comprising endogenous non-digestible carbohydrates and lignins, is an essential component of plants [23]. The types of fiber differ in their anaerobic fermentability, viscosity, chemical structure, and solubility in water [27]. Such fiber consists of the polymers of carbohydrates, combining monomeric units (three or more), which are not hydrolyzed/absorbed in the human gut when exposed to digestive enzymes [1]. Dietary fiber is classified into two categories, soluble and insoluble. Soluble dietary fiber is preferable because it is metabolized by the AIM, producing SCFAs [28]. Firmicutes and Actinobacteria species are considered to especially respond to dietary fiber [1,29].
The endogenous components of plant-based foods have been well studied for the last few decades, in which dietary fibers positively affect the human host. Meta-analysis showed that the consumption of galacto-oligosaccharides and fructans may enhance the biodiversity of Lactobacillus and Bifidobacterium species in the gut [30]. Dietary fiber directly influences the production of SCFAs in the human gut (lumen) [11,31]. An increase in dietary fiber increases the production of SCFAs and vice versa. Low intake of dietary fiber can also affect the production of amino acids and mucins, which reduces energy production for metabolic activity [1,29]. Previous studies have shown that high fiber intake leads to higher production of SCFAs (acetate, propionate, and butyrate) and vice versa [31]. In a pilot study on obese volunteers investigating rice bran/cooked navy beans (rich in dietary fiber), the results showed that the number of SCFAs (acetate and butyrate) was increased while the Firmicutes to Bacteroidetes proportion was decreased [31,32]. Previous studies investigated whether dietary fiber from whole grains and/or vegetables/fruits affects inflammatory markers and the composition of GM. Whole grain showed a significant decrease in lipopolysaccharide (LPS) and tumor necrosis factor α (TNF-α), while vegetables/fruits showed substantial changes in interleukin-6 (IL-6) [33,34].
2.2. Prebiotic Inulin
Inulin belongs to a class of dietary fiber called fructans, which are produced in plants [35,36]. Being prebiotic, the components of fructans are effectively modulated in the AIM (with Bifidobacterium spp. dominating) and positively respond in T2D patients via the production of SCFAs (acetic and propionic acids) in the ileum of the GIT [37]. Inulin-type fructans (ITFs) are systemically beneficial, promoting AIM growth and producing H2S, CO2, and organic acids. Fructans also have beneficial effects on metabolic syndrome, including T2D [38]. ITFs modulate GM and increase the production of SCFAs (acetic and propionic acids) [38], which improves the level of hemoglobin Alc by accelerating glucagon-like peptide-1 (GLP-1) production, resulting in the reduction of harmful compounds such as H2S and indole [39]. Moreover, ITFs may also regulate inflammation associated with LPS, IL-6, TNF-α, and interferon-γ [40,41].
2.3. Resistant Starch
Starches are complex polysaccharides in the form of grains stored in roots, seeds, and fruits [42,43]. These polysaccharides are present in the human diet in maize, cassava, potatoes, rice, and wheat [44,45]. Regarding digestibility, starches are classified into three classes: resistant starch (RS), slowly digestible starch (SDS), and rapidly digestible starch (RDS) [46,47]. RS is the fraction of starch that is indigestible by gut digestive enzymes, fermented by the AIM, and known to produce SCFAs [48,49,50]. The term “resistant starch” was first used by Englyst in the 1980s [51], and the efficiency of resistant starch with regard to prebiotics and lipid/glucose metabolism was studied concerning the gut environment [52,53,54,55,56,57,58]. As a component of functional foods, an indigestible portion of RS is categorized as dietary fiber. RS-1, RS-2, RS-3, RS-4, and RS-5 are sub-types of RS, among which RS-1 is substantially inaccessible, i.e., it has intact cell walls (encapsulated) that prevent access by digestive enzymes [59]. RS-2 comprises starch granules with crystalline polymers (B- or C-); this type of starch lacks water channels, and due to the condensed surface, it provides fewer sites for digestive enzymes [52]. RS-3 contains retrograded starch, which is normally found in cooked food (plant-based), and the retrograde/double helix structure of RS-3 starch molecules prevents attachment to digestive enzymes [60]. The functional group RS-4 acts by restraining the attachment of digestive enzymes [61]. The configuration of amylose-lipid complexes in RS-5 prevents it from fitting into the binding pockets of digestive enzymes [62]. Foods containing high RS content are significantly beneficial for human health because the fermentation of RS in the colon produces SCFAs by AIM, and the quantity of acetic, butyric, and propionic acids is higher than the quantity of iso-butyric, valerian, and iso-valeric acids [63]. Previous studies showed numerous effects of SCFAs on human health, including reduced cholesterol and triglyceride levels in blood and providing a significant amount of energy to colonocytes, which balance the status of the colonic epithelial lining [64,65,66,67]. In addition, SCFAs are beneficial in terms of glucose reduction and insulin secretion, and show positive effects on T2D [64,68,69,70,71].
3. Dietary Fiber, Inflammatory Markers, and T2D
T2D is among the major diseases associated with a low level of inflammatory processes, characterized by amendments in the secretion of cytokines [72]. The amount of inflammatory markers (IL-6, TNF-α, and LPS) in T2D is increased, which is associated with dysfunction in insulin resistance and β-cell activity, and the amount of LPS in diabetic patients is twice as high as that in healthy individuals [73]. A high-fat diet is associated with metabolic endotoxemia caused by serum LPS, resulting in obesity and insulin resistance [74], and high serum LPS enhances TNF-α and inhibits insulin signals [75]. An excess amount of ANK-α indirectly inhibits insulin signaling by serine-307 phosphorylation in the substrate of the insulin receptor [76]. According to scientific reports, the composition of the diet can positively affect the inflammatory process; Lactobacillus spp. and Bifidobacterium spp., which are stimulated by dietary fiber, show anti-inflammatory properties [77]. Dietary fiber at 40 g/day can reduce the level of TNF-α [78].
4. Effects of Fructose on SCFAs and T2D
Sugar is an important source of energy in our daily diet, and there is increasing evidence that high sugar intake causes a number of major diet-related health problems, such as T2D and obesity [79,80]. Dietary factors influence blood glucose homeostasis in T2D; blood glucose levels rise when fructose is converted into glucose in the liver. This conversion takes time, so a small portion of fructose is converted into glucose, resulting in a lower increase in blood glucose levels [81]; therefore, the glycemic index of fructose is only 23 [82]. In addition to contributing to blood glucose homeostasis, fructose has also been shown to improve glycemic control at moderate levels [83,84]. The health effects of fructose are closely related to the consumption amount. Ultimately, it was determined that a high-fructose diet and a certain gut microbiota profile may be associated with the inflammation of the liver, pancreas, and colon. With low or inadequate fructose intake, no adverse effects were found on body weight, fasting blood glucose, histology, gut microbiota, or colonic SCFA levels [85,86,87]. Some evidence showed that fructose causes insulin resistance in the liver, which can negatively impact blood glucose homeostasis [88].
5. Effects of Lipids on SCFAs and T2D
A lipid molecule is mostly made up of repeating units named fatty acids. There are two types of fatty acids, saturated and unsaturated. Humans get most of their energy from fatty acids, which are the main components of triacylglycerols found in oils and fats [89,90]. Long-term consumption of a high-fat diet affects gut microbiota composition in animal models as well as in humans, which directly impacts SCFA production and host health [91]. High-fat diets containing medium-chain fatty acids, monounsaturated fatty acids, and polyunsaturated fatty acids, low-fat diets containing long-chain fatty acids, and diets with high Bacteroidetes or Firmicutes ratios were associated with increased SCFA production [92].
6. Short-Chain Fatty Acids (SCFAs)
SCFAs are organic acids produced in the human gut, where the AIM resides [70]. Quantitatively, these fatty acids are measured in millimoles, and they are predominately represented by acetate, butyrate, and propionate [93]. These three SCFAs are discussed in the current review. The dietary carbon flow is based on SCFAs [94], and their production is fairly well understood and characterized [95,96]. The ratio and concentration of SCFAs depend on the microbial composition and the substrate (dietary fiber) provided to the GM [97]. The molar ratio of acetate, propionate, and butyrate is 3:1:1. SCFAs constitute 90–95% of the colon, whereas formic acid is present in a smaller proportion [93]. As a result of antibiotic treatment depleting the microbiota, mice were found to produce lower amounts of SCFAs, compared to mice that did not receive antibiotics [98]. A diet rich in prebiotics may be particularly effective at increasing SCFA production in diabetes [99]. According to previous studies, individuals with T2D have lower proportions of microbiota species producing butyrate [99,100]. Some of the beneficial properties of SCFAs that positively affect human health are shown in Table 1.

Table 1.
Important health benefits of short-chain fatty acids in modulating gut microbiota.
6.1. The Contribution of Gut Microbiota Producing SCFAs
Dietary fibers are resistant to gut digestive enzymes, which contribute to the production of SCFAs during colonic fermentation [104]. Acetate, propionate, and butyrate are dominant SCFAs in the gut [77]. These fatty acids are composed of 1–6 carbon atoms and are naturally saturated [29]. Present-day research has shown a significant role for AIM, and the metabolites produced during dietary fiber fermentation positively contribute to T2D [105]. Gut intestinal microbiota, including Clostridiales spp. SS3/4, Roseburia inulinivorans, Roseburia intestinalis, Faecalibacterium prausnitzii, and Eubacterium rectale, produce butyrate, which has a protective role in T2D, even though these species are decreased in diabetes [106]. In addition, oral administration of Clostridium butyricum in obese diabetic rats was found to modulate gut microbiota to produce butyrate, leading to reduced proportions of Bacteroides and Firmicutes spp. [107].
In diet-induced diabetes, chitosan and antibiotics targeting Gram-negative intestinal microbes may be considered antidiabetic agents [108,109]. Cross-feeding GM metabolizes lactate into acetate, propionate, and butyrate in the gut fermentation process, in which propionate and butyrate are produced in limited quantities owing to selected GM, while acetate is a regular product in the gut [94]. Propionate is produced during the fermentation of propiogenic substrate (fucose/rhamnose) by Akkermansia municiphilla, whereas butyrate is produced through RS fermentation by Eubacterium hallii, Eubacterium rectale, Faecalibacterium prausnitzii, and Ruminococcus bromii in the gut; moreover, butyrogenic bacteria ferment pyruvate, lactate, and acetate into butyrate [93]. Acetate, propionate, and butyrate are the energy sources for the human body. Butyrate is directly utilized in the liver, heart, brain, and colon; propionate is used for gluconeogenesis in the liver, and acetate is used as fuel in peripheral tissues [110].
The responsiveness of free fatty acid receptors (FFAR-2 and -3) is proportional to the length of the carbon chain. For example, acetate and propionate are more responsive to FFAR-2, whereas butyrate and propionate are more responsive to FFAR-3 [111]. Medium (FFAR-1) and long-chain (FFAR-4) fatty acids were found to positively respond to inflammation and insulin secretion [112]. FFAR-1 enhances specific pancreatic β-cell activity, while in T2D, this activity is downregulated, resulting in FFAR-1 inhibition and insulin resistance [113]. FFAR-4 boosts these fatty acids (unsaturated) to stimulate glucagon-like peptide-1 (GLP-1), secreting insulin from β-cells [114]. Propionate and butyrate may positively regulate obesity and T2D when administered orally [115,116,117].
6.2. Production of SCFAs via Anaerobic Bacterial Pathways and the Role of Akkermansia Muciniphila in T2D
The non-digestible carbohydrates are hydrolyzed by the AIM into monosaccharides and oligosaccharides during anaerobic fermentation in the colon [118]. For the metabolization of monosaccharides into phosphoenolpyruvate (PEP), the Embden–Meyerhof–Parnas pathway (sugars containing 6-c) and the pentose phosphate pathway (sugars containing 5-c) are utilized [95]. Eventually, organic acids/alcohols are formed during PEP fermentation. Nicotinamide adenine dinucleotide (NAD) + hydrogen (H) (NADH) is produced during the reaction of an acidic protein, glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Three pathways contribute to the disposal of excess reducing equivalents, as presented in Figure 2A. First is the traditional fermentation pathway, in which lactate/ethanol is produced from the reduction of pyruvate. Second, pyruvate is reduced to acetyl-CoA (ACA) and NADH to NAD+ [119]. The second pathway produces excess amounts of H2 molecules by using two major routes, pyruvate (exergonic) and NADH (endergonic) via ferredoxin oxidoreductase and hydrogenase, respectively. Despite depleting/consuming H2 molecules, the AIM is a primary participant in the fermentation process when H2 pressure in the large intestine (lumen) is low [120]. Third, the fundamental electron transport chain (ETC) proceeds with anaerobes, starting with PEP carboxylation and the reduction of oxaloacetate into fumarate [121]. The electrons are accepted by fumarate from NADH; NADH dehydrogenase and fumarate reductase constituted an ordinary electron transfer chain (OETC) [121,122]. NADH-dehydrogenase contributes to the transport of protons across the cell membrane, resulting in the chemiosmotic synthesis of ATP. Succinate (produced by fumarate reductase) is transformed into methylmalonate once the preferential load of CO2 is reduced. PEP can also be recycled from oxaloacetate through the carboxylation process.
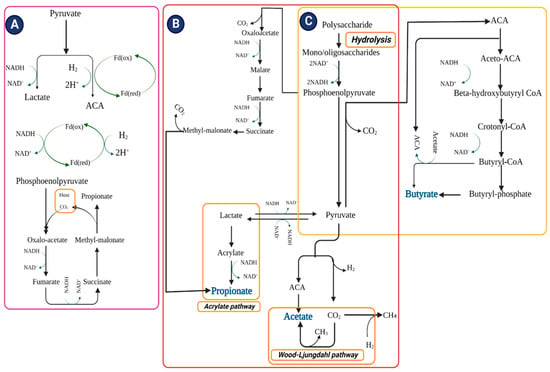
Figure 2.
Schematic presentation of three pathways that contribute to disposing of excess reducing equivalents. (A) Reduction of pyruvate into lactate, thereby reducing NADH, pyruvate; ferredoxin oxidoreductase and hydrogenase/NADH; ferredoxin oxidoreductase and hydrogenase dispose of reducing equivalents into molecular hydrogen and NADH is reduced via electron transport chain. (B) Acetate is formed directly from acetyl CoA through the Wood–Ljungdahl pathway. Propionate is formed from PEP via succinate decarboxylation pathway or acrylate pathway while reducing propionate. (C) Butyrate is shaped by condensing acetyl CoA (two molecules) in the presence of butyrate kinase or by employing exogenously derived acetate through butyryl-CoA: acetate-CoA transferase. Abbreviations: NADH; nicotinamide adenine dinucleotide (NAD) + hydrogen (H): ACA; acetyl coenzyme A: H; Hydrogen: CO2; carbon dioxide: CH3; methyl radical: CH4; methane.
SCFAs are the end product of the fermentation pathways. Pyruvate is transformed into ACA, releasing H2 and CO2 molecules. Hydrolysis of ACA leads to the formation of acetate, or it can also be produced by the Wood–Ljungdahl pathway utilizing CO2, wherein CO2 is reduced to CO coupled with CoASH and a methyl group and converted to ACA [123,124]. Propionate is formed either by utilizing PEP via OETC or by reducing lactate to propionate via the acrylate pathway [95]. These pathways accommodate supplementary NADH associated with lactate fermentation (Figure 2B). The condensation of ACA (2 molecules) results in the formation of butyrate, which is subsequently reduced to butyryl CoA (Figure 2C). ACA is produced from lactate, and then lactate is utilized by gut bacteria to produce butyrate [125]. Two pathways are involved in the formation of butyrate: the traditional pathway uses phosphotransbutyrylase and butyrate kinase to convert butyryl CoA into butyrate, accompanying ATP formation, and in the alternative pathway, butyryl CoA is converted to butyrate via butyryl-CoA: acetate CoA transferase [126,127]. The exogenic utilization of acetate to form butyrate and ACA involves cross-feeding among acetate and butyrate-producing bacteria [128,129]; the human GM prefers the alternative over the traditional pathway [126].
The symbiotic association between GM and the human body is significant in SCFA production [130]. The primary metabolites (H2 molecules) produced to get acetate must be utilized by secondary fermenters to reduce the burden of these molecules and accelerate the oxidation of NADH via primary fermenters [131]. The human body provides the CO2 molecules required in the OETC, and an average of 0.7 kg/day of CO2 is produced by the human organism [132]. By exchanging SCFA anions, some of that production is secreted into the gut (lumen) as HCO3, which is likely a significant pH-regulating mechanism, since protons in the gut (lumen) generated during the formation of SCFAs are neutralized by bicarbonate to produce CO2 [131]. Subsequently, much is known about the biochemistry of SCFA production from carbohydrates via the AIM. However, further study is still needed to determine whether SCFAs, as the significant output of indigestible carbohydrates via the AIM, have beneficial effects in T2D.
Akkermansia muciniphila is the only representative Gram-negative Verrucomicrobia inhabiting human intestinal mucosa [133]. In the studies by Derrien, gene sequence analysis revealed that multiple genes are associated with mucin encoding, and a single chromosome containing 2176 genes with 55.8% GC content was found in the MucT type strain of A. muciniphila (ATCC BAA-835 1/4 CIP107961T) [134,135]. This immobile, oval-shaped microorganism is purely anaerobic and contains chemical organotrophic material that can endure low levels of oxygen. The enzymes produced by A. muciniphila were responsible for the breakdown of mucin, and the mucin in the mucosal layer of the epithelium was used as a source of carbon and nitrogen. In order to release the sulfate, A. muciniphila splits these compounds into acetic and propionic compounds [136,137]. According to an analysis of its 16SrRNA signature, A. muciniphila makes up 3 to 5% of the gut microbiome even in healthy adults, but the amount depends on several factors. Age has been closely associated with stability in humans. This species begins to colonize at a young age and ranges from 5.0 to 8.8 log cells/g in a year, which is comparable to the adult stage, although it decreases with age [138,139]. The combined effects of an excess amount of A. muciniphila supplementation can positively affect metabolic disorders including T2D, and early vancomycin therapy may help control the progression of autoimmune diabetes by early colonization of the intestinal tract with A. muciniphila [140,141].
6.3. Effects of SCFAs on T2D
SCFAs are metabolites of gut microbe fermentation that result from indigestible dietary fiber and may have a beneficial role in T2D [142]. Compared to normal animals, diabetic rodents that consumed a high-fat diet with streptozotocin showed lower levels of acetate, propionate, and butyrate [143,144]. It was found that T2D patients had lower fecal butyrate and propionate concentrations, as well as acetate concentrations, than healthy subjects [145]. Improved insulin secretion/sensitivity, reduced fat accumulation, intestinal gluconeogenesis (IGN) triggering, and inflammation are the mechanisms by which SCFAs can positively affect T2D (Figure 3) [70,146]. A study using homeostatic model assessment of insulin resistance (HOMA-IR) observed an adverse correlation between blood insulin levels and total SCFAs, including acetate and propionate [147]. In vitro and in vivo studies showed that propionate can enhance the release of glucose-stimulated insulin, sustain β-cell mass by decreasing trans-differentiation in α-cells, obstruct apoptosis, and assist in proliferation [70,148]. Moreover, it was shown in mouse models that butyrate improved insulin sensitivity [116,149]. These mechanisms support energy consumption and boost mitochondrial functions [116].
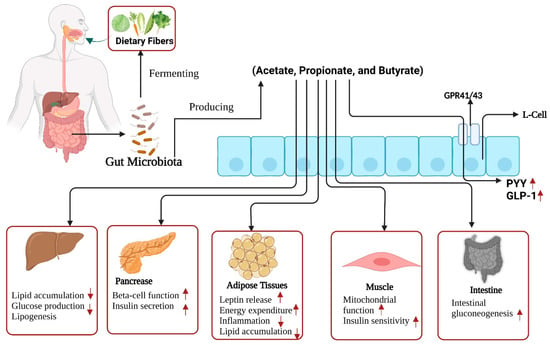
Figure 3.
Gut microbiota ferments dietary fiber and produces short-chain fatty acids (SCFAs) acetate, propionate, and butyrate. These SCFAs may facilitate the production/release of GLP-1 and PYY from enteroendocrine (L-cells) and activate adipose tissue to release leptin hormone. These fatty acids also enhance insulin sensitivity and mitochondrial functions in muscle cells, promote pancreatic functions, including insulin secretion and beta cell activity, and promote intestinal gluconeogenesis. In the liver, lipid accumulation, and glucose production are reduced. Arrows pointing upwards indicate an increase, and arrows pointing downwards indicate a decrease. Abbreviations: PYY; peptide YY: GLP-1; glucagon-like peptide-1: GPR-41; G-protein receptors-41: GPR-43; G-protein receptors-43.
Propionate- or butyrate-induced IGN affects glucose homeostasis, the cAMP-dependent pathway, and the gut–brain neural circuit [150]. Acetate enhances the suppression of lipogenesis in the liver and decreases lipid aggregation in adipose tissues, while glucose transporter-4 genes and myoglobin are enhanced in the abdominal muscles of diabetic rats [23]. The peroxisome proliferator-activated receptor-α (PPAR-α) gene was upregulated in the presence of acetate, which may suppress body fat aggregation [151,152,153]. Furthermore, SCFA supplementation reduces hepatic steatosis and body weight [154]. In vitro and in vivo models showed that SCFAs either enhance the release of peptide YY (PYY) and GLP-1 from L-cells (entero-endocrine), or promote the release of leptin hormone satiation in adipose tissues through G-protein receptors (GPR-41 and/or GPR-43) [155,156,157,158]. SCFAs promote lipid oxidation and energy consumption and were found to increase fasting fat oxidation and PYY concentration during colonic infusion in obese subjects [159]. Butyrate may weaken inflammation generated by the interaction of macrophages and adipocytes by decreasing lipolysis and obstructing inflammatory signals [160]. These fatty acids showed beneficial effects on T2D by reducing the production of TNK-α, IL-6, and monocyte chemoattractant protein-1 (MCP-1); nuclear factor kappa-B (NF-κB) activity was also constrained. Propionate had a positive influence on T2D, participating in the downregulation of inflammatory chemokines and cytokines, such as CC chemokine ligand-5 (CCL-5) and TNF-α [161].
7. Fast Foods
Fast foods are a commercial term used to describe foods sold in restaurants and stores that contain frozen, pre-cooked, or pre-heated ingredients and are sold as takeout [162]. Fast-food consumption is associated with higher energy, fat, sodium, and sugar intake, along with a lower intake of fruits, vegetables, and fibers [163]. Fast foods also tend to have higher energy density and lower nutritional quality compared to home-cooked meals and recommended diets [164,165]. China is one of the most populous countries, and the consumption of fast food is increasing day by day. As a result of the rapid growth of the fast-food industry and fast-food consumption in China, public health concerns have arisen about adverse health effects, such as obesity [166,167].
7.1. Effects of Fast Foods on Gut Microbiota and SCFAs
Fast foods contain low dietary fiber and high fats, which negatively influence the gut microbiota. The composition of gut microbiota is also affected by the quantity and quality of dietary fats [168]. In addition, fast foods are one of the main sources of toxic heavy metals in humans, especially children [169]. The non-essential metals chromium (Cr), cadmium (Cd), nickel (Ni), and lead (Pb) are toxic when they bioaccumulate in tissues and cause inflammation and other effects [170]. In mice that were fed high-fat, low-fiber diets, Bacteroides were less likely to develop and Firmicutes and Proteobacteria were more likely to develop [171]. The gut microbiota plays a very important role in food absorption and mild inflammation, contributing to the development of obesity and diabetes mellitus. Several metabolic pathways are influenced by gut microbiota metabolites (SCFAs), including insulin signaling, incretin production, and inflammation [172,173,174]. In general, fast-food products tend to contain large amounts of manufactured trans-fatty acids, and in people with diabetes, particularly those who eat a diet with high trans-fatty acids, more pro-inflammatory molecules are produced [175,176,177].
Cooking or heat treatment can significantly change the composition and structure of food; in fact, the physicochemical properties of food can be altered by heat, which can degrade antimicrobial compounds [178]. The heating process leads to the production of new compounds, some of which has prebiotic properties and affects the composition of gut bacteria. For instance, a relative decrease in bacterial groups such as Lactobacillus, Bifidobacterium, Akkermansia, Parasutterella, Barnesiella Dorea, Oscillibacter, and Alistipes was observed in animals fed with melanoidin-enriched malt [174,179]. Thus, the amount of fiber is affected by the consumption of fast food, and the number of gut microbes decreases, leading to a decrease in SCFAs.
7.2. Effect of Fast Foods on Diabetes
The term “junk food” refers to a variety of processed foods, fast foods, and ready-made snacks. Fast foods, which are heavily processed, have an adverse effect on health. Eating fast food and eating out are major risk factors in terms of poor diet quality, higher calories, fat intake, and lower dietary micronutrient density [180]. Currently, there are no government policies controlling fast-food pricing or advertising in some countries, leading to the opening of new global chains. Eating fast food twice a week has been shown to increase the risk of insulin resistance and T2D [181]. Obesity, abdominal fat gain, lipid and lipoprotein disorders, impaired insulin, glucose homeostasis, systemic inflammation, as well as oxidative stress, have been associated with frequent fast-food consumption [182]. A positive correlation between fast-food restaurants and the prevalence of diabetes was found in all counties except those with high poverty or middle minority populations [183]. A previous study showed that consuming excess calories shortens the lifespan, while moderate calorie restriction slows the aging process and protects the body and brain from age-related damage [184]. T2D is strongly associated with overweight and obesity. Animal studies have shown that nitrosamines in fast food are toxic to beta cells and increase the risk of T2D [185]. A previous study found that primiparous women who ate more fast food before pregnancy had an increased risk of developing diabetes during pregnancy and giving birth to a child with low birth weight [186]. Several chronic metabolic disorders may develop due to the consumption of fast foods, including hyperglycemia, glycosuria, hyperlipemia, negative nitrogen balance, and sometimes ketonemia, and junk food consumption causes over 90% of T2D cases [187].
8. Conclusions and Future Perspectives
Promoting health and preventing disease rely on maintaining a balance between the intestinal microbiome, genetic factors, environmental factors, and dietary conditions that affect substrate availability. Intestinal bacteria produce SCFAs as metabolites, and their concentration depends on the composition and the population size of these microorganisms. In addition to their effects on digestion, many studies have investigated how SCFAs produced by intestinal microbiomes affect organs and tissues elsewhere in the body. In addition, regular consumption of fast food negatively affects the production of SCFAs, and GM survival is responsible for SCFA production. Dietary fiber, as an essential component of foods, can be classified into soluble and insoluble forms. Soluble dietary fiber is resistant to gut-digesting enzymes and fermented by the AIM, resulting in the production of SCFAs (acetate, propionate, and butyrate).
In this review, the effectiveness of dietary fiber in producing SCFAs in the colon by the AIM as well as its health-promoting effects on T2D patients were discussed. Several pathways lead to the production of acetate, propionate, and butyrate, including the Wood–Ljungdahl pathway, the succinate decarboxylation pathway, and the acrylate pathway. Both in vitro and in vivo studies have demonstrated that SCFAs increase the release of PYY and GLP-1 from L-cells and promote leptin hormone satiation in adipose tissue via G-protein receptors such as GPR (41 and 43). SCFAs have been shown to have beneficial effects on T2D by reducing the production of TNK-α, IL-6, MCP-1, and NF-κB; and propionate has a positive impact on T2D through the downregulation of inflammatory chemokines and cytokines such as CCL-5 and TNF-α. These combined effects lead to a positive influence on T2D. In the future, the effectiveness of GM intervention in T2D will be verified by clinical trials, and the advantages will be explored. There is an urgent need for research in this area for human populations.
Author Contributions
Data Curation, Validation, Writing—Original Draft, Investigation, M.M.; Investigation, Resources, Supervision, Conceptualization, Project Administration, Funding Acquisition, L.Q.; Investigation, Conceptualization, Resources, Writing—Review and Editing, Funding Acquisition, Y.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This review was supported by Guizhou Science and Technology Program Qian Ke He Ping Tai Ren Cai [2018] 5254, [2018] 5781, Qian Ke Zhong Yin Di [2022] 4033, Qian Ke He Zhicheng [2022] Zhongdian 007, Qian Ke He Zhicheng [2021] yiban175, Qian Ke He Jichu [2019]1071, Qian Ke He Zhicheng [2020] 1Y171), Establishment of Guizhou Modern Agriculture Industry Technical System (Characteristic Coarse Cereals) Qian Cai Nong [2018]81 and Key Agricultural Technology Extension Project of Guizhou Province ([2017] 106).
Institutional Review Board Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.
Acknowledgments
Thanks to Biorender software for supporting figure formation (app.biorender.com accessed on 1 July 2022).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. The impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef] [PubMed]
- Kreznar, J.H.; Keller, M.P.; Traeger, L.L.; Rabaglia, M.E.; Schueler, K.L.; Stapleton, D.S.; Zhao, W.; Vivas, E.I.; Yandell, B.S.; Broman, A.T. Host genotype and gut microbiome modulate insulin secretion and diet-induced metabolic phenotypes. Cell Rep. 2017, 18, 1739–1750. [Google Scholar] [CrossRef]
- Federici, M. Gut microbiome and microbial metabolites: A new system affecting metabolic disorders. J. Endocrinol. Investig. 2019, 42, 1011–1018. [Google Scholar] [CrossRef]
- Rothschild, D.; Weissbrod, O.; Barkan, E.; Kurilshikov, A.; Korem, T.; Zeevi, D.; Costea, P.I.; Godneva, A.; Kalka, I.N.; Bar, N. Environment dominates over host genetics in shaping human gut microbiota. Nature 2018, 555, 210–215. [Google Scholar] [CrossRef]
- Joseph, N.; Vasodavan, K.; Saipudin, N.A.; Yusof, B.N.M.; Kumar, S.; Nordin, S.A. Gut microbiota and short-chain fatty acids (SCFAs) profiles of normal and overweight school children in Selangor after probiotics administration. J. Funct. Foods 2019, 57, 103–111. [Google Scholar] [CrossRef]
- Eastwood, M.A.; Kay, R.M. An hypothesis for the action of dietary fiber along the gastrointestinal tract. Am. J. Clin. Nutr. 1979, 32, 364–367. [Google Scholar] [CrossRef]
- Soliman, G.A. Dietary fiber, atherosclerosis, and cardiovascular disease. Nutrients 2019, 11, 1155. [Google Scholar] [CrossRef]
- Harvey, R.A.; Ferrier, D.R. Biochemistry; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2011. [Google Scholar]
- Yousafzai, M.U.R.; Nangarhar, A. Dietary fibers and their effects on health. Res. Rev. Int. J. Multidiscip. 2021, 06, 35–42. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Xue, X.; Lu, C.; Ma, J. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 2018, 359, 1151–1156. [Google Scholar] [CrossRef]
- Cai, X.; Yu, H.; Liu, L.; Lu, T.; Li, J.; Ji, Y.; Le, Z.; Bao, L.; Ma, W.; Xiao, R. Milk Powder Co-Supplemented with Inulin and Resistant Dextrin Improves Glycemic Control and Insulin Resistance in Elderly Type 2 Diabetes Mellitus: A 12-Week Randomized, Double-Blind, Placebo-Controlled Trial. Mol. Nutr. Food Res. 2018, 62, 1800865. [Google Scholar] [CrossRef]
- Partula, V.; Deschasaux, M.; Druesne-Pecollo, N.; Latino-Martel, P.; Desmetz, E.; Chazelas, E.; Kesse-Guyot, E.; Julia, C.; Fezeu, L.K.; Galan, P. Associations between consumption of dietary fibers and the risk of cardiovascular diseases, cancers, type 2 diabetes, and mortality in the prospective NutriNet-Santé cohort. Am. J. Clin. Nutr. 2020, 112, 195–207. [Google Scholar] [CrossRef]
- Kimura, Y.; Yoshida, D.; Hirakawa, Y.; Hata, J.; Honda, T.; Shibata, M.; Sakata, S.; Uchida, K.; Kitazono, T.; Ninomiya, T. Dietary fiber intake and risk of type 2 diabetes in a general Japanese population: The Hisayama Study. J. Diabetes Investig. 2021, 12, 527–536. [Google Scholar] [CrossRef]
- Muhammad, M.; Zhu, Y.; Wen, A.; Liu, N.; Qin, L. Phenolic profile, alpha-amylase inhibitory activity, and in vitro glycemic index of adzuki beans. Front. Nutr. 2022, 9, 1063602. [Google Scholar] [CrossRef]
- Xu, S.; Qin, L.; Mazhar, M.; Zhu, Y. Functional components profile and glycemic index of kidney beans. Front. Nutr. 2022, 9, 1044427. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific opinion on dietary reference values for carbohydrates and dietary fibre. EFSA J. 2010, 8, 1462. [Google Scholar]
- Nishida, C.; Uauy, R.; Kumanyika, S.; Shetty, P. The joint WHO/FAO expert consultation on diet, nutrition and the prevention of chronic diseases: Process, product and policy implications. Public Health Nutr. 2004, 7, 245–250. [Google Scholar] [CrossRef]
- Nie, Q.; Hu, J.; Chen, H.; Geng, F.; Nie, S. Arabinoxylan ameliorates type 2 diabetes by regulating the gut microbiota and metabolites. Food Chem. 2022, 371, 131106. [Google Scholar] [CrossRef]
- Markowiak-Kopeć, P.; Śliżewska, K. The effect of probiotics on the production of short-chain fatty acids by human intestinal microbiome. Nutrients 2020, 12, 1107. [Google Scholar] [CrossRef]
- Tazoe, H.; Otomo, Y.; Kaji, I.; Tanaka, R.; Karaki, S.; Kuwahara, A. Roles of short-chain fatty acids receptors, GPR41 and GPR43 on colonic functions. J. Physiol. Pharmacol. 2008, 59, 251–262. [Google Scholar]
- Galisteo, M.; Duarte, J.; Zarzuelo, A. Effects of dietary fibers on disturbances clustered in the metabolic syndrome. J. Nutr. Biochem. 2008, 19, 71–84. [Google Scholar] [CrossRef]
- Saboo, B.; Misra, A.; Kalra, S.; Mohan, V.; Aravind, S.; Joshi, S.; Chowdhury, S.; Sahay, R.; Kesavadev, J.; John, M. Role and importance of high fiber in diabetes management in India. Diabetes Metab. Syndr. Clin. Res. Rev. 2022, 16, 102480. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Hung, E.S.; Cullum, A.; Allen, R.E.; Aggett, P.J.; Dyson, P.; Forouhi, N.G.; Greenwood, D.C.; Pryke, R.; Taylor, R. Lower carbohydrate diets for adults with type 2 diabetes. Br. J. Nutr. 2022, 127, 1352–1357. [Google Scholar] [CrossRef] [PubMed]
- Houghton, D.; Hardy, T.; Stewart, C.; Errington, L.; Day, C.P.; Trenell, M.I.; Avery, L. Systematic review assessing the effectiveness of dietary intervention on gut microbiota in adults with type 2 diabetes. Diabetologia 2018, 61, 1700–1711. [Google Scholar] [CrossRef] [PubMed]
- Leylabadlo, H.E.; Sanaie, S.; Heravi, F.S.; Ahmadian, Z.; Ghotaslou, R. From role of gut microbiota to microbial-based therapies in type 2-diabetes. Infect. Genet. Evol. 2020, 81, 104268. [Google Scholar] [CrossRef]
- Deehan, E.C.; Zhang, Z.; Riva, A.; Armet, A.M.; Perez-Muñoz, M.E.; Nguyen, N.K.; Krysa, J.A.; Seethaler, B.; Zhao, Y.-Y.; Cole, J. Elucidating the role of the gut microbiota in the physiological effects of dietary fiber. Microbiome 2022, 10, 77. [Google Scholar] [CrossRef]
- Beam, A.; Clinger, E.; Hao, L. Effect of diet and dietary components on the composition of the gut microbiota. Nutrients 2021, 13, 2795. [Google Scholar] [CrossRef]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Bäckhed, F. From dietary fiber to host physiology: Short-chain fatty acids as key bacterial metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef]
- So, D.; Whelan, K.; Rossi, M.; Morrison, M.; Holtmann, G.; Kelly, J.T.; Shanahan, E.R.; Staudacher, H.M.; Campbell, K.L. Dietary fiber intervention on gut microbiota composition in healthy adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2018, 107, 965–983. [Google Scholar] [CrossRef]
- Claesson, M.J.; Jeffery, I.B.; Conde, S.; Power, S.E.; O’connor, E.M.; Cusack, S.; Harris, H.; Coakley, M.; Lakshminarayanan, B.; O’sullivan, O. Gut microbiota composition correlates with diet and health in the elderly. Nature 2012, 488, 178–184. [Google Scholar] [CrossRef]
- Sheflin, A.M.; Borresen, E.C.; Kirkwood, J.S.; Boot, C.M.; Whitney, A.K.; Lu, S.; Brown, R.J.; Broeckling, C.D.; Ryan, E.P.; Weir, T.L. Dietary supplementation with rice bran or navy bean alters gut bacterial metabolism in colorectal cancer survivors. Mol. Nutr. Food Res. 2017, 61, 1500905. [Google Scholar] [CrossRef]
- Kopf, J.C.; Suhr, M.J.; Clarke, J.; Eyun, S.-I.; Riethoven, J.-J.M.; Ramer-Tait, A.E.; Rose, D.J. Role of whole grains versus fruits and vegetables in reducing subclinical inflammation and promoting gastrointestinal health in individuals affected by overweight and obesity: A randomized controlled trial. Nutr. J. 2018, 17, 72. [Google Scholar] [CrossRef]
- Jang, S.-E.; Kim, K.-A.; Han, M.J.; Kim, D.-H. Doenjang, a fermented Korean soybean paste, inhibits lipopolysaccharide production of gut microbiota in mice. J. Med. Food 2014, 17, 67–75. [Google Scholar] [CrossRef]
- Roberfroid, M.B. Introducing inulin-type fructans. Br. J. Nutr. 2005, 93, S13–S25. [Google Scholar] [CrossRef]
- Roberfroid, M. Inulin and oligofructose: Health benefits and claims–a critical review. J. Nutr. 2007, 137, 2493S–2502S. [Google Scholar] [CrossRef]
- Birkeland, E.; Gharagozlian, S.; Birkeland, K.I.; Valeur, J.; Måge, I.; Rud, I.; Aas, A.-M. Prebiotic effect of inulin-type fructans on faecal microbiota and short-chain fatty acids in type 2 diabetes: A randomised controlled trial. Eur. J. Nutr. 2020, 59, 3325–3338. [Google Scholar] [CrossRef]
- Man, S.; Liu, T.; Yao, Y.; Lu, Y.; Ma, L.; Lu, F. Friend or foe? The roles of inulin-type fructans. Carbohydr. Polym. 2021, 252, 117155. [Google Scholar] [CrossRef]
- Agus, A.; Clément, K.; Sokol, H. Gut microbiota-derived metabolites as central regulators in metabolic disorders. Gut 2021, 70, 1174–1182. [Google Scholar] [CrossRef]
- Dehghan, P.; Gargari, B.P.; Jafar-Abadi, M.A.; Aliasgharzadeh, A. Inulin controls inflammation and metabolic endotoxemia in women with type 2 diabetes mellitus: A randomized-controlled clinical trial. Int. J. Food Sci. Nutr. 2014, 65, 117–123. [Google Scholar] [CrossRef]
- Dehghan, P.; Gargari, B.P.; Jafar-Abadi, M.A. Oligofructose-enriched inulin improves some inflammatory markers and metabolic endotoxemia in women with type 2 diabetes mellitus: A randomized controlled clinical trial. Nutrition 2014, 30, 418–423. [Google Scholar] [CrossRef]
- Farooq, U.; Di Mattia, C.; Faieta, M.; Flamminii, F.; Pittia, P. Colloidal properties and stability of olive oil-in water emulsions stabilized by starch particles. Ital. J. Food Sci. 2021, 33, 1–10. [Google Scholar] [CrossRef]
- Keskin, S.O.; Ali, T.M.; Ahmed, J.; Shaikh, M.; Siddiq, M.; Uebersax, M.A. Physico-chemical and functional properties of legume protein, starch, and dietary fiber—A review. Legume Sci. 2022, 4, e117. [Google Scholar] [CrossRef]
- Tan, L.; Kong, L. Starch-guest inclusion complexes: Formation, structure, and enzymatic digestion. Crit. Rev. Food Sci. Nutr. 2020, 60, 780–790. [Google Scholar] [CrossRef]
- Tian, S.; Sun, Y. Influencing factor of resistant starch formation and application in cereal products: A review. Int. J. Biol. Macromol. 2020, 149, 424–431. [Google Scholar] [CrossRef]
- Englyst, H.N.; Kingman, S.M.; Cummings, J. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 46, S33–S50. [Google Scholar]
- Englyst, K.; Goux, A.; Meynier, A.; Quigley, M.; Englyst, H.; Brack, O.; Vinoy, S. Inter-laboratory validation of the starch digestibility method for determination of rapidly digestible and slowly digestible starch. Food Chem. 2018, 245, 1183–1189. [Google Scholar] [CrossRef]
- DeMartino, P.; Cockburn, D.W. Resistant starch: Impact on the gut microbiome and health. Curr. Opin. Biotechnol. 2020, 61, 66–71. [Google Scholar] [CrossRef]
- Dobranowski, P.A.; Stintzi, A. Resistant starch, microbiome, and precision modulation. Gut Microbes 2021, 13, 1926842. [Google Scholar] [CrossRef]
- Tan, F.P.; Beltranena, E.; Zijlstra, R.T. Resistant starch: Implications of dietary inclusion on gut health and growth in pigs: A review. J. Anim. Sci. Biotechnol. 2021, 12, 124. [Google Scholar] [CrossRef]
- Englyst, H.; Wiggins, H.S.; Cummings, J. Determination of the non-starch polysaccharides in plant foods by gas-liquid chromatography of constituent sugars as alditol acetates. Analyst 1982, 107, 307–318. [Google Scholar] [CrossRef]
- Bendiks, Z.A.; Knudsen, K.E.; Keenan, M.J.; Marco, M.L. Conserved and variable responses of the gut microbiome to resistant starch type 2. Nutr. Res. 2020, 77, 12–28. [Google Scholar] [CrossRef]
- Byrne, C.; Chambers, E.; Morrison, D.; Frost, G. The role of short chain fatty acids in appetite regulation and energy homeostasis. Int. J. Obes. 2015, 39, 1331–1338. [Google Scholar] [CrossRef]
- Cerqueira, F.M.; Photenhauer, A.L.; Pollet, R.M.; Brown, H.A.; Koropatkin, N.M. Starch digestion by gut bacteria: Crowdsourcing for carbs. Trends Microbiol. 2020, 28, 95–108. [Google Scholar] [CrossRef]
- Coppola, S.; Avagliano, C.; Calignano, A.; Berni Canani, R. The protective role of butyrate against obesity and obesity-related diseases. Molecules 2021, 26, 682. [Google Scholar] [CrossRef]
- Harris, K.F. An introductory review of resistant starch type 2 from high-amylose cereal grains and its effect on glucose and insulin homeostasis. Nutr. Rev. 2019, 77, 748–764. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, M.; Ma, Q.; Tian, B.; Nie, C.; Chen, Z.; Li, J. Health beneficial effects of resistant starch on diabetes and obesity via regulation of gut microbiota: A review. Food Funct. 2020, 11, 5749–5767. [Google Scholar] [CrossRef]
- Xia, W.; Zhang, K.; Su, L.; Wu, J. Microbial starch debranching enzymes: Developments and applications. Biotechnol. Adv. 2021, 50, 107786. [Google Scholar] [CrossRef]
- Kaur, A.; Chen, T.; Green, S.J.; Mutlu, E.; Martin, B.R.; Rumpagaporn, P.; Patterson, J.A.; Keshavarzian, A.; Hamaker, B.R. Physical inaccessibility of a resistant starch shifts mouse gut microbiota to butyrogenic firmicutes. Mol. Nutr. Food Res. 2019, 63, 1801012. [Google Scholar] [CrossRef]
- Patel, H.; Royall, P.G.; Gaisford, S.; Williams, G.R.; Edwards, C.H.; Warren, F.J.; Flanagan, B.M.; Ellis, P.R.; Butterworth, P.J. Structural and enzyme kinetic studies of retrograded starch: Inhibition of α-amylase and consequences for intestinal digestion of starch. Carbohydr. Polym. 2017, 164, 154–161. [Google Scholar] [CrossRef]
- Zia-ud-Din; Xiong, H.; Fei, P. Physical and chemical modification of starches: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 2691–2705. [Google Scholar] [CrossRef]
- Hasjim, J.; Lee, S.-O.; Hendrich, S.; Setiawan, S.; Ai, Y.; Jane, J.-l. Molecular Diversity and Health Benefits of Carbohydrates from Cereals and Pulses. Cereal Chem. 2010, 87, 257–262. [Google Scholar] [CrossRef]
- Deehan, E.C.; Yang, C.; Perez-Muñoz, M.E.; Nguyen, N.K.; Cheng, C.C.; Triador, L.; Zhang, Z.; Bakal, J.A.; Walter, J. Precision microbiome modulation with discrete dietary fiber structures directs short-chain fatty acid production. Cell Host Microbe 2020, 27, 389–404.e6. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Du, C.; Jiang, W.; Wang, L.; Du, S.-K. The preparation, formation, fermentability, and applications of resistant starch. Int. J. Biol. Macromol. 2020, 150, 1155–1161. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.N.; Yao, Y.; Ju, S.Y. Short chain fatty acids and fecal microbiota abundance in humans with obesity: A systematic review and meta-analysis. Nutrients 2019, 11, 2512. [Google Scholar] [CrossRef] [PubMed]
- McLoughlin, R.F.; Berthon, B.S.; Jensen, M.E.; Baines, K.J.; Wood, L.G. Short-chain fatty acids, prebiotics, synbiotics, and systemic inflammation: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2017, 106, 930–945. [Google Scholar] [CrossRef] [PubMed]
- Metzler-Zebeli, B.; Canibe, N.; Montagne, L.; Freire, J.; Bosi, P.; Prates, J.A.; Tanghe, S.; Trevisi, P. Resistant starch reduces large intestinal pH and promotes fecal lactobacilli and bifidobacteria in pigs. Animal 2019, 13, 64–73. [Google Scholar] [CrossRef]
- González Hernández, M.A.; Canfora, E.E.; Jocken, J.W.; Blaak, E.E. The short-chain fatty acid acetate in body weight control and insulin sensitivity. Nutrients 2019, 11, 1943. [Google Scholar] [CrossRef]
- Huang, H.-W.; Hsu, C.-P.; Wang, C.-Y. Healthy expectations of high hydrostatic pressure treatment in food processing industry. J. Food Drug Anal. 2020, 28, 1–13. [Google Scholar] [CrossRef]
- Mandaliya, D.K.; Seshadri, S. Short Chain Fatty Acids, pancreatic dysfunction and type 2 diabetes. Pancreatology 2019, 19, 617–622. [Google Scholar] [CrossRef]
- Roy, R.; Nguyen-Ngo, C.; Lappas, M. Short-chain fatty acids as novel therapeutics for gestational diabetes. J. Mol. Endocrinol. 2020, 65, 21–34. [Google Scholar] [CrossRef]
- Esser, N.; Legrand-Poels, S.; Piette, J.; Scheen, A.J.; Paquot, N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res. Clin. Pract. 2014, 105, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Creely, S.J.; McTernan, P.G.; Kusminski, C.M.; Fisher, F.M.; Da Silva, N.; Khanolkar, M.; Evans, M.; Harte, A.; Kumar, S. Lipopolysaccharide activates an innate immune system response in human adipose tissue in obesity and type 2 diabetes. Am. J. Physiol.-Endocrinol. Metab. 2007, 292, E740–E747. [Google Scholar] [CrossRef] [PubMed]
- Pedro, M.N.; Magro, D.O.; da Silva, E.U.P.P.; Guadagnini, D.; Santos, A.; de Jesus Pedro, R.; Saad, M.J.A. Plasma levels of lipopolysaccharide correlate with insulin resistance in HIV patients. Diabetol. Metab. Syndr. 2018, 10, 5. [Google Scholar] [CrossRef] [PubMed]
- Peraldi, P.; Hotamisligil, G.S.; Buurman, W.A.; White, M.F.; Spiegelman, B.M. Tumor necrosis factor (TNF)-α inhibits insulin signaling through stimulation of the p55 TNF receptor and activation of sphingomyelinase. J. Biol. Chem. 1996, 271, 13018–13022. [Google Scholar] [CrossRef]
- Copps, K.D.; Hancer, N.J.; Opare-Ado, L.; Qiu, W.; Walsh, C.; White, M.F. Irs1 serine 307 promotes insulin sensitivity in mice. Cell Metab. 2010, 11, 84–92. [Google Scholar] [CrossRef]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar] [CrossRef]
- Bodinham, C.; Smith, L.; Thomas, E.L.; Bell, J.D.; Swann, J.R.; Costabile, A.; Russell-Jones, D.; Umpleby, A.M.; Robertson, M.D. Efficacy of increased resistant starch consumption in human type 2 diabetes. Endocr. Connect. 2014, 3, 75–84. [Google Scholar] [CrossRef]
- Laughlin, M.R. Normal roles for dietary fructose in carbohydrate metabolism. Nutrients 2014, 6, 3117–3129. [Google Scholar] [CrossRef]
- Taskinen, M.-R.; Packard, C.J.; Borén, J. Dietary fructose and the metabolic syndrome. Nutrients 2019, 11, 1987. [Google Scholar] [CrossRef]
- Teff, K.L.; Elliott, S.S.; Tschöp, M.; Kieffer, T.J.; Rader, D.; Heiman, M.; Townsend, R.R.; Keim, N.L.; D’alessio, D.; Havel, P.J. Dietary fructose reduces circulating insulin and leptin, attenuates postprandial suppression of ghrelin, and increases triglycerides in women. J. Clin. Endocrinol. Metab. 2004, 89, 2963–2972. [Google Scholar] [CrossRef]
- Segal, M.S.; Gollub, E.; Johnson, R.J. Is the fructose index more relevant with regards to cardiovascular disease than the glycemic index? Eur. J. Nutr. 2007, 46, 406–417. [Google Scholar] [CrossRef] [PubMed]
- Sievenpiper, J.L.; de Souza, R.J.; Cozma, A.I.; Chiavaroli, L.; Ha, V.; Mirrahimi, A. Fructose vs. glucose and metabolism: Do the metabolic differences matter? Curr. Opin. Lipidol. 2014, 25, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Cozma, A.I.; Sievenpiper, J.L.; De Souza, R.J.; Chiavaroli, L.; Ha, V.; Wang, D.D.; Mirrahimi, A.; Yu, M.E.; Carleton, A.J.; Di Buono, M. Effect of fructose on glycemic control in diabetes: A systematic review and meta-analysis of controlled feeding trials. Diabetes Care 2012, 35, 1611–1620. [Google Scholar] [CrossRef] [PubMed]
- Kolderup, A.; Svihus, B. Fructose metabolism and relation to atherosclerosis, type 2 diabetes, and obesity. J. Nutr. Metab. 2015, 2015, 823081. [Google Scholar] [CrossRef]
- de Farias Lelis, D.; Andrade, J.M.O.; Almenara, C.C.P.; Broseguini-Filho, G.B.; Mill, J.G.; Baldo, M.P. High fructose intake and the route towards cardiometabolic diseases. Life Sci. 2020, 259, 118235. [Google Scholar] [CrossRef] [PubMed]
- Hannou, S.A.; Haslam, D.E.; McKeown, N.M.; Herman, M.A. Fructose metabolism and metabolic disease. J. Clin. Investig. 2018, 128, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Havel, P.J. Dietary fructose: Implications for dysregulation of energy homeostasis and lipid/carbohydrate metabolism. Nutr. Rev. 2005, 63, 133–157. [Google Scholar] [CrossRef] [PubMed]
- Hawkesworth, S.; Dangour, A.D.; Johnston, D.; Lock, K.; Poole, N.; Rushton, J.; Uauy, R.; Waage, J. Feeding the world healthily: The challenge of measuring the effects of agriculture on health. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 3083–3097. [Google Scholar] [CrossRef] [PubMed]
- Kearney, J. Food consumption trends and drivers. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2793–2807. [Google Scholar] [CrossRef]
- Machate, D.J.; Figueiredo, P.S.; Marcelino, G.; Guimarães, R.d.C.A.; Hiane, P.A.; Bogo, D.; Pinheiro, V.A.Z.; Oliveira, L.C.S.d.; Pott, A. Fatty acid diets: Regulation of gut microbiota composition and obesity and its related metabolic dysbiosis. Int. J. Mol. Sci. 2020, 21, 4093. [Google Scholar] [CrossRef]
- Portincasa, P.; Bonfrate, L.; Vacca, M.; De Angelis, M.; Farella, I.; Lanza, E.; Khalil, M.; Wang, D.Q.-H.; Sperandio, M.; Di Ciaula, A. Gut microbiota and short chain fatty acids: Implications in glucose homeostasis. Int. J. Mol. Sci. 2022, 23, 1105. [Google Scholar] [CrossRef] [PubMed]
- Ríos-Covián, D.; Ruas-Madiedo, P.; Margolles, A.; Gueimonde, M.; De Los Reyes-gavilán, C.G.; Salazar, N. Intestinal short chain fatty acids and their link with diet and human health. Front. Microbiol. 2016, 7, 185. [Google Scholar] [CrossRef] [PubMed]
- Morrison, D.J.; Preston, T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 2016, 7, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Miller, T.L.; Wolin, M.J. Pathways of acetate, propionate, and butyrate formation by the human fecal microbial flora. Appl. Environ. Microbiol. 1996, 62, 1589–1592. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Links between diet, gut microbiota composition and gut metabolism. Proc. Nutr. Soc. 2015, 74, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Havenaar, R. Intestinal health functions of colonic microbial metabolites: A review. Benef. Microbes 2011, 2, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Robertson, M.D. Prebiotics and type 2 diabetes: Targeting the gut microbiota for improved glycaemic control? Pract. Diabetes 2020, 37, 133–137. [Google Scholar] [CrossRef]
- Karlsson, F.H.; Tremaroli, V.; Nookaew, I.; Bergström, G.; Behre, C.J.; Fagerberg, B.; Nielsen, J.; Bäckhed, F. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature 2013, 498, 99–103. [Google Scholar] [CrossRef]
- Li, Q.; Chang, Y.; Zhang, K.; Chen, H.; Tao, S.; Zhang, Z. Implication of the gut microbiome composition of type 2 diabetic patients from northern China. Scientific reports 2020, 10, 5450. [Google Scholar] [CrossRef]
- Reichardt, N.; Duncan, S.H.; Young, P.; Belenguer, A.; McWilliam Leitch, C.; Scott, K.P.; Flint, H.J.; Louis, P. Phylogenetic distribution of three pathways for propionate production within the human gut microbiota. ISME J. 2014, 8, 1323–1335. [Google Scholar] [CrossRef]
- Vital, M.; Howe, A.C.; Tiedje, J.M. Revealing the bacterial butyrate synthesis pathways by analyzing (meta) genomic data. MBio 2014, 5, e00889-00814. [Google Scholar] [CrossRef] [PubMed]
- Serpa, J.; Caiado, F.; Carvalho, T.; Torre, C.; Gonçalves, L.G.; Casalou, C.; Lamosa, P.; Rodrigues, M.; Zhu, Z.; Lam, E.W. Butyrate-rich colonic microenvironment is a relevant selection factor for metabolically adapted tumor cells. J. Biol. Chem. 2010, 285, 39211–39223. [Google Scholar] [CrossRef] [PubMed]
- Roy, C.C.; Kien, C.L.; Bouthillier, L.; Levy, E. Short-chain fatty acids: Ready for prime time? Nutr. Clin. Pract. 2006, 21, 351–366. [Google Scholar] [CrossRef] [PubMed]
- Brunkwall, L.; Orho-Melander, M. The gut microbiome as a target for prevention and treatment of hyperglycaemia in type 2 diabetes: From current human evidence to future possibilities. Diabetologia 2017, 60, 943–951. [Google Scholar] [CrossRef]
- Qin, J.; Li, Y.; Cai, Z.; Li, S.; Zhu, J.; Zhang, F.; Liang, S.; Zhang, W.; Guan, Y.; Shen, D. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 2012, 490, 55–60. [Google Scholar] [CrossRef]
- Jia, L.; Li, D.; Feng, N.; Shamoon, M.; Sun, Z.; Ding, L.; Zhang, H.; Chen, W.; Sun, J.; Chen, Y.Q. Anti-diabetic effects of Clostridium butyricum CGMCC0313. 1 through promoting the growth of gut butyrate-producing bacteria in type 2 diabetic mice. Sci. Rep. 2017, 7, 7046. [Google Scholar] [CrossRef]
- Patel, S.; Mandaliya, D.; Prajapati, B.; Kumar, S.; Seshadri, S. Cefdinir Microsphere modulated microflora and liver immunological response to diet induced diabetes in mice. Endocr. Metab. Immune Disord.-Drug Targets (Former. Curr. Drug Targets-Immune Endocr. Metab. Disord.) 2019, 19, 349–357. [Google Scholar] [CrossRef]
- Jena, P.K.; Singh, S.; Prajapati, B.; Nareshkumar, G.; Mehta, T.; Seshadri, S. Impact of targeted specific antibiotic delivery for gut microbiota modulation on high-fructose-fed rats. Appl. Biochem. Biotechnol. 2014, 172, 3810–3826. [Google Scholar] [CrossRef]
- Fleming, S.; Fitch, M.; DeVries, S.; Liu, M.; Kight, C. Nutrient utilization by cells isolated from rat jejunum, cecum and colon. J. Nutr. 1991, 121, 869–878. [Google Scholar] [CrossRef]
- Brown, A.J.; Goldsworthy, S.M.; Barnes, A.A.; Eilert, M.M.; Tcheang, L.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.; Kinghorn, I.; Fraser, N.J. The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 2003, 278, 11312–11319. [Google Scholar] [CrossRef]
- Itoh, Y.; Kawamata, Y.; Harada, M.; Kobayashi, M.; Fujii, R.; Fukusumi, S.; Ogi, K.; Hosoya, M.; Tanaka, Y.; Uejima, H. Free fatty acids regulate insulin secretion from pancreatic β cells through GPR40. Nature 2003, 422, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Kristinsson, H.; Bergsten, P.; Sargsyan, E. Free fatty acid receptor 1 (FFAR1/GPR40) signaling affects insulin secretion by enhancing mitochondrial respiration during palmitate exposure. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2015, 1853, 3248–3257. [Google Scholar] [CrossRef] [PubMed]
- Hirasawa, A.; Tsumaya, K.; Awaji, T.; Katsuma, S.; Adachi, T.; Yamada, M.; Sugimoto, Y.; Miyazaki, S.; Tsujimoto, G. Free fatty acids regulate gut incretin glucagon-like peptide-1 secretion through GPR120. Nat. Med. 2005, 11, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.V.; Frassetto, A.; Kowalik Jr, E.J.; Nawrocki, A.R.; Lu, M.M.; Kosinski, J.R.; Hubert, J.A.; Szeto, D.; Yao, X.; Forrest, G. Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS ONE 2012, 7, e35240. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Yin, J.; Zhang, J.; Ward, R.E.; Martin, R.J.; Lefevre, M.; Cefalu, W.T.; Ye, J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 2009, 58, 1509–1517. [Google Scholar] [CrossRef]
- Guo, Y.; Xiao, Z.; Wang, Y.; Yao, W.; Liao, S.; Yu, B.; Zhang, J.; Zhang, Y.; Zheng, B.; Ren, B. Sodium butyrate ameliorates streptozotocin-induced type 1 diabetes in mice by inhibiting the HMGB1 expression. Front. Endocrinol. 2018, 9, 630. [Google Scholar] [CrossRef]
- Tang, R.; Li, L. Modulation of short-chain fatty acids as potential therapy method for type 2 diabetes mellitus. Can. J. Infect. Dis. Med. Microbiol. 2021, 2021, 6632266. [Google Scholar] [PubMed]
- Fischbach, M.A.; Sonnenburg, J.L. Eating for two: How metabolism establishes interspecies interactions in the gut. Cell Host Microbe 2011, 10, 336–347. [Google Scholar] [CrossRef]
- Macfarlane, S.; Macfarlane, G.T. Regulation of short-chain fatty acid production. Proc. Nutr. Soc. 2003, 62, 67–72. [Google Scholar] [CrossRef]
- Macy, J.M.; Ljungdahl, L.G.; Gottschalk, G. Pathway of succinate and propionate formation in Bacteroides fragilis. J. Bacteriol. 1978, 134, 84–91. [Google Scholar] [CrossRef]
- Macy, J.M.; Probst, I. The biology of gastrointestinal bacteroides. Annu. Rev. Microbiol. 1979, 33, 561–594. [Google Scholar] [CrossRef] [PubMed]
- Pryde, S.E.; Duncan, S.H.; Hold, G.L.; Stewart, C.S.; Flint, H.J. The microbiology of butyrate formation in the human colon. FEMS Microbiol. Lett. 2002, 217, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Ragsdale, S.W.; Pierce, E. Acetogenesis and the Wood–Ljungdahl pathway of CO2 fixation. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2008, 1784, 1873–1898. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Louis, P.; Flint, H.J. Lactate-utilizing bacteria, isolated from human feces, that produce butyrate as a major fermentation product. Appl. Environ. Microbiol. 2004, 70, 5810–5817. [Google Scholar] [CrossRef]
- Louis, P.; Flint, H.J. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol. Lett. 2009, 294, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Barcenilla, A.; Stewart, C.S.; Pryde, S.E.; Flint, H.J. Acetate utilization and butyryl coenzyme A (CoA): Acetate-CoA transferase in butyrate-producing bacteria from the human large intestine. Appl. Environ. Microbiol. 2002, 68, 5186–5190. [Google Scholar] [CrossRef]
- Venema, K. Role of gut microbiota in the control of energy and carbohydrate metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 432–438. [Google Scholar] [CrossRef]
- Duncan, S.H.; Holtrop, G.; Lobley, G.E.; Calder, A.G.; Stewart, C.S.; Flint, H.J. Contribution of acetate to butyrate formation by human faecal bacteria. Br. J. Nutr. 2004, 91, 915–923. [Google Scholar] [CrossRef]
- Malard, F.; Dore, J.; Gaugler, B.; Mohty, M. Introduction to host microbiome symbiosis in health and disease. Mucosal Immunol. 2021, 14, 547–554. [Google Scholar] [CrossRef]
- Den Besten, G.; Van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.-J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef]
- El-Khoury, A.E.; Sánchez, M.; Fukagawa, N.K.; Gleason, R.E.; Young, V.R. Similar 24-h pattern and rate of carbon dioxide production, by indirect calorimetry vs. stable isotope dilution, in healthy adults under standardized metabolic conditions. J. Nutr. 1994, 124, 1615–1627. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, N.; Tan, H.-Y.; Li, S.; Zhang, C.; Feng, Y. Function of Akkermansia muciniphila in obesity: Interactions with lipid metabolism, immune response and gut systems. Front. Microbiol. 2020, 11, 219. [Google Scholar] [CrossRef] [PubMed]
- Derrien, M.; Vaughan, E.E.; Plugge, C.M.; de Vos, W.M. Akkermansia muciniphila gen. nov., sp. nov., a human intestinal mucin-degrading bacterium. Int. J. Syst. Evol. Microbiol. 2004, 54, 1469–1476. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Q.; Feng, S.; Arjan, N.; Chen, W. A next generation probiotic, Akkermansia muciniphila. Crit. Rev. Food Sci. Nutr. 2019, 59, 3227–3236. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Wang, M.M.; Kulinich, A.; Yao, H.L.; Ma, H.Y.; Martínez, J.E.; Duan, X.C.; Chen, H.; Cai, Z.P.; Flitsch, S.L. Biochemical characterisation of the neuraminidase pool of the human gut symbiont Akkermansia muciniphila. Carbohydr. Res. 2015, 415, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Ottman, N.; Huuskonen, L.; Reunanen, J.; Boeren, S.; Klievink, J.; Smidt, H.; Belzer, C.; De Vos, W.M. Characterization of outer membrane proteome of Akkermansia muciniphila reveals sets of novel proteins exposed to the human intestine. Front. Microbiol. 2016, 7, 1157. [Google Scholar] [CrossRef] [PubMed]
- Collado, M.C.; Derrien, M.; Isolauri, E.; de Vos, W.M.; Salminen, S. Intestinal integrity and Akkermansia muciniphila, a mucin-degrading member of the intestinal microbiota present in infants, adults, and the elderly. Appl. Environ. Microbiol. 2007, 73, 7767–7770. [Google Scholar] [CrossRef]
- Wang, L.; Christophersen, C.T.; Sorich, M.J.; Gerber, J.P.; Angley, M.T.; Conlon, M.A. Low relative abundances of the mucolytic bacterium Akkermansia muciniphila and Bifidobacterium spp. in feces of children with autism. Appl. Environ. Microbiol. 2011, 77, 6718–6721. [Google Scholar] [CrossRef]
- Hansen, C.; Krych, L.; Nielsen, D.; Vogensen, F.; Hansen, L.; Sørensen, S.; Buschard, K.; Hansen, A. Early life treatment with vancomycin propagates Akkermansia muciniphila and reduces diabetes incidence in the NOD mouse. Diabetologia 2012, 55, 2285–2294. [Google Scholar] [CrossRef]
- Corb Aron, R.; Abid, A.; Vesa, C.; Nechifor, A.; Behl, T.; Ghitea, T.; Munteanu, M.; Fratila, O.; Andronie-Cioara, F.; Toma, M. Recognizing the Benefits of Pre-/Probiotics in Metabolic Syndrome and Type 2 Diabetes Mellitus Considering the Influence of Akkermansia muciniphila as a Key Gut Bacterium. Microorganisms 2021, 9, 618. [Google Scholar] [CrossRef]
- y Abreu, A.T.A.; Milke-García, M.; Argüello-Arévalo, G.; Calderón-de la Barca, A.; Carmona-Sánchez, R.; Consuelo-Sánchez, A.; Coss-Adame, E.; García-Cedillo, M.; Hernández-Rosiles, V.; Icaza-Chávez, M. Dietary fiber and the microbiota: A narrative review by a group of experts from the Asociación Mexicana de Gastroenterología. Rev. Gastroenterol. Méx. (Engl. Ed.) 2021, 86, 287–304. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Pan, M.; Pan, S.; Li, W.; Zhong, Y.; Hu, J.; Nie, S. Effects of insoluble and soluble fibers isolated from barley on blood glucose, serum lipids, liver function and caecal short-chain fatty acids in type 2 diabetic and normal rats. Food Chem. Toxicol. 2020, 135, 110937. [Google Scholar] [CrossRef]
- Cao, Y.; Yao, G.; Sheng, Y.; Yang, L.; Wang, Z.; Yang, Z.; Zhuang, P.; Zhang, Y. JinQi Jiangtang tablet regulates gut microbiota and improve insulin sensitivity in type 2 diabetes mice. Journal of diabetes research 2019, 2019, 1872134. [Google Scholar] [CrossRef] [PubMed]
- Adachi, K.; Sugiyama, T.; Yamaguchi, Y.; Tamura, Y.; Izawa, S.; Hijikata, Y.; Ebi, M.; Funaki, Y.; Ogasawara, N.; Goto, C. Gut microbiota disorders cause type 2 diabetes mellitus and homeostatic disturbances in gut-related metabolism in Japanese subjects. J. Clin. Biochem. Nutr. 2019, 64, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; Jocken, J.W.; Blaak, E.E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 2015, 11, 577–591. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Adachi, K.; Sugiyama, T.; Shimozato, A.; Ebi, M.; Ogasawara, N.; Funaki, Y.; Goto, C.; Sasaki, M.; Kasugai, K. Association of intestinal microbiota with metabolic markers and dietary habits in patients with type 2 diabetes. Digestion 2016, 94, 66–72. [Google Scholar] [CrossRef]
- Pingitore, A.; Chambers, E.S.; Hill, T.; Maldonado, I.R.; Liu, B.; Bewick, G.; Morrison, D.J.; Preston, T.; Wallis, G.A.; Tedford, C. The diet-derived short chain fatty acid propionate improves beta-cell function in humans and stimulates insulin secretion from human islets in vitro. Diabetes Obes. Metab. 2017, 19, 257–265. [Google Scholar] [CrossRef]
- Sanna, S.; van Zuydam, N.R.; Mahajan, A.; Kurilshikov, A.; Vich Vila, A.; Võsa, U.; Mujagic, Z.; Masclee, A.A.; Jonkers, D.M.; Oosting, M. Causal relationships among the gut microbiome, short-chain fatty acids and metabolic diseases. Nat. Genet. 2019, 51, 600–605. [Google Scholar] [CrossRef]
- De Vadder, F.; Kovatcheva-Datchary, P.; Goncalves, D.; Vinera, J.; Zitoun, C.; Duchampt, A.; Bäckhed, F.; Mithieux, G. Microbiota-generated metabolites promote metabolic benefits via gut-brain neural circuits. Cell 2014, 156, 84–96. [Google Scholar] [CrossRef]
- Kondo, T.; Kishi, M.; Fushimi, T.; Kaga, T. Acetic acid upregulates the expression of genes for fatty acid oxidation enzymes in liver to suppress body fat accumulation. J. Agric. Food Chem. 2009, 57, 5982–5986. [Google Scholar] [CrossRef]
- Yamashita, H.; Maruta, H.; Jozuka, M.; Kimura, R.; Iwabuchi, H.; Yamato, M.; Saito, T.; Fujisawa, K.; Takahashi, Y.; Kimoto, M. Effects of acetate on lipid metabolism in muscles and adipose tissues of type 2 diabetic Otsuka Long-Evans Tokushima Fatty (OLETF) rats. Biosci. Biotechnol. Biochem. 2009, 73, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, H.; Fujisawa, K.; Ito, E.; Idei, S.; Kawaguchi, N.; Kimoto, M.; Hiemori, M.; Tsuji, H. Improvement of obesity and glucose tolerance by acetate in Type 2 diabetic Otsuka Long-Evans Tokushima Fatty (OLETF) rats. Biosci. Biotechnol. Biochem. 2007, 71, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
- Den Besten, G.; Bleeker, A.; Gerding, A.; van Eunen, K.; Havinga, R.; van Dijk, T.H.; Oosterveer, M.H.; Jonker, J.W.; Groen, A.K.; Reijngoud, D.-J. Short-chain fatty acids protect against high-fat diet–induced obesity via a PPARγ-dependent switch from lipogenesis to fat oxidation. Diabetes 2015, 64, 2398–2408. [Google Scholar] [CrossRef] [PubMed]
- Tolhurst, G.; Heffron, H.; Lam, Y.S.; Parker, H.E.; Habib, A.M.; Diakogiannaki, E.; Cameron, J.; Grosse, J.; Reimann, F.; Gribble, F.M. Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein–coupled receptor FFAR2. Diabetes 2012, 61, 364–371. [Google Scholar] [CrossRef]
- Larraufie, P.; Martin-Gallausiaux, C.; Lapaque, N.; Dore, J.; Gribble, F.; Reimann, F.; Blottiere, H. SCFAs strongly stimulate PYY production in human enteroendocrine cells. Sci. Rep. 2018, 8, 74. [Google Scholar] [CrossRef]
- Xiong, Y.; Miyamoto, N.; Shibata, K.; Valasek, M.A.; Motoike, T.; Kedzierski, R.M.; Yanagisawa, M. Short-chain fatty acids stimulate leptin production in adipocytes through the G protein-coupled receptor GPR41. Proc. Natl. Acad. Sci. USA 2004, 101, 1045–1050. [Google Scholar] [CrossRef]
- Al-Lahham, S.a.H.; Roelofsen, H.; Priebe, M.; Weening, D.; Dijkstra, M.; Hoek, A.; Rezaee, F.; Venema, K.; Vonk, R.J. Regulation of adipokine production in human adipose tissue by propionic acid. Eur. J. Clin. Investig. 2010, 40, 401–407. [Google Scholar] [CrossRef]
- Canfora, E.E.; van der Beek, C.M.; Jocken, J.W.; Goossens, G.H.; Holst, J.J.; Olde Damink, S.W.; Lenaerts, K.; Dejong, C.H.; Blaak, E.E. Colonic infusions of short-chain fatty acid mixtures promote energy metabolism in overweight/obese men: A randomized crossover trial. Sci. Rep. 2017, 7, 2360. [Google Scholar] [CrossRef]
- Ohira, H.; Fujioka, Y.; Katagiri, C.; Mamoto, R.; Aoyama-Ishikawa, M.; Amako, K.; Izumi, Y.; Nishiumi, S.; Yoshida, M.; Usami, M. Butyrate attenuates inflammation and lipolysis generated by the interaction of adipocytes and macrophages. J. Atheroscler. Thromb. 2013, 20, 425–442. [Google Scholar] [CrossRef]
- Al-Lahham, S.A.; Roelofsen, H.; Rezaee, F.; Weening, D.; Hoek, A.; Vonk, R.; Venema, K. Propionic acid affects immune status and metabolism in adipose tissue from overweight subjects. Eur. J. Clin. Investig. 2012, 42, 357–364. [Google Scholar] [CrossRef]
- Xiao, A.; Yang, S.; Iqbal, Q. Factors affecting purchase intentions in generation Y: An empirical evidence from fast food industry in Malaysia. Adm. Sci. 2018, 9, 4. [Google Scholar] [CrossRef]
- Fleischhacker, S.E.; Evenson, K.R.; Rodriguez, D.A.; Ammerman, A.S. A systematic review of fast food access studies. Obes. Rev. 2011, 12, e460–e471. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, J.F.; Lin, B.-H.; Frazao, E. Role of food prepared away from home in the American diet, 1977–1978 versus 1994-96: Changes and consequences. J. Nutr. Educ. Behav. 2002, 34, 140–150. [Google Scholar] [CrossRef]
- Mancino, L.; Todd, J.E.; Guthrie, J.; Lin, B.-H. Food Away from Home and Childhood Obesity. Curr. Obes. Rep. 2014, 3, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, L.; Xue, H.; Qu, W. A review of the growth of the fast food industry in China and its potential impact on obesity. Int. J. Environ. Res. Public Health 2016, 13, 1112. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, L.; Zhu, J.; Gao, L.; Wang, Y. Growing fast food consumption and obesity in Asia: Challenges and implications. Soc. Sci. Med. 2021, 269, 113601. [Google Scholar] [CrossRef]
- Cândido, F.G.; Valente, F.X.; Grześkowiak, Ł.M.; Moreira, A.P.B.; Rocha, D.M.U.P.; Alfenas, R.d.C.G. Impact of dietary fat on gut microbiota and low-grade systemic inflammation: Mechanisms and clinical implications on obesity. Int. J. Food Sci. Nutr. 2018, 69, 125–143. [Google Scholar] [CrossRef]
- Nazmul Haque, M.; Towhidul Islam, M.; Tariqul Hassan, M.; Shekhar, H.U. Determination of heavy metal contents in frequently consumed fast foods of Bangladesh. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2019, 89, 543–549. [Google Scholar] [CrossRef]
- Bahreini Esfahani, N.; Jafari, M.; Moravejolahkami, A.R. Heavy metals concentration and target hazard quotients assessment through the consumption of fish muscle Ctenopharyngodon Idella (Cyprinidae) from markets in Ahvaz province, Iran. Nutr. Food Sci. 2020, 50, 529–537. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, M.; Pang, X.; Zhao, Y.; Wang, L.; Zhao, L. Structural resilience of the gut microbiota in adult mice under high-fat dietary perturbations. ISME J. 2012, 6, 1848–1857. [Google Scholar] [CrossRef]
- Suceveanu, A.I.; Stoian, A.P.; Parepa, I.; Voinea, C.; Hainarosie, R.; Manuc, D.; Nitipir, C.; Mazilu, L.; Suceveanu, A.P. Gut microbiota patterns in obese and type 2 diabetes (T2D) patients from romanian black sea coast region. Rev. Chim. 2018, 69, 2260–2267. [Google Scholar] [CrossRef]
- Sroka-Oleksiak, A.; Młodzińska, A.; Bulanda, M.; Salamon, D.; Major, P.; Stanek, M.; Gosiewski, T. Metagenomic analysis of duodenal microbiota reveals a potential biomarker of dysbiosis in the course of obesity and type 2 diabetes: A pilot study. J. Clin. Med. 2020, 9, 369. [Google Scholar] [CrossRef] [PubMed]
- Nova, E.; Gómez-Martinez, S.; González-Soltero, R. The influence of dietary factors on the gut microbiota. Microorganisms 2022, 10, 1368. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, A.; Frazer, Z.A.; Hansbro, P.M.; Yang, I.A. COPD and the gut-lung axis: The therapeutic potential of fibre. J. Thorac. Dis. 2019, 11, S2173. [Google Scholar] [CrossRef] [PubMed]
- Wood, L.G.; Garg, M.L.; Gibson, P.G. A high-fat challenge increases airway inflammation and impairs bronchodilator recovery in asthma. J. Allergy Clin. Immunol. 2011, 127, 1133–1140. [Google Scholar] [CrossRef]
- Bohlouli, J.; Moravejolahkami, A.R.; Ganjali Dashti, M.; Balouch Zehi, Z.; Hojjati Kermani, M.A.; Borzoo-Isfahani, M.; Bahreini-Esfahani, N. COVID-19 and fast foods consumption: A review. Int. J. Food Prop. 2021, 24, 203–209. [Google Scholar] [CrossRef]
- Carmody, R.N.; Bisanz, J.E.; Bowen, B.P.; Maurice, C.F.; Lyalina, S.; Louie, K.B.; Treen, D.; Chadaideh, K.S.; Maini Rekdal, V.; Bess, E.N. Cooking shapes the structure and function of the gut microbiome. Nat. Microbiol. 2019, 4, 2052–2063. [Google Scholar] [CrossRef]
- Aljahdali, N.; Gadonna-Widehem, P.; Anton, P.M.; Carbonero, F. Gut microbiota modulation by dietary barley malt melanoidins. Nutrients 2020, 12, 241. [Google Scholar] [CrossRef]
- Lee, K.W.; Song, W.O.; Cho, M.S. Dietary quality differs by consumption of meals prepared at home vs. outside in Korean adults. Nutr. Res. Pract. 2016, 10, 294–304. [Google Scholar] [CrossRef]
- Mohiuddin, A.K. Fast food addiction: A major public health issue. J. Nutr. Food Process. 2020, 3, 1. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Mirmiran, P.; Azizi, F. Fast food pattern and cardiometabolic disorders: A review of current studies. Health Promot. Perspect. 2015, 5, 231. [Google Scholar] [CrossRef] [PubMed]
- Haynes-Maslow, L.; Leone, L.A. Examining the relationship between the food environment and adult diabetes prevalence by county economic and racial composition: An ecological study. BMC Public Health 2017, 17, 648. [Google Scholar] [CrossRef] [PubMed]
- Fuhrman, J. The hidden dangers of fast and processed food. Am. J. Lifestyle Med. 2018, 12, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Beigrezaei, S.; Ghiasvand, R.; Feizi, A.; Iraj, B. Relationship between dietary patterns and incidence of type 2 diabetes. Int. J. Prev. Med. 2019, 10, 122. [Google Scholar]
- Wen, L.M.; Simpson, J.M.; Rissel, C.; Baur, L.A. Maternal “junk food” diet during pregnancy as a predictor of high birthweight: Findings from the healthy beginnings trial. Birth 2013, 40, 46–51. [Google Scholar] [CrossRef]
- Bodicoat, D.H.; Carter, P.; Comber, A.; Edwardson, C.; Gray, L.J.; Hill, S.; Webb, D.; Yates, T.; Davies, M.J.; Khunti, K. Is the number of fast-food outlets in the neighbourhood related to screen-detected type 2 diabetes mellitus and associated risk factors? Public Health Nutr. 2015, 18, 1698–1705. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).